Salvia mellifera—How Does It Alleviate Chronic Pain?
Abstract
1. Introduction
2. The Medicine
3. Case Reports: Use of Black Sage Sun Tea in Pain Patients
4. Discussion
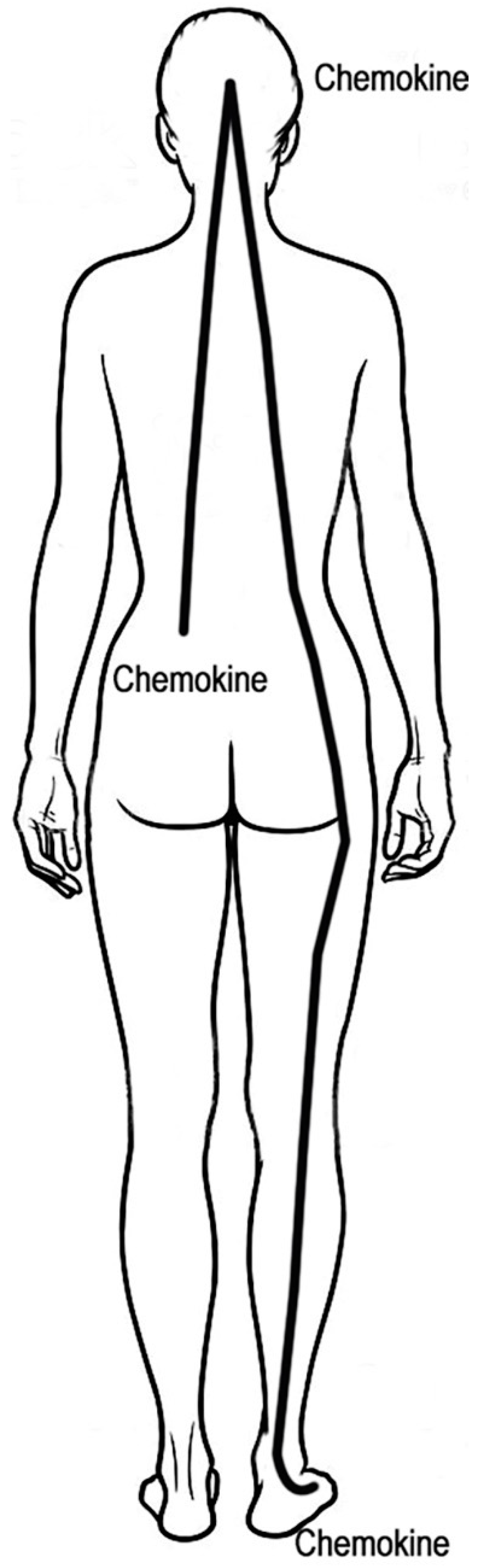
4.1. How Does Black Sage Sun Tea Work?
4.2. The Placebo Effect
4.3. The Exercise Effect
4.4. Does Black Sage Sun Tea Actually Cure Chronic Pain?
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Garcia, C.; Adams, J. Healing with Medicinal Plants of the West–Cultural and Scientific Basis for Their Use, 3rd ed.; Abedus Press: La Crescenta, CA, USA, 2016; ISBN 978-0-9763091-9-2. [Google Scholar]
- Adams, J.; Garcia, C. Palliative care among Chumash people. Evid. Based Complement. Altern. Med. 2005, 2, 143–147. [Google Scholar] [CrossRef]
- De Martino, L.; Roscigno, G.; Mancini, E.; De Falco, E.; De Feo, V. Chemical composition and antigerminative activity of the essential oils of five Salvia species. Molecules 2010, 15, 735–746. [Google Scholar] [CrossRef]
- Gonzalez, A.; Andres, L.; Aguiar, Z.; Luis, J. Diterpenes from Salvia mellifera and their biogenetic significance. Phytochemistry 1992, 31, 1297–1305. [Google Scholar] [CrossRef]
- Nahin, R. Estimates of pain prevalence and severity in adults: United States, 2012. J. Pain 2015, 16, 769–780. [Google Scholar] [CrossRef]
- Pak, J.; Yong, R.; Kaye, A.; Uman, R. Chronification of pain: Mechanisms, current understanding, and clinical implications. Curr. Pain Headache Rep. 2018, 22, 9. [Google Scholar] [CrossRef]
- Voscopoulos, C.; Lema, M. When does acute pain become chronic? Br. J. Anaesth. 2010, 105, i69–i85. [Google Scholar] [CrossRef]
- Adams, J. Chronic pain–can it be cured? J. Pharm. Drug Dev. 2017, 4, 105–109. [Google Scholar]
- Adams, J. The effects of yin, yang and qi in the skin on pain. Medicines 2016, 3, 5. [Google Scholar] [CrossRef]
- Adams, J.; Wang, X. Control of pain with topical plant medicines. Asian Pac. Trop. Biomed. 2015, 5, 93–95. [Google Scholar] [CrossRef]
- Fontaine, P.; Wong, V.; Williams, T.; Garcia, C.; Adams, J. Chemical Composition and Antinociceptive Activity of California Sagebrush (Artemisia californica). J. Pharmacogn. Phytother. 2013, 5, 1–11. [Google Scholar]
- Adams, J. The use of California sagebrush (Artemisia californica) liniment to control pain. Pharmaceuticals 2012, 5, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Coderre, T. Topical drug therapies for neuropathic pain. Exp. Opin. Pharmacother. 2018, 19, 1211–1220. [Google Scholar] [CrossRef] [PubMed]
- Keppel Hesselink, J.; Kopsky, D.; Bhaskar, A. Skin matters! The role of keratinocytes in nociception: A rational argument for the development of topical analgesics. J. Pain Res. 2017, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Fang, Y.; Liu, D.; Chen, P.; Mei, Y. cAMP /PKA pathways and S56 phosphorylation are involved in AA/PGE2 induced increases in rNaV1.4 current. PLoS ONE 2015, 10, e0140715. [Google Scholar] [CrossRef] [PubMed]
- Adams, J. Chronic pain two cures. OBM Integr. Complement. Med. submitted.
- Parsadaniantz, S.M.; Rivat, C.; Rostène, W.; Réaux-Le Goazigo, A. Opioid and chemokine receptor crosstalk: A promising target for pain therapy? Nat. Rev. Neurosci. 2015, 16, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Cook, A.; Christensen, A.; Tewari, D.; McMahon, S.; Hamilton, J. Immune Cytokines and Their Receptors in Inflammatory Pain. Trends Immunol. 2018, 39, 240–255. [Google Scholar] [CrossRef]
- Sun, S.; Dong, X. TRP channels and itch. Semin. Immunopathol. 2016, 38, 293–307. [Google Scholar] [CrossRef]
- Xu, H.; Blair, N.T.; Clapham, D.E. Camphor activates and strongly desensitizes the transient receptor potential vanilloid subtype 1 channel in vanilloid-independent mechanism. J. Neurosci. 2005, 25, 8924–8937. [Google Scholar] [CrossRef]
- Vriens, J.; Appendino, G.; Nilius, B. Pharmacology of Vanilloid transient receptor potential cation channels. Mol. Pharmacol. 2009, 75, 1262–1279. [Google Scholar] [CrossRef]
- Vogt-Eisele, A.K.; Weber, K.; Sherkheli, M.A.; Vielhaber, G.; Panten, J.; Gisselmann, G.; Hatt, H. Monoterpenoid agonists of TRPV3. Br. J. Pharmacol. 2007, 151, 530–540. [Google Scholar] [CrossRef]
- Games, E.; Guerreiro, M.; Santana, F.; Pinheiro, N.; de Oliveira, E.; Lopes, F.; Olivo, C.; Tibério, L.; Martins, M.; Lago, H.; et al. Structurally related monoterpenes p-cymene, carvacrol and thymol isolated from essential oil from leaves of Lippia sidoides Cham. (Verbenaceae) protect mice against elastase-induced emphysema. Molecules 2016, 21, 1390. [Google Scholar] [CrossRef]
- Kim, D.; Lee, H.; Jeon, Y.; Han, Y.; Kee, J.; Kim, H.; Shin, H.; Kang, J.; Lee, B.; Kim, S.; et al. Alpha-pinene exhibits anti-inflammatory activity through the suppression of MAPKs and the NF-κB pathway in mouse peritoneal macrophages. Am. J. Chin. Med. 2015, 43, 731–742. [Google Scholar] [CrossRef]
- Luis, J.; Andres, L.; Perales, A. C-16 hydroxylated abietane diterpenes from Salvia mellifera. Absolute configuration and biogenetic implications. Tetrahedron 1993, 49, 4993–5000. [Google Scholar] [CrossRef]
- Luo, M.; Yang, J.; Li, M. Tanshinone IIA attenuates interleukin-17A induced systemic sclerosis patient-derived dermal vascular smooth muscle cell activation via inhibition of the extracellular signal regulated kinase signaling pathway. Clinics 2015, 70, 250–256. [Google Scholar]
- Wang, Y.; Wei, D.; Lai, Z.; Le, Y. Triptolide inhibits CC chemokines expressed in rat adjuvant induced arthritis. Int. Immunopharmacol. 2006, 6, 1825–1832. [Google Scholar] [CrossRef]
- Zhou, L.; Hu, Q.; Sui, H.; Ci, S.; Wang, Y.; Liu, X.; Liu, N.; Yin, P.; Qin, J.; Li, Q. Tanshinone IIA inhibits angiogenesis through down regulation of COX2 in human colorectal cancer. Asian Pac. J. Cancer Prev. 2012, 13, 4453–4458. [Google Scholar] [CrossRef]
- Bannuru, R.; McAlindon, T.; Sullivan, M.; Wong, J.; Kent, D.; Schmid, C. Effectiveness and implications of alternative placebo treatments a systematic review and network meta-analysis of osteoarthritis trials. Ann. Intern. Med. 2015, 163, 365–372. [Google Scholar] [CrossRef]
- Becker, S.; Navratilova, E.; Nees, F.; Van Damme, S. Emotional and motivational pain processing: Current state of knowledge and perspectives in translational research. Pain Res. Manag. 2018, 2018, 5457870. [Google Scholar] [CrossRef]
- Jaehne, E.; Baune, B. Effects of chemokine receptor signalling on cognition-like, emotion-like and sociability behaviours of CCR6 and CCR7 knockout mice. Behav. Brain Res. 2014, 261, 31–39. [Google Scholar] [CrossRef]
- Mansour, A.R.; Farmer, M.A.; Baliki, M.N.; Vania Apkarian, A. Chronic pain: The role of learning and brain plasticity. Restor. Neurol. Neurosci. 2014, 32, 129–139. [Google Scholar] [PubMed]
- Haringman, J.; Kraan, M.; Smeets, T.; Zwinderman, K.; Tak, P. Chemokine blockade and chronic inflammatory disease: Proof of concept in patients with rheumatoid arthritis. Ann. Rheum. Dis. 2003, 62, 715–721. [Google Scholar] [CrossRef] [PubMed]
- Barry, S.; Simtchouk, S.; Durrer, C.; Jung, M.; Little, J. Short term exercise training alters leukocyte chemokine receptors in obese patients. Med. Sci. Sports Exerc. 2017, 49, 1631–1640. [Google Scholar] [CrossRef] [PubMed]
- Troseid, M.; Lappegard, K.; Claudi, T.; Damas, J.; Morkrid, L.; Brendberg, R.; Mollnes, T. Exercise reduces plasma levels of the chemokines MCP-1 and IL-8 in subjects with the metabolic syndrome. Eur. Heart J. 2014, 25, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Crane, J.; MacNeil, L.; Lally, J.; Ford, R.; Bujak, A.; Brar, I.; Kemp, B.; Raha, S.; Steinberg, G.; Tarnopolsky, M. Exercise stimulated interleukin-15 is controlled by AMPK and regulates skin metabolism and aging. Aging Cell 2015, 14, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Bouchaud, G.; Gehrke, S.; Krieg, C.; Kolios, A.; Hafner, J.; Navarini, A.; French, L.; Boyman, O. Epidermal IL-15Ralpha acts as an endogenous antagonist of psoriasiform inflammation in mouse and man. J. Exp. Med. 2013, 210, 2105–2117. [Google Scholar] [CrossRef] [PubMed]
- Schechter, D.; Smith, A.; Beck, J.; Karim, R.; Azen, S. Outcomes of a mind-body treatment program for chronic back pain with no distinct structural pathology—A case series of patients diagnosed and treated as tension myositis syndrome. Altern. Ther. Health Med. 2007, 13, 26–35. [Google Scholar]
| Age | Site of Pain | Family History of Pain | Duration of Pain | Duration of Pain Relief |
|---|---|---|---|---|
| 20 | Legs | No | A few months | 1 day |
| 28 | Back | No | Several months | Several months |
| 62 | Knee | No | 2 years | Several months |
| 47 | Foot | No | 1 month | Partial |
| 55 | Foot | No | Several months | Partial |
| 48 | Shoulder | No | 2 months | Several months |
| 56 | Leg arthritis | DTS | Several years | 1 day |
| 47 | Hand arthritis | DTS | Several years | 1 day |
| 54 | Hip arthritis | DTS | Several years | 1 day |
| 49 | Neck | No | Several years | Several months |
| 68 | Neck | No | Several months | Several months |
| 70 | Neck | DTS | Several months | Several weeks |
| 37 | Foot | No | Several years | Several months |
| 70 | Torso | No | Several weeks | Several months |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adams, J.D.; Guhr, S.; Villaseñor, E. Salvia mellifera—How Does It Alleviate Chronic Pain? Medicines 2019, 6, 18. https://doi.org/10.3390/medicines6010018
Adams JD, Guhr S, Villaseñor E. Salvia mellifera—How Does It Alleviate Chronic Pain? Medicines. 2019; 6(1):18. https://doi.org/10.3390/medicines6010018
Chicago/Turabian StyleAdams, James David, Steven Guhr, and Enrique Villaseñor. 2019. "Salvia mellifera—How Does It Alleviate Chronic Pain?" Medicines 6, no. 1: 18. https://doi.org/10.3390/medicines6010018
APA StyleAdams, J. D., Guhr, S., & Villaseñor, E. (2019). Salvia mellifera—How Does It Alleviate Chronic Pain? Medicines, 6(1), 18. https://doi.org/10.3390/medicines6010018






