Effect of Age of Agave tequilana Weber Blue Variety on Quality and Authenticity Parameters for the Tequila 100% Agave Silver Class: Evaluation at the Industrial Scale Level
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
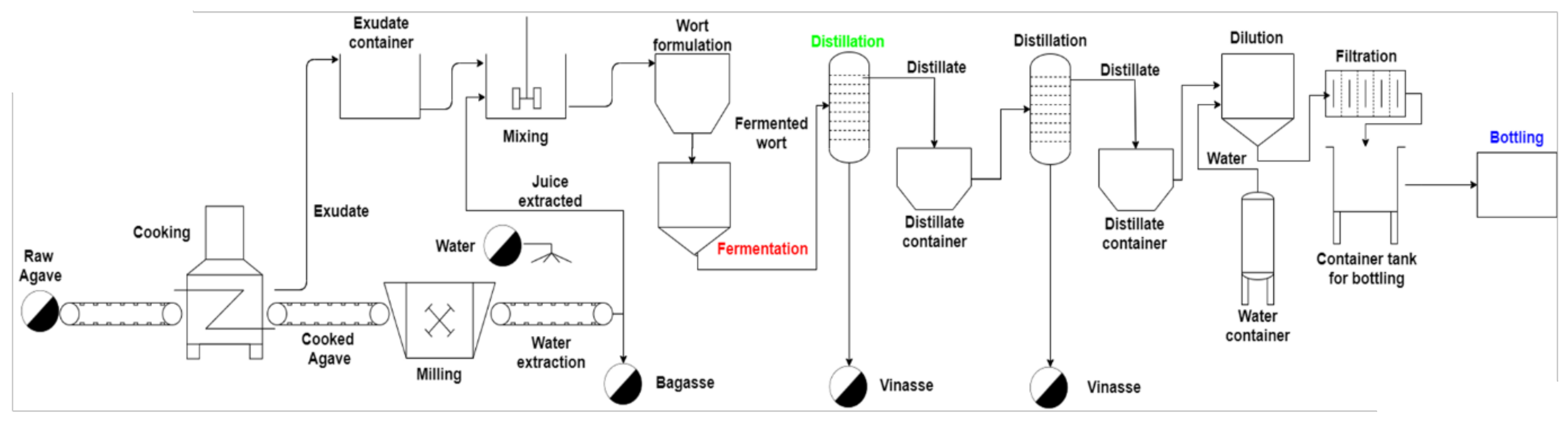
2.2. Industrial-Scale Tequila Production
2.2.1. Raw Material (Agave tequilana Weber Blue Variety)
2.2.2. Hydrolysis and Milling Processes
2.2.3. Fermentation
2.2.4. Distillation
2.2.5. Final Conditioning and Bottling
2.3. Chemical Analysis
2.3.1. Total Reducing Sugars (TRS)
2.3.2. Alcoholic Content
2.3.3. Gas Chromatography
2.3.4. High-Performance Liquid Chromatography
2.3.5. Determination of the Isotopic Ratios of Carbon 13 (δ13C) and Oxygen 18 (δ18O)
3. Results and Discussion
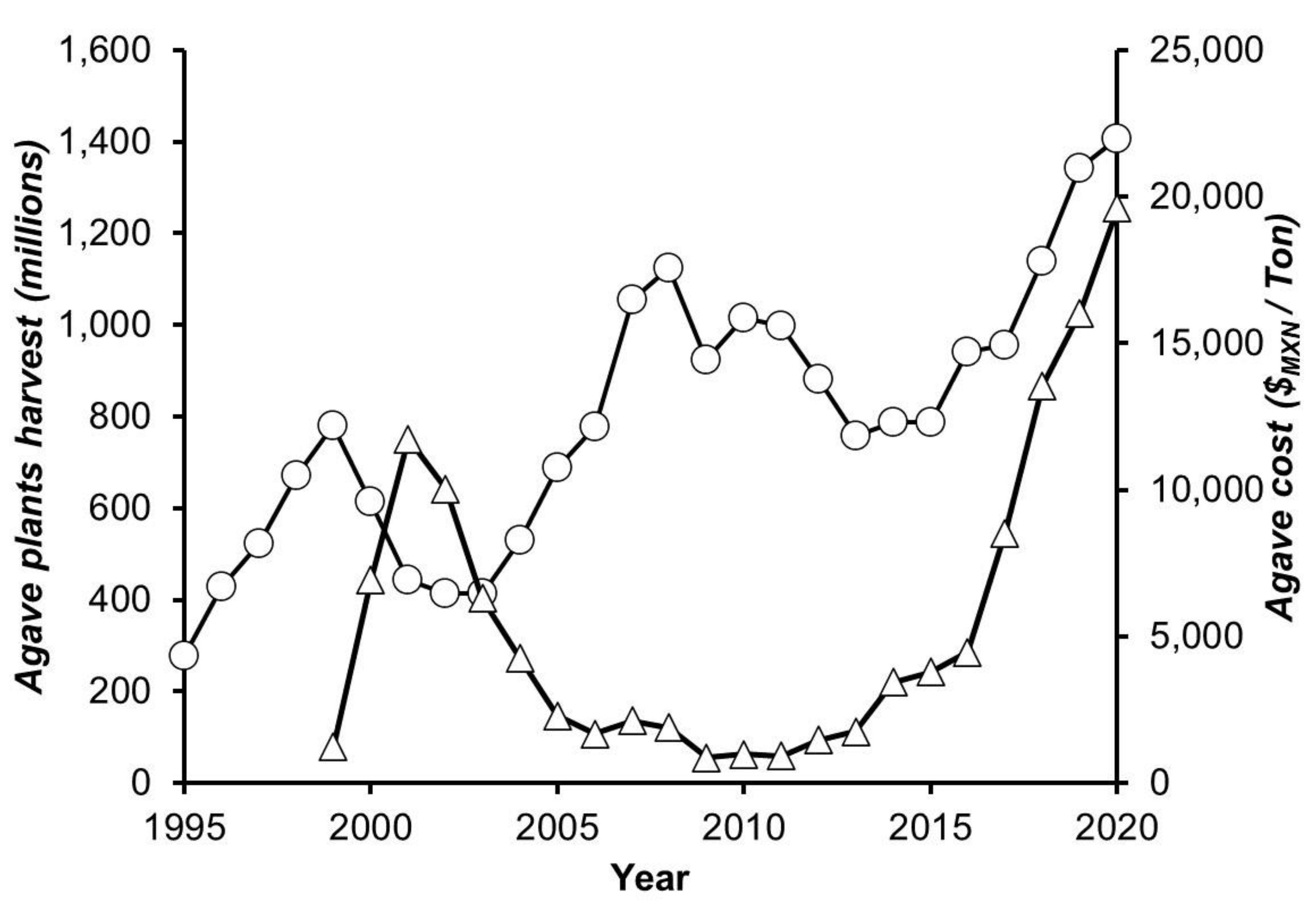
3.1. Analysis of Harvest, Cooking, and Milling Processes
3.2. Fermentation Stage
3.3. Distillation Stage
3.4. Final Product Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IIEG. Exportaciones de Tequila: Un Estudio de Caso para la Obtención de Ponderadores de Comercio Exterior; IIEG Jalisco: Zapopan, Mexico, 2020. [Google Scholar]
- Orozco Martínez, J.L. Panorámica Actual de la Industria Tequilera. In Manual del Técnico Tequilero; Consejo Regulador del Tequila: Guadalajara, Mexico, 2019; pp. 7–39. [Google Scholar]
- Del Real Laborde, J.I. Agave, Materia Prima del Tequila. In Manual del Técnico Tequilero; Consejo Regulador del Tequila: Guadalajara, Mexico, 2019; pp. 128–157. [Google Scholar]
- Ruiz-Pérez, A.; Pérez-Castañeda, J.I.; Castañeda-Guzmán, R.; Pérez-Ruiz, S.J. Determination of tequila quality by photoacoustic analysis. Int. J. Thermophys. 2013, 34, 1695–1702. [Google Scholar] [CrossRef]
- Dirección General de Normas. Norma Oficial Mexicana: Bebidas Alcohólicas—Tequila—Especificaciones. In Diario Oficial de la Federación Mexico; The Norma Oficial Mexicana (NOM): NOM-006-SCFI-2012; Dirección General de Normas: Mexico City, Mexico, 2012. [Google Scholar]
- Soto-Castro, D.; Perez-Herrera, A.; Garcia-Sanchez, E.; Santiago-Garcia, P.A. Identification and quantification of bioactive compounds in agave potatorum zucc. leaves at different stages of development and a preliminary biological assay. Waste Biomass Valoriz. 2021, 12, 4537–4547. [Google Scholar] [CrossRef]
- Mellado-Mojica, E.; de la Vara, L.E.; Lopez, M.G. Fructan active enzymes (FAZY) activities and biosynthesis of fructooligosaccharides in the vacuoles of Agave tequilana Weber Blue variety plants of different age. Planta 2017, 245, 265–281. [Google Scholar] [CrossRef] [PubMed]
- Nava-Cruza, N.Y.; Medina-Moralesa, M.A.; Martineza, J.L.; Rodrigueza, R.; Aguilara, C.N. Agave biotechnology: An overview. Crit. Rev. Biotechnol. 2015, 35, 546–559. [Google Scholar] [CrossRef] [PubMed]
- Arrizon, J.; Morel, S.; Gschaedler, A.; Monsan, P. Comparison of the water-soluble carbohydrate composition and fructan structures of Agave tequilana plants of different ages. Food Chem. 2010, 122, 123–130. [Google Scholar] [CrossRef]
- Pinos-Rodriguez, J.M.; Zamudio, M.; Gonzalez, S.S. The effect of plant age on the chemical composition of fresh and ensiled Agave salmiana leaves. S. Afr. J. Anim. Sci. 2008, 38, 43–50. [Google Scholar] [CrossRef]
- Aldrete-Herrera, P.I.; Lopez, M.G.; Medina-Torres, L.; Ragazzo-Sanchez, J.A.; Calderon-Santoyo, M.; Gonzalez-Avila, M.; Ortiz-Basurto, R.I. Physicochemical composition and apparent degree of polymerization of fructans in five wild Agave varieties: Potential industrial use. Foods 2019, 8, 404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aleixandre, J.L.; Lizama, V.; Alvarez, I.; García, M.J. Varietal differentiation of red wines in the Valencian region (Spain). J. Agric. Food Chem. 2002, 50, 751–755. [Google Scholar] [CrossRef]
- Rapp, A. Volatile flavour of wine: Correlation between instrumental analysis and sensory perception. Food/Nahrung 1998, 42, 351–363. [Google Scholar] [CrossRef]
- Sabon, I.; de Revel, G.; Kotseridis, Y.; Bertrand, A. Determination of volatile compounds in Grenache wines in relation with different terroirs in the Rhone Valley. J. Agric. Food Chem. 2002, 50, 6341–6345. [Google Scholar] [CrossRef]
- Pinal, L.; Cornejo, E.; Arellano, M.; Herrera, E.; Nuñez, L.; Arrizon, J.; Gschaedler, A. Effect of Agave tequilana age, cultivation field location and yeast strain on tequila fermentation process. J. Ind. Microbiol. Biotechnol. 2009, 36, 655–661. [Google Scholar] [CrossRef]
- Arrizon, J.; Fiore, C.; Acosta, G.; Romano, P.; Gschaedler, A. Fermentation behaviour and volatile compound production by agave and grape must yeasts in high sugar Agave tequilana and grape must fermentations. Antonie Van Leeuwenhoek 2006, 89, 181–189. [Google Scholar] [CrossRef]
- Cedeño, M.C. Tequila production. Crit. Rev. Biotechnol. 1995, 15, 1–11. [Google Scholar] [CrossRef]
- Pinal, L.; Cedeño, M.; Gutiérrez, H.; Alvarez-jacobs, J. Fermentation parameters influencing higher alcohol production in the tequila process. Biotechnol. Lett. 1997, 19, 45–47. [Google Scholar] [CrossRef]
- Prado-Ramírez, R.; Gonzáles-Alvarez, V.; Pelayo-Ortiz, C.; Casillas, N.; Estarrón, M.; Gómez-Hernández, H.E. The role of distillation on the quality of tequila. Int. J. Food Sci. Technol. 2005, 40, 701–708. [Google Scholar] [CrossRef]
- Dirección General de Normas. Bebidas Alcohólicas—Determinación de Azúcares, Azúcares Reductores Directos y Azucares Reductores Totales—Métodos de Ensayo (Prueba). In Diario Oficial de la Federación Mexico; The Norma Oficial Mexicana (NOM): NMX-V-006-NORMEX-2019; Dirección General de Normas: Mexico City, Mexico, 2020. [Google Scholar]
- Dirección General de Normas. Bebidas Alcohólicas—Determinación del Contenido Alcohólico (por Ciento de Alcohol en Volumen a 20 °C) (% Alc. Vol.)—Métodos de Ensayo (Prueba). In Diario Oficial de la Federación Mexico; The Norma Oficial Mexicana (NOM): NMX-V-013-NORMEX-2019; Dirección General de Normas: Mexico City, Mexico, 2020. [Google Scholar]
- Dirección General de Normas. Bebidas Alcohólicas—Determinación de Aldehídos, Ésteres, Metanol, y Alcoholes Superiores—Métodos en Ensayo (Prueba). In Diario Oficial de la Federación Mexico; The Norma Oficial Mexicana (NOM): NMX-V-005-NORMEX-2018; Dirección General de Normas: Mexico City, Mexico, 2020. [Google Scholar]
- Dirección General de Normas. Bebidas Alcohólicas—Determinación de Furfural—Métodos de Ensayo (Prueba). In Diario Oficial de la Federación Mexico; The Norma Oficial Mexicana (NOM): NMX-V-004-NORMEX-2018; Dirección General de Normas: Mexico City, Mexico, 2020. [Google Scholar]
- International Organisation of Vine and Wine. Determination of the Deuterium Distribution in Ethanol Derived from Fermentation of Grape Musts, Concentrated Grape Musts, Rectified Concentrated Grape Musts and Wines by Application of Nuclear Magnetic Resonance (Snif-Nmr/Rmn-Fins); OIV-OENO 426-2011; Fractionnement Isotop: Paris, France, 2011. [Google Scholar]
- Fonseca-Aguiñaga, R.; Gómez-Ruiz, H.; Miguel-Cruz, F.; Romero-Cano, L.A. Analytical characterization of tequila (silver class) using stable isotope analyses of C, O and atomic absorption as additional criteria to determine authenticity of beverage. Food Control. 2020, 112, 107161. [Google Scholar] [CrossRef]
- Van Roermund, C.W.T.; Waterham, H.R.; Ijlst, L.; Wanders, R.J.A. Fatty acid metabolism in Saccharomyces cerevisiae. Cell. Mol. Life Sci. 2003, 60, 1838–1851. [Google Scholar] [CrossRef]
- Maeda, I.; Delessert, S.; Hasegawa, S.; Seto, Y.; Zuber, S.; Poirier, Y. The peroxisomal acyl-CoA thioesterase pte1p from Saccharomyces cerevisiae is required for efficient degradation of short straight chain and branched chain fatty acids. J. Biol. Chem. 2006, 281, 11729–11735. [Google Scholar] [CrossRef] [Green Version]
- Mancilla-Margalli, N.A.; Lopez, M.G. Generation of Maillard compounds from inulin during the thermal processing of Agave tequilana Weber var. azul. J. Agric. Food Chem. 2002, 50, 806–812. [Google Scholar] [CrossRef]
- Eder, M.; Sanchez, I.; Brice, C.; Camarasa, C.; Legras, J.L.; Dequin, S. QTL mapping of volatile compound production in Saccharomyces cerevisiae during alcoholic fermentation. BMC Genom. 2018, 19, 166. [Google Scholar] [CrossRef]
- Arellano, M.; Pelayo, C.; Ramirez, J.; Rodriguez, I. Characterization of kinetic parameters and the formation of volatile compounds during the tequila fermentation by wild yeasts isolated from agave juice. J. Ind. Microbiol. Biotechnol. 2008, 35, 835–841. [Google Scholar] [CrossRef]
- Amaya-Delgado, L.; Herrera-López, E.J.; Arrizon, J.; Arellano-Plaza, M.; Gschaedler, A. Performance evaluation of Pichia kluyveri, Kluyveromyces marxianus and Saccharomyces cerevisiae in industrial tequila fermentation. World J. Microbiol. Biotechnol. 2013, 29, 875–881. [Google Scholar] [CrossRef]
- Prado-Jaramillo, N.; Estarrón-Espinosa, M.; Escalona-Buendía, H.; Cosío-Ramírez, R.; Martín-del-Campo, S.T. Volatile compounds generation during different stages of the tequila production process. A preliminary study. LWT Food Sci. Technol. 2015, 61, 471–483. [Google Scholar] [CrossRef]
- Aguilar-Cisneros, B.O.; López, M.; Richling, E.; Heckel, F.; Schreier, P. Tequila authenticity assessment by headspace SPME-HRGC-IRMS Analysis of 13 C/12 C and 18 O/16 O ratios of ethanol. J. Agric. Food Chem. 2002, 50, 7520–7523. [Google Scholar] [CrossRef]
- Hoefs, J. Stable Isotope Geochemistry, 7th ed.; Springer: Cham, Switzerland, 2015; ISBN 978-3-319-19716-6. [Google Scholar]
- Park, R.; Epstein, S. Carbon isotope fractionation during photosynthesis. Geochim. Cosmochim. Acta 1960, 21, 110–126. [Google Scholar] [CrossRef]
- Fonseca-Aguiñaga, R.; Warren-Vega, W.M.; Miguel-Cruz, F.; Romero-Cano, L.A. Isotopic characterization of 100% Agave tequila (silver, aged and extra-aged class) for its use as an additional parameter in the determination of the authenticity of the beverage maturation time. Molecules 2021, 26, 1719. [Google Scholar] [CrossRef]
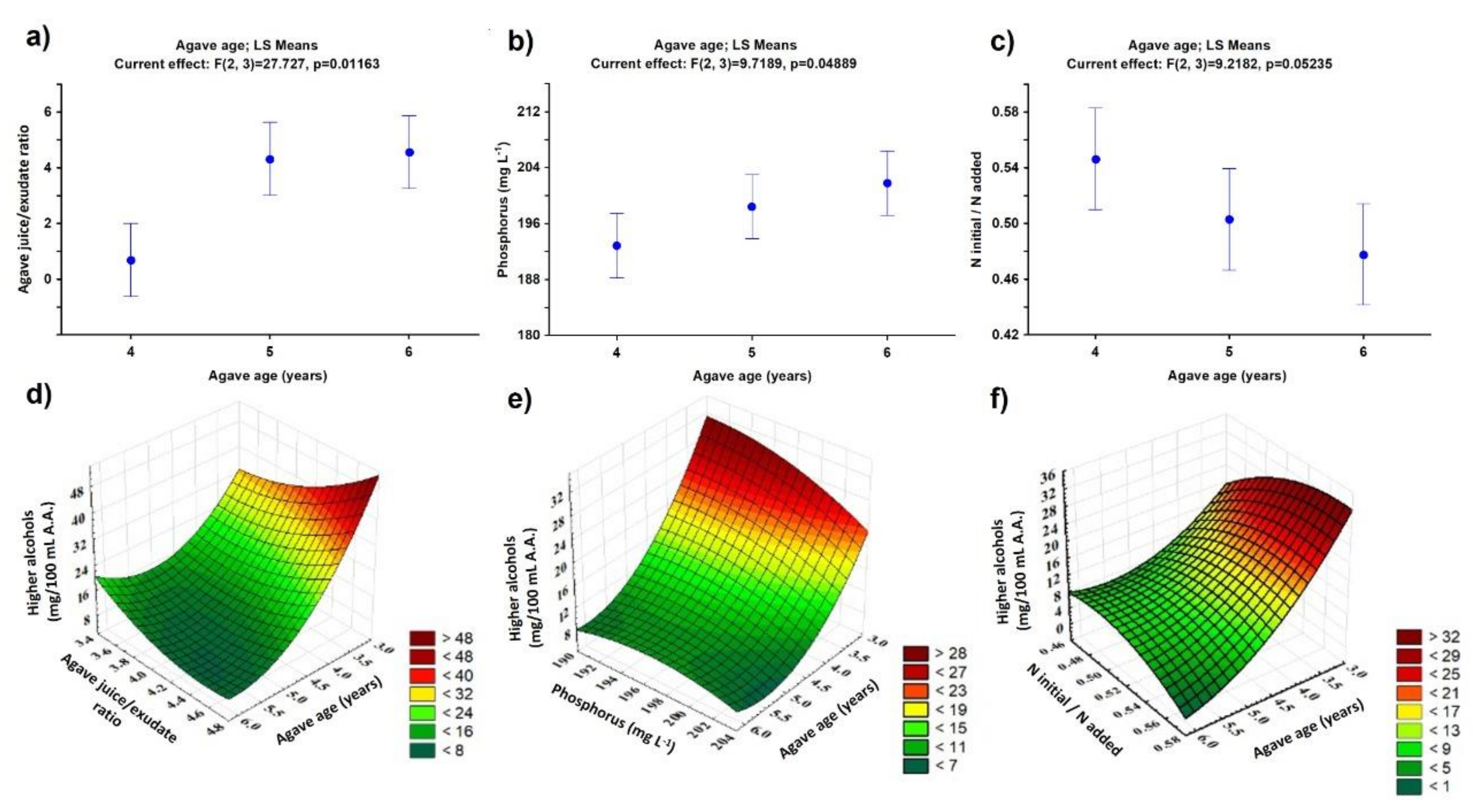
| Age (Years) | Height (cm) | Diameter (cm) | Average Heart wt. (kg) | TRS (%) | Sugars | Percentage in Must | Nitrogen Content in Juice (mg L−1) | ||
|---|---|---|---|---|---|---|---|---|---|
| °Brix | Juice (%) | Exudate (%) | Initial | Added | |||||
| 4 | 37.5 ± 1.10 | 41.0 ± 2.20 | 18.4 ± 0.60 | 15.21 ± 0.35 | 19.4 ± 0.60 | 78.8 ± 0.90 | 20.9 ± 0.90 | 95.4 ± 2.80 | 174.4 ± 2.80 |
| 5 | 40.5 ± 1.60 | 43.0 ± 1.70 | 20.7 ± 0.80 | 18.84 ± 0.45 | 23.0 ± 0.90 | 80.9 ± 0.50 | 18.7 ± 0.50 | 90.4 ± 1.50 | 179.4 ± 1.50 |
| 6 | 46.5 ± 2.30 | 49.5 ± 2.40 | 25.5 ± 1.30 | 19.21 ± 0.35 | 24.2 ± 1.20 | 81.8 ± 0.70 | 17.9 ± 0.70 | 87.4 ± 0.64 | 182.4 ± 0.60 |
| Congeners (mg/100 mL A.A.) | Agave Age (Years) | ||
|---|---|---|---|
| Batch 1 (4 Years Agave) | Batch 2 (5 Years Agave) | Batch 3 (6 Years Agave) | |
| Fermentation | |||
| Methanol | 0.08 ± 0.11 | 1.25 ± 0.40 | 0.13 ± 0.19 |
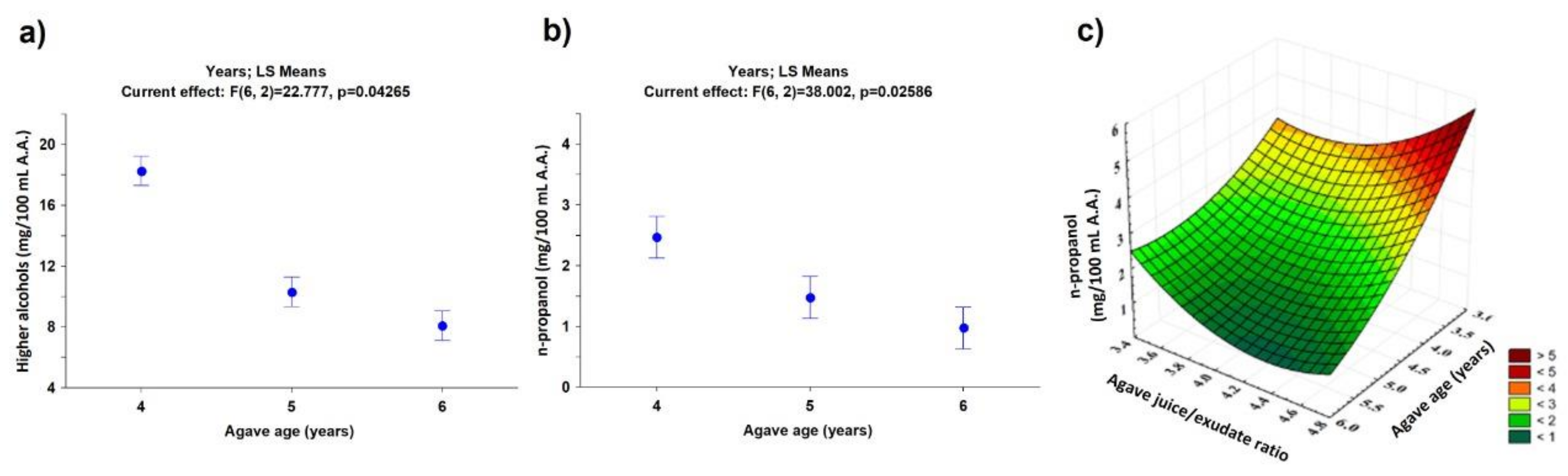
| Higher alcohols | 18.26 ± 0.26 | 10.30 ± 0.61 | 8.05 ± 0.31 |
| Esters | 0.00 | 0.00 | 0.00 |
| Aldehydes | 0.00 | 0.00 | 0.00 |
| Furfural | 0.00 | 0.00 | 0.00 |
| First distillation | |||
| Methanol | 573.90 ± 21.96 | 520.26 ± 8.66 | 481.06 ± 3.14 |
| Higher alcohols | 338.56 ± 18.53 | 315.12 ± 3.61 | 294.85 ± 5.90 |
| Esters | 11.65 ± 0.13 | 19.93 ± 1.38 | 20.41 ± 1.52 |
| Aldehydes | 7.36 ± 7.16 | 1.15 ± 0.28 | 0.83 ± 0.05 |
| Furfural | 0.71 ± 0.08 | 2.08 ± 0.03 | 3.36 ± 0.36 |
| Second distillation | |||
| Methanol | 244.11 ± 32.75 | 217.35 ± 17.59 | 192.19 ± 18.79 |
| Higher alcohols | 293.68 ± 7.55 | 282.54 ± 25.77 | 247.31 ± 1.25 |
| Esters | 34.32 ± 4.75 | 30.32 ± 12.17 | 27.03 ± 5.05 |
| Aldehydes | 4.76 ± 0.11 | 3.53 ± 0.94 | 3.25 ± 0.04 |
| Furfural | 0.42 ± 0.12 | 0.51 ± 0.04 | 0.34 ± 0.04 |
| Higher Alcohol (mg/100 mL A.A.) | Agave Age (Years) | ||
|---|---|---|---|
| Batch 1 (4 Years Agave) | Batch 2 (5 Years Agave) | Batch 3 (6 Years Agave) | |
| Fermentation | |||
| 2-butanol | 0.00 | 0.00 | 0.00 |
| n-propanol | 2.47 ± 0.18 | 1.48 ± 0.09 | 0.97 ± 0.16 |
| 2-methyl-1-propanol | 4.85 ± 0.37 | 4.08 ± 0.19 | 3.82 ± 0.09 |
| n-butanol | 0.00 | 0.00 | 0.00 |
| 3-methyl-1-butanol | 10.94 ± 0.30 | 4.75 ± 0.32 | 3.29 ±0.06 |
| First distillation | |||
| 2-butanol | 0.00 | 0.00 | 0.00 |
| n-propanol | 90.83 ± 0.17 | 78.94 ± 0.60 | 79.00 ± 1.66 |
| 2-methyl-1-propanol | 69.86 ± 1.76 | 65.55 ± 0.75 | 64.58 ± 0.87 |
| n-butanol | 0.00 | 0.00 | 0.00 |
| 3-methyl-1-butanol | 177.87 ± 16.60 | 170.63 ± 3.77 | 151.27 ± 3.37 |
| Second distillation | |||
| 2-butanol | 0.62 ± 0.12 | 0.54 ± 0.13 | 0.18 ± 0.08 |
| n-propanol | 61.21 ± 0.40 | 58.32 ± 5.45 | 46.04 ± 3.39 |
| 2-methyl-1-propanol | 63.19 ± 4.36 | 57.18 ± 4.09 | 52.63 ± 0.91 |
| n-butanol | 0.73 ± 0.11 | 0.69 ± 0.21 | 0.54 ± 0.11 |
| 3-methyl-1-butanol | 167.43 ± 11.31 | 165.25 ± 15.88 | 147.42 ± 1.27 |
| Age (years) | Methanol (mg/100 mL A.A.) | Higher Alcohols (mg/100 mL A.A.) | Esters (mg/100 mL A.A.) | Aldehydes (mg/100 mL A.A.) | Furfural (mg/100 mL A.A.) | δ 13CVPDB (‰) | δ 18OVSMOW (‰) |
|---|---|---|---|---|---|---|---|
| 4 | 244.11 ± 32.75 | 293.68 ± 7.55 | 34.32 ± 4.75 | 4.76 ± 0.11 | 0.42 ± 0.12 | −13.37 ± 0.05 | 20.91 ± 1.72 |
| 5 | 217.35 ± 17.59 | 282.54 ± 25.77 | 30.32 ± 12.17 | 3.53 ± 0.94 | 0.51 ± 0.04 | −13.36 ± 0.13 | 20.11 ± 0.81 |
| 6 | 192.19 ± 18.79 | 247.31 ± 1.25 | 27.03 ± 5.05 | 3.25 ± 0.04 | 0.34 ± 0.04 | −13.49 ± 0.17 | 20.56 ± 1.25 |
| Age (years) | 2-Butanol (mg/100 mL A.A.) | N-Propanol (mg/100 mL A.A.) | 2-Methyl-1-propanol (mg/100 mL A.A.) | 1-Butanol (mg/100 mL A.A.) | 3-Methyl-1-butanol (mg/100 mL A.A.) |
|---|---|---|---|---|---|
| 4 | 0.62 ± 0.12 | 61.21 ± 0.40 | 63.19 ± 4.36 | 0.73 ± 0.11 | 167.43 ± 11.31 |
| 5 | 0.54 ± 0.13 | 58.32 ± 5.45 | 57.18 ± 4.09 | 0.69 ± 0.21 | 165.25 ± 15.88 |
| 6 | 0.18 ± 0.08 | 46.04 ± 3.39 | 52.63 ± 0.91 | 0.54 ± 0.11 | 147.42 ± 1.27 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acosta-Salazar, E.; Fonseca-Aguiñaga, R.; Warren-Vega, W.M.; Zárate-Guzmán, A.I.; Zárate-Navarro, M.A.; Romero-Cano, L.A.; Campos-Rodríguez, A. Effect of Age of Agave tequilana Weber Blue Variety on Quality and Authenticity Parameters for the Tequila 100% Agave Silver Class: Evaluation at the Industrial Scale Level. Foods 2021, 10, 3103. https://doi.org/10.3390/foods10123103
Acosta-Salazar E, Fonseca-Aguiñaga R, Warren-Vega WM, Zárate-Guzmán AI, Zárate-Navarro MA, Romero-Cano LA, Campos-Rodríguez A. Effect of Age of Agave tequilana Weber Blue Variety on Quality and Authenticity Parameters for the Tequila 100% Agave Silver Class: Evaluation at the Industrial Scale Level. Foods. 2021; 10(12):3103. https://doi.org/10.3390/foods10123103
Chicago/Turabian StyleAcosta-Salazar, Efraín, Rocío Fonseca-Aguiñaga, Walter M. Warren-Vega, Ana I. Zárate-Guzmán, Marco A. Zárate-Navarro, Luis A. Romero-Cano, and Armando Campos-Rodríguez. 2021. "Effect of Age of Agave tequilana Weber Blue Variety on Quality and Authenticity Parameters for the Tequila 100% Agave Silver Class: Evaluation at the Industrial Scale Level" Foods 10, no. 12: 3103. https://doi.org/10.3390/foods10123103
APA StyleAcosta-Salazar, E., Fonseca-Aguiñaga, R., Warren-Vega, W. M., Zárate-Guzmán, A. I., Zárate-Navarro, M. A., Romero-Cano, L. A., & Campos-Rodríguez, A. (2021). Effect of Age of Agave tequilana Weber Blue Variety on Quality and Authenticity Parameters for the Tequila 100% Agave Silver Class: Evaluation at the Industrial Scale Level. Foods, 10(12), 3103. https://doi.org/10.3390/foods10123103










