Hexanal as a Predictor of Development of Oxidation Flavor in Cured and Uncured Deli Meat Products as Affected by Natural Antioxidants
Abstract
1. Introduction
2. Materials and Methods
2.1. Production of Deli Meat Products with Natural Antioxidants
2.2. Solid-Phase Microextraction Gas-Chromatography (SPME-GC)
2.3. Sensory Analysis
2.4. Measurement of pH
2.5. Statistical Analysis
3. Results and Discussion
3.1. Headspace Hexanal and pH
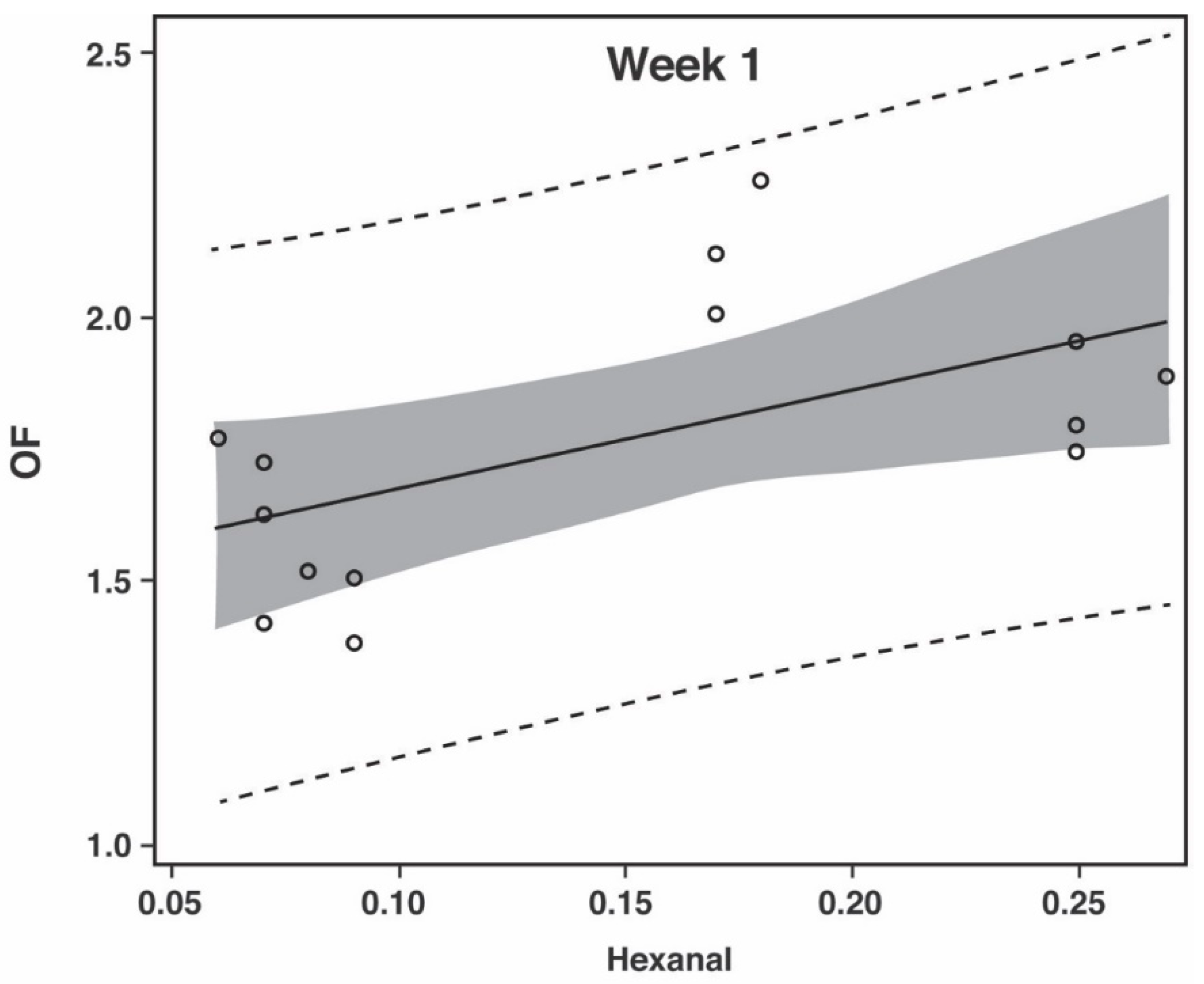
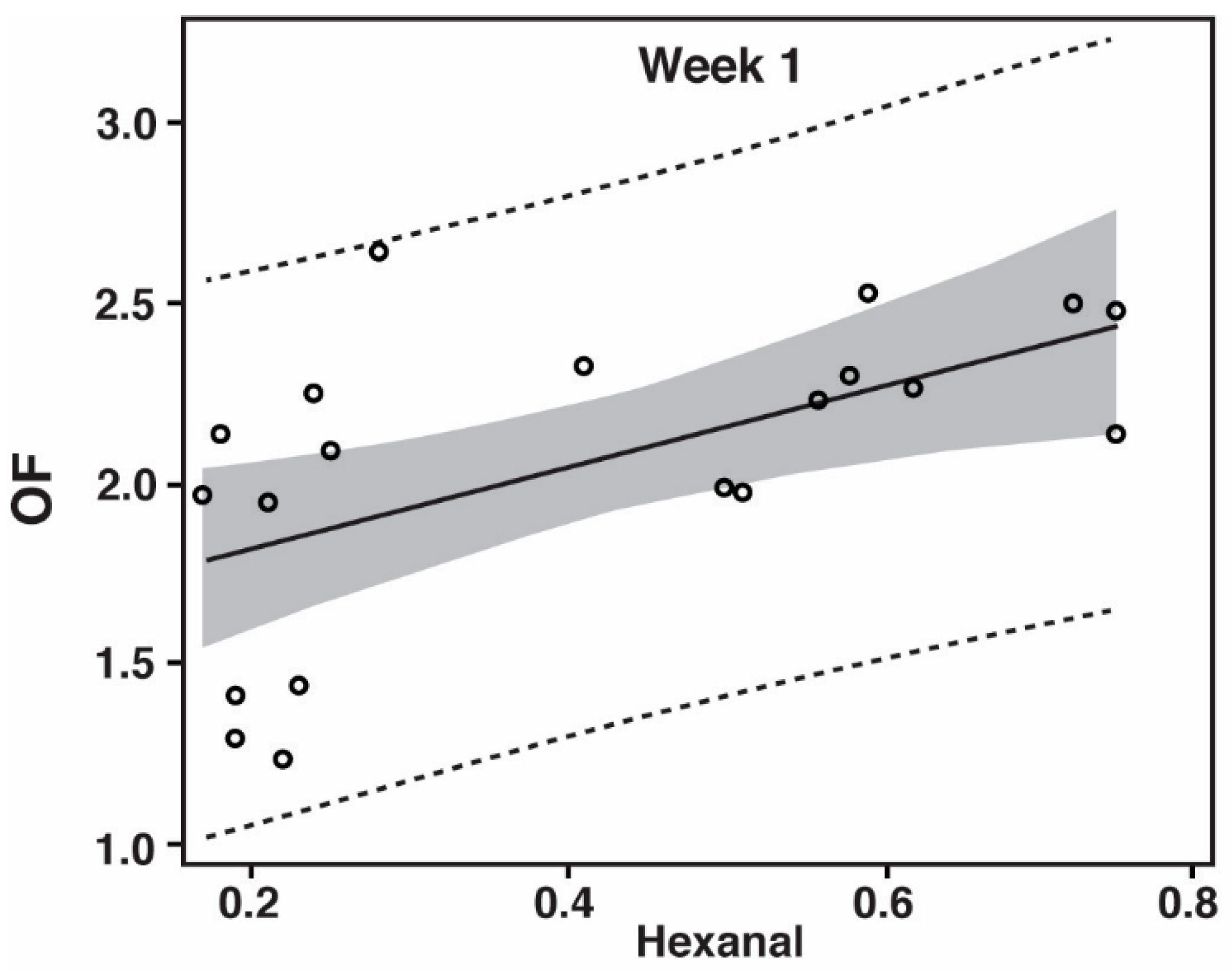
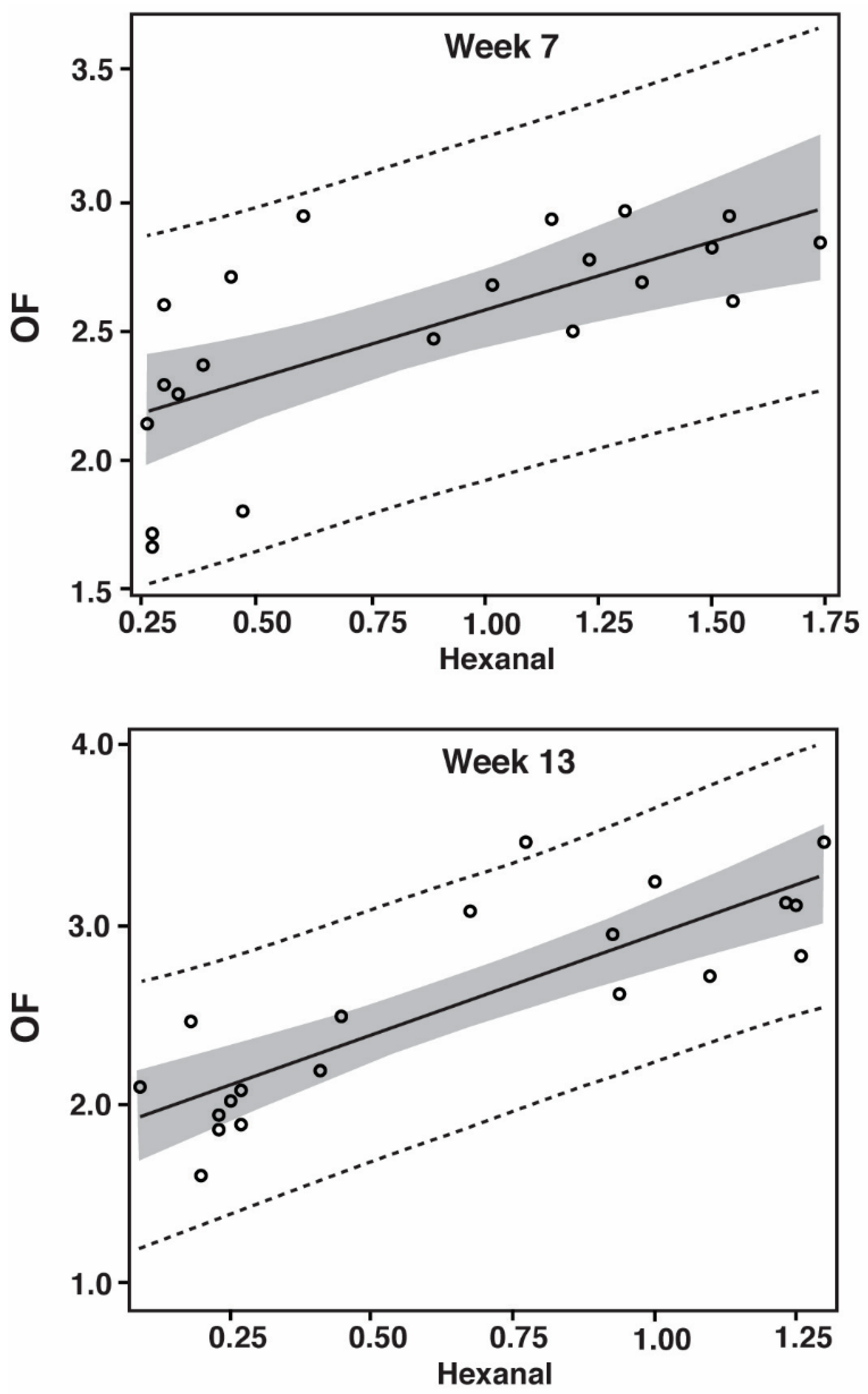
3.2. Oxidation Flavor (OF)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Lee, E.J.; Ahn, D.U. Quality characteristics of irradiated turkey breast rolls formulated with plum extract. Meat Sci. 2005, 71, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Pires, M.A.; Munekata, P.E.S.; Villanueva, N.D.M.; Tonin, F.G.; Baldin, J.C.; Rocha, Y.J.P.; Carvalho, L.T.; Rodrigues, I.; Trindade, M.A. The Antioxidant Capacity of Rosemary and Green Tea Extracts to Replace the Carcinogenic Antioxidant (BHA) in Chicken Burgers. J. Food Qual. 2017, 2017, 2409527. [Google Scholar] [CrossRef]
- Rababah, T.; Hettiarachchy, N.; Horax, R.; Cho, M. Thiobarbituric Acid Reactive Substances and Volatile Compounds in Chicken Breast Meat Infused with Plant Extracts and Subjected to Electron Beam Irradiation. Poult. Sci. 2006, 85, 1107–1113. [Google Scholar] [CrossRef]
- Sampaio, G.R.; Saldanha, T.; Soares, R.A.M.; Torres, E.A.F.S. Effect of natural antioxidant combinations on lipid oxidation in cooked chicken meat during refrigerated storage. Food Chem. 2012, 135, 1383–1390. [Google Scholar] [CrossRef]
- Vaithiyanathan, S.; Naveena, B.M.; Muthukumar, M.; Girish, P.S.; Kondaiah, N. Effect of dipping in pomegranate (Punica granatum) fruit juice phenolic solution on the shelf life of chicken meat under refrigerated storage (4 °C). Meat Sci. 2011, 88, 409–414. [Google Scholar] [CrossRef]
- Lee, C.-H.; Reed, J.D.; Richards, M.P. Ability of Various Polyphenolic Classes from Cranberry to Inhibit Lipid Oxidation in Mechanically Separated Turkey and Cooked Ground Pork. J. Muscle Foods 2006, 17, 248–266. [Google Scholar] [CrossRef]
- Hayes, J.E.; Stepanyan, V.; O’Grady, M.; Allen, P.; Kerry, J. Evaluation of the effects of selected phytochemicals on quality indices and sensorial properties of raw and cooked pork stored in different packaging systems. Meat Sci. 2010, 85, 289–296. [Google Scholar] [CrossRef]
- Rojas, M.C.; Brewer, M.S. Effect of Natural Antioxidants on Oxidative Stability of Cooked, Refrigerated Beef and Pork. J. Food Sci. 2007, 72, S282–S288. [Google Scholar] [CrossRef] [PubMed]
- Rojas, M.C.; Brewer, M.S. Effect of natural antioxidants on oxidative stability of frozen, vacuum-packaged beef and pork. J. Food Qual. 2008, 31, 173–188. [Google Scholar] [CrossRef]
- Schilling, M.W.; Pham, A.J.; Williams, J.B.; Xiong, Y.L.; Dhowlaghar, N.; Tolentino, A.C.; Kin, S. Changes in the physiochemical, microbial, and sensory characteristics of fresh pork sausage containing rosemary and green tea extracts during retail display. Meat Sci. 2018, 143, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Lara, M.S.; Gutierrez, J.I.; Timón, M.; Andrés, A.I. Evaluation of two natural extracts (Rosmarinus officinalis L. and Melissa officinalis L.) as antioxidants in cooked pork patties packed in MAP. Meat Sci. 2011, 88, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Sasse, A.; Colindres, P.; Brewer, M.S. Effect of Natural and Synthetic Antioxidants on the Oxidative Stability of Cooked, Frozen Pork Patties. J. Food Sci. 2009, 74, S30–S35. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, T.L.; Kerry, J.P.; Kerry, J.F.; Lynch, P.B.; Buckley, D.J. Evaluation of the antioxidant potential of natural food/plant extracts as compared with synthetic antioxidants and vitamin E in raw and cooked pork patties. Meat Sci. 2001, 58, 45–52. [Google Scholar] [CrossRef]
- Jayathilakan, K.; Sharma, G.K.; Radhakrishna, K.; Bawa, A.S. Antioxidant potential of synthetic and natural antioxidants and its effect on warmed-over-flavour in different species of meat. Food Chem. 2007, 105, 908–916. [Google Scholar] [CrossRef]
- Laguerre, M.; Bayrasy, C.; Panya, A.; Weiss, J.; McClements, D.; Lecomte, J.; Decker, E.; Villeneuve, P. What Makes Good Antioxidants in Lipid-Based Systems? The Next Theories Beyond the Polar Paradox. Crit. Rev. Food Sci. Nutr. 2015, 55, 183–201. [Google Scholar] [CrossRef]
- Ribeiro, J.S.; Santos, M.J.M.C.; Silva, L.K.R.; Pereira, L.C.L.; Santos, I.A.; da Silva Lannes, S.C.; da Silva, M.V. Natural antioxidants used in meat products: A brief review. Meat Sci. 2019, 148, 181–188. [Google Scholar] [CrossRef]
- Candan, T.; Ba Datli, A. Use of Natural Antioxidants in Poultry Meat. Celal Bayar Univ. Fen Bilimleri Derg. 2017, 13, 279–291. [Google Scholar]
- Borella, T.G.; Peccin, M.M.; Mazon, J.M.; Roman, S.S.; Cansian, R.L.; Soares, M.B.A. Effect of rosemary (Rosmarinus officinalis) antioxidant in industrial processing of frozen-mixed hamburger during shelf life. J. Food Process. Preserv. 2019, 43, e14092. [Google Scholar] [CrossRef]
- Oswell, N.J.; Thippareddi, H.; Pegg, R.B. Practical use of natural antioxidants in meat products in the U.S.: A review. Meat Sci. 2018, 145, 469–479. [Google Scholar] [CrossRef]
- Munekata, P.E.S.; Gullón, B.; Pateiro, M.; Tomasevic, I.; Domínguez, R.; Lorenzo, J.M. Natural Antioxidants from Seeds and Their Application in Meat Products. Antioxidants 2020, 9, 815. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Pateiro, M.; Domínguez, R.; Barba, F.J.; Putnik, P.; Kovačević, D.B.; Shpigelman, A.; Granato, D.; Franco, D. Berries extracts as natural antioxidants in meat products: A review. Food Res. Int. 2018, 106, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Bak, K.H.; Rankin, S.A.; Richards, M.P. Hexanal as a marker of oxidation flavour in sliced and uncured deli turkey with and without phosphates using rosemary extracts. Int. J. Food Sci. Technol. 2020. [Google Scholar] [CrossRef]
- Brewer, M.S. Natural Antioxidants: Sources, Compounds, Mechanisms of Action, and Potential Applications. Compr. Rev. Food Sci. Food Saf. 2011, 10, 221–247. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Lipid oxidation and improving the oxidative stability. Chem. Soc. Rev. 2010, 39, 4067–4079. [Google Scholar] [CrossRef]
- Jiang, J.; Xiong, Y.L. Natural antioxidants as food and feed additives to promote health benefits and quality of meat products: A review. Meat Sci. 2016, 120, 107–117. [Google Scholar] [CrossRef]
- Estévez, M.; Morcuende, D.; Ventanas, S. Determination of Oxidation. In Handbook of Muscle Foods Analysis, 1st ed.; Nollet, L.M.L., Toldrá, F., Eds.; CRC Press: Boca Raton, FL, USA, 2009; pp. 221–239. [Google Scholar]
- Shahidi, F.; Zhong, Y. Lipid Oxidation: Measurement methods. In Bailey’s Industrial Oil and Fat Products; Shahidi, F., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005; pp. 357–385. [Google Scholar]
- Drumm, T.D.; Spanier, A.M. Changes in the content of lipid autoxidation anad sulfur-containing compounds in cooked beef during storage. J. Agric. Food Chem. 1991, 39, 336–343. [Google Scholar] [CrossRef]
- Elmore, J. Aroma. In Handbook of Muscle Foods Analysis, 1st ed.; Nollet, L.M.L., Toldrá, F., Eds.; CRC Press: Boca Raton, FL, USA, 2009; pp. 241–262. [Google Scholar]
- Lawless, H. The sense of smell in food quality and sensory evaluation. J. Food Qual. 1991, 14, 33–60. [Google Scholar] [CrossRef]
- Singham, P.; Birwal, P.; Yadav, B.K. Importance of Objective and Subjective Measurement of Food Quality and their Inter-relationship. J. Food Process. Technol. 2015, 6, 1–7. [Google Scholar] [CrossRef]
- Caul, J.F. The Profile Method of Flavor Analysis. In Advances in Food Research; Mrak, E.M., Stewart, G.F., Eds.; Academic Press: Cambridge, MA, USA, 1957; Volume 7, pp. 1–40. [Google Scholar]
- Igene, J.O.; Yamauchi, K.; Pearson, A.M.; Gray, J.I.; Aust, S.D. Mechanisms by which nitrite inhibits the development of warmed-over flavour (WOF) in cured meat. Food Chem. 1985, 18, 1–18. [Google Scholar] [CrossRef]
- Trout, G.R.; Dale, S. Prevention of Warmed-Over Flavor in Cooked Beef: Effect of Phosphate Type, Phosphate Concentration, a Lemon Juice/Phosphate Blend, and Beef Extract. J. Agric. Food Chem. 1990, 38, 665–669. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Revisiting the Polar Paradox Theory: A Critical Overview. J. Agric. Food Chem. 2011, 59, 3499–3504. [Google Scholar] [CrossRef] [PubMed]
- Feiner, G. Additives: Phosphates, salts (sodium chloride and potassium chloride, citrate, lactate) and hydrocolloids. In Meat Products Handbook. Practical Science and Technology; Woodhead Publishing Limited: Cambridge, UK, 2006; pp. 72–88. [Google Scholar]
- Papadima, S.N.; Arvanitoyannis, I.; Bloukas, J.G.; Fournitzis, G.C. Chemometric model for describing Greek traditional sausages. Meat Sci. 1999, 51, 271–277. [Google Scholar] [CrossRef]
- Hamasaki, Y.; Ayaki, M.; Fuchu, H.; Sugiyama, M.; Morita, H. Behavior of psychrotrophic lactic acid bacteria isolated from spoiling cooked meat products. Appl. Environ. Microbiol. 2003, 69, 3668–3671. [Google Scholar] [CrossRef]
- Shahidi, F.; Pegg, R.B. Hexanal as an indicator of meat flavor deterioration. J. Food Lipids 1994, 1, 177–186. [Google Scholar] [CrossRef]
- Shahidi, F.; Yun, J.; Rubin, L.J.; Wood, D.F. The Hexanal Content as an Indicator of Oxidative Stability and Flavour Acceptability in Cooked Ground Pork. Can. Inst. Food Sci. Technol. J. 1987, 20, 104–106. [Google Scholar] [CrossRef]
- Angelo, A.J.S.; Vercellotti, J.R.; Legendre, M.G.; VinnelT, C.H.; Kuan, J.W.; James, C., Jr.; Dupuy, H.P. Chemical and Instrumental Analyses of Warmed-Over Flavor in Beef. J. Food Sci. 1987, 52, 1163–1168. [Google Scholar] [CrossRef]

| Treatment | Antioxidant | Level (mg/kg) | Hexanal (mg/kg Meat) | |||||
|---|---|---|---|---|---|---|---|---|
| Week 1 | Week 7 | Week 13 | ||||||
| Estimate | SE | Estimate | SE | Estimate | SE | |||
| T1 | Control | 0 | 0.14 a | 0.01 | 0.16 a | 0.02 | 0.16 a | 0.01 |
| T2 | Guardian 09 | 200 | 0.09 b | 0.01 | 0.13 ab | 0.02 | 0.13 ab | 0.01 |
| T3 | Guardian 09 | 400 | 0.08 b | 0.01 | 0.14 ab | 0.02 | 0.10 b | 0.01 |
| T4 | Guardian 09 | 600 | 0.09 b | 0.01 | 0.11 bc | 0.02 | 0.09 bc | 0.01 |
| T5 | StabilEnhance OSR D 2.5 | 200 | 0.08 b | 0.01 | 0.11 bc | 0.02 | 0.10 b | 0.01 |
| T6 | StabilEnhance OSR D 2.5 | 400 | 0.08 b | 0.01 | 0.08 c | 0.02 | 0.06 c | 0.01 |
| T7 | StabilEnhance OSR D 2.5 | 600 | 0.08 b | 0.01 | 0.10 bc | 0.02 | 0.10 bc | 0.01 |
| Treatment | Antioxidant | Level (mg/kg) | Hexanal (mg/kg Meat) | ||||
|---|---|---|---|---|---|---|---|
| Week 1 | Week 7 | ||||||
| Estimate | SE | Estimate | SE | ||||
| P | T1 | Control | 0 | 0.07 c | 0.01 | 0.81 bcde | 0.17 |
| T2 | Guardian 09 | 400 | 0.07 c | 0.01 | 0.23 g | 0.17 | |
| T3 | Guardian 09 | 600 | 0.06 c | 0.01 | 0.35 efg | 0.17 | |
| T4 | Guardian 09 | 800 | 0.07 c | 0.01 | 0.30 fg | 0.17 | |
| T5 | StabilEnhance OSR D 2.5 | 400 | 0.09 c | 0.01 | 0.71 cdef | 0.17 | |
| T6 | StabilEnhance OSR D 2.5 | 600 | 0.09 c | 0.01 | 0.68 cdefg | 0.17 | |
| T7 | StabilEnhance OSR D 2.5 | 800 | 0.08 c | 0.01 | 0.52 defg | 0.17 | |
| nP | T1 | Control | 0 | 0.27 a | 0.01 | 1.52 a | 0.17 |
| T2 | Guardian 09 | 400 | 0.17 b | 0.01 | 0.87 bcd | 0.17 | |
| T3 | Guardian 09 | 600 | 0.17 b | 0.01 | 0.90 bcd | 0.17 | |
| T4 | Guardian 09 | 800 | 0.18 b | 0.01 | 1.01 bc | 0.17 | |
| T5 | StabilEnhance OSR D 2.5 | 400 | 0.25 a | 0.01 | 1.12 abc | 0.17 | |
| T6 | StabilEnhance OSR D 2.5 | 600 | 0.25 a | 0.01 | 1.08 abc | 0.17 | |
| T7 | StabilEnhance OSR D 2.5 | 800 | 0.25 a | 0.01 | 1.25 ab | 0.17 | |
| Treatment | Antioxidant | Level (mg/kg) | Hexanal (mg/kg meat) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Week 1 | Week 7 | Week 13 | |||||||
| Estimate | SE | Estimate | SE | Estimate | SE | ||||
| P | T1 | Control | 0 | 0.19 fg | 0.04 | 0.47 hi | 0.06 | 0.27 gh | 0.06 |
| T2 | Guardian 20S | 300 | 0.22 fg | 0.04 | 0.27 jk | 0.06 | 0.20 h | 0.06 | |
| T3 | Guardian 20S | 400 | 0.19 fg | 0.04 | 0.27 jk | 0.06 | 0.23 h | 0.06 | |
| T4 | Guardian 20S | 500 | 0.23 fg | 0.04 | 0.26 k | 0.06 | 0.23 h | 0.06 | |
| T5 | Guardian 09 | 400 | 0.24 fg | 0.04 | 0.44 hij | 0.06 | 0.09 h | 0.06 | |
| T6 | Guardian 09 | 600 | 0.18 g | 0.04 | 0.30 ijk | 0.06 | 0.18 h | 0.06 | |
| T7 | Guardian 09 | 800 | 0.28 f | 0.04 | 0.60 h | 0.06 | 0.46 f | 0.06 | |
| T8 | StabilEnhance OSR 4 | 400 | 0.25 fg | 0.04 | 0.33 ijk | 0.06 | 0.41 fg | 0.06 | |
| T9 | StabilEnhance OSR 4 | 600 | 0.21 fg | 0.04 | 0.38 ijk | 0.06 | 0.27 gh | 0.06 | |
| T10 | StabilEnhance OSR 4 | 800 | 0.17 g | 0.04 | 0.30 ijk | 0.06 | 0.25 gh | 0.06 | |
| nP | T1 | Control | 0 | 0.75 a | 0.04 | 1.55 b | 0.06 | 1.26 ab | 0.06 |
| T2 | Guardian 20S | 300 | 0.51 d | 0.04 | 1.20 de | 0.06 | 0.94 cd | 0.06 | |
| T3 | Guardian 20S | 400 | 0.50 de | 0.04 | 1.02 fg | 0.06 | 1.10 bc | 0.06 | |
| T4 | Guardian 20S | 500 | 0.41 e | 0.04 | 0.89 g | 0.06 | 1.23 ab | 0.06 | |
| T5 | Guardian 09 | 400 | 0.58 cd | 0.04 | 1.15 ef | 0.06 | 1.00 c | 0.06 | |
| T6 | Guardian 09 | 600 | 0.59 cd | 0.04 | 1.50 bc | 0.06 | 1.25 ab | 0.06 | |
| T7 | Guardian 09 | 800 | 0.56 cd | 0.04 | 1.31 de | 0.06 | 0.77 de | 0.06 | |
| T8 | StabilEnhance OSR 4 | 400 | 0.62 bc | 0.04 | 1.54 b | 0.06 | 0.67 e | 0.06 | |
| T9 | StabilEnhance OSR 4 | 600 | 0.75 a | 0.04 | 1.35 cd | 0.06 | 1.30 a | 0.06 | |
| T10 | StabilEnhance OSR 4 | 800 | 0.72 ab | 0.04 | 1.74 a | 0.06 | 0.93 cd | 0.06 | |
| Significance of Correlation between Hexanal and Oxidation Flavor | |||
|---|---|---|---|
| Week 1 | Week 7 | Week 13 | |
| CDT | p = 0.7960 | p = 0.0379 | p = 0.4269 |
| CF | p = 0.0312 | p = 0.0005 | --- |
| PP | p = 0.0071 | p = 0.0009 | p < 0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bak, K.H.; Richards, M.P. Hexanal as a Predictor of Development of Oxidation Flavor in Cured and Uncured Deli Meat Products as Affected by Natural Antioxidants. Foods 2021, 10, 152. https://doi.org/10.3390/foods10010152
Bak KH, Richards MP. Hexanal as a Predictor of Development of Oxidation Flavor in Cured and Uncured Deli Meat Products as Affected by Natural Antioxidants. Foods. 2021; 10(1):152. https://doi.org/10.3390/foods10010152
Chicago/Turabian StyleBak, Kathrine Holmgaard, and Mark P. Richards. 2021. "Hexanal as a Predictor of Development of Oxidation Flavor in Cured and Uncured Deli Meat Products as Affected by Natural Antioxidants" Foods 10, no. 1: 152. https://doi.org/10.3390/foods10010152
APA StyleBak, K. H., & Richards, M. P. (2021). Hexanal as a Predictor of Development of Oxidation Flavor in Cured and Uncured Deli Meat Products as Affected by Natural Antioxidants. Foods, 10(1), 152. https://doi.org/10.3390/foods10010152





