ShenLingLan Influences the Attachment and Migration of Ovarian Cancer Cells Potentially through the GSK3 Pathway
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Growth Assay
2.2. Protein Arrays for Detecting Phosphorylation Changes
2.3. Electric Cell-Substrate Impedance Sensing (ECIS)-Based Analyses on Cell Adhesion and Cell Migration
2.4. Ovarian Cancer Cohort, Analysis of GSK3Β Transcript
2.5. Quantitative Analysis of Gene Transcripts in Cells and Tissues
3. Results
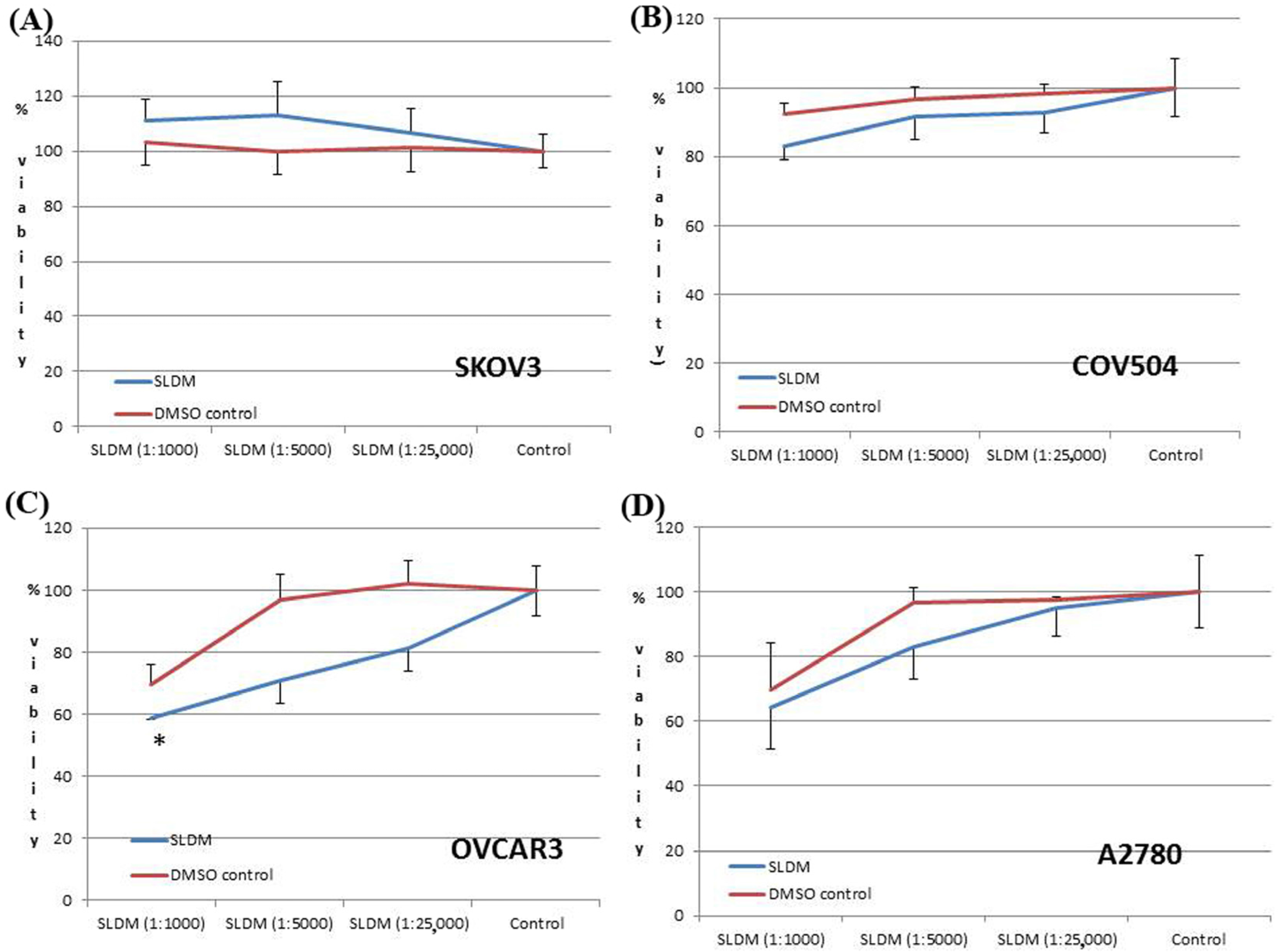
3.1. The Effect of SLDM Treatment on Ovarian Cancer Cell Viabiability
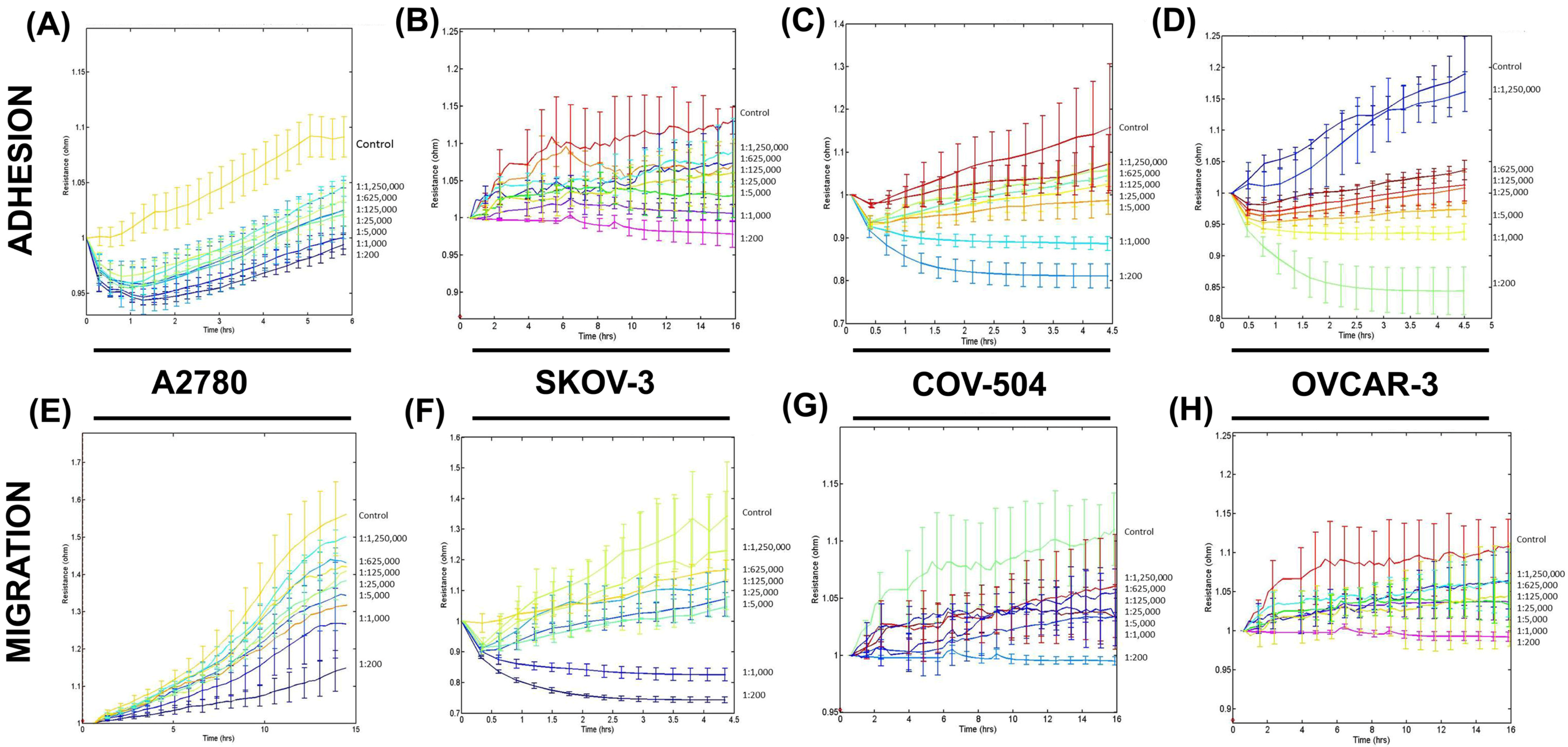
3.2. SLDM Had a Profound Impact on Cellular Adhesion and Migration of Ovarian Cancer Cells
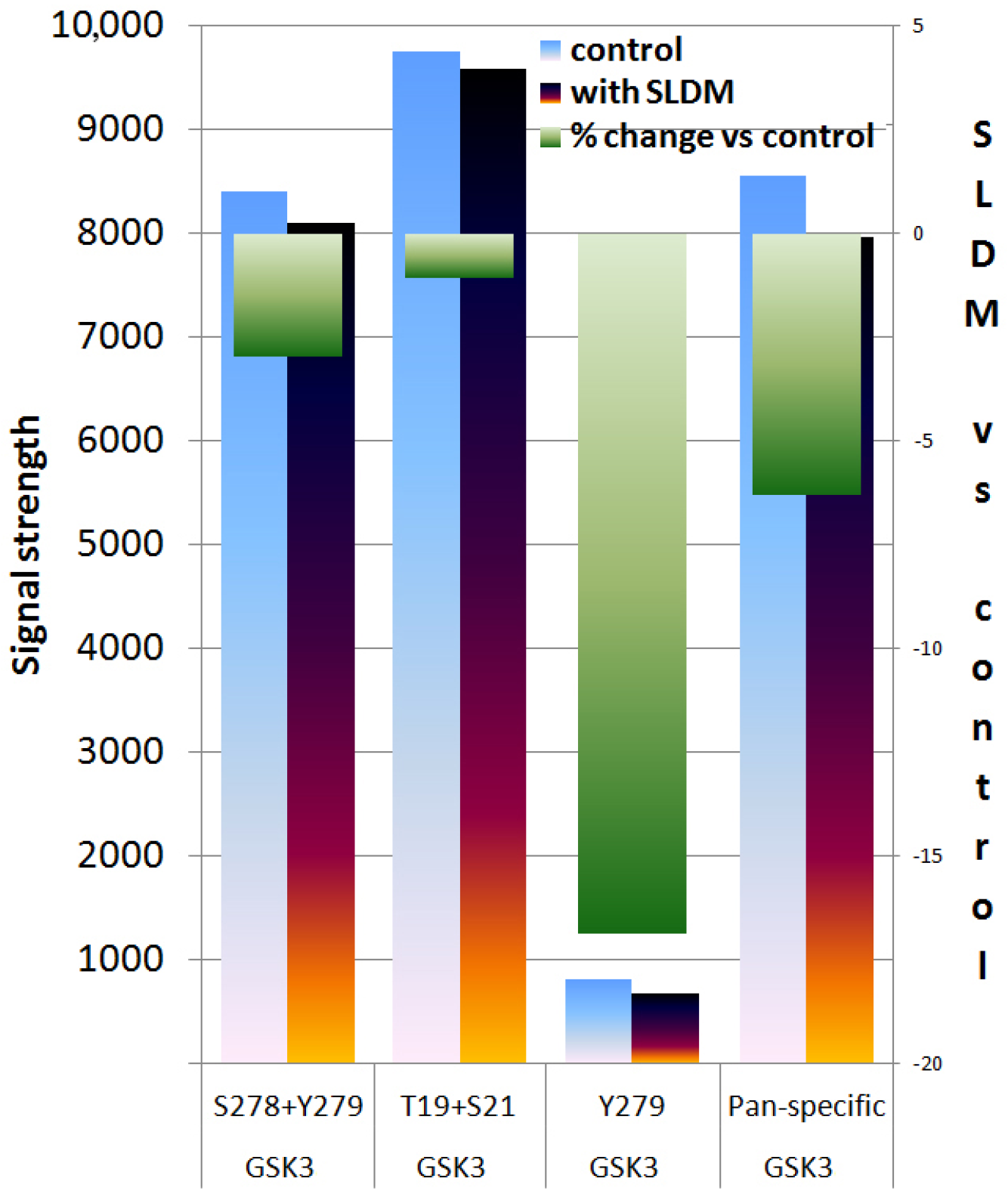
3.3. Protein Kinase Array Revealed that SLDM Inhibited Phosphorylation of GSK3 in Ovarian Cancer Cells
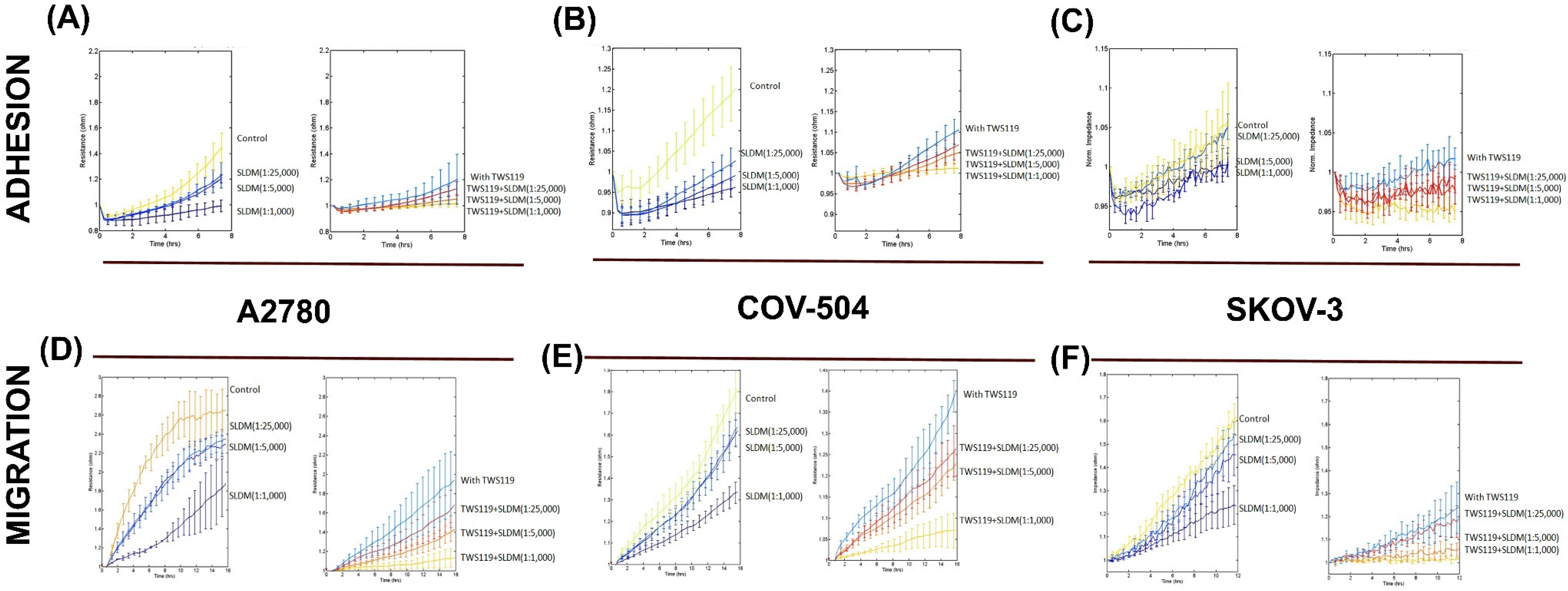
3.4. Impact of SLDM and GSK3 Kinase Inhibitor on Cell Migration
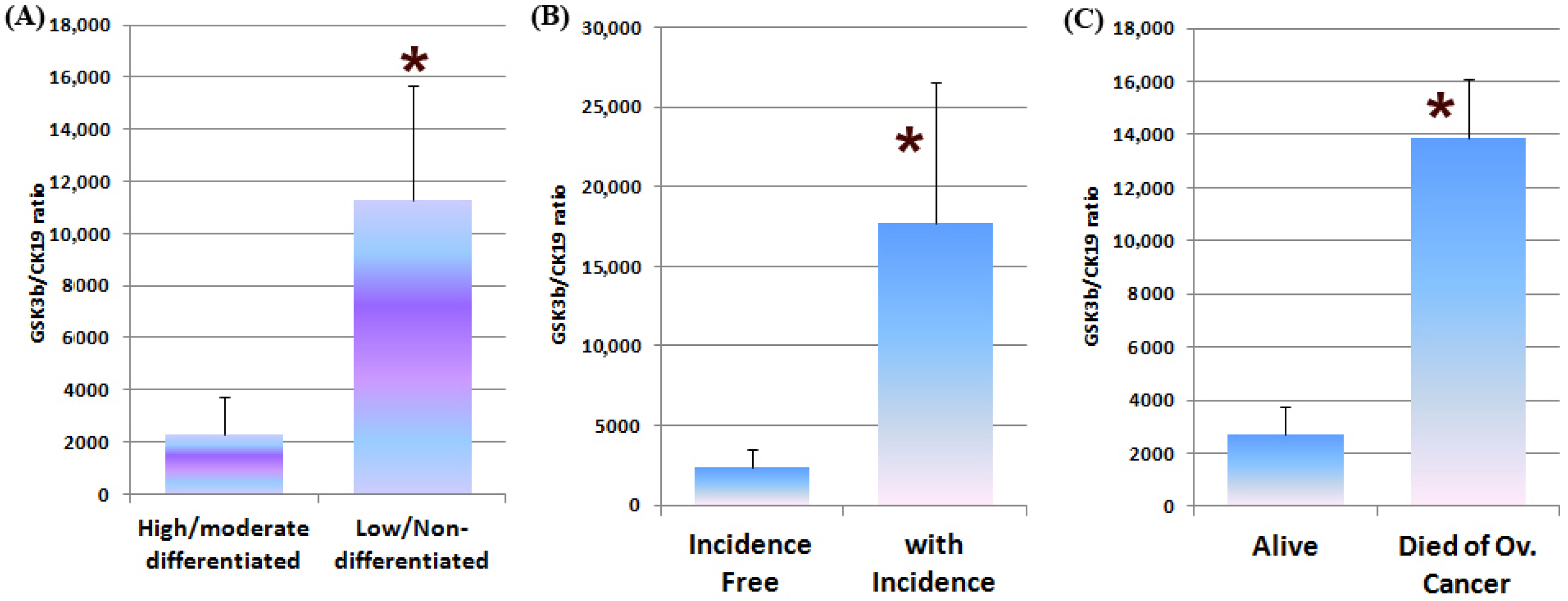
3.5. GSK3β Expression in Human Ovarian Cancer and the Clinical Implications
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cancer Research UK. Available online: http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/ovarian-cancer/incidence#ref-2 (accessed on 26 August 2016).
- Jasen, P. From the “silent killer” to the “whispering disease”: Ovarian cancer and the uses of metaphor. Med. Hist. 2009, 53, 489–512. [Google Scholar] [CrossRef] [PubMed]
- Maringe, C.; Walters, S.; Butler, J.; Coleman, M.P.; Hacker, N.; Hanna, L.; Mosgaard, B.J.; Nordin, A.; Rosen, B.; Engholm, G.; et al. Stage at diagnosis and ovarian cancer survival: Evidence from the international cancer benchmarking partnership. Gynecol. Oncol. 2012, 127, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Fan, G.; Zhang, S.; Gao, Y.; Greer, P.A.; Tonks, N.K. Hgf-independent regulation of met and gab1 by nonreceptor tyrosine kinase fer potentiates metastasis in ovarian cancer. Genes Dev. 2016, 30, 1542–1557. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Woodgett, J. Glycogen synthase kinase-3 and cancer: Good cop, bad cop? Cancer Cell 2008, 14, 351–353. [Google Scholar] [CrossRef] [PubMed]
- McCubrey, J.A.; Davis, N.M.; Abrams, S.L.; Montalto, G.; Cervello, M.; Basecke, J.; Libra, M.; Nicoletti, F.; Cocco, L.; Martelli, A.M.; et al. Diverse roles of gsk-3: Tumor promoter-tumor suppressor, target in cancer therapy. Adv. Biol. Regul. 2014, 54, 176–196. [Google Scholar] [CrossRef] [PubMed]
- Cao, Q.; Lu, X.; Feng, Y.J. Glycogen synthase kinase-3beta positively regulates the proliferation of human ovarian cancer cells. Cell Res. 2006, 16, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Shakoori, A.; Ougolkov, A.; Yu, Z.W.; Zhang, B.; Modarressi, M.H.; Billadeau, D.D.; Mai, M.; Takahashi, Y.; Minamoto, T. Deregulated gsk3beta activity in colorectal cancer: Its association with tumor cell survival and proliferation. Biochem. Biophys. Res. Commun. 2005, 334, 1365–1373. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Wang, L.; Gou, S.M.; Wang, T.L.; Zhang, M.; Liu, T.; Wang, C.Y. Shrna silencing glycogen synthase kinase-3 beta inhibits tumor growth and angiogenesis in pancreatic cancer. Cancer Lett. 2012, 316, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Peng, J.; Zhang, H.; Mondesire, W.H.; Jian, W.; Mills, G.B.; Hung, M.C.; Meric-Bernstam, F. Role of glycogen synthase kinase 3beta in rapamycin-mediated cell cycle regulation and chemosensitivity. Cancer Res. 2005, 65, 1961–1972. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lam, J.B.; Lam, K.S.; Liu, J.; Lam, M.C.; Hoo, R.L.; Wu, D.; Cooper, G.J.; Xu, A. Adiponectin modulates the glycogen synthase kinase-3beta/beta-catenin signaling pathway and attenuates mammary tumorigenesis of mda-mb-231 cells in nude mice. Cancer Res. 2006, 66, 11462–11470. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, C. What are the bona fide gsk3 substrates? Int. J. Alzheimer's Dis. 2011, 2011, 505607. [Google Scholar]
- Eom, T.Y.; Jope, R.S. Gsk3 beta n-terminus binding to p53 promotes its acetylation. Mol. Cancer 2009, 8, 14. [Google Scholar] [CrossRef] [PubMed]
- Hoeflich, K.P.; Luo, J.; Rubie, E.A.; Tsao, M.S.; Jin, O.; Woodgett, J.R. Requirement for glycogen synthase kinase-3beta in cell survival and nf-kappab activation. Nature 2000, 406, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wu, X. Shenlinglan in the treatment of chronic hepatitis-b, a clinical investigation of 30 patients cohort. LiaoNian J. Tradit. Chin. Med. 2006, 33, 60. [Google Scholar]
- Jiang, W.G.; Ye, L.; Ji, K.; Frewer, N.; Ji, J.; Mason, M.D. Inhibitory effects of yangzheng xiaoji on angiogenesis and the role of the focal adhesion kinase pathway. Int. J. Oncol. 2012, 41, 1635–1642. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Ji, K.; Frewer, N.; Ji, J.; Jiang, W.G. Impact of yangzheng xiaoji on the adhesion and migration of human cancer cells: The role of the akt signalling pathway. Anticancer Res. 2012, 32, 2537–2543. [Google Scholar] [PubMed]
- Li Sun, J.R.; Yei, T.; Gao, X. Yang capsule in treatment of hepatocellular carcinoma with interventional chemotherapy random double-blind, multicenter clinical study. World Chin. J. 2013, 8, 688–691. [Google Scholar]
- Cui, X.; Ma, W.; Bi, X. Effect of yangzhengxiaoji capsule on cellular immune function in patients with advanced gastric cancer chemotherapy. Chin. J. Diffic. Complicat. Cases 2011, 10, 703–704. [Google Scholar]
- Xue Kan, S.F.; Ji, J. Meta-anlaysis of the safety of yangzhengxiaoji capsule for the treatment of cancer and precancerosis. Chin. J. Clin. Oncol. 2013, 40, 1318–1323. [Google Scholar]
- Jiang, W.G.; Ye, L.; Ruge, F.; Owen, S.; Martin, T.; Sun, P.H.; Sanders, A.J.; Lane, J.; Satherley, L.; Weeks, H.P.; et al. Yangzheng xiaoji exerts anti-tumour growth effects by antagonising the effects of hgf and its receptor, cmet, in human lung cancer cells. J. Transl. Med. 2015, 13, 280. [Google Scholar] [CrossRef] [PubMed]
- Nazarenko, I.A.; Bhatnagar, S.K.; Hohman, R.J. A closed tube format for amplification and detection of DNA based on energy transfer. Nucleic Acid Res. 1997, 25, 2516–2521. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.G.; Watkins, G.; Lane, J.; Cunnick, G.H.; Douglas-Jones, A.; Mokbel, K.; Mansel, R.E. Prognostic value of rho gtpases and rho guanine nucleotide dissociation inhibitors in human breast cancers. Clin. Cancer Res. 2003, 9, 6432–6440. [Google Scholar] [PubMed]
- Davies, S.R.; Davies, M.L.; Sanders, A.; Parr, C.; Torkington, J.; Jiang, W.G. Differential expression of the ccn family member wisp-1, wisp-2 and wisp-3 in human colorectal cancer and the prognostic implications. Int. J. Oncol. 2010, 36, 1129–1136. [Google Scholar] [PubMed]
- Sun, T.; Rodriguez, M.; Kim, L. Glycogen synthase kinase 3 in the world of cell migration. Dev. Growth Differ. 2009, 51, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P.; Sleeman, J.P. Complex networks orchestrate epithelial-mesenchymal transitions. Nat. Rev. Mol. Cell Biol. 2006, 7, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Yu, S.X.; Lu, Y.; Bast, R.C., Jr.; Woodgett, J.R.; Mills, G.B. Phosphorylation and inactivation of glycogen synthase kinase 3 by protein kinase A. Proc. Natl. Acad. Sci. USA 2000, 97, 11960–11965. [Google Scholar] [CrossRef] [PubMed]
- Doble, B.W.; Woodgett, J.R. Gsk-3: Tricks of the trade for a multi-tasking kinase. J. Cell Sci. 2003, 116, 1175–1186. [Google Scholar] [CrossRef] [PubMed]
- Meijer, L.; Flajolet, M.; Greengard, P. Pharmacological inhibitors of glycogen synthase kinase 3. Trends Pharmacol. Sci. 2004, 25, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Cao, Q.; Feng, Y.J. Glycogen synthase kinase-3beta (gsk-3beta) promotes proliferation of ovarian cancer cells in vitro. Zhonghua Zhong Liu Za Zhi 2006, 28, 804–809. [Google Scholar] [PubMed]
- Fu, Y.; Hu, D.; Qiu, J.; Xie, X.; Ye, F.; Lu, W.G. Overexpression of glycogen synthase kinase-3 in ovarian carcinoma cells with acquired paclitaxel resistance. Int. J. Gynecol. Cancer 2011, 21, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Wang, X.; Cheng, X.; Ye, F.; Xie, X.; Lu, W. Clinicopathological and biological significance of aberrant activation of glycogen synthase kinase-3 in ovarian cancer. Onco Targets Ther. 2014, 7, 1159–1168. [Google Scholar] [PubMed]
| Clinical Information | Patient Numbers (n) |
|---|---|
| Stage | |
| 1 | 3 |
| 2 | 3 |
| 3 | 23 |
| 4 | 6 |
| Differentiation | |
| Low | 21 |
| Moderate | 1 |
| High | 2 |
| Tumour Subtype | |
| Adenocarcinoma | 44 |
| Transitional | 2 |
| Carcinoma | 1 |
| Small Cell Ccarcinoma | 1 |
| Nonadenoma | 5 |
| Metastasis | |
| No metastasis | 36 |
| Metastasis | 16 |
| Incidence | |
| Incidence free | 25 |
| With incidence | 27 |
| Survival | |
| Alive | 36 |
| Died | 9 |
| Gene Target | Primer Sequence (5′-3′) |
|---|---|
| GSK-3β | |
| Forward | AACAGGACATTTCACCTCAG |
| Reverse | ACTGAACCTGACCGTACAGGTGTATACTCCAGCAGACG |
| CK-19 | |
| Forward | AGCCACTACTACACGACCAT |
| Reverse | ACTGAACCTGACCGTACATCGATCTGCAGGACAATC |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Owen, S.; Ruge, F.; Gao, Y.; Yang, Y.; Hou, J.; Chen, J.; Feng, Y.; Zhang, H.; Gao, Y.; Wang, H.; et al. ShenLingLan Influences the Attachment and Migration of Ovarian Cancer Cells Potentially through the GSK3 Pathway. Medicines 2017, 4, 10. https://doi.org/10.3390/medicines4010010
Owen S, Ruge F, Gao Y, Yang Y, Hou J, Chen J, Feng Y, Zhang H, Gao Y, Wang H, et al. ShenLingLan Influences the Attachment and Migration of Ovarian Cancer Cells Potentially through the GSK3 Pathway. Medicines. 2017; 4(1):10. https://doi.org/10.3390/medicines4010010
Chicago/Turabian StyleOwen, Sioned, Fiona Ruge, Yunong Gao, Ying Yang, Jianqing Hou, Jian Chen, Yi Feng, Huiming Zhang, Yong Gao, Hongtao Wang, and et al. 2017. "ShenLingLan Influences the Attachment and Migration of Ovarian Cancer Cells Potentially through the GSK3 Pathway" Medicines 4, no. 1: 10. https://doi.org/10.3390/medicines4010010
APA StyleOwen, S., Ruge, F., Gao, Y., Yang, Y., Hou, J., Chen, J., Feng, Y., Zhang, H., Gao, Y., Wang, H., Wei, C., Wu, Y., & Jiang, W. G. (2017). ShenLingLan Influences the Attachment and Migration of Ovarian Cancer Cells Potentially through the GSK3 Pathway. Medicines, 4(1), 10. https://doi.org/10.3390/medicines4010010







