Correlations between Polyacetylene Concentrations in Carrot (Daucus carota L.) and Various Soil Parameters
Abstract
:1. Introduction
2. Materials and Methods
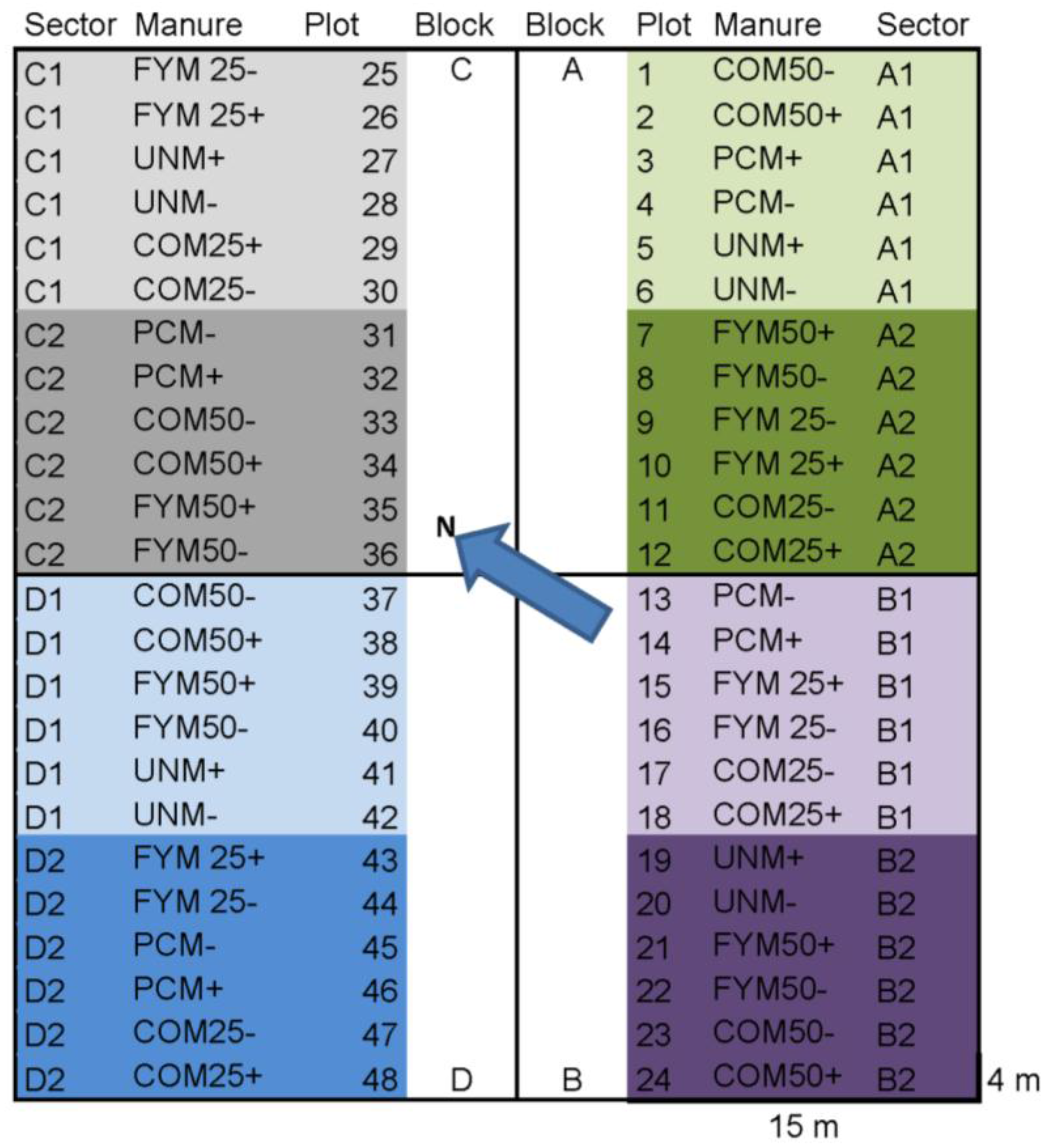
2.1. Sampling and Post-Harvest Treatment
2.2. Analysis
2.2.1. Polyacetylenes in carrots
2.2.2. Soluble Sugars in carrots
2.2.3. Soil
2.3. Treatment of Data
3. Results
3.1. Soil Conditions within the Field Trial
3.2. Soil Conditions, Polyacetylenes, Soluble Sugars and Carrot Size
3.3. Correlations between Soil Parameters
3.4. Soil Parameters and Polyacetylenes
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| FaDOH | falcarindiol |
| FaDOAc | falcarindiol-3-acetate |
| FaOH | falcarinol |
| FaTP | falcarinol-type polyacetylenes |
| P (Al) | available phosphorus |
| K (Al) | available potassium |
| Mg (Al) | available magnesium |
| Ca (Al) | available calcium |
| K (HCl) | total potassium |
| P (HCl) | total phosphorus |
| Cu (HCl) | total copper |
| PCA | principal component analysis. |
References
- Zidorn, C.; Jöhrer, K.; Ganzera, M.; Schubert, B.; Sigmund, E.M.; Mader, J.; Greil, R.; Ellmerer, E.P.; Stuppner, H. Polyacetylenes from the Apiaceae vegetables carrot, celery, fennel, parsley, and parsnip and their cytotoxic activities. J. Agric. Food Chem. 2005, 53, 2518–2523. [Google Scholar] [CrossRef] [PubMed]
- Christensen, L.P.; Brandt, K. Bioactive polyacetylenes in food plants of the Apiaceae family: Occurrence, bioactivity and analysis. J. Pharm. Biomed. Anal. 2006, 41, 683–693. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, H.; Katano, M.; Yamatoto, H.; Fujito, H.; Mori, M.; Takata, K. Cytotoxic activity of polyacetylene compounds in Panax ginseng C.A. Meyer. Chem. Pharm. Bull. 1990, 38, 2480–2482. [Google Scholar] [CrossRef]
- Hansen, S.L.; Purup, S.; Christensen, L.P. Bioactivity of falcarinol and the influence of processing and storage on its content in carrots (Daucus carota L.). J. Sci. Food Agric. 2003, 83, 1010–1017. [Google Scholar] [CrossRef]
- Czepa, A.; Hofmann, T. Structural and sensory characterization of compounds contributing to the bitter off-taste of carrots (Daucus carota L.) and carrot puree. J. Agric. Food Chem. 2003, 51, 3865–3873. [Google Scholar] [CrossRef] [PubMed]
- Garrod, B.; Lea, P.; Lewis, B.G. Studies on the mechanism of action of the antifungal compound falcarindiol. New Phytol. 1979, 83, 463–471. [Google Scholar] [CrossRef]
- Zaini, R.G.; Brandt, K.; Clench, M.R.; Le Maitre, C.L. Effects of bioactive compounds from carrots (Daucus carota L.), polyacetylenes, beta-carotene and lutein on human lymphoid leukaemia cells. Anti-Cancer Agent Med. Chem. 2012, 12, 640–652. [Google Scholar] [CrossRef]
- Schmiech, L.; Alayrac, C.; Witulski, B.; Hofmann, T. Structure determination of bisacetylenic oxylipins in carrots (Daucus carota L.) and enantioselective synthesis of falcarindiol. J. Agric. Food Chem. 2009, 57, 11030–11040. [Google Scholar] [CrossRef] [PubMed]
- Minto, R.E.; Blacklock, B.J. Biosynthesis and function of polyacetylenes and allied products. Prog. Lipid Res. 2008, 47, 233–306. [Google Scholar] [CrossRef] [PubMed]
- Czepa, A.; Hofmann, T. Quantitative studies and sensory analyses on the influence of cultivar, spatial tissue distribution, and industrial processing on the bitter off-taste of carrots (Daucus carota L.) and carrot products. J. Agric. Food Chem. 2004, 52, 4508–4514. [Google Scholar] [CrossRef] [PubMed]
- Baranska, M.; Schulz, H. Spatial tissue distribution of polyacetylenes in carrot root. Analyst 2005, 130, 855–859. [Google Scholar] [CrossRef] [PubMed]
- Garrod, B.; Lewis, B.G. Location of the antifungal compound falcarindiol in carrot root tissue. Trans. Br. Mycol. Soc. 1979, 72, 515–517. [Google Scholar] [CrossRef]
- Dawid, C.; Dunemann, F.; Schwab, W.; Nothnagel, T.; Hofmann, T. Bioactive C17-polyacetylenes in carrots (Daucus carota L.): Current knowledge and future perspectives. J. Agric. Food Chem. 2015, 63, 9211–9222. [Google Scholar] [CrossRef] [PubMed]
- Kidmose, U.; Hansen, S.L.; Christensen, L.P.; Edelenbos, M.; Larsen, E.; Norbaek, R. Effects of genotype, root size, storage, and processing on bioactive compounds in organically grown carrots (Daucus carota L.). J. Food Sci. 2004, 69, S388–S394. [Google Scholar] [CrossRef]
- Pferschy-Wenzig, E.M.; Getzinger, V.; Kunert, O.; Woelkart, K.; Zahrl, J.; Bauer, R. Determination of falcarinol in carrot (Daucus carota L.) genotypes using liquid chromatography/mass spectrometry. Food Chem. 2009, 114, 1083–1090. [Google Scholar] [CrossRef]
- Lund, E.D.; White, J.M. Polyacetylenes in normal and water-stressed ‘Orlando gold’ carrots (Daucus carota). J. Sci. Food Agric. 1990, 51, 507–516. [Google Scholar] [CrossRef]
- Garrod, B.; Lewis, B.G. Effect of falcarindiol on hyphal growth of Mycocentrospora acerina. Trans. Br. Mycol. Soc. 1982, 78, 533–536. [Google Scholar] [CrossRef]
- Lewis, B.G.; Davies, W.P.; Garrod, B. Wound-healing in carrot roots in relation to infection by Mycocentrospora acerina. Ann. Appl. Biol. 1981, 99, 35–42. [Google Scholar] [CrossRef]
- Louarn, S.; Nawrocki, A.; Edelenbos, M.; Jensen, D.F.; Jensen, O.N.; Collinge, D.B.; Jensen, B. The influence of the fungal pathogen Mycocentrospora acerina on the proteome and polyacetylenes and 6-methoxymellein in organic and conventionally cultivated carrots (Daucus carota) during post harvest storage. J. Proteom. 2012, 75, 962–977. [Google Scholar] [CrossRef] [PubMed]
- Olsson, K.; Svensson, R. The influence of polyacetylenes on the susceptibility of carrots to storage diseases. J. Phytopathol. 1996, 144, 441–447. [Google Scholar] [CrossRef]
- Aguiló-Aguayo, I.; Brunton, N.; Rai, D.K.; Balagueró, E.; Hossain, M.B.; Valverde, J. Polyacetylene levels in carrot juice, effect of pH and thermal processing. Food Chem. 2014, 152, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Rawson, A.; Brunton, N.P.; Rai, D.K.; McLoughlin, P.; Tiwari, B.K.; Tuohy, M.G. Stability of falcarinol type polyacetylenes during processing of Apiaceae vegetables. Trends Food Sci. Tech. 2013, 30, 133–141. [Google Scholar] [CrossRef]
- Seljåsen, R.; Kristensen, H.L.; Lauridsen, C.; Wyss, G.S.; Kretzschmar, U.; Birlouez-Aragone, I.; Kahl, J. Quality of carrots as affected by pre- and postharvest factors and processing. J. Sci. Food Agric. 2013, 93, 2611–2626. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, U.; Cummins, E. Factors influencing levels of phytochemicals in selected fruit and vegetables during pre- and post-harvest food processing operations. Food Res. Int. 2013, 50, 497–506. [Google Scholar] [CrossRef]
- Ubbor, S.C.; Uzowuru, O. Effect of processing methods on the phytochemical content of selected food materials. J. Sustain. Agric. Environ. 2013, 14, 133–141. [Google Scholar]
- Zhang, Y.; Liu, X.C.; Wang, Y.; Zhao, F.; Sun, Z.; Liao, X. Quality comparison of carrot juices processed by high-pressure processing and high-temperature short-time processing. Innov. Food Sci. Emerg. Technol. 2016, 33, 135–144. [Google Scholar] [CrossRef]
- Kjellenberg, L.; Johansson, E.; Gustavsson, K.E.; Olsson, M.E. Effects of harvesting date and storage on the amounts of polyacetylenes in carrots, Daucus carota. J. Agric. Food Chem. 2010, 58, 11703–11708. [Google Scholar] [CrossRef] [PubMed]
- Kjellenberg, L.; Johansson, E.; Gustavsson, K.E.; Olsson, M.E. Polyacetylenes in fresh and stored carrots (Daucus carota): Relations to root morphology and sugar content. J. Sci. Food Agric. 2012, 92, 1748–1754. [Google Scholar] [CrossRef] [PubMed]
- Kjellenberg, L.; Johansson, E.; Gustavsson, K.-E.; Granstedt, A.; Olsson, M.E. Influence of organic manures on carrot (Daucus carota L.) crops grown in a long-term field experiment in Sweden. Renew. Agric. Food Syst. 2016, 31, 258–268. [Google Scholar] [CrossRef]
- Rosenfeld, H.J.; Samuelsen, R.T. The effect of soil-relationships and temperature on sensory and chemical quality parameters of carrots (Daucus carota L.). Acta Hortic. 2000, 514, 123–132. [Google Scholar] [CrossRef]
- Simon, P.W.; Peterson, C.E.; Lindsay, R.C. Genotype, soil, and climate effects on sensory and objective components of carrot flavor. J. Am. Soc. Hort. Sci. 1982, 107, 644–648. [Google Scholar]
- Kramer, M.; Bufler, G.; Nothnagel, T.; Carle, R.; Kammerer, D.R. Effects of cultivation conditions and cold storage on the polyacetylene contents of carrot (Daucus carota L.) and parsnip (Pastinaca sativa L.). J. Hortic. Sci. Biotechnol. 2012, 87, 101–106. [Google Scholar] [CrossRef]
- Christensen, L.P.; Kreutzmann, S. Determination of polyacetylenes in carrot roots (Daucus carota L.) by high-performance liquid chromatography coupled with diode array detection. J. Sep. Sci. 2007, 30, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Schachtman, D.P.; Reid, R.J.; Ayling, S.M. Phosphorus uptake by plants: From soil to cell. Plant Physiol. 1998, 116, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Dechassa, N.; Schenk, M.K.; Claassen, N.; Steingrobe, B. Phosphorus efficiency of cabbage (Brassica oleraceae L. Var. Capitata), carrot (Daucus carota L.), and potato (Solanum tuberosum L.). Plant Soil 2003, 250, 215–224. [Google Scholar]
- Plenchette, C.; Fortin, J.A.; Furlan, V. Growth responses of several plant species to mycorrhizae in a soil of moderate p-fertility. Plant Soil 1983, 70, 199–209. [Google Scholar] [CrossRef]
- Villegas, J.; Fortin, J.A. Phosphorus solubilization and pH changes as a result of the interactions between soil bacteria and arbuscular mycorrhizal fungi on a medium containing NO3 as nitrogen source. Can. J. Bot. 2002, 80, 571–576. [Google Scholar] [CrossRef]
- Gosling, P.; Hodge, A.; Goodlass, G.; Bending, G.D. Arbuscular mycorrhizal fungi and organic farming. Agric. Ecosyst. Environ. 2006, 113, 17–35. [Google Scholar] [CrossRef]
- Harrier, L.A.; Watson, C.A. The potential role of arbuscular mycorrhizal fungi in the bioprotection of plants against soil-borne pathogens in organic and/or other sustainable farming systems. Pest. Manag. Sci. 2004, 60, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Ngosong, C.; Jarosch, M.; Raupp, J.; Neumann, E.; Ruess, L. The impact of farming practice on soil microorganisms and arbuscular mycorrhizal fungi: Crop type versus long-term mineral and organic fertilization. Appl. Soil Ecol. 2010, 46, 134–142. [Google Scholar] [CrossRef]
- Lewis, B.G.; Garrod, B.; Sullivan, C. Accumulation of antifungal compounds on wound surfaces of carrot root tissue, Daucus carota, falcarindiol, 6-methoxymellein, wound healing, invading fungi. Trans. Br. Mycol. Soc. 1983, 80, 183–184. [Google Scholar] [CrossRef]
- Garrod, B.; Lewis, B.G.; Coxon, D.T. Cis-heptadeca-1,9-diene-4,6-diyne-3,8-diol, an antifungal polyacetylene from carrot root tissue. Physiol. Plant Pathol. 1978, 13, 241–246. [Google Scholar] [CrossRef]
- Lund, E.D.; White, J.M. Stress-related compounds in water-stressed carrots and celery. Proc. Fla. State Hortic. Soc. 1986, 99, 100–102. [Google Scholar]
- Nilsson, T. Growth and chemical composition of carrots as influenced by the time of sowing and harvest. J. Agric. Sci. 1987, 108, 459–468. [Google Scholar] [CrossRef]
- Goris, M.A. Glucide metabolism in cultivated carrot roots (nataise-type) during their vegetative cycles. Qual. Plant. Mater. Veg. 1969, 18, 307. [Google Scholar]
- Nilsson, T. Carbohydrate composition during long-term storage of carrots influenced by the time of harvest. J. Hort. Sci. 1987, 62, 191–203. [Google Scholar] [CrossRef]
- Koch, K. Sucrose metabolism: Regulatory mechanisms and pivotal roles in sugar sensing and plant development. Curr. Opin. Plant Biol. 2004, 7, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Suojala, T. Pre- and Postharvest Development of carrot Yield and Quality. Ph.D. Thesis, Department of Plant Production, University of Helsinki, Helsingfors, Finland, 2000. [Google Scholar]
- Suojala, T. Variation in sugar content and composition of carrot storage roots at harvest and during storage. Sci. Hortic. 2000, 85, 1–19. [Google Scholar] [CrossRef]
- Goris, M.A. The sugars in the root of the cultivated carrot (cultivar nantaise demi-longue) seasonal and climatological variations, distributions in the tissues, alteration during storage. Qual. Plant. Mater. Veg. 1969, 18, 283–306. [Google Scholar]

| Harvest Date | 26 August | 10 September | 24 September |
|---|---|---|---|
| Harvest number | 1 | 2 | 3 |
| Cultivation days | 104 | 119 | 133 |
| Precipitation, mm 1 | 153 | 154 | 175 |
| Degree-days °C 1 | 1874 | 2098 | 2277 |
| Weight, g | 30 ± 8 c | 66 ± 18 b | 109 ± 22 a |
| FaDOH, μg/g DW 2 | 503 ± 87 a | 529 ± 203 a | 340 ± 178 b |
| FaDOAc, μg/g DW 2 | 50 ± 13 a | 54 ± 27 a | 32 ± 24 b |
| FaOH, μg/g DW 2 | 104 ± 29 a | 118 ± 41 a | 69 ± 58 b |
| Sector | N | pH | Tot C% | Tot N% | P (Al) mg/100 g | P (HCl) mg/100 g | K (Al) mg/100 g | K (HCl) mg/100 g | Mg (Al) mg/100 g | K (Al)/Mg (Al) | Ca (Al) mg/100 g | Cu (HCl) mg/1000 g |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A1 | 6 | 5.9 ± 0.1 cd | 2.05 ± 0.1 d | 0.25 ± 0.02 ab | 2.0 ± 0.3 bc | 47.8 ± 2 b | 13.2 ± 1 d | 230 ± 12 cd | 22.1 ± 3 e | 0.60 ± 0.09 bc | 197 ± 13 e | 17.2 ± 2 bc |
| A2 | 6 | 5.8 ± 0.1 d | 2.35 ± 0.2 abc | 0.27 ± 0.02 ab | 2.3 ± 0.4 abc | 52.3 ± 3 a | 14.6 ± 1 cd | 254 ± 14 b | 26.6 ± 1 cd | 0.54 ± 0.02 c | 213 ± 6 d | 20.2 ± 1 a |
| B1 | 6 | 6.0 ± 0.1 c | 2.43 ± 0.2 ab | 0.28 ± 0.03 a | 2.1 ± 0.4 abc | 54.7 ± 2 a | 14.6 ± 1 cd | 285 ± 11 a | 29.9 ± 2 ab | 0.49 ± 0.02 d | 236 ± 8 c | 20.2 ± 1 a |
| B2 | 6 | 6.3 ± 0.2 a | 2.15 ± 021 cd | 0.28 ± 0.06 a | 2.4 ± 0.6 a | 47.1 ± 2 b | 16.1 ± 1 bc | 301 ± 16 a | 29.3 ± 1 abc | 0.55 ± 0.03 bc | 289 ± 14 a | 18.4 ± 3 abc |
| C1 | 6 | 5.9 ± 0.1 cd | 2.33 ± 0.1 bc | 0.25 ± 0.03 ab | 2.2 ± 0.2 abc | 55.3 ± 4 a | 14.5 ± 2 cd | 227 ± 11 cd | 19.3 ± 2 e | 0.75 ± 0.07 a | 193 ± 11 e | 17.8 ± 1 bc |
| C2 | 6 | 5.8 ± 0.1 d | 2.55 ± 0.2 ab | 0.28 ± 0.02 a | 2.5 ± 0.4 abc | 53.5 ± 4 a | 14.8 ± 3 cd | 219 ± 16 d | 25.6 ± 4 d | 0.58 ± 0.06 bc | 214 ± 13 d | 18.9 ± 2 ab |
| D1 | 6 | 6.0 ± 0.1 c | 2.57 ± 0.3 a | 0.27 ± 0.03 ab | 2.7 ± 0.7 ab | 51.5 ± 5 ab | 18.4 ± 2 a | 246 ± 24 bc | 31.3 ± 4 a | 0.59 ± 0.04 bc | 242 ± 17 c | 19.1 ± 2 ab |
| D2 | 6 | 6.1 ± 0.1 b | 2.05 ± 0.1 d | 0.24 ± 0.02 b | 1.7 ± 0.3 c | 42.0 ± 5 c | 17.2 ± 1 ab | 264 ± 23 b | 27.9 ± 1 bcd | 0.62 ± 0.04 b | 267 ± 11 b | 16.7 ± 1 c |
| Total | 48 | 6.0 ± 0.2 | 2.3 ± 0.3 | 0.27 ± 0.03 | 2.3 ± 0.6 | 50.5 ± 5 | 15.4 ± 2 | 253 ± 31 | 26.5 ± 4 | 0.59 ± 0.09 | 253 ± 31 | 18.6 ± 2 |
| Harvest | Sector | FaDOH μg/g DW 1 | FaDOAc μg/g DW 1 | FaOH μg/g DW 1 | FaOH/FaTP% | Fructose mg/g DW | Glucose mg/g DW | Sucrose mg/g DW | Weight g |
|---|---|---|---|---|---|---|---|---|---|
| 26 August | A1 | 528 ab | 44 ab | 91 cd | 13.8 bc | 194 a | 237 a | 61 a | 33.5 a |
| A2 | 483 ab | 41 b | 113 abc | 17.9 ab | 171 ab | 206 ab | 72 a | 33.1 a | |
| B1 | 473 ab | 44 ab | 81 d | 13.9 bc | 147 bc | 175 bc | 39 b | 30.7 a | |
| B2 | 494 ab | 47 ab | 78 d | 12.7 c | 136 c | 170 bc | 41 b | 25.7 a | |
| C1 | 575 a | 55 ab | 126 ab | 16.8 abc | 129 c | 164 c | 41 b | 28.7 a | |
| C2 | 552 a | 59 a | 140 a | 19.4 a | 123 c | 151 c | 44 b | 32.3 a | |
| D1 | 479 ab | 52 ab | 99 bcd | 15.7 abc | 121 c | 150 c | 36 b | 30.0 a | |
| D2 | 439 b | 57 ab | 98 cd | 16.5 abc | 117 c | 142 c | 34 b | 28.1 a | |
| 10 September | A1 | 362 d | 36 c | 113 abc | 22.5 ab | 155 ab | 190 ab | 162 a | 63.3 a |
| A2 | 381 cd | 31 c | 84 c | 16.8 bc | 144 ab | 176 b | 150 a | 75.4 a | |
| B1 | 446 cd | 51 bc | 99 bc | 16.9 bc | 133 b | 160 b | 132 ab | 58.8 a | |
| B2 | 420 cd | 48 bc | 140 ab | 22.9 a | 147 ab | 183 ab | 151 a | 67.0 a | |
| C1 | 898 a | 95 a | 155 a | 13.5 c | 174 a | 219 a | 104 b | 59.7 a | |
| C2 | 760 b | 71 b | 129 abc | 13.5 c | 144 ab | 179 b | 131 ab | 68.5 a | |
| D1 | 493 c | 52 bc | 107 abc | 16.3 c | 176 a | 218 a | 155 a | 65.6 a | |
| D2 | 470 cd | 48 bc | 120 abc | 18.3 abc | 154 ab | 188 ab | 108 b | 66.8 a | |
| 24 September | A1 | 129 b | 7 b | 10 c | 6.8 c | 139 b | 163 a | 166 a | 106.1 a |
| A2 | 181 b | 11 b | 16 c | 8.0 c | 139 b | 159 a | 159 a | 116.4 a | |
| B1 | 198 b | 14 b | 23 c | 9.5 c | 145 ab | 169 a | 139 abc | 111.2 a | |
| B2 | 237 b | 15 b | 30 c | 10.6 c | 142 ab | 169 a | 124 bc | 107.1 a | |
| C1 | 547 a | 53 a | 107 b | 15.2 b | 136 b | 161 a | 122 c | 106.2 a | |
| C2 | 484 a | 47 a | 111 ab | 17.0 ab | 141 ab | 161 a | 158 ab | 119.6 a | |
| D1 | 465 a | 49 a | 110 ab | 18.1 ab | 139 b | 166 a | 160 a | 104.7 a | |
| D2 | 477 a | 58 a | 145 a | 21.3 a | 152 a | 170 a | 139 abc | 97.8 a |
| pH | P (Al) | K (Al) | Mg (Al) | K/Mg | Ca (Al) | K (HCl) | P (HCl) | Cu (HCl) | Tot C | Tot N | FaDOH | FADOAc | FaOH | FaTP | FaOH/FaTP | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH | 1 | |||||||||||||||
| P (Al) | 0.19 | 1 | ||||||||||||||
| K (Al) | 0.43 ** | 0.34 * | 1 | |||||||||||||
| Mg (Al) | 0.48 ** | 0.30 * | 0.67 ** | 1 | ||||||||||||
| K/Mg | −0.15 | 0.00 | 0.18 | −0.61 ** | 1 | |||||||||||
| Ca (Al) | 0.82 ** | 0.24 | 0.56 ** | 0.75 ** | −0.38 ** | 1 | ||||||||||
| K (HCl) | 0.60 ** | −0.10 | 0.21 | 0.51 ** | −0.41 ** | 0.65 ** | 1 | |||||||||
| P (HCl) | −0.51 ** | 0.14 | −0.23 | −0.14 | 0.01 | −0.47 ** | −0.07 | 1 | ||||||||
| Cu (HCl) | −0.26 | 0.05 | 0.06 | 0.35 * | −0.38 ** | −0.05 | 0.31 * | 0.61 ** | 1 | |||||||
| Tot C | −.030 * | 0.47 ** | 0.28 | 0.34 * | −0.10 | −0.10 | −0.23 | 0.62 ** | 0.46 ** | 1 | ||||||
| Tot N | −0.08 | 0.49 ** | 0.07 | 0.21 | −0.18 | 0.06 | 0.03 | 0.24 | 0.14 | 0.45 ** | 1 | |||||
| FaDOH | −0.26 | 0.04 | −0.07 | −0.42 ** | 0.50 ** | −0.37 * | −0.40 ** | 0.44 ** | −0.06 | 0.22 | −0.02 | 1 | ||||
| FADOAc | −0.15 | 0.12 | 0.01 | −0.29 * | 0.44 ** | −0.22 | −0.22 | 0.38 ** | −0.05 | 0.16 | −0.01 | 0.84 ** | 1 | |||
| FaOH | 0.07 | 0.15 | 0.01 | −0.24 | 0.35 * | −0.02 | −0.02 | 0.08 | −0.12 | −0.14 | 0.12 | 0.45 ** | 0.45 ** | 1 | ||
| FaTP | −0.33 * | 0.06 | −0.14 | −0.52 ** | 0.55 ** | −0.47 ** | −0.44 ** | 0.42 ** | −0.09 | 0.14 | 0.01 | 0.97 ** | 0.84 ** | 0.57 ** | 1 | |
| FaOH/FaTP | 0.24 | 0.14 | −0.03 | 0.03 | −0.08 | 0.23 | 0.26 | −0.36 * | −0.11 | −0.42 ** | 0.07 | −0.52 ** | −0.37 ** | 0.48 ** | −0.35 * | 1 |
| Soil trait | Quartiles | FaDOH μg/g DW 1 | FaDOAc μg/g DW 1 | FaOH μg/g DW 1 | Total FaTP μg/g DW 1 | FaOH/FaTP% | Root weight, g | |
|---|---|---|---|---|---|---|---|---|
| Total carbon % | 1.9–2.1 | 17 | 464 a | 50 ab | 120 a | 634 a | 19.8 a | 61 a |
| 2.2–2.3 | 10 | 491 a | 41 b | 113 a | 645 a | 18.2 ab | 70 a | |
| 2.4–2.5 | 12 | 608 a | 65 a | 129 a | 803 a | 16.5 ab | 66 a | |
| 2.6–2.9 | 9 | 586 a | 62 ab | 106 a | 754 a | 14.2 b | 68 a | |
| Phosphorus (HCl) mg/100 g | 37–47 | 12 | 449 b | 50 ab | 124 a | 622 b | 20.1 a | 65 a |
| 47–51 | 12 | 447 b | 37 b | 114 a | 598 b | 19.1 ab | 65 a | |
| 51–55 | 13 | 583 ab | 64 a | 111 a | 758 ab | 16.0 ab | 68 a | |
| 55–59 | 11 | 640 a | 66 a | 126 a | 832 a | 15.0 b | 65 a | |
| Potassium (HCl) mg/100 g | 198–230 | 12 | 632 a | 62 a | 115 a | 809 a | 15.9 a | 66 a |
| 230–248 | 13 | 584 ab | 60 a | 131 a | 774 ab | 17.3 a | 69 a | |
| 249–278 | 11 | 442 b | 45 a | 101 a | 588 b | 17.4 a | 62 a | |
| 279–314 | 12 | 445 b | 49 a | 124 a | 618 ab | 19.8 a | 64 a | |
| Potassium (Al)/ Magnesium (Al), % | 46–53 | 12 | 456 b | 48 b | 104 a | 607 b | 17.4 a | 64 a |
| 53–57 | 12 | 494 ab | 48 b | 113 a | 654 b | 17.4 a | 69 a | |
| 57–63 | 13 | 520 ab | 51 ab | 120 a | 622 ab | 18.2 a | 64 a | |
| 63–84 | 11 | 656 a | 71 a | 137 a | 865 a | 17.3 a | 67 a |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kjellenberg, L.; Johansson, E.; Gustavsson, K.-E.; Granstedt, A.; Olsson, M.E. Correlations between Polyacetylene Concentrations in Carrot (Daucus carota L.) and Various Soil Parameters. Foods 2016, 5, 60. https://doi.org/10.3390/foods5030060
Kjellenberg L, Johansson E, Gustavsson K-E, Granstedt A, Olsson ME. Correlations between Polyacetylene Concentrations in Carrot (Daucus carota L.) and Various Soil Parameters. Foods. 2016; 5(3):60. https://doi.org/10.3390/foods5030060
Chicago/Turabian StyleKjellenberg, Lars, Eva Johansson, Karl-Erik Gustavsson, Artur Granstedt, and Marie E. Olsson. 2016. "Correlations between Polyacetylene Concentrations in Carrot (Daucus carota L.) and Various Soil Parameters" Foods 5, no. 3: 60. https://doi.org/10.3390/foods5030060
APA StyleKjellenberg, L., Johansson, E., Gustavsson, K.-E., Granstedt, A., & Olsson, M. E. (2016). Correlations between Polyacetylene Concentrations in Carrot (Daucus carota L.) and Various Soil Parameters. Foods, 5(3), 60. https://doi.org/10.3390/foods5030060







