Evaluation of Different Dentifrice Compositions for Increasing the Hardness of Demineralized Enamel: An in Vitro Study
Abstract
:1. Introduction
2. Materials and Methods
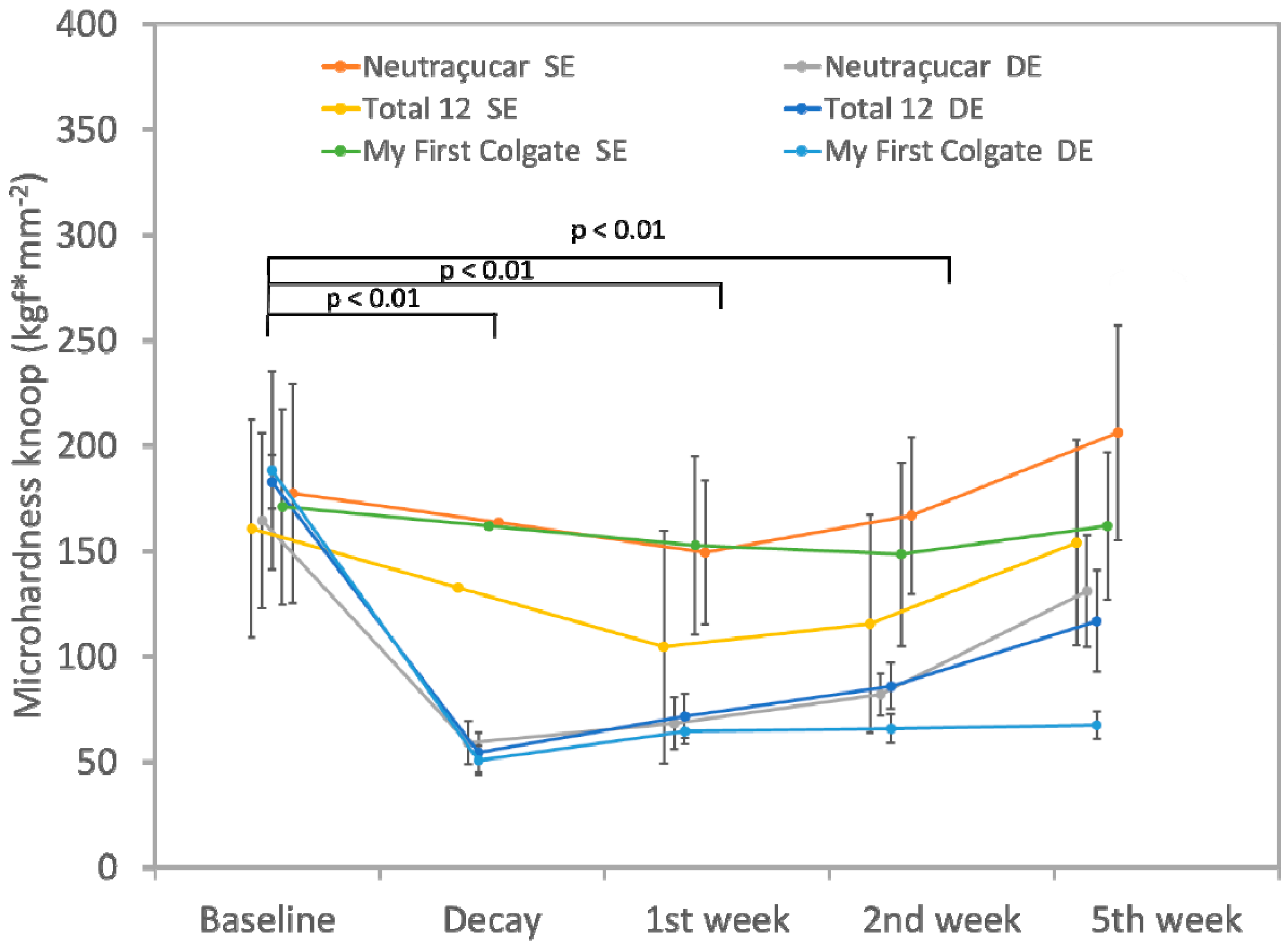
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cummins, D. Dental caries: A disease which remains a public health concern in the 21st century. The exploration of a breakthrough technology for caries prevention. J. Clin. Dent. 2013, 24, A1–A14. [Google Scholar] [PubMed]
- Featherstone, J.D. The caries balance: Contributing factors and early detection. J. Calif. Dent. Assoc. 2003, 31, 129–133. [Google Scholar] [PubMed]
- Featherstone, J.D. Caries prevention and reversal based on the caries balance. Pediatr. Dent. 2006, 28, 128–132. [Google Scholar] [PubMed]
- Arends, J.; Christoffersen, J. The nature of early caries lesions in enamel. J. Dent. Res. 1986, 65, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Souza, M.LR.; Cury, J.A.; Tenuta, L.M.A.; Zhang, Y.P.; Mateo, L.R.; Cummins, D. Comparing the efficacy of a dentifrice containing 1.5% arginine and 1450 ppm fluoride to a dentifrice containing fluoride alone in the management of primary root caries. J. Dent. 2013, 41, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Xu, P.; Zhou, X.; Deng, M.; Cheng, L.; Li, M.; Li, Y.; Xu, X. Arginine promotes fluoride uptake into artificial carious lesions in vitro. Aust. Dent. J. 2015, 60, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Huang, Z.; Mei, L.; Li, G.; Li, H. Anti-Caries Effect of Arginine-Containing Formulations in vivo: A Systematic Review and Meta-Analysis. Caries Res. 2015, 49, 606–617. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, M.M.; Burne, R.A. Caries prevention by arginine metabolism in oral biofilms: Translating science into clinical success. Curr. Oral Health Rep. 2014, 1, 79–85. [Google Scholar] [CrossRef]
- Nascimento, M.M.; Gordan, V.V.; Garvan, C.W.; Browngardt, C.M.; Burne, R.A. Correlations of oral bacterial arginine and urea catabolism with caries experience. Oral Microbiol. Immunol. 2009, 24, 89–95. [Google Scholar] [CrossRef]
- Nascimento, M.M.; Liu, Y.; Kalra, R.; Perez, S.; Adewumi, A.; Xu, X. Arginine metabolismo may confer caries resistance in children. J. Dent. Res. 2012, 91, 691. [Google Scholar]
- Acevedo, A.M.; Machedo, C.; Rivera, L.E.; Wolff, M.; Kleinberg, I. The inhibitory effect of an arginine bicarbonate/calcium carbonate Cavistat-containing dentifrice on the development of dental caries in Venezuelan school children. J. Clin. Dent. 2005, 16, 63–70. [Google Scholar] [PubMed]
- Yin, W.; Hu, D.Y.; Li, X.; Fan, X.; Zhang, Y.P.; Pretty, I.A.; Mateo, L.R.; Cummins, D.; Elwood, R.P. The anti-caries efficacy of a dentifrice containing 1.5% arginine and 1450 ppm fluoride as sodium monofluorophosphate assessed using quantitative light-induced fluorescence (QLF). J. Dent. 2013, 41, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Srisilapanan, P.; Korwanich, N.; Yin, W.; Cheunsuwonkui, C.; Mateo, L.R.; Zhang, Y.P.; Cummins, D.; Elwood, R.P. Comparison of the efficacy of a dentifrice containing 1.5% arginine and 1450 ppm fluoride to a dentifrice containing 1450 ppm fluoride alone in the management of early coronal caries as assessed using quantitative light-induced fluorescence. J. Dent. 2013, 41, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Vieira, A.E.; Delbem, A.C.; Sassaki, K.T.; Rodrigues, E.; Cury, J.A.; Cunha, R.F. Fluoride dose response in pH-cycling models using bovine enamel. Caries Res. 2005, 39, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Featherstone, J.D.B.; O’Reily, M.M.; Shariati, M.; Brugler, S. Enhancement of Remineralization In Vitro and In Vivo. In Leach AS. Factors Relating to Demineralization and Remineralization of the Teeth; IRL Press: Oxford, UK, 1986; pp. 23–24. [Google Scholar]
- Featherstone, J.D.B.; ten Cate, J.M.; Shariati, M.; Arends, J. Comparison of artificial caries-like lesions by quantitative microradiography and microhardness profiles. Caries Res. 1983, 17, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Marinho, V.C.; Higgins, J.P.; Sheiham, A.; Logan, S. Fluoride toothpastes for preventing dental caries in children and adolescents. Cochrane Database Syst. Rev. 2009, 1, CD002278. [Google Scholar] [CrossRef] [PubMed]
- Metropolitan Waterworks Authority. Fluoride Content of Tap Water in Bangkok; Metropolitan Waterworks Authority: Bangkok, Thailand, 2007. [Google Scholar]
- Walsh, T.; Worthington, H.V.; Glenny, A.M.; Appelbe, P.; Marinho, V.C.; Shi, X. Fluoride toothpastes of different concentrations for preventing dental caries in children and adolescents. Cochrane Database Syst. Rev. 2010, 1, CD007868. [Google Scholar] [CrossRef]
- Hu, D.Y.; Yin, W.; Li, X.; Feng, Y.; Zhang, Y.P.; Cummins, D.; Mateo, L.R.; Elwood, R.P. A clinical investigation of the efficacy of a dentifrice containing 1.5% arginine and 1450 ppm fluoride, as monofluorophosphate, in a calcium base on primary root caries. J. Clin. Dent. 2013, 24, A23–A31. [Google Scholar]
- Ammari, A.; Bloch-Zupan, A.; Ashley, P. Systematic review of studies comparing the anti-caries efficacy of children’s toothpaste containing 600 ppm of fluoride or less with high fluoride toothpastes of 1000 ppm or above. Caries Res. 2003, 37, 85–92. [Google Scholar] [CrossRef]
- Suyama, E.; Tamura, T.; Ozawa, T. Remineralization and acid resistance of enamel lesions after chewing gum containing fluoride extracted from green tea. Aust. Dent. J. 2011, 56, 394–400. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, S.; Pei, D.; Du, X.; Ouyang, X.; Huang, C. Effect of an 8.0% arginine and calcium carbonate in-office desensitizing paste on the microtensile bond strength of self-etching dental adhesives to human dentin. Am. J. Dent. 2012, 25, 281–286. [Google Scholar] [PubMed]
- Yassen, G.H.; Platt, J.A.; Hara, A.T. Bovine teeth as substitute for human teeth in dental research: A review of literature. J. Oral Sci. 2011, 53, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Rüttermann, S.; Braun, A.; Janda, R. Shear bond strength and fracture analysis of human vs. bovine teeth. PLoS ONE 2013, 8, e59181. [Google Scholar] [CrossRef]
- Teruel, J.D.D.; Alcolea, A.; Hernández, A.; Ruiz, A.J. Comparison of chemical composition of enamel and dentine in human, bovine, porcine and ovine teeth. Arch. Oral Biol. 2015, 60, 768–775. [Google Scholar] [CrossRef] [PubMed]
| Study Groups | Type of Enamel | Main Components of the Dentifrice | Commercial Name |
|---|---|---|---|
| Neutra DE | DE | 1.5% arginine, insoluble calcium compound, 1450 ppm fluoride as sodium monofluorophosphate. | Neutraçucar Colgate® |
| Neutra SE | SE | 1.5% arginine, insoluble calcium compound, 1450 ppm fluoride as sodium monofluorophosphate. | Neutraçucar Colgate® |
| Total DE | DE | 1450 ppm fluoride | Colgate Total 12® |
| Total SE | SE | 1450 ppm fluoride | Colgate Total 12® |
| My First DE | DE | No Fluoride | My First Colgate® |
| My First SE | SE | No Fluoride | My First Colgate® |
| Name | Composition | Batch Number |
|---|---|---|
| Demineralizing solution | Acetic acid, Ca(OH)2, sodium acetate, phosphoric acid, KOH or HCl in order to regulate pH, and distilled water up to complete 1 l | NR 3046-1 |
| Remineralizing solution | Hydrochloric acid, Ca(OH)2, calcium chloride, tris buffer, KOH or HCl in order to regulate pH, and distilled water up to complete 1 l | NR 3046-0 |
| Total 12 | H2O, glycerin, sorbitol, hydrated silica, sodium lauryl sulphate, copolymer PVM/MA, cellulose gum, sodium hydroxide, propylene glycol, carrageenan, triclosan, sodium saccharin, sodium fluoride 0.32% (1450 ppm) | 5280BR123B |
| Neutraçucar | Calcium cabonate H2O, glycerin, arginine (1.5%), sodium bicarbonate, sodium lauryl ether sulphate, monofluoride phosphate (1.1%, 450 ppm), aroma, cellulose gum, sodium pyrophosphate, benzylic ethanol, saccharin, sodium hydroxide | L3331001021 |
| My first Colgate | H2O, sodic sarcina, sorbitol, poloxamer 407, cellulose gum, aroma, citric acid | 5026USC11A |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, P.H.C.; Oliveira, M.R.C.; Oliveira, L.H.C.; Sfalcin, R.A.; Pinto, M.M.; Rosa, E.P.; Deana, A.M.; Horliana, A.C.R.T.; César, P.F.; Bussadori, S.K. Evaluation of Different Dentifrice Compositions for Increasing the Hardness of Demineralized Enamel: An in Vitro Study. Dent. J. 2019, 7, 14. https://doi.org/10.3390/dj7010014
Oliveira PHC, Oliveira MRC, Oliveira LHC, Sfalcin RA, Pinto MM, Rosa EP, Deana AM, Horliana ACRT, César PF, Bussadori SK. Evaluation of Different Dentifrice Compositions for Increasing the Hardness of Demineralized Enamel: An in Vitro Study. Dentistry Journal. 2019; 7(1):14. https://doi.org/10.3390/dj7010014
Chicago/Turabian StyleOliveira, Pedro Henrique Cabral, Marcia Regina Cabral Oliveira, Luiz Henrique Cabral Oliveira, Ravana Angelini Sfalcin, Marcelo Mendes Pinto, Ellen Perin Rosa, Alessandro Melo Deana, Anna Carolina Ratto Tempestini Horliana, Paulo Francisco César, and Sandra Kalil Bussadori. 2019. "Evaluation of Different Dentifrice Compositions for Increasing the Hardness of Demineralized Enamel: An in Vitro Study" Dentistry Journal 7, no. 1: 14. https://doi.org/10.3390/dj7010014
APA StyleOliveira, P. H. C., Oliveira, M. R. C., Oliveira, L. H. C., Sfalcin, R. A., Pinto, M. M., Rosa, E. P., Deana, A. M., Horliana, A. C. R. T., César, P. F., & Bussadori, S. K. (2019). Evaluation of Different Dentifrice Compositions for Increasing the Hardness of Demineralized Enamel: An in Vitro Study. Dentistry Journal, 7(1), 14. https://doi.org/10.3390/dj7010014





