Effects of Lonicera japonica Flower Bud Extract on Citrobacter rodentium-Induced Digestive Tract Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. LJF Extract (LJFE) Preparation
2.2. Bacteria
2.3. Animal Study
2.4. Analysis of Ex Vivo Effects of LJFE on Immune Functions
2.5. Statistical Analysis
3. Results
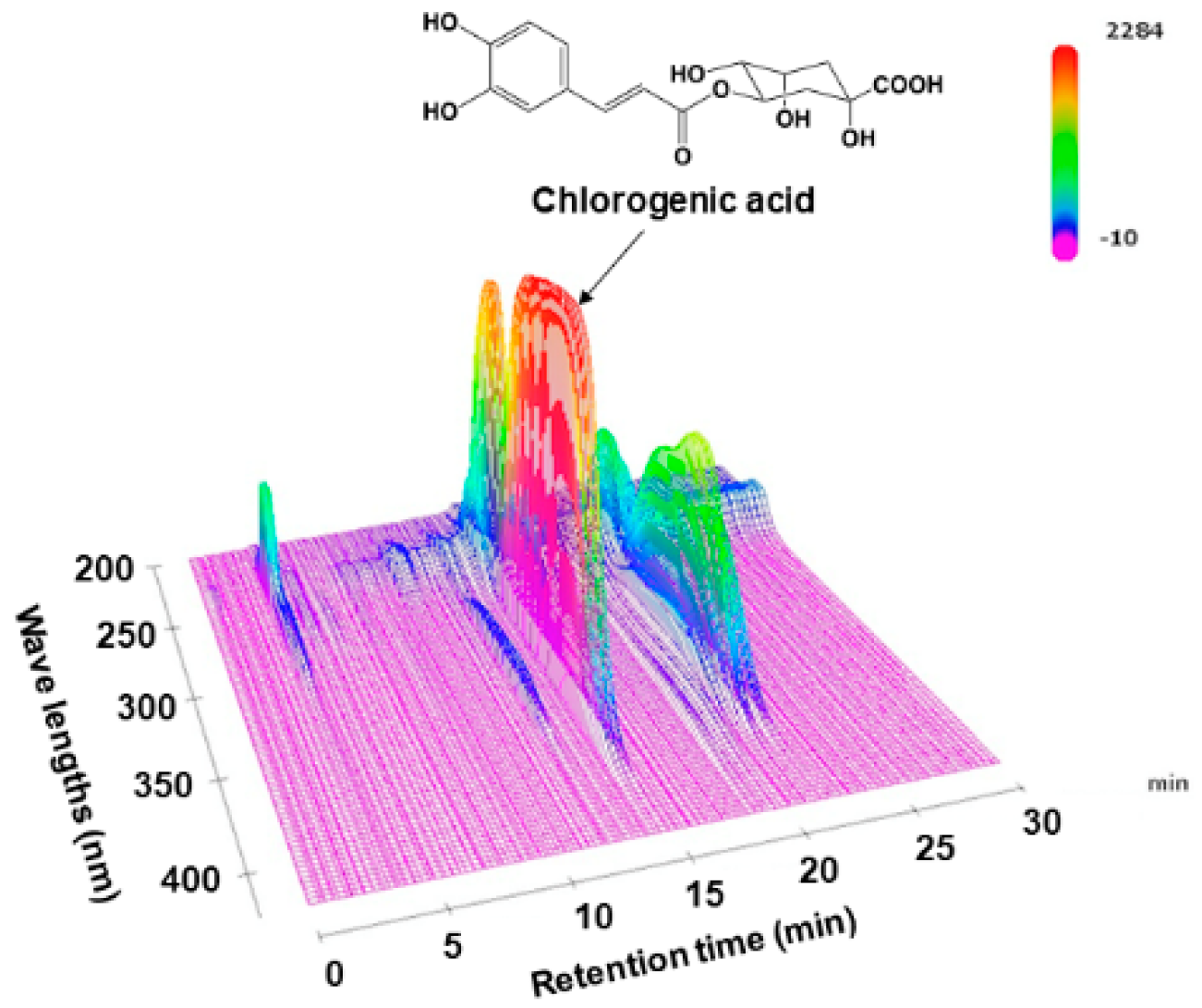
3.1. HPLC Chromatogram of LJFE
3.2. In Vitro Effects of LJFE against Citrobacter rodentium
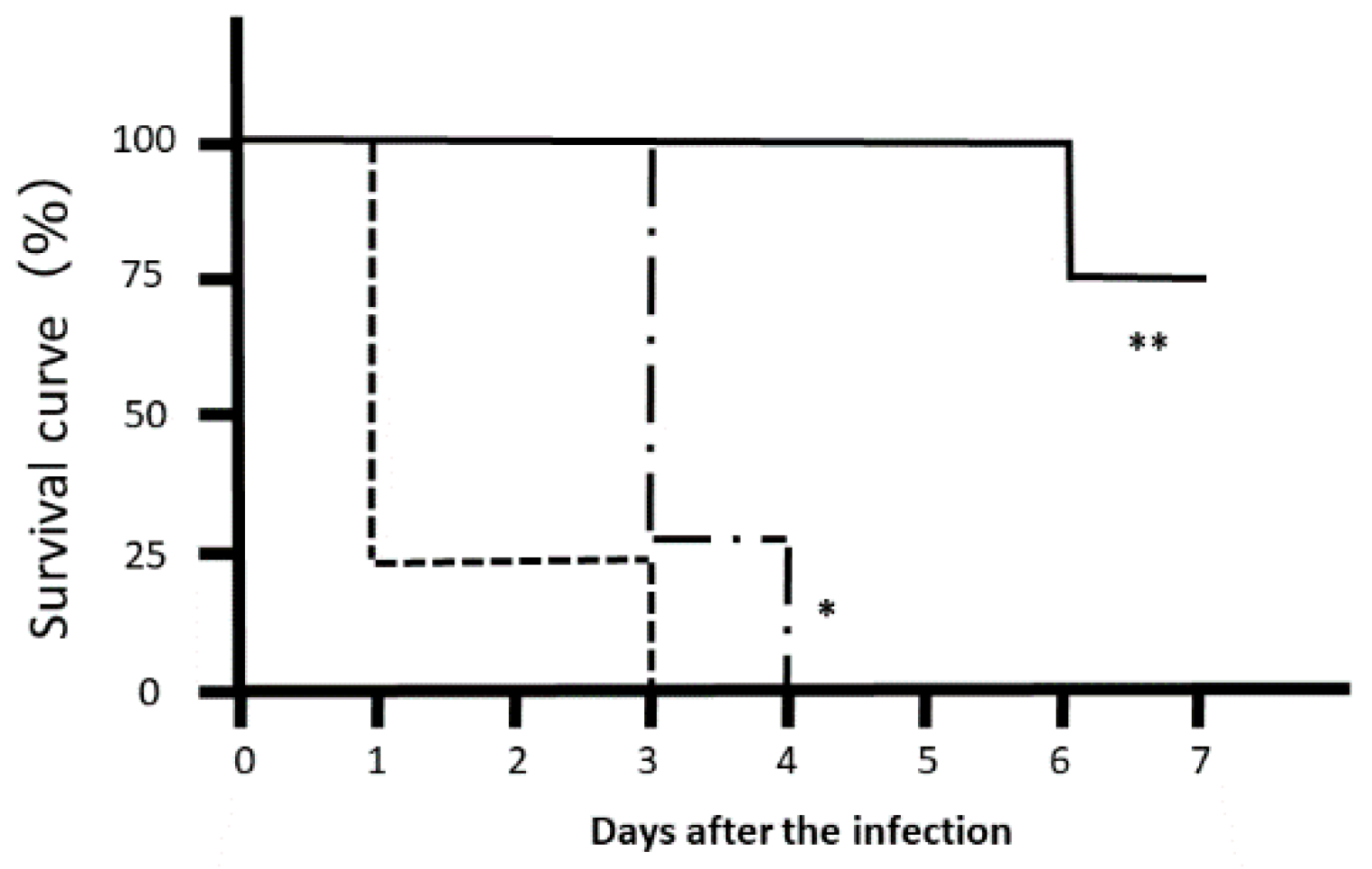
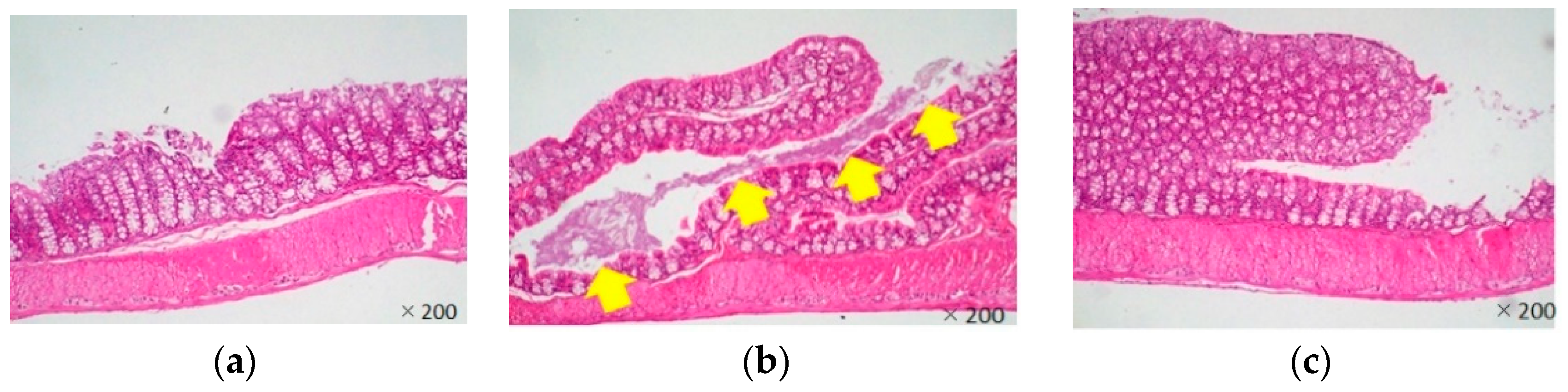
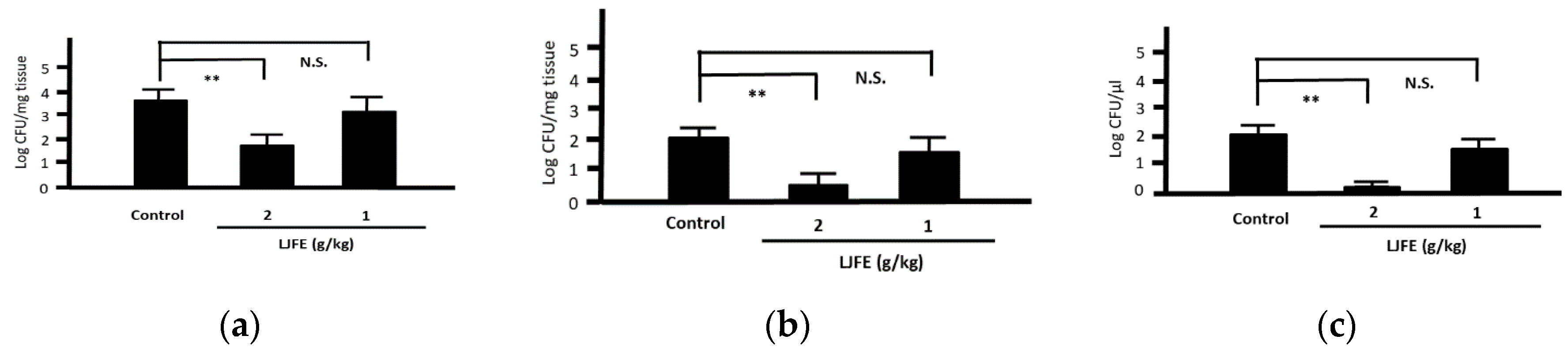
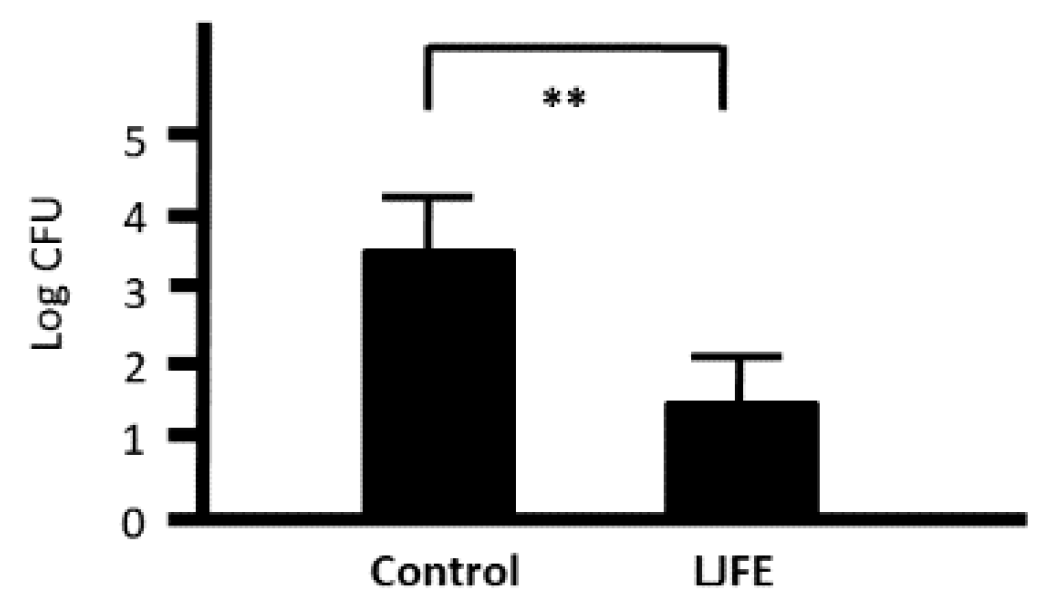
3.3. In Vivo Effects of LJFE against Citrobacter rodentium in a Mouse Model
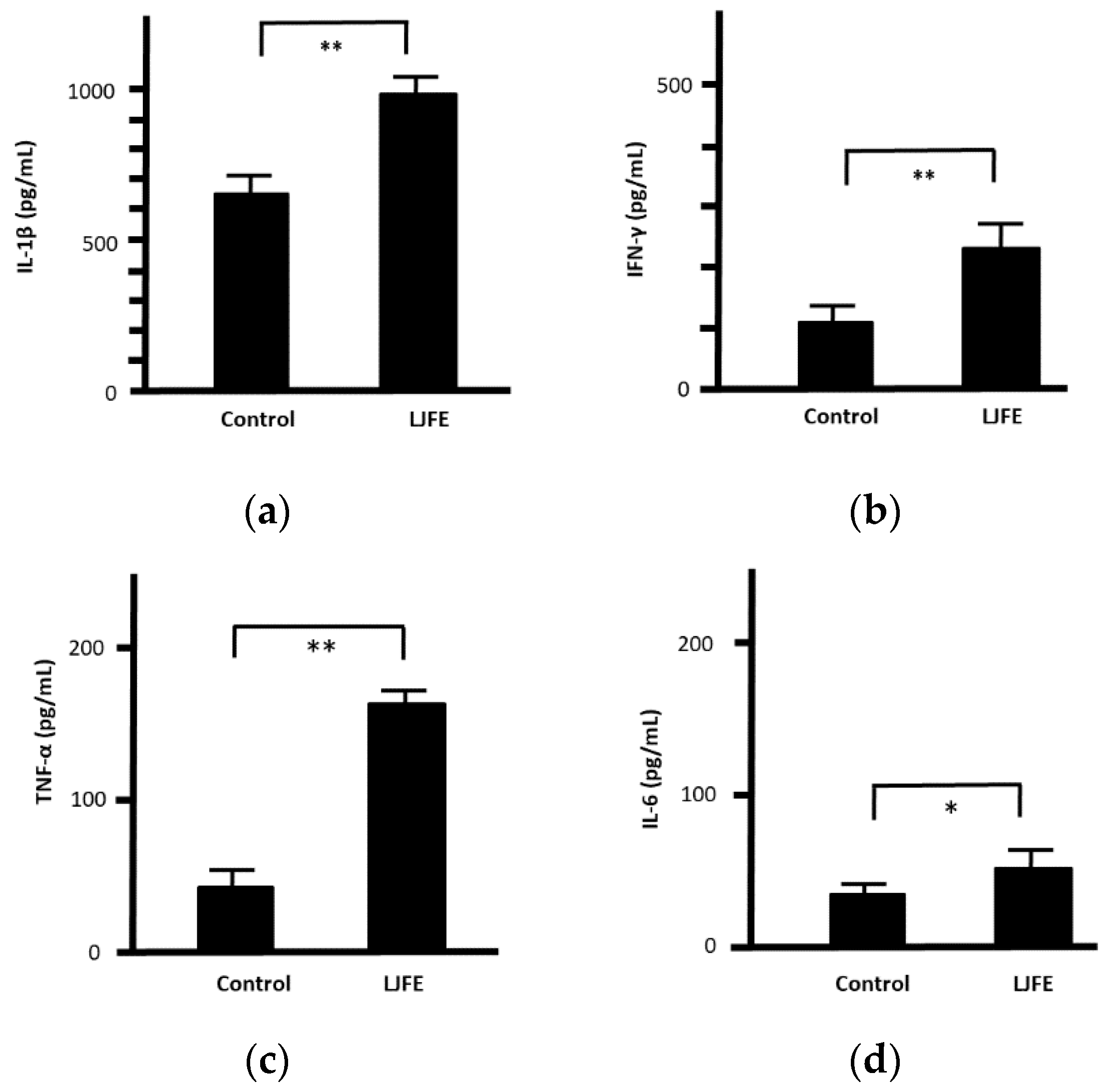
3.4. Effects of LJFE on Serum Cytokine Levels of Mice
3.5. Ex Vivo Effects of LJFE on the Phagocytic Activity of Peritoneal Macrophages against Citrobacter rodentium in Mice
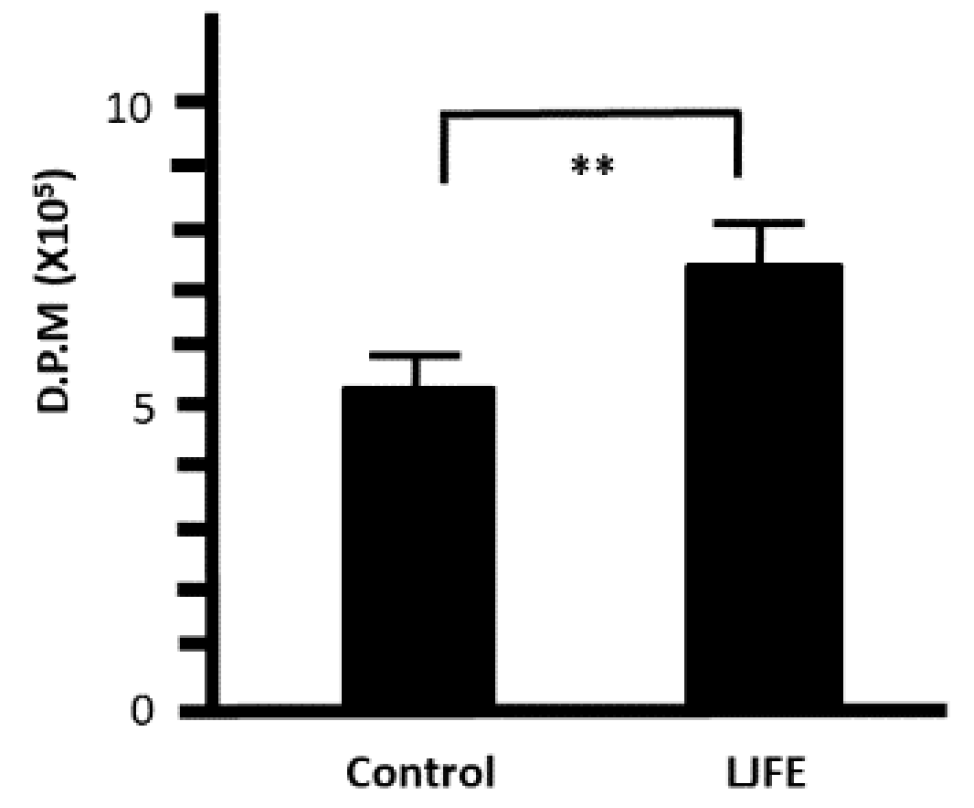
3.6. Ex Vivo Effects of LJFE on Peritoneal Macrophage Proliferation in Mice
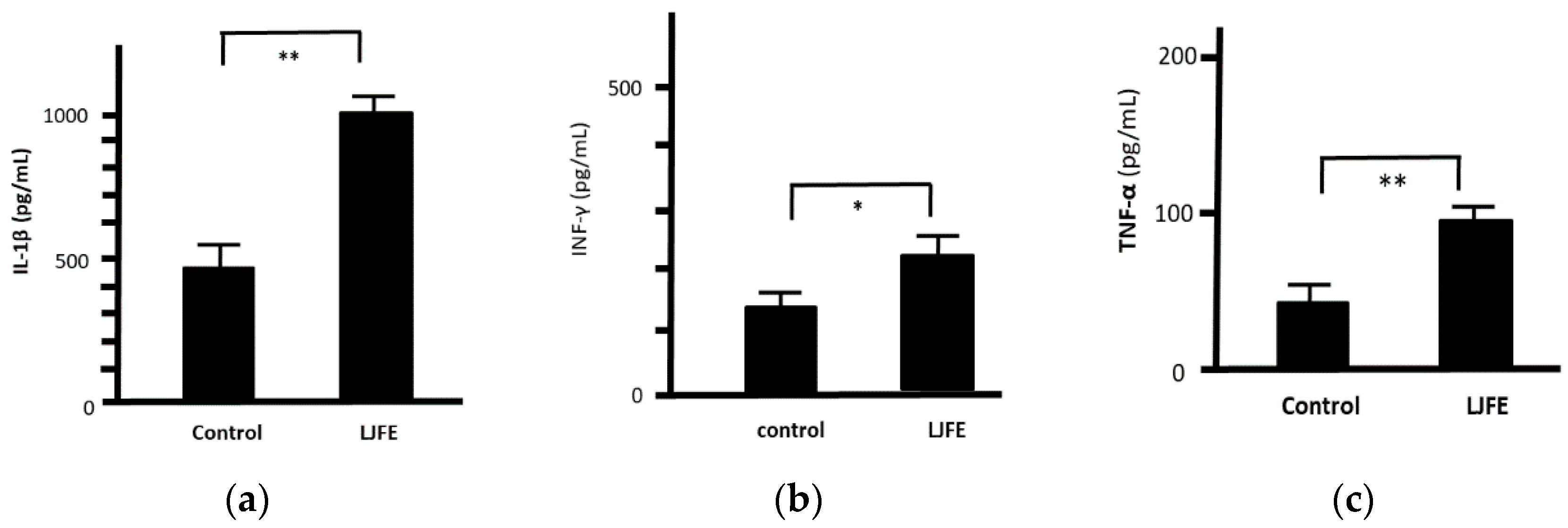
3.7. Ex Vivo Effects of LJFE on Cytokine Secretions from Peritoneal Macrophages in Mice
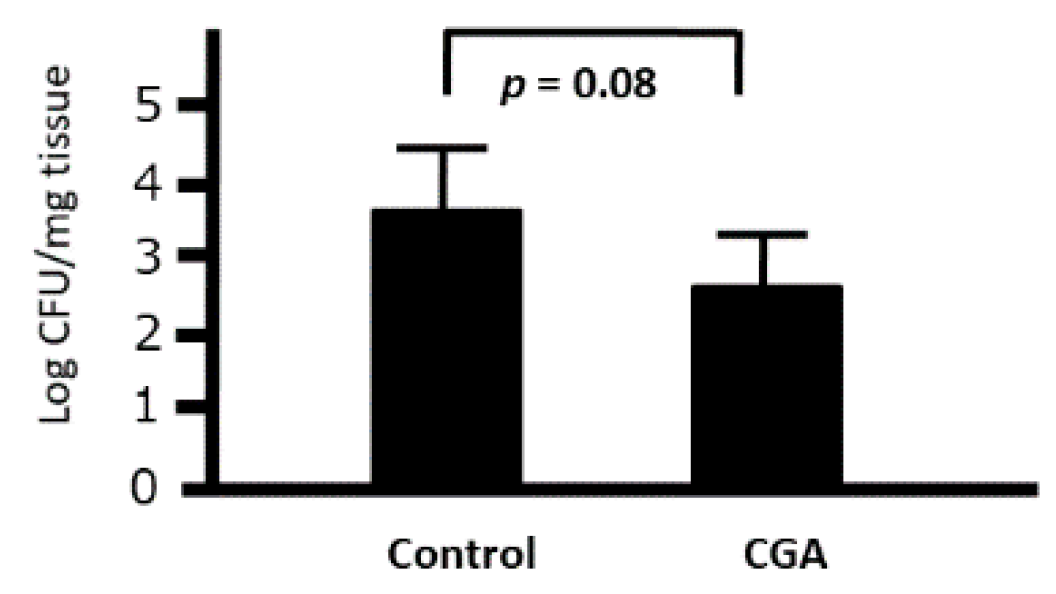
3.8. Ex Vivo Effects of CGA on the Phagocytic Activity of Peritoneal Macrophages against Citrobacter rodentium in Mice
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Navaneethan, U.; Giannella, R.A. Infectious colitis. Curr. Opin. Gastroenterol. 2011, 27, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Melton-Celsa, A.; Mohawk, K.; Louise Teel, L.; O’Brien, A. Pathogenesis of shiga-toxin producing Escherichia coli. Curr. Top. Microbiol. Immunol. 2012, 357, 67–103. [Google Scholar] [PubMed]
- Bush, K.; Bradford, P.A. Epidemiology of β-lactamase-producing pathogens. Clin. Microbiol. Rev. 2020, 33, e00047-19. [Google Scholar] [CrossRef] [PubMed]
- Halat, D.H.; Moubareck, C.A. The current burden of carbapenemases: Review of significant properties and dissemination among gram-negative bacteria. Antibiotics 2020, 9, 186. [Google Scholar] [CrossRef]
- Kakoullis, L.; Papachristodoulou, E.; Chra, P.; Panos, G. Shiga toxin-induced haemolytic uraemic syndrome and the role of antibiotics: A global overview. J. Infect. 2019, 79, 75–94. [Google Scholar] [CrossRef]
- Shang, X.; Pan, H.; Li, M.; Miao, X.; Ding, H. Lonicera japonica Thunb: Ethnopharmacology, phytochemistry and pharmacology of an important traditional Chinese medicine. J. Ethnopharmacol. 2011, 138, 1–21. [Google Scholar] [CrossRef]
- Luo, H.; Tang, Q.L.; Shang, Y.X.; Liang, S.B.; Yang, M.; Robinson, N.; Liu, J.P. Can Chinese medicine be used for prevention of corona virus disease 2019 (COVID-19)? A review of historical classics, research evidence and current prevention programs. Chin. J. Integr. Med. 2020, 26, 243–250. [Google Scholar] [CrossRef]
- Kao, S.T.; Liu, C.J.; Yeh, C.C. Protective and immunomodulatory effect of flos Lonicerae japonicae by augmenting IL-10 expression in a murine model of acute lung inflammation. J. Ethnopharmacol. 2015, 168, 108–115. [Google Scholar] [CrossRef]
- Mullineaux-Sanders, C.; Sanchez-Garrido, J.; Hopkins, E.G.D.; Shenoy, A.R.; Barry, R.; Frankel, G. Citrobacter rodentium-host-microbiota interactions: Immunity, bioenergetics and metabolism. Nat. Rev. Microbiol. 2019, 17, 701–715. [Google Scholar] [CrossRef]
- Eriguchi, Y.; Takashima, S.; Oka, H.; Shimoji, S.; Nakamura, K.; Uryu, H.; Shimoda, S.; Iwasaki, H.; Shimono, N.; Ayabe, T.; et al. Graft-versus-host disease disrupts intestinal microbial ecology by inhibiting Paneth cell production of α-defensins. Blood 2012, 120, 223–231. [Google Scholar] [CrossRef]
- Chang, Z.F.; Jia, D.X.; James, B. Chinese Material Media (International Standard Library of Chinese Medicine); People’s Medical Publishing House: Beijing, China, 2014; pp. 139–141. [Google Scholar]
- Minami, M.; Konishi, T.; Takase, H.; Jiang, Z.; Arai, T.; Makino, T. Effect of Shin’iseihaito (Xinyiqingfeitang) on acute Streptococcus pneumoniae murine sinusitis via macrophage activation. Evid. Based Complement. Altern. Med. 2017, 2017, 4293291. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Lee, Y.J.; Park, S.N. Synergistic antimicrobial effect of Lonicera japonica and Magnolia obovata extracts and potential as a plant-derived natural preservative. J. Microbiol. Biotechnol. 2018, 28, 1814–1822. [Google Scholar] [CrossRef] [PubMed]
- Li, R.J.; Kuang, X.P.; Wang, W.J.; Wan, C.P.; Li, W.X. Comparison of chemical constitution and bioactivity among different parts of Lonicera japonica Thunb. J. Sci. Food Agric. 2020, 100, 614–622. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Dong, Q.; Kan, X.; Peng, L.; Xu, X.; Fang, Y.; Yang, J. Immunomodulatory activity of a novel polysaccharide from Lonicera japonica in immunosuppressed mice induced by cyclophosphamide. PLoS ONE 2018, 13, e0204152. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Roh, E.; Kim, H.S.; Baek, S.I.; Choi, N.S.; Kim, N.; Hwang, B.Y.; Han, S.B.; Kim, Y. Inhibition of IRAK-4 activity for rescuing endotoxin LPS-induced septic mortality in mice by lonicerae flos extract. Biochem. Biophys. Res. Commun. 2013, 442, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Zhang, L.; Huang, H. Study on the isolation of active constituents in Lonicera japonica and the mechanism of their anti-upper respiratory tract infection action in children. Afr. Health Sci. 2015, 15, 1295–1301. [Google Scholar] [CrossRef]
- Wu, H.Z.; Luo, J.; Yin, Y.X.; Wei, Q. Effects of chlorogenic acid, an active compound activating calcineurin, purified from Flos Lonicerae on macrophage. Acta Pharmacol. Sin. 2004, 25, 1685–1689. [Google Scholar]
- Lee, C.H.; Yoon, S.J.; Lee, S.M. Chlorogenic acid attenuates high mobility group box 1 (HMGB1) and enhances host defense mechanisms in murine sepsis. Mol. Med. 2013, 18, 1437–1448. [Google Scholar] [CrossRef]
- Krakauer, T. The polyphenol chlorogenic acid inhibits staphylococcal exotoxin-induced inflammatory cytokines and chemokines. Immunopharmacol. Immunotoxicol. 2002, 24, 113–119. [Google Scholar] [CrossRef]
- Shan, J.; Fu, J.; Zhao, Z.; Kong, X.; Huang, H.; Luo, L.; Yin, Z. Chlorogenic acid inhibits lipopolysaccharide-induced cyclooxygenase-2 expression in RAW264.7 cells through suppressing NF-kappaB and JNK/AP-1 activation. Int. Immunopharmacol. 2009, 9, 1042–1048. [Google Scholar] [CrossRef]
- Hwang, S.J.; Kim, Y.W.; Park, Y.; Lee, H.J.; Kim, K.W. Anti-inflammatory effects of chlorogenic acid in lipopolysaccharide-stimulated RAW 264.7 cells. Inflamm. Res. 2014, 63, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Park, S.Y.; Park, Y.L.; Myung, D.S.; Rew, J.S.; Joo, Y.E. Chlorogenic acid suppresses lipopolysaccharide-induced nitric oxide and interleukin-1β expression by inhibiting JAK2/STAT3 activation in RAW264.7 cells. Mol. Med. Rep. 2017, 16, 9224–9232. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Bae, H.; Lee, G.; Hong, B.G.; Yoo, H.H.; Lim, S.J.; Lee, K.; Kim, J.; Ryu, B.; Lee, B.J.; et al. Prophylactic effects of Lonicera japonica extract on dextran sulphate sodium-induced colitis in a mouse model by the inhibition of the Th1/Th17 response. Br. J. Nutr. 2013, 109, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Yu, X.; Sun, H.; Zhang, W.; Liu, G.; Zhu, L. Flos lonicerae flavonoids attenuate experimental ulcerative colitis in rats via suppression of NF-κB signaling pathway. Naunyn Schmiedebergs Arch. Pharmacol. 2020, 3. [Google Scholar] [CrossRef] [PubMed]
- Peterson, C.T.; Sharma, V.; Uchitel, S.; Denniston, K.; Chopra, D.; Mills, P.J.; Peterson, S.N. Prebiotic potential of herbal medicines used in digestive health and disease. J. Altern. Complement. Med. 2018, 24, 656–665. [Google Scholar] [CrossRef]
- Rahman, N.; Ahmad, M.; Riaz, M.; Mehjabeen; Jahan, N.; Ahmad, R. Phytochemical, antimicrobial, insecticidal and brine shrimp lethality bioassay of the crude methanolic extract of Ajuga parviflora Benth. Pak. J. Pharm. Sci. 2013, 26, 751–756. [Google Scholar]
- Chehregani, A.; Atri, M.; Yousefi, S.; Albooyeh, Z.; Mohsenzadeh, F. Essential oil variation in the populations of Artemisia spicigera from northwest of Iran: Chemical composition and antibacterial activity. Pharm. Biol. 2013, 51, 246–252. [Google Scholar] [CrossRef][Green Version]
- Alam, N.; Hossain, M.; Mottalib, M.A.; Sulaiman, S.A.; Gan, S.H.; Khalil, M.I. Methanolic extracts of Withania somnifera leaves, fruits and roots possess antioxidant properties and antibacterial activities. BMC Complement. Altern. Med. 2012, 12, 175. [Google Scholar] [CrossRef]
- Kumarasamy, Y.; Nahar, L.; Byres, M.; Delazar, A.; Sarker, S.D. The assessment of biological activities associated with the major constituents of the methanol extract of ‘wild carrot’ (Daucus carota L.) seeds. J. Herb. Pharmacother. 2005, 5, 61–72. [Google Scholar]
- Zhang, R.; Gilbert, S.; Yao, X.; Vallance, J.; Steinbrecher, K.; Moriggl, R.; Zhang, D.; Eluri, M.; Chen, H.; Cao, H.; et al. Natural compound methyl protodioscin protects against intestinal inflammation through modulation of intestinal immune responses. Pharmacol. Res. Perspect. 2015, 3, e00118. [Google Scholar] [CrossRef]
- Li, H.J.; Li, P.; Ye, W.C. Determination of five major iridoid glucosides in Flos Lonicerae by high-performance liquid chromatography coupled with evaporative light scattering detection. J. Chromatogr. A 2003, 1008, 167–172. [Google Scholar] [CrossRef]
- Ziyan, L.; Yongmei, Z.; Nan, Z.; Ning, T.; Baolin, L. Evaluation of the anti-inflammatory activity of luteolin in experimental animal models. Planta Med. 2007, 73, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.A.; Kim, W.K.; Park, H.J.; Kang, S.S.; Lee, S.K. Anti-proliferative activity of hydnocarpin, a natural lignan, is associated with the suppression of Wnt/β-catenin signaling pathway in colon cancer cells. Bioorg. Med. Chem. Lett. 2013, 23, 5511–5514. [Google Scholar] [CrossRef]
- Vimberg, V.; Kuzma, M.; Stodůlková, E.; Novák, P.; Bednárová, L.; Šulc, M.; Gažák, R. Hydnocarpin-type flavonolignans: Semisynthesis and inhibitory effects on Staphylococcus aureus biofilm formation. J. Nat. Prod. 2015, 78, 2095–2103. [Google Scholar] [CrossRef] [PubMed]
- Kwak, W.J.; Han, C.K.; Chang, H.W.; Kim, H.P.; Kang, S.S.; Son, K.H. Loniceroside C, an antiinflammatory saponin from Lonicera japonica. Chem. Pharm. Bull. 2003, 51, 333–335. [Google Scholar] [CrossRef]
- Wang, J.W.; Pan, Y.B.; Cao, Y.Q.; Wang, C.; Jiang, W.D.; Zhai, W.F.; Lu, J.G. Loganin alleviates LPS-activated intestinal epithelial inflammation by regulating TLR4/NF-κB and JAK/STAT3 signaling pathways. Kaohsiung J. Med. Sci. 2020, 36, 257–264. [Google Scholar]
- Kang, O.H.; Choi, J.G.; Lee, J.H.; Kwon, D.Y. Luteolin isolated from the flowers of Lonicera japonica suppresses inflammatory mediator release by blocking NF-kappaB and MAPKs activation pathways in HMC-1 cells. Molecules 2010, 15, 385–398. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Minami, M.; Makino, T. Effects of Lonicera japonica Flower Bud Extract on Citrobacter rodentium-Induced Digestive Tract Infection. Medicines 2020, 7, 52. https://doi.org/10.3390/medicines7090052
Minami M, Makino T. Effects of Lonicera japonica Flower Bud Extract on Citrobacter rodentium-Induced Digestive Tract Infection. Medicines. 2020; 7(9):52. https://doi.org/10.3390/medicines7090052
Chicago/Turabian StyleMinami, Masaaki, and Toshiaki Makino. 2020. "Effects of Lonicera japonica Flower Bud Extract on Citrobacter rodentium-Induced Digestive Tract Infection" Medicines 7, no. 9: 52. https://doi.org/10.3390/medicines7090052
APA StyleMinami, M., & Makino, T. (2020). Effects of Lonicera japonica Flower Bud Extract on Citrobacter rodentium-Induced Digestive Tract Infection. Medicines, 7(9), 52. https://doi.org/10.3390/medicines7090052





