A Heteroaromatically Functionalized Hexamolybdate
Abstract
:1. Introduction
2. Results and Discussion
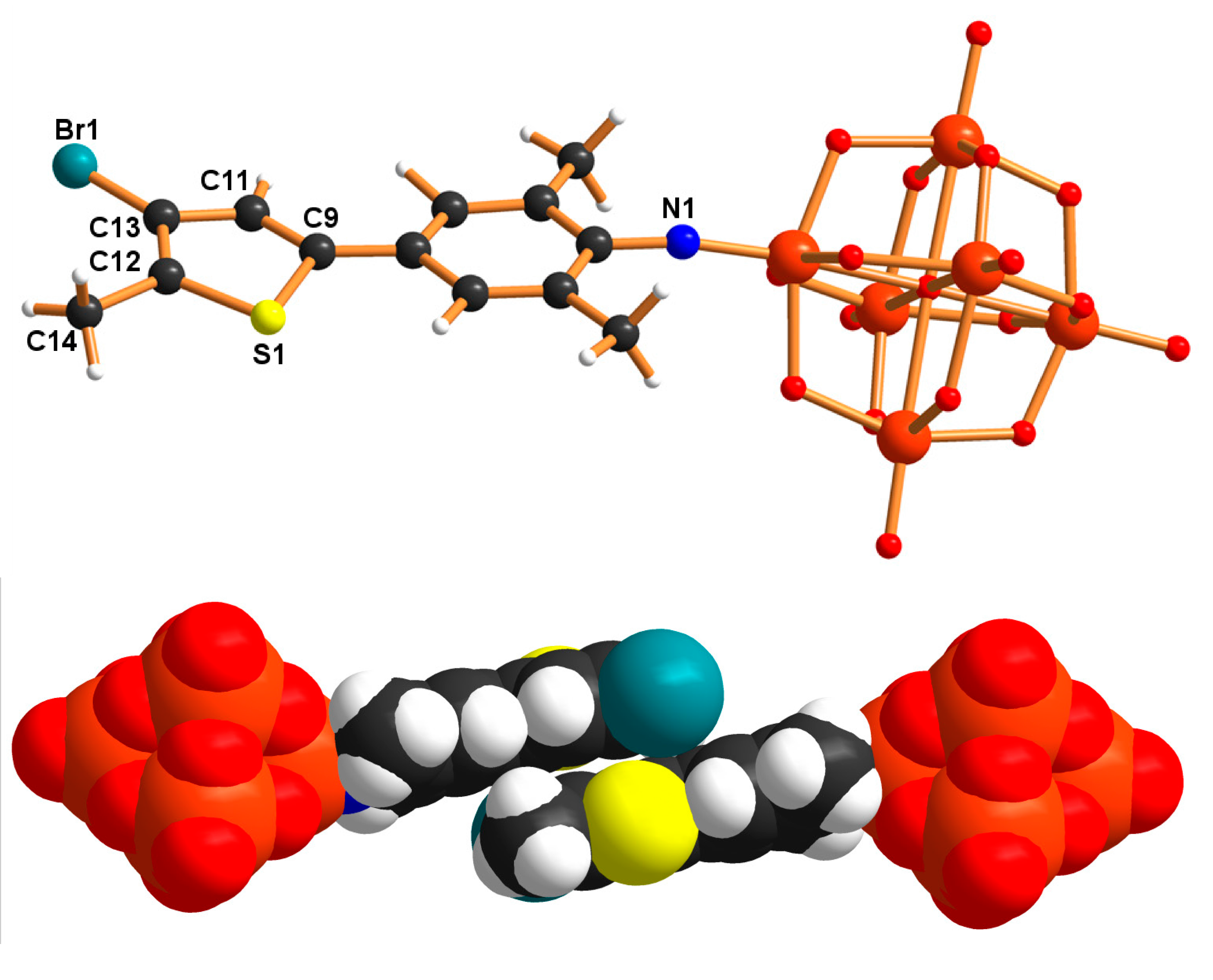
3. Conclusions
Acknowledgments
Author Contributions
Appendix
A1. Instrumentation
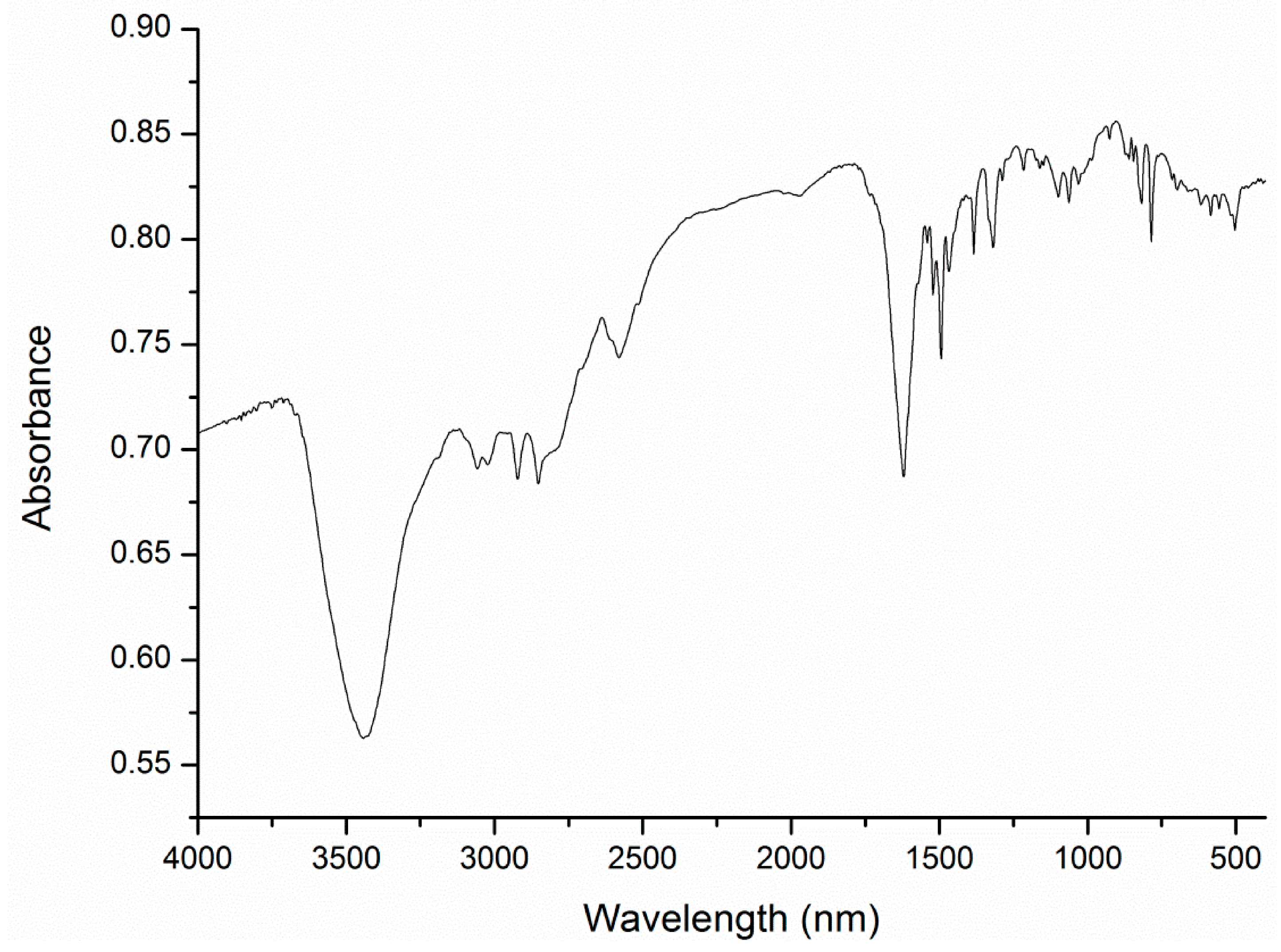
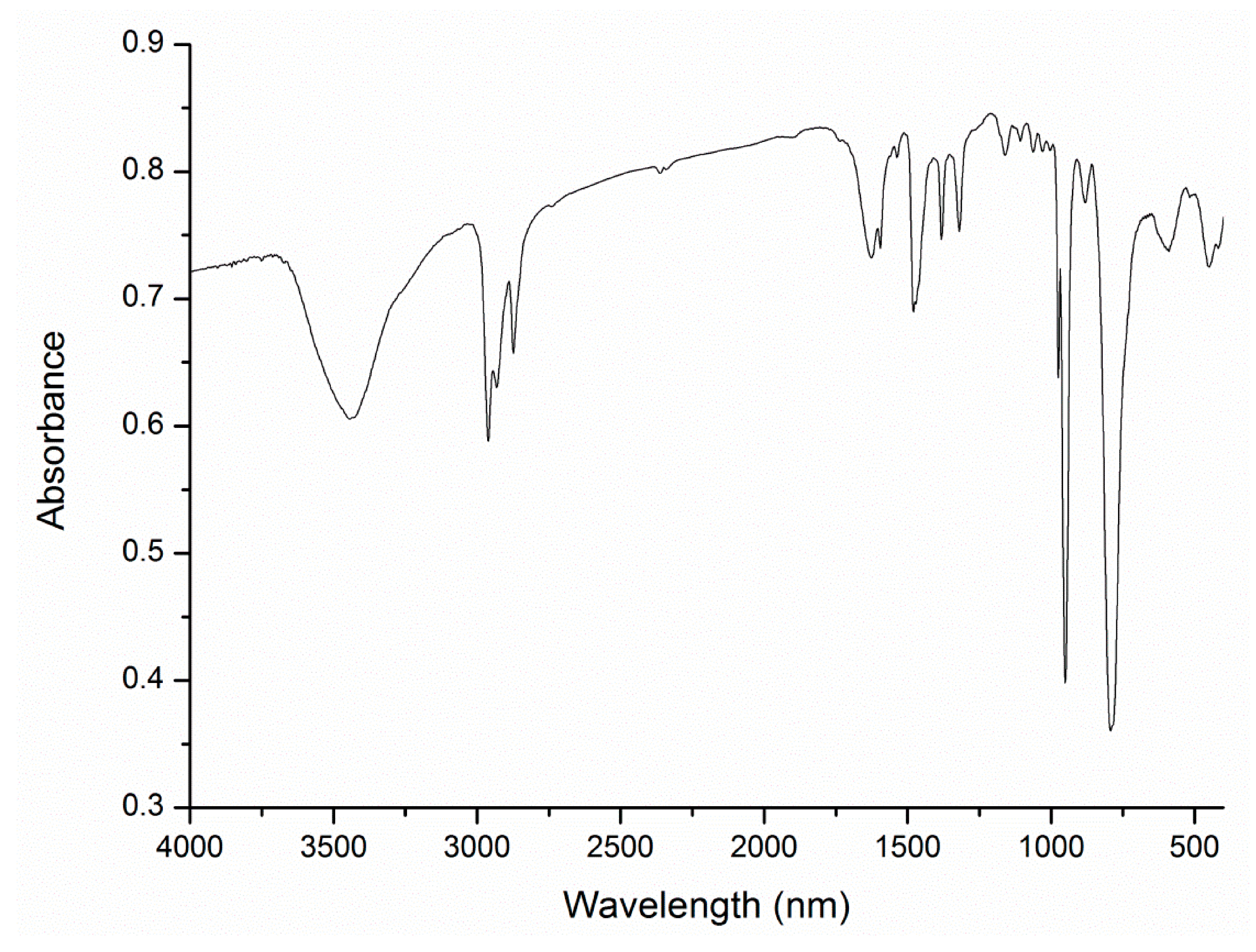
A1.1. FT-IR Spectroscopy (KBr Disc)
A1.2. Elemental Analysis
A1.3. Microwave Reactor
A1.4. UV-Vis Spectrometer
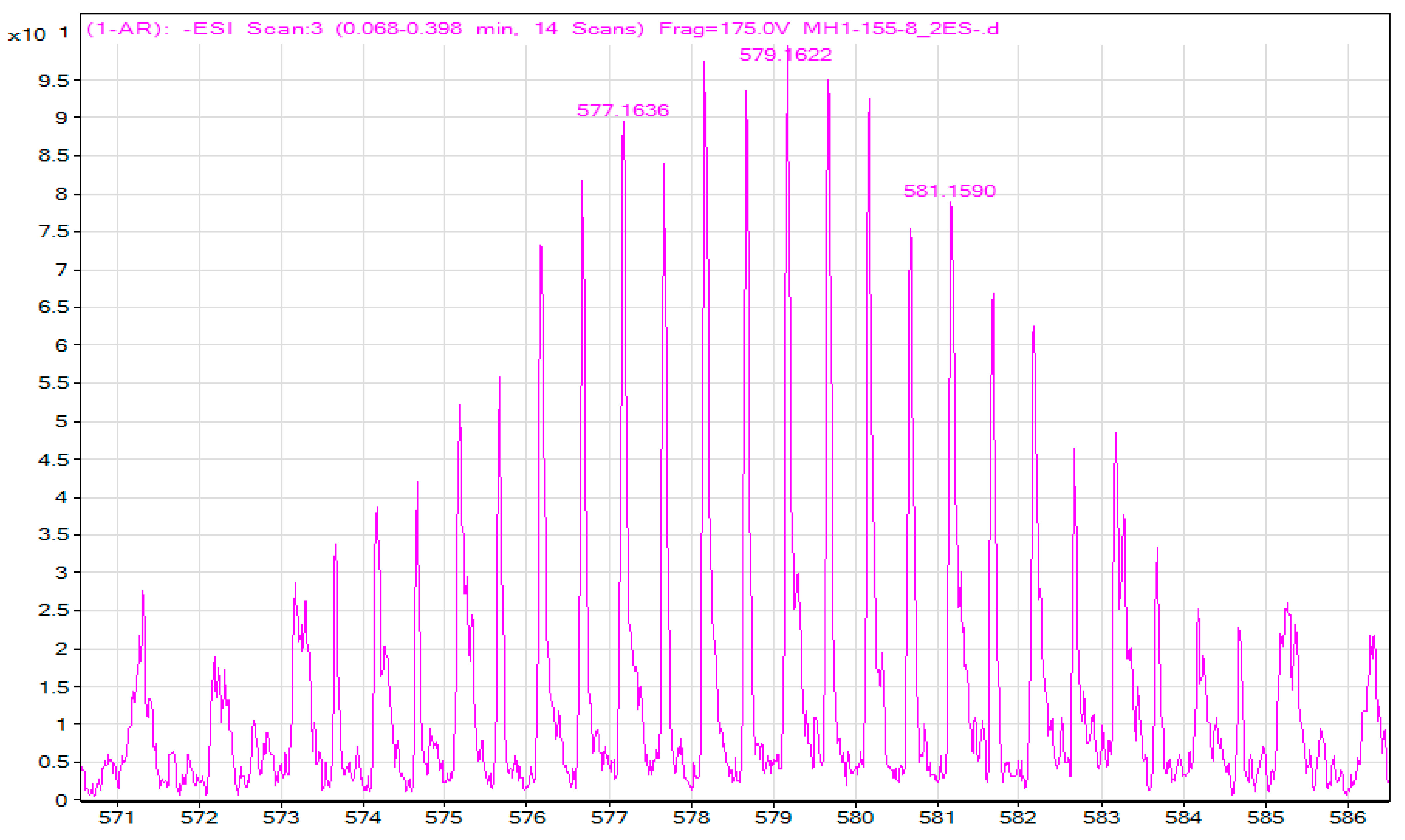
A1.5. Mass Spectroscopy (ESI)
A1.6. Thermogravimetric Analysis (TGA)
A1.7. NMR Spectroscopy
A1.8. Column Chromatography
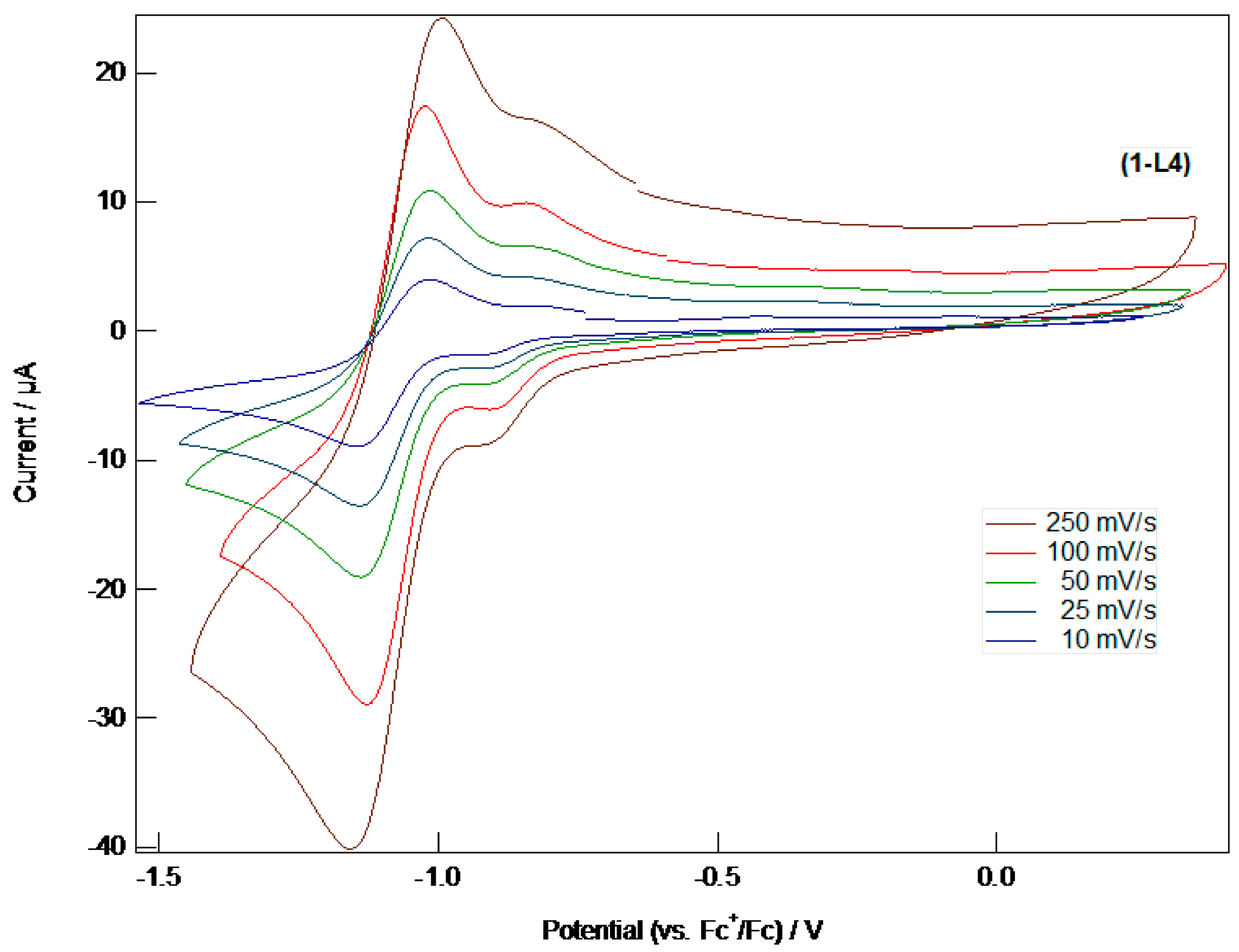
A1.9. Electrochemistry
A2. Experimental Section
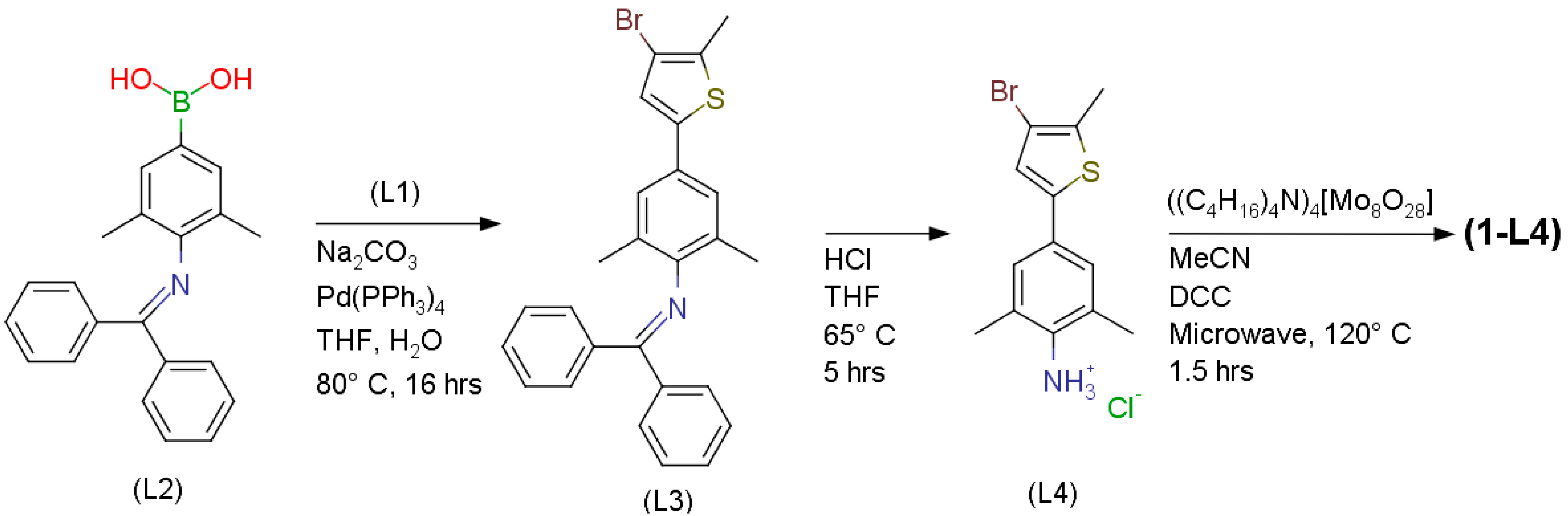
Synthesis of (L3): N-[4-(4-bromo-5-methylthiophen-2-yl)-2,6-dimethylphenyl]-1,1-diphenylmethanimine
Synthesis of (L4): (4-bromo-5-methylthiophen-2-yl)-2,6-dimethylaniline·HCl
Synthesis of (1-L4): Mo6O18(4-(4-bromo-5-methylthiophen-2-yl)-2,6-dimethylaniline)
A3. Characterization Section
A3.1. Mass Spectroscopy
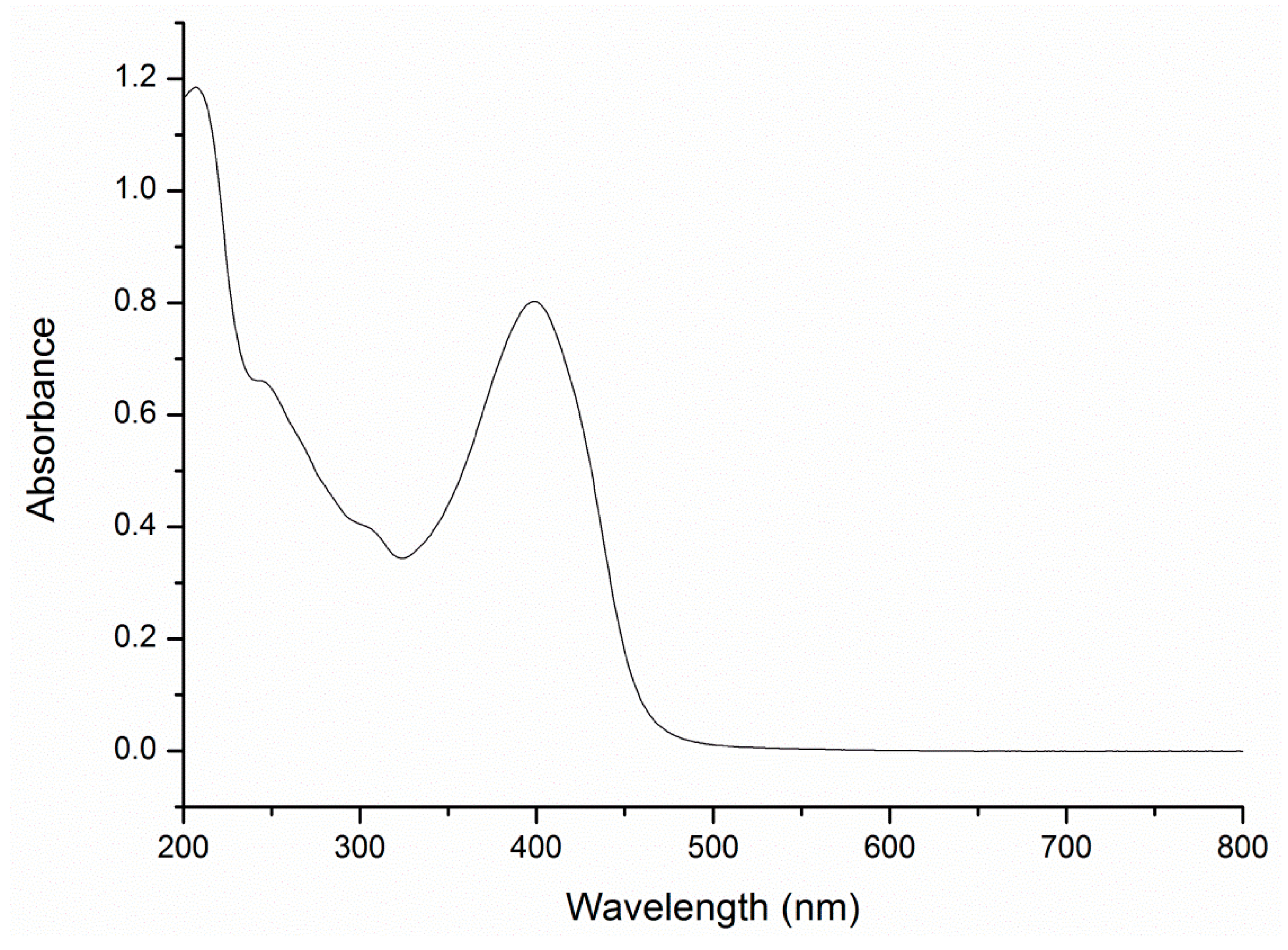
A3.2. Electronic Spectroscopy
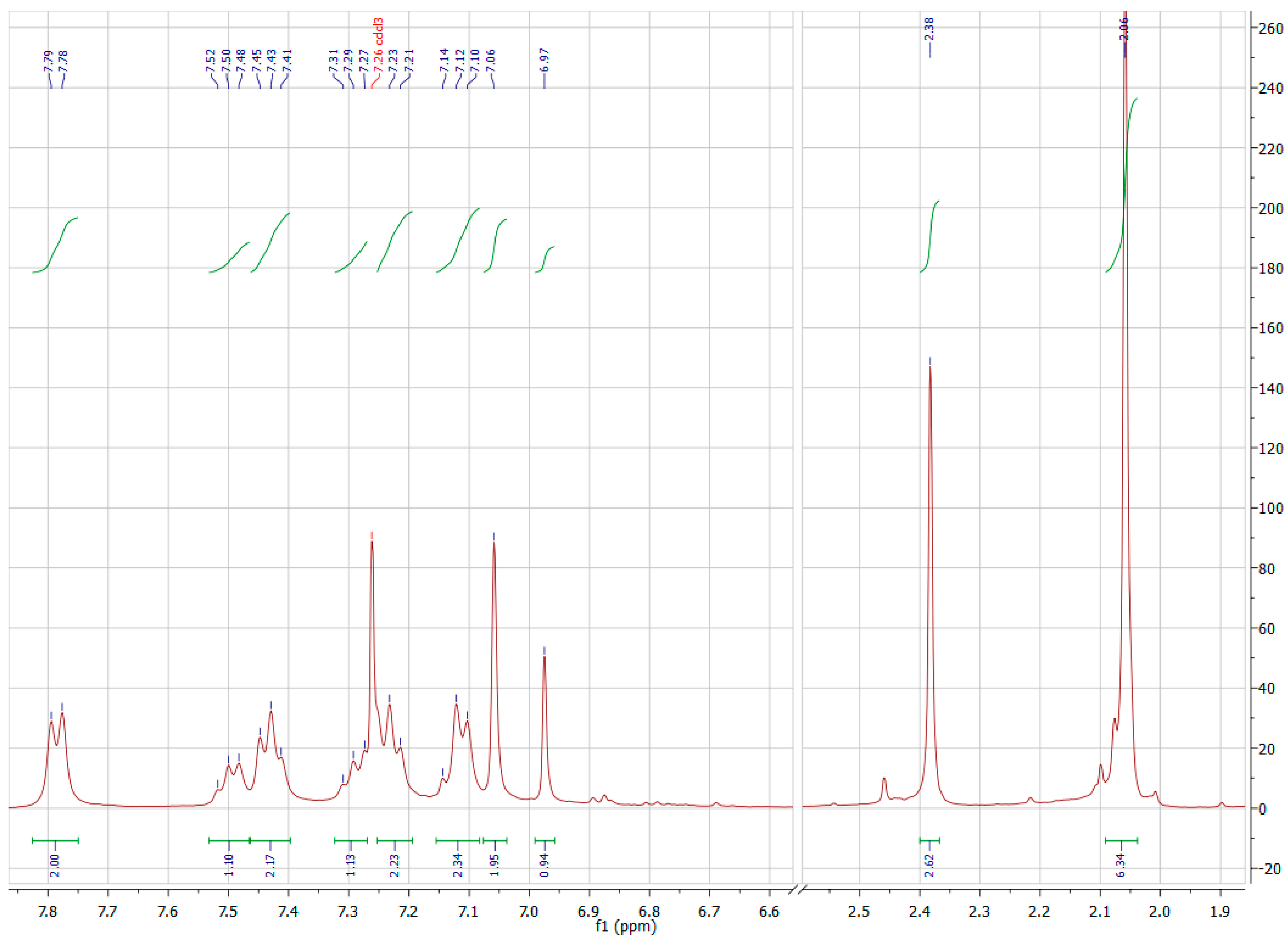
A3.3. Nuclear Magnetic Resonance Spectroscopy
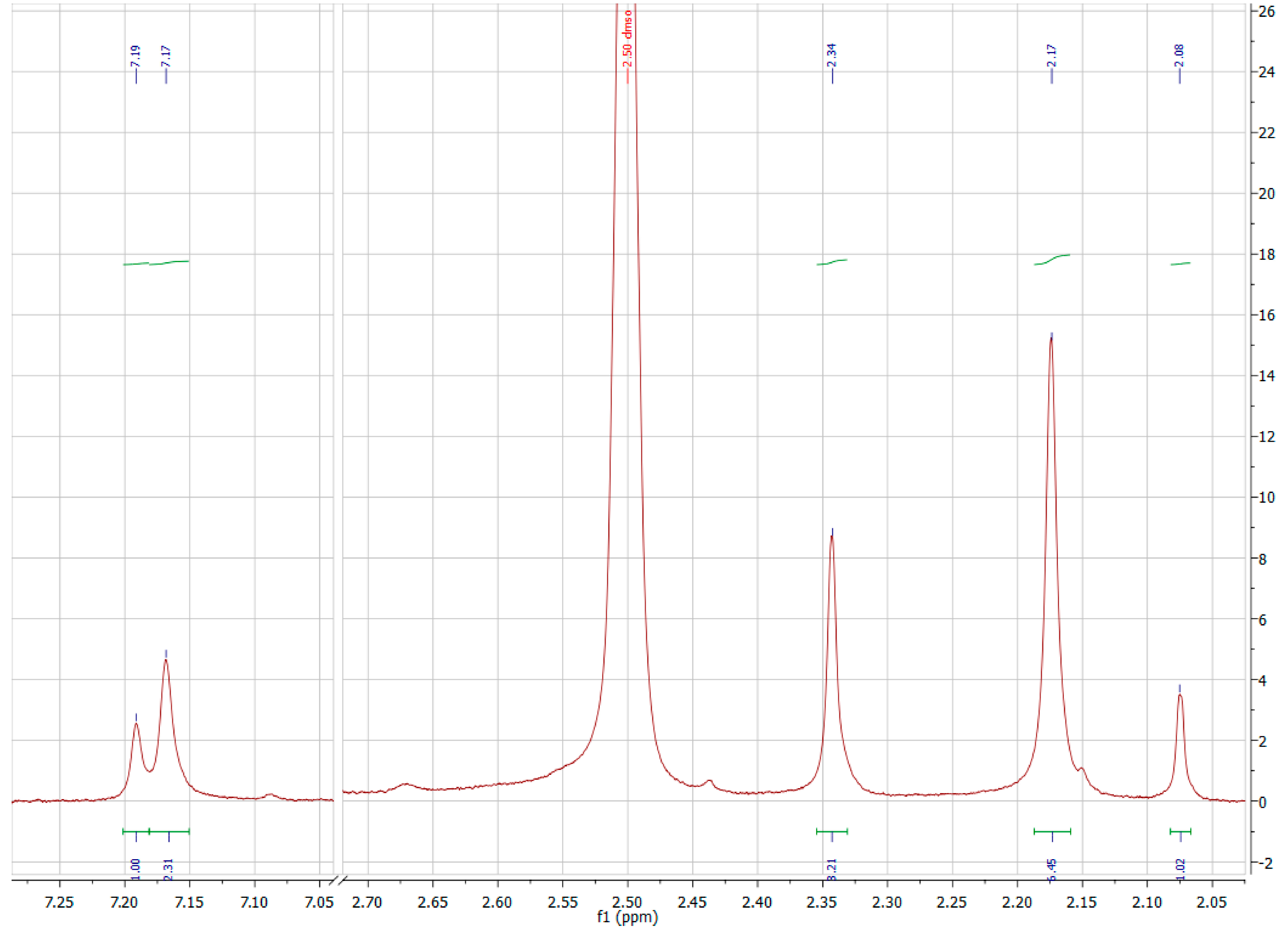
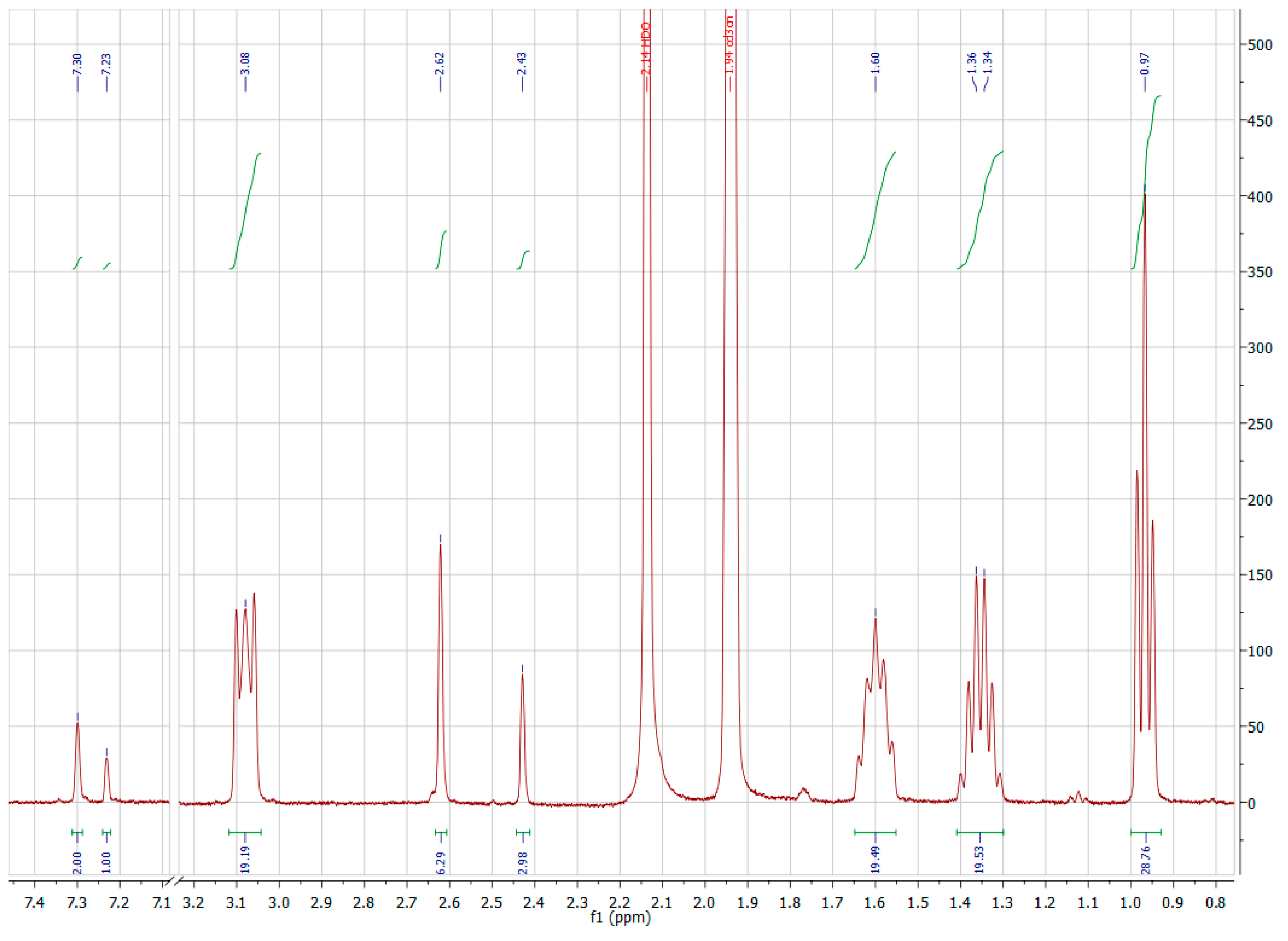
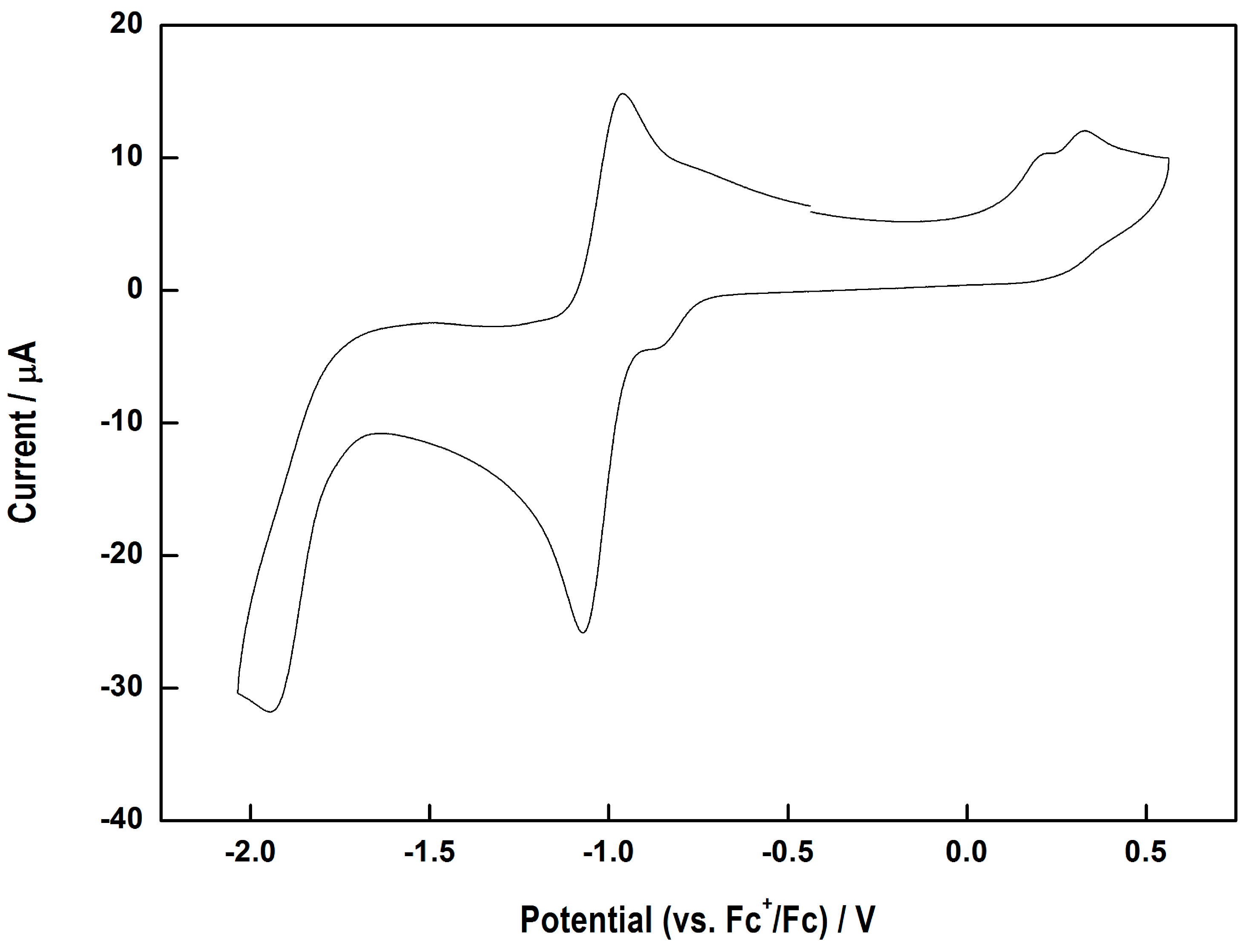
A3.4. Electrochemistry
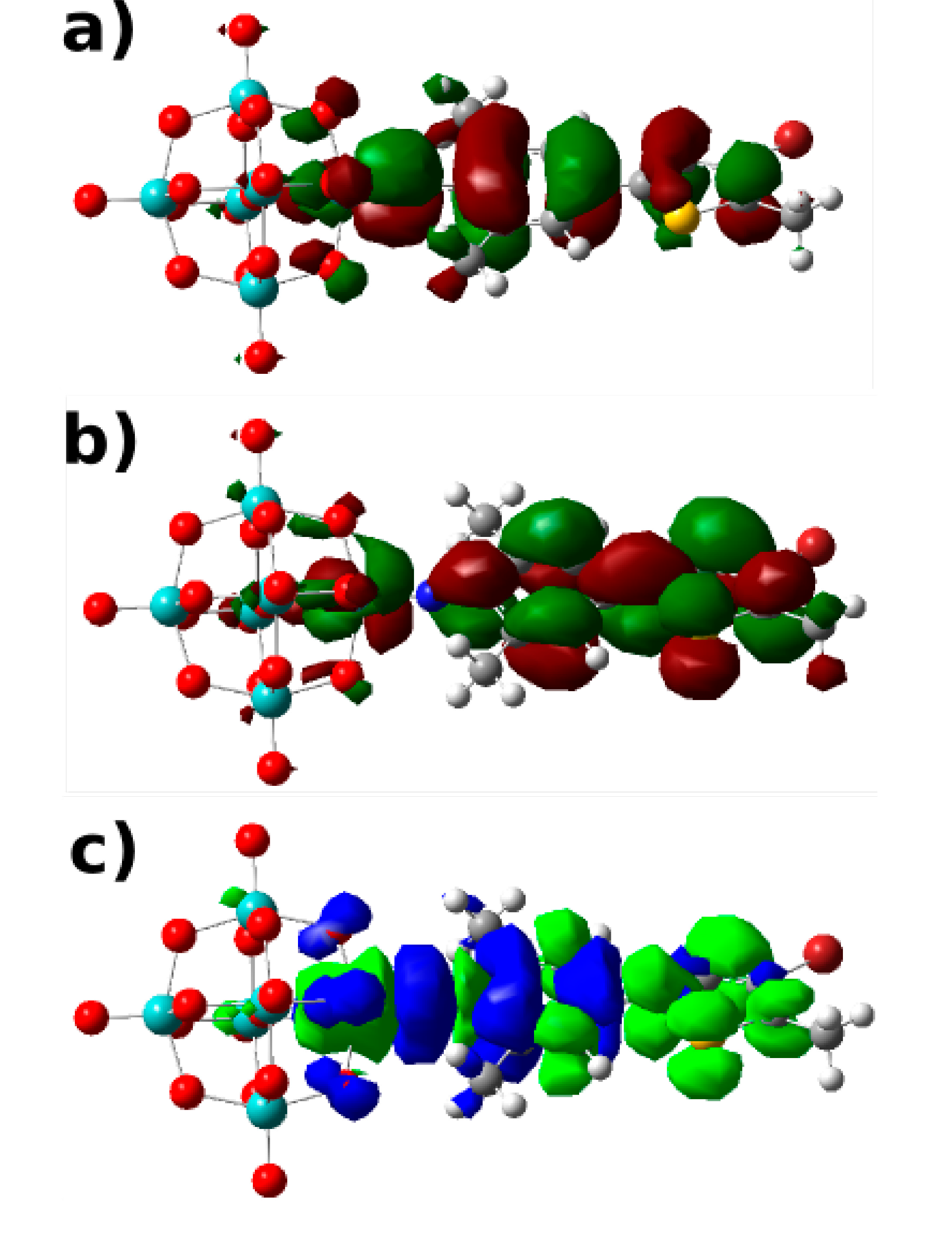
A3.5. Computational Details
A3.6. Cartesian Coordinates
| Cartesian Coordinates (Å) | |||
|---|---|---|---|
| H | 3.76704225 | 2.03847542 | 0.30065425 |
| C | 8.79961983 | −2.01560166 | 0.41244729 |
| H | 8.87077619 | −2.73103735 | −0.41655383 |
| H | 9.69833234 | −1.39231526 | 0.40394112 |
| H | 8.78913391 | −2.59102756 | 1.34634501 |
| C | 0.96256278 | 0.13987740 | −0.04620859 |
| C | 1.56254262 | −1.14797725 | −0.14917984 |
| C | 1.77530740 | 1.29901199 | 0.10936217 |
| C | 2.94484244 | −1.24373548 | −0.09804489 |
| H | 3.40180409 | −2.22830515 | −0.19445924 |
| C | 1.10163712 | 2.63869224 | 0.22587839 |
| H | 0.50481002 | 2.85061645 | −0.66856868 |
| H | 1.83866424 | 3.43685064 | 0.36309774 |
| H | 0.39680786 | 2.64829174 | 1.06459060 |
| C | 7.59386438 | −1.14230232 | 0.28740460 |
| C | 6.18839822 | 0.74582657 | 0.03186394 |
| H | 5.97589682 | 1.79675983 | −0.11316036 |
| C | 3.15321125 | 1.15160124 | 0.15798309 |
| C | 3.77009862 | −0.11209650 | 0.05490087 |
| C | 0.67156702 | −2.34846316 | −0.30992473 |
| H | −0.01800377 | −2.43524581 | 0.53732293 |
| H | 1.26335447 | −3.26621562 | −0.38903123 |
| H | 0.03677621 | −2.24804774 | −1.19713465 |
| C | 5.21655957 | −0.23465473 | 0.11026329 |
| C | 7.50055059 | 0.21728381 | 0.13429621 |
| N | −0.37806453 | 0.25874441 | −0.08920770 |
| O | −6.30236442 | 1.57923483 | −1.41436027 |
| O | −4.37228812 | 0.26702048 | −0.09343012 |
| O | −2.55751051 | 1.59712153 | −1.44792530 |
| O | −6.29892939 | 1.59573674 | 1.21226320 |
| O | −4.41902666 | 2.91549342 | −0.08453437 |
| O | −2.55711831 | 1.61203372 | 1.24718283 |
| O | −4.42922706 | 0.27669877 | −2.74280726 |
| O | −6.29344006 | −1.04499882 | 1.23892385 |
| O | −4.43233273 | 0.25667581 | 2.55667783 |
| O | −6.29498094 | −1.05586386 | −1.40947046 |
| O | −2.55700529 | −1.08720380 | −1.42217494 |
| O | −4.46304402 | 3.15757998 | −2.93033868 |
| O | −2.55518400 | −1.06732613 | 1.24923151 |
| O | −4.45481996 | 3.10375150 | 2.79799406 |
| O | −8.47713797 | 0.24262438 | −0.08541032 |
| O | −4.44104229 | −2.38607044 | −0.10306189 |
| O | −4.42260783 | −2.62719372 | 2.74380416 |
| O | −4.43177656 | −2.57234737 | −2.98509932 |
| S | 5.98186692 | −1.79775572 | 0.31727666 |
| BR | 9.04012899 | 1.35225338 | 0.04645216 |
| MO | −4.45217513 | 1.95216728 | −1.72269566 |
| MO | −6.77320756 | 0.24249817 | −0.08640557 |
| MO | −4.44116305 | 1.89427511 | 1.59437427 |
| MO | −2.16185379 | 0.29537333 | −0.10077312 |
| MO | −4.40705584 | −1.42246356 | 1.53532479 |
| MO | −4.41479636 | −1.36432115 | −1.77989579 |
A3.7. Crystallography
| Parameter | L3 | 1-L4 |
|---|---|---|
| Formula | C26H22NSBr | Mo6O18N3C45H84SBr |
| Fw | 460.41 | 1642.76 |
| Crystal Class | Monoclinic | Monoclinic |
| Space Group | P21/n | P21/n |
| a (Å) | 12.5989(5) | 11.03023(16) |
| b (Å) | 11.5694(5) | 16.9232(2) |
| c (Å) | 15.2670(6) | 32.4507(5) |
| α (°) | 90 | 90 |
| β (°) | 101.954(4) | 98.8238(13) |
| γ (°) | 90 | 90 |
| V (Å3) | 2177.07(16) | 5985.79(15) |
| Z | 4 | 4 |
| T (K) | 130.01(10) | 130.01(10) |
| μ (mm−1) | 1.996 | 11.658 |
| reflns measd | 13729 | 105913 |
| unique reflns | 5687 | 12538 |
| R1 [I > 2σ(I)] | 0.0497 | 0.0898 |
| wR2(all data) | 0.1727 | 0.2471 |
A3.8. Thermal Gravimetric Analysis
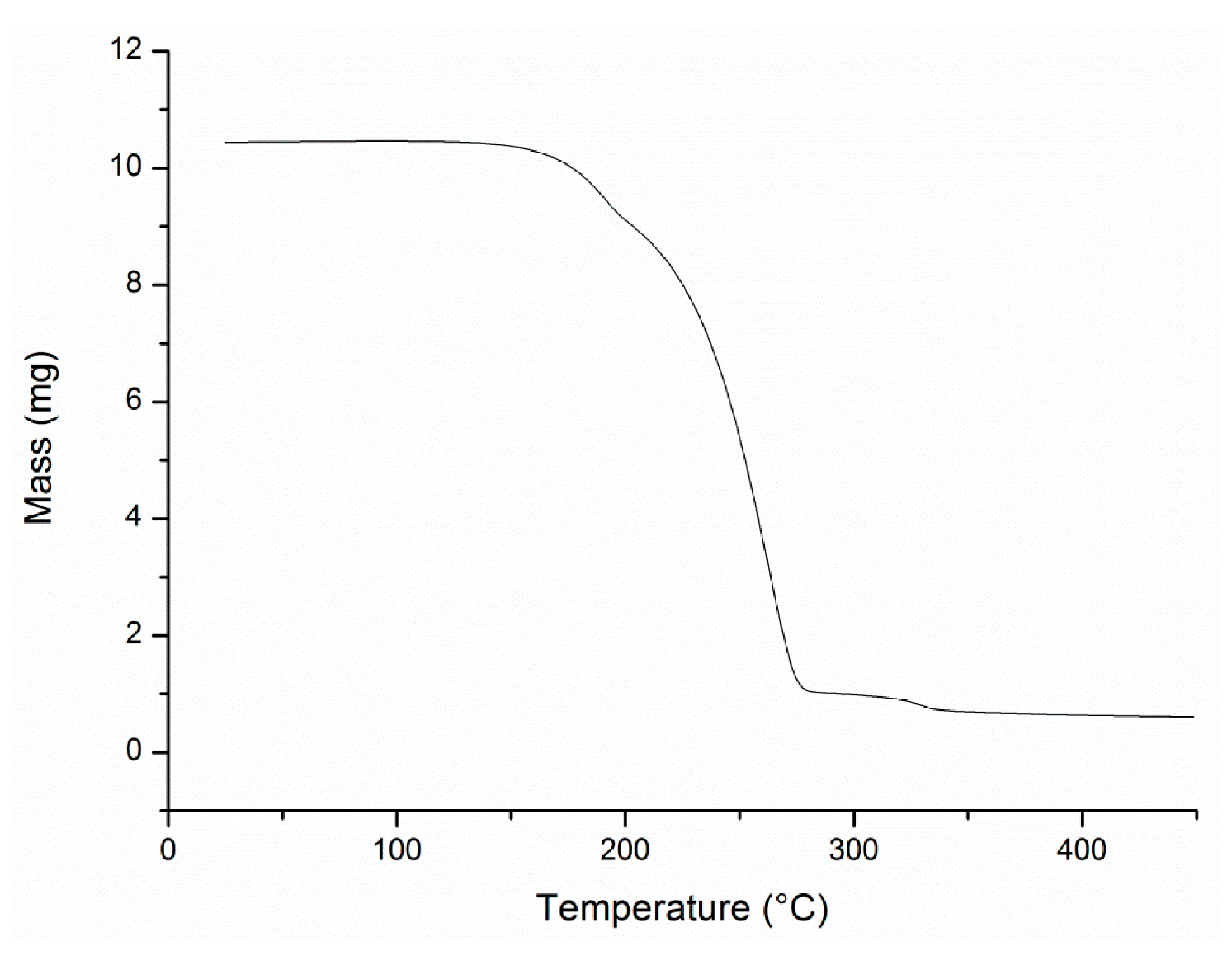
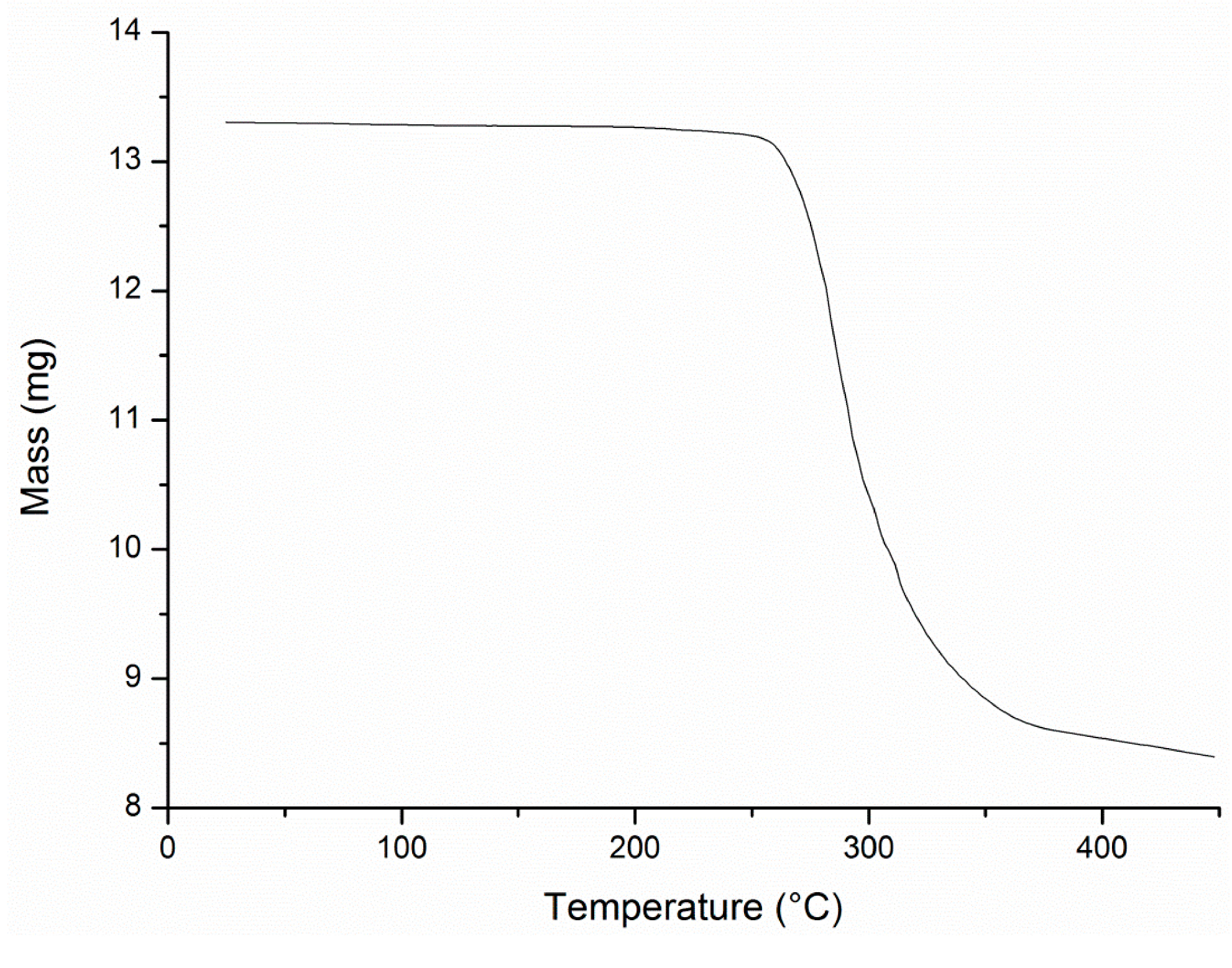
Conflicts of Interest
References
- An, H.Y.; Wang, E.B.; Xiao, D.R.; Li, Y.G.; Su, Z.M.; Xu, L. Chiral 3D architectures with helical channels constructed from polyoxometalate clusters and copper-amino acid complexes. Angew. Chem. Int. Ed. Engl. 2006, 45, 904–908. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Song, Y.; Cronin, L.; Liu, T. Self-assembly of organic-inorganic hybrid amphiphilic surfactants with large polyoxometalates as polar head groups. J. Am. Chem. Soc. 2008, 130, 14408–14409. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.F.; McMillan, N.; Long, D.L.; Thiel, J.; Ding, Y.; Chen, H.; Gadegaard, N.; Cronin, L. Design of hydrophobic polyoxometalate hybrid assemblies beyond surfactant encapsulation. Chemistry 2008, 14, 2349–2354. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; McMillan, N.; Long, D.; Kane, S.; Malm, J.; Riehle, M.O.; Pradeep, C. P.; Gadegaard, N.; Cronin, L. Micropatterned surfaces with covalently grafted unsymmetrical polyoxometalate-hybrid clusters lead to selective cell adhesion. J. Am. Chem. Soc. 2009, 131, 1340–1341. [Google Scholar] [CrossRef] [PubMed]
- Aronica, C.; Chastanet, G.; Zueva, E.; Borshch, S.A.; Clemente-Juan, J.M.; Luneau, D. A mixed-valence polyoxovanadate(III,IV) cluster with a calixarene cap exhibiting ferromagnetic V(III)-V(IV) interactions. J. Am. Chem. Soc. 2008, 130, 2365–2371. [Google Scholar] [CrossRef] [PubMed]
- Pan, D.; Chen, J.; Tao, W.; Nie, L.; Yao, S. Polyoxometalate-modified carbon nanotubes: New catalyst support for methanol electro-oxidation. Langmuir 2006, 22, 5872–5876. [Google Scholar] [CrossRef] [PubMed]
- Strong, J.B.; Yap, G.P.A.; Ostrander, R.; Liable-Sands, L.M.; Rheingold, A.L.; Thouvenot, R.; Gouzerh, P.; Maatta, E.A. A New Class of Functionalized Polyoxometalates: Synthetic, Structural, Spectroscopic, and Electrochemical Studies of Organoimido Derivatives of [Mo6O19]2−. J. Am. Chem. Soc. 2000, 122, 639–649. [Google Scholar] [CrossRef]
- Proust, A.; Thouvenot, R.; Chaussade, M.; Robert, F.; Gouzerh, P. Phenylimido derivatives of [Mo6O19]2−: Syntheses, X-ray structures, vibrational, electrochemical, 95Mo and 14N NMR studies. Inorg. Chim. Acta 1994, 224, 81–95. [Google Scholar] [CrossRef]
- Moore, A.R.; Kwen, H.; Beatty, A.M.; Maatta, E.A. Organoimido-polyoxometalates as polymer pendants. Chem. Commun. 2000, 996, 1793–1794. [Google Scholar] [CrossRef]
- Wei, Y.; Xu, B.; Barnes, C.L.; Peng, Z. An Efficient and Convenient Reaction Protocol to Organoimido Derivatives of Polyoxometalates. J. Am. Chem. Soc. 2001, 123, 4083–4084. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Li, Q.; Ge, N.; Wei, Y.; Wang, Y.; Wang, P.; Guo, H. An Easy Route to Monofunctionalized Organoimido Derivatives of the Lindqvist Hexamolybdate. Eur. J. Inorg. Chem. 2004, 2004, 2819–2822. [Google Scholar] [CrossRef]
- Li, Q.; Wu, P.; Xia, Y.; Wei, Y.; Guo, H. Synthesis, spectroscopic studies and crystal structure of a polyoxoanion cluster incorporating para-bromophenylimido ligand, (Bu4N)2[Mo6O18(NC6H4Br-p)]. J. Organomet. Chem. 2006, 691, 1223–1228. [Google Scholar] [CrossRef]
- Xu, B.; Wei, Y.; Barnes, C.L. Hybrid Molecular Materials Based on and Organic Conjugated Systems. Angew. Chem. Int. Ed. Engl. 2001, 40, 2290–2292. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Wei, Y.; Xu, B.; Cheung, C.F.C.; Peng, Z.; Powell, D.R. Hybrid molecular dumbbells: bridging polyoxometalate clusters with an organic pi-conjugated rod. Angew. Chem. Int. Ed. Engl. 2002, 41, 1566–1568. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Nelson, J.A.; Lu, M.; Xie, B.; Peng, Z.; Powell, D.R. Charge-transfer hybrids containing covalently bonded polyoxometalates and ferrocenyl units. Inorg. Chem. 2004, 43, 6408–6413. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Xie, B.; Kang, J.; Chen, F.; Peng, Z. Synthesis of Main-Chain Polyoxometalate-Containing Hybrid Polymers and Their Applications in Photovoltaic Cells. Chem. Mater. 2005, 17, 402–408. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, L.; Hao, J.; Yin, P.; Zhang, J.; Li, Q.; Zhu, L.; Wei, Y. Palladium-catalyzed Heck reaction of polyoxometalate-functionalised aryl iodides and bromides with olefins. Chem. Eur. J. 2009, 15, 3076–3080. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Peng, Z.; Wei, Y.; Powell, D.R. Polyoxometalates covalently bonded with terpyridine ligands. Chem. Commun. 2003, 375, 2562–2563. [Google Scholar] [CrossRef]
- Hirata, S.; Head-Gordon, M. Time-dependent density functional theory within the Tamm-Dancoff approximation. Chem. Phys. Lett. 1999, 314, 291–299. [Google Scholar] [CrossRef]
- Yanai, T.; Tew, D.P.; Handy, N.C. A new hybrid exchange-correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem. Phys. Lett. 2004, 393, 51–57. [Google Scholar] [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.; Perdew, J.; Staroverov, V.; Scuseria, G. Climbing the Density Functional Ladder: Nonempirical Meta-Generalized Gradient Approximation Designed for Molecules and Solids. Phys. Rev. Lett. 2003, 91, 146401. [Google Scholar] [CrossRef] [PubMed]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef] [PubMed]
- Grimme, S.; Ehrlich, S.; Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 2011, 32, 1456–1465. [Google Scholar] [CrossRef] [PubMed]
- Goerigk, L.; Grimme, S. Assessment of TD-DFT methods and of various spin scaled CIS(D) and CC2 versions for the treatment of low-lying valence excitations of large organic dyes. J. Chem. Phys. 2010, 132, 184103. [Google Scholar] [CrossRef]
- Janjua, M.R.S.A. Quantum mechanical design of efficient second-order nonlinear optical materials based on heteroaromatic imido-substituted hexamolybdates: First theoretical framework of POM-based heterocyclic aromatic rings. Inorg. Chem. 2012, 51, 11306–11314. [Google Scholar] [CrossRef] [PubMed]
- Melcamu, Y.Y.; Wen, S.; Yan, L.; Zhang, T.; Su, Z. Theoretical investigation of second-order nonlinear optical response by linking hexamolybdate with graphene in the donor-acceptor (D-A) framework. Mol. Simul. 2013, 39, 214–219. [Google Scholar] [CrossRef]
- Osakai, T.; Himeno, S.; Saito, A.; Hori, T. Electrochemical reduction of hexamolybdate(2−) ion in acidic aqueous-organic media. J. Electroanal. Chem. Interfacial Electrochem. 1990, 285, 209–221. [Google Scholar] [CrossRef]
- MarvinSketch. Available online: http://www.chemaxon.com (accessed on 27 April 2015).
- Borg, S.J.; Best, S.P. Spectroelectrochemical cell for the study of interactions between redox-activated species and moderate pressures of gaseous substrates. J. Electroanal. Chem. 2002, 535, 57–64. [Google Scholar] [CrossRef]
- Lee, B.Y.; Kwon, H.Y.; Lee, S.Y.; Na, S.J.; Han, S.I.; Yun, H.; Lee, H.; Park, Y.W. Bimetallic anilido-aldimine zinc complexes for epoxide/CO2 copolymerization. J. Am. Chem. Soc. 2005, 127, 3031–3037. [Google Scholar] [CrossRef] [PubMed]
- Furche, F.; Ahlrichs, R.; Hättig, C.; Klopper, W.; Sierka, M.; Weigend, F. Turbomole. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2014, 4, 91–100. [Google Scholar] [CrossRef]
- Neese, F. The ORCA program system. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2012, 2, 73–78. [Google Scholar] [CrossRef]
- Lin, Y.; Li, G.; Mao, S.; Chai, J. Long-Range Corrected Hybrid Density Functionals with Improved Dispersion Corrections. J. Chem. Theory Comput. 2013, 9, 263–272. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Healey, M.R.; Best, S.P.; Goerigk, L.; Ritchie, C. A Heteroaromatically Functionalized Hexamolybdate. Inorganics 2015, 3, 82-100. https://doi.org/10.3390/inorganics3020082
Healey MR, Best SP, Goerigk L, Ritchie C. A Heteroaromatically Functionalized Hexamolybdate. Inorganics. 2015; 3(2):82-100. https://doi.org/10.3390/inorganics3020082
Chicago/Turabian StyleHealey, Merinda R., Stephen P. Best, Lars Goerigk, and Chris Ritchie. 2015. "A Heteroaromatically Functionalized Hexamolybdate" Inorganics 3, no. 2: 82-100. https://doi.org/10.3390/inorganics3020082
APA StyleHealey, M. R., Best, S. P., Goerigk, L., & Ritchie, C. (2015). A Heteroaromatically Functionalized Hexamolybdate. Inorganics, 3(2), 82-100. https://doi.org/10.3390/inorganics3020082





