Paper-Based Microfluidic Chips for Food Hazard Factor Detection: Fabrication, Modification, and Application
Abstract
1. Introduction
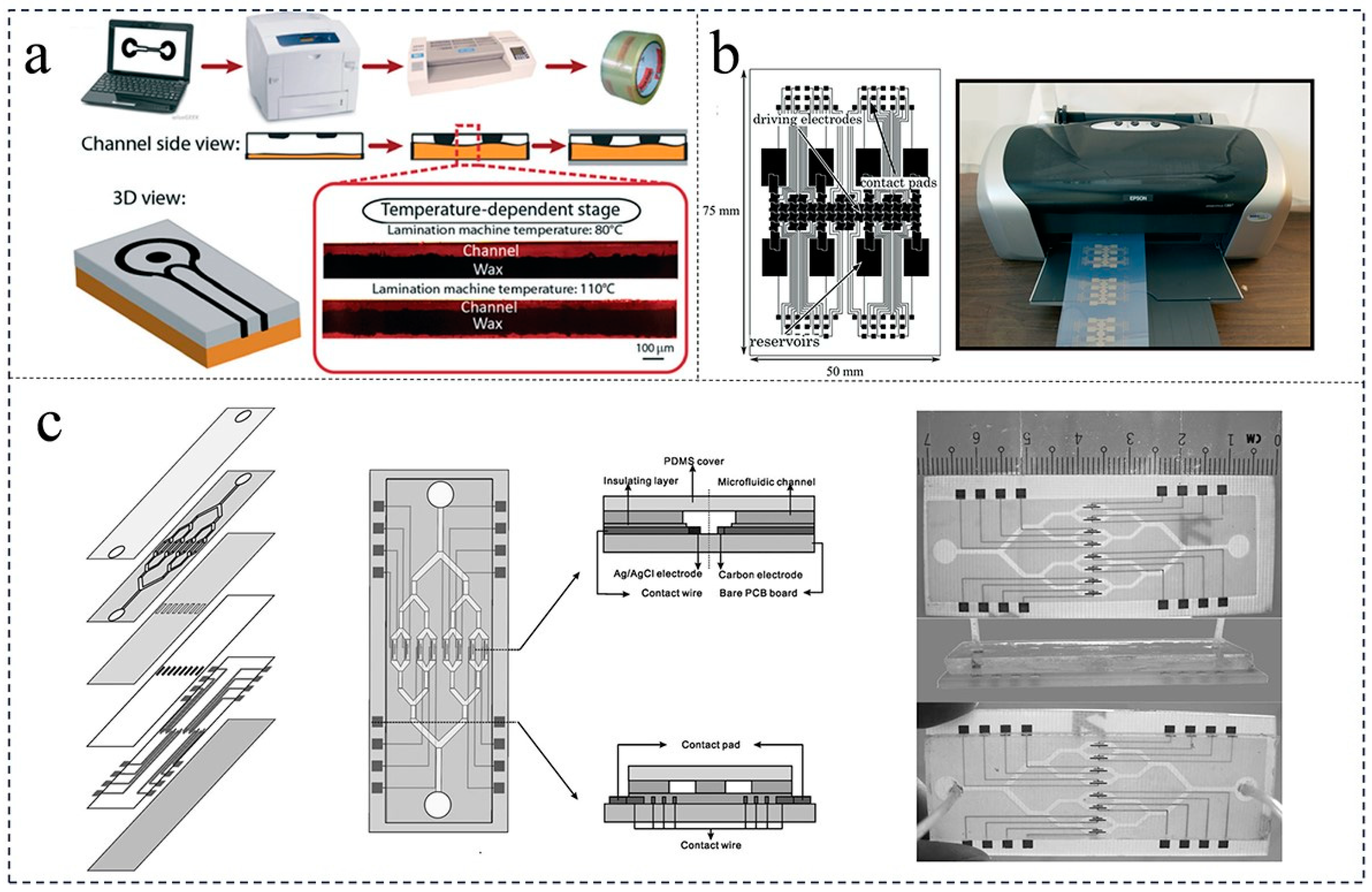
2. Fabrication Strategy of Paper-Based Microfluidic Chips
2.1. Wax Printing Method
2.2. Etching Method

2.3. Cutting Method
3. Surface Functionalization Modification of Paper-Based Chips
3.1. Electrostatic Adsorption Method
3.2. Covalent Bond Modification Method
3.3. Embedding Method
4. Analysis Method of Paper-Based Microfluidic Chips
4.1. Electrochemical Method
4.2. Optical Analysis
5. Application of Integrated Paper-Based Microfluidic Chips for Rapid Food Safety Detection

| Target | Chip Material | Detection Method | Limits of Detection (LOD) | Real Sample Application | Property | Reference |
|---|---|---|---|---|---|---|
| nitrite | paper | electrochemical analysis | 1.06 μM | — | low cost, simple, and reproducible | [46] |
| nitrite | paper | colorimetric analysis | 19.2 mg/kg | pork | high selectivity, sensitivity, environment-friendly, and suitable for on-site measurement | [60] |
| glucose | paper | electrochemical analysis | 0.12 μM | — | high sensitivity and good selectivity | [61] |
| Ascorbic acid | paper | colorimetric analysis | 0.406 μmol/L | — | reliable and sensitive | [64] |
| heavy metal ions | paper | fluorescence | 0.035 µg/L, 0.056 µg/L | water | good linear relationship | [65] |
| 2,4-dichloro phenoxy acetic acid | paper | fluorescence | 90 nM | soybean sprouts | fast response, high precision, practical availability, and good reproducibility | [66] |
| E. coli and Enterococcus | paper | electrochemical and colorimetric analysis | 81 μΜ and 119 μM | — | fast response, and high sensitivity | [67] |
| E. coli | paper | fluorescence and colorimetric analysis | 100 CFU/mL and 44 CFU/mL | — | rapid, portable, and sensitive | [68] |
| methyl parathion | paper | SERE | 0.011 μg/cm2 | apple | low cost and on-site inspection | [69] |
| pesticide thiram | paper | SERE | 0.024 ppm, 600 ng/cm2 | water and apple juice, apple peel | high sensitivity and environment-friendly | [70] |
| peanut allergen Ara h1 | paper | electrochemical analysis | 11.8 ng/mL | cookies, milk, and bread | specificity, sensitivity, and good stability | [74] |
| sulfite | paper | colorimetric analysis | 78 μM | wines | environment-friendly and high sensitivity | [79] |
| allergens | paper | colorimetric analysis | 0.246 KUA/L | — | efficient, accurate, and sensitive | [80] |
| glucose | paper | colorimetric analysis | 3.12 mM | fruits | low cost and highly sensitive | [82] |
| paraoxon | paper | colorimetric analysis | 1 μM | milk and apple juice | environment-friendly, and high selectivity | [83] |
| trichlorfon | paper | spectrophotometric | 1.65 mg/mL | vegetable | rapid and sensitive | [84] |
| trichlorfon | paper | colorimetric analysis | 0.0406 mg/L | — | repeatability, specificity, good stability | [85] |
| Salmonella | paper | electrochemical analysis | 5 cells/mL | apple juice | simple and accessible | [87] |
| Hg2+, Ag+ and NEO | paper | fluorescence | 121 nM, 47 nM, 153 nM | — | low cost, simple, and multiplexed | [88] |
| glyphosate | paper | fluorescence | 4.19 nM | — | rapid, on-site inspection | [89] |
| Pb | paper | fluorescence | 18.3 nM | — | rapid, on-site inspection, and high selectivity | [90] |
6. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hermann, C.A.; Duerkop, A.; Baeumner, A.J. Food safety analysis enabled through biological and synthetic materials: A critical review of current trends. Anal. Chem. 2018, 91, 569–587. [Google Scholar] [CrossRef] [PubMed]
- Jiao, K.; Liu, G.; Zuo, X.; Li, J.; Fan, C. Bioassay development for public health emergency. Innovation 2022, 3, 100331. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Chen, H.; Sun, B. Recent progress in food flavor analysis using gas chromatography–ion mobility spectrometry (GC–IMS). Food Chem. 2020, 315, 126158. [Google Scholar] [CrossRef]
- Nie, Q.; Nie, S. High-performance liquid chromatography for food quality evaluation. In Evaluation Technologies for Food Quality; Woodhead Publishing: Sawston, UK, 2019. [Google Scholar]
- Wang, X.; Wang, S.; Cai, Z. The latest developments and applications of mass spectrometry in food-safety and quality analysis. TrAC Trends Anal. Chem. 2013, 52, 170–185. [Google Scholar] [CrossRef]
- De Medici, D.; Kuchta, T.; Knutsson, R.; Angelov, A.; Auricchio, B.; Barbanera, M.; Diaz-Amigo, C.; Fiore, A.; Kudirkiene, E.; Hohl, A. Rapid methods for quality assurance of foods: The next decade with polymerase chain reaction (PCR)-based food monitoring. Food Anal. Methods 2015, 8, 255–271. [Google Scholar] [CrossRef]
- Salihah, N.T.; Hossain, M.M.; Lubis, H.; Ahmed, M.U. Trends and advances in food analysis by real-time polymerase chain reaction. J. Food Sci. Technol. 2016, 53, 2196–2209. [Google Scholar] [CrossRef]
- Su, W.; Gao, X.; Jiang, L.; Qin, J. Microfluidic platform towards point-of-care diagnostics in infectious diseases. J. Chromatogr. A 2015, 1377, 13–26. [Google Scholar] [CrossRef]
- Su, W.; Liang, D.; Tan, M. Microfluidic strategies for sample separation and rapid detection of food allergens. Trends Food Sci. Technol. 2021, 110, 213–225. [Google Scholar] [CrossRef]
- Su, W.; Liang, D.; Tan, M. Nucleic acid-based detection for foodborne virus utilizing microfluidic systems. Trends Food Sci. Technol. 2021, 113, 97–109. [Google Scholar] [CrossRef]
- Cai, Z.; Fan, S.; Sun, X.; Mo, X.; Yang, G. Novel microfluidic device for measurable residual disease detection in acute leukemia. Innovation 2023, 4, 100408. [Google Scholar] [CrossRef]
- Kfla, B.; Ag, A.; Chh, C. Paper/polymer composited microfluidic platform for screening cell viability and protein expression under a chemical gradient environment. Talanta 2019, 205, 120124. [Google Scholar]
- Cate, D.M.; Adkins, J.A.; Mettakoonpitak, J.; Henry, C.S. Recent Developments in Paper-Based Microfluidic Devices. Anal. Chem. 2015, 87, 19–41. [Google Scholar] [CrossRef] [PubMed]
- Ys, A.; Bsbb, C.; Mgk, B. Lab-on-paper for all-in-one molecular diagnostics (LAMDA) of zika, dengue, and chikungunya virus from human serum. Biosens. Bioelectron. 2020, 165, 112400. [Google Scholar]
- Al-Ghanim, S.M.; Tamimi, A.F.; Owiedi, A.A.; Al-Qudah, A. Anesthesia and electrocorticography for epilepsy surgery: A jordanian experience. Middle East J. Anaesthesiol. 2009, 30, 31–37. [Google Scholar]
- Chaiyo, S.; Apiluk, A.; Siangproh, W.; Chailapakul, O. High sensitivity and specificity simultaneous determination of lead, cadmium and copper using μPAD with dual electrochemical and colorimetric detection. Sens. Actuators B Chem. 2016, 233, 540–549. [Google Scholar] [CrossRef]
- Lee, J.; Yoo, Y.K.; Lee, D.; Kim, C.; Kim, K.H.; Lee, S.; Kwak, S.; Kang, J.Y.; Kim, H.; Yoon, D.S. Origami paper-based sample preconcentration using sequentially driven ion concentration polarization. Lab Chip 2021, 21, 867–874. [Google Scholar] [CrossRef]
- Gao, H.; Yan, C.; Wu, W.; Li, J. Application of Microfluidic Chip Technology in Food Safety Sensing. Sensors 2020, 20, 1792. [Google Scholar] [CrossRef]
- Lu, Y.; Shi, W.; Qin, J.; Lin, B. Fabrication and characterization of paper-based microfluidics prepared in nitrocellulose membrane by wax printing. Anal. Chem. 2010, 82, 329–335. [Google Scholar] [CrossRef]
- Jain, P.; Chakma, B.; Patra, S.; Goswami, P. Potential Biomarkers and Their Applications for Rapid and Reliable Detection of Malaria. BioMed Res. Int. 2014, 2014, 852645. [Google Scholar] [CrossRef]
- Songjaroen, T.; Dungchai, W.; Chailapakul, O.; Henry, C.S.; Laiwattanapaisal, W. Blood separation on microfluidic paper-based analytical devices. Lab Chip 2012, 12, 3392. [Google Scholar] [CrossRef]
- Olkkonen, J.; Lehtinen, K.; Erho, T. Flexographically printed fluidic structures in paper. Anal. Chem. 2010, 82, 10246–10250. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Cordero, J.L.; Kurzbuch, D.; Benito-Lopez, F.; Diamond, D.; Lee, L.P.; Ricco, A.J. Optically addressable single-use microfluidic valves by laser printer lithography. Lab Chip 2010, 10, 2680–2687. [Google Scholar] [CrossRef] [PubMed]
- Dixon, C.; Ng, A.H.; Fobel, R.; Miltenburg, M.B.; Wheeler, A.R. An inkjet printed, roll-coated digital microfluidic device for inexpensive, miniaturized diagnostic assays. Lab Chip 2016, 16, 4560–4568. [Google Scholar] [CrossRef]
- Bsoul, A.; Sheng, P.; Cretu, E.; Stoeber, B.; Walus, K. Design, microfabrication, and characterization of a moulded PDMS/SU-8 inkjet dispenser for a Lab-on-a-Printer platform technology with disposable microfluidic chip. Lab Chip 2016, 16, 3351–3361. [Google Scholar] [CrossRef]
- Pearce, J.M.; Anzalone, N.C.; Heldt, C.L. Open-Source Wax RepRap 3-D Printer for Rapid Prototyping Paper-Based Microfluidics. Jala J. Assoc. Lab. Autom. 2016, 21, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, T.; Bercovici, M. 1000-fold sample focusing on paper-based microfluidic devices. Lab Chip 2014, 14, 4465–4474. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.W.; Li, Z.; Jie, H.; Kadri, N.A.; Feng, X.; Fei, L.; Pingguan-Murphy, B. Fabrication of dielectrophoretic microfluidic chips using a facile screen-printing technique for microparticle trapping. J. Micromech. Microeng. 2015, 25, 105015. [Google Scholar] [CrossRef][Green Version]
- Bhattacharjee, N.; Urrios, A.; Kang, S.; Folch, A. The upcoming 3D-printing revolution in microfluidics. Lab Chip 2016, 16, 1720–1742. [Google Scholar] [CrossRef]
- Gul, J.Z.; Sajid, M.; Rehman, M.M.; Siddiqui, G.U.; Shah, I.; Kim, K.-H.; Lee, J.-W.; Choi, K.H. 3D printing for soft robotics—A review. Sci. Technol. Adv. Mater. 2018, 19, 243–262. [Google Scholar] [CrossRef]
- Van-Thai, T.; Wei, Y.; Wei, L.; Yang, H.; Du, H. Preparing of Interdigitated Microelectrode Arrays for AC Electrokinetic Devices Using Inkjet Printing of Silver Nanoparticles Ink. Micromachines 2017, 8, 106. [Google Scholar]
- Song, W.; Tang, Y.; Qian, C.; Kim, B.J.; Liao, Y.; Yu, D.-G. Electrospinning spinneret: A bridge between the visible world and the invisible nanostructures. Innovation 2023, 4, 100381. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Li, C.-M.; Zhang, Y.-F.; Cao, X.-D.; Gan, Y. Screen-printed microfluidic device for electrochemical immunoassay. Lab Chip 2007, 7, 1752–1758. [Google Scholar] [CrossRef] [PubMed]
- Gaal, G.; Mendes, M.; de Almeida, T.P.; Piazzetta, M.H.; Gobbi, Â.L.; Riul, A., Jr.; Rodrigues, V. Simplified fabrication of integrated microfluidic devices using fused deposition modeling 3D printing. Sens. Actuators B Chem. 2017, 242, 35–40. [Google Scholar] [CrossRef]
- Liu, Y.-Q.; Xiong, Z.; Zhang, Y.-L.; Liu, H. Breakthroughs in projection-enabled additive manufacturing: From novel strategies to cutting-edge applications. Innovation 2023, 4, 100395. [Google Scholar] [CrossRef] [PubMed]
- Kataoka, E.M.; Murer, R.C.; Santos, J.M.; Carvalho, R.M.; Eberlin, M.N.; Augusto, F.; Poppi, R.J.; Gobbi, A.L.; Hantao, L.W. Simple, expendable, 3D-printed microfluidic systems for sample preparation of petroleum. Anal. Chem. 2017, 89, 3460–3467. [Google Scholar] [CrossRef]
- Raj, N.; Breedveld, V.; Hess, D. Fabrication of fully enclosed paper microfluidic devices using plasma deposition and etching. Lab Chip 2019, 19, 3337–3343. [Google Scholar] [CrossRef]
- Si, Z.; Li, J.; Ma, L.; Cai, D.; Li, S.; Baeyens, J.; Degrève, J.; Nie, J.; Tan, T.; Qin, P. The ultrafast and continuous fabrication of a polydimethylsiloxane membrane by ultraviolet-induced polymerization. Angew. Chem. 2019, 131, 17335–17339. [Google Scholar] [CrossRef]
- Lin, D.; Li, B.; Qi, J.; Ji, X.; Yang, S.; Wang, W.; Chen, L. Low cost fabrication of microfluidic paper-based analytical devices with water-based polyurethane acrylate and their application for bacterial detection. Sens. Actuators B Chem. 2020, 303, 127213. [Google Scholar] [CrossRef]
- Abe, K.; Suzuki, K.; Citterio, D. Inkjet-Printed Microfluidic Multianalyte Chemical Sensing Paper. Anal. Chem. 2008, 80, 6928–6934. [Google Scholar] [CrossRef]
- Shangguan, J.W.; Liu, Y.; Pan, J.; Xu, B.Y.; Chen, H.Y. Microfluidic PDMS on paper (POP) devices. Lab Chip 2016, 17, 120. [Google Scholar] [CrossRef]
- Dungchai, W.; Chailapakul, O.; Henry, C.S. A low-cost, simple, and rapid fabrication method for paper-based microfluidics using wax screen-printing. Analyst 2010, 136, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Ding, Y.; Xie, D.; Hu, D.; Li, P.; Li, X.; Xue, W.; Jin, L.; Song, B. Design, synthesis, and antiviral activity of novel rutin derivatives containing 1, 4-pentadien-3-one moiety. Eur. J. Med. Chem. 2015, 92, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Kao, P.K.; Hsu, C.C. Battery-Operated, Portable, and Flexible Air Microplasma Generation Device for Fabrication of Microfluidic Paper-Based Analytical Devices on Demand. Anal. Chem. 2014, 86, 8757. [Google Scholar] [CrossRef] [PubMed]
- Renault, C.; Anderson, M.J.; Crooks, R.M. Electrochemistry in hollow-channel paper analytical devices. J. Am. Chem. Soc. 2014, 136, 4616–4623. [Google Scholar] [CrossRef]
- Driesche, S.; Lucklum, F.; Bunge, F.; Vellekoop, M.J. 3D Printing Solutions for Microfluidic Chip-to-World Connections. Micromachines 2018, 9, 71. [Google Scholar] [CrossRef]
- Wang, Z.K.; Zheng, H.Y.; Lim, R.; Wang, Z.F.; Lam, Y.C. Improving surface smoothness of laser-fabricated microchannels for microfluidic application. J. Micromech. Microeng. 2011, 21, 095008. [Google Scholar] [CrossRef]
- Khatri, V.; Halász, K.; Trandafilović, L.V.; Dimitrijević-Branković, S.; Mohanty, P.; Djoković, V.; Csóka, L. ZnO-modified cellulose fiber sheets for antibody immobilization. Carbohydr. Polym 2014, 109, 139–147. [Google Scholar] [CrossRef]
- Elsharkawy, M.; Schutzius, T.M.; Megaridis, C.M. Inkjet patterned superhydrophobic paper for open-air surface microfluidic devices. Lab Chip 2014, 14, 1168–1175. [Google Scholar] [CrossRef]
- Yetisen, A.K.; Akram, M.S.; Lowe, C.R. Paper-based microfluidic point-of-care diagnostic devices. Lab Chip 2013, 13, 2210–2251. [Google Scholar] [CrossRef]
- Ju, Q.; Noor, M.O.; Krull, U.J. Paper-based biodetection using luminescent nanoparticles. Analyst 2016, 141, 2838–2860. [Google Scholar] [CrossRef]
- Ahmed, S.; Bui, M.P.N.; Abbas, A. Paper-based chemical and biological sensors: Engineering aspects. Biosens. Bioelectron. Int. J. Prof. Involv. Res. Technol. Appl. Biosensers Relat. Devices 2016, 77, 249–263. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, C.; Tomar, L.K.; Singh, H. Surface modification of cellulose filter paper by glycidyl methacrylate grafting for biomolecule immobilization: Influence of grafting parameters and urease immobilization. J. Appl. Polym. Ence 2010, 111, 1381–1390. [Google Scholar] [CrossRef]
- Sun, X.; Jian, Y.; Wang, H.; Ge, S.; Yan, M.; Yu, J. Ultrasensitive microfluidic paper-based electrochemical biosensor based on molecularly imprinted film and boronate affinity sandwich assay for glycoprotein detection. ACS Appl. Mater. Interfaces 2019, 11, 16198–16206. [Google Scholar] [CrossRef] [PubMed]
- Hossain, S.; Brennan, J.D. β-Galactosidase-based colorimetric paper sensor for determination of heavy metals. Anal. Chem. 2011, 83, 8772–8778. [Google Scholar] [CrossRef] [PubMed]
- Luckham, R.E.; Brennan, J.D. Bioactive paper dipstick sensors for acetylcholinesterase inhibitors based on sol–gel/enzyme/gold nanoparticle composites. Analyst 2010, 135, 2028–2035. [Google Scholar] [CrossRef]
- Wang, J.; Bowie, D.; Zhang, X.; Filipe, C.; Pelton, R.; Brennan, J.D. Morphology and Entrapped Enzyme Performance in Inkjet-Printed Sol–Gel Coatings on Paper. Chem. Mater. 2014, 26, 1941–1947. [Google Scholar] [CrossRef]
- Wang, P.; Wang, M.; Zhou, F.; Yang, G.; Qu, L.; Miao, X. Development of a paper-based, inexpensive, and disposable electrochemical sensing platform for nitrite detection. Electrochem. Commun. 2017, 81, 74–78. [Google Scholar] [CrossRef]
- Guadarrama-Fernández, L.; Novell, M.; Blondeau, P.; Andrade, F.J. A disposable, simple, fast and low-cost paper-based biosensor and its application to the determination of glucose in commercial orange juices. Food Chem. 2018, 265, 64–69. [Google Scholar] [CrossRef]
- Trofimchuk, E.; Hu, Y.; Nilghaz, A.; Hua, M.Z.; Lu, X. Development of paper-based microfluidic device for the determination of nitrite in meat. Food Chem. 2020, 316, 126396. [Google Scholar] [CrossRef]
- Zhang, Z.; Ma, X.; Jia, M.; Li, B.; Rong, J.; Yang, X. Deposition of CdTe quantum dots on microfluidic paper chips for rapid fluorescence detection of pesticide 2, 4-D. Analyst 2019, 144, 1282–1291. [Google Scholar] [CrossRef]
- Lopez-Ruiz, N.; Curto, V.F.; Erenas, M.M.; Benito-Lopez, F.; Diamond, D.; Palma, A.J.; Capitan-Vallvey, L.F. Smartphone-based simultaneous pH and nitrite colorimetric determination for paper microfluidic devices. Anal. Chem. 2014, 86, 9554–9562. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Chen, Q.; Kutsanedzie, F.Y.; Yang, M.; Ouyang, Q.; Jiang, H. Highly sensitive and label-free determination of thiram residue using surface-enhanced Raman spectroscopy (SERS) coupled with paper-based microfluidics. Anal. Methods 2017, 9, 6186–6193. [Google Scholar] [CrossRef]
- Guan, H.; Du, S.; Han, B.; Zhang, Q.; Wang, D. A rapid and sensitive smartphone colorimetric sensor for detection of ascorbic acid in food using the nanozyme paper-based microfluidic chip. LWT-Food Sci. Technol. 2023, 184, 115043. [Google Scholar] [CrossRef]
- Santangelo, M.F.; Shtepliuk, I.; Filippini, D.; Puglisi, D.; Vagin, M.; Yakimova, R.; Eriksson, J. Epitaxial Graphene Sensors Combined with 3D-Printed Microfluidic Chip for Heavy Metals Detection. Sensors 2019, 19, 2393. [Google Scholar] [CrossRef]
- Zhang, Z.; Ma, X.; Li, B.; Zhao, J.; Qi, J.; Hao, G.; Jianhui, R.; Yang, X. Fluorescence detection of 2, 4-dichlorophenoxyacetic acid by ratiometric fluorescence imaging on paper-based microfluidic chips. Analyst 2020, 145, 963–974. [Google Scholar] [CrossRef] [PubMed]
- Adkins, J.A.; Boehle, K.; Friend, C.; Chamberlain, B.; Bisha, B.; Henry, C.S. Colorimetric and Electrochemical Bacteria Detection Using Printed Paper- and Transparency-Based Analytic Devices. Anal. Chem. 2017, 89, 3613–3621. [Google Scholar] [CrossRef]
- Wang, C.; Gao, X.; Wang, S.; Liu, Y. A smartphone-integrated paper sensing system for fluorescent and colorimetric dual-channel detection of foodborne pathogenic bacteria. Anal. Bioanal. Chem. 2020, 412, 611–620. [Google Scholar] [CrossRef]
- Xie, J.; Li, L.; Khan, I.M.; Wang, Z.; Ma, X. Flexible paper-based SERS substrate strategy for rapid detection of methyl parathion on the surface of fruit. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 231, 118104. [Google Scholar] [CrossRef]
- Martins, N.C.; Fateixa, S.; Fernandes, T.; Nogueira, H.I.; Trindade, T. Inkjet printing of Ag and polystyrene nanoparticle emulsions for the one-step fabrication of hydrophobic paper-based surface-enhanced Raman scattering substrates. ACS Appl. Nano Mater. 2021, 4, 4484–4495. [Google Scholar] [CrossRef]
- Chen, Q.; Cheung, K.; Kong, S.; Zhou, J.; Kwan, Y.; Wong, C.; Ho, H. An integrated lab-on-a-disc for automated cell-based allergen screening bioassays. Talanta 2012, 97, 48–54. [Google Scholar] [CrossRef]
- Berti, F.; Laschi, S.; Palchetti, I.; Rossier, J.S.; Reymond, F.; Mascini, M.; Marrazza, G. Microfluidic-based electrochemical genosensor coupled to magnetic beads for hybridization detection. Talanta 2009, 77, 971–978. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-Y.; Huang, C.-H.; Park, J.; Pathania, D.; Castro, C.M.; Fasano, A.; Weissleder, R.; Lee, H. Integrated magneto-chemical sensor for on-site food allergen detection. ACS Nano 2017, 11, 10062–10069. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Han, X.; Chen, S.; Yang, J.; Wang, Y.; Li, H.; Wang, S. Paper-based microfluidic device for selective detection of peanut allergen Ara h1 applying black phosphorus-Au nanocomposites for signal amplification. Talanta 2024, 267, 125188. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.-Y.; Chou, S.-T.; Chen, C.-H.; Chou, S.-Y.; Wu, J.-H.; Chen, Y.-C.; Lee, G.-B. An automatic integrated microfluidic system for allergy microarray chips. Analyst 2018, 143, 2285–2292. [Google Scholar] [CrossRef]
- Sundhoro, M.; Agnihotra, S.R.; Amberger, B.; Augustus, K.; Khan, N.D.; Barnes, A.; BelBruno, J.; Mendecki, L. An electrochemical molecularly imprinted polymer sensor for rapid and selective food allergen detection. Food Chem. 2021, 344, 128648. [Google Scholar] [CrossRef]
- Martinez, A.W.; Phillips, S.T.; Butte, M.J.; Whitesides, G.M. Patterned paper as a platform for inexpensive, low-volume, portable bioassays. Angew. Chem. 2007, 119, 1340–1342. [Google Scholar] [CrossRef]
- Ge, L.; Yan, J.; Song, X.; Yan, M.; Ge, S.; Yu, J. Three-dimensional paper-based electrochemiluminescence immunodevice for multiplexed measurement of biomarkers and point-of-care testing. Biomaterials 2012, 33, 1024–1031. [Google Scholar] [CrossRef]
- Arroyo, M.J.; de Orbe-Payá, I.; Ortega-Munoz, M.; Vilar-Tenorio, J.; Gallego, D.; Mohr, G.J.; Capitán-Vallvey, L.F.; Erenas, M.M. Capillary microfluidic platform for sulfite determination in wines. Sens. Actuators B Chem. 2022, 359, 131549. [Google Scholar] [CrossRef]
- Yang, Y.; Zeng, X.; Fu, C.; Tan, L.; Yang, N.; Liu, Y.; Shen, Q.; Wei, J.; Yu, C.; Lu, C. Paper-based microfluidics and tailored gold nanoparticles for visual colorimetric detection of multiplex allergens. Anal. Chim. Acta 2023, 1272, 341497. [Google Scholar] [CrossRef]
- Zhou, J.; Li, B.; Qi, A.; Shi, Y.; Qi, J.; Xu, H.; Chen, L. ZnSe quantum dot based ion imprinting technology for fluorescence detecting cadmium and lead ions on a three-dimensional rotary paper-based microfluidic chip. Sens. Actuators B Chem. 2020, 305, 127462. [Google Scholar] [CrossRef]
- Li, Z.; Zhu, Y.; Zhang, W.; Xu, C.; Zhao, Y. A Low-Cost and High Sensitive Paper-Based Microfluidic Device for Rapid Detection of Glucose in Fruit. Food Anal. Methods 2016, 10, 666–674. [Google Scholar] [CrossRef]
- Hossain, S.; Luckham, R.E.; Mcfadden, M.J.; Brennan, J.D. Reagentless bidirectional lateral flow bioactive paper sensors for detection of pesticides in beverage and food samples. Anal. Chem. 2009, 81, 9055–9064. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Yang, T.; Zhang, W.; Ren, C.; Zhang, J.; Zhang, Y.; Cui, T.; Yue, W. Rapid detection of trichlorfon residues by a microfluidic paper-based phosphorus-detection chip (μPPC). New J. Chem. 2019, 43, 7194–7197. [Google Scholar] [CrossRef]
- Yang, N.; Wang, P.; Xue, C.Y.; Sun, J.; Mao, H.P.; Oppong, P.K. A portable detection method for organophosphorus and carbamates pesticide residues based on multilayer paper chip. J. Food Process Eng. 2018, 41, e12867. [Google Scholar] [CrossRef]
- Qiang, G.; Li, Z.; Niu, M. A pilot study on PCR-based detection of four foodborne pathogenic microorganisms. J. Food Meas. Charact. 2017, 12, 675–682. [Google Scholar]
- Silva, N.F.; Almeida, C.M.; Magalhães, J.M.; Gonçalves, M.P.; Freire, C.; Delerue-Matos, C. Development of a disposable paper-based potentiometric immunosensor for real-time detection of a foodborne pathogen. Biosens. Bioelectron. 2019, 141, 111317. [Google Scholar] [CrossRef]
- Zhang, Y.; Peng, Z.; Ye, B.C. A low-cost and simple paper-based microfluidic device for simultaneous multiplex determination of different types of chemical contaminants in food. Biosens. Bioelectron. 2015, 68, 14–19. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, Z.; Xu, S.; Da, L.; Lin, D.; Jiang, C. Enzyme-free and rapid visual quantitative detection for pesticide residues utilizing portable smartphone integrated paper sensor. J. Hazard. Mater. 2022, 436, 129320. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, Y.; Yang, X.; Yuan, X.; Zhang, J. Pb (II) inhibits CRISPR/Cas12a activation and application for paper-based microfluidic biosensor assisted by smartphone. Sens. Actuators B Chem. 2023, 398, 134732. [Google Scholar] [CrossRef]
- Ji, Q.; Xin-Xia, F.; Dong-Mei, D.; Hai-Bo, H.; Li-Qiang, L. Progress in rapid detection techniques using paper-based platforms for food safety. Chin. J. Anal. Chem. 2020, 48, 1616–1624. [Google Scholar]
- Bougadi, E.T.; Kalogianni, D.P. based DNA biosensor for food authenticity testing. Food Chem. 2020, 322, 126758. [Google Scholar] [CrossRef] [PubMed]
- Muthukumar, R.; Kapoor, A.; Balasubramanian, S.; Vaishampayan, V.; Gabhane, M. Detection of adulteration in sunflower oil using paper-based microfluidic lab-on-a-chip devices. Mater. Today Proc. 2021, 34, 496–501. [Google Scholar] [CrossRef]
- Zhong, J.; Shi, J.; Amundadottir, L.T. Artificial intelligence and improved early detection for pancreatic cancer. Innovation 2023, 4, 100457. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, M.; Zhang, G.; Song, J.; Tan, M.; Su, W. Paper-Based Microfluidic Chips for Food Hazard Factor Detection: Fabrication, Modification, and Application. Foods 2023, 12, 4107. https://doi.org/10.3390/foods12224107
Liang M, Zhang G, Song J, Tan M, Su W. Paper-Based Microfluidic Chips for Food Hazard Factor Detection: Fabrication, Modification, and Application. Foods. 2023; 12(22):4107. https://doi.org/10.3390/foods12224107
Chicago/Turabian StyleLiang, Meiqi, Guozhi Zhang, Jie Song, Mingqian Tan, and Wentao Su. 2023. "Paper-Based Microfluidic Chips for Food Hazard Factor Detection: Fabrication, Modification, and Application" Foods 12, no. 22: 4107. https://doi.org/10.3390/foods12224107
APA StyleLiang, M., Zhang, G., Song, J., Tan, M., & Su, W. (2023). Paper-Based Microfluidic Chips for Food Hazard Factor Detection: Fabrication, Modification, and Application. Foods, 12(22), 4107. https://doi.org/10.3390/foods12224107










