Importance of Rocks and Their Weathering Products on Groundwater Quality in Central-East Cameroon
Abstract
:1. Introduction
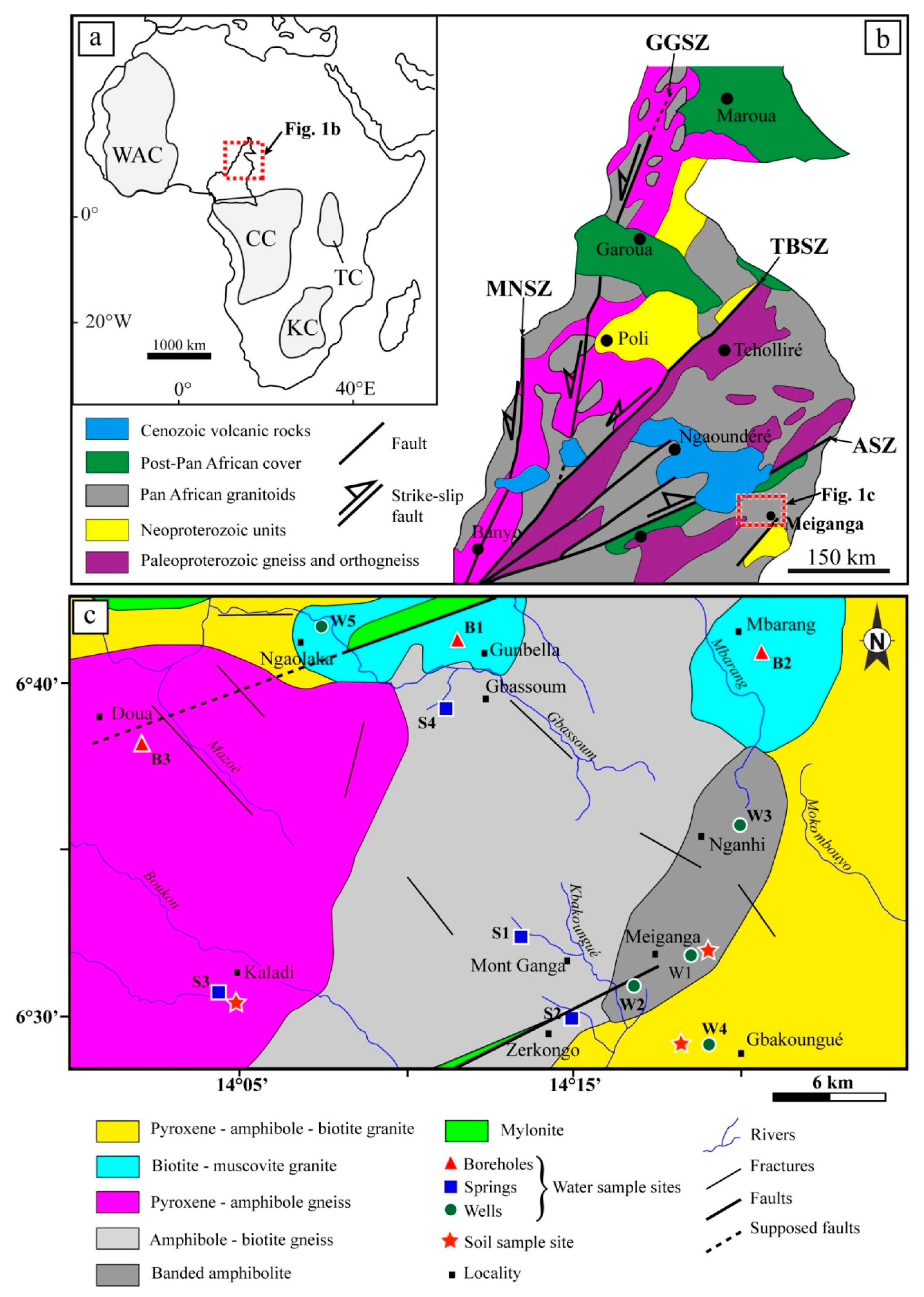
2. Location and Hydrogeological Background of the Study Area
3. Methods of Study
3.1. Sample Collection
3.2. Analytical Procedures
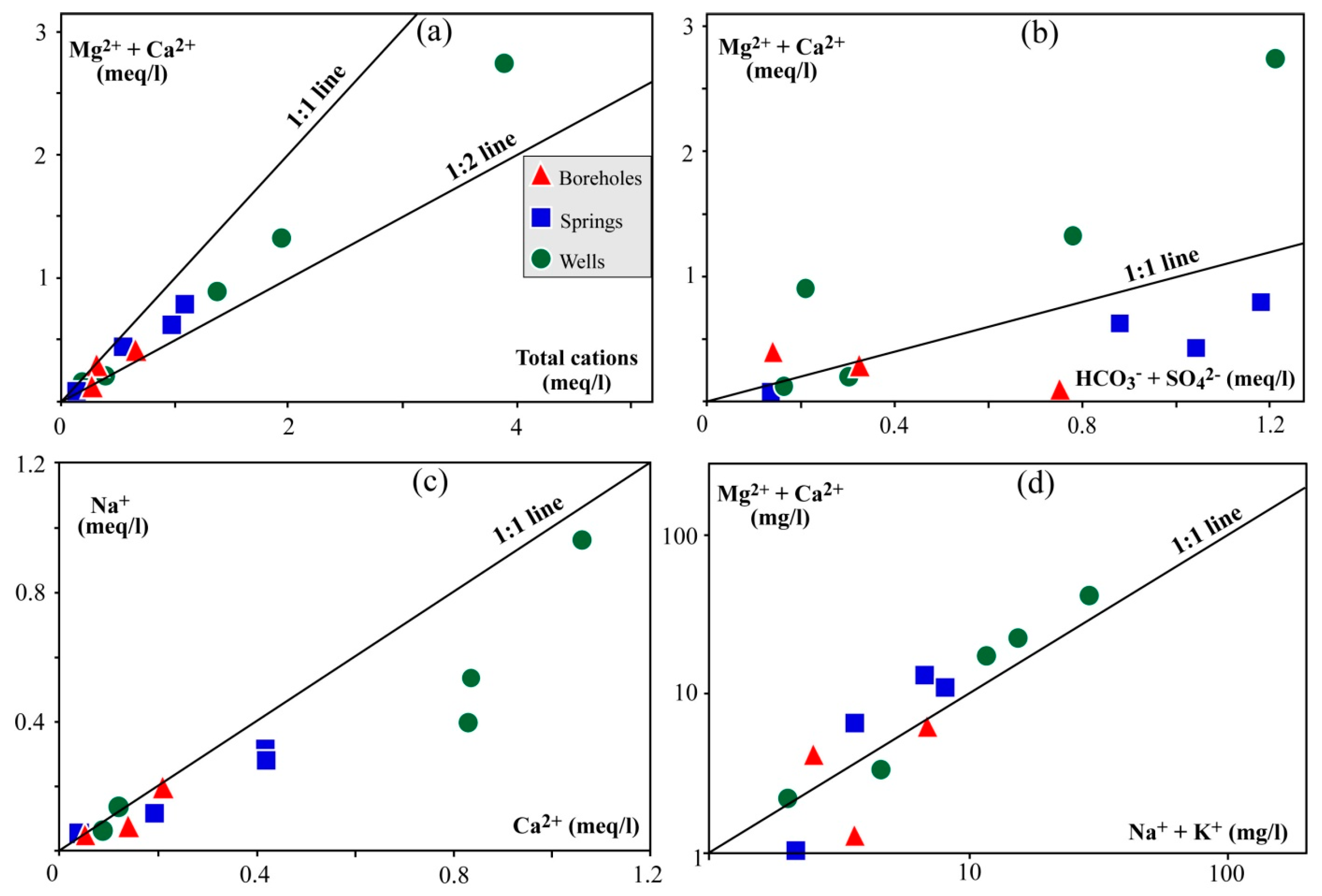
4. Results
4.1. Petrographic and Geochemical Characterisation of Geologic Formations
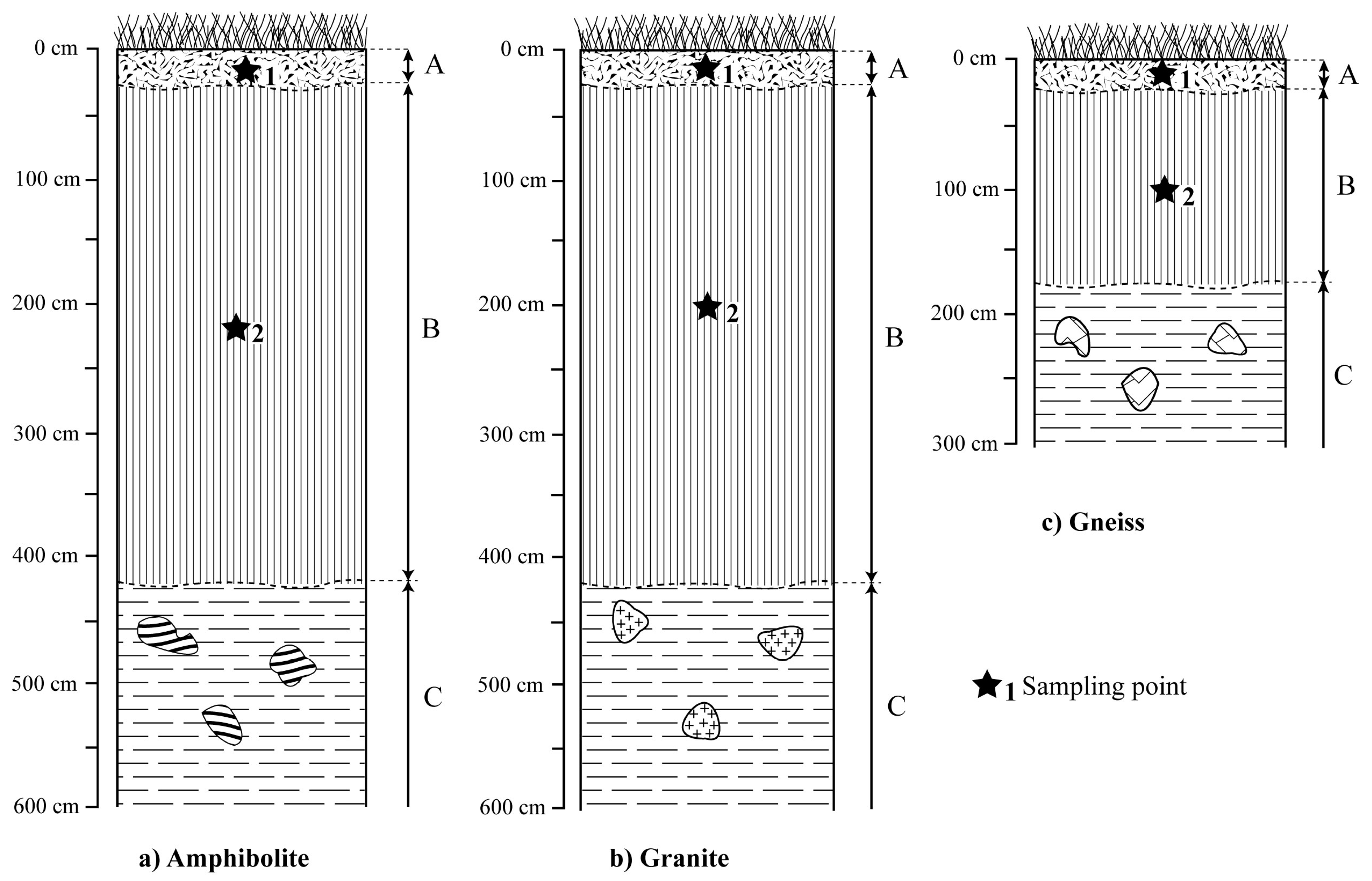
4.2. Morphological Characteristics of Weathering Material
- -
- a humiferous Ap horizon. Its thickness is about 25 cm on each bedrock. It is dark, characterized by a sandy, clayey texture, a lumpy structure and high matrix porosity. Few nodules are present and there are many rootlets. The boundary is progressive and regular;
- -
- a red B horizon. Its thickness is about 150 cm on gneiss and about 400 cm on amphibolite and granite (Figure 2). It is characterized by a clayey texture and a blocky structure. Nodules are noted on all bedrock and their contents are below 10%. The boundary is progressive and regular;
- -
- a reddish C horizon in which the structure of the bedrock is well preserved.
4.3. Physicochemical Characteristics of Soil Solutions
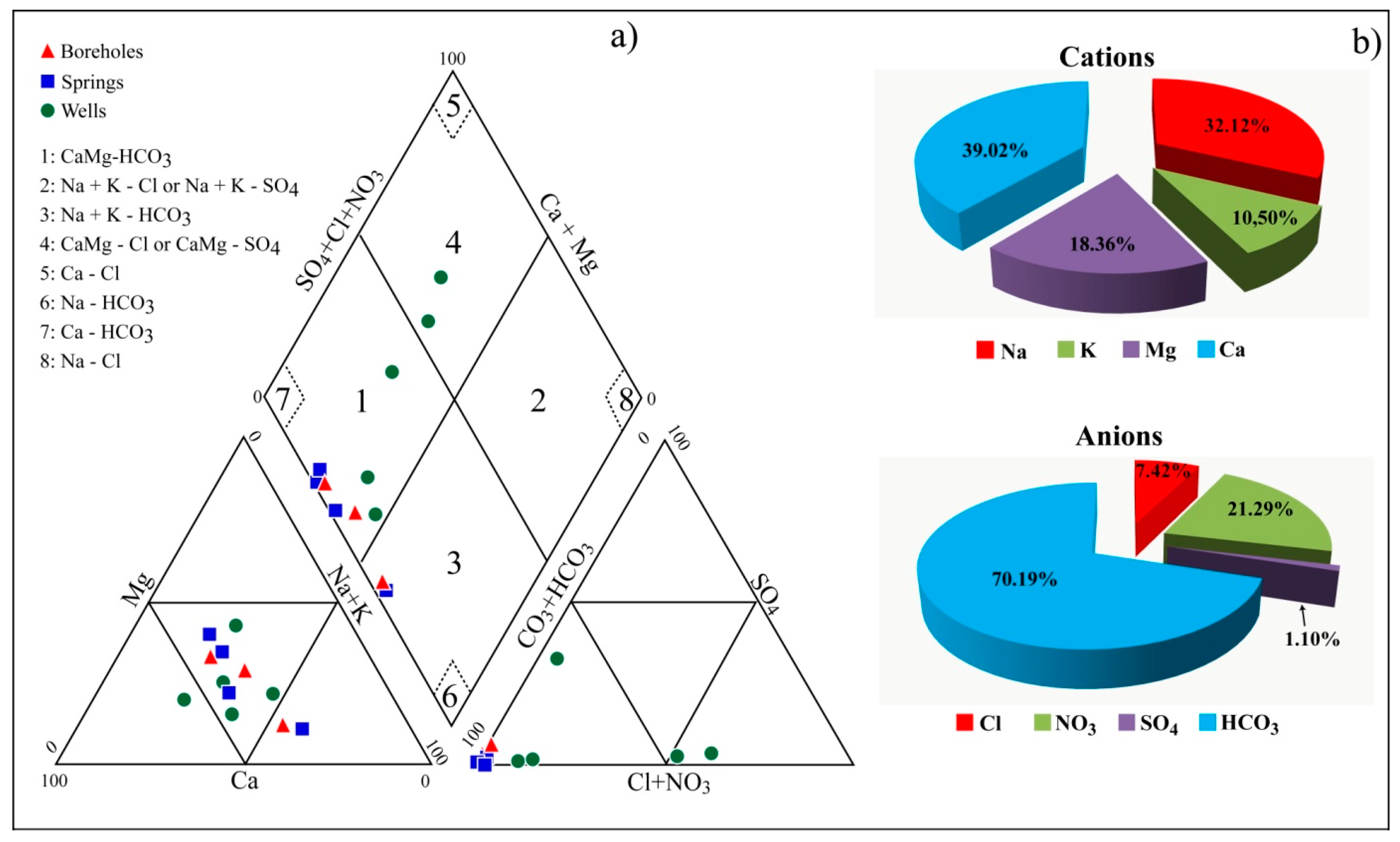
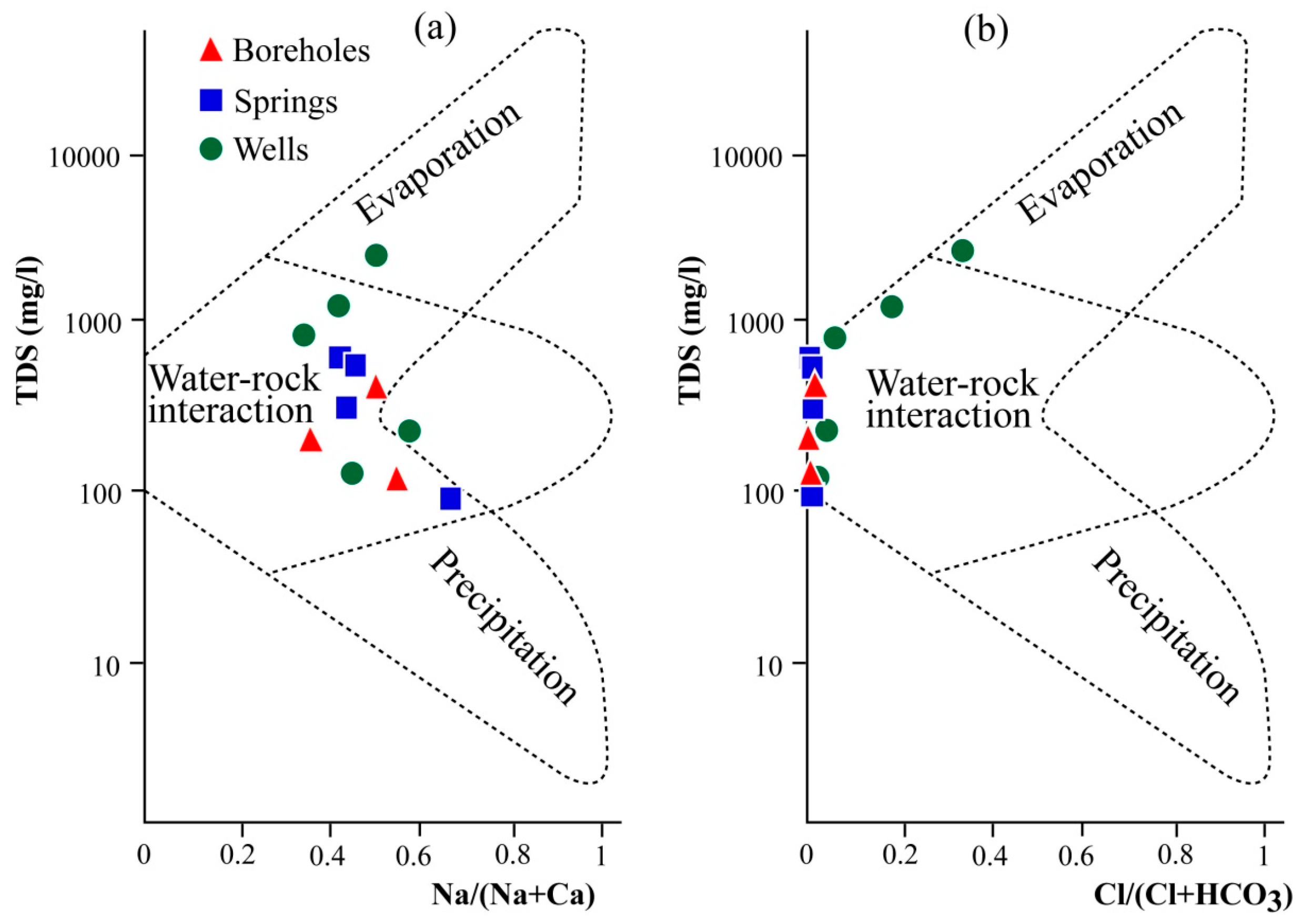
4.4. Physicochemical Characteristics of Water Samples
5. Discussion
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Oki, A.O.; Akana, T.S. Quality Assessment of Groundwater in Yenagoa, Niger Delta, Nigeria. Geosciences 2016, 6, 1–12. [Google Scholar]
- Ndiomo, E.D.; Mengue Mbom, A.; Assako Assako, R. Remote sensing and rational interventions for the urban development in Africa. The case of Yaounde in Cameroon (Central Africa). In Proceedings of the ISPRS, Tempe, AZ, USA, 14–16 March 2005; p. 5. [Google Scholar]
- Ngnikam, E.; Mougoue, B.; Tietche, F. Eau, Assainissement et impact sur la santé: Étude de cas d’un écosystème urbain à Yaoundé. In Proceedings of the Actes des JSIRAUF, Hanoi, Vietnam, 6–9 November 2007. [Google Scholar]
- Kringel, R.; Rechenburgc, A.; Kuitcha, D.; Fouépé, A.; Bellenberg, S.; Kengne, I.M.; Fomo, M.A. Mass balance of nitrogen and potassiumin urban groundwater in Central Africa, Yaounde/Cameroon. Sci. Total Environ. 2016, 547, 382–395. [Google Scholar] [CrossRef] [PubMed]
- Ayoade, J.O. Tropical Hydrology and Water Resources; Macmillan Ltd.: London, UK, 2003; p. 276. [Google Scholar]
- Salbu, B.; Steinnes, E. Trace Elements in Natural Water; CRC Press. Inc.: Boca Raton, FL, USA, 1995; p. 302. [Google Scholar]
- Nono, A.; Temgoua, E.; Likeng, J.D.H.; Djouokouo, T.J.P. Influence de la nature lithologique et des structures géologiques sur la qualité des eaux souterraines dans le versant Nord des Monts Bambouto: Cas du village Balepo et ses environs. Afr. Geosci. Rev. 2008, 15, 149–162. [Google Scholar]
- Jackson, T.A. The Biogeochemical and Ecological Significance of Interactions between Colloidal Minerals and Trace Elements. In Environmental Interactions of Clays; Rae, J., Parker, A., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 273. [Google Scholar]
- Temgoua, E.; Djeuda, T.H.B.; Tanawa, E.; Guenat, C.; Pfeifer, H.R. Groundwater fluctuations and footslope ferricrete soils in the humid tropical zone of southern Cameroon. Hydrol. Proc. 2005, 19, 3097–3111. [Google Scholar] [CrossRef]
- Nahon, D. Cuirasses Ferrugineuses et Encroûtement Calcaires au Sénégal Occidental et en Mauritanie. Système Évolutifs: Géochimie, Structures, Relais et Coexistence; Mémoire Société Géologique: Strasbourg, France, 1976; p. 232. [Google Scholar]
- Drever, J.I.; Zobrist, S. Chemical weathering of silicate rocks as a function of evolution in the southem Swiss Alps. Geochim. Cosmochim. Acta 1992, 56, 3209–3216. [Google Scholar] [CrossRef]
- World Health Organization and United Nations Children’s Fund (WHO/UNICEF). Coverage Estimates: Improved Sanitation, Cameroon. Joint Monitoring Programme for Water Supply and Sanitation. 2008. Available online: http://documents.wssinfo.orgdownload/id_document (15 September 2008).
- Ako, A.A.; Takem, E.G.E.; Nkeng, E.G. Water resources management and integrated water resources management (IWRM) in Cameroon. Water Resour. Manag. 2010, 24, 871–888. [Google Scholar] [CrossRef]
- Djeuda, T.H.B.; Tanawa, E.; Ngnikam, E. L’eau au Cameroun; Tome I: Approvisionnement en Potable; Presse Universitaire Yaoundé: Yaoundé, Cameroun, 2001; p. 359. [Google Scholar]
- Suchel, J.B. Les Climats du Cameroun. Tome III. Ph.D. Thesis, Université de St Etienne, St. Etienne, France, 1987; p. 1186. [Google Scholar]
- Olivry, J.C. Fleuves et Rivières du Cameroun; Collection “Monographies Hydrologiques ORSTOM”; ORSTOM: Marseille, France, 1986; p. 547. [Google Scholar]
- Lasserre, M. Carte Géologique de Reconnaissance à L’échelle 1/500 000, Territoire du Cameroun, Ngaoundéré-Est, Dir; Mines Géol. Cameroun: Yaoundé, Cameroun, 1961. [Google Scholar]
- Toteu, S.F.; Van Schmus, R.W.; Penaye, J.; Michard, A. New U–Pb and Sm–Nd data from North-Central Cameroon and its bearing on the Pre-Pan-African history of Central Africa. Precamb. Res. 2001, 108, 45–73. [Google Scholar] [CrossRef]
- Ganwa, A.A.; Frisch, W.; Siebel, W.; Shang, K.C.; Ondoa, M.J.; Satir, M.; Numbem, T.J. Zircon 207Pb/206Pb evaporation ages of Pan-African metasedimentary rocks in the Kombé-II area (Bafia Group, Cameroun): Constraints on protolith age and provenance. C. R. Géosci. 2008, 340, 211–222. [Google Scholar] [CrossRef]
- Ganwa, A.A.; Siebel, W.; Frisch, W.; Kongnyuy Shang, C. Geochemistry of magmatic rocks and time constraints on deformational phases and shear zone slip in the Méiganga area, central Cameroon. Int. Geol. Rev. 2011, 53, 759–784. [Google Scholar] [CrossRef]
- Ganwa, A.A.; Siebel, W.; Kongnyuy Shang, C.; Seguem, N.; Ekodeck, G.E. New Constraints from Pb-Evaporation Zircon Ages of the Méiganga Amphibole-Biotite Gneiss, Central Cameroon, on Proterozoic Crustal Evolution. Int. J. Geosci. 2011, 2, 138–147. [Google Scholar]
- Toteu, S.F.; Penaye, J.; Poudjom-Djomani, Y. Geodynamic evolution of the Pan-African belt in central Africa with special reference to Cameroon. Can. J. Earth Sci. 2004, 41, 73–85. [Google Scholar] [CrossRef]
- Baise, D. Guide pour la Description des Sols; Institut National de la Recherche Agronomique (INRA): Paris, France, 1995. [Google Scholar]
- Verma, B.C.; Swaminathan, K.; Sud, K.C. An improved turbidimetric procedure for determination of sulphate in plants and soils. Talanta 1977, 29, 49–50. [Google Scholar] [CrossRef]
- Walkey, A.; Black, I.A. Determination of organic matter in soil. Soil Sci. 1934, 37, 549–556. [Google Scholar]
- Bray, R.H.; Kurtz, L.T. Determination of total organic and available forms of phosphorus in soils. Soil Sci. 1945, 59, 22–229. [Google Scholar] [CrossRef]
- Kamtchueng, B.T.; Fantong, W.Y.; Ueda, A.; Tiodjio, E.R.; Anazawa, K.; Wirmvem, M.J.; Mvondo, J.O.; Nkamdjou, S.L.; Kusakabe, M.; Ohba, T.; et al. Assessment of shallow groundwater in Lake Nyos catchment (Cameroon, Central-Africa): Implications for hydrogeochemical controls and uses. Environ. Earth Sci. 2014, 72, 3663–3678. [Google Scholar] [CrossRef]
- Kamtchueng, B.T.; Fantong, W.Y.; Wirmvem, M.J.; Tiodjio, R.E.; Takounjou, A.F.; Ndam Ngoupayou, J.R.; Kusakabe, M.; Zhang, J.; Ohba, T.; Tanyileke, G.; et al. Hydrogeochemistry and quality of surface water and groundwater in the vicinity of Lake Monoun, West Cameroon: Approach from multivariate statistical analysis and stable isotopic characterization. Environ. Monit. Assess. 2016, 524, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Freeze, R.A.; Cherry, J.A. Groundwater; Prentice Hall: Englewood Cliffs, NJ, USA, 1979; p. 604. [Google Scholar]
- Takem, G.E.; Kuitcha, D.; Ako, A.A.; Mafany, G.T.; Takounjou-Fouepe, A.; Ndjama, J.; Ntchancho, R.; Ateba, B.H.; Chandrasekharam, D.; Ayonghe, S.N. Acidification of shallow groundwater in the unconfined sandy aquifer of the city of Douala, Cameroon, Western Africa: Implications for groundwater quality and use. Environ. Earth Sci. 2015, 61, 287–298. [Google Scholar] [CrossRef]
- Wirmvem, M.J.; Ohba, T.; Fantong, W.Y.; Ayonghe, S.N.; Suila, J.Y.; Asaah, A.N.E.; Tanyileke, G.; Hell, J.V. Hydrogeochemistry of shallow groundwater and surface water in the Ndop plain, NorthWest Cameroon. Afr. J. Environ. Sci. Technol. 2013, 7, 518–530. [Google Scholar]
- Ako, A.A.; Shimada, J.; Hosono, T.; Ichiyanagi, K.; Nkeng, G.E.; Fantong, W.Y.; Takem, G.E.E.; Njila, N.R. Evaluation of groundwater quality and its suitability for drinking, domestic, and agricultural uses in the Banana Plain (Mbanga, Njombe, Penja) of the Cameroon Volcanic Line. Environ. Geochem. Health 2011, 33, 559–575. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Guidelines for Drinkingwater Quality: Training Pack; WHO: Geneva, Switzerland, 2004. [Google Scholar]
- Piper, A.M. A graphical interpretation of water-analysis. Trans. Am. Geophys. Union 1944, 25, 914–928. [Google Scholar] [CrossRef]
- Hem, J.D. Study and Interpretation of the Chemical Characteristics of Natural Water; U.S Geological Survey Water-Supply Paper; U.S Geological Survey: Reston, VA, USA, 1989; p. 272. [Google Scholar]
- Nesbitt, H.W.; Wilson, R.E. Recent chemical weathering of basalts. Am. J. Sci. 1992, 292, 740–777. [Google Scholar] [CrossRef]
- Herczeg, A. Can major ion chemistry be used estimated groundwater residence time in basaltic aquifer? In Proceedings of the 9th International Symposium on Water–Rock Interaction, Taupo, New Zealand, 30 March–3 April 1998; Cidu, R., Ed.; Balkema: Rotterdam, The Netherlands, 1998; pp. 529–532. [Google Scholar]
- Subramani, T.; Rajmohan, N.; Elango, L. Groundwater geochemistry and identification of hydrogeochemical processes in hard rock region, Southern India. Environ. Monit. Assess. 2010, 162, 123–137. [Google Scholar] [CrossRef] [PubMed]
- Tanyileke, G.; Kusakabe, M.; Evans, W.C. Chemical and isotopic characteristics of fluids along the Cameroon Volcanic Line, Cameroon. J. Afr. Earth Sci. 1996, 22, 433–441. [Google Scholar] [CrossRef]
- Ako, A.A.; Shimada, J.; Hosono, T.; Kagabu, M.; Ayuk, A.R.; Nkeng, G.E.; Takem, G.E.E.; Takounjou, A.L.F. Spring water quality and usability in the Mount Cameroon area revealed by hydrogeochemistry. Environ. Geochem. Health 2012. [Google Scholar] [CrossRef] [PubMed]
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater, and Pollution, 2nd ed.; Balkema Publishers: Rotterdam, The Netherlands, 2005; p. 649. [Google Scholar]
- Demlie, M.; Wohnlich, S.; Wisotzky, F.; Gizaw, B. Groundwater recharge, flow and hydrogeochemical evolution in a complex volcanic aquifer system, central Ethiopia. Hydrogeol. J. 2007, 15, 1169–1181. [Google Scholar] [CrossRef]
- Gibbs, R.J. Mechanisms controlling world water chemistry. Science 1970, 17, 1088–1090. [Google Scholar] [CrossRef] [PubMed]
- Korjinski, D. Differential mobility of component of metasomatic zoning in metamorphism. In Proceedings of the 18th International Geology Congress, London, UK, 25 August–1 September 1948; pp. 5–7. [Google Scholar]
- Pédro, G. Essai sur la caractérisation géochimique des différents processus zonaux résultant de l’altération des roches superficielles (cycle alumino-silicique). C. R. Acad. Sci. 1966, 262, 1828–1831. [Google Scholar]
- Claquin, T.; Schulz, M.; Balkanski, Y. Modeling the mineralogy of atmospheric dust sources. J. Geophys. Res. 1999, 104, 22243–22256. [Google Scholar] [CrossRef]
- Caquineau, S.; Gaudichet, A.; Gomes, L.; Legrand, M. Mineralogy of Saharan dust transported over northwestern tropical Atlantic Ocean in relation to source regions. J. Geophys. Res. 2002, 107. [Google Scholar] [CrossRef]
- Journet, E.; Balkanski, Y.; Harrison, S.P. A new data set of soil mineralogy for dust-cycle modeling. Atmos. Chem. Phys. 2014, 14, 3801–3816. [Google Scholar] [CrossRef]
- Stoch, L.; Sikora, W. Transformations of micas in the process of kaolinitisation of granite and gneisses. Clay Clay Miner. 1976, 24, 156–162. [Google Scholar] [CrossRef]
- Matini, L.; Tathy, C.; Moutou, J.M. Seasonal groundwater quality variation in Brazzaville, Congo. Res. J. Chem. Sci. 2012, 2, 7–14. [Google Scholar]
- Hounslow, A.W. Water Quality Data: Analysis and Interpretation; Lewis Publishers: Boca Raton, FL, USA, 1995; p. 416. [Google Scholar]
- Srinivasamoorthy, K.; Chidambaram, S.; Prasanna, M.V.; Vasanthavihar, M.; Peter, J.; Anandhan, P. Identification of major sources controlling groundwater chemistry from a hard rock terrain—A case study from Mettur taluk, Salem district, Tamil Nadu. J. Earth Syst. Sci. 2008, 117, 49–58. [Google Scholar] [CrossRef]
- Fisher, R.S.; Mulican, W.F., III. Hydrochemical evolution of sodium-sulfate and sodium-chloride groundwater beneath the Northern Chihuahuan Desert, Trans-Pecos, Texas, USA. Hydrogeol. J. 1997, 10, 455–474. [Google Scholar] [CrossRef]
- Jankowski, J.; Acworth, R.I.; Shekarforoush, S. Reverse ionexchange in deeply weathered porphyritic dacite fractured aquifer system, Yass, New South Wales, Austria. In Proceedings of the 9th International Symposium on Water–Rock Interaction, Taupo, New Zealand, 30 March–3 April 1998; Arehart, G.B., Hulston, J.R., Eds.; Balkema: Rotterdam, The Netherlands, 1998; pp. 243–246. [Google Scholar]
- Kumar, M.; Kumari, K.; Ramanathan, A.I.; Saxena, R. A comparative evaluation of groundwater suitability for irrigation and drinking purposes in two intensively cultivated districts of Punjab, India. Environ. Geol. 2007, 53, 553–574. [Google Scholar] [CrossRef]
- Fantong, W.Y.; Satake, H.; Ayonghe, S.N.; Aka, F.T.; Asai, K. Hydrogeochemical controls and usability of groundwater in the semi-arid Mayo Tsanaga River Basin: Far north province, Cameroon. Environ. Geol. 2009, 58, 1281–1293. [Google Scholar] [CrossRef]
- Hendry, M.J.; Wassenaar, L.L. Controls on the distribution of majors ions in pore waters of a thick surficial aquitard. Water Resour. Res. 2000, 36, 503–513. [Google Scholar] [CrossRef]

| Rocks | Px-Amph Gneiss 1 | Bt-Amph Gneiss 2 | Bt-Musc Granite 3 | Px Granite 3 | Amphibolite | ||||
|---|---|---|---|---|---|---|---|---|---|
| Samples | Ka1 | Go1 | Me7 | Me5 | Mi2 | NPg3 | MB | Me6 | MG |
| SiO2 | 60.72 | 69.30 | 59.41 | 61.66 | 75.46 | 73.45 | 68.8 | 68.34 | 62.73 |
| TiO2 | 0.65 | 0.46 | 0.92 | 0.48 | 0.15 | 0.08 | 0.41 | 0.41 | 0.82 |
| Al2O3 | 16.08 | 15.04 | 16.12 | 20.07 | 13.64 | 13.89 | 15.32 | 15.12 | 13.02 |
| Fe2O3 | 6.66 | 4.07 | 7.23 | 3.18 | 1.44 | 0.80 | 3.04 | 3.35 | 6.72 |
| MnO | 0.11 | 0.04 | 0.12 | 0.04 | 0.036 | 0.023 | 0.087 | 0.07 | 0.15 |
| MgO | 3.22 | 1.36 | 3.59 | 1.20 | 0.23 | 0.20 | 1.42 | 1.27 | 4.46 |
| CaO | 6.11 | 2.68 | 5.69 | 2.76 | 0.83 | 0.70 | 1.86 | 2.44 | 7.15 |
| Na2O | 4.2 | 3.58 | 3.35 | 4.21 | 3.26 | 3.00 | 4.09 | 3.86 | 2.97 |
| K2O | 1.45 | 3.46 | 2.59 | 6.33 | 5.27 | 6.75 | 4.99 | 4.50 | 1.33 |
| P2O5 | 0.2 | 0.19 | 0.26 | 0.22 | 0.05 | 0.13 | 0.19 | 0.19 | 0.18 |
| LOI | 1.02 | 0.59 | 0.92 | 0.75 | 0.59 | 0.25 | 0.64 | 0.92 | 0.59 |
| Total | 100.42 | 100.77 | 100.2 | 100.9 | 101.15 | 98.46 | 101.48 | 101.09 | 100.11 |
| Na2O/K2O | 2.90 | 1.03 | 1.29 | 0,67 | 0.62 | 0.44 | 0.82 | 0.86 | 2.23 |
| MgO/CaO | 0.53 | 0.51 | 0.63 | 0,43 | 0.28 | 0.29 | 0.76 | 0.52 | 0.62 |
| Na2O + K2O | 5.65 | 7.04 | 5.94 | 10,54 | 8.53 | 9.75 | 9.08 | 8.36 | 4.29 |
| MgO + CaO | 9.33 | 4.04 | 9.28 | 3,96 | 1.06 | 0.9 | 3.28 | 3.71 | 11.61 |
| (MgO + CaO)/ (Na2O + K2O) | 1.65 | 0.57 | 1.56 | 0.38 | 0.12 | 0.09 | 0.36 | 0.44 | 2.70 |
| Geologic Formations | Position | pHwater | pHKCl | OC (%) | OM (%) | P (mg/kg) | EC (µS/cm) | Ntot (%) | SO42− (%) | NO3-N (%) | Cl− (%) | Ca2+ | Mg2+ | Na+ | K+ |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (cmol(+)/kg) | |||||||||||||||
| Granite | 1 | 5.3 | 4.5 | 0.60 | 1.03 | 40.14 | 74 | 0.13 | 0.42 | 0.022 | 0.011 | 1.42 | 0.08 | 0.15 | 0.03 |
| 2 | 5.3 | 4.8 | 0.09 | 0.16 | 8.68 | 97 | 0.07 | 0.11 | 0.009 | 0.015 | 0.14 | 0.04 | 0.23 | 0.04 | |
| Amphibolite | 1 | 6.3 | 6.1 | 0.55 | 0.94 | 71.48 | 82 | 0.17 | 0.36 | 0.017 | 0.013 | 0.20 | 0.10 | 0.02 | 0.05 |
| 2 | 6.7 | 6.3 | 0.18 | 0.31 | 58.26 | 84 | 0.16 | 0.24 | 0.011 | 0.013 | 1.71 | 0.15 | 0.10 | 0.88 | |
| Gneiss | 1 | 5.9 | 5.4 | 0.36 | 0.63 | 14.74 | 75 | 0.08 | 0.30 | 0.016 | 0.010 | 0.72 | 0.28 | 0.28 | 0.02 |
| 2 | 5.5 | 4.7 | 0.36 | 0.63 | 2.68 | 53 | 0.09 | 0.32 | 0.015 | 0.007 | 0.20 | 0.08 | 0.23 | 0.16 | |
| Min | 5.30 | 4.50 | 0.09 | 0.16 | 2.68 | 53.00 | 0.07 | 0.11 | 0.009 | 0.007 | 0.14 | 0.04 | 0.02 | 0.02 | |
| Max | 6.70 | 6.30 | 0.60 | 1.03 | 71.48 | 97.00 | 0.17 | 0.42 | 0.022 | 0.015 | 1.71 | 0.28 | 0.28 | 0.88 | |
| Mean | 5.83 | 5.30 | 0.36 | 0.62 | 32.66 | 77.50 | 0.12 | 0.29 | 0.015 | 0.012 | 0.73 | 0.12 | 0.17 | 0.20 | |
| SD | 0.52 | 0.69 | 0.18 | 0.31 | 25.86 | 13.30 | 0.04 | 0.10 | 0.004 | 0.003 | 0.62 | 0.08 | 0.09 | 0.31 | |
| CV (%) | 9.00 | 13.10 | 51.10 | 50.30 | 79.20 | 17.20 | 33.40 | 33.60 | 28.00 | 22.30 | 85.50 | 64.10 | 52.60 | 157.20 | |
| pHeau | pHKCl | CO | MO | P | EC | Ntot | SO42− | NO3-N | Cl− | Ca2+ | Mg2+ | Na+ | K+ | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pHeau | 1 | |||||||||||||
| pHKCl | 0.977 * | 1 | ||||||||||||
| CO | −0.090 | −0.112 | 1 | |||||||||||
| MO | −0.097 | −0.121 | 1.000 * | 1 | ||||||||||
| EG | 0.199 | 0.064 | −0.129 | −0.121 | ||||||||||
| P | 0.711 | 0.731 | 0.391 | 0.376 | 1 | |||||||||
| CE | 0.172 | 0.317 | −0.442 | −0.454 | 0.304 | 1 | ||||||||
| Ntot | 0.705 | 0.682 | 0.425 | 0.412 | 0.967 * | 0.100 | 1 | |||||||
| SO42− | 0.022 | −0.061 | 0.953 * | 0.955 * | 0.371 | −0.605 | 0.459 | 1 | ||||||
| NO3-N | −0.227 | −0.279 | 0.958 * | 0.960 * | 0.235 | −0.480 | 0.274 | 0.946 * | 1 | |||||
| Cl− | 0.223 | 0.364 | −0.375 | −0.389 | 0.429 | 0.979 * | 0.250 | −0.540 | −0.448 | 1 | ||||
| Ca2+ | 0.368 | 0.236 | 0.070 | 0.065 | 0.411 | 0.062 | 0.447 | 0.255 | 0.214 | 0.069 | 1 | |||
| Mg2+ | 0.426 | 0.387 | 0.033 | 0.041 | −0.005 | −0.118 | −0.075 | 0.146 | 0.102 | −0.254 | 0.271 | 1 | ||
| Na+ | −0.590 | −0.620 | −0.382 | −0.366 | −0.944 * | −0.231 | −0.952 * | −0.335 | −0.184 | −0.400 | −0.214 | 0.292 | 1 | |
| K+ | 0.716 | 0.606 | −0.440 | −0.447 | 0.387 | 0.107 | 0.474 | −0.224 | −0.440 | 0.162 | 0.643 | 0.109 | −0.326 | 1 |
| Geologic Formations | Locality | Sample ID | pH | EC | TDS | TSS | Alk. | F− | Cl− | NO3− | PO43− | SO42− | HCO3− | Na+ | K+ | Mg2+ | Ca2+ | NH4+ | TH |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| UC | µS/cm | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | |||
| Ampibolite | Mokolo | W1 | 6.31 | 215 | 123 | 65.2 | 736 | 0.06 | 9.34 | 34.22 | 0 | 1.81 | 44.90 | 12.23 | 3.41 | 5.80 | 16.83 | 0.21 | 65.86 |
| Kpok-ko | W2 | 5.88 | 40.1 | 23 | 21.6 | 303 | 0.05 | 0.72 | 1.11 | 0 | 0.05 | 18.48 | 3.20 | 1.31 | 0.93 | 2.36 | 0.22 | 9.72 | |
| Ganhi | W3 | 6.33 | 436 | 249 | 21.2 | 723 | 0.10 | 33.94 | 91.13 | 0 | 0.66 | 73.44 | 22.05 | 6.74 | 20.39 | 21.21 | 0.35 | 136.65 | |
| Granite | Gbakoungué | W4 | 5.93 | 20.4 | 12 | 11.6 | 156 | 0.03 | 0.13 | 1.34 | 0 | 0.08 | 9.52 | 1.46 | 0.54 | 0.35 | 1.79 | 0.23 | 5.92 |
| Ngaolaka | W5 | 8.12 | 136.2 | 78 | 13.2 | 1378 | 0.68 | 0.47 | 0.06 | 0 | 3.45 | 8.62 | 9.21 | 2.55 | 0.74 | 16.69 | 0.26 | 44.79 | |
| Gunbella | B1 | 5.93 | 34.2 | 20 | 16 | 307 | 0.04 | 0.05 | 0.06 | 0.01 | 0.09 | 19.48 | 1.57 | 0.94 | 1.10 | 2.80 | 0 | 11.51 | |
| Mbarang | B2 | 5.92 | 21.4 | 12 | 26.8 | 122 | 0.02 | 0.07 | 0.14 | 0.03 | 0.13 | 45.90 | 1.18 | 2.45 | 0.25 | 0.99 | 0.06 | 3.49 | |
| Gneiss | Mont ganga | S1 | 7.2 | 102.4 | 59 | 15.2 | 1183 | 0.06 | 0.03 | 0.02 | 0 | 0.01 | 72.16 | 6.34 | 0.66 | 4.48 | 8.52 | 0.11 | 39.67 |
| Zerkongo | S2 | 6.53 | 94.1 | 54 | 14.4 | 876 | 0.07 | 0.16 | 0.03 | 0.02 | 0.09 | 53.44 | 7.05 | 1.11 | 2.54 | 8.46 | 0.15 | 31.56 | |
| Kaladi | S3 | 5.4 | 15.1 | 9 | 15.2 | 140 | 0.01 | 0.08 | 0.02 | 0.09 | 0.01 | 8.54 | 1.43 | 0.69 | 0.15 | 0.76 | 0.10 | 2.54 | |
| Gbassoum | S4 | 6.08 | 56.6 | 32 | 16.2 | 534 | 0.05 | 0.04 | 1.01 | 0 | 0.13 | 63.16 | 2.85 | 0.84 | 2.91 | 3.80 | 0.06 | 21.46 | |
| Doua | B3 | 6.31 | 69.5 | 40 | 13.6 | 668 | 0.07 | 0.06 | 0.12 | 0.27 | 0.14 | 8.54 | 4.25 | 2.55 | 1.96 | 4.26 | 0 | 18.66 | |
| WHO Standard (2004) | DL | 6.5–8.5 | 500 | 500 | - | - | - | 200 | 10 | 5 | 200 | 200 | <20 | 12 | 50 | 75 | - | 100 | |
| PL | 9.2 | 1400 | 1000 | 1500 | 200 | 1.5 | 600 | 50 | 50 | 400 | 300 | 200 | 100 | 150 | 200 | 0.5 | 500 | ||
| Statistical data | |||||||||||||||||||
| Min | 5.4 | 15.1 | 9 | 11.6 | 122 | 0.01 | 0.03 | 0.02 | 0 | 0.01 | 8.54 | 1.18 | 0.54 | 0.15 | 0.76 | 0 | 2.54 | ||
| Max | 8.12 | 436 | 249 | 65.2 | 1378 | 0.68 | 33.94 | 91.13 | 0.27 | 3.45 | 73.44 | 22.05 | 6.74 | 20.39 | 21.21 | 0.35 | 136.65 | ||
| Mean | 6.33 | 103.42 | 59.25 | 20.85 | 593.83 | 0.1 | 3.76 | 10.77 | 0.08 | 0.55 | 35.51 | 6.07 | 1.98 | 3.47 | 7.37 | 0.15 | 32.65 | ||
| SD | 0.68 | 114.67 | 65.45 | 13.99 | 395.26 | 0.17 | 9.45 | 25.96 | 0.08 | 1.00 | 24.94 | 5.87 | 1.70 | 5.38 | 6.80 | 0.11 | 36.38 | ||
| CV (%) | 11 | 111 | 110 | 67 | 67 | 167 | 251 | 241 | 220 | 180 | 70 | 97 | 86 | 155 | 92 | 72 | 111.40 | ||
| pH | EC | TDS | TSS | Alk | F− | Cl− | NO3− | PO43− | SO4− | HCO3− | Na+ | NH4+ | K+ | Mg2+ | Ca2+ | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T | ||||||||||||||||
| pH | 1 | |||||||||||||||
| EC | 0.943 * | 1 | ||||||||||||||
| TDS | 0.943 * | 1.000 * | 1 | |||||||||||||
| TSS | −0.600 | −0.371 | −0.371 | 1 | ||||||||||||
| Alk | 1.000 * | 0.943 * | 0.943 * | −0.600 | 1 | |||||||||||
| F− | 1.000 * | 0.943 * | 0.943 * | −0.600 | 1.000 | 1 | ||||||||||
| Cl− | 0.543 | 0.600 | 0.600 | 0.086 | 0.543 | 0.543 | 1 | |||||||||
| NO3− | 0.086 | 0.314 | 0.314 | 0.657 | 0.086 | 0.086 | 0.257 | 1 | ||||||||
| PO43− | −0.516 | −0.516 | −0.516 | 0.030 | −0.516 | −0.516 | −0.213 | −0.334 | 1 | |||||||
| SO4− | 0.829 | 0.771 | 0.771 | −0.314 | 0.829 | 0.829 | 0.829 | 0.200 | −0.273 | 1 | ||||||
| HCO3− | −0.143 | 0.086 | 0.086 | 0.771 | −0.143 | −0.143 | 0.143 | 0.771 | −0.577 | −0.143 | 1 | |||||
| Na+ | 0.943 * | 1.000 * | 1.000 * | −0.371 | 0.943 * | 0.943 * | 0.600 | 0.314 | −0.516 | 0.771 | 0.086 | 1 | ||||
| NH4+ | 0.609 | 0.667 | 0.667 | 0.174 | 0.609 | 0.609 | 0.638 | 0.609 | −0.832 | 0.638 | 0.609 | 0.667 | 1 | |||
| K+ | 0.600 | 0.714 | 0.714 | −0.086 | 0.600 | 0.600 | 0.829 | 0.257 | 0.091 | 0.771 | −0.086 | 0.714 | 0.348 | 1 | ||
| Mg2+ | 0.371 | 0.600 | 0.600 | 0.086 | 0.371 | 0.371 | −0.029 | 0.600 | −0.395 | 0.029 | 0.486 | 0.600 | 0.348 | 0.257 | 1 | |
| Ca2+ | 0.943 * | 1.000 * | 1.000 * | −0.371 | 0.943* | 0.943* | 0.600 | 0.314 | −0.516 | 0.771 | 0.086 | 1.000 * | 0.667 | 0.714 | 0.600 | 1 |
| Geologic Formations | Sample ID | (Ca + Mg)/Total Cations | (Na + K)/Total Cations | (Ca + Mg)/(HCO3 + SO4) | (Ca + Mg)/HCO3 | Na/Cl | Na/Ca |
|---|---|---|---|---|---|---|---|
| Ampibolite | W1 | 0.68 | 0.32 | 1.69 | 1.78 | 2.04 | 0.63 |
| W2 | 0.53 | 0.45 | 0.67 | 0.67 | 7.00 | 1.17 | |
| W3 | 0.70 | 0.29 | 2.26 | 2.28 | 1.00 | 0.91 | |
| Granite | W4 | 0.60 | 0.35 | 0.75 | 0.75 | 15.00 | 0.67 |
| W5 | 0.65 | 0.34 | 4.24 | 6.36 | 30.77 | 0.48 | |
| B1 | 0.72 | 0.28 | 0.72 | 0.72 | 70 | 0.50 | |
| B2 | 0.39 | 0.61 | 0.09 | 0.09 | 25 | 1.00 | |
| Gneiss | S1 | 0.72 | 0.27 | 0.67 | 0.67 | 280 | 0.67 |
| S2 | 0.64 | 0.35 | 0.72 | 0.72 | 62 | 0.74 | |
| S3 | 0.36 | 0.57 | 0.36 | 0.36 | 30 | 1.50 | |
| S4 | 0.75 | 0.25 | 0.41 | 0.41 | 120 | 0.63 | |
| B3 | 0.59 | 0.41 | 2.64 | 2.64 | 95 | 0.90 | |
| Statistical data | |||||||
| Min | 0.36 | 0.25 | 0.09 | 0.36 | 1 | 0.48 | |
| Max | 0.75 | 0.61 | 4.24 | 6.36 | 280 | 1.50 | |
| Mean | 0.61 | 0.37 | 1.27 | 1.45 | 61.48 | 0.82 | |
| SD | 0.12 | 0.11 | 1.16 | 1.66 | 75.25 | 0.28 | |
| CV | 19.90 | 29.70 | 92.20 | 114.40 | 122.4 | 34.9 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dedzo, M.G.; Tsozué, D.; Mimba, M.E.; Teddy, F.; Nembungwe, R.M.; Linida, S. Importance of Rocks and Their Weathering Products on Groundwater Quality in Central-East Cameroon. Hydrology 2017, 4, 23. https://doi.org/10.3390/hydrology4020023
Dedzo MG, Tsozué D, Mimba ME, Teddy F, Nembungwe RM, Linida S. Importance of Rocks and Their Weathering Products on Groundwater Quality in Central-East Cameroon. Hydrology. 2017; 4(2):23. https://doi.org/10.3390/hydrology4020023
Chicago/Turabian StyleDedzo, Merlin Gountié, Désiré Tsozué, Mumbfu Ernestine Mimba, Fulbert Teddy, Romio Mofor Nembungwe, and Sylvie Linida. 2017. "Importance of Rocks and Their Weathering Products on Groundwater Quality in Central-East Cameroon" Hydrology 4, no. 2: 23. https://doi.org/10.3390/hydrology4020023
APA StyleDedzo, M. G., Tsozué, D., Mimba, M. E., Teddy, F., Nembungwe, R. M., & Linida, S. (2017). Importance of Rocks and Their Weathering Products on Groundwater Quality in Central-East Cameroon. Hydrology, 4(2), 23. https://doi.org/10.3390/hydrology4020023





