Structure, Physicochemical Property, and Functional Activity of Dietary Fiber Obtained from Pear Fruit Pomace (Pyrus ussuriensis Maxim) via Different Extraction Methods
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Preparation of Pomace
2.3. Preparation of Samples
2.4. Routine Constituents
2.5. Physicochemical Property Analysis
2.5.1. Water Holding Capacity (WHC), Water Binding Capacity (WBC), and Oil Holding Capacity (OHC)
2.5.2. Water Swelling Capacity (WSC)
2.6. Monosaccharide Composition
2.7. Molecular Weight Distribution
2.8. Structural Analysis
2.9. Functional Characteristics
2.9.1. Antioxidant Capacity
2.9.2. Cholesterol Adsorption Capacity (CAC)
2.9.3. Nitrite Adsorption Capacity (NAC)
2.9.4. Glucose Adsorption Capacity (GAC)
2.9.5. Inhibitory Effect of SDF on α-Amylase (α-AAIR)
2.10. Statistical Analysis
3. Results and Discussion
3.1. Proximate Composition
3.2. Physicochemical Properties
3.3. Monosaccharide Composition of SDFs
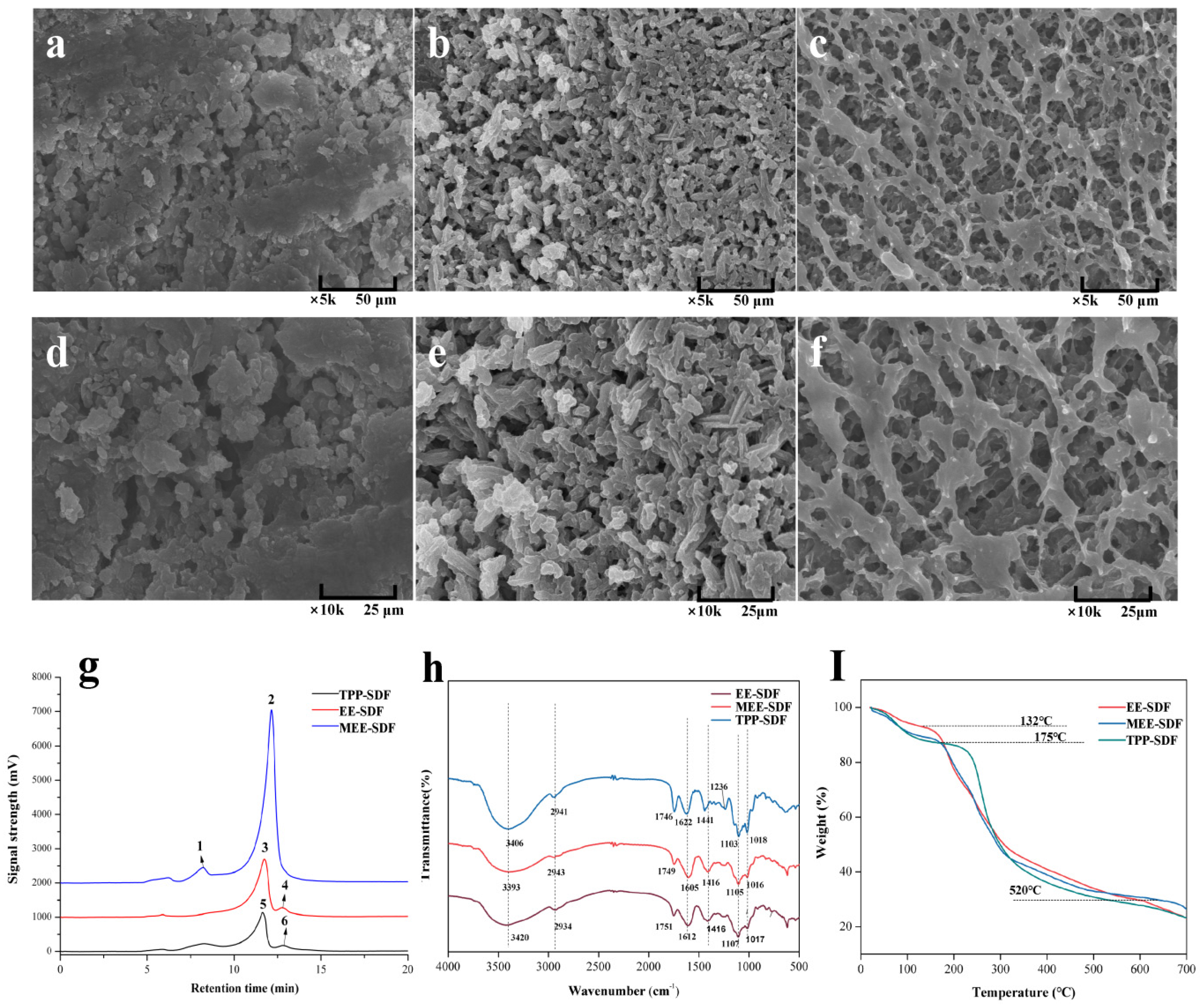
3.4. Structural Characterization of SDFs
3.5. Functional Characteristics of SDFs
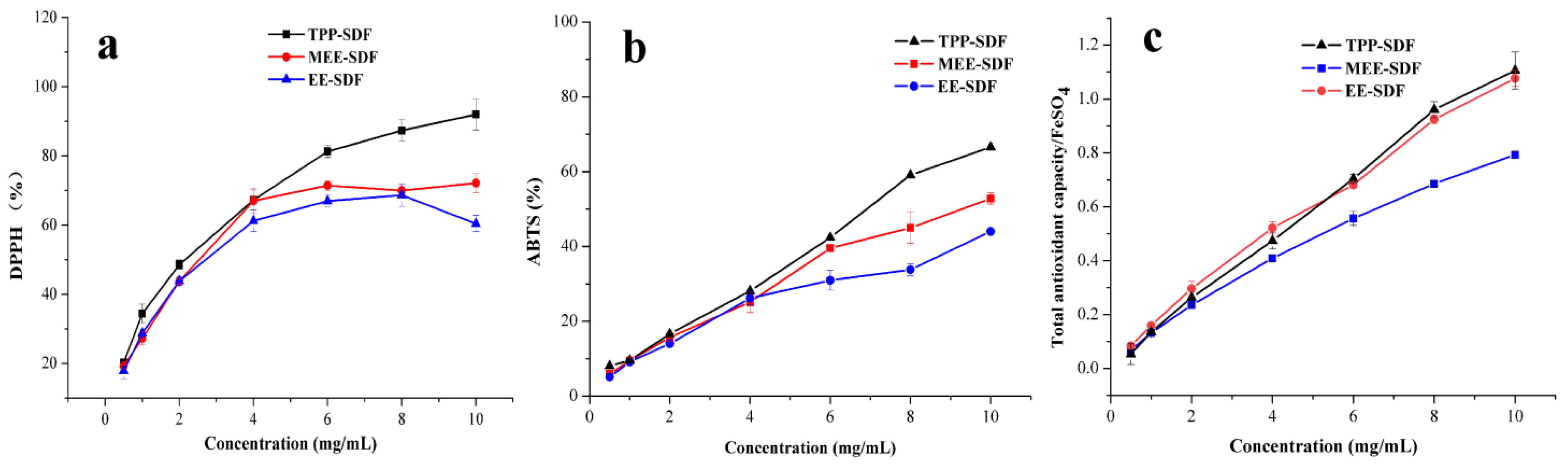
3.5.1. Antioxidant Activities
3.5.2. Cholesterol Adsorption Capacity (CAC)
3.5.3. Nitrite Adsorption Capacity (NAC)
3.5.4. Glucose Adsorption Capacity (GAC)
3.5.5. Inhibitory Effect of SDF on α-Amylase (α-AAIR)
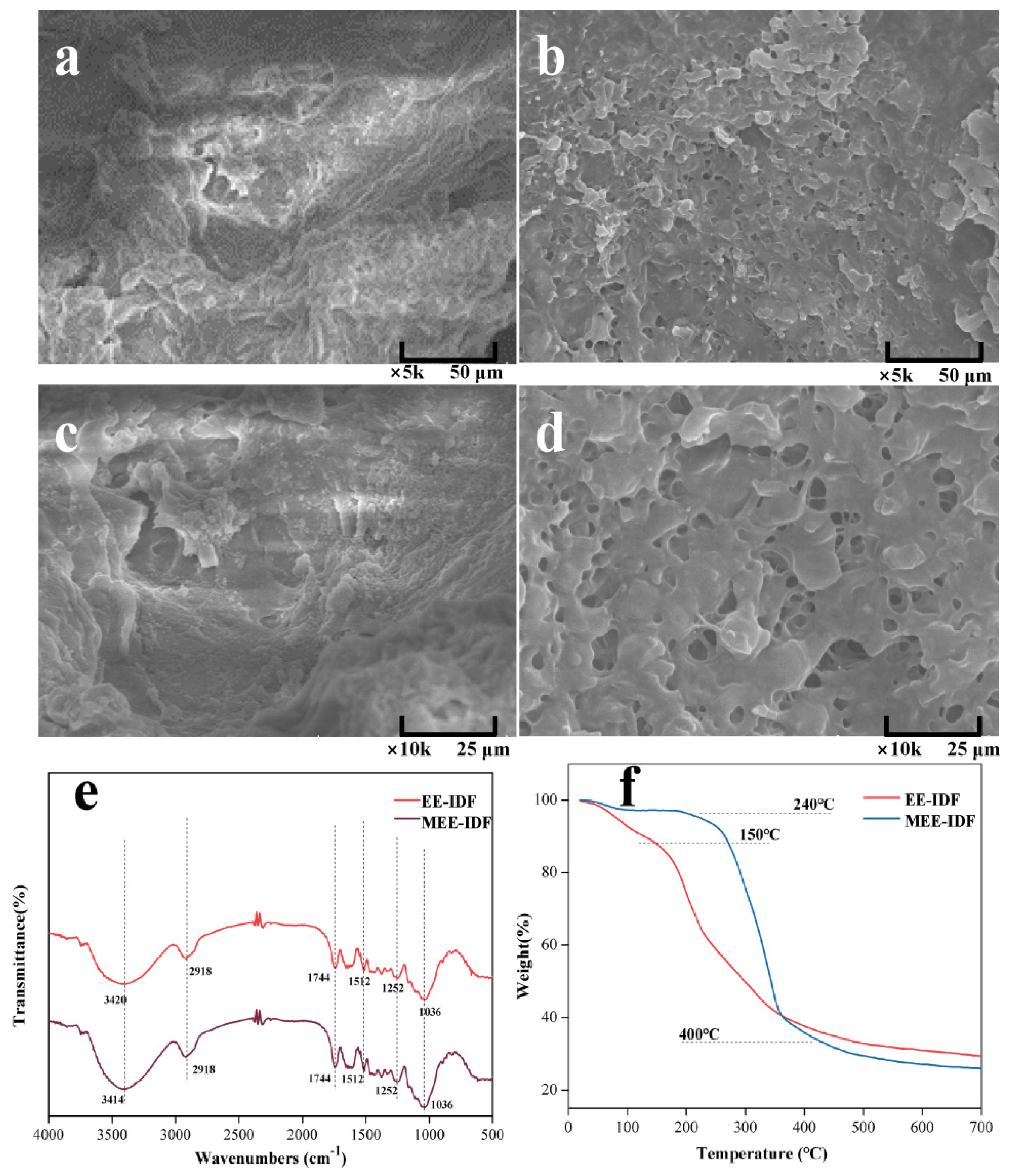
3.6. Structural Characteristics of IDFs
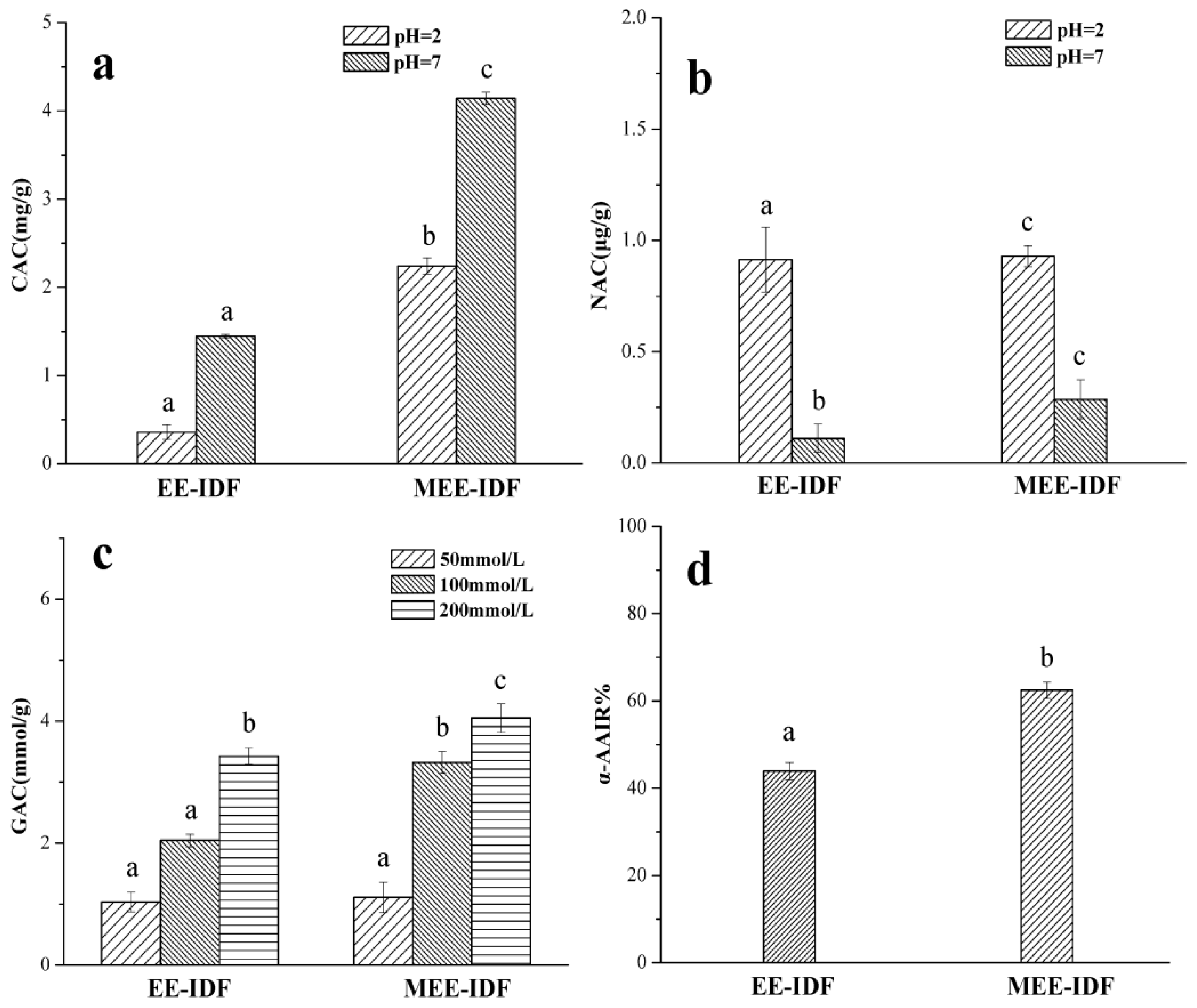
3.7. Functional Activity of IDFs
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stephen, A.M.; Champ, M.M.J.; Cloran, S.J.; Fleith, M.; Lieshout, L.V.; Mejborn, H.; Burley, V.J. Dietary fibre in Europe: Current state of knowledge on definitions, sources, recommendations, intakes and relationships to health. Nutr. Res. Rev. 2017, 30, 149–190. [Google Scholar] [CrossRef] [PubMed]
- Theuwissen, E.; Mensink, R.P. Water-soluble dietary fibers and cardiovascular disease. Physiol. Behav. 2008, 94, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Feng, D.; Planinic, P.; Ebersole, J.L.; Lyons, T.J.; Alexander, J.M. Dietary blueberry and soluble fiber supplementation reduces risk of gestational diabetes in women with obesity in a randomized controlled trial. J. Nutr. 2021, 151, 1128–1138. [Google Scholar] [CrossRef]
- do Prado, S.B.R.; Minguzzi, B.T.; Hoffmann, C.; Fabi, J.P. Modulation of human gut microbiota by dietary fibers from unripe and ripe papayas: Distinct polysaccharide degradation using a colonic in vitro fermentation model. Food Chem. 2021, 348, 129071. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Song, R.; Li, Y.; Zhang, W.; Wan, Z.; Wang, Y.; Zhang, H.; Han, S. Effects of oat fiber intervention on cognitive behavior in ldlr−/− mice modeling atherosclerosis by targeting the microbiome–gut–brain axis. J. Agric. Food Chem. 2020, 68, 14480–14491. [Google Scholar] [CrossRef]
- Raza, G.S.; Maukonen, J.; Makinen, M.; Niemi, P.; Niiranen, L.; Hibberd, A.A.; Poutanen, K.; Buchert, J.; Herzig, K.H. Hypocholesterolemic effect of the lignin-rich insoluble pomace of brewer’s spent grain in mice fed a high-fat diet. J. Agric. Food Chem. 2018, 67, 1104–1114. [Google Scholar] [CrossRef]
- Yang, L.; Lin, Q.; Han, L.; Wang, Z.; Luo, M.; Kang, W.; Liu, J.; Wang, J.; Ma, T.; Liu, H. Soy hull dietary fiber alleviates inflammation in balb/c mice by modulating the gut microbiota and suppressing the TLR-4/NF-κB signaling pathway. Food Funct. 2020, 11, 5965–5975. [Google Scholar] [CrossRef]
- Reynolds, A.; Mann, J.; Cummings, J.; Winter, N.; Mete, E.; Morenga, L.T. Carbohydrate quality and human health: A series of systematic reviews and meta-analyses. Lancet 2019, 393, 434–445. [Google Scholar] [CrossRef] [Green Version]
- Zhao, L.; Zhang, F.; Ding, X.; Wu, G.; Lam, Y.Y.; Wang, X.; Fu, H.; Xue, X.; Lu, C.; Ma, J.; et al. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science 2018, 359, 1151–1156. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Fang, X.; Chen, H.; Han, Y.; Liu, R.; Wu, W.; Gao, H. Retarding effect of dietary fibers from bamboo shoot (Phyllostachys edulis) in hyperlipidemic rats induced by a high-fat diet. Food Funct. 2021, 12, 4696–4706. [Google Scholar] [CrossRef]
- Benitez, V.; Rebollo-Hernanz, M.; Hernanz, S.; Chantres, S.; Aguilera, Y.; Martin-Cabrejas, M.A. Coffee parchment as a new dietary fiber ingredient: Functional and physiological characterization. Food Res. Int. 2019, 122, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Cabrejas, A.; Laclaustra, M.; Guallar-Castillón, P.; Casasnovas, J.A.; Jarauta, E.; Sandoval-Insausti, H.; Donat-Vargas, C.; Moreno-Franco, B. High-quality intake of carbohydrates is associated with lower prevalence of subclinical atherosclerosis in femoral arteries: The AWHS study. Clin. Nutr. 2021, 40, 3883–3889. [Google Scholar] [CrossRef]
- León-González, A.J.; Jara-Palacios, M.J.; Abbas, M.; Heredia, F.J.; Schini-Kerth, V.B. Role of epigenetic regulation on the induction of apoptosis in Jurkat leukemia cells by white grape pomace rich in phenolic compounds. Food Funct. 2017, 8, 4062–4069. [Google Scholar] [CrossRef]
- Macagnan, F.T.; Da Silva, L.P.; Hecktheuer, L.H. Dietary fiber: The scientific search for an ideal definition and methodology of analysis, and its physiological importance as a carrier of bioactive compounds. Food Res. Int. 2016, 85, 144–154. [Google Scholar] [CrossRef]
- Huang, Y.; Ruan, G.; Qin, Z.; Li, H.; Zheng, Y. Antioxidant activity measurement and potential antioxidant peptides exploration from hydrolysates of novel continuous microwave-assisted enzymolysis of the Scomberomorus niphonius protein. Food Chem. 2017, 223, 89–95. [Google Scholar] [CrossRef]
- Yan, J.K.; Wang, Y.Y.; Qiu, W.Y.; Ma, H.; Wang, Z.B.; Wu, J.Y. Three-phase partitioning as an elegant and versatile platform applied to nonchromatographic bioseparation processes. Crit. Rev. Food Sci. 2018, 58, 2416–2431. [Google Scholar] [CrossRef] [PubMed]
- Gulati, S.; Misra, A.; Pandey, R.M. Effects of 3 g of soluble fiber from oats on lipid levels of Asian Indians-a randomized controlled, parallel arm study. Lipids Health Dis. 2017, 16, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Qiu, D.; Guo, J.; Yu, H.; Yan, J.; Yang, S.; Li, X.; Zhang, Y.; Sun, J.; Cong, J.; He, S.; et al. Antioxidant phenolic compounds isolated from wild Pyrus ussuriensis Maxim. fruit peels and leaves. Food Chem. 2018, 241, 182–187. [Google Scholar] [CrossRef] [PubMed]
- AOAC. AOAC official method 968.28. In Total Sugars in Molasses as Invert Sugar; Association of Official Analytical Chemists: Washington, DC, USA, 1969. [Google Scholar]
- AOAC. AOAC official method 991.43. In Total, Soluble, and Insoluble Dietary Fiber in Foods; Association of Official Analytical Chemists: Washington, DC, USA, 1996. [Google Scholar]
- Ma, M.; Mu, T. Effects of extraction methods and particle size distribution on the structural, physicochemical, and functional properties of dietary fiber from deoiled cumin. Food Chem. 2016, 194, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Sowbhagya, H.B.; Suma, P.F.; Mahadevamma, S.; Tharanathan, R.N. Spent residue from cumin–a potential source of dietary fiber. Food Chem. 2007, 104, 1220–1225. [Google Scholar] [CrossRef]
- Bender, A.B.B.; Speroni, C.S.; Moro, K.I.B.; Morisso FD, P.; dos Santos, D.R.; da Silva, L.P.; Penna, N.G. Effects of micronization on dietary fiber composition, physicochemical properties, phenolic compounds, and antioxidant capacity of grape pomace and its dietary fiber concentrate. LWT-Food Sci. Technol. 2020, 117, 108652. [Google Scholar] [CrossRef]
- Jia, M.Y.; Chen, J.J.; Liu, X.Z.; Xie, M.Y.; Nie, S.P.; Chen, Y.; Xie, J.; Yu, Q. Structural characteristics and functional properties of soluble dietary fiber from defatted rice bran obtained through Trichoderma viride fermentation. Food Hydrocoll. 2019, 94, 468–474. [Google Scholar] [CrossRef]
- Hua, M.; Lu, J.; Qu, D.; Liu, C.; Zhang, L.; Li, S.; Chen, J.; Sun, Y. Structure, physicochemical properties and adsorption function of insoluble dietary fiber from ginseng residue: A potential functional ingredient. Food Chem. 2019, 286, 522–529. [Google Scholar] [CrossRef]
- Peerajit, P.; Chiewchan, N.; Devahastin, S. Effects of pretreatment methods on health-related functional properties of high dietary fibre powder from lime residues. Food Chem. 2012, 132, 1891–1898. [Google Scholar] [CrossRef]
- Benítez, V.; Rebollo-Hernanz, M.; Aguilera, Y.; Bejerano, S.; Cañas, S.; Martín-Cabrejas, M.A. Extruded coffee parchment shows enhanced antioxidant, hypoglycaemic, and hypolipidemic properties by releasing phenolic compounds from the fibre matrix. Food Funct. 2021, 12, 1097–1110. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Li, T.; Liu, C.H.; Zheng, L. Effects of high hydrostatic pressure and superfine grinding treatment on physicochemical/functional properties of pear pomace and chemical composition of its soluble dietary fibre. LWT-Food Sci. Technol. 2019, 107, 171–177. [Google Scholar] [CrossRef]
- Adapa, S.; Dingeldein, H.; Schmidt, K.A.; Herald, T.J. Rheological properties of ice cream mixes and frozen ice creams containing fat and fat replacers. J. Dairy Sci. 2000, 83, 2224–2229. [Google Scholar] [CrossRef]
- Hussain, S.; Jie, L.; Jin, W.; Yan, S.; Wang, Q. Effect of micronisation on dietary fibre content and hydration properties of lotus node powder fractions. Int. J. Food Sci. Technol. 2018, 53, 590–598. [Google Scholar] [CrossRef]
- Deng, M.; Lin, Y.; Dong, L.; Jia, X.; Shen, Y.; Liu, L.; Chi, J.; Huang, F.; Zhang, M.; Zhang, R. Physicochemical and functional properties of dietary fiber from pummelo (Citrus grandis L. Osbeck) and grapefruit (Citrus paradisi Mcfad) cultivars. Food Biosci. 2021, 40, 100890. [Google Scholar] [CrossRef]
- Kikuchi, Y.; Nozaki, S.; Makita, M.; Yokozuka, S.; Fukudome, S.I.; Yanagisawa, T.; Aoe, S. Effects of whole grain wheat bread on visceral fat obesity in Japanese subjects: A randomized double-blind study. Plant Food Hum. Nutr. 2018, 73, 161–165. [Google Scholar] [CrossRef]
- Jacometti, G.; Mello, L.; Nascimento, P.; Sueiro, A.C.; Yamashita, F.; Mali, S. The physicochemical properties of fibrous residues from the agro industry. LWT-Food Sci. Technol. 2015, 62, 138–143. [Google Scholar] [CrossRef]
- Zhang, W.M.; Zeng, G.L.; Pan, Y.G.; Chen, W.; Huang, W.; Cheen, H.; Li, Y. Properties of soluble dietary fiber-polysaccharide from papaya peel obtained through alkaline or ultrasound-assisted alkaline extraction. Carbohyd. Polym. 2017, 172, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Y.; Ma, H.; Ding, Z.C.; Yang, Y.; Wang, W.H.; Zhang, H.N.; Yan, J.K. Three-phase partitioning for the direct extraction and separation of bioactive exopolysaccharides from the cultured broth of Phellinus baumii. Int. J. Biol. Macromol. 2019, 123, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Kapusniak, K.; Lubas, K.; Wojcik, M.; Rosicka-Kaczmarek, J.; Pavlyuk, V.; Kluziak, K.; Gonçalves, I.; Lopes, J.; Coimbra, M.A. Janusz Kapusniak. Effect of continuous and discontinuous microwave-assisted heating on starch-derived dietary fiber production. Molecules 2021, 26, 5619. [Google Scholar] [CrossRef]
- Wang, Z.B.; Pei, J.J.; Ma, H.L.; Cai, P.F.; Yan, J.K. Effect of extraction media on preliminary characterizations and antioxidant activities of Phellinus linteus polysaccharides. Carbohyd. Polym. 2014, 109, 49–55. [Google Scholar] [CrossRef]
- Yang, M.; Wu, L.R.; Cao, C.J.; Wang, S.Y.; Zhang, D.M. Improved function of bamboo shoot fiber by high-speed shear dispersing combined with enzyme treatment. Int. J. Food Sci. Technol. 2019, 54, 844–853. [Google Scholar] [CrossRef]
- Silva, I.; Machado, F.; Moreno, M.J.; Nunes, C.; Coimbra, M.A.; Coreta-Gomes, F. Polysaccharide Structures and Their Hypocholesterolemic Potential. Molecules 2021, 26, 4559. [Google Scholar] [CrossRef]
- Yan, J.K.; Yu, Y.B.; Wang, C.; Cai, W.D.; Yang, Y.; Zhang, H.N. Production, physicochemical characteristics, and in vitro biological activities of polysaccharides obtained from fresh bitter gourd (Momordica charantia L.) via room temperature extraction techniques. Food Chem. 2021, 337, 127798. [Google Scholar] [CrossRef]
- Wang, W.; Ma, X.; Jiang, P.; Hu, L.; Zhi, Z.; Chen, J.; Ding, T.; Ye, X.; Liu, D. Characterization of pectin from grapefruit peel: A comparison of ultrasound-assisted and conventional heating extractions. Food Hydrocoll. 2016, 61, 730–739. [Google Scholar] [CrossRef]
- Yan, X.; Ye, R.; Chen, Y. Blasting extrusion processing: The increase of soluble dietary fiber content and extraction of soluble-fiber polysaccharides from wheat bran. Food Chem. 2015, 180, 106–115. [Google Scholar] [CrossRef]
- Shen, M.; Ge, Y.; Kang, Z.; Quan, Z.; Wang, J.; Xiao, J.; Wang, W.; Cao, L. Yield and Physicochemical Properties of Soluble Dietary Fiber Extracted from Untreated and Steam Explosion-Treated Black Soybean Hull. J. Chem. 2019, 2019, 9736479. [Google Scholar] [CrossRef]
- Chau, C.F.; Huang, Y.L.; Lee, M.H. In vitro hypoglycemic effects of different insoluble fibre-rich fractions prepared from the peel of Citrus sinensis L.cv. Liu cheng. J. Agric. Food Chem. 2003, 51, 6623–6626. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Y.; Zhu, J.; Ma, H.; Ding, Z.D.; Li, L.; Yan, J.K. Antidiabetic activity of a polysaccharide-protein complex from Asian Clam (Corbicula fluminea) in streptozotoxin-induced diabetic rats and its underlying mechanism. Food Funct. 2019, 10, 5574–5586. [Google Scholar] [CrossRef] [PubMed]
- Gan, J.P.; Peng, G.Y.; Liu, S.; Hu, X.; Wang, X.; Guo, S.; Xie, J.; Chen, Y.; Yu, Q. Comparison of structural, functional and in vitro in vitro digestion properties of bread incorporated with grapefruit peel soluble dietary fibers prepared by three microwave-assisted modifications. Food Funct. 2020, 11, 6458–6466. [Google Scholar] [CrossRef]
- Wang, L.; Xu, H.; Yuan, F.; Pan, Q.; Fan, R.; Gao, Y. Physicochemical characterization of five types of citrus dietary fibers. Biocatal. Agric. Biotechnol. 2015, 4, 250–258. [Google Scholar] [CrossRef]
- Jiang, Y.; Yin, H.; Zheng, Y.; Wang, D.; Liu, Z.; Deng, Y.; Zhao, Y. Structure, physicochemical and bioactive properties of dietary fibers from Akebia trifoliata (Thunb.) Koidz. seeds using ultrasonication/shear emulsifying/microwave-assisted enzymatic extraction. Food Res. Int. 2020, 136, 109348. [Google Scholar] [CrossRef]


| Proximate Composition (g/100 g) | PUPP | EE-SDF | MEE-SDF | TPP-SDF | EE-IDF | MEE-IDF |
|---|---|---|---|---|---|---|
| Dietary fiber | 79.90 ± 1.02 e | 96.23 ± 0.86 c | 97.45 ± 0.72 b | 96.63 ± 0.81 c | 95.78 ± 1.05 d | 97.96 ± 0.83 a |
| Ash | 0.80 ± 0.03 c | 0.67 ± 0.16 e | 0.75 ± 0.20 d | 0.55 ± 0.27 f | 1.18 ± 0.22 a | 1.06 ± 0.17 b |
| Protein | 2.21 ± 0.05 a | 1.14 ± 0.14 c | 1.09 ± 0.09 d | 0.85 ± 0.06 e | 1.28 ± 0.17 b | 1.26 ± 0.12 b |
| Moisture | 3.18 ± 0.07 a | 1.06 ± 0.12 e | 1.21 ± 0.10 d | 1.55 ± 0.11 b | 1.18 ± 0.07 b | 1.28 ± 0.14 c |
| Total sugar | 13.80 ± 0.17 d | 28.58 ± 0.14 c | 35.68 ± 0.22 b | 39.20 ± 0.17 a | 5.80 ± 0.16 f | 6.97 ± 0.20 e |
| Yield (%) | - | 5.80 ± 0.16 d | 7.68 ± 0.22 c | 3.97 ± 0.21 e | 71.21 ± 0.55 a | 67.81 ± 0.66 b |
| WHC (g/g) | - | 3.85 ± 0.23 d | 4.49 ± 0.54 b | 5.11 ± 0.29 a | 4.35 ± 0.15 c | 4.29 ± 0.18 c |
| WBC (g/g) | - | 3.40 ± 0.04 c | 3.54 ± 0.15 b | 4.05 ± 0.31 a | 0.94 ± 0.19 d | 0.88 ± 0.50 d |
| WSC (mL/g) | - | 2.03 ± 0.11 d | 2.93 ± 0.30 b | 3.48 ± 0.14 a | 1.67 ± 0.40 e | 2.20 ± 0.25 c |
| OHC (g/g) | - | 3.56 ± 0.22 d | 4.19 ± 0.10 b | 3.85 ± 0.33 c | 3.59 ± 0.13 d | 4.59 ± 0.29 a |
| Monosaccharide Composition% | EE-SDF | MEE-SDF | TPP-SDF |
|---|---|---|---|
| Fuc | 0.91 ± 0.13 b | 0.97 ± 0.14 b | 1.55 ± 0.21 a |
| Rha | 3.06 ± 0.14 c | 3.77 ± 0.17 b | 4.69 ± 0.25 a |
| Ara | 11.22 ± 0.47 c | 13.97 ± 0.56 b | 21.51 ± 0.82 a |
| Gal | 14.51 ± 0.64 b | 12.79 ± 0.59 c | 16.16 ± 0.71 a |
| Glc | 33.11 ± 2.13 a | 21.11 ± 1.66 b | 16.99 ± 1.07 c |
| Xyl | 4.54 ± 0.35 b | 3.76 ± 0.28 c | 6.20 ± 0.51 a |
| Man | 12.24 ± 0.65 b | 10.03 ± 0.59 c | 14.64 ± 0.67 a |
| Fru | 0.40 ± 0.05 a | 0.40 ± 0.02 a | 0.35 ± 0.03 b |
| Rib | 0.13 ± 0.04 c | 0.39 ± 0.06 a | 0.21 ± 0.04 b |
| Gal-UA | 19.41 ± 1.43 b | 32.39 ± 2.07 a | 16.87 ± 1.87 c |
| Glc-UA | 0.48 ± 0.11 b | 0.43 ± 0.08 b | 0.63 ± 0.07 a |
| Man-UA | ND | ND | 0.20 ± 0.01 a |
| Sample | Peak Number | RT (min) | Mn (Da) | Mw (Da) | D (Mw/Mn) | Area (%) |
|---|---|---|---|---|---|---|
| EE-SDF | 1 | 11.74 | 2888 | 486,841 | 168.57 | 87.91 |
| 2 | 12.77 | 31 | 135 | 4.35 | 10.17 | |
| MEE-SDF | 3 | 8.22 | 12,081,875 | 20,760,280 | 1.72 | 4.27 |
| 4 | 12.15 | 584 | 5432 | 9.28 | 92.90 | |
| TPP-SDF | 5 | 11.32 | 5932 | 15,760 | 2.66 | 86.10 |
| 6 | 12.79 | 119 | 185 | 1.55 | 5.48 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, F.; Ren, X.; Du, B.; Chen, L.; Yu, Z.; Yang, Y. Structure, Physicochemical Property, and Functional Activity of Dietary Fiber Obtained from Pear Fruit Pomace (Pyrus ussuriensis Maxim) via Different Extraction Methods. Foods 2022, 11, 2161. https://doi.org/10.3390/foods11142161
Peng F, Ren X, Du B, Chen L, Yu Z, Yang Y. Structure, Physicochemical Property, and Functional Activity of Dietary Fiber Obtained from Pear Fruit Pomace (Pyrus ussuriensis Maxim) via Different Extraction Methods. Foods. 2022; 11(14):2161. https://doi.org/10.3390/foods11142161
Chicago/Turabian StylePeng, Fei, Xin Ren, Bin Du, Linan Chen, Zuoqing Yu, and Yuedong Yang. 2022. "Structure, Physicochemical Property, and Functional Activity of Dietary Fiber Obtained from Pear Fruit Pomace (Pyrus ussuriensis Maxim) via Different Extraction Methods" Foods 11, no. 14: 2161. https://doi.org/10.3390/foods11142161
APA StylePeng, F., Ren, X., Du, B., Chen, L., Yu, Z., & Yang, Y. (2022). Structure, Physicochemical Property, and Functional Activity of Dietary Fiber Obtained from Pear Fruit Pomace (Pyrus ussuriensis Maxim) via Different Extraction Methods. Foods, 11(14), 2161. https://doi.org/10.3390/foods11142161






