Comparison of Flocculation Methods for Sodium Alginate and Characterization of Its Structure and Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Instruments
2.3. Experimental Methods
2.3.1. Optimization of the Alkaline Treatment Process of LN
Single-Factor Experiment on Alkaline Treatment Process
Orthogonal Testing of Alkaline Treatment Processes
2.3.2. Effects of Different Flocculation Processes on the Extraction Yields of Sodium Alginate
2.3.3. Comparison of the Extraction Yields of Sodium Alginate from Different Parts of LN
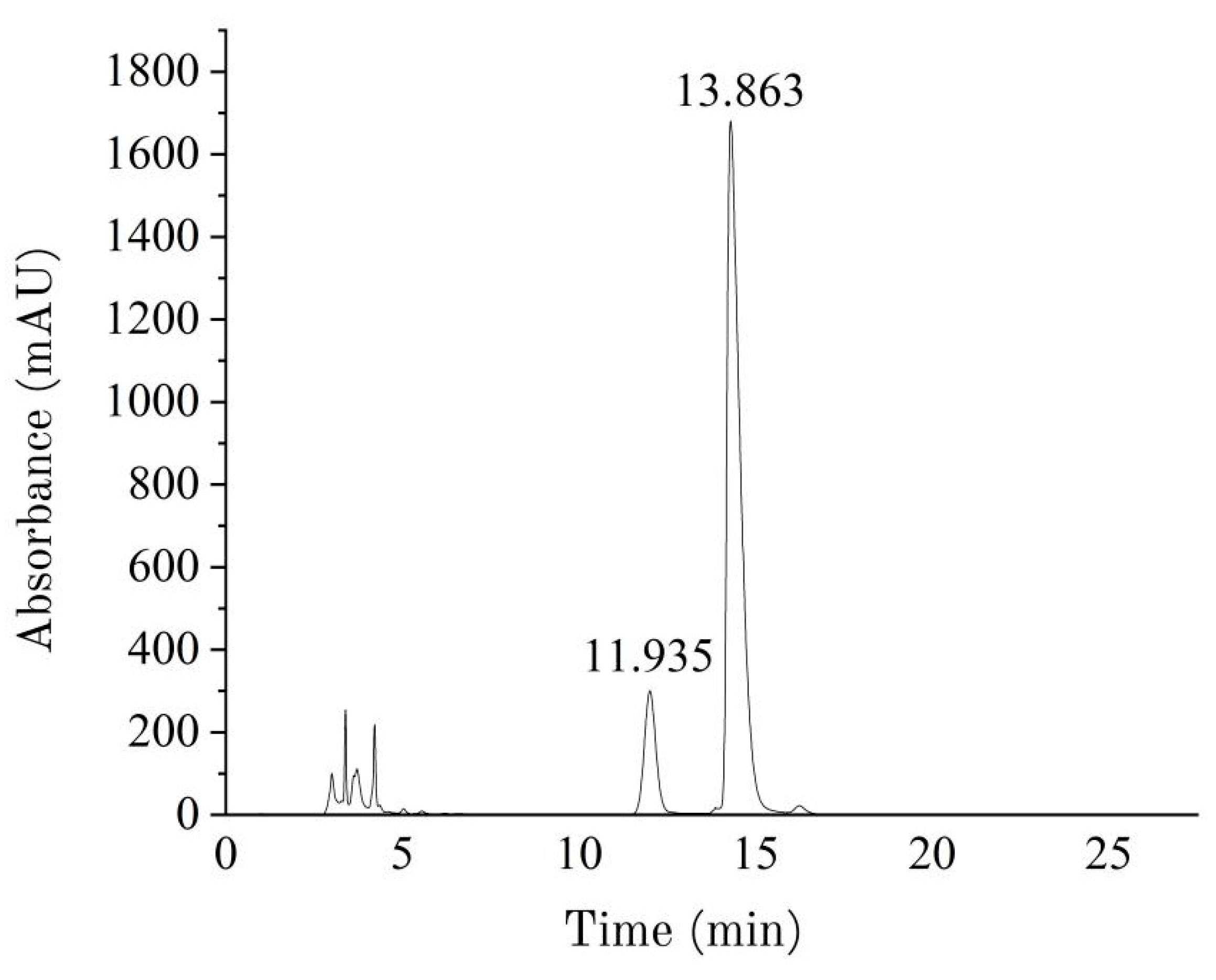
2.3.4. Determination of Molecular Weight
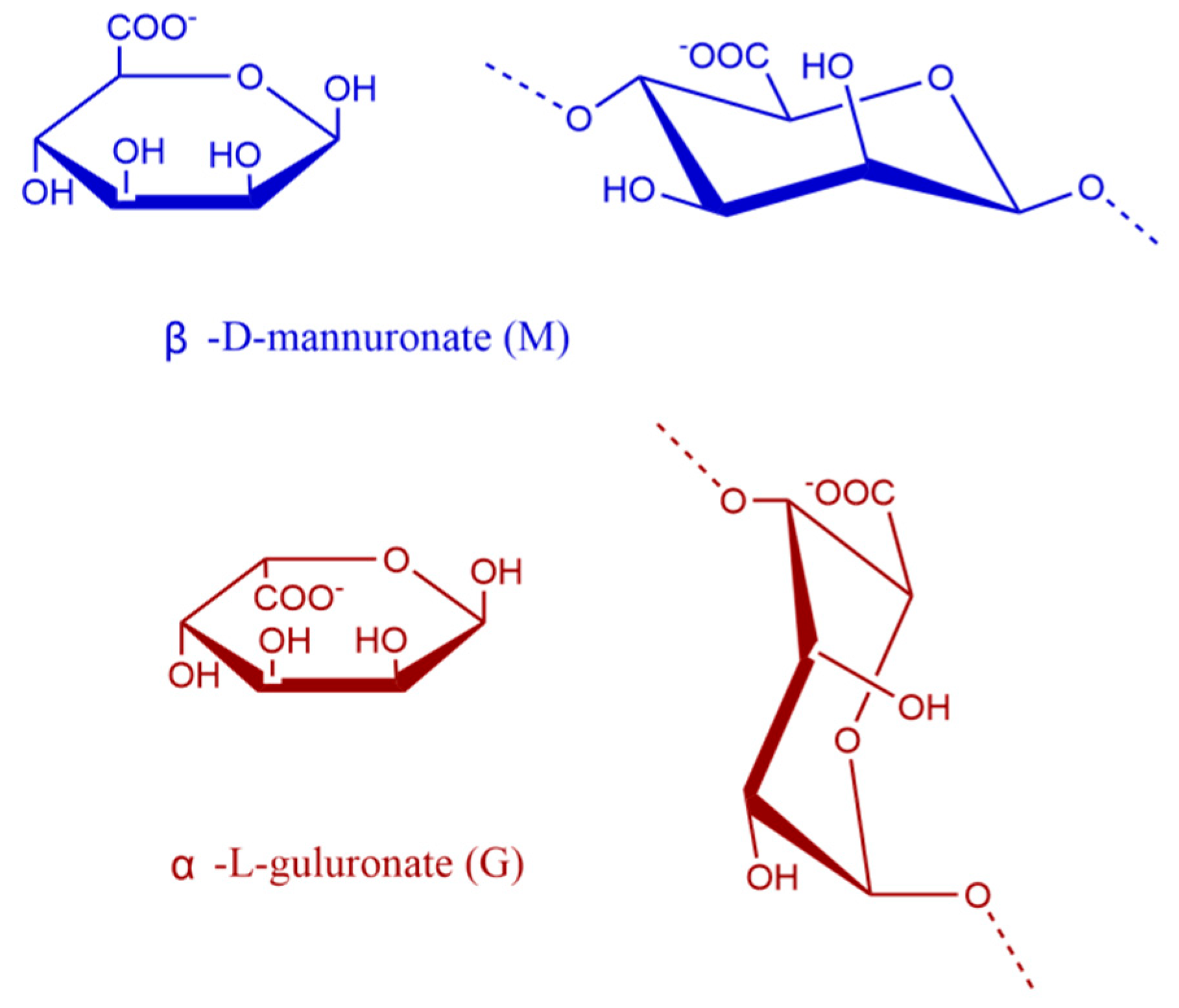
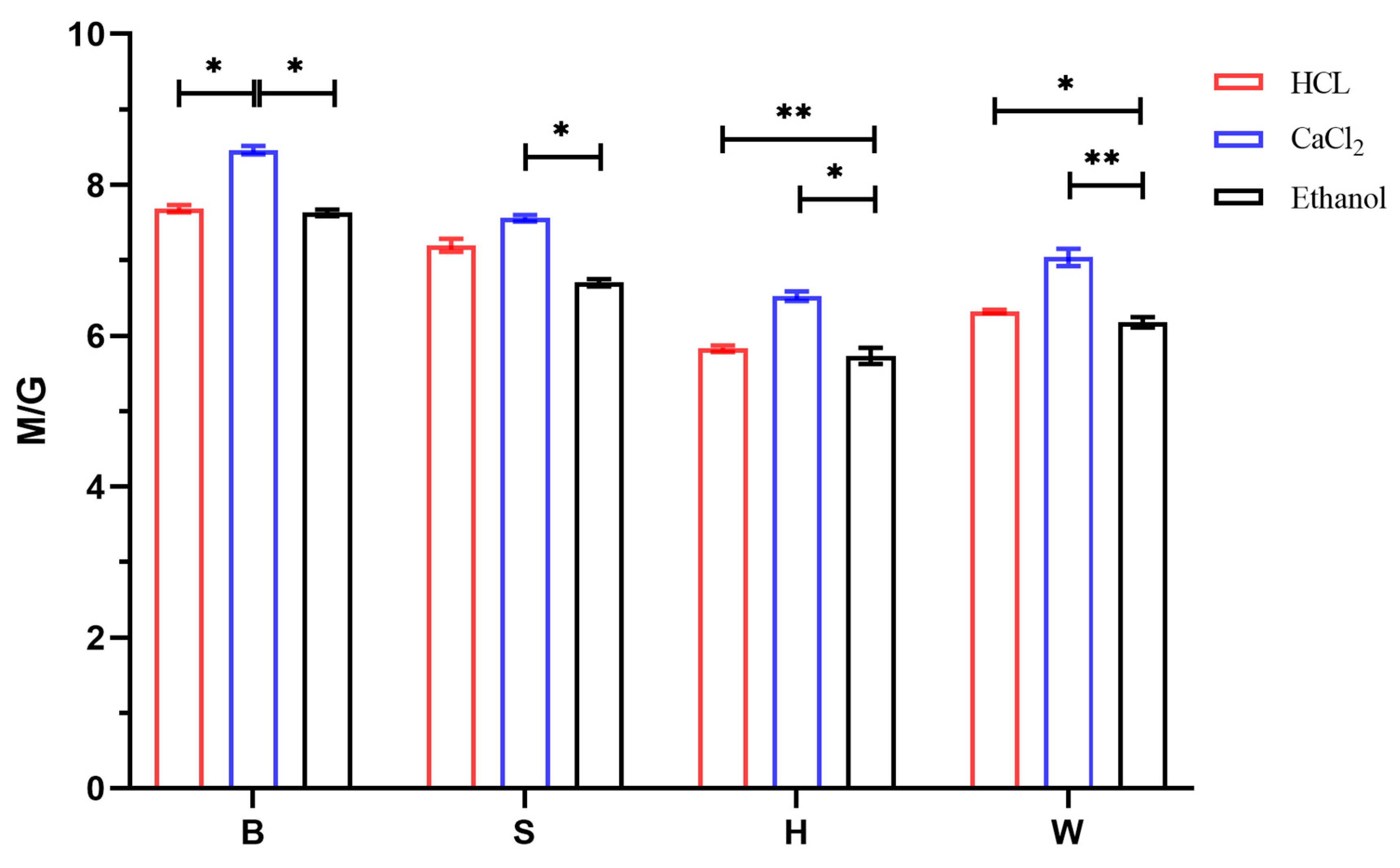
2.3.5. Determination of M/G Values
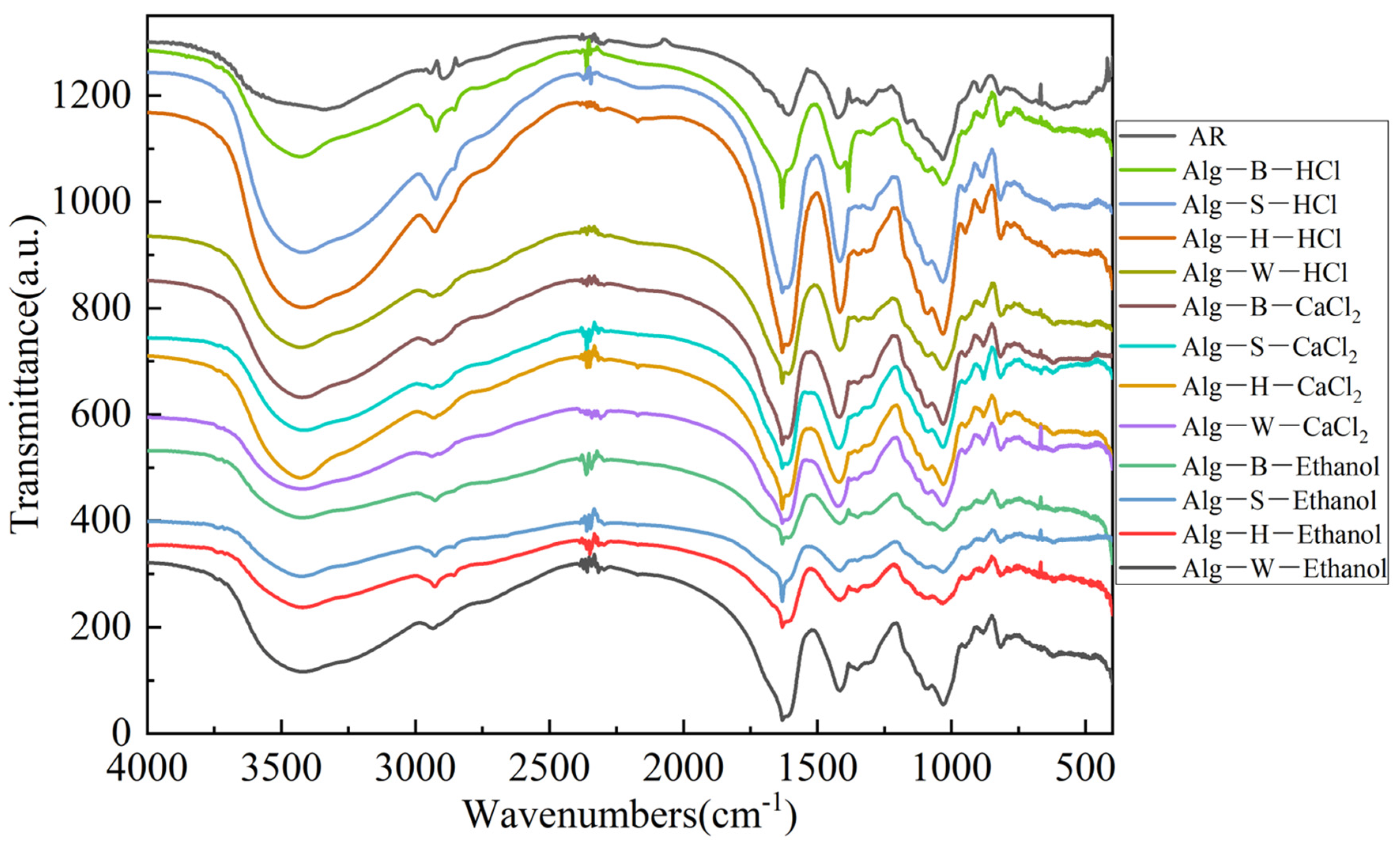
2.3.6. Infrared Spectroscopy
2.3.7. Determination of Physical Properties of Sodium Alginate Samples
2.3.8. Statistical Analysis
3. Results and Discussion
3.1. Optimal Process Determination
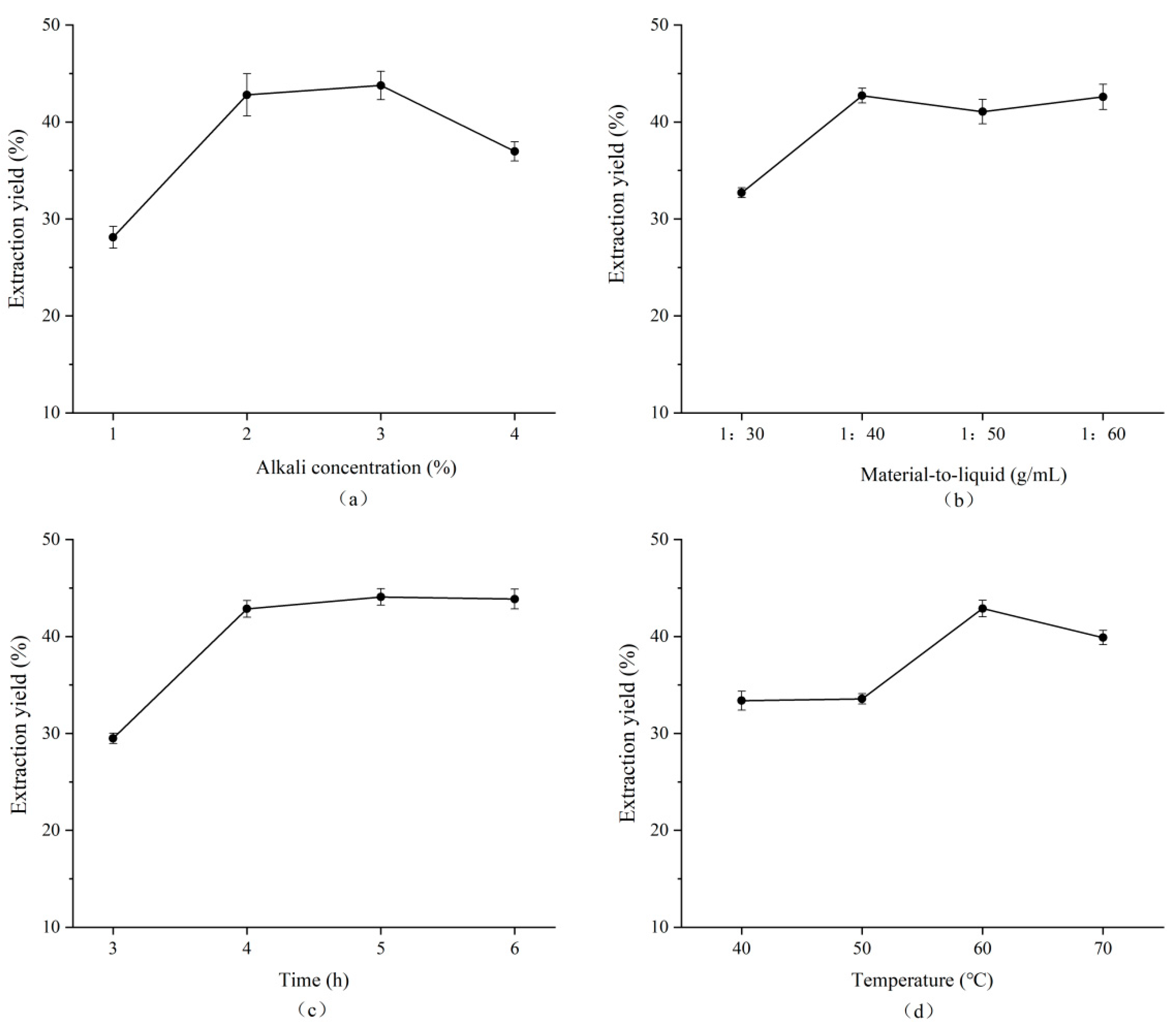
3.1.1. Single-Factor Experiments on Alkaline Treatment Processes
3.1.2. Results of Orthogonal Experimental Optimization of Alkaline Treatment Process
3.2. Extraction Yield of Sodium Alginate
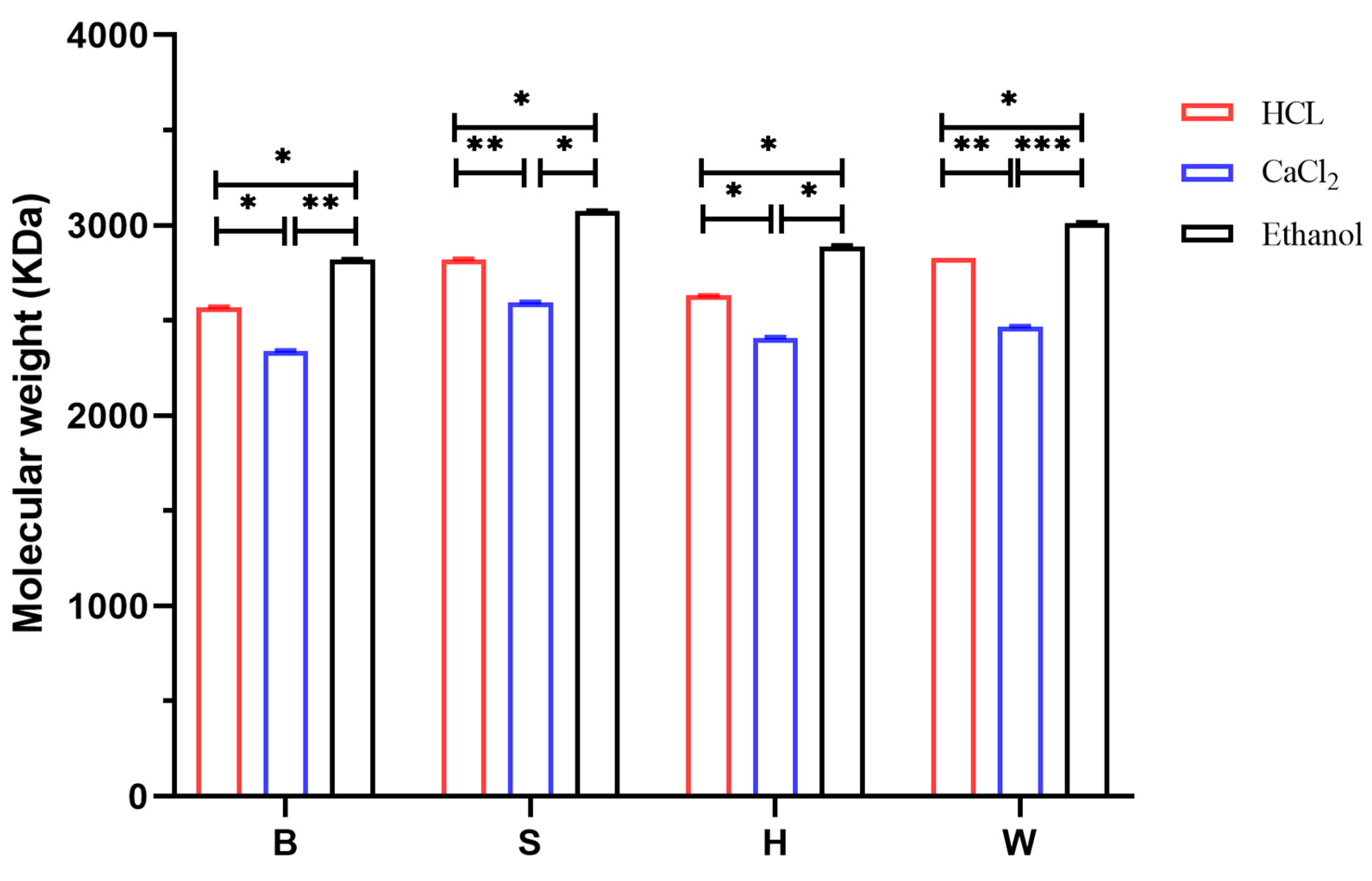
3.3. Analysis of Molecular Weight Results
3.4. Analysis of M/G Results
3.5. Analysis of Infrared Spectroscopy Results
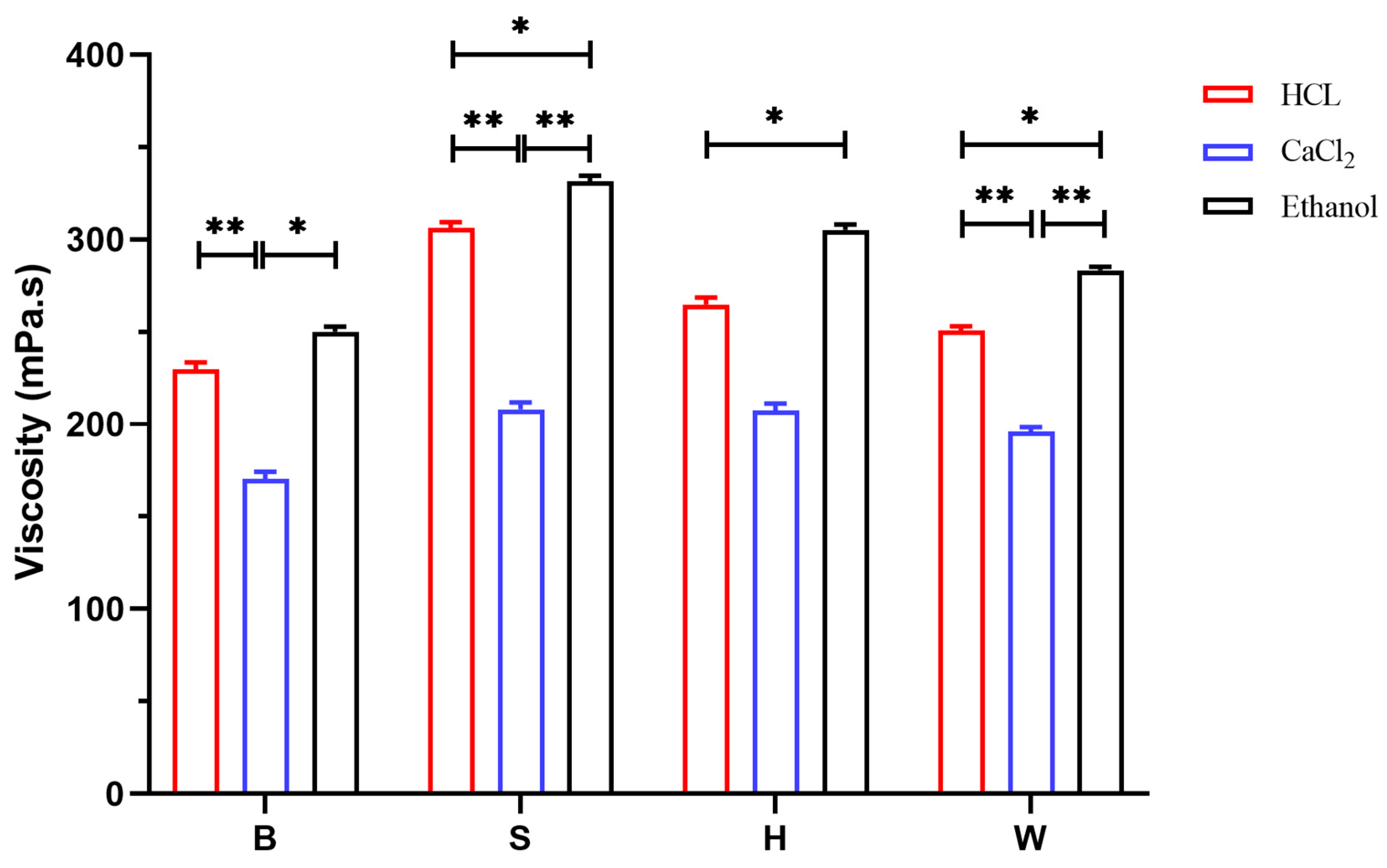
3.6. Analysis of Physical Properties of Sodium Alginate Samples
3.6.1. Viscosity Result Analysis
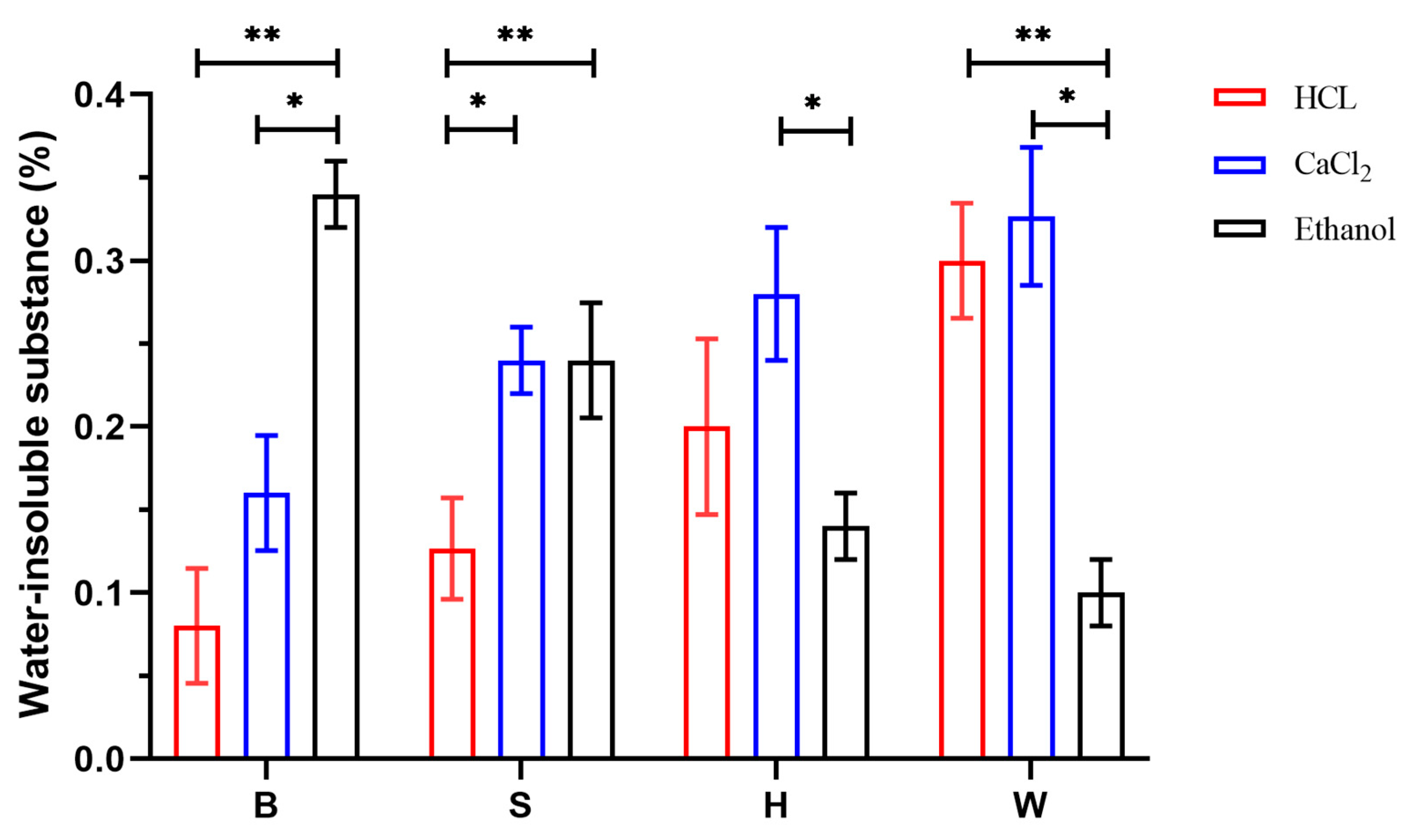
3.6.2. Analysis of Results of Water-Insoluble Substance Determination
3.6.3. Analysis of Other Physicochemical Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lim, S.J.; Wan Aida, W.M.; Schiehser, S.; Rosenau, T.; Böhmdorfer, S. Structural elucidation of fucoidan from Cladosiphon okamuranus (Okinawa mozuku). Food Chem. 2019, 272, 222–226. [Google Scholar] [CrossRef]
- Madsen, M.; Westh, P.; Khan, S.; Ipsen, R.; Almdal, K.; Aachmann, F.L.; Svensson, B. Impact of Alginate Mannuronic-Guluronic Acid Contents and pH on Protein Binding Capacity and Complex Size. Biomacromolecules 2021, 22, 649–660. [Google Scholar] [CrossRef] [PubMed]
- Cid, A.H.; Flores, I.M.; Pizarro, F.J.; Castillo, A.X.; Barros, E.D.; Moreno-Piraján, C.J.; Ortiz, A.C. Mechanisms of Cu2+ biosorption on Lessonia nigrescens dead biomass: Functional groups interactions and morphological characterization. J. Environ. Chem. Eng. 2018, 6, 2696–2704. [Google Scholar] [CrossRef]
- Meena, S.; Vidya, K.M.; Abhishek, D.T.; Ramyaa, L.T.S. Optimization and characterization of Alginic acid synthesized from a novel strain of Pseudomonas stutzeri. Biotechnol. Rep. 2020, 27, e00517. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Verma, C.; Singh, P.; Mukhopadhyay, S.; Gupta, A.; Gupta, B. Alginate based biomaterials for hemostatic applications: Innovations and developments. Int. J. Biol. Macromol. 2024, 262, 130771. [Google Scholar] [CrossRef]
- Orhotohwo, L.O.; Lucci, P.; Aquilanti, L.; Jaiswal, A.K.; Pacetti, D.; Jaiswal, D. Development and optimisation of sodium alginate and chitosan edible films with sea fennel by-product extract for active food packaging. LWT 2025, 226, 117960. [Google Scholar] [CrossRef]
- Bertagnolli, C.; Espindola, D.P.A.; Kleinübing, J.S.; Tasic, L.; Meuris Gurgel, C.D.S. Sargassum filipendula alginate from Brazil: Seasonal influence and characteristics. Carbohydr. Polym. 2014, 111, 619–623. [Google Scholar] [CrossRef]
- Xu, N.; Wang, Z.; Duan, R.; Zhang, J.; Liu, Y.; Fan, J.; Zhao, L.; Cong, W. Research Progress of Algin Digestion Technology. Technol. Innov. Appl. 2023, 13, 5–9+15. (In Chinese) [Google Scholar]
- Zhang, H.; Cheng, J.; Ao, Q. Preparation of Alginate-Based Biomaterials and Their Applications in Biomedicine. Mar. Drugs 2021, 19, 264. [Google Scholar] [CrossRef]
- Gomez, G.C.; Lambrecht, P.V.M.; Lozano, E.J.; Rinaudo, M.; Villar, M.A. Influence of the extraction–purification conditions on final properties of alginates obtained from brown algae (Macrocystis pyrifera). Int. J. Biol. Macromol. 2009, 44, 365–371. [Google Scholar] [CrossRef]
- Tiemi, M.N.; Rustici, L.C.; Camila, Y.; Sawada, N.N.S.; Freitas, M.L.C.; Zanini, B.C.C.; Guilherme, B.L. Optimal conditions for alkaline treatment of alginate extraction from the brown seaweed Sargassum cymosum C. Agardh by response surface methodology. Appl. Food Res. 2022, 2, 100141. [Google Scholar] [CrossRef]
- Queiroz, O.D.P.L.; Aroucha, M.M.E.; Santos, D.G.K.F.; Leite, R.H.D.L. Optimization of alginate extraction conditions from the brown seaweed Dictyota mertensii using a central composite design. Algal Res. 2024, 79, 103482. [Google Scholar] [CrossRef]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–489. [Google Scholar] [CrossRef]
- Leyton, A.; Pezoa-Conte, R.; Barriga, A.; Buschmann, A.H.; Mäki-Arvela, P.; Mikkola, J.P.; Lienqueo, M.E. Identification and efficient extraction method of phlorotannins from the brown seaweed Macrocystis pyrifera using an orthogonal experimental design. Algal Res. 2016, 16, 201–16208. [Google Scholar] [CrossRef]
- Leticia, M.; Mauro, G.; Ignacio, H.; Jorge, S.; Ramón, M. Impact of drying on the sodium alginate obtained after polyphenols ultrasound-assisted extraction from Ascophyllum nodosum seaweeds. Carbohydr. Polym. 2021, 272, 118455. [Google Scholar] [CrossRef] [PubMed]
- Rajan, K.D.; Lingwen, Z.; Hao, L.; Jie, L.; Shubing, Z. Purification and characterization of alginate extracted from Sargassum hemiphyllum and its antioxidant and wound healing efficacy. Food Biosci. 2024, 62, 105228. [Google Scholar] [CrossRef]
- Doh, H.; Dunno, D.K.; Whiteside, S.W. Preparation of novel seaweed nanocomposite film from brown seaweeds Laminaria japonica and Sargassum natans. Food Hydrocoll. 2020, 105, 105744. [Google Scholar] [CrossRef]
- Hylenne, B.; Amparo, L.; Antonio, M.; José, F.M. Overview of alginate extraction processes: Impact on alginate molecular structure and techno-functional properties. Trends Food Sci. Technol. 2023, 140, 104142. [Google Scholar] [CrossRef]
- Fernando, S.P.I.; Lee, W.; Han, J.E.; Ahn, G. Alginate-based nanomaterials: Fabrication techniques, properties, and applications. Chem. Eng. J. 2020, 391, 123823. [Google Scholar] [CrossRef]
- Khajouei, A.R.; Keramat, J.; Hamdami, N.; Ursu, A.V.; Delattre, C.; Laroche, C.; Gardarin, C.; Lecerf, D.; Desbrières, J.; Djelveh, G.; et al. Extraction and characterization of an alginate from the Iranian brown seaweed Nizimuddinia zanardini. Int. J. Biol. Macromol. 2018, 118, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.; Li, Z.; Chen, M.; Sun, Z.; Ling, Y.; Jiang, J.; Huang, C. Structural elucidation and protective role of a polysaccharide from Sargassum fusiforme on ameliorating learning and memory deficiencies in mice. Carbohydr. Polym. 2016, 139, 150–158. [Google Scholar] [CrossRef]
- Lim, S.J.; Wan Aida, W.M.; Maskat, M.Y.; Latip, J.; Badri, K.H.; Hassan, O.; Yamin, B.M. Characterisation of fucoidan extracted from Malaysian Sargassum binderi. Food Chem. 2016, 209, 267–273. [Google Scholar] [CrossRef]
- Sun, Q.; Li, Y.; Ni, L.; Li, Y.; Cui, Y.; Jiang, S.; Xie, E.; Du, J.; Deng, F.; Dong, C. Structural characterization and antiviral activity of two fucoidans from the brown algae Sargassum henslowianum. Carbohydr. Polym. 2020, 229, 115487. [Google Scholar] [CrossRef]
- Haixia, C.; Min, Z.; Bijun, X. Quantification of uronic acids in tea polysaccharide conjugates and their antioxidant properties. J. Agric. Food Chem. 2004, 52, 3333–3336. [Google Scholar] [CrossRef] [PubMed]
- Brovko, O.; Palamarchuk, I.; Gorshkova, N.; Chukhchin, D. Investigation of interpolymer complexes of fucoidan with sodium alginate in solutions and films. J. Appl. Phycol. 2025, 37, 539–551. [Google Scholar] [CrossRef]
- Eduardo, C.; Agustín, F.; Araceli, O. Sustainable exploitation of macroalgae species from Chilean coast: Characterization and food applications. Algal Res. 2021, 57, 102349. [Google Scholar] [CrossRef]
- Rashedy, S.H.; Abd El Hafez, M.S.M.; Dar, M.A.; Cotas, J.; Pereira, L. Evaluation and Characterization of Alginate Extracted from Brown Seaweed Collected in the Red Sea. Appl. Sci. 2021, 11, 6290. [Google Scholar] [CrossRef]
- Yamashita, C.; Moraes, F.C.I.; Ferreira, G.A.; Branco, C.C.Z.; Branco, L.G. Multi-response optimization of alginate bleaching technology extracted from brown seaweeds by an eco-friendly agent. Carbohydr. Polym. 2021, 251, 116992. [Google Scholar] [CrossRef]
- Taotao, L.; Jiachao, X.; Xiaoting, F. Comparison on the quality of sodium alginate extracted by calcificationprecipitation and acidulation precipitation. Sci. Technol. Food Ind. 2011, 32, 314. [Google Scholar]
- Mohammed, A.; Bissoon, R.; Bajnath, E.; Mohammed, K.; Lee, T.; Bissram, M.; John, N.; Jalsa, N.K.; Lee, K.Y.; Ward, K. Multistage extraction and purification of waste Sargassum natans to produce sodium alginate: An optimization approach. Carbohydr. Polym. 2018, 198, 109–118. [Google Scholar] [CrossRef]
- Mazumder, A.; Holdt, S.L.; De Francisci, D.; Morales, M.A.; Mishra, H.N.; Angelidaki, L. Extraction of alginate from Sargassum muticum: Process optimization and study of its functional activities. J. Appl. Phycol. 2016, 28, 625–3634. [Google Scholar] [CrossRef]
- Fawzy, A.M.; Gomaa, M.; Hifney, F.A.; Khayria, M.A.G. Optimization of alginate alkaline extraction technology from Sargassum latifolium and its potential antioxidant and emulsifying properties. Carbohydr. Polym. 2017, 157, 1903–1912. [Google Scholar] [CrossRef]
- Fertah, M.; Belfkira, A.; Dahmane, M.E.; Taourirte, M.; Brouillette, F. Extraction and characterization of sodiumalginate from Moroccan Laminaria digitata brown seaweed. Arab. J. Chem. 2017, 10, S3707–S3714. [Google Scholar] [CrossRef]
- Craigie, J.S.; Morris, E.R.; Rees, D.A.; Thom, D. Alginate block structure in phaeophyceae from Nova Scotia: Variation with species, environment and tissue type. Carbohydr. Polym. 1984, 4, 237–252. [Google Scholar] [CrossRef]
- Eslami, Z.; Elkoun, S.; Ahyaya, L.; Robert, M. Influence of M/G ratio and molecular weight on water sorption and glass transition behavior of alginate films. Int. J. Biol. Macromol. 2025, 315, 144639. [Google Scholar] [CrossRef]
- Roya, K.A.; Javad, K.; Nasser, H.; Ursu, A.V.; Cedric, D.; Christine, G.; Didier, L.; Jacques, D.; Gholamreza, D.; Philippe, M. Effect of high voltage electrode discharge on the physicochemical characteristics of alginate extracted from an Iranian brown seaweed (Nizimuddinia zanardini). Algal Res. 2021, 56, 102326. [Google Scholar] [CrossRef]
- Jiao, W.; Chen, W.; Mei, Y.; Yun, Y.; Wang, B.; Zhong, Q.; Chen, H.; Chen, W. Effects of Molecular Weight and Guluronic Acid/Mannuronic Acid Ratio on the Rheological Behavior and Stabilizing Property of Sodium Alginate. Molecules 2019, 24, 4374. [Google Scholar] [CrossRef]
- Jones, C.T.; Bakshani, C.; Lelenaite, I.; Mravec, J.; Kračun, S.K.; Pearson, J.; Wilcox, M.D.; Hardouin, K.; Kridi, S.; Hervé, C.; et al. Spatiotemporal regulation of alginate sub-structures at multiple scales revealed by monoclonal antibodies. Cell Surf. 2025, 13, 100136. [Google Scholar] [CrossRef]
- Khan, A.A.M.; Hasan, M.M.; Hossain, K.M.; Adhikery, D.; Hakim, M.; Mohanta, L.C.; Sharif, A.S.M.; Sarker, A.K. Extraction and characteristic properties analyses of sodium alginate derived from the Sargassum oligocystum brown seaweed alga of the Bay of Bengal. Next Mater. 2025, 6, 100417. [Google Scholar] [CrossRef]
- Mohammed, A.; Rivers, A.; Stuckey, C.D.; Ward, K. Alginate extraction from Sargassum seaweed in the Caribbean region: Optimization using response surface methodology. Carbohydr. Polym. 2020, 245, 116419. [Google Scholar] [CrossRef]
- Martínez, L.M.F.; Torres, M.L.; Castro, S.D.; León, F.C.; Plancarte, C.H.; Barrita, P.F.D.J.C. Sodium alginate from Playa del Carmen Sargassum: Extraction, characterization, and rheological analysis of solutions and hydrogels. Int. J. Biol. Macromol. 2025, 317, 144803. [Google Scholar] [CrossRef]
- Zahira, B.; Soukaina, K.; Samir, A.E.; Chaimaa, K.; Fouad, B.; Charafeddine, J.; Abdeltif, R.; Brahim, S.; Vitor, V. AbdeltifIsolation and FTIR-ATR and 1H NMR Characterization of Alginates from the Main Alginophyte Species of the Atlantic Coast of Morocco. Molecules 2020, 25, 335–4335. [Google Scholar]
- Kyle, J.; Jerome, R.; Leonie, B.; Lalehvash, M. Technology comparison for sequential extraction of fucoidan and sodium alginate from Ascophyllum nodosum using a glycerol and choline chloride solvent. Clean. Eng. Technol. 2024, 18, 100707. [Google Scholar]
- Noelia, M.S.; Bernard, B.; Ian, S. Sodium alginate and alginic acid as pharmaceutical excipients for tablet formulation: Structure-function relationship. Carbohydr. Polym. 2021, 270, 118399. [Google Scholar] [CrossRef] [PubMed]
- Gromovykh, I.T.; Tarasova, A.I.; Zefirov, V.V.; Gulin, A.A.; Ivanenko, I.P.; Molchanov, V.S.; Kharitonova, E.P.; Kiselyova, O.I. Composites of bacterial cellulose and alginate produced in situ: The impact of viscosity and temperature on the microscale morphology. Carbohydr. Polym. 2025, 357, 123495. [Google Scholar] [CrossRef] [PubMed]
- Andrea, D.; Silvia, V.; Alloisio, M.; Castellano, M. Rheological properties of sodium alginate solutions in the presence of added salt: An application of Kulicke equation. Rheol. Acta 2020, 59, 365–374. [Google Scholar]
- Kenny, M.H.; Reynolds, M.C.; Vaquero, G.M.; Feeney, L.E. Keeping an eye on alginate: Innovations and opportunities for sustainable production and diverse applications. Carbohydr. Polym. 2025, 366, 123902. [Google Scholar] [CrossRef] [PubMed]
- GB 1886.243-2016; National Food Safety Standard for Food Additive: Sodium Alginate. Standardization Administration of the People’s Republic of China: Beijing, China, 2016.

| Factor | ||||
|---|---|---|---|---|
| A | B | C | D | |
| Level | Alkali Concentration | Time | Material-to-Liquid Ratio | Temperature |
| (%) | (h) | (g/mL) | (°C) | |
| 1 | 2 | 3 | 1:30 | 50 |
| 2 | 3 | 4 | 1:40 | 60 |
| 3 | 4 | 5 | 1:50 | 70 |
| Factor | Metric | |||||
|---|---|---|---|---|---|---|
| A | B | C | D | |||
| Number | Alkali Concentration | Time | Material-Liquid Ratio | Temperature | Extraction Yield | |
| (%) | (h) | (g/mL) | (°C) | (%) | ||
| 1 | 2 | 3 | 1:30 | 50 | 36.87 | |
| 2 | 2 | 4 | 1:40 | 60 | 43.03 | |
| 3 | 2 | 5 | 1:50 | 70 | 37.91 | |
| 4 | 3 | 3 | 1:40 | 70 | 37.30 | |
| 5 | 3 | 4 | 1:50 | 50 | 32.72 | |
| 6 | 3 | 5 | 1:30 | 60 | 33.40 | |
| 7 | 4 | 3 | 1:50 | 60 | 41.89 | |
| 8 | 4 | 4 | 1:30 | 70 | 40.93 | |
| 9 | 4 | 5 | 1:40 | 50 | 33.61 | |
| K1 | 117.81 | 116.06 | 111.20 | 103.20 | ||
| K2 | 103.42 | 116.68 | 113.94 | 118.32 | ||
| K3 | 116.43 | 104.92 | 112.52 | 116.14 | ||
| k1 | 39.27 | 38.69 | 37.07 | 34.40 | ||
| k2 | 34.47 | 38.89 | 37.98 | 39.44 | ||
| k3 | 38.81 | 34.97 | 37.51 | 38.71 | ||
| R | 4.80 | 3.92 | 0.91 | 5.04 | ||
| Optimal solution | A1 | B2 | C2 | D2 | ||
| Sample Name | Part of LN | Sedimentation Method | Extraction Yield % |
|---|---|---|---|
| Alg-H-Ethanol | Holdfast | Ethanol | 40.62 ± 0.52 de |
| Alg-H-CaCl2 | Holdfast | Calcium | 38.50 ± 0.61 h |
| Alg-H-HCl | Holdfast | Acid | 39.43 ± 0.64 g |
| Alg-S-Ethanol | Stipe | Ethanol | 46.02 ± 0.59 a |
| Alg-S-CaCl2 | Stipe | Calcium | 41.23 ± 0.77 d |
| Alg-S-HCl | Stipe | Acid | 43.96 ± 0.39 b |
| Alg-B-Ethanol | Blade | Ethanol | 44.77 ± 0.58 b |
| Alg-B-CaCl2 | Blade | Calcium | 39.54 ± 0.63 fg |
| Alg-B-HCl | Blade | Acid | 40.08 ± 0.42 efg |
| Alg-W-Ethanol | Whole Seaweed | Ethanol | 42.96 ± 0.37 c |
| Alg-W-CaCl2 | Whole Seaweed | Calcium | 39.84 ± 0.49 efg |
| Alg-W-HCl | Whole Seaweed | Acid | 40.51 ± 0.47 def |
| Sample Name | Color | State | pH | Moisture (%) | Transmittance (%) |
|---|---|---|---|---|---|
| Alg-B-HCl | Light yellow brown | Flocculent | 7.49 ± 0.15 cde | 6.48 ± 0.20 bcd | 98.64 ± 0.22 b |
| Alg-S-HCl | Light yellow brown | Flocculent | 7.53 ± 0.10 cd | 5.90 ± 0.05 ef | 98.75 ± 0.10 b |
| Alg-H-HCl | Light yellow brown | Flocculent | 7.86 ± 0.11 a | 5.91 ± 0.28 ef | 98.71 ± 0.07 b |
| Alg-W-HCl | Light yellow brown | Flocculent | 7.33 ± 0.12 e | 6.65 ± 0.36 abcd | 99.71 ± 0.16 a |
| Alg-B-CaCl2 | Light yellow brown | Flocculent | 7.64 ± 0.07 bc | 7.10 ± 0.29 a | 99.55 ± 0.23 a |
| Alg-S-CaCl2 | Light yellow brown | Flocculent | 7.88 ± 0.06 a | 6.66 ± 0.27 abcd | 99.61 ± 0.10 a |
| Alg-H-CaCl2 | Light yellow brown | Flocculent | 7.52 ± 0.10 cd | 6.81 ± 0.15 ab | 99.49 ± 0.18 a |
| Alg-W-CaCl2 | Light yellow brown | Flocculent | 7.73 ± 0.11 ab | 5.54 ± 0.30 f | 99.77 ± 0.19 a |
| Alg-B-Ethanol | Brown | Flocculent | 7.87 ± 0.04 a | 6.30 ± 0.04 cde | 97.57 ± 0.24 c |
| Alg-S-Ethanol | Brown | Flocculent | 7.36 ± 0.05 de | 6.73 ± 0.37 abc | 98.54 ± 0.09 b |
| Alg-H-Ethanol | Brown | Flocculent | 7.39 ± 0.05 de | 5.79 ± 0.21 f | 98.56 ± 0.14 b |
| Alg-W-Ethanol | Brown | Flocculent | 7.86 ± 0.10 a | 6.24 ± 0.15 de | 97.61 ± 0.12 c |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, Y.; Dong, M.; Lei, X.; Xu, Z.; Sun, J.; Zhao, Y.; Ma, Y.; Zhou, H.; Liu, S.; He, Y.; et al. Comparison of Flocculation Methods for Sodium Alginate and Characterization of Its Structure and Properties. Foods 2025, 14, 2970. https://doi.org/10.3390/foods14172970
Shi Y, Dong M, Lei X, Xu Z, Sun J, Zhao Y, Ma Y, Zhou H, Liu S, He Y, et al. Comparison of Flocculation Methods for Sodium Alginate and Characterization of Its Structure and Properties. Foods. 2025; 14(17):2970. https://doi.org/10.3390/foods14172970
Chicago/Turabian StyleShi, Yuxin, Mingna Dong, Xuhui Lei, Zhiying Xu, Jiyan Sun, Yingying Zhao, Yichao Ma, Hui Zhou, Shu Liu, Yunhai He, and et al. 2025. "Comparison of Flocculation Methods for Sodium Alginate and Characterization of Its Structure and Properties" Foods 14, no. 17: 2970. https://doi.org/10.3390/foods14172970
APA StyleShi, Y., Dong, M., Lei, X., Xu, Z., Sun, J., Zhao, Y., Ma, Y., Zhou, H., Liu, S., He, Y., Wang, Q., & Ren, D. (2025). Comparison of Flocculation Methods for Sodium Alginate and Characterization of Its Structure and Properties. Foods, 14(17), 2970. https://doi.org/10.3390/foods14172970






