Probiotics: A Promising Role in Dental Health
Abstract
:1. Introduction
2. Foreseen and Research Targeted Activities of Probiotics
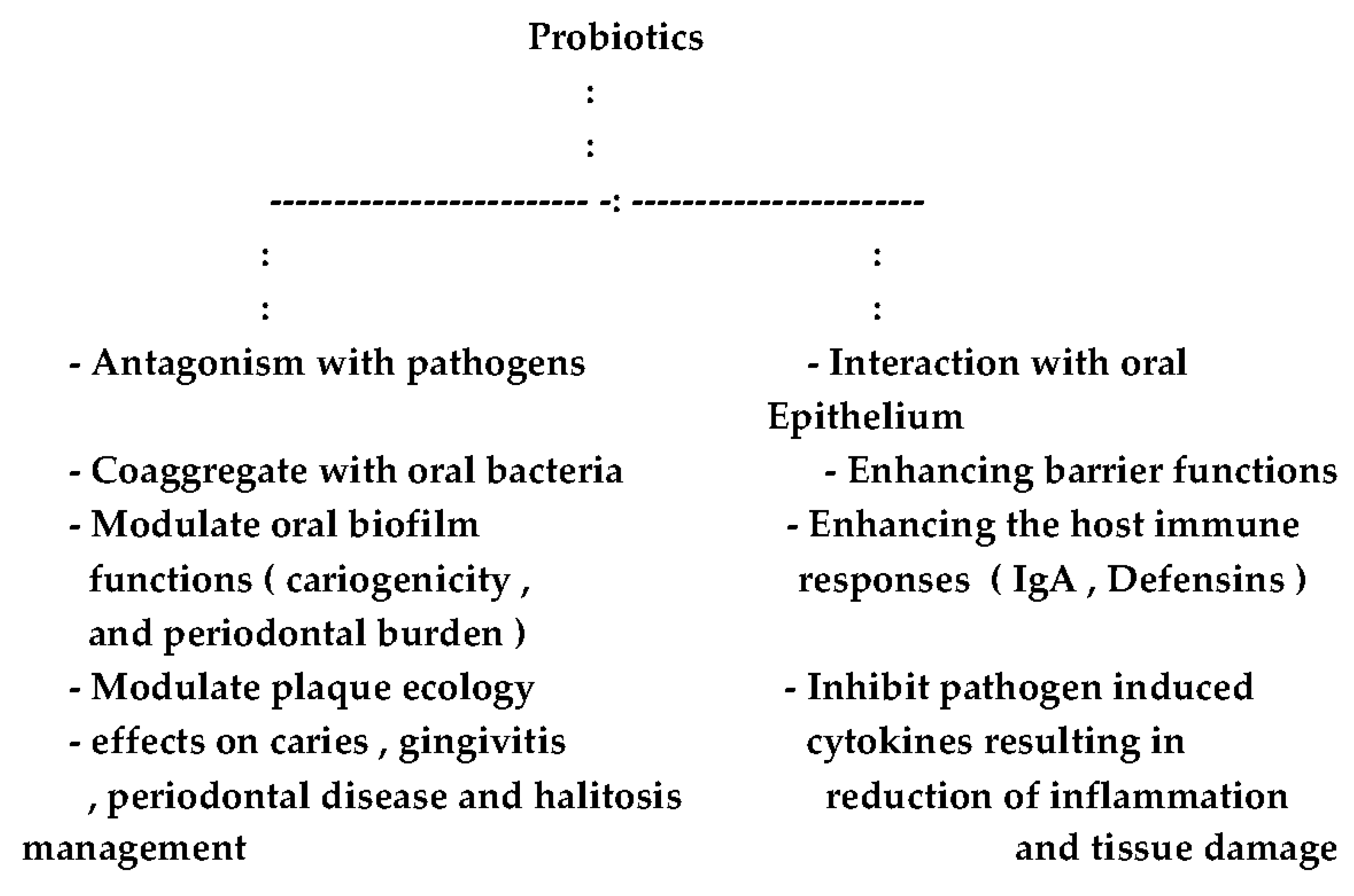
3. Possible Roles of Probiotics in the Oral Cavity
- (1)
- Antagonism with pathogens;
- (2)
- Aggregation with oral bacteria; and
- (3)
- Interaction with oral epithelium.
4. Oral Microbiota Characteristics
5. The Oral Cavity and Indigenous Probiotics
6. Halitosis and Probiotics
7. Oral Fungal Infections
8. Designer Probiotics as a Base for Living Therapeutics
9. Concluding Remarks and Future Directions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wade, W.G. The oral microbiome in health and disease. Pharmacol. Res. 2013, 69, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Shomark, D.K.; Allen, S.J. The microbiota and disease reviewing the links between the oral microbiome, aging and Alzheimer’s disease. J. Alzheimer’s Dis. 2015, 43, 725–738. [Google Scholar]
- Scannapieco, F.A. The oral microbiome: Its role in health and in oral and systemic infections. Clin. Microbiol. News 2013, 35, 163–199. [Google Scholar] [CrossRef]
- Gatej, S.; Gully, N.; Gibson, R.; Bartold, P.M. Probiotics and periodontitis—A literature review. J. Int. Acad. Periodontol. 2012, 19, 42–50. [Google Scholar]
- Stamatova, I.; Meurman, J.H. Probiotics: Health benefits in the mouth. Am. J. Dent. 2009, 22, 329–338. [Google Scholar] [PubMed]
- Kobayashi, R.; Kobayashi, T.; Sakai, F.; Hosoya, T.; Yamamoto, M.; Kurita-Ochiai, T. Oral administration of Lactobacillus gasseri SBT 2055 is effective in preventing Porphyromonas gingivalis—Accelerated periodontal disease. Sci. Rep. 2017, 7, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Vandenplas, Y.; Huys, G.; Daube, G. Probiotics: An update. J. Pediatr. 2015, 91, 6–21. [Google Scholar] [CrossRef] [PubMed]
- Selle, K.; Klaenhammer, T.R. Genomic and phenotypic evidence for probiotic influences of Lactobacillus gasseri on human health. FEMS Micrbiol. Rev. 2013, 37, 915–935. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G. Periodontitis from microbial immune subversion to systemic inflammation. Nat. Rev. Immunol. 2015, 15, 30–44. [Google Scholar] [CrossRef] [PubMed]
- Maseda, L.; Kulik, E.M.; Hauser-Gerpach, I.; Ramseier, A.M.; Filipi, A.; Waltimo, T. Antimicrobial activity of Streptococcus salivarius K12 on bacteria involved in oral malodor. Arch. Oral Biol. 2012, 57, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, M.R.; Kragelund, C.; Jansen, P.; Keller, M.K.; Twetman, S. Probiotic Lactobacillus reuteri has antifungal effects on oral Candida species In Vitro. Arch. Oral Biol. 2017, 9, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Manning, J.; Dunne, E.; Wescombe, P.; Hale, J.; Mullholland, E.; Tagg, J. Investigation of S. salivarius mediated inhibition of pneumococcal adherence to pharyngeal cells. BMC Microbiol. 2016, 16, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Schwendicke, F.; Korte, F.; Drofer, C.; Kneist, S.; El-Sayed, F.; Paris, S. Inhibition of Streptococcus mutans growth and biofilm formation by probiotics In Vitro. Caries Res. 2017, 51, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.C.; Huang, C.S.; Ou-Yang, L.W.; Lin, S.Y. Probiotic Lactobacillus paracaesi effect on cariogenic flora. Clin. Oral Investig. 2017, 15, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Schwendicke, F.; Dorfer, C.; Kneist, F.; Meyer-Lueckel, H.; Paris, S. Cariogenic effect of Lactobacillus rhamnosus GG in dental biofilm model. Caries Res. 2014, 48, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Gruner, D.; Paris, S.; Schwendicke, F. Probiotics for managing caries and periodontitis: Systematic review and meta-analysis. J. Dent. 2016, 48, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Fong, F.L.; Shah, N.P.; Kirjavainen, P.; El-Nezami, H. Mechanisms of action of probiotic bacteria on intestinal and systemic immunities and antigen-presenting cells. Int. Rev. Immunol. 2016, 35, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Oliveria, L.F.F.; Salvador, S.L.; Silva, P.H.F.; Furlaneto, F.A.; Figuiredo, L.; Casarin, R.; Ervolino, E.; Palioto, D.; Souza, S.; Taba, M.; et al. Benefits of Bifidobacterium animalis subsp. lactis Probiotic in Experimental Periodontitis. J. Periodontl. 2017, 88, 197–208. [Google Scholar] [CrossRef] [PubMed]
- Para, D.; Martinez, J.A. Amino acid uptake from probiotic milk in lactose intolerance subjects. Br. J. Nutr. 2007, 98, 5101–5104. [Google Scholar]
- Burton, J.P.; Chilcott, C.N.; Moore, C.J.; Speiser, G.; Tagg, J.R. A preliminary study of the effect of probiotic Streptococcus salivarius K12 on oral malodour parameters. J. Appl. Microbiol. 2006, 100, 754–764. [Google Scholar] [CrossRef] [PubMed]
- Alok, A.; Singh, I.D.; Singh, S.; Kishore, M.; Jha, P.C.; Iqbal, M.A. A new era of biotherapy. Adv. Biomed. Res. 2017, 6, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Toshimitsu, T.; Ozaki, S.; Mochizuki, J.; Furuichi, K.; Asami, Y. Effects of Lactobacillus plantarum strain OLL 2712 culture conditions on the anti-inflammatory activities for murine immune cells and obese type 2 diabetic mice. Appl. Environ. Microbiol. 2017, 83, e03001-16. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, D.S.; Rault, L.; Berkova, N.; Le Loir, Y.; Evens, S. Inhibition of Staphylococcus aureus invasion into bovine mammary epithelial cells by contact with live Lactobacillus casei. Appl. Environ. Microbiol. 2013, 79, 877–885. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, S.; Lau, C.S.; Chamberlain, R.S. Probiotics and symbiotics decrease post-operation sepsis in elective gastrointestinal surgical patients: A meta-analysis. J. Gastrointest. Surg. 2016, 20, 1123–1131. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Dhanda, S. Mechnistic insight of probiotic derived anticancer pharmaceuticals: A road forward for cancer therapeutics. Nutr. Cancer 2017, 69, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Laleman, I.; Teughels, W. Probiotics in dental practice: A review. Quintessence Int. 2015, 46, 255–264. [Google Scholar] [PubMed]
- Collado, M.C.; Meriluoto, J.; Salminen, S. Adhesion and aggregation properties of probiotic and pathogen strains. Eur. Food Restechnol. 2008, 226, 1065–1075. [Google Scholar] [CrossRef]
- Mahasneh, A.M.; Hamdan, S.; Mahasneh, S.A. Probiotic properties of Lactobacillus species isolated from local traditionally fermented products. Jordan J. Biol. Sci. 2015, 8, 81–87. [Google Scholar] [CrossRef]
- Mahasneh, A.M.; Mahasneh, S.A. Probiotic characterization of lactic acid bacteria isolated from local fermented vegetables (Makdoos). Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 1673–1686. [Google Scholar] [CrossRef]
- Bonifait, I.; Chandad, F.; Grenier, D. Probiotics for oral health: Myth or reality? J. Can. Dent. Assoc. 2009, 75, 585–590. [Google Scholar] [PubMed]
- Nudelman, P.; Frazao, J.V.; Vieira, T.I.; Balth, C.F.; Andrade, M.M.; Alexandria, A.K.; Cruz, A.G.; Fonseca, A.; Maia, L.C. The performance of fermented sheep milk and ice cream sheep milk in inhibiting enamel loss. Food Res. Int. 2017, 97, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Thomas, I.V. Probiotics—The journey continues. Int. J. Dairy Technol. 2016, 69, 469–480. [Google Scholar] [CrossRef]
- Ben Taheur, F.; Kouidhi, B.; Fdhila, K.; Elabed, H.; Ben Salama, R.; Mahduan, K.; Bakharouf, A.; Chaieb, K. Antibacterial and antibiofilm activity of probiotic bacteria against oral pathogens. Microb. Pathog. 2016, 97, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Samot, J.; Badet, C. Antibacterial activity of probiotic candidate for oral health. Anaerobe 2013, 19, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Kim, Y.-J. A copmparative study on cariogenic biofilm model for preventing dental caries. Arch. Microbiol. 2014, 196, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Schwendicke, F.; Horb, K.; Kneist, S.; Dorfer, C.; Paris, S. Effects of heat-inactivated bifidobacterium BB12 on cariogenicity of Streptococcus mutans In Vitro. Arch. Oral Biol. 2014, 59, 1384–1390. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Uneda, M.; Hatano, Y.; Iwamoto, T.; Masuo, Y.; Hirofugi, T. Enterococcus faecium WB 2000 inhibits biofilm formation by oral cariogenic streptococci. Int. J. Dent. 2011, 2011, 834151. [Google Scholar] [CrossRef] [PubMed]
- Bensalama, R.; Kouidhi, B.; Zmantar, T.; Chaieb, K.; Bakharouf, A. Antilisterial and biofilm activities of potential probiotic Lactobacillus strains isolated from Tunisian traditional fermented foods. J. Food Saf. 2013, 33, 8–16. [Google Scholar] [CrossRef]
- Singh, V.P.; Malhotra, N.; Apratim, A.; Verma, M. Assessment and management of halitosis. Dent. Update 2015, 42, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Castalonga, M.; Herzberg, M. The oral microbiome and the immunobiology of periodontal disease and caries. Immunol. Lett. 2014, 162, 22–38. [Google Scholar] [CrossRef] [PubMed]
- Anusha, R.L.; Umar, D.; Basheer, D.; Baroudi, K. The magic of magic bugs in oral cavity: Probiotics. J. Adv. Pharm. Technol. Res. 2015, 6, 43–47. [Google Scholar] [PubMed]
- Russel, D.A.; Ross, R.P.; Fitzgerald, G.F.; Santon, C. Metabolic activities and probiotic potential of bifidobacteria. Int. J. Food Microbiol 2011, 149, 88–105. [Google Scholar] [CrossRef] [PubMed]
- Marsh, P.D.; Zura, E. Dental biofilms: Ecological interactions inhealth disease. J. Clin. Periodontol. 2017, 44, S12–S22. [Google Scholar] [CrossRef] [PubMed]
- Ereshefsky, M.; Pedroso, M. Rethinking evolutionary individuality. Proc. Natl. Acad. Sci. USA 2015, 112, 10126–10132. [Google Scholar] [CrossRef] [PubMed]
- Burczynska, A.; Dziewit, L.; Decewit, L.; Decewicz, P.; Struzycka, I.; Wroblewska, M. Application of metagenomic analyses in dentistry and novel strategy enabling complex insight into microbial diversity of the oral cavity. Pol. J. Microbiol. 2017, 66, 9–15. [Google Scholar] [CrossRef]
- Perez-Chaparro, P.J.; Concalves, C.; Figueirado, P.; Faveri, M.; Louo, E.; Tamashiro, N.; Durate, P.; Feres, M. Newly identified pathogens associated with periodontitis: A systematic review. J. Dent. Res. 2014, 93, 846–858. [Google Scholar] [CrossRef] [PubMed]
- Roberts, F.A.; Darveau, R.P. Microbial protection and virulence in periodontal tissue as a function of polymicrobial communities: Symbiosis and dysbiosis. Periodontology 2015, 69, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Ng, H.M.; Kin, L.X.; Dashper, S.G.; Slakeski, N.; Butler, C.A.; Reynolds, E.C. Bacterial interactions in pathogenic subgingival plaque. Microb. Pathog. 2016, 94, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Mark-Welsh, J.L.; Rosseti, B.J.; Rieken, C.W.; Dewhirst, F.W.; Boristy, G.G. Biogeography of a human oral microbiome at the micro scale. Proc. Natl. Acad. Sci. USA 2016, 113, E791–E800. [Google Scholar] [CrossRef] [PubMed]
- Hagishengallis, G. Immunomicrobial pathogenesis of periodontitis: Keystones, pathobionnts and host response. Trends Immunol. 2014, 35, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Schincaglia, G.P.; Hong, B.Y.; Rosania, A.; Barasz, J.; Thompson, A.; Soube, T.; Panagakos, F.; Burleson, J.A.; Dongari-Bagtzoglou, A.; Diaz, P. Clinical, immune, and microbiome traits of gingivitis and peri-implant mucositis. J. Dent. Res. 2017, 96, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Diaz, P.I.; Xie, Z.; Sobae, T.; Thompson, A.; Biyikoglu, B.; Rikers, A.; Ikonomou, I.; Dongari-Bagtazoslou, A. Synergistic interaction between Candida albicans and commensal oral streptococci in a novel In Vitro mucosal model. Infect. Immun. 2012, 80, 620–632. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.H.; Seers, C.A.; Dashper, S.G.; Mitchell, H.L.; Pyke, J.S.; Meuric, V.; Slakeski, N.; Cleal, S.M.; Chambers, J.L.; McConville, M.; et al. Porphyromonas gingivalis and Treponema denticola exhibits metabolic symbiosis. PLoS Pathog. 2014, 10, e1003955. [Google Scholar] [CrossRef] [PubMed]
- Oukoda, T.; Kokubu, E.; Kawana, T.; Saito, A.; Okuda, K.; Ishihara, K. Synergy in biofilm formation between Fusobacterium nucleatum and Prevotella species. Anaerobe 2012, 18, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Keijser, B.J.; Zaura, E.; Huse, S.M.; Van der Vossen, J.M.; Schuren, F.H.; Montigen, R.C.; Ten Cate, J.M.; Crielaad, V.V. Pyrosequencing analysis of the oral microflora of healthy adults. J. Dent. Res. 2008, 87, 1016–1020. [Google Scholar] [CrossRef] [PubMed]
- Rudney, J.D. Saliva and dental plaque. Adv. Dent. Res. 2000, 14, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Burne, R.A.; Quivey, R.G.; Marquis, R.E. Physiologic homeostasis and stress responses in oral biofilms. Meth. Enzymol. 1999, 310, 441–460. [Google Scholar] [PubMed]
- Bernaedeau, M.; Venoux, J.P.; Henri-Dubernet, S.; Gueguen, M. Safety assessment of dairy microorganisms: The Lactobacillus genus. Int. J. Food Microbiol. 2008, 85, 88–94. [Google Scholar]
- Yli-Knuuttila, H.; Snall, J.; Kari, K.; Meurman, J.H. Colonization of Lactobacillus rhamnosus GC in the oral cavity. Oral Microbiol. Immunol. 2006, 21, 129–131. [Google Scholar] [CrossRef] [PubMed]
- Koll-Klais, P.; Mandar, R.; Leibur, E.; Marcotte, H.; Hammarstrom, L.; Mikelsaar, M. Oral lactobacilli in chronic periodontitis and periodontal health: Species composition and antimicrobial activity. Oral Microbiol. Immunol. 2005, 20, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Bizzini, B.; Pizzo, G.; Scapagnini, G. Probiotics and oral health. Curr. Pharm. Des. 2012, 18, 5522–5531. [Google Scholar] [CrossRef] [PubMed]
- Scully, C.; Greenman, J. Halitosis (breath odour). Periodontology 2012, 48, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Islam, B.; Khan, S.; Khan, A. Dental caries: From infection to prevention. Med. Sci. Monit. 2007, 13, 196–205. [Google Scholar]
- Ruiz-Martinez, R.C.; Bedani, R.; Saad, S.M. Scientific evidence for probiotics and prebiotics: An update for current prospectives and future challenges. Br. J. Nutr. 2015, 114, 1993–2015. [Google Scholar] [CrossRef] [PubMed]
- Sookkhee, S.; Chulasiri, M.; Pradyabreud, W. Lactic acid bacteria from healthy oral cavity of Thai volunteers, inhibition of oral pathogens. J. Appl. Microbiol. 2001, 90, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G. The inflammophilic character of the periodontitis-associated microbiota. Mol. Oral Microbiol. 2014, 29, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Bartold, P.M.; Van Dyke, T.E. Periodontitis: A host mediated disruption of microbial homeostasis. Unlearning learned concept. Periodontology 2013, 62, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Wade, W. Has the use of molecular methods for the characterization of the human oral microbiome changed our understanding of the role of bacteria in the pathogenesis of periodontal disease? J. Clin. Periodontol. 2011, 38, 7–16. [Google Scholar] [CrossRef] [PubMed]
- De Geest, S.; Laleman, I.; Teughels, W.; Dekeyser, C.; Quirynen, M. Periodental diseases as a source of halitosis: A review of the evidence and treatment approaches for dentists and dental hygienists. Periodontology 2016, 71, 213–227. [Google Scholar] [CrossRef] [PubMed]
- De Boever, E.H.; Loesche, W.J. Assessing the contribution of anaerobic microflora of the tongue to oral malodor. Am. Dent. Assoc. 1995, 126, 1384–1393. [Google Scholar] [CrossRef]
- Aung, E.; Ueno, M.; Zaitsu, T.; Furukawa, S.; Kawaguchi, Y. Effectiveness of three oral hygiene regimens on oral malodor reduction: A randomized clinical trial. Trials 2015, 16, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N. Oral microbiome metabolism: From who are they? To what are they doing? J. Dent. Res. 2015, 94, 1628–1637. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.S.; Kim, B.J.; Yang, K.H.; Oh, J.H. Effect of Weissella cibaria isolates on the formation of S. mutans biofilms. Cries Res. 2006, 40, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.S.; Kim, B.J.; Chung, J.; Lee, H.C.; Oh, J.H. Inhibitory effect of Weissella cibaria isolates on the production of volatile sulfur compounds. J. Clin. Periodontol. 2006, 33, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Stingu, C.S.; Escherich, K.; Rodloff, A.C.; Jentsch, H. Periodontitis is associated with loss of colonization by Streptococcus sanguinis. J. Med. Microbiol. 2008, 57, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Vesty, A.; Biswas, K.; Taylor, M.W.; Gear, K.; Douglas, R.G. Evaluating the impact of DNA extraction-method on the representation of human oral bacterial and fungal communities. PLoS ONE 2007, 12, e0169877. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Dongari-Bagtzoglou, A. Shaping the oral microbiota: Interaction of opportunistic fungi with oral bacteria and the host. Curr. Opin. Microbiol. 2015, 26, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Krom, B.P.; Kidwai, S.; Ten Cate, J.M. Candida and other fungal species: Forgotten players of healthy oral microbiota. J. Dent. Res. 2017, 93, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Ghannoum, M.A.; Jurevic, R.J.; Mukherjee, P.K.; Cui, F.; Sikaroodi, M.; Naqavi, M. Characterization of the oral fungal microbiome (mycobiome) in healthy individuals. PLoS Pathog. 2010, 6, e1000713. [Google Scholar] [CrossRef] [PubMed]
- Barros, P.P.; Ribeiro, F.C.; Rossoni, R.D. Influence of Candida krusei and Candida glabrata on Candida albicans gene expression In Vitro biofilms. Arch. Oral Biol. 2016, 64, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Sardi, J.S.; Scorzoni, I.; Bernardi, T.; Fusco-Almeida, A.M.; Mandis Giannini, M.J. Candida species: Current epidemiology, pathogenicity, biofilm formation natural antifungal products and new therapeutic options. J. Med. Microbiol. 2013, 62, 10–24. [Google Scholar] [CrossRef] [PubMed]
- Wade, W.G. Characterization of the human oral microbiome. J. Oral Biosci. 2013, 55, 143–148. [Google Scholar] [CrossRef]
- Darveau, R.P. Periodontitis: A poly microbial disruption of host homeostasis. Nat. Rev. Microbiol. 2010, 8, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Samaranayake, L.P. Superficial oral fungal infections. Curr. Opin. Dent. 1991, 1, 415–422. [Google Scholar] [PubMed]
- Matsubara, V.H.; Bandara, H.M.; Marcia, P.; Mayer, A.; Samaranayake, P. Probiotics as antifungals in mucosal candidiasis. Clin. Infect. Dis. 2016, 62, 1143–1153. [Google Scholar] [PubMed]
- Miceli, M.H.; Diaz, J.A.; Lee, S.A. Emerging opportunistic yeast infections. Lancet Inf. Dis. 2011, 11, 42–51. [Google Scholar] [CrossRef]
- Oever, J.C.; Netea, M.G. The bacteriome-mycobiome interactions and antifungal host defense. Eur. J. Immunol. 2014, 44, 3182–3191. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, V.H.; Wang, Y.; Bandara, H.M.; Samaranayake, L.P. Probiotic lactobacilli inhibit early stages of Candida albicans biofilm development by reducing their growth, cell adhesion and filamentation. Appl. Microbiol. Biotechnol. 2016, 100, 6415–6426. [Google Scholar] [CrossRef] [PubMed]
- Gungor, C.E.; Kirizioglu, Z.; Kivanc, M. Probiotics can they be used to improve oral health? Benef. Microbes 2015, 6, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Prodeep, K.; Kuttapa, M.A.; Prasna, K.P. Probiotics and oral health: An update. SADJ 2014, 69, 20–24. [Google Scholar]
- Haukioja, A. Probiotics and oral health. Eur. J. Dent. 2010, 4, 348–355. [Google Scholar] [PubMed]
- Wescombe, P.A.; Hale, J.D.; Heng, N.C. Developing oral probiotics from Streptococcus salivarius. Future Microbiol. 2012, 7, 1355–1371. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Tomaro-Duchesneau, C.; Tabrizian, M.; Prakash, S. Probiotics as oral health biotherapeutics. Expert Opin. Biol. Ther. 2013, 12, 1207–1220. [Google Scholar] [CrossRef] [PubMed]
- Paton, A.W. Bioengineered microbes in disease therapy. Trends Mol. Med. 2012, 18, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M. Bioengineered probiotics as a new hope for health and disease: Potential and prospects. Future Microbiol. 2015, 11, 585–600. [Google Scholar] [CrossRef] [PubMed]
- Maxmen, A. Living therapeutics: Scientists genetically modify bacteria to deliver drugs. Nat. Med. 2017, 25, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Mansour, N.M.; Abdelaziz, S.A. Oral immunization of mice with engineered Lactobacillus gasseri NM713 strain expressing Streptococcus pyogenes M6 antigen. Microbiol. Immunol. 2016, 60, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B. Recombinant Lactococcus lactis NZ 9000 secretes a bioactive kisspeptin that inhibits proliferation and migration of human colon carcinoma HT-29 cells. Microb. Cell Fact. 2016, 15, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Mahasneh, A.M.; Abbas, M.M. Probiotics: The possible alternative to disease therapy. In Microbial Biotechnolgy: Progress and Trends; CRC Press: Boca Raton, FL, USA, 2015; pp. 213–238. [Google Scholar]
| Probiotic | Activity | References |
|---|---|---|
| S. salivarius K12 | Antagonism | [10] |
| L. reuteri | Coaggregation | [11] |
| S. salivarius K12, M18 | Interaction withepithelium | [12] |
| L. acidophilus LA-5 | Modulation of biofilm | [13] |
| L. casei LC-11 | Reduction of cariogenic biofilm potential | [13] |
| L. paracasei | Caries management | [14,15] |
| Lactobacilli sp | Periodontal control | [16] |
| Bifidobacterium sp | Gingivitis management | [16] |
| L. rhamnosus GG | Modulation of immune response | [17] |
| Bifidobacterium | Improved resistance | [16,18] |
| Animalis subsp. lacis | to oral infections |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahasneh, S.A.; Mahasneh, A.M. Probiotics: A Promising Role in Dental Health. Dent. J. 2017, 5, 26. https://doi.org/10.3390/dj5040026
Mahasneh SA, Mahasneh AM. Probiotics: A Promising Role in Dental Health. Dentistry Journal. 2017; 5(4):26. https://doi.org/10.3390/dj5040026
Chicago/Turabian StyleMahasneh, Sari A., and Adel M. Mahasneh. 2017. "Probiotics: A Promising Role in Dental Health" Dentistry Journal 5, no. 4: 26. https://doi.org/10.3390/dj5040026
APA StyleMahasneh, S. A., & Mahasneh, A. M. (2017). Probiotics: A Promising Role in Dental Health. Dentistry Journal, 5(4), 26. https://doi.org/10.3390/dj5040026




