X-Optogenetics and U-Optogenetics: Feasibility and Possibilities
Abstract
:1.Introduction
Optogenetics
2. Methodology
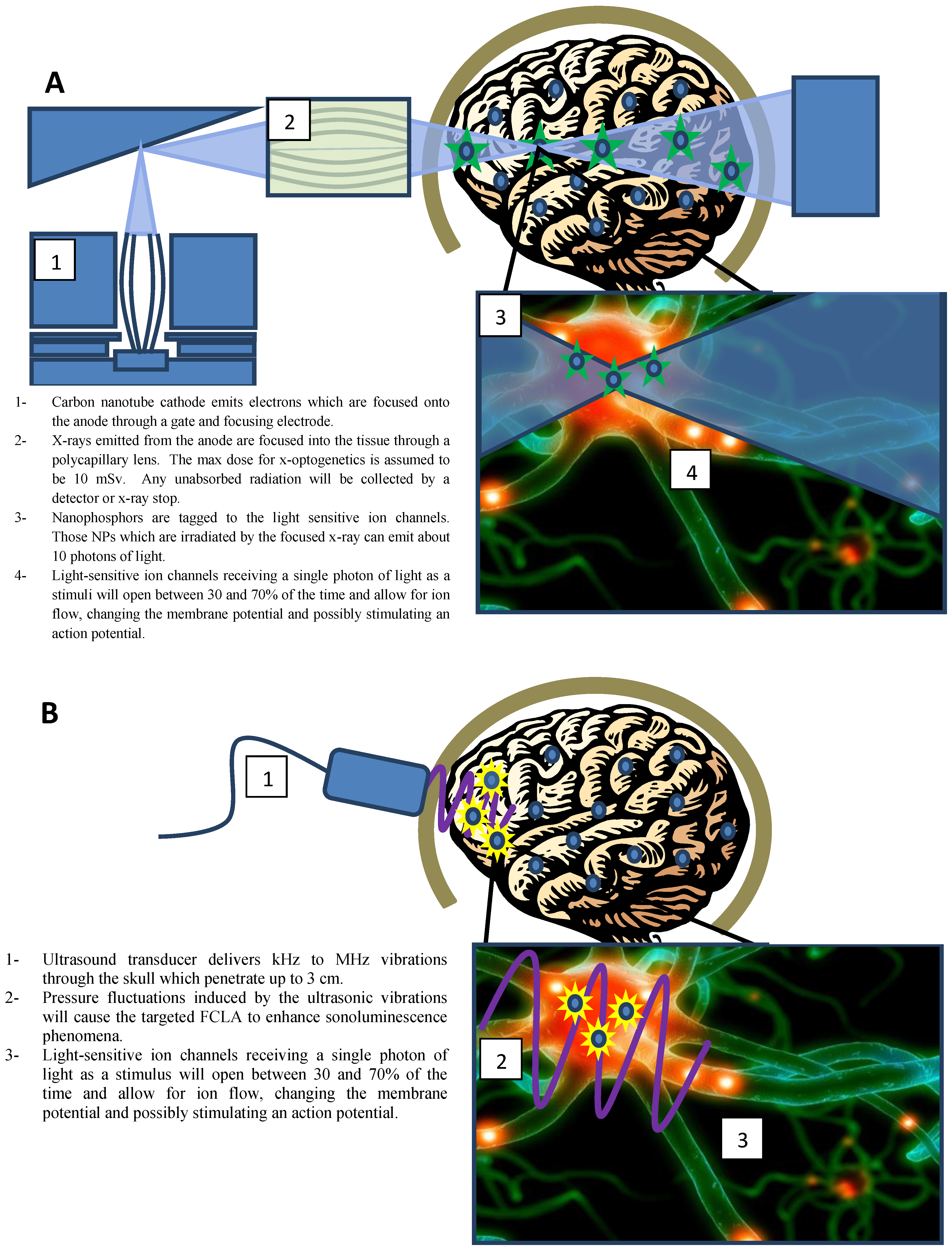
2.1. Key Elements for X-Optogenetics
2.1.1. X-Ray Excitable Nanophosphors
| Formula | Source | Emission Maximum (nm) | Excitation Wavelength (nm) | Conversion Efficiency (%) | Size (nm) | Dispersible | Toxicity |
|---|---|---|---|---|---|---|---|
| Gd2O2S:Eu3+ (Tb3+) | [19,20,21,22] | 620 (545) | <310 | 15 | 50–300 | Yes, PGA-PEG | Low |
| Y2O3:Eu3+ | [23,24,25] | 610 | <310 | 80 | 10–50 | Yes | -- |
| LiGa5O8:Cr3+ | [26,27] | 716 | <310 | -- | 50–150 | Yes, PEI | Low |
| Gd2O2S:Yb(8),Er(1) | [28] | 500–700 | 980 | 25 | 4 µm | Yes | Low |
| NaMF4 :Yb3+/Ln3+ | [29] | 510–560 | 980 | -- | 60 | Yes, DSPE-PEG-COOH | Low |
| La(OH)3:Eu3+ | [30] | 597, 615 | 280 | -- | 3.5 | Yes, PEG | Low |
| NaYF4:Yb/Er | [31] | 520, 540, 654 | 980 | -- | 33 ± 1 | Yes, citrate | -- |
| NaYF4:40%Eu3+ | [32] | 592 | 394 | -- | 28 | Yes, PAA | Low |
| NaYF4:40%Tb3+ | [32] | 545 | 368 | -- | 28 | Yes, PAA | Low |
| cit-NaLuF4:Yb,Tm | [33] | 800 | 980 | -- | 25 | Yes, citric acid | Low |
| Ba2SiO4 | [34] | 505 | 350 | 38.6 | 40–50 | -- | -- |
| Na2Sr2Al2PO4F9: Eu3+ | [35] | 593, 619 | 393 | -- | 35.26 | -- | Non-toxic materials |
| BaMgAl10O17:Eu2+ | [36] | 450 | 147 | -- | 62, 85, 115, 160, 450 | -- | -- |
| Sr2CeO4 | [37] | 467–485 | 240–360 | -- | 45 | -- | -- |
| LiCaPO4:Eu2+0.03 | [38] | 476 | 375 | Quantum Efficiency: 53.7, 67.6 | Yes, PEG-P | -- | |
| PEG-Er-Y2O3 | [39] | 660 | 980 | -- | 30–60 | Yes, PEG | Low |
| GdVO4:Eu3+ | [40] | 620 | 330 | -- | 6 | Yes | Low |
2.1.2. Nanoparticle Targeting
2.1.3. X-Ray Focusing
| Ion Channel/Pump | Channelrhodopsin 2 (ChR2) | Halorhodopsin (NphR) | Archeorhodopsin (Arch) |
|---|---|---|---|
| Channel/Pump Mass | 30 kDa [51] | 30 kDa [52] | 28 kDa [53] |
| Minimum Channel/Pump Radius (assuming spherical) | 2.58–2.72 nm [54] | 2.05 nm [54] | 2.00 nm [54] |
| Intensity | 2–20 mW/mm2 [55] | 5.4 ± 0.2 mW/mm2 [56] | <10 mW/mm2 [56] |
| Wavelength | 488 nm [55] | 532 nm [57] | 532 nm [57] |
| Pulse Train | 5 ms, 40 Hz [55] | 15 s illumination [57] | 15 s illumination [57] |
| Depolarizing/Hyperpolarizing | Depolarizing | Hyperpolarizing | Hyperpolarizing |
| Possible Nanophosphors | BaMgAl10O17:Eu2+ LiCaPO4:Eu2+0.03 | Gd2O2S:Tb3+ (combination doping?) | Gd2O2S:Tb3+ (combination doping?) |
| Hardware Specifications/Involved Components | SOURCE: Carbon Nanotube (peak ~8 keV, pulsing capability) FOCUSING ELEMENT: polycapillary lens OR Fresnel zone plate | ||
2.1.4. X-ray Carbon Nano-Tube (CNT) Sources
2.1.5. X-Ray Dose
2.2. X-Optogenetics Safety Analysis
2.3. U-Optogenetics via Sonoluminescence
3. Discussions and Conclusions
Author Contributions
Conflicts of Interest
References
- Fenno, L.; Yizhar, O.; Deisseroth, K. The development and application of optogenetics. Annu. Rev. Neurosci. 2011, 34, 389–412. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Xing, D.; Tan, S.; Tang, Y.; Ueda, K. In vivo sonoluminescence imaging with the assistance of FCLA. Phys. Med. Biol. 2002, 47, 1535. [Google Scholar] [CrossRef] [PubMed]
- Optogenetics: Controlling the brain with light. Available online: http://video.mit.edu/watch/optogenetics-controlling-the-brain-with-light-7659/ (accessed on 27 September 2014).
- Adams, A. Optogenetics earns Deisseroth Keio Prize in Medicine. Available online: https://med.stanford.edu/news/all-news/2014/09/optogenetics-earns-deisseroth-keio-prize-in-medicine.html (accessed on 26 September 2014).
- Knopfel, T.; Boyden, E.S. Optogenetics: Tools for Controlling and Monitoring Neuronal Activity; Elsevier: Oxford, UK, 2012. [Google Scholar]
- Grossman, N.; Poher, V.; Grubb, M.; Kennedy, G.; Nikolic, K.; McGovern, B.; Berlinguer, R.; Palmini, R.; Gong, Z.; Drakakis, E.; Neil, M.; et al. Multi-site optical excitation using ChR2 and micro-LED array. J. Neural Eng 2010. [Google Scholar] [CrossRef]
- Farah, N.; Reutsky, I.; Shoham, S. Patterned optical activation of retinal ganglion cells. Conf Proc IEEE Eng Med Biol Soc 2007, 2007, 6368–6370. [Google Scholar]
- Lutz, C.; Otis, T.; DeSars, V.; Charpak, S.; DiGregorio, D.; Emiliani, V. Holographic photolysis of caged neurotransmitters. Nat Methods 2008. [Google Scholar] [CrossRef]
- Clements, I.P.; Gnadea, A.G.; Rusha, A.D.; Pattena, C.D.; Twomeya, M.C.; Kravitzb, A.V. Miniaturized LED sources for in vivo optogenetic experimentation. Proc. SPIE 2013, 8586, 85860X–1. [Google Scholar]
- Entcheva, E.; Williams, J.C. Channelrhodopsin2 Current During the Action Potential:[ldquo] Optical AP Clamp [rdquo] and Approximation. Sci. Rep. 2014. [Google Scholar] [CrossRef]
- Fessenden, J. EUREKA grant to fund development of new “optogenetic” technique for mapping neural networks at UMMS. Available online: http://www.eurekalert.org/pub_releases/2013-10/uomm-egt102413.php (accessed on 26 September 2014).
- Jagdeo, J.R.; Adams, L.E.; Brody, N.I.; Siegel, D.M. Transcranial red and near infrared light transmission in a cadaveric model. PLoS One 2012, 7, e47460. [Google Scholar] [CrossRef] [PubMed]
- Stolik, S.; Delgado, J.A.; Perez, A.; Anasagasti, L. Measurement of the penetration depths of red and near infrared light in human “ex vivo” tissues. J. Photochem. Photobiol. B Biol. 2000, 57, 90–93. [Google Scholar] [CrossRef]
- Sowell, E.R.; Thompson, P.M.; Leonard, C.M.; Welcome, S.E.; Kan, E.; Toga, A.W. Longitudinal mapping of cortical thickness and brain growth in normal children. J. Neurosci. 2004, 24, 8223–8231. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Pratx, G.; Carpenter, C.M.; Liu, H.; Cheng, Z.; Gambhir, S.S.; Xing, L. Synthesis and Radioluminescence of PEGylated Eu3+-doped Nanophosphors as Bioimaging Probes. Adv. Mater. 2011, 23, H195–H199. [Google Scholar] [CrossRef] [PubMed]
- Shuba, Y.M. Use of scintillator-based nanoparticles for in vivo control of light-sensitive bioactive molecules 2014. Patent US20140219922, 2014. [Google Scholar]
- Stark, G. X-ray. Available online: http://www.britannica.com/EBchecked/topic/650351/X-ray (accessed on 27 October 2014).
- Ahmad, M.; Pratx, G.; Bazalova, M.; Xing, L. X-ray luminescence and x-ray fluorescence computed tomography: new molecular imaging modalities. IEEE Access 2014. [Google Scholar] [CrossRef]
- Innovation in Dispersible Nanomaterials. Available online: http://www.oceannanotech.com/product.php?cid=92&pid=286 (accessed on 27 October 2014).
- Chen, H.; Moore, T.; Qi, B.; Colvin, D.C.; Jelen, E.K.; Hitchcock, D.A.; He, J.; Mefford, O.T.; Gore, J.C.; Alexis, F. Monitoring pH-triggered drug release from radioluminescent nanocapsules with X-ray excited optical luminescence. ACS Nano 2013, 7, 1178–1187. [Google Scholar] [CrossRef] [PubMed]
- Moore, T.L.; Wang, F.; Chen, H.; Grimes, S.W.; Anker, J.N.; Alexis, F. Polymer-Coated Radioluminescent Nanoparticles for Quantitative Imaging of Drug Delivery. Adv. Funct. Mater. 2014, 24, 5815–5823. [Google Scholar] [CrossRef]
- Gorokhova, E.I.; Demidenko, V.A.; Mikhrin, S.B.; Rodnyi, P.A.; Van Eijk, C.W.E. Luminescence and scintillation properties of Gd2O2S: Tb Ce ceramics. IEEE Trans. Nucl. Sci. 2005, 52, 3129–3132. [Google Scholar] [CrossRef]
- Jadhav, A.P.; Pawar, A.U.; Pal, U.; Kang, Y.S. Red emitting Y2O3:Eu3+ nanophosphors with >80% down conversion efficiency. J. Mater. Chem. C 2014. [Google Scholar] [CrossRef]
- Dhanaraj, J.; Jagannathan, R.; Kutty, T.R.N.; Lu, C.-H. Photoluminescence characteristics of Y2O3: Eu3+ nanophosphors prepared using sol-gel thermolysis. J. Phys. Chem. B 2001, 105, 11098–11105. [Google Scholar] [CrossRef]
- Van Hao, B.; Huy, P.T.; Khiem, T.N.; Ngan, N.T.T.; Duong, P.H. Synthesis of Y2O3: Eu3+ micro-and nanophosphors by sol-gel process. J. Phys. Conf. Series 2009, 187, 12074. [Google Scholar] [CrossRef]
- Liu, F.; Yan, W.; Chuang, Y.-J.; Zhen, Z.; Xie, J.; Pan, Z. Photostimulated near-infrared persistent luminescence as a new optical read-out from Cr3+-doped LiGa5O8. Sci. Rep. 2013. [Google Scholar] [CrossRef]
- Chuang, Y.-J.; Zhen, Z.; Zhang, F.; Liu, F.; Mishra, J.P.; Tang, W.; Chen, H.; Huang, X.; Wang, L.; Chen, X. Photostimulable Near-Infrared Persistent Luminescent Nanoprobes for Ultrasensitive and Longitudinal Deep-Tissue Bio-Imaging. Theranostics 2014, 4, 1112. [Google Scholar] [CrossRef] [PubMed]
- Ajithkumar, G.; Yoo, B.; Goral, D.E.; Hornsby, P.J.; Lin, A.-L.; Ladiwala, U.; Dravid, V.P.; Sardar, D.K. Multimodal bioimaging using a rare earth doped Gd2O2S:Yb/Er phosphor with upconversion luminescence and magnetic resonance properties. J. Mater. Chem. B 2013, 1, 1561–1572. [Google Scholar] [CrossRef]
- Li, L.; Zhang, R.; Yin, L.; Zheng, K.; Qin, W.; Selvin, P.R.; Lu, Y. Biomimetic Surface Engineering of Lanthanide‐Doped Upconversion Nanoparticles as Versatile Bioprobes. Angew. Chemie 2012, 124, 6225–6229. [Google Scholar] [CrossRef]
- Sun, C.; Carpenter, C.; Pratx, G.; Xing, L. Facile synthesis of amine-functionalized Eu3+-doped La(OH)3 nanophosphors for bioimaging. Nanoscale Res. Lett 2011, 6, 24. [Google Scholar]
- Cao, T.; Yang, T.; Gao, Y.; Yang, Y.; Hu, H.; Li, F. Water-soluble NaYF4:Yb/Er upconversion nanophosphors: Synthesis, characteristics and application in bioimaging. Inorg. Chem. Commun. 2010, 13, 392–394. [Google Scholar] [CrossRef]
- Olesiak-Banska, J.; Nyk, M.; Kaczmarek, D.; Matczyszyn, K.; Pawlik, K.; Samoc, M. Synthesis and optical properties of water-soluble fluoride nanophosphors co-doped with Eu3+ and Tb3+. Opt. Mater. (Amst). 2011, 33, 1419–1423. [Google Scholar] [CrossRef]
- Sun, Y.; Peng, J.; Feng, W.; Li, F. Upconversion nanophosphors NaluF4:Yb, Tm for lymphatic imaging in vivo by real-time upconversion luminescence imaging under ambient light and high-resolution X-ray CT. Theranostics 2013, 3, 346. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-Y.; Huang, C.K.; Hung, W.B.; Sun, K.W.; Chen, T.M. Efficiency improvement of Si solar cells using metal-enhanced nanophosphor fluorescence. Sol. Energy Mater. Sol. Cells 2014, 120, 168–174. [Google Scholar] [CrossRef]
- Dhoble, S.J.; Shinde, K.N. Ce3+ and Eu3+ activtated Na2Sr2Al2PO4F9 nanophosphor. Adv. Mater. Lett. 2011, 2, 349. [Google Scholar]
- Yadav, R.S.; Pandey, S.K.; Pandey, A.C. Blue-Shift and Enhanced Photoluminescence in BaMgAl10O17:Eu2+ Nanophosphor under VUV Excitation for PDPs Application. Mater. Sci. Appl. 2010, 1, 25. [Google Scholar]
- Niyaz Parvin, S.; Poornachandra Rao, N.V.; Subba Rao, B.; Murthy, K.V.R. Photoluminescence Studies on Sr2CeO4 Nanophosphor. Wourld J. Chem. 2011, 6, 114–117. [Google Scholar]
- Minsung, K.; Makoto, K.; Hideki, K.; Masato, K. A Highly Luminous LiCaPO4: Eu2+ Phosphor Synthesized by a Solution Method Employing a Water-Soluble Phosphate Ester. Opt. Photonics J. 2013. [Google Scholar] [CrossRef]
- Zako, T.; Nagata, H.; Terada, N.; Sakono, M.; Soga, K.; Maeda, M. Improvement of dispersion stability and characterization of upconversion nanophosphors covalently modified with PEG as a fluorescence bioimaging probe. J. Mater. Sci. 2008, 43, 5325–5330. [Google Scholar] [CrossRef]
- Dong, K.; Ju, E.; Liu, J.; Han, X.; Ren, J.; Qu, X. Ultrasmall biomolecule-anchored hybrid GdVO 4 nanophosphors as a metabolizable multimodal bioimaging contrast agent. Nanoscale 2014, 6, 12042–12049. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Jiang, X. Influence of particle size on transport of methotrexate across blood brain barrier by polysorbate 80-coated polybutylcyanoacrylate nanoparticles. Int. J. Pharm. 2006, 310, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, K.; Sarkar, S.; Jagajjanani Rao, K.; Paria, S. Core/shell nanoparticles in biomedical applications. Adv. Colloid Interface Sci. 2014, 209, 8–39. [Google Scholar] [CrossRef] [PubMed]
- Yizhar, O.; Fenno, L.E.; Davidson, T.J.; Mogri, M.; Deisseroth, K. Optogenetics in neural systems. Neuron 2011, 71, 9–34. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, J.M.; Berry, C.C.; Riehle, M.O.; Curtis, A.S.G. Nanoparticle targeting at cells. Langmuir 2006, 22, 3286–3293. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Drummond, D.C.; Zou, H.; Hayes, M.E.; Adams, G.P.; Kirpotin, D.B.; Marks, J.D. Impact of single-chain Fv antibody fragment affinity on nanoparticle targeting of epidermal growth factor receptor-expressing tumor cells. J. Mol. Biol. 2007, 371, 934–947. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowski, K.M.; Dul, D.T.; Korecki, P. X-ray imaging inside the focal spot of polycapillary optics using the coded aperture concept. Opt. Express 2013, 21, 2920–2927. [Google Scholar] [CrossRef] [PubMed]
- Romanov, A.Y. Optic parameters of a middle-focus Kumakhov lens for hard X-rays. Tech. Phys. Lett. 2005, 31, 200–201. [Google Scholar] [CrossRef]
- Cong, W.; Xi, Y.; Wang, G. X-Ray Fluorescence Computed Tomography With Polycapillary Focusing. IEEE Access 2014. [Google Scholar] [CrossRef]
- Di Fabrizio, E.; Romanato, F.; Gentili, M.; Cabrini, S.; Kaulich, B.; Susini, J.; Barrett, R. High-efficiency multilevel zone plates for keV X-rays. Nature 1999, 401, 895–898. [Google Scholar] [CrossRef]
- Nagel, G.; Szellas, T.; Huhn, W.; Kateriya, S.; Adeishvili, N.; Berthold, P.; Ollig, D.; Hegemann, P.; Bamberg, E. Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc. Natl. Acad. Sci. 2003, 100, 13940–13945. [Google Scholar] [CrossRef] [PubMed]
- Agrisera Antibodies Product Information. Available online: http://www.agrisera.com/cgi-bin/ibutik/SkapaFaktura.pl?SkrivPDF=J&funk=visa_artikel&artnr=AS121851&Friendly_Grupp=bacterial-insect-and-fungal-&Friendly=nphr-halorhodopsin-&artgrp=28&Sprak=en (accessed on 5 November 2014).
- Uegaki, K.; Sugiyama, Y.; Mukohata, Y. Archaerhodopsin-2, from Halobacterium sp. aus-2 further reveals essential amino acid residues for light-driven proton pumps. Arch. Biochem. Biophys. 1991, 286, 107–110. [Google Scholar] [CrossRef] [PubMed]
- Erickson, H.P. Size and shape of protein molecules at the nanometer level determined by sedimentation, gel filtration, and electron microscopy. Biol. Proced. Online 2009, 11, 32–51. [Google Scholar] [CrossRef] [PubMed]
- Arenkiel, B.R.; Peca, J.; Davison, I.G.; Feliciano, C.; Deisseroth, K.; Augustine, G.J.; Ehlers, M.D.; Feng, G. In vivo light-induced activation of neural circuitry in transgenic mice expressing channelrhodopsin-2. Neuron 2007, 54, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Mattis, J.; Tye, K.M.; Ferenczi, E.A.; Ramakrishnan, C.; O’Shea, D.J.; Prakash, R.; Gunaydin, L.A.; Hyun, M.; Fenno, L.E.; Gradinaru, V. Principles for applying optogenetic tools derived from direct comparative analysis of microbial opsins. Nat. Methods 2012, 9, 159–172. [Google Scholar] [CrossRef]
- Raimondo, J.V.; Kay, L.; Ellender, T.J.; Akerman, C.J. Optogenetic silencing strategies differ in their effects on inhibitory synaptic transmission. Nat. Neurosci. 2012, 15, 1102–1104. [Google Scholar] [CrossRef] [PubMed]
- Yue, G.Z.; Qiu, Q.; Gao, B.; Cheng, Y.; Zhang, J.; Shimoda, H.; Chang, S.; Lu, J.P.; Zhou, O. Generation of continuous and pulsed diagnostic imaging x-ray radiation using a carbon-nanotube-based field-emission cathode. Appl. Phys. Lett. 2002, 81, 355. [Google Scholar] [CrossRef]
- Radiation Dose in X-Ray and CT Exams. Available online: http://www.radiologyinfo.org/en/safety/?pg=sfty_xray (accessed on 27 October 2014).
- Energy and Work Unit Conversion. Available online: http://www.unit-conversion.info/energy.html (accessed on 10 November 2014).
- Hegemann, P.; Möglich, A. Channelrhodopsin engineering and exploration of new optogenetic tools. Nat. Methods 2011, 8, 39–42. [Google Scholar] [CrossRef] [PubMed]
- Frenzel, J.; Schultes, H. Luminescence in water carrying supersonic waves. Z. Phys. C 1934, 27, 421–424. [Google Scholar]
- Hilgenfeldt, S.; Grossmann, S.; Lohse, D. A simple explanation of light emission in sonoluminescence. Nature 1999, 398, 402–405. [Google Scholar] [CrossRef]
- Zhou, J.; Xing, D.; Chen, Q. Enhancement of Fluoresceinyl Cypridina Luciferin Analog Chemiluminescence by Human Serum Albumin for Singlet Oxygen Detection. Photochem. Photobiol. 2006, 82, 1058. [Google Scholar] [CrossRef] [PubMed]
- Christensen, D.A. Ultrasonic bioinstrumentation; JW Sons: New York, NY, USA, 1988. [Google Scholar]
- Shen, C.; Xu, J.; Fang, N.X.; Jing, Y. Anisotropic Complementary Acoustic Metamaterial for Canceling out Aberrating Layers. Phys. Rev. X 2014, 4, 41033. [Google Scholar]
- Jia, Z.; Valiunas, V.; Lu, Z.; Bien, H.; Liu, H.; Wang, H.-Z.; Rosati, B.; Brink, P.R.; Cohen, I.S.; Entcheva, E. Stimulating Cardiac Muscle by Light Cardiac Optogenetics by Cell Delivery. Circ. Arrhythmia Electrophysiol. 2011, 4, 753–760. [Google Scholar] [CrossRef]
- Berndt, A.; Lee, S. Y.; Ramakrishnan, C.; Deisseroth, K. Structure-Guided Transformation of Channelrhodopsin into a Light-Activated Chloride Channel. Science (80-. ). 2014, 344, 420–424. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, G.; Cheng, Y.; Gao, B.; Qiu, Q.; Lee, Y.Z.; Lu, J.P.; Zhou, O. Stationary scanning x-ray source based on carbon nanotube field emitters. Appl. Phys. Lett. 2005, 86, 184104. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berry, R.; Getzin, M.; Gjesteby, L.; Wang, G. X-Optogenetics and U-Optogenetics: Feasibility and Possibilities. Photonics 2015, 2, 23-39. https://doi.org/10.3390/photonics2010023
Berry R, Getzin M, Gjesteby L, Wang G. X-Optogenetics and U-Optogenetics: Feasibility and Possibilities. Photonics. 2015; 2(1):23-39. https://doi.org/10.3390/photonics2010023
Chicago/Turabian StyleBerry, Rachel, Matthew Getzin, Lars Gjesteby, and Ge Wang. 2015. "X-Optogenetics and U-Optogenetics: Feasibility and Possibilities" Photonics 2, no. 1: 23-39. https://doi.org/10.3390/photonics2010023
APA StyleBerry, R., Getzin, M., Gjesteby, L., & Wang, G. (2015). X-Optogenetics and U-Optogenetics: Feasibility and Possibilities. Photonics, 2(1), 23-39. https://doi.org/10.3390/photonics2010023





