Influence of Heat Treatments on Carotenoid Content of Cherry Tomatoes
Abstract
:1. Introduction
2. Experimental
2.1. Samples Preparation: Raw, Home-Processed and Canned Tomatoes
2.2. Reagents
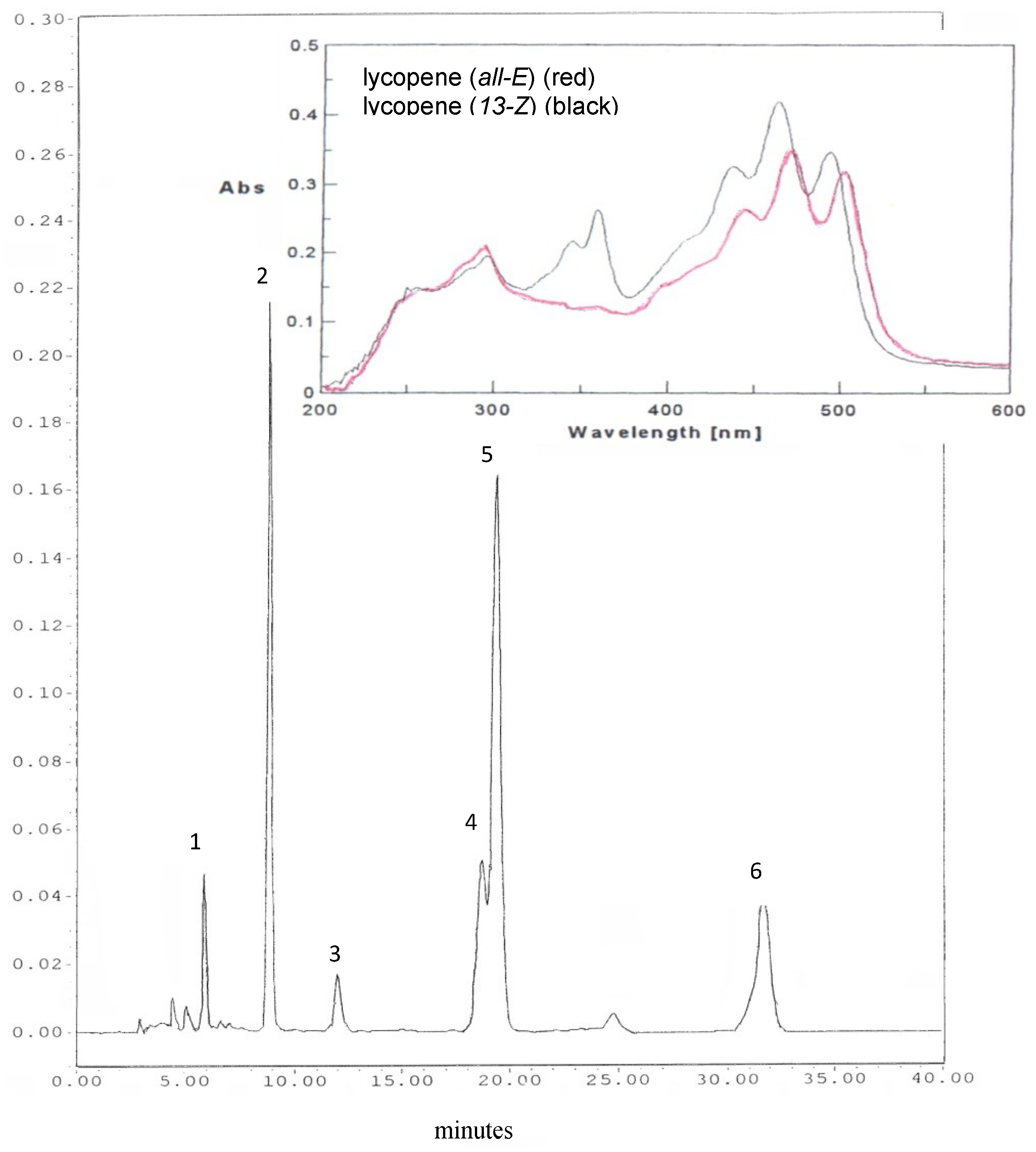
2.3. Extraction and HPLC Analysis of Carotenoids
2.4. Standard Solutions
2.5. Statistical Analysis
3. Results and Discussion
| Samples | Moisture | Lutein | Lycopene | β-Carotene |
|---|---|---|---|---|
| g/100 g | mg/100 g f.w. | mg/100 g f.w. | mg/100 g f.w. | |
| Raw | 93.70 ± 0.10 | 0.11 ± 0.00 | 5.12 ± 0.35 | 1.00 ± 0.05 |
| Canned | 92.50 ± 0.16 | 0.15 ± 0.01 | 11.60 ± 0.18 | 0.75 ± 0.01 |
| Carotenoid (mg/100 g f.w.) | ||||||
|---|---|---|---|---|---|---|
| Whole | Pulp | Skin | ||||
| Raw | Home-processed | Raw | Home-processed | Raw | Home-processed | |
| Lutein | 0.11 ± 0.00 | 0.10 ± 0.01 | 0.12 ± 0.01 | 0.09 ± 0.01 * | 0.09 ± 0.01 | 0.08 ± 0.00 |
| Lycopene | 5.12 ± 0.35 | 5.58 ± 0.35 | 4.54 ± 0.11 | 5.39 ± 0.26 ** | 19.24 ± 0.76 | 12.25 ± 1.11 *** |
| β-Carotene | 1.00 ± 0.05 | 0.93± 0.06 | 1.07 ± 0.01 | 1.01 ± 0.01 ** | 1.38 ± 0.04 | 1.15 ± 0.04 ** |
4. Conclusions
Conflicts of Interest
References
- La Vecchia, C. Tomatoes, lycopene intake, and digestive tract and female hormone-related neoplasm. Exp. Biol. Med. (Maywood) 2002, 227, 860–863. [Google Scholar]
- Obulesu, M.; Dowlathabad, M.R.; Bramhachari, P.V. Carotenoids and Alzheimer’s disease: An insight into therapeutic role of retinoids in animal models. Neurochem. Int. 2011, 59, 535–541. [Google Scholar] [CrossRef]
- Koh, W.P.; Yuan, J.M.; Wang, R.; Lee, Y.P.; Lee, B.L.; Yu, M.C; Ong, C.N. Plasma carotenoids and risk of acute myocardial infarction in the Singapore Chinese Health Study. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 685–690. [Google Scholar] [CrossRef]
- Hung, R.J.; Zhang, Z.F.; Rao, J.Y.; Pantuck, A.; Reuter, V.E.; Heber, D.; Lu, Q.Y. Protective effects of plasma carotenoids on the risk of bladder cancer. J. Urol. 2006, 176, 1192–1197. [Google Scholar] [CrossRef]
- Giovannucci, E. Tomato products, lycopene, and prostate cancer: A review of the epidemiological literature. J. Nutr. 2005, 135, 2030S–2031S. [Google Scholar]
- Palozza, P.; Simone, R.E.; Catalano, A.; Mele, M.C. Tomato lycopene and lung cancer prevention: From experimental to human studies. Cancers 2011, 3, 2333–2357. [Google Scholar] [CrossRef]
- Sesso, H.D.; Liu, S.; Gaziano, M.; Buring, J.E. Dietary lycopene, tomato-based food products and cardiovascular disease in women. J. Nutr. 2003, 133, 2336–2341. [Google Scholar]
- Kohlmeier, L.; Kark, J.D.; Gomez-Garcia, E.; Martin, B.C.; Steck, S.E.; Kardinaal, A.F.; Ringstad, J.; Thamm, M.; Masaev, V.; Riemersma, R.; et al. Lycopene and myocardial infarction risk in the EURAMIC study. Am. J. Epidemiol. 1997, 146, 618–626. [Google Scholar] [CrossRef]
- Li, H.; Deng, Z.; Liu, R.; Loewen, S.; Tsao, R. Carotenoid compositions of coloured tomato cultivars and contribution to antioxidant activities and protection against H2O2-induced cell death in H9c2. Food Chem. 2013, 136, 878–888. [Google Scholar] [CrossRef]
- Khachik, F.; Carvalho, L.; Bernstein, P.S.; Muir, G.J.; Zhao, D.Y.; Katz, N.B. Chemistry, distribution, and metabolism of tomato carotenoids and their impact on human health. Exp. Biol. Med. (Maywood) 2002, 227, 845–851. [Google Scholar]
- Ong, A.S.H.; Tee, E.S. Natural sources of carotenoids from plants and oils. Methods Enzymol. 1992, 213, 142–167. [Google Scholar] [CrossRef]
- Sies, H.; Stahl, W. Vitamins E and C, beta-carotene, and other carotenoids as antioxidants. Am. J. Clin. Nutr. 1995, 62, 1315S–1321S. [Google Scholar]
- Abushita, A.A.; Hebshi, E.A.; Daood, H.G.; Biacs, P.A. Determination of antioxidant vitamins in tomatoes. Food Chem. 1997, 60, 207–212. [Google Scholar] [CrossRef]
- Giovannucci, E.; Ascherio, A.; Rimm, E.B.; Stampfer, M.J.; Colditz, G.A.; Willett, W.C. Intake of carotenoids and retinol in relation to risk of prostate cancer. J. Natl. Cancer Inst. 1995, 87, 1767–1776. [Google Scholar] [CrossRef]
- Giovannucci, E. Tomatoes, tomato-based products, lycopene, and cancer: Review of the epidemiologic literature. J. Natl. Cancer Inst. 1999, 91, 317–331. [Google Scholar] [CrossRef]
- Etminan, M.; Takkouche, B.; Caamano-Isorna, F. The role of tomato products and lycopene in the prevention of prostate cancer: A meta-analysis of observational studies. Cancer Epidemiol. Biomarkers Prev. 2004, 13, 340–345. [Google Scholar]
- Gärtner, C.; Stahl, W.; Sies, H. Lycopene is more bioavailable from tomato paste than from fresh tomatoes. Am. J. Clin. Nutr. 1997, 66, 116–122. [Google Scholar]
- Porrini, M.; Riso, P.; Testolin, G. Absorption of lycopene from single or daily portions of raw and processed tomato. Br. J. Nutr. 1998, 80, 353–361. [Google Scholar] [CrossRef]
- Abushita, A.A.; Daood, H.G.; Biacs, P.A. Change in carotenoids and antioxidant vitamins in tomato as a function of varietal and technological factors. J. Agric. Food Chem. 2000, 48, 2075–2081. [Google Scholar] [CrossRef]
- Thompson, K.A.; Marshall, M.R.; Sims, C.A.; Wei, C.I.; Sargent, S.A.; Scott, J.W. Cultivar, maturity, and heat treatment on lycopene content in tomatoes. J. Food Sci. 2000, 65, 791–795. [Google Scholar] [CrossRef]
- Dietz, J.M.; Sri Kantha, S; Erdman, J.W., Jr. Reversed phase HPLC analysis of alpha- and beta-carotene from selected raw and cooked vegetables. Plant Foods Hum. Nutr. 1988, 38, 333–341. [Google Scholar] [CrossRef]
- Dewanto, V.; Wu, X.; Adom, K.K.; Liu, R.H. Thermal processing enhances the nutritional value of tomatoes by increasing total antioxidant activity. J. Agric. Food Chem. 2002, 50, 3010–3014. [Google Scholar] [CrossRef]
- Hsu, K.C.; Tan, F.J.; Chi, H.Y. Evaluation of microbial inactivation and physicochemical properties of pressurized tomato juice during refrigerated storage. LWT Food Sci. Technol. 2008, 41, 367–375. [Google Scholar] [CrossRef]
- Georgé, S.; Tourniaire, F.; Gautier, H.; Goupy, P.; Rock, E.; Caris-Veyrat, C. Changes in the contents of carotenoids, phenolic compounds and vitamin C during technical processing and lyophilisation of red and yellow tomatoes. Food Chem. 2011, 124, 1603–1611. [Google Scholar] [CrossRef]
- Van het Hof, K.H.; de Boer, B.C.J.; Tijburg, L.B.; Lucius, B.R.; Zijp, I.; West, C.E.; Hautvast, J.G.; Westrate, J.A. Carotenoid bioavailability in humans from tomatoes processed in different ways determined from the carotenoids response in the triglyceride-rich lipoprotein fraction of plasma after a single consumption and in plasma after four days of consumption. J. Nutr. 2000, 130, 1189–1196. [Google Scholar]
- Courraud, J.; Berger, J.; Cristol, J.P.; Avallone, S. Stability and bioaccessibility of different forms of carotenoids and vitamin A during in vitro digestion. Food Chem. 2013, 136, 871–877. [Google Scholar] [CrossRef]
- Plaza, L.; Sánchez-Moreno, C.; de Ancos, B.; Elez-Martínez, P.; Martín-Belloso, O.; Cano, M.P. Carotenoid and flavanone content during refrigerated storage of orange juice processed by high-pressure, pulsed electric fields and low pasteurization. LWT Food Sci. Technol. 2011, 44, 834–839. [Google Scholar] [CrossRef]
- Hornero-Mendez, D.; Mínguez-Mosquera, M.I. Bioaccessibility of carotenes from carrots: Effect of cooking and addition of oil. Innov. Food Sci. Emerg. Technol. 2007, 8, 407–412. [Google Scholar] [CrossRef]
- Van Buggenhout, S.; Alminger, M.; Lemmens, L.; Colle, I.; Knockaert, G.; Moelants, K.; van Loey, A.; Hendrickx, M. In vitro approaches to estimate the effect of food processing on carotenoid bioavailability need thorough understanding of process induced microstructural changes. Trends Food Sci. Technol. 2010, 21, 607–618. [Google Scholar] [CrossRef]
- Rodriguez-Amaya, D.B. Changes in carotenoids during processing and storage of food. Arch. Latinoam. Nutr. 1999, 49, 38S–47S. [Google Scholar]
- Nguyen, M.L.; Schwartz, S.J. Lycopene stability during food processing. Proc. Soc. Exp. Biol. Med. 1998, 218, 101–105. [Google Scholar] [CrossRef]
- Shierlie, J.; Bretzel, W.; Buhler, I.; Faccin, N.; Hess, D; Steiner, K.; Schuep, W. Content and isomeric ratios of lycopene in food and human blood plasma. Food Chem. 1997, 59, 459–465. [Google Scholar] [CrossRef]
- Knockaert, G.; Pulissery, S.K.; Colle, I.; van Buggenhout, S.; Hendrickx, M.; van Loey, A. Lycopene degradation, isomerization and in vitro bioaccessibility in high pressure homogenized tomato puree containing oil: Effect of additional thermal and high pressure processing. Food Chem. 2012, 135, 1290–1297. [Google Scholar] [CrossRef]
- Anguelova, T.; Warthesen, J. Lycopene stability in tomato powders. J. Food Sci. 2000, 65, 67–70. [Google Scholar] [CrossRef]
- Demiray, E.; Tulek, Y.; Yilmaz, Y. Degradation kinetics of lycopene, β-carotene and ascorbic acid in tomatoes during hot air drying. LWT Food Sci. Technol. 2013, 50, 172–176. [Google Scholar] [CrossRef]
- Tangwongchai, R.; Ledward, D.A.; Ames, J.M. Effect of high-pressure treatment on the texture of cherry tomato. J. Agric. Food Chem. 2000, 48, 1434–1441. [Google Scholar] [CrossRef]
- Squarcina, N.; Sandei, L.; Carpi, G.; Rovere, P. Evaluation of different technological treatments on cherry tomatoes by means of a laboratory texturometer. Ind. Conserve 1999, 74, 35–39. [Google Scholar]
- Ciampa, A; Dell’Abate, M.T.; Masetti, O.; Valentini, M.; Sequi, P. Seasonal chemical-hysical changes of PGI Pachino cherry tomatoes detected by magnetic resonance imaging (MRI). Food Chem. 2010, 122, 1253–1260. [Google Scholar] [CrossRef]
- Leonardi, C.; Ambrosino, P.; Esposito, F.; Fogliano, V. Antioxidative activity and carotenoid and tomatine contents in different typologies of fresh consumption tomatoes. J. Agric. Food Chem. 2000, 48, 4723–4727. [Google Scholar] [CrossRef]
- Raffo, A.; Leonardi, C.; Fogliano, V.; Ambrosino, P; Salucci, M.; Gennaro, L.; Bugianesi, R.; Giuffrida, F.; Quaglia, G. Nutritional value of cherry tomatoes (Lycopersicon esculentum cv. Naomi F1) harvested at different ripening stages. J. Agric. Food Chem. 2002, 50, 6550–6556. [Google Scholar] [CrossRef]
- Raffo, A.; la Malfa, G.; Fogliano, V.; Maiani, G.; Quaglia, G. Seasonal variations in antioxidant components of cherry tomatoes (Lycopersicon esculentum cv. Naomi F1). J. Food Comp. Anal. 2006, 19, 11–19. [Google Scholar] [CrossRef]
- Bugianesi, R.; Salucci, M.; Leonardi, C.; Ferracane, R.; Catasta, G.; Azzini, E.; Maiani, G. Effect of domestic cooking on human bioavailability of naringenin, chlorogenic acid, lycopene and beta-carotene in cherry tomatoes. Eur. J. Nutr. 2004, 43, 360–366. [Google Scholar] [CrossRef]
- Zanfini, A.; Corbini, G.; la Rosa, C.; Dreassi, E. Antioxidant activity of tomato lipophilic extracts and interactions between carotenoids and α-tocopherol in synthetic mixtures. LWT Food Sci. Technol. 2010, 43, 67–72. [Google Scholar] [CrossRef]
- Sharma, S.K.; le Maguer, M. Lycopene in tomatoes and tomato pulp fractions. Ital. J. Food Sci. 1996, 8, 107–113. [Google Scholar]
- Clinton, S.K. Lycopene: Chemistry, biology and implications for human health and disease. Nutr. Rev. 1998, 56, 35–51. [Google Scholar] [CrossRef]
- Tonucci, L.H.; Holden, J.M.; Beecher, G.R.; Khachik, F.; Davis, C.S.; Mulokozi, G. Carotenoid content of thermally processed tomato-based food products. J. Agric. Food Chem. 1995, 43, 579–586. [Google Scholar] [CrossRef]
- Hart, D.J.; Scott, K.J. Development and evaluation of an HPLC method for the analysis of carotenoids in foods, and the measurement of the carotenoids content of vegetables and fruits commonly consumed in the UK. Food Chem. 1995, 54, 101–111. [Google Scholar] [CrossRef]
- Holland, B.; Unwin, I.D.; Buss, D.H. Vegetables, Herbs and Spices. In The composition of Food, 4th ed.; McCance & Widdowson’s: Cambridge, UK, 1991; p. 133. [Google Scholar]
- Adalid, A.M.; Rosello, S.; Nuez, F. Evaluation and selection of tomato accessions (Solanum section Lycopersicon) for content of lycopene, β-carotene and ascorbic acid. J. Food Comp. Anal. 2010, 23, 613–618. [Google Scholar] [CrossRef]
- Al-Wandawi, H.; Abdul-Rahman, M.; Al-Shaikhly, K. Tomato processing wastes as essential raw material source. J. Agric. Food Chem. 1985, 33, 804–807. [Google Scholar] [CrossRef]
- Nguyen, M.; Francis, D.; Schwartz, S. Thermal isomerization susceptibility of carotenoids in different tomato varieties. J. Sci. Food Agric. 2001, 81, 910–917. [Google Scholar] [CrossRef]
- Deming, D.M.; Baker, D.H.; Erdman, J.W., Jr. The relative vitamin A value of 9-cis β-carotene is less and that of 13-cis β-carotene may be greater than the accepted 50% that of all-trans β-carotene in gerbils. J. Nutr. 2002, 132, 2709–2712. [Google Scholar]
- Böhm, V; Puspitasari-Nienaber, N.L.; Ferruzzi, M.G.; Schwartz, S.G. Trolox equivalent antioxidant capacity of different geometrical isomers of α-carotene, β-carotene, lycopene and zeaxanthin. J. Agric. Food Chem. 2002, 50, 221–226. [Google Scholar] [CrossRef]
- Müller, L; Goupy, P.; Fröhlich, K.; Dangles, O.; Caris-Veyrat, C.; Böhm, V. Comparative study on antioxidant activity of lycopene (Z)-isomers in different assays. J. Agric. Food Sci. 2011, 59, 4504–4511. [Google Scholar] [CrossRef]
- Clinton, S.K.; Emenhiser, C.; Schwartz, S.J.; Bostwick, D.G.; Williams, A.W.; Moore, B.J.; Erdman, J.W., Jr. cis-trans lycopene isomers, carotenoids, and retinol in the human prostate. Cancer Epidemiol. Biomarkers Prev. 1996, 5, 823–833. [Google Scholar]
- Boileau, T.W.; Boileau, A.C.; Erdman, J.W., Jr. Bioavailability of all-trans and cis-isomers of lycopene. Exp. Biol. Med. (Maywood) 2002, 227, 914–919. [Google Scholar]
- Lambelet, P.; Richelle, M.; Bortlik, K.; Franceschi, F.; Giori, A.M. Improving the stability of lycopene Z-isomers in isomerised tomato extracts. Food Chem. 2009, 112, 156–161. [Google Scholar] [CrossRef]
- Unlu, N.Z.; Bohn, T.; Francis, D.M.; Nagaraja, H.N.; Clinton, S.K.; Schwartz, S. Lycopene form heat-induced cis-isomer-rich tomato sauce is more bioavailable than from all-trans-rich tomato sauce in human subjects. Br. J. Nutr. 2007, 98, 140–146. [Google Scholar] [CrossRef]
- Beecher, G.R. Nutrient content of tomatoes and tomato products. Proc. Soc. Exp. Biol. Med. 1998, 218, 98–100. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
D'Evoli, L.; Lombardi-Boccia, G.; Lucarini, M. Influence of Heat Treatments on Carotenoid Content of Cherry Tomatoes. Foods 2013, 2, 352-363. https://doi.org/10.3390/foods2030352
D'Evoli L, Lombardi-Boccia G, Lucarini M. Influence of Heat Treatments on Carotenoid Content of Cherry Tomatoes. Foods. 2013; 2(3):352-363. https://doi.org/10.3390/foods2030352
Chicago/Turabian StyleD'Evoli, Laura, Ginevra Lombardi-Boccia, and Massimo Lucarini. 2013. "Influence of Heat Treatments on Carotenoid Content of Cherry Tomatoes" Foods 2, no. 3: 352-363. https://doi.org/10.3390/foods2030352
APA StyleD'Evoli, L., Lombardi-Boccia, G., & Lucarini, M. (2013). Influence of Heat Treatments on Carotenoid Content of Cherry Tomatoes. Foods, 2(3), 352-363. https://doi.org/10.3390/foods2030352




