Coupling Environmental Whole Mixture Toxicity Screening with Unbiased RNA-Seq Reveals Site-Specific Biological Responses in Zebrafish
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Sampling Methods
2.3. Chemical Analysis
2.4. Zebrafish Rearing, Exposure, and Morphological Assessment
2.5. RNA Isolation
2.6. Sequencing Data Pipeline
2.7. RNA-Seq Analysis
3. Results and Discussion
3.1. Chemical Characterization of PSD Extracts: Parallel PAH Contamination, Divergent Qualitative Screen
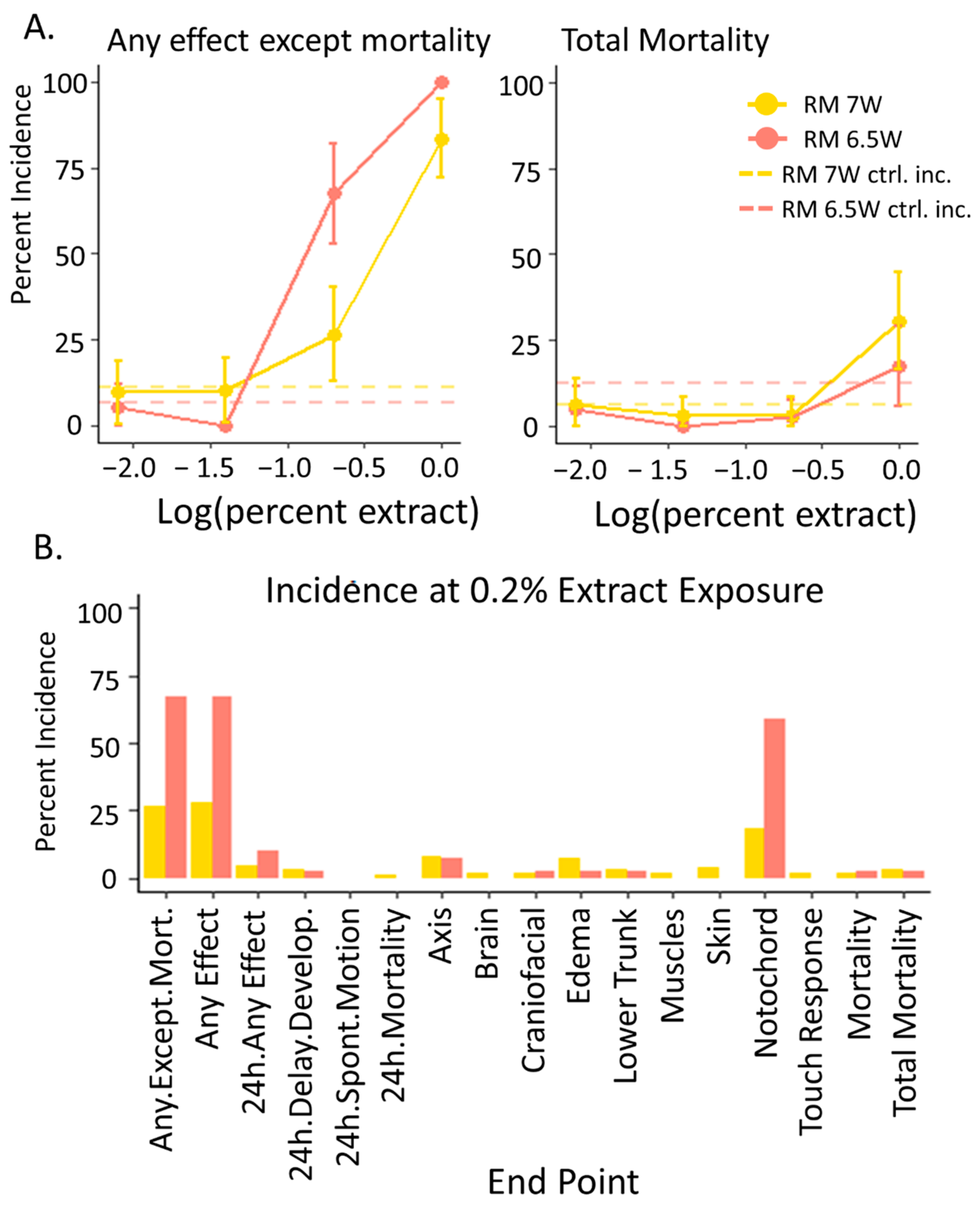
3.2. Developmental Toxicity: RM 6.5W Was More Toxic Than RM 7W
3.3. RNA-Seq: Direct Comparison of PHSS Extract Responses
3.3.1. RM 6.5W Elicits a More Robust Gene Expression Response than RM 7W
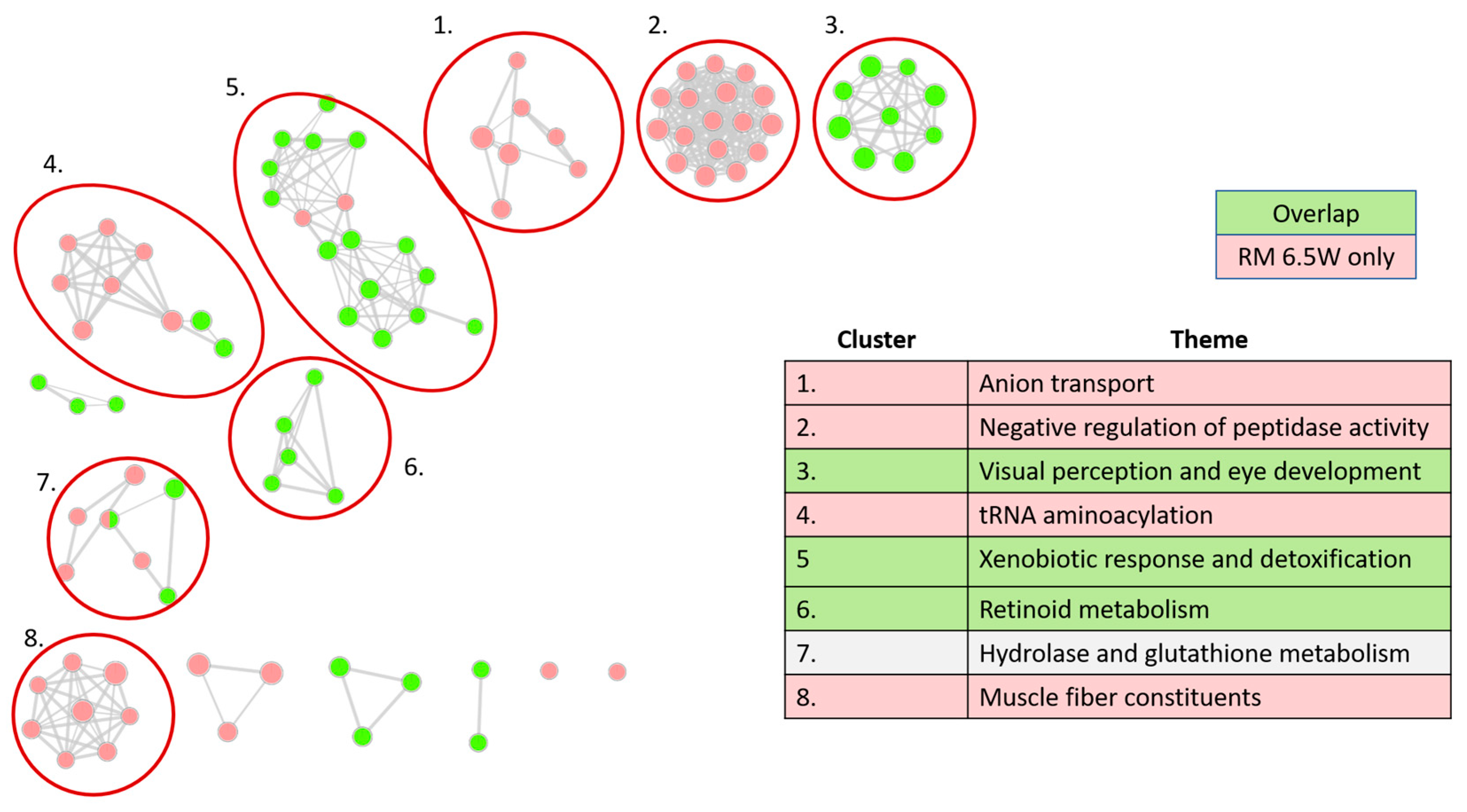
3.3.2. Distinct Themes Underly the Transcriptional Response to PHSS Extract Exposures
3.3.3. Xenobiotic Metabolism Glutathione Processes Are among the Expected GO Network Clusters
3.3.4. Visual System Development and Muscle Fiber Related Genes Are Disrupted
3.4. RNA-Seq: Comparison of Mixtures to Individual PAH and OPAH Exposure Responses
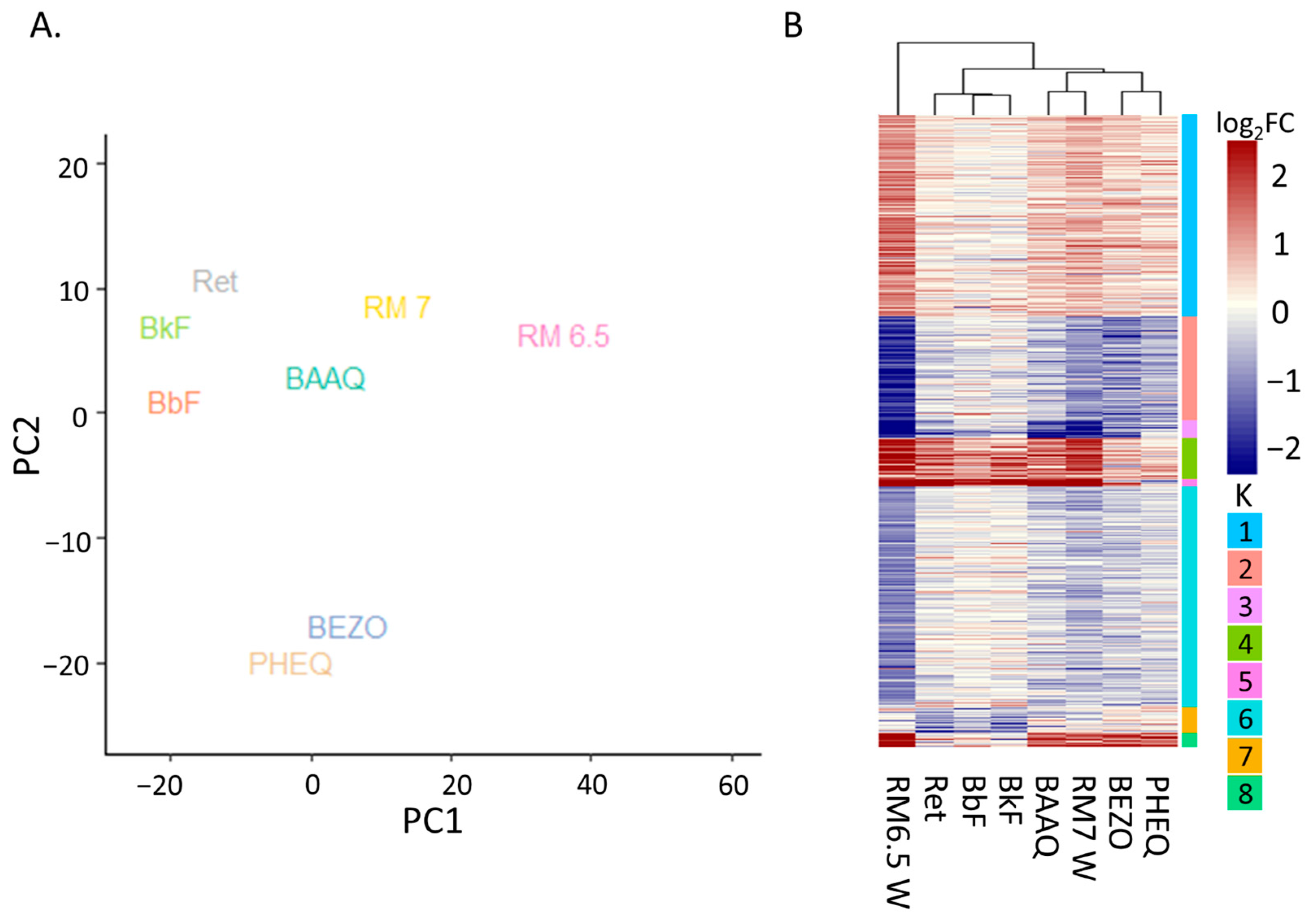
3.4.1. Principal Analysis Indicates AHR as Second Strongest Determinants of Variation
3.4.2. Differential Expression Heatmap Contains AHR Transcription and OPAH Related Clusters Yet PHSS Extract Exposure Remains Distinct
3.4.3. There Is Significant Overlap between Individual Constituent and Mixture GO Terms but Muscle Fiber Related Genes Are without a Match
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- USEPA. Portland Harbor Remedial Investigation Report, Final; USEPA: Washington, DC, USA, 2016. [Google Scholar]
- USEPA. Revised Working Draft Sampling Plan for Pre-Remedial Design, Baseline and Long-Term Monitoring; USEPA: Washington, DC, USA, 2017. [Google Scholar]
- Allan, S.E.; Smith, B.W.; Tanguay, R.L.; Anderson, K.A. Bridging environmental mixtures and toxic effects. Environ. Toxicol. Chem. 2012, 31, 2877–2887. [Google Scholar] [CrossRef]
- Hillwalker, W.E.; Allan, S.E.; Tanguay, R.L.; Anderson, K.A. Exploiting lipid-free tubing passive samplers and embryonic zebrafish to link site specific contaminant mixtures to biological responses. Chemosphere 2010, 79, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Lubin, A.; Otterstrom, J.; Hoade, Y.; Bjedov, I.; Stead, E.; Whelan, M.; Gestri, G.; Paran, Y.; Payne, E. A versatile, automated and high-throughput drug screening platform for zebrafish embryos. Biol. Open 2021, 10, bio058513. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.G.; Lohmann, R.; Fernandez, L.A.; MacFarlane, J.K. Polyethylene Devices: Passive Samplers for Measuring Dissolved Hydrophobic Organic Compounds in Aquatic Environments. Environ. Sci. Technol. 2007, 41, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Carls, M.G.; Holland, L.G.; Short, J.W.; Heintz, R.A.; Rice, S.D. Monitoring polynuclear aromatic hydrocarbons in aqueous environments with passive low-density polyethylene membrane devices. Environ. Toxicol. Chem. 2004, 23, 1416–1424. [Google Scholar] [CrossRef]
- Minick, D.J.; Anderson, K.A. Diffusive flux of PAHs across sediment-water and water-air interfaces at urban superfund sites. Environ. Toxicol. Chem. 2017, 36, 2281–2289. [Google Scholar] [CrossRef] [PubMed]
- Some non-heterocyclic polycyclic aromatic hydrocarbons and some related exposures. IARC Monogr. Eval. Carcinog Risks Hum. 2010, 92, 1–853.
- Boffetta, P.; Jourenkova, N.; Gustavsson, P. Cancer risk from occupational and environmental exposure to polycyclic aromatic hydrocarbons. Cancer Causes Control 1997, 8, 444–472. [Google Scholar] [CrossRef]
- Brette, F.; Shiels, H.A.; Galli, G.L.J.; Cros, C.; Incardona, J.P.; Scholz, N.L.; Block, B.A. A Novel Cardiotoxic Mechanism for a Pervasive Global Pollutant. Sci. Rep. 2017, 7, 41476. [Google Scholar] [CrossRef]
- Das, L.; Patel, B.; Patri, M. Adolescence benzo[a]pyrene treatment induces learning and memory impairment and anxiolytic like behavioral response altering neuronal morphology of hippocampus in adult male Wistar rats. Toxicol. Rep. 2019, 6, 1104–1113. [Google Scholar] [CrossRef]
- Incardona, J.P. Molecular Mechanisms of Crude Oil Developmental Toxicity in Fish. Arch. Environ. Contam Toxicol. 2017, 73, 19–32. [Google Scholar] [CrossRef] [PubMed]
- McCallister, M.M.; Maguire, M.; Ramesh, A.; Aimin, Q.; Liu, S.; Khoshbouei, H.; Aschner, M.; Ebner, F.F.; Hood, D.B. Prenatal exposure to benzo(a)pyrene impairs later-life cortical neuronal function. Neurotoxicology 2008, 29, 846–854. [Google Scholar] [CrossRef] [PubMed]
- Melendez-Colon, V.J.; Luch, A.; Seidel, A.; Baird, W.M. Formation of stable DNA adducts and apurinic sites upon metabolic activation of bay and fjord region polycyclic aromatic hydrocarbons in human cell cultures. Chem. Res. Toxicol. 2000, 13, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Novosad, J.; Fiala, Z.; Borska, L.; Krejsek, J. Immunosuppressive effect of polycyclic aromatic hydrocarbons by induction of apoptosis of pre-B lymphocytes of bone marrow. Acta Med. (Hradec Kralove) 2002, 45, 123–128. [Google Scholar] [CrossRef]
- Denison, M.; Helferich, W. The Ah Receptor Signal Transduction Pathw ay. In Toxicant-Receptor Interactions; CRC Press: Boca Raton, FL, USA, 1998; pp. 15–46. [Google Scholar]
- Miao, W.; Hu, L.; Scrivens, P.J.; Batist, G. Transcriptional regulation of NF-E2 p45-related factor (NRF2) expression by the aryl hydrocarbon receptor-xenobiotic response element signaling pathway: Direct cross-talk between phase I and II drug-metabolizing enzymes. J. Biol. Chem. 2005, 280, 20340–20348. [Google Scholar] [CrossRef] [PubMed]
- Nebert, D.W.; Dalton, T.P.; Okey, A.B.; Gonzalez, F.J. Role of aryl hydrocarbon receptor-mediated induction of the CYP1 enzymes in environmental toxicity and cancer. J. Biol. Chem. 2004, 279, 23847–23850. [Google Scholar] [CrossRef] [PubMed]
- Shankar, P.; Garcia, G.R.; La Du, J.K.; Sullivan, C.M.; Dunham, C.L.; Goodale, B.C.; Waters, K.M.; Stanisheuski, S.; Maier, C.S.; Thunga, P.; et al. The Ahr2-Dependent wfikkn1 Gene Influences Zebrafish Transcriptome, Proteome, and Behavior. Toxicol. Sci. 2022, 187, 325–344. [Google Scholar] [CrossRef]
- Shankar, P.; Geier, M.C.; Truong, L.; McClure, R.S.; Pande, P.; Waters, K.M.; Tanguay, R.L. Coupling Genome-wide Transcriptomics and Developmental Toxicity Profiles in Zebrafish to Characterize Polycyclic Aromatic Hydrocarbon (PAH) Hazard. Int. J. Mol. Sci. 2019, 20, 2570. [Google Scholar] [CrossRef]
- Yeager, R.L.; Reisman, S.A.; Aleksunes, L.M.; Klaassen, C.D. Introducing the “TCDD-inducible AhR-Nrf2 gene battery”. Toxicol. Sci. 2009, 111, 238–246. [Google Scholar] [CrossRef]
- Abdel-Shafy, H.I.; Mansour, M.S. A review on polycyclic aromatic hydrocarbons: Source, environmental impact, effect on human health and remediation. Egypt. J. Pet. 2016, 25, 107–123. [Google Scholar] [CrossRef]
- Sower, G.J.; Anderson, K.A. Spatial and Temporal Variation of Freely Dissolved Polycyclic Aromatic Hydrocarbons in an Urban River Undergoing Superfund Remediation. Environ. Sci. Technol. 2008, 42, 9065–9071. [Google Scholar] [CrossRef]
- Anderson, K.A.; Sethajintanin, D.; Sower, G.; Quarles, L. Field trial and modeling of uptake rates of in situ lipid-free polyethylene membrane passive sampler. Environ. Sci. Technol. 2008, 42, 4486–4493. [Google Scholar] [CrossRef] [PubMed]
- Matzke, M.M.; Allan, S.E.; Anderson, K.A.; Waters, K.M. An approach for calculating a confidence interval from a single aquatic sample for monitoring hydrophobic organic contaminants. Environ. Toxicol. Chem. 2012, 31, 2888–2892. [Google Scholar] [CrossRef] [PubMed]
- Garry Mallard, J.R. Automated Mass Spectral Deconvolution and Identification System; US National Institute of Standards and Technology: Gaithersburg, MD, USA, 1997; p. 57. [Google Scholar]
- Goodale, B.C.; La Du, J.; Tilton, S.C.; Sullivan, C.M.; Bisson, W.H.; Waters, K.M.; Tanguay, R.L. Ligand-Specific Transcriptional Mechanisms Underlie Aryl Hydrocarbon Receptor-Mediated Developmental Toxicity of Oxygenated PAHs. Toxicol. Sci. 2015, 147, 397–411. [Google Scholar] [CrossRef]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data, Version 0.11.9; Babraham Bioinformatics: Cambridge, UK, 2015. [Google Scholar]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Putri, G.H.; Anders, S.; Pyl, P.T.; Pimanda, J.E.; Zanini, F. Analysing high-throughput sequencing data in Python with HTSeq 2.0. Bioinformatics 2022, 38, 2943–2945. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environemnt for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Raudvere, U.; Kolberg, L.; Kuzmin, I.; Arak, T.; Adler, P.; Peterson, H.; Vilo, J. g:Profiler: A web server for functional enrichment analysis and conversions of gene lists (2019 update). Nucleic Acids Res. 2019, 47, W191–W198. [Google Scholar] [CrossRef]
- Harris, M.A.; Clark, J.; Ireland, A.; Lomax, J.; Ashburner, M.; Foulger, R.; Eilbeck, K.; Lewis, S.; Marshall, B.; Mungall, C.; et al. The Gene Ontology (GO) database and informatics resource. Nucleic Acids Res. 2004, 32, D258–D261. [Google Scholar] [CrossRef]
- Merico, D.; Isserlin, R.; Stueker, O.; Emili, A.; Bader, G.D. Enrichment map: A network-based method for gene-set enrichment visualization and interpretation. PLoS ONE 2010, 5, e13984. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]
- Geier, M.C.; Chlebowski, A.C.; Truong, L.; Massey Simonich, S.L.; Anderson, K.A.; Tanguay, R.L. Comparative developmental toxicity of a comprehensive suite of polycyclic aromatic hydrocarbons. Arch. Toxicol. 2018, 92, 571–586. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Siddens, L.K.; Heine, L.K.; Sampson, D.A.; Yu, Z.; Fischer, K.A.; Löhr, C.V.; Tilton, S.C. Comparative mechanisms of PAH toxicity by benzo[a]pyrene and dibenzo[def,p]chrysene in primary human bronchial epithelial cells cultured at air-liquid interface. Toxicol. Appl. Pharmacol. 2019, 379, 114644. [Google Scholar] [CrossRef]
- Hayes, J.D.; Chanas, S.A.; Henderson, C.J.; McMahon, M.; Sun, C.; Moffat, G.J.; Wolf, C.R.; Yamamoto, M. The Nrf2 transcription factor contributes both to the basal expression of glutathione S-transferases in mouse liver and to their induction by the chemopreventive synthetic antioxidants, butylated hydroxyanisole and ethoxyquin. Biochem. Soc. Trans. 2000, 28, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Greiling, T.M.; Clark, J.I. The transparent lens and cornea in the mouse and zebra fish eye. Semin. Cell Dev. Biol. 2008, 19, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Bejjani, B.A.; Xu, L.; Armstrong, D.; Lupski, J.R.; Reneker, L.W. Expression patterns of cytochrome P4501B1 (Cyp1b1) in FVB/N mouse eyes. Exp. Eye Res. 2002, 75, 249–257. [Google Scholar] [CrossRef]
- Doshi, M.; Marcus, C.; Bejjani, B.A.; Edward, D.P. Immunolocalization of CYP1B1 in normal, human, fetal and adult eyes. Exp. Eye Res. 2006, 82, 24–32. [Google Scholar] [CrossRef]
- Williams, A.L.; Bohnsack, B.L. Neural crest derivatives in ocular development: Discerning the eye of the storm. Birth Defects Res. C Embryo Today 2015, 105, 87–95. [Google Scholar] [CrossRef]
- Williams, A.L.; Eason, J.; Chawla, B.; Bohnsack, B.L. Cyp1b1 Regulates Ocular Fissure Closure Through a Retinoic Acid-Independent Pathway. Investig. Ophthalmol. Vis. Sci. 2017, 58, 1084–1097. [Google Scholar] [CrossRef]
- Rottbauer, W.; Wessels, G.; Dahme, T.; Just, S.; Trano, N.; Hassel, D.; Burns, C.G.; Katus, H.A.; Fishman, M.C. Cardiac myosin light chain-2: A novel essential component of thick-myofilament assembly and contractility of the heart. Circ. Res. 2006, 99, 323–331. [Google Scholar] [CrossRef]
- Sheikh, F.; Lyon, R.C.; Chen, J. Getting the skinny on thick filament regulation in cardiac muscle biology and disease. Trends Cardiovasc. Med. 2014, 24, 133–141. [Google Scholar] [CrossRef]
- Korashy, H.M.; El-Kadi, A.O. The role of aryl hydrocarbon receptor in the pathogenesis of cardiovascular diseases. Drug Metab. Rev. 2006, 38, 411–450. [Google Scholar] [CrossRef] [PubMed]
- Rigaud, C.; Eriksson, A.; Rokka, A.; Skaugen, M.; Lihavainen, J.; Keinänen, M.; Lehtivuori, H.; Vehniäinen, E.R. Retene, pyrene and phenanthrene cause distinct molecular-level changes in the cardiac tissue of rainbow trout (Oncorhynchus mykiss) larvae, part 2-Proteomics and metabolomics. Sci. Total Environ. 2020, 746, 141161. [Google Scholar] [CrossRef] [PubMed]
- Geier, M.C.; James Minick, D.; Truong, L.; Tilton, S.; Pande, P.; Anderson, K.A.; Teeguardan, J.; Tanguay, R.L. Systematic developmental neurotoxicity assessment of a representative PAH Superfund mixture using zebrafish. Toxicol. Appl. Pharmacol. 2018, 354, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Truong, L.; Reif, D.M.; St Mary, L.; Geier, M.C.; Truong, H.D.; Tanguay, R.L. Multidimensional in vivo hazard assessment using zebrafish. Toxicol. Sci. 2014, 137, 212–233. [Google Scholar] [CrossRef]
- Haendel, M.A.; Tilton, F.; Bailey, G.S.; Tanguay, R.L. Developmental toxicity of the dithiocarbamate pesticide sodium metam in zebrafish. Toxicol. Sci. 2004, 81, 390–400. [Google Scholar] [CrossRef] [PubMed]
- Tilton, F.; La Du, J.K.; Vue, M.; Alzarban, N.; Tanguay, R.L. Dithiocarbamates have a common toxic effect on zebrafish body axis formation. Toxicol. Appl. Pharmacol. 2006, 216, 55–68. [Google Scholar] [CrossRef]
- Lundstedt, S.; White, P.A.; Lemieux, C.L.; Lynes, K.D.; Lambert, I.B.; Oberg, L.; Haglund, P.; Tysklind, M. Sources, fate, and toxic hazards of oxygenated polycyclic aromatic hydrocarbons (PAHs) at PAH-contaminated sites. Ambio 2007, 36, 475–485. [Google Scholar] [CrossRef]
- Wassenberg, D.M.; Di Giulio, R.T. Synergistic embryotoxicity of polycyclic aromatic hydrocarbon aryl hydrocarbon receptor agonists with cytochrome P4501A inhibitors in Fundulus heteroclitus. Environ. Health Perspect. 2004, 112, 1658–1664. [Google Scholar] [CrossRef]
- Misaki, K.; Matsui, S.; Matsuda, T. Metabolic enzyme induction by HepG2 cells exposed to oxygenated and nonoxygenated polycyclic aromatic hydrocarbons. Chem. Res. Toxicol. 2007, 20, 277–283. [Google Scholar] [CrossRef]
- Knecht, A.L.; Goodale, B.C.; Truong, L.; Simonich, M.T.; Swanson, A.J.; Matzke, M.M.; Anderson, K.A.; Waters, K.M.; Tanguay, R.L. Comparative developmental toxicity of environmentally relevant oxygenated PAHs. Toxicol. Appl. Pharmacol. 2013, 271, 266–275. [Google Scholar] [CrossRef] [PubMed]



| Detection | ||
|---|---|---|
| Chemical | RM 7W | RM 6.5W |
| o,p’-DDD | X | X |
| p,p’-DDE | X | X |
| Hexachlorobenzene | X | X |
| PCB65 | X | |
| PCB118X | X | |
| Tonalide | X | |
| Benzoflourenone | X | |
| benzanthrone | X | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rude, C.I.; Tidwell, L.G.; Tilton, S.C.; Waters, K.M.; Anderson, K.A.; Tanguay, R.L. Coupling Environmental Whole Mixture Toxicity Screening with Unbiased RNA-Seq Reveals Site-Specific Biological Responses in Zebrafish. Toxics 2023, 11, 201. https://doi.org/10.3390/toxics11030201
Rude CI, Tidwell LG, Tilton SC, Waters KM, Anderson KA, Tanguay RL. Coupling Environmental Whole Mixture Toxicity Screening with Unbiased RNA-Seq Reveals Site-Specific Biological Responses in Zebrafish. Toxics. 2023; 11(3):201. https://doi.org/10.3390/toxics11030201
Chicago/Turabian StyleRude, Christian I., Lane G. Tidwell, Susan C. Tilton, Katrina M. Waters, Kim A. Anderson, and Robyn L. Tanguay. 2023. "Coupling Environmental Whole Mixture Toxicity Screening with Unbiased RNA-Seq Reveals Site-Specific Biological Responses in Zebrafish" Toxics 11, no. 3: 201. https://doi.org/10.3390/toxics11030201
APA StyleRude, C. I., Tidwell, L. G., Tilton, S. C., Waters, K. M., Anderson, K. A., & Tanguay, R. L. (2023). Coupling Environmental Whole Mixture Toxicity Screening with Unbiased RNA-Seq Reveals Site-Specific Biological Responses in Zebrafish. Toxics, 11(3), 201. https://doi.org/10.3390/toxics11030201









