Concentration of Immunoglobulins in Microfiltration Permeates of Skim Milk: Impact of Transmembrane Pressure and Temperature on the IgG Transmission Using Different Ceramic Membrane Types and Pore Sizes
Abstract
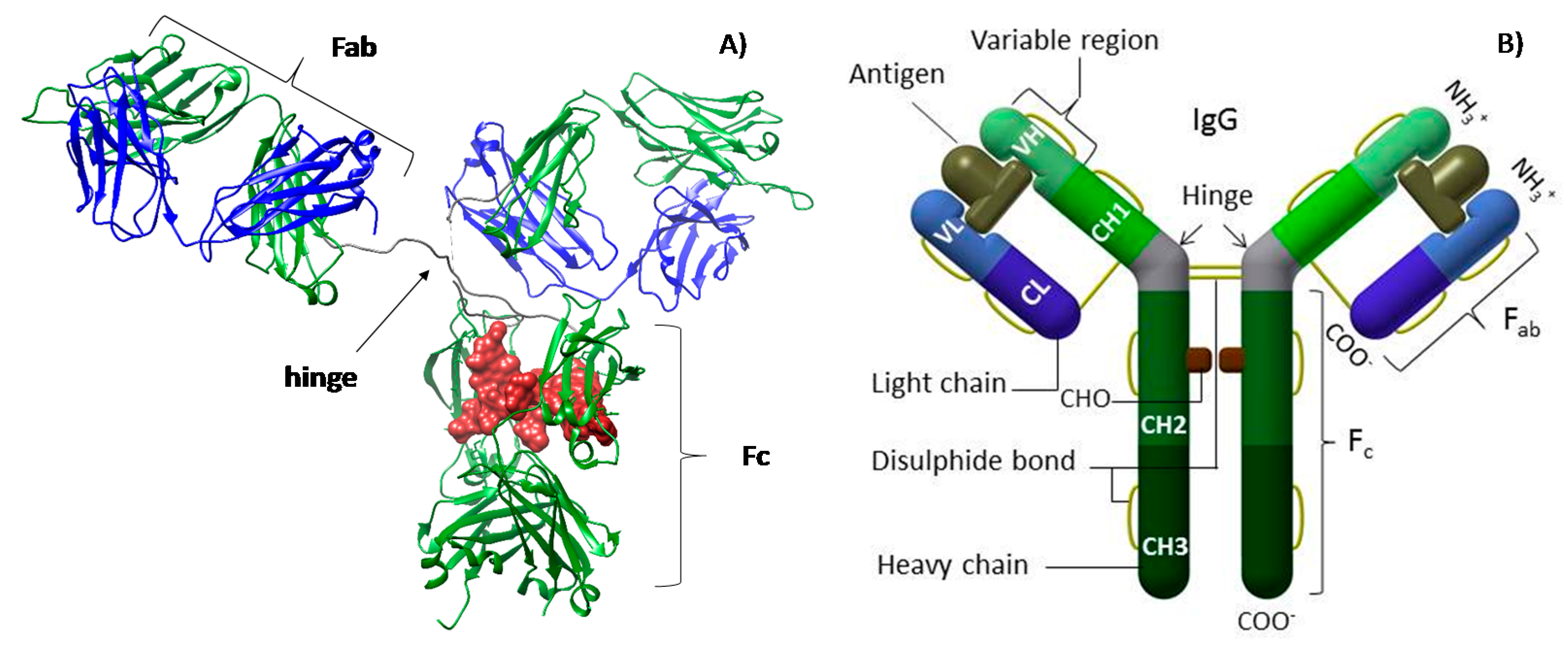
:1. Introduction
2. Materials and Methods
2.1. Microfiltration of Skim Milk
2.2. Operating Conditions during Microfiltration
2.3. Measurement of Immunoglobulin G with Reversed-Phase High-Performance Liquid Chromatography
2.4. Measurement of Casein with RP-HPLC
3. Results and Discussion
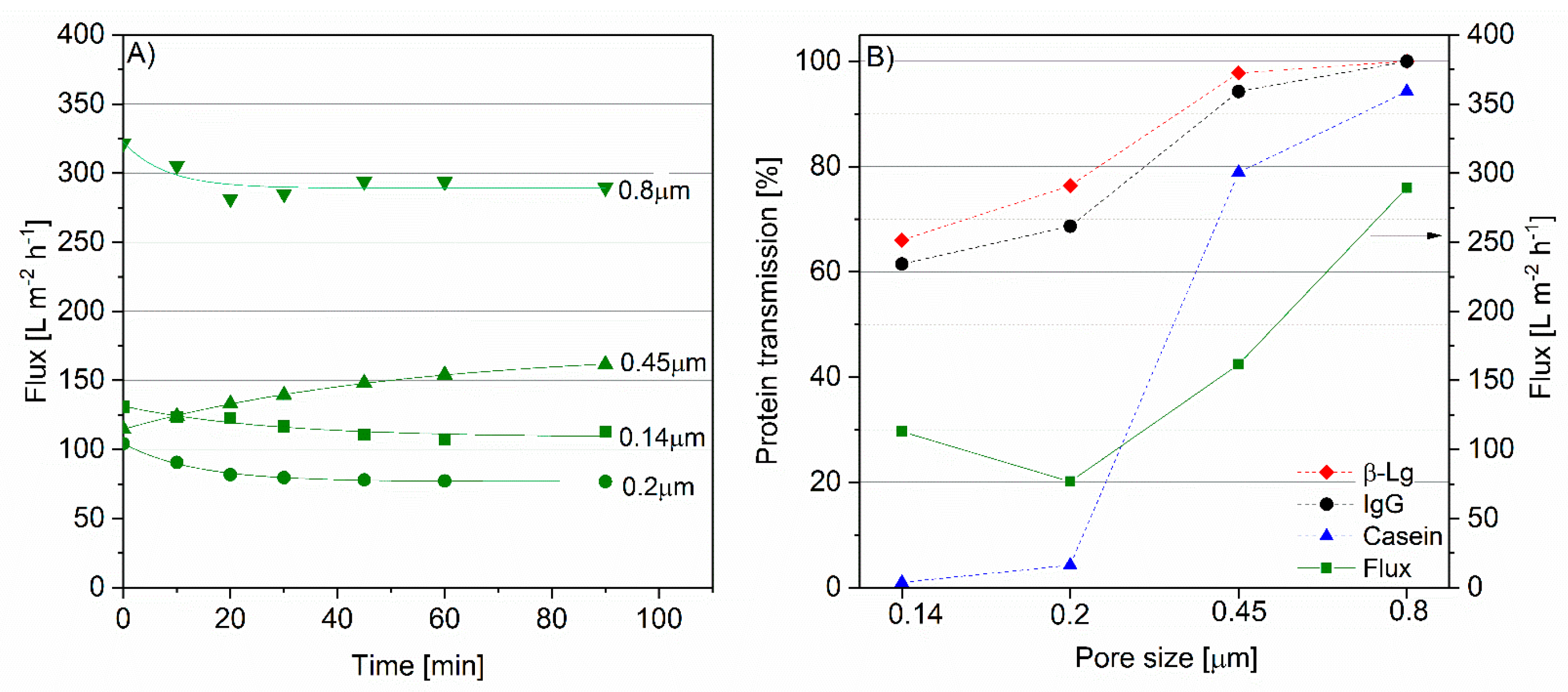
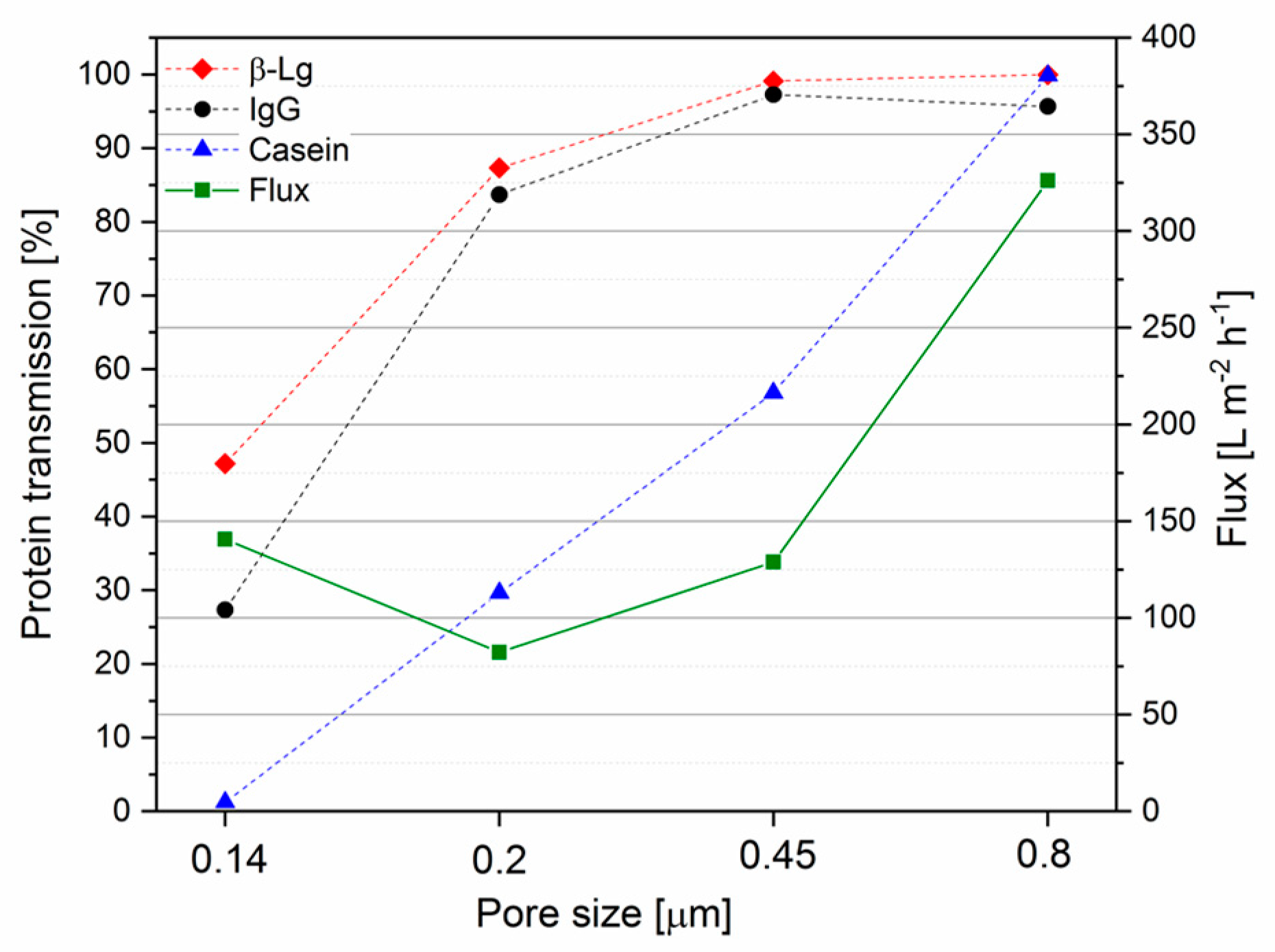
3.1. IgG Transmission as a Function of Time and Pore Size
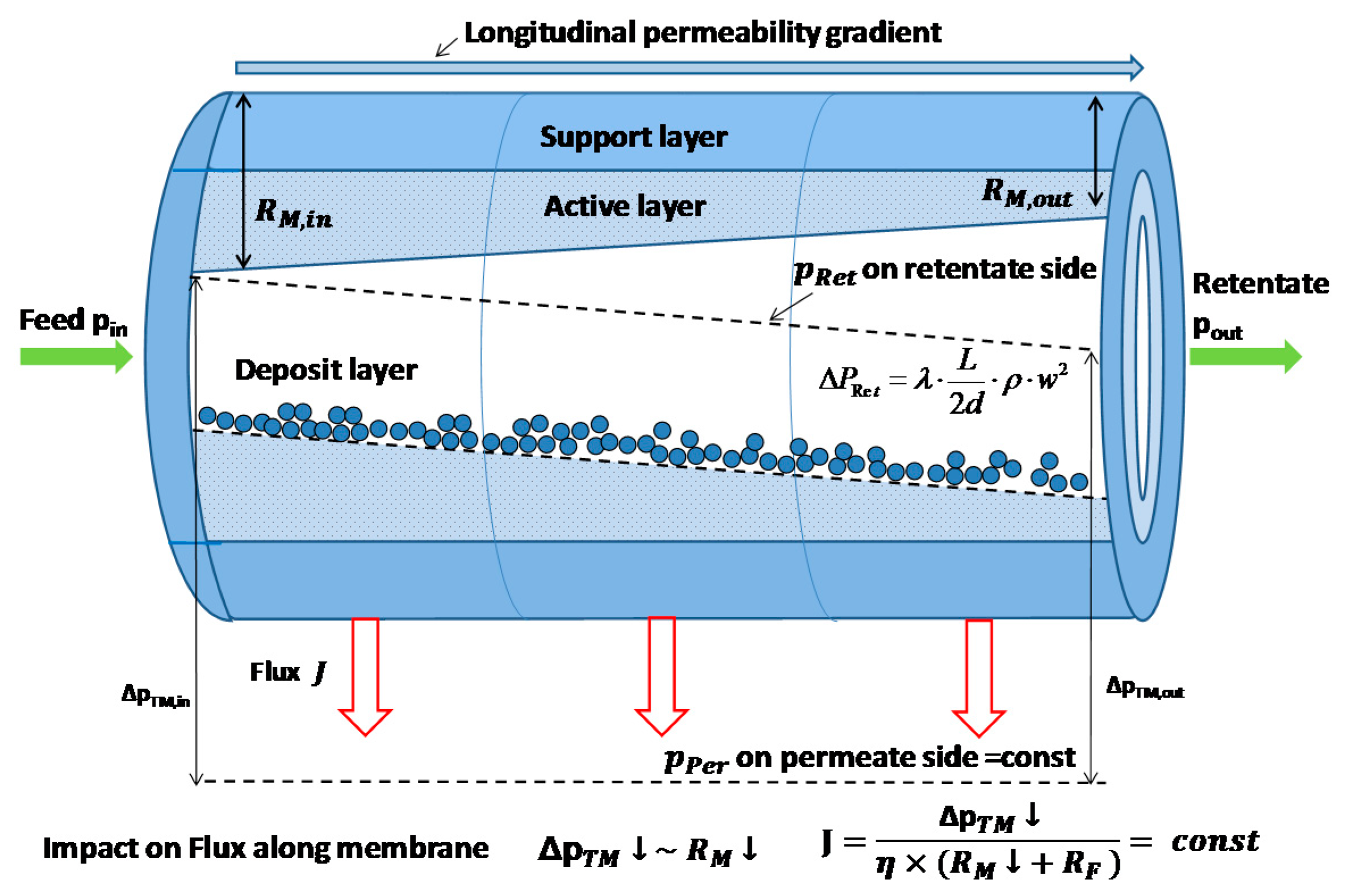
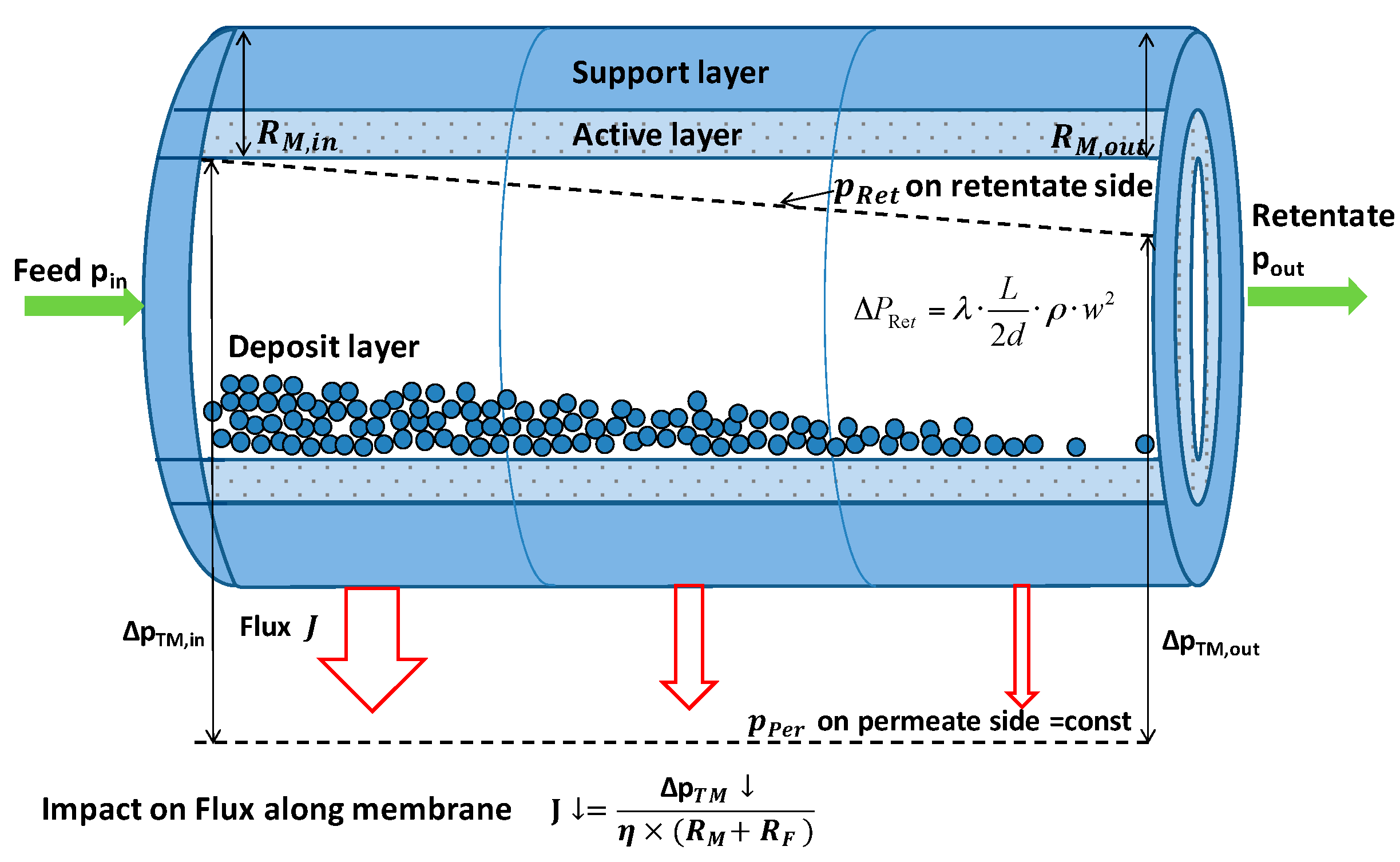
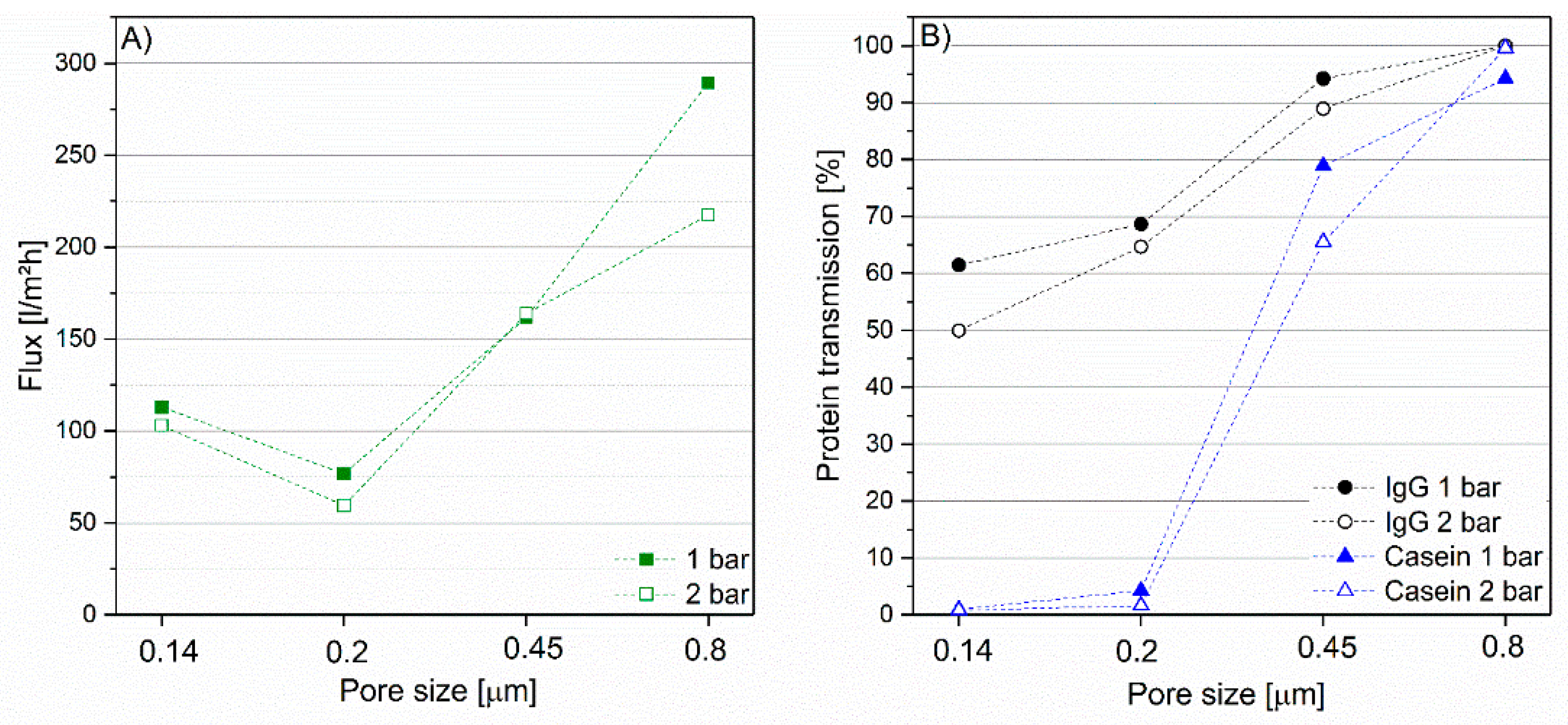
3.2. Impact of Membrane Type on IgG Transmission and Flux
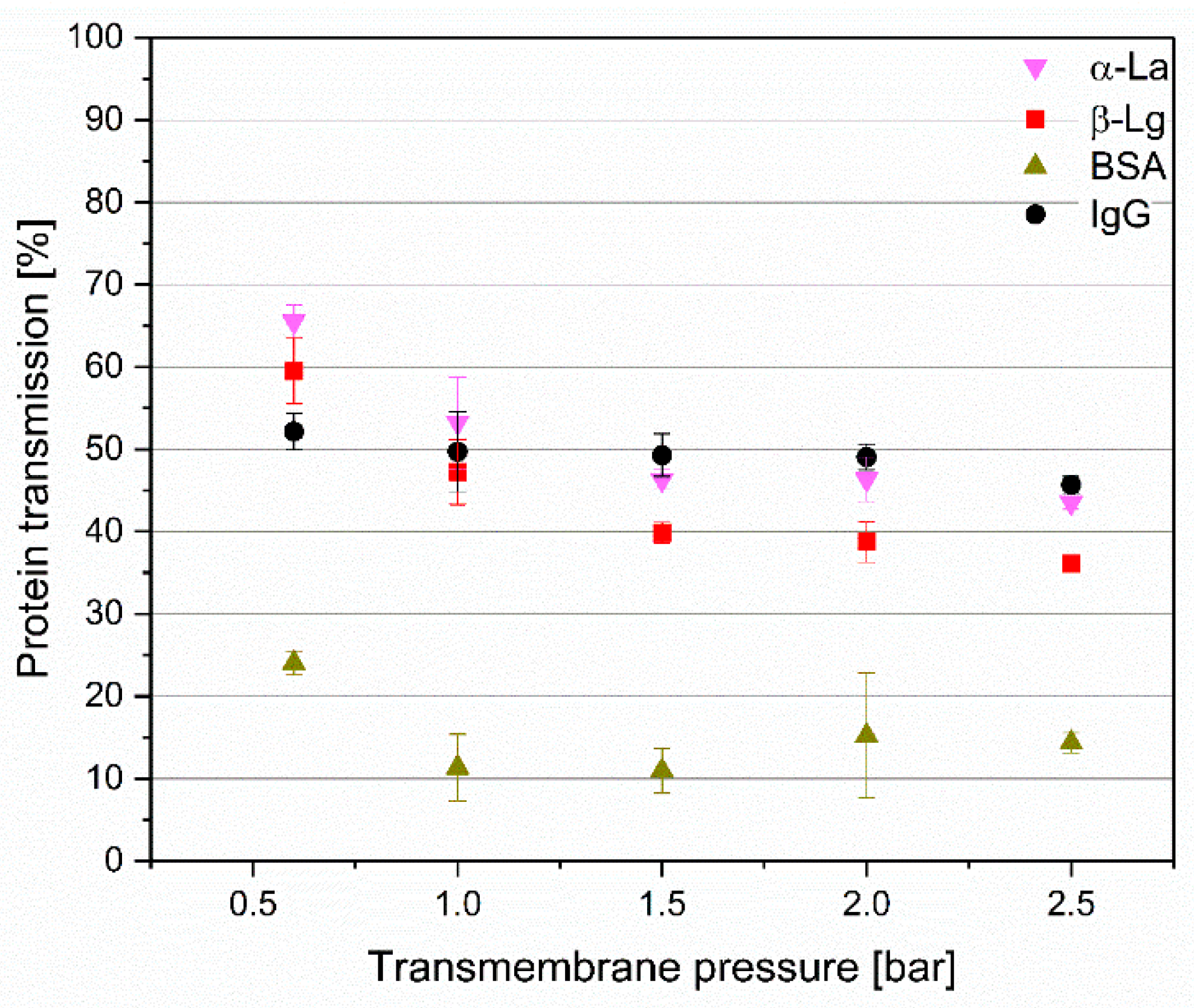
3.3. Impact of Transmembrane Pressure on IgG Transmissionand Flux at Different Pore Sizes
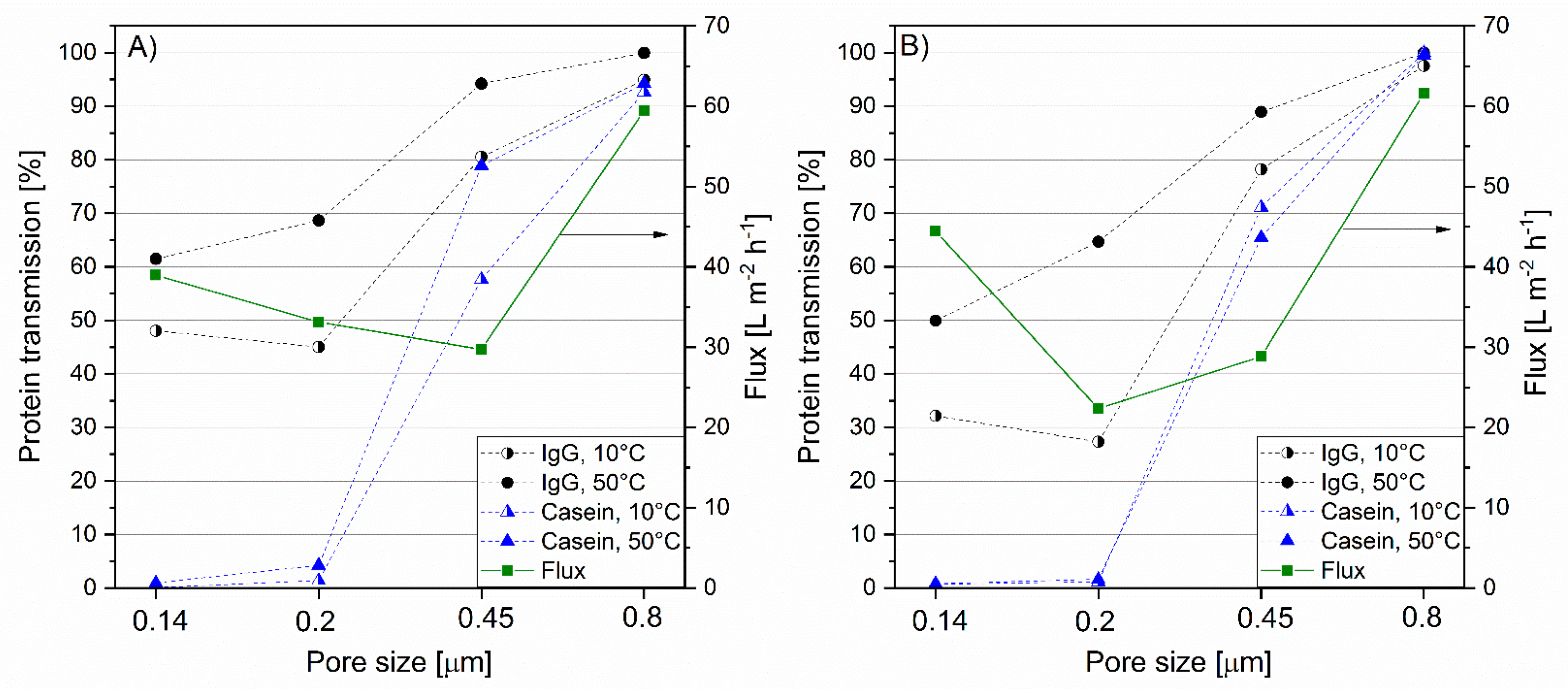
3.4. Impact of Temperature on IgG Transmission and Flux
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Korhonen, H.; Marnila, P.; Gill, H.S. Bovine milk antibodies for health. Br. J. Nutr. 2000, 84, 135–146. [Google Scholar] [CrossRef]
- Steele, J.; Sponseller, J.; Schmidt, D.; Cohen, O.; Tzipori, S. Hyperimmune bovine colostrum for treatment of GI infections: A review and update on Clostridium difficile. Hum. Vanccines Immunother. 2013, 9, 1565–1568. [Google Scholar] [CrossRef] [PubMed]
- Marnila, P.; Korhonen, H. Encyclopedia of Dairy Sciences, 2nd ed.; Fuquay, J.W., Ed.; Academic Press: San Diego, CA, USA, 2011; pp. 807–815. [Google Scholar]
- Van Dissel, J.T.; de Groot, N.; Hensgens, C.M.; Numan, S.; Kuijper, E.J.; Veldkamp, P.; van’t Wout, J. Bovine antibody-enriched whey to aid in the prevention of a relapse of Clostridium difficile-associated diarrhoea: Preclinical and preliminary clinical data. J. Med. Microbiol. 2005, 54, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Young, K.W.H.; Munro, I.C.; Taylor, S.L.; Veldkamp, P.; van Dissel, J.T. The safety of whey protein concentrate derived from the milk of cows immunized against Clostridium difficile. Regul. Toxicol. Pharmacol. 2007, 47, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Numan, S.C.; Veldkamp, P.; Kuijper, E.J.; van den Berg, R.J.; van Dissel, J.T. Clostridium difficile-associated diarrhoea: Bovine anti-Clostridium difficile whey protein to help aid the prevention of relapses. Gut 2007, 56, 888–889. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Chang, H. Effect of Thermal Protectants on the Stability of Bovine Milk Immunoglobulin G. J. Agric. Food Chem. 1998, 46, 3570–3576. [Google Scholar] [CrossRef]
- Mainer, G.; Sanchez, L.; Ena, J.M.; Calvo, M. Kinetic and Thermodynamic Parameters for Heat Denaturation of Bovine Milk IgG, IgA and IgM. J. Food Sci. 1997, 62, 1034–1038. [Google Scholar] [CrossRef]
- Saboyainsta, L.V.; Maubois, J.-L. Current developments of microfiltration technology in the dairy industry. Le Lait 2000, 80, 541–553. [Google Scholar] [CrossRef] [Green Version]
- Gosch, T.; Apprich, S.; Kneifel, W.; Novalin, S. Improved isolation of bioactive components of bovine colostrum using cross-flow microfiltration. Int. J. Dairy Technol. 2013, 66, 175–181. [Google Scholar] [CrossRef]
- Le Berre, O.; Daufin, G. Skimmilk crossflow microfiltration performance versus permeation flux to wall shear stress ratio. J. Membr. Sci. 1996, 117, 261–270. [Google Scholar] [CrossRef]
- Kulozik, U.; Kersten, M. New Ways for the Fractionation of Dairy and Minor Constituents Using UTP-Membrane Technology. Bull. Int. Dairy Fed. 2002, 374, 37–42. [Google Scholar]
- Jimenez-Lopez, A.J.E.; Leconte, N.; Dehainault, O.; Geneste, C.; Fromont, L.; Gésan-Guiziou, G. Role of milk constituents on critical conditions and deposit structure in skimmilk microfiltration (0.1 μm). Sep. Purif. Technol. 2008, 61, 33–43. [Google Scholar] [CrossRef]
- Jimenez-Lopez, A.J.E.; Leconte, N.; Garnier-Lambrouin, F.; Bouchoux, A.; Rousseau, F.; Gésan-Guiziou, G. Ionic strength dependence of skimmed milk microfiltration: Relations between filtration performance, deposit layer characteristics and colloidal properties of casein micelles. J. Membr. Sci. 2011, 369, 404–413. [Google Scholar] [CrossRef]
- Piry, A.; Kühnl, W.; Tolkach, A.; Ripperger, S.; Kulozik, U. Length dependency of flux and protein permeation in crossflow microfiltration of skimmed milk. J. Membr. Sci. 2008, 325, 887–894. [Google Scholar] [CrossRef]
- Piry, A.; Heino, A.; Kühnl, W.; Grein, T.; Ripperger, S.; Kulozik, U. Effect of membrane length, membrane resistance, and filtration conditions on the fractionation of milk proteins by microfiltration. J. Dairy Sci. 2012, 95, 1590–1602. [Google Scholar] [CrossRef] [PubMed]
- Kühnl, W.; Piry, A.; Kaufmann, V.; Grein, T.; Ripperger, S.; Kulozik, U. Impact of colloidal interactions on the flux in cross-flow microfiltration of milk at different pH values: A surface energy approach. J. Membr. Sci. 2010, 352, 107–115. [Google Scholar] [CrossRef]
- Steinhauer, T.; Kühnl, W.; Kulozik, U. Impact of protein interactions and transmembrane pressure on physical properties of filter cakes formed during filtrations of skim milk. Procedia Food Sci. 2011, 1, 886–892. [Google Scholar] [CrossRef]
- Steinhauer, T.; Lonfat, J.; Hager, I.; Gebhardt, R.; Kulozik, U. Effect of pH, transmembrane pressure and whey proteins on the properties of casein micelle deposit layers. J. Membr. Sci. 2015, 493, 452–459. [Google Scholar] [CrossRef]
- Adams, M.C.; Barbano, D.M. Serum protein removal from skim milk with a 3-stage, 3× ceramic Isoflux membrane process at 50 °C. J. Dairy Sci. 2013, 96, 2020–2034. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, C.E.; Abrahamsen, R.K.; Rukke, E.-O.; Johansen, A.-G.; Schüller, R.B.; Skeie, S.B. Optimization of protein fractionation by skim milk microfiltration: Choice of ceramic membrane pore size and filtration temperature. J. Dairy Sci. 2016, 99, 6164–6179. [Google Scholar] [CrossRef] [PubMed]
- Piot, M.; Fauquant, J.; Madec, M.N.; Maubois, J.L. Preparation of serocolostrum by membrane microfiltration. Le Lait 2004, 84, 333–341. [Google Scholar] [CrossRef] [Green Version]
- Le Berre, O.; Daufin, G. Microfiltration (0·1 μm) of milk: Effect of protein size and charge. J. Dairy Res. 1998, 65, 443–455. [Google Scholar] [CrossRef]
- Kessler, H.G.; Beyer, H.J. Thermal denaturation of whey proteins and its effect in dairy technology. Int. J. Biol. Macromol. 1991, 13, 165–173. [Google Scholar] [CrossRef]
- Toro-Sierra, J.; Tolkach, A.; Kulozik, U. Fractionation of a-Lactalbumin and ß-Lactoglobulin from Whey Protein Isolate Using Selective Thermal Aggregation, an Optimized Membrane Separation Procedure and Resolubilization Techniques at Pilot Plant Scale. Food Bioprocess Technol. 2013, 6, 1032–1043. [Google Scholar] [CrossRef]
- Bonfatti, V.; Grigoletto, L.; Cecchinato, A.; Gallo, L.; Carnier, P. Validation of a new reversed-phase high-performance liquid chromatography method for separation and quantification of bovine milk protein genetic variants. J. Chromatogr. A 2008, 1195, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Bonizzi, I.; Buffoni, J.N.; Feligini, M. Quantification of bovine casein fractions by direct chromatographic analysis of milk. Approaching the application to a real production context. J. Chromatogr. A 2009, 1216, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, H.W.; Cavacini, L. Structure and function of immunoglobulins. J. Allergy Clin. Immunol. 2010, 125, S41–S52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gapper, L.W.; Copestake, D.E.J.; Otter, D.E.; Indyk, H.E. Analysis of bovine immunoglobulin G in milk, colostrum and dietary supplements: A review. Anal. Bioanal. Chem. 2007, 389, 93–109. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Saphire, E.O.; Parren, P.W.; Pantophlet, R.; Zwick, M.B.; Morris, G.M.; Rudd, P.M.; Dwek, R.A.; Stanfield, R.L.; Burton, D.R.; Wilson, I.A. Crystal structure of a neutralizing human IGG against HIV-1: A template for vaccine design. Science 2001, 293, 1155–1159. [Google Scholar] [CrossRef] [PubMed]
- Piry, A. Untersuchung zur Längenanhängigkeit der Filtrationsleistung bei der Fraktionierung von Milchproteinen Mittels Mikrofiltration. Ph.D. Thesis, Technische Universität München, München, Germany, 2011. [Google Scholar]
- Saxena, A.; Tripathi, B.P.; Kumar, M.; Shahi, V.K. Membrane-based techniques for the separation and purification of proteins: An overview. Adv. Colloid Interf. Sci. 2009, 145, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Brans, G.; Schroën, C.G.P.H.; van der Sman, R.G.M.; Boom, R.M. Membrane fractionation of milk: State of the art and challenges. J. Membr. Sci. 2004, 243, 263–272. [Google Scholar] [CrossRef]
- Skrzypek, M.; Burger, M. Isoflux® ceramic membranes—Practical experiences in dairy industry. Conf. Fouling Clin. Flux Theor. Appl. 2010, 250, 1095–1100. [Google Scholar] [CrossRef]
- Kulozik, U.; Kersten, M. Fraktionieren von Proteinen mittels Mikrofiltration. CIT 2001, 73, 1622–1625. [Google Scholar] [CrossRef]
- Samuelsson, G.; Dejmek, P.; Trägårdh, G.; Paulsson, M. Minimizing whey protein retention in cross-flow microfiltration of skim milk. Int. Dairy J. 1997, 7, 237–242. [Google Scholar] [CrossRef]
- Espina, V.S.; Jaffrin, M.Y.; Ding, L.H. Comparison of rotating ceramic membranes and polymeric membranes in fractionation of milk proteins by microfiltration. Desalination 2009, 245, 714–722. [Google Scholar] [CrossRef]
- Farrell, H.M.; Jimenez-Flores, R.; Bleck, G.T.; Brown, E.M.; Butler, J.E.; Creamer, L.K.; Hicks, C.L.; Hollar, C.M.; Ng-Kwai-Hang, K.F.; Swaisgood, H.E. Nomenclature of the Proteins of Cows’ Milk—Sixth Revision. J. Dairy Sci. 2004, 87, 1641–1674. [Google Scholar] [CrossRef]
- Coppola, L.E.; Molitor, M.S.; Rankin, S.A.; Lucey, J.A. Comparison of milk-derived whey protein concentrates containing various levels of casein. Int. J. Dairy Technol. 2014, 67, 467–473. [Google Scholar] [CrossRef]
- Hurt, E.E.; Adams, M.C.; Barbano, D.M. Microfiltration of skim milk and modified skim milk using a 0.1-µm ceramic uniform transmembrane pressure system at temperatures of 50, 55, 60, and 65 °C. J. Dairy Sci. 2015, 98, 765–780. [Google Scholar] [CrossRef] [PubMed]
- Dalgleish, D.G.; Corredig, M. The Structure of the Casein Micelle of Milk and Its Changes during Processing. Annu. Rev. Food Sci. Technol. 2012, 3, 449–467. [Google Scholar] [CrossRef] [PubMed]
- Huppertz, T.; Hennebel, J.-B.; Considine, T.; Shakeel-Ur-Rehman; Kelly, A.L.; Fox, P.F. A method for the large-scale isolation of β-casein. Food Chem. 2006, 99, 45–50. [Google Scholar] [CrossRef]
- Kessler, H.G. Lebensmittel-und Bioverfahrenstechnik. Molkereitechnologie; Technische Universität München-Weihnstephan: München, Germany, 1996. [Google Scholar]
- Kersten, M.; Kulozik, U. Proteinfraktionierung mittels Membrantrennverfahren; VDI-Verlag: Munich, Germany, 2000. [Google Scholar]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heidebrecht, H.-J.; Toro-Sierra, J.; Kulozik, U. Concentration of Immunoglobulins in Microfiltration Permeates of Skim Milk: Impact of Transmembrane Pressure and Temperature on the IgG Transmission Using Different Ceramic Membrane Types and Pore Sizes. Foods 2018, 7, 101. https://doi.org/10.3390/foods7070101
Heidebrecht H-J, Toro-Sierra J, Kulozik U. Concentration of Immunoglobulins in Microfiltration Permeates of Skim Milk: Impact of Transmembrane Pressure and Temperature on the IgG Transmission Using Different Ceramic Membrane Types and Pore Sizes. Foods. 2018; 7(7):101. https://doi.org/10.3390/foods7070101
Chicago/Turabian StyleHeidebrecht, Hans-Jürgen, José Toro-Sierra, and Ulrich Kulozik. 2018. "Concentration of Immunoglobulins in Microfiltration Permeates of Skim Milk: Impact of Transmembrane Pressure and Temperature on the IgG Transmission Using Different Ceramic Membrane Types and Pore Sizes" Foods 7, no. 7: 101. https://doi.org/10.3390/foods7070101



