Deriving Soil Quality Criteria of Chromium Based on Species Sensitivity Distribution Methodology
Abstract
1. Introduction
2. Materials and Methods
2.1. Soils and Chemicals
2.2. Short-Term Toxicity Tests
2.3. Species Sensitivity Distribution (SSD)
2.4. Statistic Analysis and Data Integration
3. Results
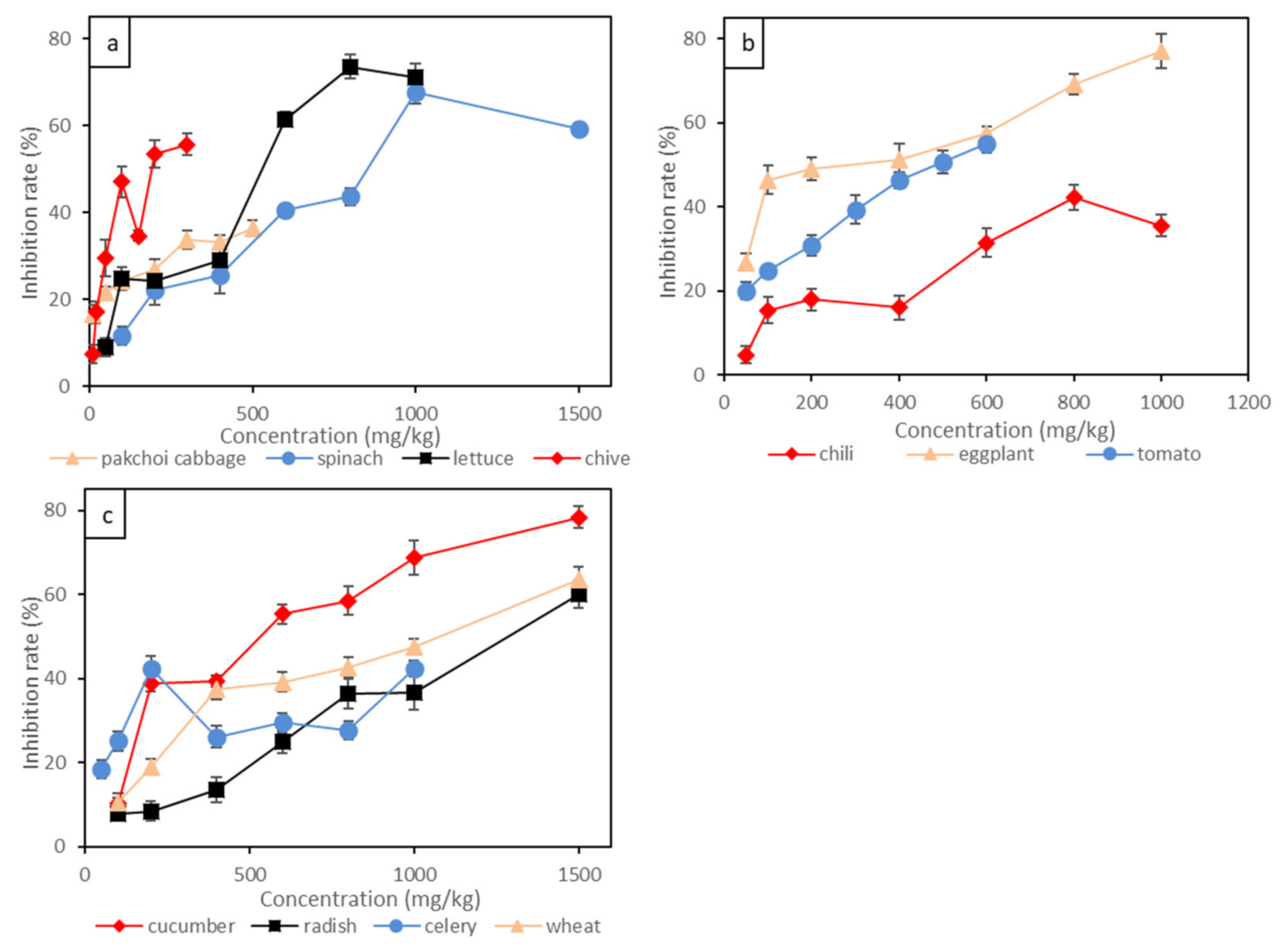
3.1. Species Sensitivity to Short-Term Toxicity of Cr
3.1.1. Short-Term Toxicity Tests of Cr(VI)
3.1.2. Short-Term Toxicity Tests of Cr(III)
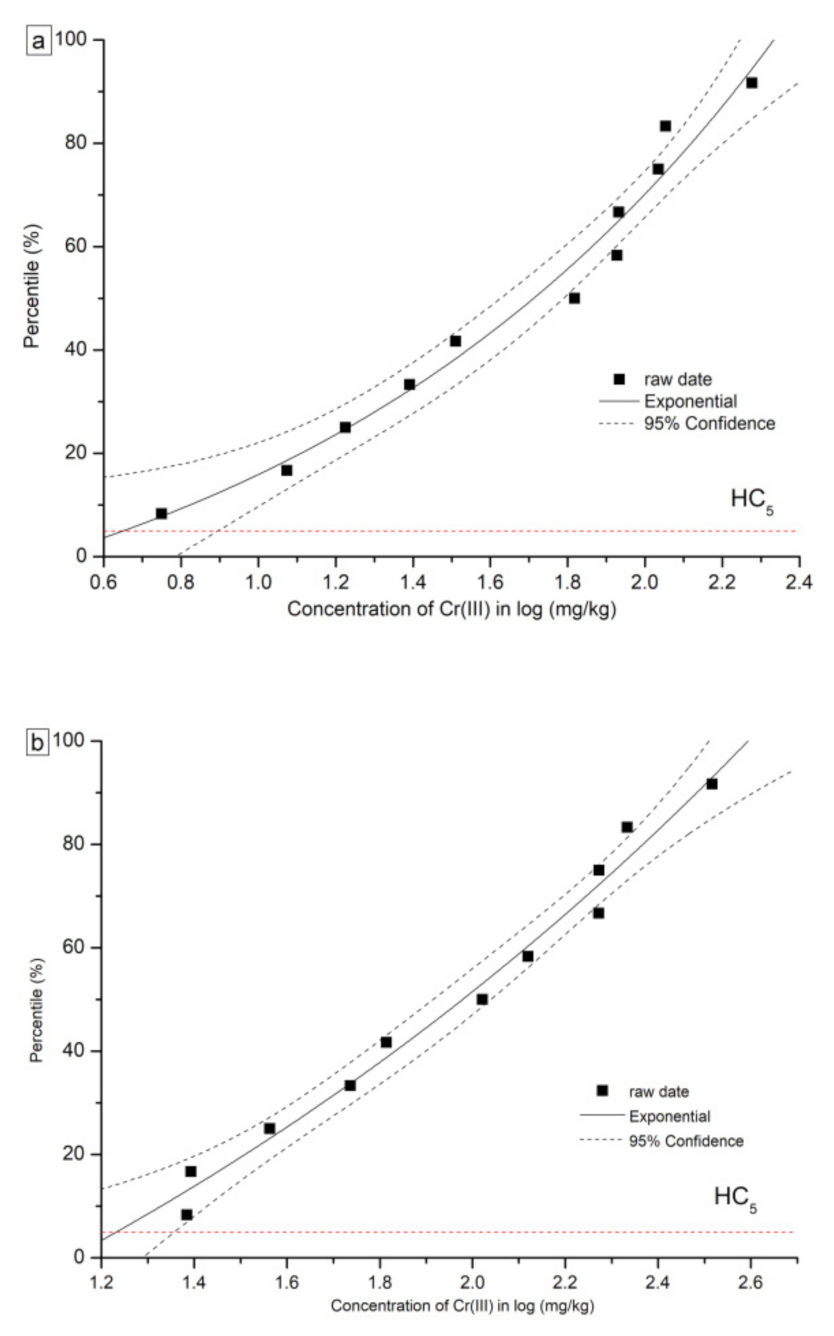
3.2. Ecological Risk Assessment Based on Species Sensitivity Distributions (SSD)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kotaś, J.; Stasicka, Z. Chromium occurrence in the environment and methods of its speciation. Environ. Pollut. 2000, 107, 263–283. [Google Scholar] [CrossRef]
- Turner, R.D.R.; Warne, M.S.J.; Dawes, L.A.; Vardy, S.; Will, G.D. Irrigated greywater in an urban sub-division as a potential source of metals to soil, groundwater and surface water. J. Environ. Manag. 2016, 183, 806–817. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, V.; Sarkar, A.; Singh, S.; Singh, P.; De Araujo, A.S.F.; Singh, R.P. Agroecological responses of heavy metal pollution with special emphasis on soil health and plant performances. Front. Environ. Sci. 2017, 5, 64. [Google Scholar] [CrossRef]
- Gabarron, M.; Faz, A.; Martinez-Martinez, S.; Acosta, J. Change in metals and arsenic distribution in soil and their bioavailability beside old tailing ponds. J. Environ. Manag. 2018, 212, 292–300. [Google Scholar] [CrossRef]
- Wang, M.; Liu, R.; Lu, X.; Zhu, Z.; Wang, H.; Jiang, L.; Liu, J.; Wu, Z. Heavy metal contamination and ecological risk assessment of swine manure irrigated vegetable soils in Jiangxi Province, China. Bull. Environ. Contam. Toxicol. 2018, 100, 634–640. [Google Scholar] [CrossRef]
- Chen, C.W.; Lim, Y.C.; Chen, C.F.; Ju, Y.R.; Dong, C.D. Spatial distribution and ecological risk assessment of sediment metals in a highly industrialized coastal zone southwestern Taiwan. Environ. Sci. Pollut. Res. 2019, 26, 14717–14731. [Google Scholar] [CrossRef]
- Ramos-Miras, J.J.; Gil, C.; Rodriguez, M.J.A.; Bech, J.; Boluda, R. Ecological risk assessment of mercury and chromium in greenhouse soils. Environ. Geochem. Health 2020, 42, 313–324. [Google Scholar] [CrossRef]
- Rastmanesh, F.; Shalbaf, F.; Moradi, R.; Prinzhofer, A. Health risk assessment of heavy metals in Ahvaz oilfield using environmental indicators. Int. J. Environ. Sci. Technol. 2020, 17, 4669–4678. [Google Scholar] [CrossRef]
- Richard, F.C.; Bourg, A.C.M. Aqueous geochemistry of chromium: A review. Water Res. 1991, 25, 807–816. [Google Scholar] [CrossRef]
- Yu, X.Z.; Gu, J.D. Accumulation and distribution of trivalent chromium and effects on hybrid willow (Salix matsudana Koidz × alba L.). Metabolism. Arch. Environ. Contam. Toxicol. 2007, 52, 503–511. [Google Scholar] [CrossRef]
- Zayed, A.; Lytle, C.M.; Terry, Q.N. Chromium accumulation, translocation and chemical speciation in vegetable crops. Planta 1998, 206, 293–299. [Google Scholar] [CrossRef]
- do Nascimento, J.L.; de Almeida, A.A.F.; Barroso, J.P.; Mangabeira, P.A.; Ahnert, D.; Sousa, A.G.; Silva, J.V.S.; Baligar, V.C. Physiological, ultrastructural, biochemical and molecular responses of young cocoa plants to the toxicity of Cr (III) in soil. Ecotox. Environ. Saf. 2018, 159, 272–283. [Google Scholar] [CrossRef]
- Sinha, V.; Pakshirajan, K.; Chaturvedi, R. Chromium tolerance, bioaccumulation and localization in plants: An overview. J. Environ. Manag. 2018, 206, 715–730. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.T.; Shen, H. Bacterial reduction of hexavalent chromium. J. Ind. Microbiol. 1995, 14, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Montes, M.O.; Peralta-Videa, J.R.; Parsons, J.G.; Diaz, B.C.; Gardea-Torresdey, J.L. Spectroscopic determination of the toxicity, absorption, reduction, and translocation of Cr(VI) in two Magnoliopsida species. Int. J. Phytorem. 2013, 15, 168–187. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.X.; Huang, G.H. Environmental Biogeochemistry and Global Environmental Changes; Science Press: Beijing, China, 2001. [Google Scholar]
- Fernandez, M.D.; Vega, M.M.; Tarazona, J.V. Risk-based ecological soil quality criteria for the characterization of contaminated soils. Combination of chemical and biological tools. Sci. Total Environ. 2006, 366, 466–484. [Google Scholar] [CrossRef]
- Zhou, Q.X.; Teng, Y.; Liu, Y. A study on soil-environmental quality criteria and standards of arsenic. Appl. Geochem. 2017, 77, 158–166. [Google Scholar] [CrossRef]
- Zhou, Q.X.; Luo, Y.; Zhu, L.Y. Scientific research on environmental benchmark values and rivision of national environmental standards in China. J. Agro-Environ. Sci. 2007, 26, 1–5. (In Chinese) [Google Scholar] [CrossRef]
- Taghipour, M.; Jalali, M. Influence of organic acids on kinetic release of chromium in soil contaminated with leather factory waste in the presence of some adsorbents. Chemosphere 2016, 155, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Shahid, M.; Shamshad, S.; Rafiq, M.; Khalid, S.; Bibi, I.; Niazi, N.K.; Dumat, C.; Rashid, M.I. Chromium speciation, bioavailability, uptake, toxicity and detoxification in soil-plant system: A review. Chemosphere 2017, 178, 513–533. [Google Scholar] [CrossRef]
- EPA, U.S. Regional Screening Levels for Chemical Contaminants at Superfund Sites; United States Environmental Protection Agency: Washington, DC, USA, 2010.
- Hose, G.C.; Van den Brink, P.J. Confirming the species-sensitivity distribution concept for endosulfan using laboratory, mesocosm, and field data. Arch. Environ. Contam. Toxicol. 2004, 47, 511–520. [Google Scholar] [CrossRef]
- Xin, Z.; Wenchao, Z.; Zhenguang, Y.; Yiguo, H.; Zhengtao, L.; Xianliang, Y.; Xiaonan, W.; Tingting, L.; Liming, Z. Species sensitivity analysis of heavy metals to freshwater organisms. Ecotoxicology 2015, 24, 1621–1631. [Google Scholar] [CrossRef]
- Wang, X.N.; Yan, Z.G.; Liu, Z.T.; Zhang, C.; Wang, W.; Li, H. Comparison of species sensitivity distributions for species from China and the USA. Environ. Sci. Pollut. Res. 2014, 21, 168–176. [Google Scholar] [CrossRef] [PubMed]
- van Straalen, N.M. Threshold models for species sensitivity distributions applied to aquatic risk assessment for zinc. Environ. Toxicol. Pharmacol. 2002, 11, 167–172. [Google Scholar] [CrossRef]
- Nagai, T.; Taya, K. Estimation of herbicide species sensitivity distribution using single-species algal toxicity data and information on the mode of action. Environ. Toxicol. Chem. 2015, 34, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.A.; Ater, M.; Sunahara, G.I.; Robidoux, P.Y. Phytotoxicity and bioaccumulation of copper and chromium using barley (Hordeum vulgare L.) in spiked artificial and natural forest soils. Ecotox. Environ. Saf. 2004, 57, 363–374. [Google Scholar] [CrossRef]
- López-Luna, J.; González-Chávez, M.C.; Esparza-García, F.J.; Rodríguez-Vázquez, R. Toxicity assessment of soil amended with tannery sludge, trivalent chromium and hexavalent chromium, using wheat, oat and sorghum plants. J. Hazard. Mater. 2009, 163, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.H.; Bradshaw, A.D. A comparison of the toxicity of heavy metals, using root elongation of rye grass, Lolium perenne. New Phytol. 1982, 91, 255–261. [Google Scholar] [CrossRef]
- Lu, R. Analytical Methods for Soils and Agricultural Chemistry; China Agricultural Science and Technology Press: Beijing, China, 1999. [Google Scholar]
- CCME. A Protocol for the Derivation of Environmental and Human Health Soil Quality Guidelines; Canadian Council of Ministers of the Environment: Winnipeg, MB, Canada, 2006.
- Schroer, A.F.W.; Belgers, J.D.M.; Brock, C.M.; Matser, A.M.; Maund, S.J.; Brink, P.J.V.D. Comparison of laboratory single species and field population-level effects of the pyrethroid insecticide λ-cyhalothrin on freshwater invertebrates. Arch. Environ. Contam. Toxicol. 2004, 46, 324–335. [Google Scholar] [CrossRef]
- Kamran, M.; Wang, D.; Alhaithloul, H.A.S.; Alghanem, S.M.; Aftab, T.; Xie, K.; Lu, Y.; Shi, C.; Sun, J.; Gu, W.; et al. Jasmonic acid-mediated enhanced regulation of oxidative, glyoxalase defense system and reduced chromium uptake contributes to alleviation of chromium (VI) toxicity in choysum (Brassica parachinensis L.). Ecotox. Environ. Saf. 2021, 208, 111758. [Google Scholar] [CrossRef]
- Kumar, P.; Tokas, J.; Singal, H.R. Amelioration of chromium(VI) toxicity in Sorghum (Sorghum bicolor L.) using glycine betaine. Sci. Rep. 2019, 9, 16020. [Google Scholar] [CrossRef]
- Ulhassan, Z.; Gill, R.A.; Huang, H.; Ali, S.; Mwamba, T.M.; Ali, B.; Huang, Q.; Hamid, Y.; Khan, A.R.; Wang, J.; et al. Selenium mitigates the chromium toxicity in Brassicca napus L. by ameliorating nutrients uptake, amino acids metabolism and antioxidant defense system. Plant Physiol. Biochem. 2019, 145, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Ai, H.; Xu, X.; Chen, K.; Niu, H.; Zhu, H.; Sun, J.; Du, D.; Chen, L. Nitric oxide alleviates toxicity of hexavalent chromium on tall fescue and improves performance of photosystem II. Ecotox. Environ. Saf. 2018, 164, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.P.; Mahajan, P.; Kaur, S.; Batish, D.R.; Kohli, R.K. Chromium toxicity and tolerance in plants. Environ. Chem. Lett. 2013, 11, 229–254. [Google Scholar] [CrossRef]
- Depault, F.; Cojocaru, M.; Fortin, F.; Chakrabarti, S.; Lemieux, N. Genotoxic effects of chromium(VI) and cadmium(II) in human blood lymphocytes using the electron microscopy in situ end-labeling (EM-ISEL) assay. Toxicol. Vitro 2006, 20, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Riaz, M.; Yasmeen, T.; Arif, M.S.; Ashraf, M.A.; Hussain, Q.; Shahzad, S.M.; Rizwan, M.; Mehmood, M.W.; Zia, A.; Mian, I.A.; et al. Variations in morphological and physiological traits of wheat regulated by chromium species in long-term tannery effluent irrigated soils. Chemosphere 2019, 222, 891–903. [Google Scholar] [CrossRef]
- Ma, Y.; Rajkumar, M.; Zhang, C.; Freitas, H. Beneficial role of bacterial endophytes in heavy metal phytoremediation. J. Environ. Manag. 2016, 174, 14–25. [Google Scholar] [CrossRef]
- Tekerlekopoulou, A.G.; Tsiflikiotou, M.; Akritidou, L.; Viennas, A.; Tsiamis, G.; Pavlou, S.; Bourtzis, K.; Vayenas, D.V. Modelling of biological Cr(VI) removal in draw-fill reactors using microorganisms in suspended and attached growth systems. Water Res. 2013, 47, 623–636. [Google Scholar] [CrossRef]
- Otitoloju, A.A. Relevance of joint action toxicity evaluations in setting realistic environmental safe limits of heavy metals. J. Environ. Manag. 2003, 67, 121–128. [Google Scholar] [CrossRef]
- MEGJ. Environmental Quality Standards for Soil Pollution; Ministry of the Environment, Government of Japan (MEGJ): Tokyo, Japan, 2001.
- RIVM. Maximum Permissible Concentrations and Negligible Concentrations for Metals, Taking Background Concentrations into Account; National Institute of Public Health and Environment: Bilthoven, The Netherlands, 1997. [Google Scholar]
- FSPR. Precaution Values for Soils Pursuant to 8 Paragraph Ederal Soil Protection Law; Federal Soil Protection and Contaminated Sites Ordiance (BBodSchV): Hannover, Germany, 1999. [Google Scholar]
- Naturvårdsverket. Nya Generella Riktvärden för Förorenad Mark. 2009. Available online: www.naturvardsverket.se/ebh (accessed on 12 October 2020).
- NEPC. Guidelines on Healthbased Investigation Levels; National Environmental Protection Council (NEPC): Sydney, Australia, 1999.
- PCD. Soil Quality Standard for Habitat and Agriculture. Pollution Control Department of Thailand (PCD). Notification of National Environmental Board No. 25. 2004. Available online: http://www.pcd.go.th/info_serv/en_reg_std_soil01.html#s1 (accessed on 12 October 2020).
- He, J.; He, H.; Yan, Z.; Gao, F.; Zheng, X.; Fan, J.; Wang, Y. Comparative analysis of freshwater species sensitivity distributions and ecotoxicity for priority pesticides: Implications for water quality criteria. Ecotox. Environ. Saf. 2019, 176, 119–124. [Google Scholar] [CrossRef]
- Sorgog, K.; Kamo, M. Quantifying the precision of ecological risk: Conventional assessment factor method vs. species sensitivity distribution method. Ecotox. Environ. Saf. 2019, 183, 109494. [Google Scholar] [CrossRef] [PubMed]
- China. The National Soil Pollution Survey Bulletin. Ministry of Environmental Protection and Ministry of Land and Resources of P. R. China. 2014. Available online: http://www.sdpc.gov.cn/fzgggz/ncjj/zhdt/201404/t20140418_607888.html (accessed on 12 October 2020). (In Chinese)


| pH | Organic Matter (g/kg) | Cation Exchange Capacity (cmol/kg) | Total Nitrogen (g/kg) | Total Phosphorus (%) |
|---|---|---|---|---|
| 7.1 | 16.77 | 29.5 | 1.48 | 1.48 |
| Species | Cr(III) mg/kg | Cr(VI) mg/kg |
|---|---|---|
| Brassica rapa chinensis | 0, 10, 50, 100, 200, 300, 400, 500 | 0, 1, 2, 4, 6, 8, 10, 20 |
| Lactuca sativa L. | 0, 50, 100, 200, 400, 600, 800, 1000 | 0, 1, 2, 4, 6, 8, 10, 20 |
| Allium schoenoprasum L. | 0, 10, 20, 50, 100, 150, 200, 300 | 0, 1, 2, 4, 6, 8, 10, 20 |
| Triticum aestivum L. | 0, 100, 200, 400, 600, 800, 1000, 1500 | 0, 1, 2.5, 5, 10, 20, 40, 60, 80 |
| Solanum lycopersicum | 0, 50, 100, 200, 300, 400, 500, 600 | 0, 1, 2, 4, 6, 8, 10, 20 |
| Raphanus sativus L. | 0, 100, 200, 400, 600, 800, 1000, 1500 | 0, 1, 2.5, 5, 10, 15, 20, 40 |
| Capsicum annuum L. | 0, 50, 100, 200, 400, 600, 800, 1000 | 0, 2.5, 5, 10, 15, 20, 30, 40 |
| Solanum melongena L. | 0, 50, 100, 200, 400, 600, 800, 1000 | 0, 2, 4, 6, 8, 10, 15, 20 |
| Spinacia oleracea L. | 0, 100, 200, 400, 600, 800, 1000, 1500 | 0, 1, 2, 5, 10, 15, 20, 30 |
| Apium graveolens L. | 0, 50, 100, 200, 400, 600, 800, 1000 | 0, 2.5, 5, 7.5, 10, 15, 30 |
| Cucumis sativus | 0, 100, 200, 400, 600, 800, 1000, 1500 | 0, 5, 10, 15, 20, 30, 40, 80 |
| Species | Rank | EC10 mg/kg | Rank | EC20 mg/kg |
|---|---|---|---|---|
| Brassica rapa chinensis | 1 | 0.65 (0.55~0.76) | 1 | 1.13 (0.97~1.33) |
| Lactuca sativa L. | 2 | 1.26 (1.10~1.44) | 2 | 1.81 (1.58~2.07) |
| Allium schoenoprasum L. | 3 | 1.42 (1.01~2.00) | 3 | 1.98 (1.41~2.80) |
| Triticum aestivum L. | 4 | 1.50 (1.18~1.91) | 4 | 2.25 (1.77~2.86) |
| Solanum lycopersicum | 5 | 1.55 (1.10~2.18) | 5 | 2.61 (1.85~3.67) |
| Raphanus sativus L. | 6 | 2.14 (1.62~2.83) | 6 | 3.19 (2.41~4.22) |
| Capsicum annuum L. | 7 | 2.63 (1.99~3.46) | 7 | 3.67 (2.78~4.83) |
| Solanum melongena L. | 8 | 2.78 (2.26~3.42) | 8 | 3.71 (3.02~4.57) |
| Spinacia oleracea L. | 9 | 3.71 (2.83~4.86) | 9 | 5.13 (3.91~6.71) |
| Apium graveolens L. | 10 | 4.39 (2.30~8.38) | 10 | 5.87 (3.07~11.22) |
| Cucumis sativus | 11 | 12.83 (10.88~15.13) | 11 | 16.62 (14.09~19.61) |
| Species | Rank | EC10 mg/kg | Rank | EC20 mg/kg |
|---|---|---|---|---|
| Brassica rapa chinensis | 1 | 5.62 (4.62~6.98) | 3 | 36.56 (29.12~46.63) |
| Allium schoenoprasum L. | 2 | 11.84 (8.53~16.45) | 1 | 24.21 (17.44~33.62) |
| Solanum melongena L. | 3 | 16.78 (13.05~22.21) | 2 | 24.71 (16.97~35.98) |
| Apium graveolens L. | 4 | 24.59 (21.02~29.54) | 4 | 54.52 (39.31~75.63) |
| Solanum lycopersicum | 5 | 32.35 (25.00~41.86) | 5 | 65.16 (50.36~84.32) |
| Lactuca sativa L. | 6 | 65.72 (41.86~103.20) | 6 | 105.10 (66.96~165.10) |
| Capsicum annuum L. | 7 | 84.68 (51.90~138.20) | 10 | 215.60 (132.10~351.70) |
| Cucumis sativus | 8 | 85.58 (67.52~108.50) | 7 | 131.70 (103.90~166.90) |
| Triticum aestivum L. | 9 | 108.30 (87.17~134.70) | 9 | 187.40 (150.80~232.90) |
| Spinacia oleracea L. | 10 | 113.10 (74.35~171.90) | 8 | 187.20 (123.00~284.70) |
| Raphanus sativus L. | 11 | 189.10 (115.90~308.30) | 11 | 328.60 (201.50~535.80) |
| Contaminants | Cr(VI) | Cr(III) | ||
|---|---|---|---|---|
| EC10 | EC20 | EC10 | EC20 | |
| Model | Slogistic1 | Slogistic3 | Exponential | Exponential |
| Formula | y = a/(1 + exp (−k × (x − xc))) | y = a/(1 + b × exp (−k × x)) | y = y0 + A × exp (R0 × x) | y = y0 + A × exp (R0 × x) |
| Parameter | a = 93.04 | a = 92.75 | y0 = −29.50 | y0 = −110.64 |
| xc = 0.30 | b = 18.30 | A = 20.69 | A = 67.19 | |
| k = 5.59 | k = 6.33 | R0 = 0.79 | R0 = 0.44 | |
| R2 | 0.97 | 0.99 | 0.97 | 0.98 |
| HC5 | 0.60 | 1.01 | 4.51 | 6.65 |
| HC10 | 0.83 | 1.34 | 16.98 | 21.33 |
| Country | Cr(VI) | Total Cr | Reference |
|---|---|---|---|
| Australia | 100 | NEPC, 1999 | |
| Canada | 0.4 | CCME, 2006 | |
| Germany | 30 (sand), 60 (loam), 100 (clay) 1 | FSPR, 1999 | |
| Sweden | 80 | Naturvårdsverket, 2009 | |
| Netherlands | 100 | RIVM, 1997 | |
| United States | 0.29 | U.S. EPA, 2010 | |
| Japan | 0.05 2 | MEGJ | |
| Thailand | 300 | PCD, 2004 | |
| Belgium | 2.5 | 34 | Carlon, 2007 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Zhou, Q.; Wang, Y.; Cheng, S.; Hao, W. Deriving Soil Quality Criteria of Chromium Based on Species Sensitivity Distribution Methodology. Toxics 2021, 9, 58. https://doi.org/10.3390/toxics9030058
Liu Y, Zhou Q, Wang Y, Cheng S, Hao W. Deriving Soil Quality Criteria of Chromium Based on Species Sensitivity Distribution Methodology. Toxics. 2021; 9(3):58. https://doi.org/10.3390/toxics9030058
Chicago/Turabian StyleLiu, Yuxia, Qixing Zhou, Yi Wang, Siwen Cheng, and Weiduo Hao. 2021. "Deriving Soil Quality Criteria of Chromium Based on Species Sensitivity Distribution Methodology" Toxics 9, no. 3: 58. https://doi.org/10.3390/toxics9030058
APA StyleLiu, Y., Zhou, Q., Wang, Y., Cheng, S., & Hao, W. (2021). Deriving Soil Quality Criteria of Chromium Based on Species Sensitivity Distribution Methodology. Toxics, 9(3), 58. https://doi.org/10.3390/toxics9030058






