Direct-Acting Oral Anticoagulants and Their Reversal Agents—An Update
Abstract
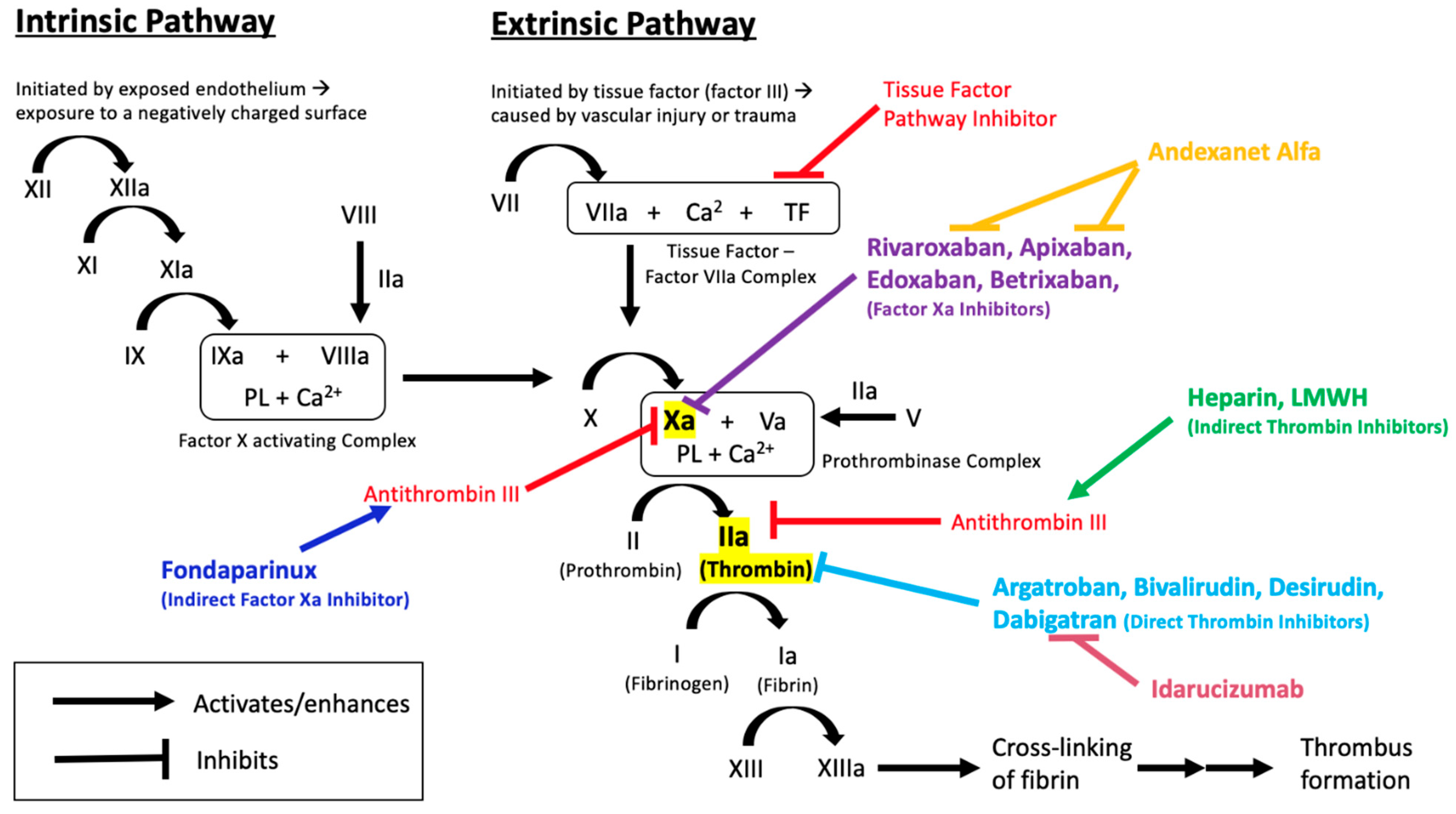
1. Introduction
2. Materials and Methods
3. Results
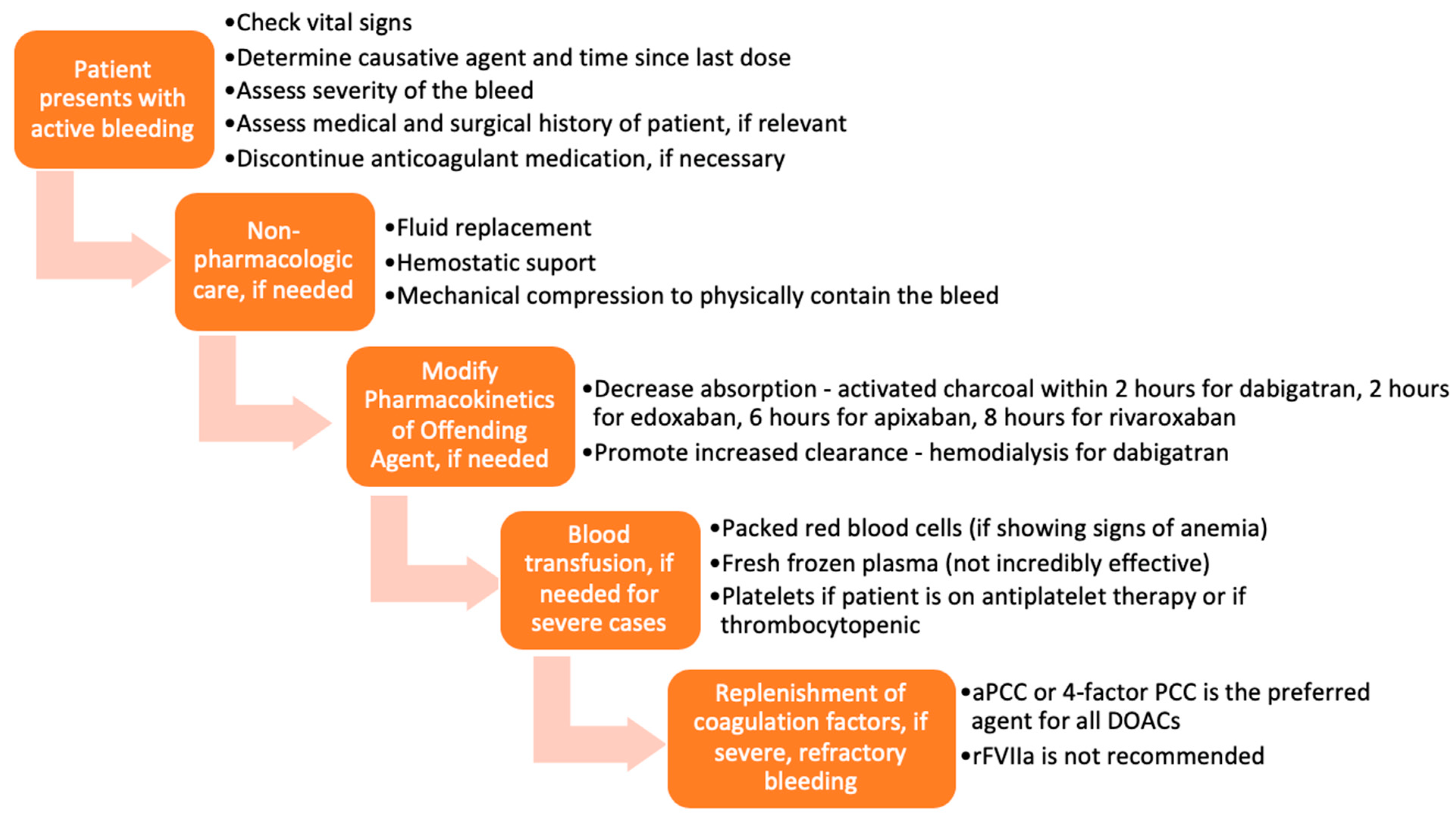
3.1. Before the Advent of Approved Reversal Agents
3.2. Direct Thrombin Inhibitor–Dabigatran—And Its Reversal
3.3. Factor Xa Inhibitors and Their Reversal
3.3.1. Rivaroxaban
3.3.2. Apixaban
3.3.3. Edoxaban
3.3.4. Betrixaban
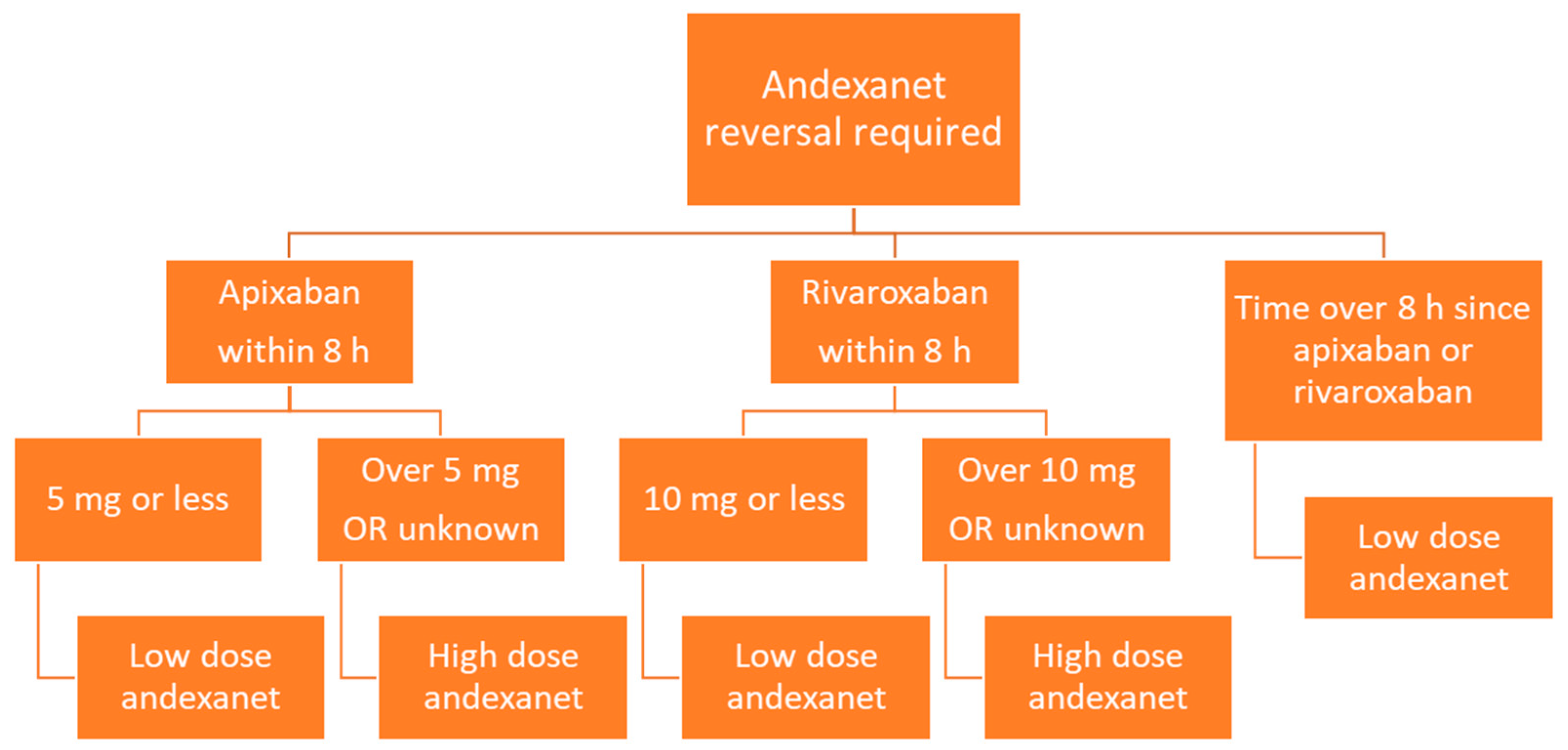
3.3.5. Andexanet Alfa
3.4. Reversal Agents in Development—Ciraparantag
4. Discussion
5. Conclusions
Funding
Conflicts of Interest
References
- Serhal, M.; Barnes, G.D. Venous thromboembolism: A clinician update. Vasc. Med. 2019, 24, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Busch, M.; Masuhr, F. Thromboprophylaxis and early antithrombotic therapy in patients with acute ischemic stroke a37nd cerebral venous and sinus thrombosis. Eur. J. Med. Res. 2004, 9, 99–206. [Google Scholar]
- Hirsh, J.; Warkentin, T.E.; Dalen, J.E.; Deykin, D.; Poller, L. Heparin: Mechanism of action, pharmacokinetics, dosing considerations, monitoring, efficacy, and safety. Chest 1995, 108, 258S–275S. [Google Scholar] [CrossRef] [PubMed]
- Sokolowska, E.; Kalaska, B.; Miklosz, J.; Mogielnicki, A. The toxicology of heparin reversal with protamine: Past, present and future. Expert Opin. Drug Metab. Toxicol. 2016, 12, 897–909. [Google Scholar] [CrossRef]
- Lee, C.J.; Ansell, J.E. Direct thrombin inhibitors. Br. J. Clin. Pharmacol. 2011, 72, 581–592. [Google Scholar] [CrossRef]
- Whitlon, D.S.; Sadowski, J.A.; Suttie, J.W. Mechanism of coumarin action: Significance of vitamin K epoxide reductase inhibition. Biochemistry 1978, 17, 1371–1377. [Google Scholar] [CrossRef]
- Wadhera, R.K.; Russell, C.E.; Piazza, G. Warfarin versus novel oral anticoagulants: How to choose? Circulation 2014, 130, e191–e193. [Google Scholar] [CrossRef]
- Yeh, C.H.; Gross, P.L.; Weitz, J.I. Evolving use of new oral anticoagulants for treatment of venous thromboembolism. Blood 2014, 124, 1020–1028. [Google Scholar] [CrossRef]
- Mekaj, Y.H.; Mekaj, A.Y.; Duci, S.B.; Miftari, E.I. New oral anticoagulants: Their advantages and disadvantages compared with vitamin K antagonists in the prevention and treatment of patients with thromboembolic events. Ther. Clin. Risk Manag. 2015, 11, 967. [Google Scholar] [CrossRef]
- Beyer-Westendorf, J.; Forster, K.; Pannach, S.; Ebertz, F.; Gelbricht, V.; Thieme, C.; Michalski, F.; Köhler, C.; Werth, S.; Sahin, K.; et al. Rates, management, and outcome of rivaroxaban bleeding in daily care: Results from the Dresden NOAC registry. Blood 2014, 124, 955–962. [Google Scholar] [CrossRef]
- Castellucci, L.A.; Cameron, C.; Le Gal, G.; Rodger, M.A.; Coyle, D.; Wells, P.S.; Clifford, T.; Gandara, E.; Wells, G.; Carrier, M. Clinical and safety outcomes associated with treatment of acute venous thromboembolism: A systematic review and meta-analysis. JAMA 2014, 312, 1122–1135. [Google Scholar] [CrossRef] [PubMed]
- Lip, G.Y.; Keshishian, A.; Kamble, S.; Pan, X.; Mardekian, J.; Horblyuk, R.; Hamilton, M. Real-world comparison of major bleeding risk among non-valvular atrial fibrillation patients initiated on apixaban, dabigatran, rivaroxaban, or warfarin. A propensity score matched analysis. Thromb. Haemost. 2016, 116, 975–986. [Google Scholar] [CrossRef]
- Dabi, A.; Koutrouvelis, A.P. Reversal strategies for intracranial hemorrhage related to direct oral anticoagulant medications. Crit. Care Res. Pract. 2018, 2018, 4907164. [Google Scholar] [CrossRef] [PubMed]
- Kaatz, S.; Kouides, P.A.; Garcia, D.A.; Spyropolous, A.C.; Crowther, M.; Douketis, J.D.; Chan, A.K.; James, A.; Moll, S.; Ortel, T.L.; et al. Guidance on the emergent reversal of oral thrombin and factor Xa inhibitors. Am. J. Hematol. 2012, 87, S141–S145. [Google Scholar] [CrossRef] [PubMed]
- Steiner, T.; Böhm, M.; Dichgans, M.; Diener, H.C.; Ell, C.; Endres, M.; Epple, C.; Grond, M.; Laufs, U.; Nickenig, G.; et al. Recommendations for the emergency management of complications associated with the new direct oral anticoagulants (DOACs), apixaban, dabigatran and rivaroxaban. Clin. Res. Cardiol. 2013, 102, 399–412. [Google Scholar] [CrossRef]
- Christos, S.; Naples, R. Anticoagulation reversal and treatment strategies in major bleeding: Update 2016. West. J. Emerg. Med. 2016, 17, 264. [Google Scholar] [CrossRef]
- Levine, M.; Goldstein, J.N. Emergency reversal of anticoagulation: Novel agents. Curr. Neurol. Neurosci. Rep. 2014, 14, 471. [Google Scholar] [CrossRef]
- Woo, J.S.; Kapadia, N.; Phanco, S.E.; Lynch, C.A. Positive outcome after intentional overdose of dabigatran. J. Med. Toxicol. 2013, 9, 192–195. [Google Scholar] [CrossRef]
- van Ryn, J.; Ruehl, D.; Priepke, H.; Hauel, N.; Wienen, W. Reversibility of the anticoagulant effect of high doses of the direct thrombin inhibitor dabigatran, by recombinant Factor VIIa or activated prothrombin complex concentrate. Haemato Logica 2008, 93, S148. [Google Scholar]
- Wang, X.; Mondal, S.; Wang, J.; Tirucherai, G.; Zhang, D.; Boyd, R.A.; Frost, C. Effect of activated charcoal on apixaban pharmacokinetics in healthy subjects. Am. J. Cardiovasc. Drugs 2014, 14, 147–154. [Google Scholar] [CrossRef]
- Ollier, E.; Hodin, S.; Lanoiselée, J.; Escal, J.; Accassat, S.; De Magalhaes, E.; Basset, T.; Bertoletti, L.; Mismetti, P.; Delavenne, X. Effect of activated charcoal on rivaroxaban complex absorption. Clin. Pharm. 2017, 56, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.C.; Sheth, N.R.; Dadzie, K.A.; Smith, S.W.; Nelson, L.S.; Hoffman, R.S.; Winchester, J.F. Hemodialysis for the treatment of pulmonary hemorrhage from dabigatran overdose. Am. J. Kidney Dis. 2013, 62, 591–594. [Google Scholar] [CrossRef] [PubMed]
- Maw, T.T.; Henry, B.L.; Singh, T. Use of hemodialysis for the treatment of intracerebral hemorrhage in patients on dabigatran with normal renal function. Clin. Nephrol. Case Stud. 2014, 2, 18. [Google Scholar] [CrossRef] [PubMed]
- Parasrampuria, D.A.; Marbury, T.; Matsushima, N.; Chen, S.; Wickremasingha, P.K.; He, L.; Dishy, V.; Brown, K.S. Pharmacokinetics, safety, and tolerability of edoxaban in end-stage renal disease subjects undergoing haemodialysis. Thromb. Haemost. 2015, 113, 719–727. [Google Scholar]
- Grandhi, R.; Newman, W.C.; Zhang, X.; Harrison, G.; Moran, C.; Okonkwo, D.O.; Ducruet, A.F. Administration of 4-factor prothrombin complex concentrate as an antidote for intracranial bleeding in patients taking direct factor Xa inhibitors. World Neurosurg. 2015, 84, 1956–1961. [Google Scholar] [CrossRef]
- Schulman, S.; Ritchie, B.; Goy, J.K.; Nahirniak, S.; Almutawa, M.; Ghanny, S. Activated prothrombin complex concentrate for dabigatran-associated bleeding. Br. J. Haematol. 2014, 164, 308–310. [Google Scholar] [CrossRef]
- Demeyere, R.; Gillardin, S.; Arnout, J.; Strengers, P.F. Comparison of fresh frozen plasma and prothrombin complex concentrate for the reversal of oral anticoagulants in patients undergoing cardiopulmonary bypass surgery: A randomized study. Vox Sang. 2010, 99, 251–260. [Google Scholar] [CrossRef]
- Dumkow, L.E.; Voss, J.R.; Peters, M.; Jennings, D.L. Reversal of dabigatran-induced bleeding with a prothrombin complex concentrate and fresh frozen plasma. Am. J. Health Syst. Pharm. 2012, 69, 1646–1650. [Google Scholar] [CrossRef]
- Cohen, H. Avoiding the misuse of fresh frozen plasma. BMJ 1993, 14, 307–395. [Google Scholar] [CrossRef][Green Version]
- Perzborn, E.; Heitmeier, S.; Laux, V.; Buchmüller, A. Reversal of rivaroxaban-induced anticoagulation with prothrombin complex concentrate, activated prothrombin complex concentrate and recombinant activated factor VII in vitro. Thromb. Res. 2014, 133, 671–681. [Google Scholar] [CrossRef]
- Martin, A.C.; Le Bonniec, B.; Fischer, A.M.; Marchand-Leroux, C.; Gaussem, P.; Samama, C.M.; Godier, A. Evaluation of recombinant activated factor VII, prothrombin complex concentrate, and fibrinogen concentrate to reverse apixaban in a rabbit model of bleeding and thrombosis. Int. J. Cardiol. 2013, 168, 4228–4233. [Google Scholar] [CrossRef] [PubMed]
- Harter, K.; Levine, M.; Henderson, S.O. Anticoagulation drug therapy: A review. West. J. Emerg. Med. 2015, 16, 11. [Google Scholar] [CrossRef] [PubMed]
- Hunt, B.J. The current place of tranexamic acid in the management of bleeding. Anaesthesia 2015, 70, 50-e18. [Google Scholar] [CrossRef] [PubMed]
- Moore, T.J.; Cohen, M.R.; Mattison, D.R. Dabigatran, bleeding, and the regulators. BMJ 2014, 349, g4517. [Google Scholar] [CrossRef]
- Stangier, J.; Rathgen, K.; Stähle, H.; Gansser, D.; Roth, W. The pharmacokinetics, pharmacodynamics and tolerability of dabigatran etexilate, a new oral direct thrombin inhibitor, in healthy male subjects. Br. J. Clin. Pharmacol. 2007, 64, 292–303. [Google Scholar] [CrossRef]
- Trocóniz, I.F.; Tillmann, C.; Liesenfeld, K.H.; Schäfer, H.G.; Stangier, J. Population pharmacokinetic analysis of the new oral thrombin inhibitor dabigatran etexilate (BIBR 1048) in patients undergoing primary elective total hip replacement surgery. J. Clin. Pharmacol. 2007, 47, 371–382. [Google Scholar] [CrossRef]
- Blech, S.; Ebner, T.; Ludwig-Schwellinger, E.; Stangier, J.; Roth, W. The metabolism and disposition of the oral direct thrombin inhibitor, dabigatran, in humans. Drug Metab. Dispos. 2008, 36, 386–399. [Google Scholar] [CrossRef]
- Stangier, J.; Stähle, H.; Rathgen, K.; Fuhr, R. Pharmacokinetics and pharmacodynamics of the direct oral thrombin inhibitor dabigatran in healthy elderly subjects. Clin. Pharm. 2008, 47, 47–59. [Google Scholar] [CrossRef]
- Alquwaizani, M.; Buckley, L.; Adams, C.; Fanikos, J. Anticoagulants: A review of the pharmacology, dosing, and complications. Curr. Emerg. Hosp. Med. Rep. 2013, 1, 83–97. [Google Scholar] [CrossRef]
- Hankey, G.J.; Eikelboom, J.W. Dabigatran etexilate: A new oral thrombin inhibitor. Circulation 2011, 123, 1436–1450. [Google Scholar] [CrossRef]
- Ebner, T.; Wagner, K.; Wienen, W. Dabigatran acylglucuronide, the major human metabolite of dabigatran: In vitro formation, stability, and pharmacological activity. Drug Metab. Dispos. 2010, 38, 1567–1575. [Google Scholar] [CrossRef] [PubMed]
- Stangier, J.; Stähle, H.; Rathgen, K.; Roth, W.; Shakeri-Nejad, K. Pharmacokinetics and pharmacodynamics of dabigatran etexilate, an oral direct thrombin inhibitor, are not affected by moderate hepatic impairment. J. Clin. Pharmacol. 2008, 48, 1411–1419. [Google Scholar] [CrossRef] [PubMed]
- Stangier, J.; Rathgen, K.; Stähle, H.; Mazur, D. Influence of renal impairment on the pharmacokinetics and pharmacodynamics of oral dabigatran etexilate. Clin. Pharm. 2010, 49, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Härtter, S.; Sennewald, R.; Nehmiz, G.; Reilly, P. Oral bioavailability of dabigatran etexilate (Pradaxa®) after co-medication with verapamil in healthy subjects. Br. J. Clin. Pharmacol. 2013, 75, 1053–1062. [Google Scholar] [CrossRef]
- Eriksson, B.I.; Dahl, O.E.; Ahnfelt, L.; Kälebo, P.; Stangier, J.; Nehmiz, G.; Hermansson, K.; Kohlbrenner, V. Dose escalating safety study of a new oral direct thrombin inhibitor, dabigatran etexilate, in patients undergoing total hip replacement: BISTRO I. J. Thromb. Haemost. 2004, 2, 1573–1580. [Google Scholar] [CrossRef]
- Beyer-Westendorf, J.; Ebertz, F.; Foerster, K.; Gelbricht, V.; Michalski, F.; Köhler, C.; Werth, S.; Endig, H.; Pannach, S.; Tittl, L.; et al. Effectiveness and safety of dabigatran therapy in daily-care patients with atrial fibrillation. Thromb. Haemost. 2015, 113, 1247–1257. [Google Scholar]
- Southworth, M.R.; Reichman, M.E.; Unger, E.F. Dabigatran and postmarketing reports of bleeding. N. Engl. J. Med. 2013, 368, 1272–1274. [Google Scholar] [CrossRef]
- Lillo-Le Louët, A.; Wolf, M.; Soufir, L.; Galbois, A.; Dumenil, A.S.; Offenstadt, G.; Samama, M.M. Life-threatening bleeding in four patients with an unusual excessive response to dabigatran: Implications for emergency surgery and resuscitation. Thromb. Haemost. 2012, 108, 583–585. [Google Scholar]
- Enriquez, A.; Baranchuk, A.; Redfearn, D.; Simpson, C.; Abdollah, H.; Michael, K. Dabigatran for the prevention and treatment of thromboembolic disorders. Exp. Rev. Cardiovasc. Ther. 2015, 13, 529–540. [Google Scholar] [CrossRef]
- Ganetsky, M.; Babu, K.M.; Salhanick, S.D.; Brown, R.S.; Boyer, E.W. Dabigatran: Review of pharmacology and management of bleeding complications of this novel oral anticoagulant. J. Med. Toxicol. 2011, 7, 281–287. [Google Scholar] [CrossRef]
- Schiele, F.; van Ryn, J.; Newsome, C.; Sepulveda, E.; Park, J.; Nar, H.; Litzenburger, T. A specific antidote for dabigatran: Functional and structural characterization. Blood 2013, 121, 3554–3562. [Google Scholar] [CrossRef] [PubMed]
- Glund, S.; Stangier, J.; Schmohl, M.; Moschetti, V.; Haazen, W.; De Smet, M.; Gansser, D.; Norris, S.; Lang, B.; Reilly, P. Idarucizumab, a specific antidote for dabigatran: Immediate, complete and sustained reversal of dabigatran induced anticoagulation in elderly and renally impaired subjects. Am. Soc. Hematol. 2014, 124, 344. [Google Scholar] [CrossRef]
- Glund, S.; Stangier, J.; Schmohl, M.; Gansser, D.; Norris, S.; van Ryn, J.; Lang, B.; Ramael, S.; Moschetti, V.; Gruenenfelder, F.; et al. Safety, tolerability, and efficacy of idarucizumab for the reversal of the anticoagulant effect of dabigatran in healthy male volunteers: A randomised, placebo-controlled, double-blind phase 1 trial. Lancet 2015, 386, 680–690. [Google Scholar] [CrossRef]
- Pollack, C.V., Jr.; Reilly, P.A.; Bernstein, R.; Dubiel, R.; Eikelboom, J.; Glund, S.; Huisman, M.V.; Hylek, E.; Kam, C.W.; Kamphuisen, P.W.; et al. Design and rationale for RE-VERSE AD: A phase 3 study of idarucizumab, a specific reversal agent for dabigatran. Thromb. Haemost. 2015, 114, 198–205. [Google Scholar]
- Pollack, C.V., Jr.; Reilly, P.A.; Eikelboom, J.; Glund, S.; Verhamme, P.; Bernstein, R.A.; Dubiel, R.; Huisman, M.V.; Hylek, E.M.; Kamphuisen, P.W.; et al. Idarucizumab for dabigatran reversal. N. Engl. J. Med. 2015, 373, 511–520. [Google Scholar] [CrossRef]
- Pollack, C.V., Jr.; Reilly, P.A.; Van Ryn, J.; Eikelboom, J.W.; Glund, S.; Bernstein, R.A.; Dubiel, R.; Huisman, M.V.; Hylek, E.M.; Kam, C.W.; et al. Idarucizumab for dabigatran reversal—Full cohort analysis. N. Engl. J. Med. 2017, 377, 431–441. [Google Scholar] [CrossRef]
- Burness, C.B. Idarucizumab: First global approval. Drugs 2015, 75, 2155–2161. [Google Scholar] [CrossRef]
- Tummala, R.; Kavtaradze, A.; Gupta, A.; Ghosh, R.K. Specific antidotes against direct oral anticoagulants: A comprehensive review of clinical trials data. Int. J. Cardiol. 2016, 214, 292–298. [Google Scholar] [CrossRef]
- Eikelboom, J.W.; Quinlan, D.J.; van Ryn, J.; Weitz, J.I. Idarucizumab: The antidote for reversal of dabigatran. Circulation 2015, 132, 2412–2422. [Google Scholar] [CrossRef]
- Hu, T.Y.; Vaidya, V.R.; Asirvatham, S.J. Reversing anticoagulant effects of novel oral anticoagulants: Role of ciraparantag, andexanet alfa, and idarucizumab. Vasc. Health Risk Manag. 2016, 2, 35. [Google Scholar]
- Simon, A.; Domanovits, H.; Ay, C.; Sengoelge, G.; Levy, J.H.; Spiel, A.O. The recommended dose of idarucizumab may not always be sufficient for sustained reversal of dabigatran. J. Thromb. Haemost. 2017, 15, 1317–1321. [Google Scholar] [CrossRef] [PubMed]
- Buchheit, J.; Reddy, P.; Connors, J.M. Idarucizumab (Praxbind) formulary review. Crit. Pathw. Cardiol. 2016, 15, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Yasaka, M.; Ikushima, I.; Harada, A.; Imazu, S.; Taniguchi, A.; Norris, S.; Gansser, D.; Stangier, J.; Schmohl, M.; Reilly, P.A. Safety, pharmacokinetics and pharmacodynamics of idarucizumab, a specific dabigatran reversal agent in healthy Japanese volunteers: A randomized study. Res. Pract. Thromb. Haemost. 2017, 1, 202–215. [Google Scholar] [CrossRef] [PubMed]
- Steele, A.P.; Lee, J.A.; Dager, W.E. Incomplete dabigatran reversal with idarucizumab. Clin. Toxicol. 2018, 56, 216–218. [Google Scholar] [CrossRef] [PubMed]
- Stecher, A.; Vene, N.; Mavri, A.; Mijovski, M.B.; Krevel, B.; Gradišek, P. Late rebound of dabigatran levels after idarucizumab reversal in two patients with severe renal failure. Eur. J. Anaesthesiol. 2017, 34, 400–402. [Google Scholar] [CrossRef]
- Alhashem, H.M.; Avendano, C.; Hayes, B.D.; Winters, M.E. Persistent life-threatening hemorrhage after administration of idarucizumab. Am. J. Emerg. Med. 2017, 35, 193. [Google Scholar] [CrossRef]
- Misselwitz, F.; Berkowitz, S.D.; Perzborn, E. The discovery and development of rivaroxaban. Ann. N. Y. Acad. Sci. 2011, 1222, 64–75. [Google Scholar] [CrossRef]
- Apostolakis, S.; Lip, G.Y.; Lane, D.A.; Shantsila, E. The quest for new anticoagulants: From clinical development to clinical practice. Cardiovasc. Ther. 2011, 29, e12–e22. [Google Scholar] [CrossRef]
- Perzborn, E.; Strassburger, J.; Wilmen, A.; Pohlmann, J.; Roehrig, S.; Schlemmer, K.H.; Straub, A. In vitro and in vivo studies of the novel antithrombotic agent BAY 59-7939—An oral, direct Factor Xa inhibitor. J. Thromb. Haemost. 2005, 3, 514–521. [Google Scholar] [CrossRef]
- Kubitza, D.; Becka, M.; Wensing, G.; Voith, B.; Zuehlsdorf, M. Safety, pharmacodynamics, and pharmacokinetics of BAY 59-7939—An oral, direct Factor Xa inhibitor—After multiple dosing in healthy male subjects. Eur. J. Clin. Pharmacol. 2005, 61, 873–880. [Google Scholar] [CrossRef]
- Ageno, W. Rivaroxaban for the prevention of venous thromboembolism following major orthopedic surgery: The RECORD trials. Expert Rev. Cardiovasc. Ther. 2009, 7, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Turpie, A.G.; Lassen, M.R.; Davidson, B.L.; Bauer, K.A.; Gent, M.; Kwong, L.M.; Cushner, F.D.; Lotke, P.A.; Berkowitz, S.D.; Bandel, T.J.; et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after total knee arthroplasty (RECORD4): A randomised trial. Lancet 2009, 373, 1673–1680. [Google Scholar] [CrossRef]
- Romualdi, E.; Donadini, M.P.; Ageno, W. Oral rivaroxaban after symptomatic venous thromboembolism: The continued treatment study (EINSTEIN-extension study). Expert Rev. Cardiovasc. Ther. 2011, 9, 841–844. [Google Scholar] [CrossRef] [PubMed]
- Prins, M.H.; Lensing, A.W.; Brighton, T.A.; Lyons, R.M.; Rehm, J.; Trajanovic, M.; Davidson, B.L.; Beyer-Westendorf, J.; Pap, Á.F.; Berkowitz, S.D.; et al. Oral rivaroxaban versus enoxaparin with vitamin K antagonist for the treatment of symptomatic venous thromboembolism in patients with cancer (EINSTEIN-DVT and EINSTEIN-PE): A pooled subgroup analysis of two randomised controlled trials. Lancet Haematol. 2014, 1, e37–e46. [Google Scholar] [CrossRef]
- Pink, J.; Pirmohamed, M.; Hughes, D.A. Comparative effectiveness of dabigatran, rivaroxaban, apixaban, and warfarin in the management of patients with nonvalvular atrial fibrillation. Clin. Pharmacol. Ther. 2013, 94, 269–276. [Google Scholar] [CrossRef]
- Rybak, I.; Ehle, M.; Buckley, L.; Fanikos, J. Efficacy and safety of novel anticoagulants compared with established agents. Ther. Adv. Hematol. 2011, 2, 175–195. [Google Scholar] [CrossRef] [PubMed]
- Trujillo, T.; Dobesh, P.P. Clinical use of rivaroxaban: Pharmacokinetic and pharmacodynamic rationale for dosing regimens in different indications. Drugs 2014, 74, 1587–1603. [Google Scholar] [CrossRef]
- Samama, M.M. The mechanism of action of rivaroxaban–an oral, direct Factor Xa inhibitor–compared with other anticoagulants. Thromb. Res. 2011, 127, 497–504. [Google Scholar] [CrossRef]
- Kubitza, D.; Becka, M.; Voith, B.; Zuehlsdorf, M.; Wensing, G. Safety, pharmacodynamics, and pharmacokinetics of single doses of BAY 59-7939, an oral, direct factor Xa inhibitor. Clin. Pharmacol. Ther. 2005, 78, 412–421. [Google Scholar] [CrossRef]
- Mueck, W.; Stampfuss, J.; Kubitza, D.; Becka, M. Clinical pharmacokinetic and pharmacodynamic profile of rivaroxaban. Clin. Pharm. 2014, 53, 1–6. [Google Scholar] [CrossRef]
- Kubitza, D.; Becka, M.; Mueck, W.; Halabi, A.; Maatouk, H.; Klause, N.; Lufft, V.; Wand, D.D.; Philipp, T.; Bruck, H. Effects of renal impairment on the pharmacokinetics, pharmacodynamics and safety of rivaroxaban, an oral, direct Factor Xa inhibitor. Br. J. Clin. Pharmacol. 2010, 70, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Weinz, C.; Schwarz, T.; Kubitza, D.; Mueck, W.; Lang, D. Metabolism and excretion of rivaroxaban, an oral, direct factor Xa inhibitor, in rats, dogs, and humans. Drug Metab. Dispos. 2009, 37, 1056–1064. [Google Scholar] [CrossRef] [PubMed]
- Wasserlauf, G.; Grandi, S.M.; Filion, K.; Eisenberg, M.J. Meta-analysis of rivaroxaban and bleeding risk. Am. J. Cardiol. 2013, 112, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Wong, P.C.; Pinto, D.J.; Zhang, D. Preclinical discovery of apixaban, a direct and orally bioavailable factor Xa inhibitor. J. Throm. Thrombolysis 2011, 31, 478–492. [Google Scholar] [CrossRef] [PubMed]
- Lassen, M.R.; Raskob, G.E.; Gallus, A.; Pineo, G.; Chen, D.; Hornick, P. Apixaban versus enoxaparin for thromboprophylaxis after knee replacement (ADVANCE-2): A randomised double-blind trial. Lancet 2010, 375, 807–815. [Google Scholar] [CrossRef]
- Lassen, M.R.; Gallus, A.S.; Pineo, G.F.; Raskob, G.E. Randomized double-blind comparison of apixaban with enoxaparin for thromboprophylaxis after knee replacement: The ADVANCE-1 trial. Blood 2008, 112, 31. [Google Scholar]
- Easton, J.D.; Lopes, R.D.; Bahit, M.C.; Wojdyla, D.M.; Granger, C.B.; Wallentin, L.; Alings, M.; Goto, S.; Lewis, B.S.; Rosenqvist, M.; et al. Apixaban compared with warfarin in patients with atrial fibrillation and previous stroke or transient ischaemic attack: A subgroup analysis of the ARISTOTLE trial. Lancet Neurol. 2012, 11, 503–511. [Google Scholar] [CrossRef]
- Diener, H.C.; Eikelboom, J.; Connolly, S.J.; Joyner, C.D.; Hart, R.G.; Lip, G.Y.; O’Donnell, M.; Hohnloser, S.H.; Hankey, G.J.; Shestakovska, O.; et al. Apixaban versus aspirin in patients with atrial fibrillation and previous stroke or transient ischaemic attack: A predefined subgroup analysis from AVERROES, a randomised trial. Lancet Neurol. 2012, 11, 225–231. [Google Scholar] [CrossRef]
- Frost, C.; Nepal, S.; Wang, J.; Schuster, A.; Byon, W.; Boyd, R.A.; Yu, Z.; Shenker, A.; Barrett, Y.C.; Mosqueda-Garcia, R.; et al. Safety, pharmacokinetics and pharmacodynamics of multiple oral doses of apixaban, a factor X a inhibitor, in healthy subjects. Br. J. Clin. Pharmacol. 2013, 76, 776–786. [Google Scholar] [CrossRef]
- Raghavan, N.; Frost, C.E.; Yu, Z.; He, K.; Zhang, H.; Humphreys, W.G.; Pinto, D.; Chen, S.; Bonacorsi, S.; Wong, P.C.; et al. Apixaban metabolism and pharmacokinetics after oral administration to humans. Drug Metab. Dispos. 2009, 37, 74–81. [Google Scholar] [CrossRef]
- Hanna, M.S.; Mohan, P.; Knabb, R.; Gupta, E.; Frost, C.; Lawrence, J.H. Development of apixaban: A novel anticoagulant for prevention of stroke in patients with atrial fibrillation. Ann. N. Y. Acad. Sci. 2014, 1329, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Hylek, E.M.; Held, C.; Alexander, J.H.; Lopes, R.D.; De Caterina, R.; Wojdyla, D.M.; Huber, K.; Jansky, P.; Steg, P.G.; Hanna, M.; et al. Major bleeding in patients with atrial fibrillation receiving apixaban or warfarin: The ARISTOTLE Trial (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation): Predictors, Characteristics, and Clinical Outcomes. J. Am. Coll. Cardiol. 2014, 63, 2141–2147. [Google Scholar] [CrossRef] [PubMed]
- Schulman, S. Advantages and limitations of the new anticoagulants. J. Intern. Med. 2014, 275, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Samama, M.M.; Mendell, J.; Guinet, C.; Le Flem, L.; Kunitada, S. In vitro study of the anticoagulant effects of edoxaban and its effect on thrombin generation in comparison to fondaparinux. Thromb. Res. 2012, 129, e77–e82. [Google Scholar] [CrossRef] [PubMed]
- Lip, G.Y.; Agnelli, G. Edoxaban: A focused review of its clinical pharmacology. Eur. Heart J. 2004, 35, 1844–1855. [Google Scholar] [CrossRef] [PubMed]
- Ogata, K.; Mendell-Harary, J.; Tachibana, M.; Masumoto, H.; Oguma, T.; Kojima, M.; Kunitada, S. Clinical safety, tolerability, pharmacokinetics, and pharmacodynamics of the novel factor Xa inhibitor edoxaban in healthy volunteers. J. Clin. Pharmacol. 2010, 50, 743–753. [Google Scholar] [CrossRef]
- Parasrampuria, D.A.; Truitt, K.E. Pharmacokinetics and pharmacodynamics of edoxaban, a non-vitamin K antagonist oral anticoagulant that inhibits clotting factor Xa. Clin. Pharm. 2016, 55, 641–655. [Google Scholar] [CrossRef]
- Mikkaichi, T.; Yoshigae, Y.; Masumoto, H.; Imaoka, T.; Rozehnal, V.; Fischer, T.; Okudaira, N.; Izumi, T. Edoxaban transport via P-glycoprotein is a key factor for the drug’s disposition. Drug Metab. Dispos. 2014, 42, 520–528. [Google Scholar] [CrossRef]
- Mendell, J.; Tachibana, M.; Shi, M.; Kunitada, S. Effects of food on the pharmacokinetics of edoxaban, an oral direct factor Xa inhibitor, in healthy volunteers. J. Clin. Pharmacol. 2011, 51, 687–694. [Google Scholar] [CrossRef]
- Fuji, T.; Fujita, S.; Kawai, Y.; Nakamura, M.; Kimura, T.; Fukuzawa, M.; Abe, K.; Tachibana, S. Efficacy and safety of edoxaban versus enoxaparin for the prevention of venous thromboembolism following total hip arthroplasty: STARS J-V. Thromb. J. 2015, 13, 27. [Google Scholar] [CrossRef]
- Giugliano, R.P.; Ruff, C.T.; Braunwald, E.; Murphy, S.A.; Wiviott, S.D.; Halperin, J.L.; Waldo, A.L.; Ezekowitz, M.D.; Weitz, J.I.; Špinar, J.; et al. Edoxaban versus warfarin in patients with atrial fibrillation. N. Engl. J. Med. 2013, 369, 2093–2104. [Google Scholar] [CrossRef] [PubMed]
- Hokusai-VTE Investigators. Edoxaban versus warfarin for the treatment of symptomatic venous thromboembolism. N. Engl. J. Med. 2013, 369, 1406–1415. [Google Scholar] [CrossRef] [PubMed]
- Chung, N.; Jeon, H.K.; Lien, L.M.; Lai, W.T.; Tse, H.F.; Chung, W.S.; Lee, T.H.; Chen, S.A. Safety of edoxaban, an oral factor Xa inhibitor, in Asian patients with non-valvular atrial fibrillation. Thromb. Haemost. 2011, 105, 535–545. [Google Scholar] [PubMed]
- Palladino, M.; Merli, G.; Thomson, L. Evaluation of the oral direct factor Xa inhibitor–betrixaban. Expert Opin. Investig. Drugs 2013, 22, 1465–1472. [Google Scholar] [CrossRef]
- Chan, N.C.; Hirsh, J.; Ginsberg, J.S.; Eikelboom, J.W. Betrixaban (PRT054021): Pharmacology, dose selection and clinical studies. Future Cardiol. 2014, 10, 43–52. [Google Scholar] [CrossRef]
- Chan, N.C.; Bhagirath, V.; Eikelboom, J.W. Profile of betrixaban and its potential in the prevention and treatment of venous thromboembolism. Vasc. Health Risk Manag. 2015, 11, 343. [Google Scholar]
- Turpie, A.G.; Bauer, K.A.; Davidson, B.L.; Fisher, W.D.; Gent, M.; Huo, M.H.; Sinha, U.; Gretler, D.D. EXPERT Study Group. A randomized evaluation of betrixaban, an oral factor Xa inhibitor, for prevention of thromboembolic events after total knee replacement (EXPERT). Thromb. Haemost. 2009, 101, 68–76. [Google Scholar]
- Connolly, S.J.; Eikelboom, J.; Dorian, P.; Hohnloser, S.H.; Gretler, D.D.; Sinha, U.; Ezekowitz, M.D. Betrixaban compared with warfarin in patients with atrial fibrillation: Results of a phase 2, randomized, dose-ranging study (Explore-Xa). Eur. Heart J. 2013, 34, 1498–1505. [Google Scholar] [CrossRef]
- Cohen, A.T.; Harrington, R.A.; Goldhaber, S.Z.; Hull, R.D.; Wiens, B.L.; Gold, A.; Hernandez, A.F.; Gibson, C.M. Extended thromboprophylaxis with betrixaban in acutely ill medical patients. N. Engl. J. Med. 2016, 375, 534–544. [Google Scholar] [CrossRef]
- Gibson, C.M.; Halaby, R.; Korjian, S.; Daaboul, Y.; Arbetter, D.F.; Yee, M.K.; Goldhaber, S.Z.; Hull, R.; Hernandez, A.F.; Lu, S.P.; et al. The safety and efficacy of full-versus reduced-dose betrixaban in the Acute Medically Ill VTE (Venous Thromboembolism) Prevention With Extended-Duration Betrixaban (APEX) trial. Am. Heart J. 2017, 185, 93–100. [Google Scholar] [CrossRef]
- U.S. National Library of Medicine. Available online: https://clinicaltrials.gov/ct2/show/NCT03346083?term=betrixaban&rank=4 (accessed on 20 August 2019).
- U.S. National Library of Medicine. Available online: https://clinicaltrials.gov/ct2/show/NCT03613402 (accessed on 20 August 2019).
- Steinberg, B.A.; Becker, R.C. Structure–function relationships of factor Xa inhibitors: Implications for the practicing clinician. J. Thromb. Thrombolysis 2014, 37, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Kaatz, S.; Bhansali, H.; Gibbs, J.; Lavender, R.; Mahan, C.E.; Paje, D.G. Reversing factor Xa inhibitors–clinical utility of andexanet alfa. J. Blood Med. 2017, 8, 141. [Google Scholar] [CrossRef] [PubMed]
- Becker, R.C. The biochemistry, enzymology and pharmacology of non-vitamin K anticoagulant drug reversal agents and antidotes. J. Thromb. Thrombolysis 2016, 41, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; DeGuzman, F.R.; Hollenbach, S.J.; Karbarz, M.J.; Abe, K.; Lee, G.; Luan, P.; Hutchaleelaha, A.; Inagaki, M.; Conley, P.B.; et al. A specific antidote for reversal of anticoagulation by direct and indirect inhibitors of coagulation factor Xa. Nat. Med. 2013, 19, 446. [Google Scholar] [CrossRef]
- Sartori, M.; Cosmi, B. Andexanet alfa to reverse the anticoagulant activity of factor Xa inhibitors: A review of design, development and potential place in therapy. J. Thromb. Thrombolysis 2018, 45, 345–352. [Google Scholar] [CrossRef]
- Lu, G.; Lin, J.P.; Curnutte, J.T.; Conley, P.B. Effect of andexanet-TFPI interaction on in vitro thrombin formation and coagulation markers in the TF-pathway. Blood 2017, 130, 629. [Google Scholar]
- Ghadimi, K.; Dombrowski, K.E.; Levy, J.H.; Welsby, I.J. Andexanet alfa for the reversal of Factor Xa inhibitor related anticoagulation. Expert Rev. Hematol. 2016, 9, 115–122. [Google Scholar] [CrossRef]
- Siegal, D.M.; Curnutte, J.T.; Connolly, S.J.; Lu, G.; Conley, P.B.; Wiens, B.L.; Mathur, V.S.; Castillo, J.; Bronson, M.D.; Leeds, J.M.; et al. Andexanet alfa for the reversal of factor Xa inhibitor activity. N. Engl. J. Med. 2015, 373, 2413–2424. [Google Scholar] [CrossRef]
- Yeh, C.H.; Fredenburgh, J.C.; Weitz, J.I. The real decoy: An antidote for factor Xa–directed anticoagulants. Circ. Res. 2013, 113, 954–957. [Google Scholar] [CrossRef]
- Milling, T.J., Jr.; Ziebell, C.M. A review of reversal of oral anticoagulants, old and new, in major bleeding and the need for urgent surgery. Trends Cardiovasc. Med. 2019, in press. [Google Scholar] [CrossRef]
- Crowther, M.; Kitt, M.; McClure, M.; Sinha, U.; Lu, G.; Karbarz, M.; Hutchaleelaha, A.; Barron, L.; Mathur, V.; Curnutte, J. Randomized, double-blind, placebo-controlled single ascending dose pharmacokinetic and pharmacodynamic study of PRT064445, a universal antidote for factor Xa inhibitors. Arter. Thromb. Vasc. Biol. 2013, 33, A10. [Google Scholar]
- U.S. National Library of Medicine. Available online: https://clinicaltrials.gov/ct2/show/NCT01758432?term=andexanet&rank=6 (accessed on 20 August 2019).
- Siegal, D.; Lu, G.; Leeds, J.M.; Karbarz, M.; Castillo, J.; Mathur, V.; Hutchaleelaha, A.; Sinha, U.; Kitt, M.; McClure, M.; et al. Safety, pharmacokinetics, and reversal of apixaban anticoagulation with andexanet alfa. Blood Adv. 2017, 1, 1827–1838. [Google Scholar] [CrossRef] [PubMed]
- Crowther, M.; Lu, G.; Conley, P.; Leeds, J.; Castillo, J.; Levy, G.; Connolly, S.; Curnutte, J. Reversal of factor XA inhibitors-induced anticoagulation in healthy subjects by andexanet alfa. Crit. Care Med. 2014, 42, A1469. [Google Scholar] [CrossRef]
- Crowther, M.; Levy, G.; Lu, G.; Leeds, J.; Barron, L.; Conley, P. ANNEXA-A: A phase 3 randomized, double-blind, placebo-controlled trial, demonstrating reversal of apixaban-induced anticoagulation in older subjects by andexanet alfa (PRT064445), a universal antidote for factor Xa (fXa) inhibitors. Circulation 2014, 130, 2105–2126. [Google Scholar]
- Connolly, S.J.; Crowther, M.; Eikelboom, J.W.; Gibson, C.M.; Curnutte, J.T.; Lawrence, J.H.; Yue, P.; Bronson, M.D.; Lu, G.; Conley, P.B.; et al. Full Study Report of Andexanet Alfa for Bleeding Associated with Factor Xa Inhibitors. N. Engl. J. Med. 2019, 380, 1326–1335. [Google Scholar] [CrossRef]
- Heo, Y.A. Andexanet alfa: First global approval. Drugs 2018, 78, 1049–1055. [Google Scholar] [CrossRef]
- Global News. Available online: http://www.globenewswire.com/news-release/2019/04/26/1810785/0/en/European-Commission-Grants-Conditional-Marketing-Authorization-for-Portola-Pharmaceuticals-Ondexxya-andexanet-alfa-the-First-and-Only-Antidote-for-the-Reversal-of-Factor-Xa-Inhibit.html (accessed on 20 August 2019).
- Cuker, A.; Burnett, A.; Triller, D.; Crowther, M.; Ansell, J.; Van Cott, E.M.; Wirth, D.; Kaatz, S. Reversal of direct oral anticoagulants: Guidance from the Anticoagulation Forum. Am. J. Hematol. 2019, 94, 697–709. [Google Scholar] [CrossRef]
- Specialy Pharmacy Times. Available online: https://www.specialtypharmacytimes.com/news/fda-oks-large-scale-commercial-production-of-andexxa (accessed on 20 August 2019).
- Grottke, O.; Akman, N.; Braunschweig, T.; Conley, P.B.; Rossaint, R. Comparison of second and first generation of andexanet alfa in a porcine polytrauma model with apixaban anticoagulation. Blood 2018, 132, 3778. [Google Scholar] [CrossRef]
- Ellington, T.M. A systematic and evidence-based review of published and pending reports of andexanet alfa. J. Pharm. Pract. 2019, in press. [Google Scholar] [CrossRef]
- U.S. National Library of Medicine. Available online: https://clinicaltrials.gov/ct2/show/NCT03310021 (accessed on 20 August 2019).
- U.S. National Library of Medicine. Available online: https://clinicaltrials.gov/ct2/show/NCT03661528 (accessed on 20 August 2019).
- U.S. National Library of Medicine. Available online: https://clinicaltrials.gov/ct2/show/NCT02329327 (accessed on 20 August 2019).
- Costin, J.; Ansell, J.; Laulicht, B.; Bakhru, S.; Steiner, S. Reversal agents in development for the new oral anticoagulants. Postgrad. Med. 2014, 126, 19–24. [Google Scholar] [CrossRef]
- Milling, T.J., Jr.; Kaatz, S. Preclinical and clinical data for factor Xa and “universal” reversal agents. Am. J. Med. 2016, 129, S80–S88. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ansell, J.E.; Bakhru, S.H.; Laulicht, B.E.; Steiner, S.S.; Grosso, M.A.; Brown, K.; Dishy, V.; Lanz, H.J.; Mercuri, M.F.; Noveck, R.J.; et al. Single-dose ciraparantag safely and completely reverses anticoagulant effects of edoxaban. Thromb. Haemost. 2017, 117, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Costin, J.C.; Laulicht, B.; Bakhru, S.; Steiner, S. PER977 reverses low molecular weight heparin in addition to IIa and Xa new oral anticoagulants. J. Am. Coll. Cardiol. 2015, 65, A2056. [Google Scholar] [CrossRef]
- Sullivan, D.W., Jr.; Gad, S.C.; Laulicht, B.; Bakhru, S.; Steiner, S. Nonclinical safety assessment of PER977: A small molecule reversal agent for new oral anticoagulants and heparins. Int. J. Toxicol. 2015, 34, 308–317. [Google Scholar] [CrossRef]
- Bakhru, S.; Laulicht, B.; Jiang, X.; Chen, L.; Pan, D.; Grosso, M.; Morishima, Y.; Brown, K.; Masumoto, H.; Costin, J.; et al. PER977: A synthetic small molecule which reverses over-dosage and bleeding by the new oral anticoagulants. Circulation 2013, 128, A18809. [Google Scholar]
- Bakhru, S.; Laulicht, B.; Jiang, X.; Chen, L.; Grosso, M.; Morishima, Y.; Brown, K.; Mercuri, M.; Masumoto, H.; Costin, J.; et al. Reversal of anticoagulant-induced bleeding in external and internal bleeding models by PER977, a small molecule anticoagulant antidote. Circulation 2014, 130, A19361. [Google Scholar]
- Laulicht, B.; Bakhru, S.; Bakhru, S.; Lee, C.; Baker, C.; Jiang, X.; Mathiowitz, E.; Costin, J. Small molecule antidote for anticoagulants. Circulation 2018, 126, A11395. [Google Scholar]
| General Class/MOA | Drug Name and Year of First Approval | Labeled Indications | Adult Dosing (with Normal Renal & Hepatic Function) | Route of Administration | Approved Reversal Agent |
|---|---|---|---|---|---|
| Vitamin K Antagonist | Warfarin (1954) |
| INR-adjusted-based dosing Goal INR is 2–3 for most patients Goal INR for mitral valve replacement is 2.5–3.5 | Oral | Vitamin K and/or Prothrombin Complex Concentrate |
| Indirect Thrombin Inhibitors | Heparin (1940s) * |
| VTE Treatment: 80 unit/kg IV bolus, then 18 unit/kg/h IV infusion VTE Prophylaxis: 5000 units q8h Target anti-Xa level 6 h post-dose: 0.3–0.7 units/mL | Injectable Intravenous or Subcutaneous | Protamine → 100% reversal |
Low Molecular Weight Heparins (LMWH):
|
| DVT Treatment: 1 mg/kg q12h OR 1.5 mg/kg q24h VTE Prophylaxis: 40 mg q24h Target anti-Xa level 4 h post-dose: 0.5–1.1 units/mL | Injectable Subcutaneous | Protamine → 60% reversal | |
| Direct thrombin Inhibitors | Argatroban (2000) |
| Prophylaxis/treatment of thrombosis in HIT: 2 mcg/kg/min and adjust based on aPTT (goal 1.5–3 times baseline) PCI: 350 mcg/kg bolus, then 25 mcg/kg/min infusion and adjust based on ACT | Injectable Intravenous | N/A |
| Bivalirudin (2000) |
| Before PCI: 0.1 mg/kg bolus, then 0.25 mg/kg/h until PCI During PCI: 0.75 mg/kg bolus, then 1.75 mg/kg/h for the duration of procedure | Injectable Intravenous | N/A | |
| Dabigatran (2010) |
| VTE Treatment (after initial therapy with a parenteral anticoagulant for 5 days): 150 mg BID Afib: 150 mg BID THA: 110 mg given 1–4 h after surgery, then 220 mg QD for 10–14 d | Oral | Idarucizumab (Praxbind) | |
| Desirudin (2003) |
| 15 mg q12h | Injectable Subcutaneous | N/A | |
| Lepirudin ** (1998) |
| 0.4 mg/kg IV bolus, followed by 0.15 mg/kg/h IV infusion for 2–10 days (or as clinically indicated) | Injectable Subcutaneous | N/A | |
| Indirect Factor Xa Inhibitor | Pentasaccharide-Fondaparinux (2001) |
| VTE Treatment: <50 kg → 5 mg QD 50–100 kg → 7.5 mg QD >100 mg → 10 mg QD VTE Prophylaxis: 2.5 mg QD | Injectable Subcutaneous | N/A |
| Factor Xa Inhibitors | Apixaban (2012) |
| VTE Treatment: 10 mg BID for 7 days, then 5 mg BID Afib: 5 mg BID; if patient has any 2 of the following then 2.5 mg BID → age ≥80, weight ≤60 kg, or SCr ≥1.5 mg/dL Knee/hip arthroplasty: 2.5 mg BID starting 12–24 h after surgery Secondary prevention: 2.5 mg BID (following 6 months of initial therapy) | Oral Tablet | Andexanet Alfa |
| Betrixaban (2017) |
| VTE prophylaxis: 160 mg as a single dose on day 1, followed by 80 mg once daily for 35 to 42 days | Oral Capsule | N/A | |
| Edoxaban (2015) |
| VTE: If >60 kg → 60 mg QD If ≤60 kg → 30 mg QD Afib: 60 mg QD | Oral Tablet | N/A | |
| Rivaroxaban (2011) |
| VTE treatment: 15 mg BID w/ food for 21 days, followed by 20 mg QD w/ food Knee/hip arthroplasty: 10 mg QD starting 6–10 h after surgery Afib: 20 mg QD w/ evening meal CAD/PAD: 2.5 mg BID (with or without aspirin) Secondary prevention: 10 mg QD (following 6 months of initial treatment) Indefinite anticoagulation: reduced intensity dosing | Oral Tablet | Andexanet Alfa |
| Administration | Low Dose | High Dose |
|---|---|---|
| Bolus | 400 mg at 30 mg/min | 800 mg at 30 mg/min |
| Infusion | 4 mg/min for 120 mins | 8 mg/min for 120 mins |
| Number of vials | Bolus = 400 mg → 4 vials Infusion = 480 mg → 5 vials Total = 9 vials of 100 mg | Bolus = 800 mg → 8 vials Infusion = 960 mg → 10 vials Total = 18 vials of 100 mg OR 9 vials of 200 mg |
| Clinical Trials Identifier | Title | Primary and Secondary Outcomes | Status | Results Available? |
|---|---|---|---|---|
| NCT01826266 | Phase I Evaluation of the Safety, Tolerability, PK, and PD Effects of a Double-Blind, Single Dose of PER977 Administered Alone, and Following a Single Dose of Edoxaban | 1º—safety, tolerability, plasma PK, and urinary PK of a range of single IV doses of PER977 | Completed December 2013 | No |
| NCT02206100 | Phase I/II Evaluation of the Safety, Tolerability, PK, and PD Effects of Single, Hourly-Repeating Escalating Doses of PER977 Following a Single Subcutaneous Dose of Enoxaparin | 1º—number of adverse events 2º—reversal of enoxaparin anticoagulation, PK of enoxaparin, PK of PER977 and its metabolite | Completed April 2014 | No |
| NCT02205905 | An Open-label, Single-dose, Non-randomized Study of the Absorption, Metabolism, and Excretion of Intravenously Administered 14C-labeled PER977 in Healthy Male Subjects (Phase I Study) | 1º—characterization of the mass balance, metabolic disposition, and identification of the metabolites and general metabolic pathways of PER977 2º—number of subjects with adverse events | Completed August 2014 | No |
| NCT02207257 | Phase II Randomized, Sequential Group, Evaluation of Ascending Reversal Doses of PER977 Administered to Subjects with Steady State Edoxaban Dosing and Re-anticoagulation With Edoxaban Following PER977 Reversal | 1º—WBCT as a measure of edoxaban anticoagulation reversal by PER977 2º—PK of PER977, PK of edoxaban administered with PER977, safety coagulation measures, and safety and tolerability of PER977 after edoxaban | Completed November 2015 | No |
| NCT02206087 | Phase I/II Evaluation of the Safety, Tolerability, PK, and PD Effects of a Single Escalating Dose of PER977 Following Administration of Unfractionated Heparin | 1º—effect of PER977 on reversal of UFH anticoagulation 2º—safety and tolerability of PER977 administered after UFH | Completed March 2016 | No |
| NCT03288454 | Phase 2 Placebo-Controlled, Single-Site, Single-Blind Study of Apixaban Reversal by Ciraparantag as Measured by WBCT | 1º—efficacy of ciraparantag in reversal of apixaban-induced anticoagulation, measured by WBCT 2º—number of participants with abnormal lab values or adverse events | Recruiting; expected to be complete in January 2019 | No |
| NCT03172910 | Phase 2 Placebo-Controlled, Single-Site, Single-Blind Study of Rivaroxaban Reversal by Ciraparantag as Measured by WBCT | 1º—efficacy of ciraparantag in reversal of rivaroxaban-induced anticoagulation, measured by WBCT 2º—incidence of adverse effects | Recruiting; expected to be complete in January 2019 | No |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kustos, S.A.; Fasinu, P.S. Direct-Acting Oral Anticoagulants and Their Reversal Agents—An Update. Medicines 2019, 6, 103. https://doi.org/10.3390/medicines6040103
Kustos SA, Fasinu PS. Direct-Acting Oral Anticoagulants and Their Reversal Agents—An Update. Medicines. 2019; 6(4):103. https://doi.org/10.3390/medicines6040103
Chicago/Turabian StyleKustos, Stephanie A., and Pius S. Fasinu. 2019. "Direct-Acting Oral Anticoagulants and Their Reversal Agents—An Update" Medicines 6, no. 4: 103. https://doi.org/10.3390/medicines6040103
APA StyleKustos, S. A., & Fasinu, P. S. (2019). Direct-Acting Oral Anticoagulants and Their Reversal Agents—An Update. Medicines, 6(4), 103. https://doi.org/10.3390/medicines6040103






