Adsorption Characteristics and Mechanism of Calcium Ions on Different Molybdenite Surfaces via Experiments and DFT Simulations
Abstract
:1. Introduction
2. Materials and Methods
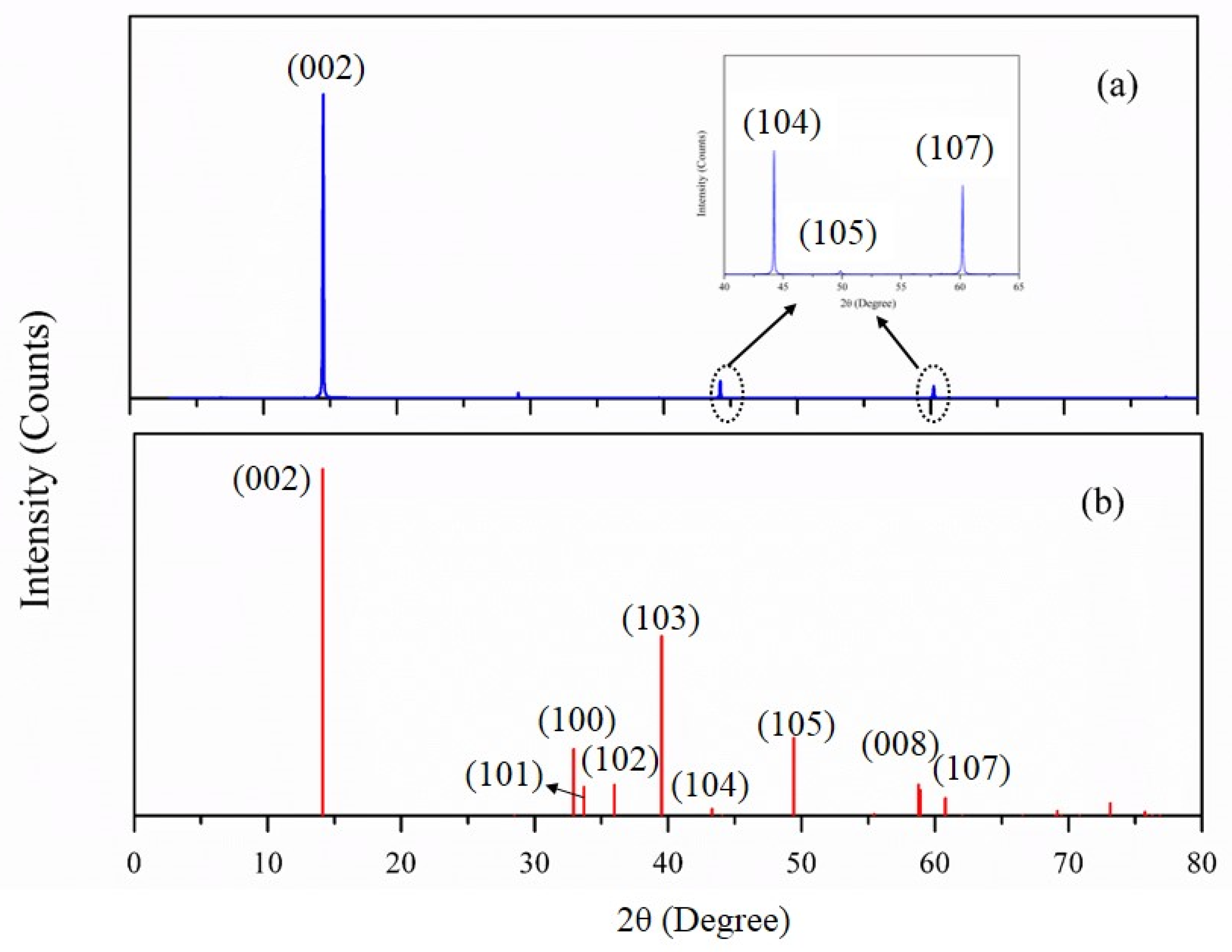
2.1. Materials and Reagents
2.2. Micro-Flotation Tests
2.3. Adsorption Experiments of Calcium Ions on Molybdenite Powders
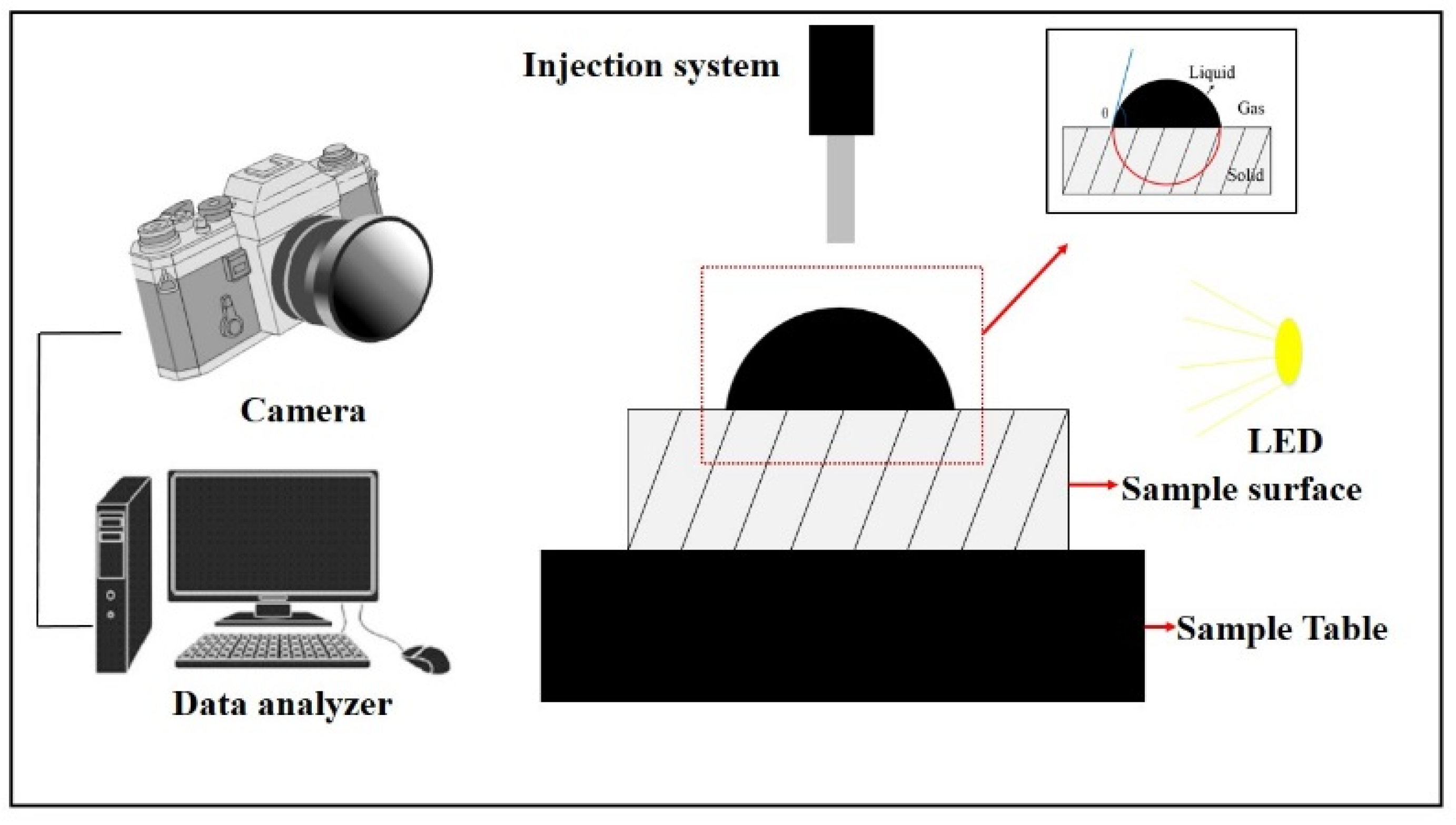
2.4. Contact Angle Measurements
2.5. Computational Methods and Models
3. Results and Discussions
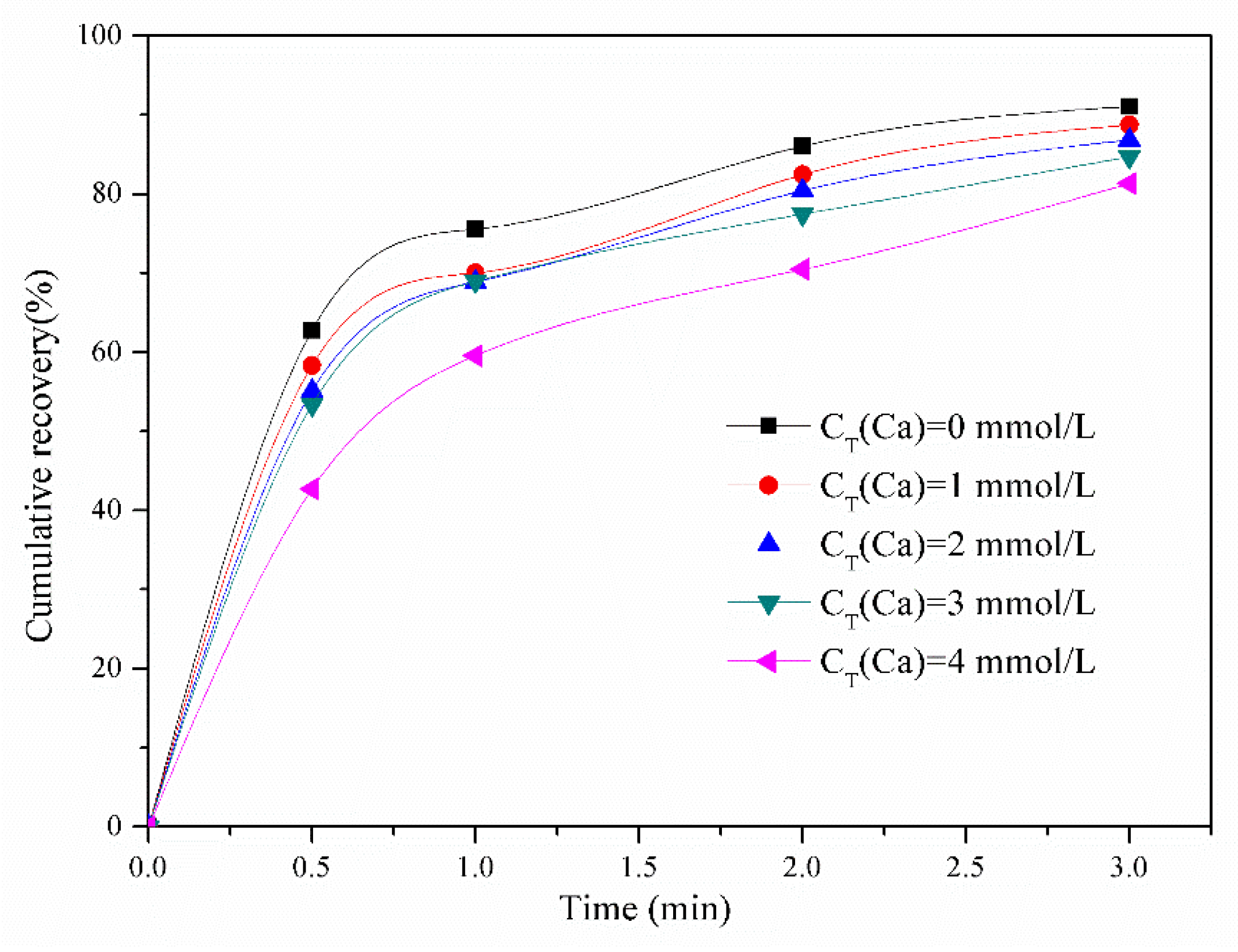
3.1. Effect of Calcium Ions on Molybdenite Flotation Performance
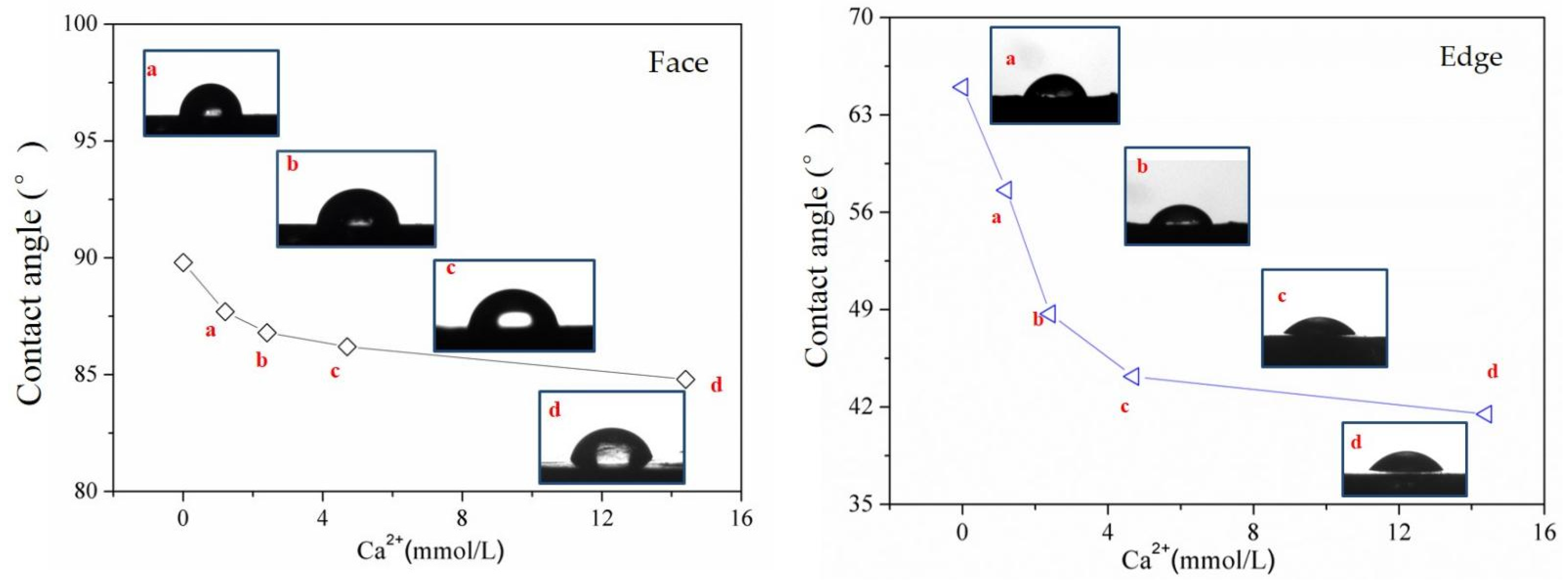
3.2. Wetting Properties of Molybdenite Different Surfaces
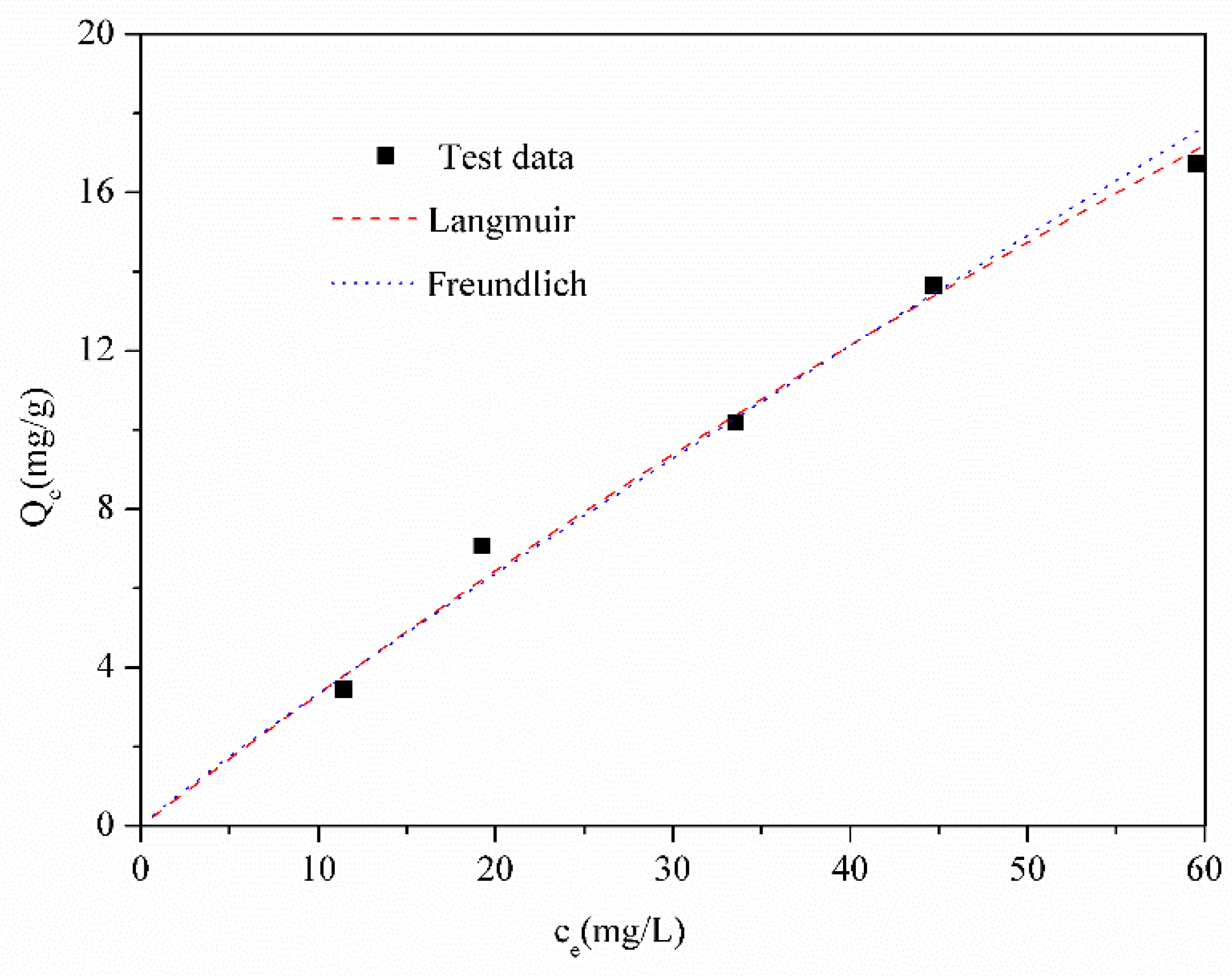
3.3. Batch Adsorption of Calcium Ions on Molybdenite
3.4. Simulation of Calcium Ions on Molybdenite Surfaces Based on DFT
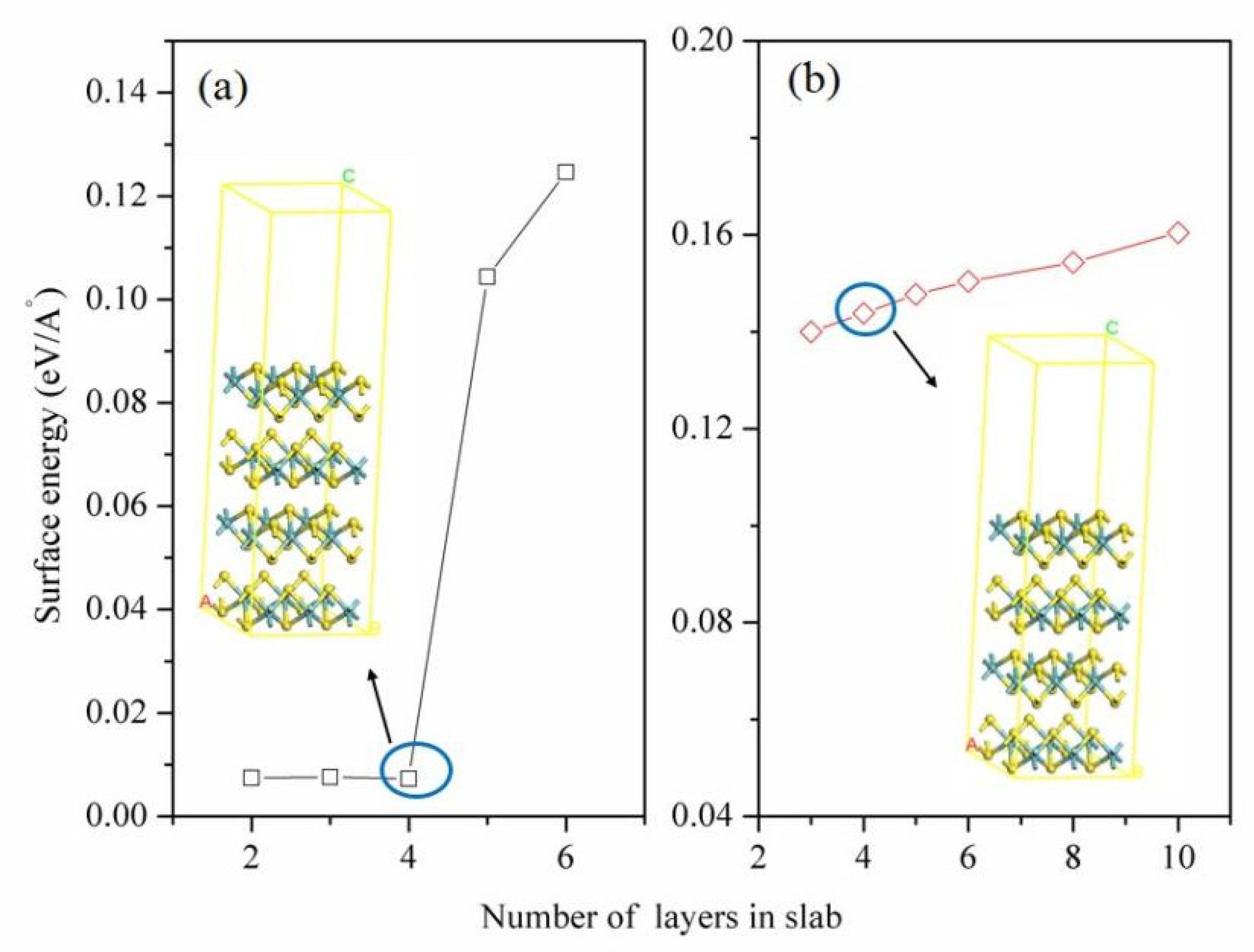
3.4.1. Building the Molybdenite (001) and (100) Surfaces
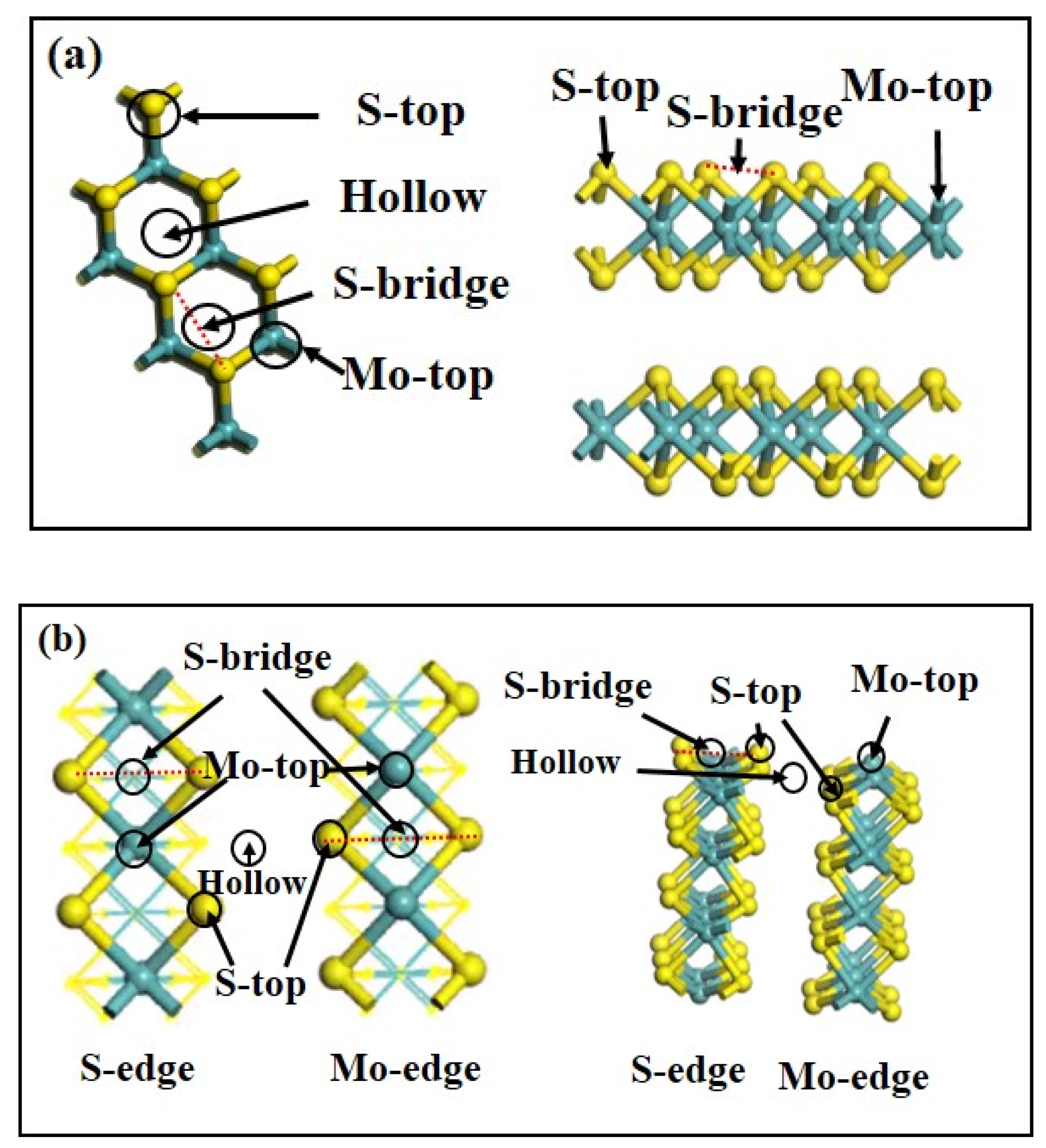
3.4.2. Adsorption Configuration of Calcium Ions on Molybdenite Different Surfaces
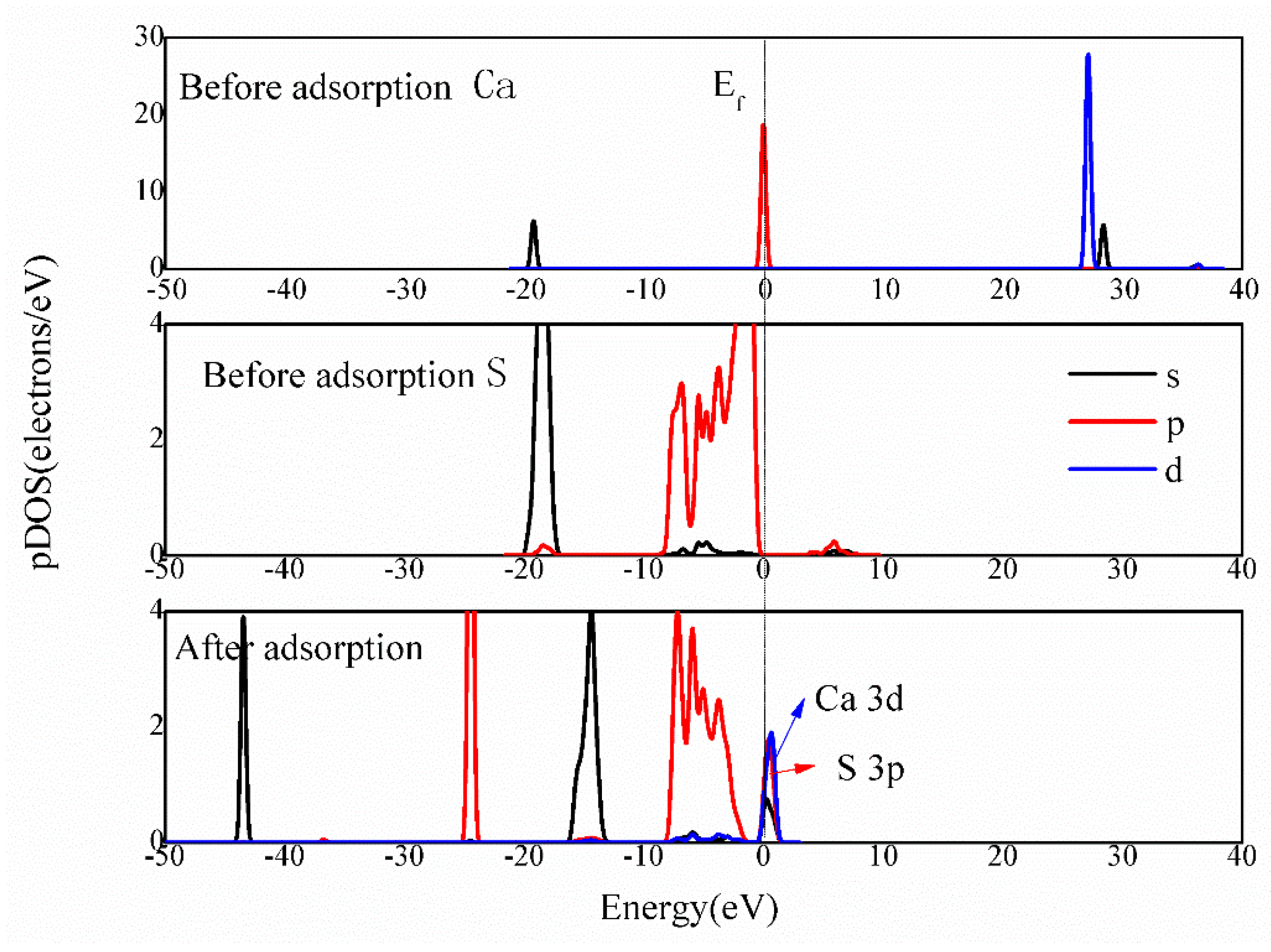
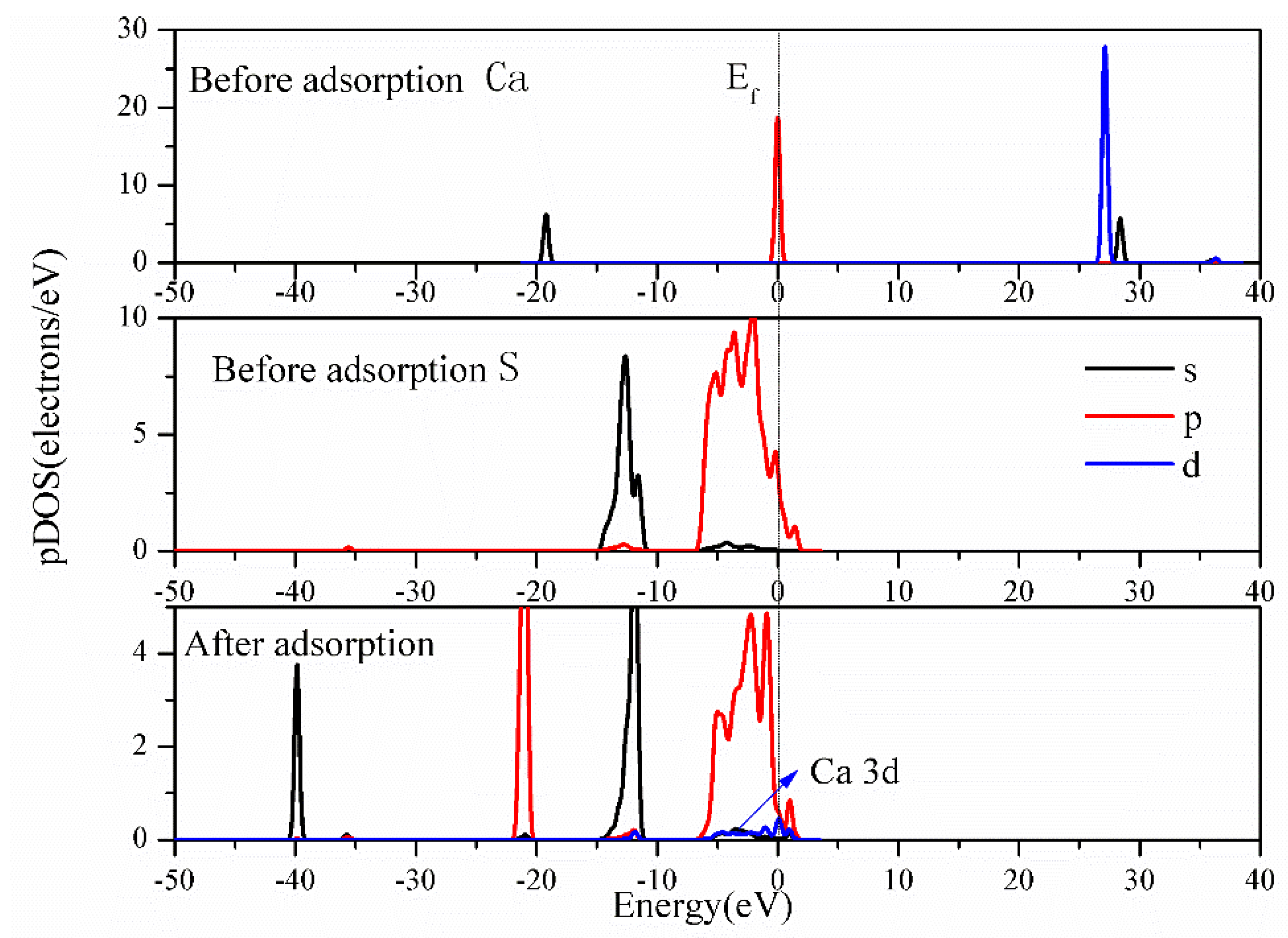
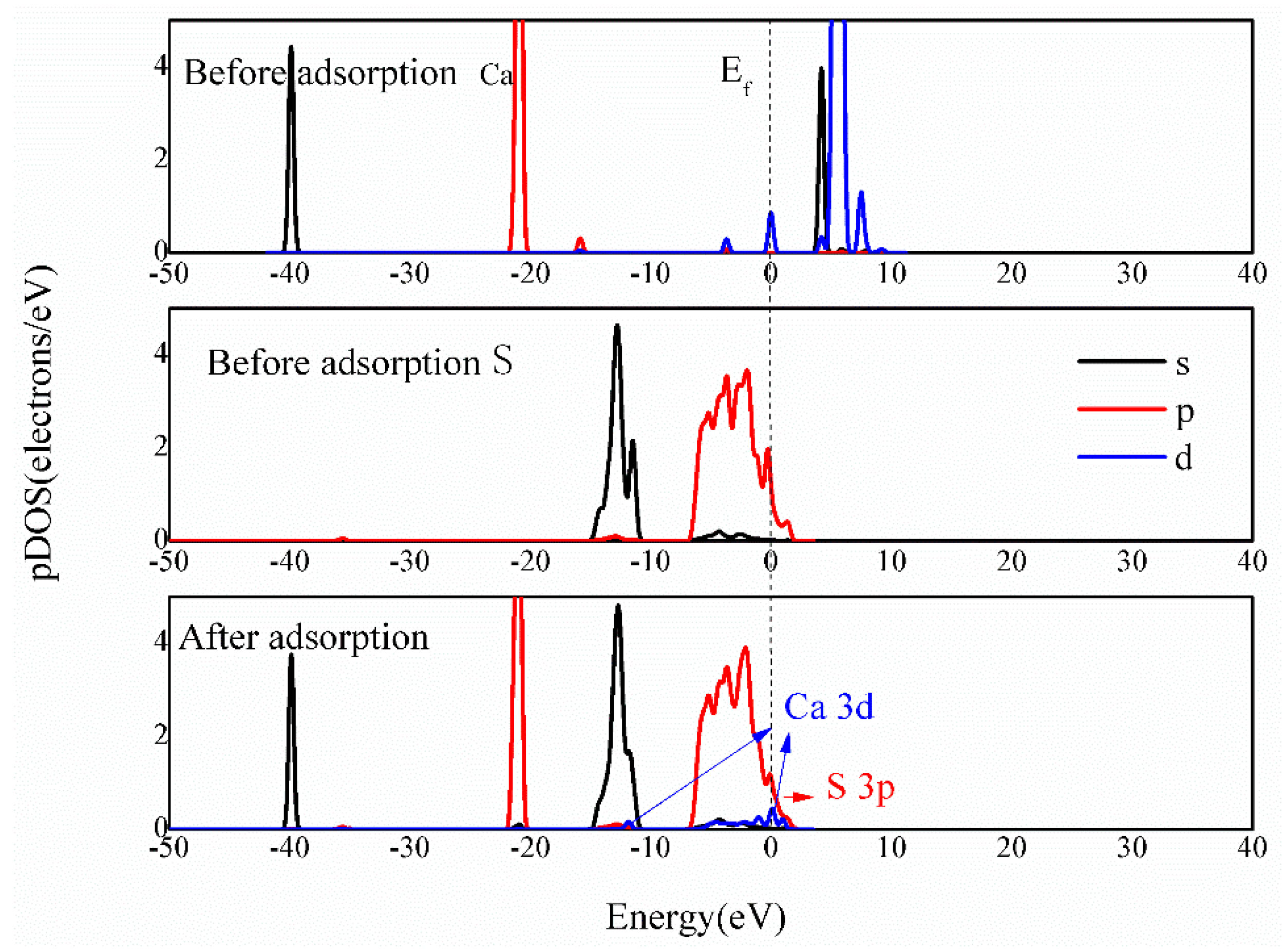
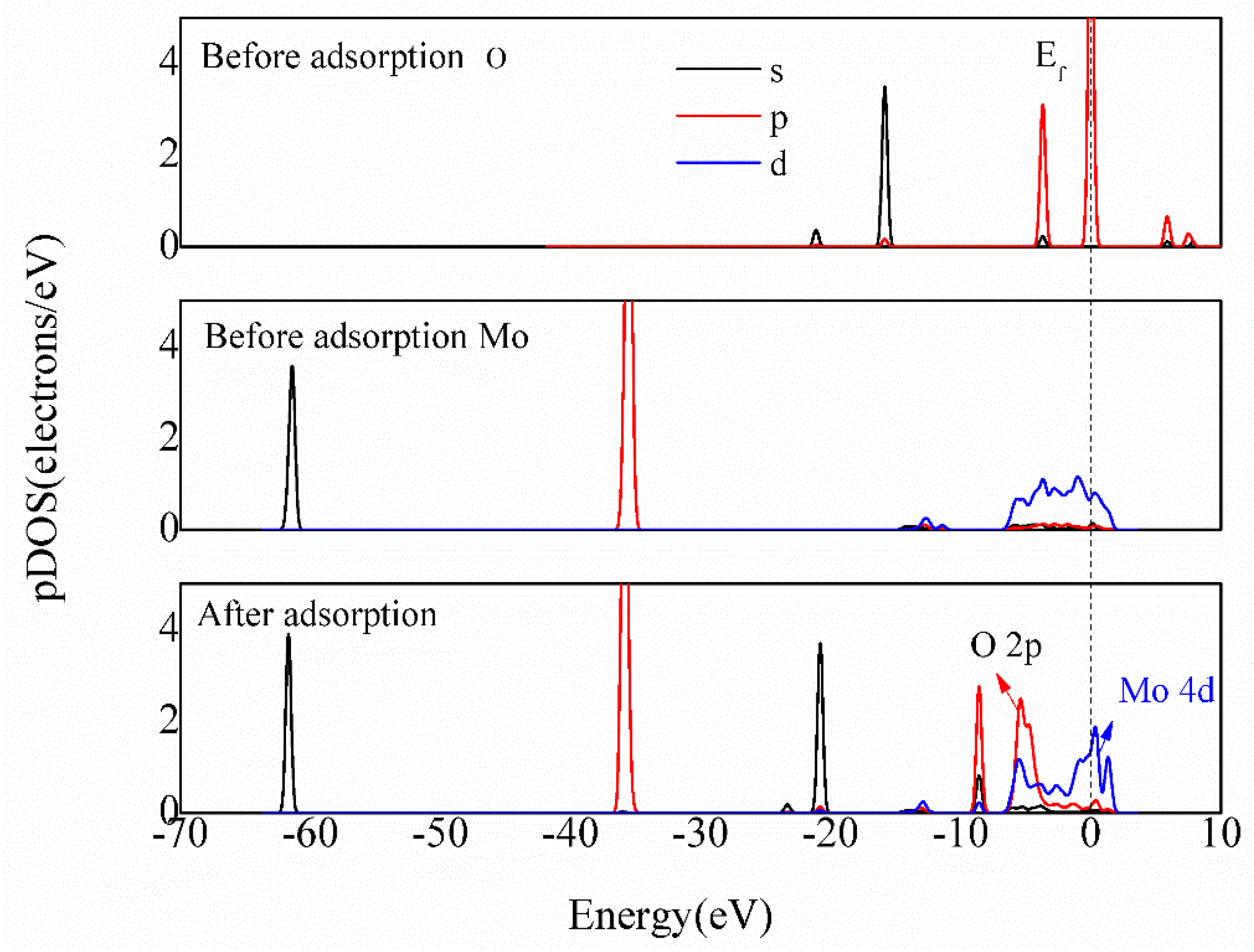
3.4.3. Bonding Mechanism Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, M.; Wei, D.; Liu, Q.; Liu, W.; Zheng, J.; Sun, H. Flotation separation of copper-molybdenum sulfides using chitosan as a selective depressant. Miner. Eng. 2015, 83, 217–222. [Google Scholar] [CrossRef]
- Suyantara, G.P.W.; Hirajima, T.; Elmahdy, A.M.; Miki, H.; Sasaki, K. Effect of kerosene emulsion in MgCl2 solution on the kinetics of bubble interactions with molybdenite and chalcopyrite. Colloids Surf. A 2016, 501, 98–113. [Google Scholar] [CrossRef]
- Ramos, O.; Castro, S.; Laskowski, J. Copper–molybdenum ores flotation in sea water: Floatability and frothability. Miner. Eng. 2013, 53, 108–112. [Google Scholar] [CrossRef]
- Yang, B.; Wang, D.; Wang, T.; Zhang, H.; Jia, F.; Song, S. Effect of Cu2+ and Fe3+ on the depression of molybdenite in flotation. Miner. Eng. 2019, 130, 101–109. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhu, H.; Yang, B.; Jia, F.; Yan, H.; Zeng, M.; Qu, H. Effect of Pb2+ on the flotation of molybdenite in the presence of sulfide ion. Results Phys. 2019, 14, 102361. [Google Scholar] [CrossRef]
- Qiu, Z.; Liu, G.; Liu, Q.; Zhong, H. Understanding the roles of high salinity in inhibiting the molybdenite flotation. Colloids Surf. A 2016, 509, 123–129. [Google Scholar] [CrossRef]
- Hirajima, T.; Suyantara, G.P.W.; Ichikawa, O.; Elmahdy, A.M.; Miki, H.; Sasaki, K. Effect of Mg2+ and Ca2+ as divalent seawater cations on the floatability of molybdenite and chalcopyrite. Miner. Eng. 2016, 96, 83–93. [Google Scholar] [CrossRef]
- Laskowski, J.S.; Castro, S.; Ramos, O. Effect of seawater main components on froth ability in the flotation of Cu-Mo sulfide ore. Physicochem. Probl. Miner. Process. 2014, 50, 17–29. [Google Scholar]
- Suyantara, G.P.W.; Hirajima, T.; Miki, H.; Sasaki, K. Floatability of molybdenite and chalcopyrite in artificial seawater. Miner. Eng. 2018, 115, 117–130. [Google Scholar] [CrossRef]
- Yang, B.; Song, S.; Lopez-Valdivieso, A. Effect of particle size on the contact angle of molybdenite powders. Min. Proc. Ext. Met. Rev. 2014, 35, 208–215. [Google Scholar] [CrossRef]
- Castro, S.C.S.; Lopez-Valdivieso, A.L.A.; Laskowski, J.L.J.S. Review of the flotation of molybdenite. Part I: Surface properties and floatability. Int. J. Miner. Process. 2016, 148, 48–58. [Google Scholar] [CrossRef]
- Yuan, D.; Cadien, K.; Liu, Q.; Zeng, H. Adsorption characteristics and mechanisms of O-Carboxymethyl chitosan on chalcopyrite and molybdenite. J. Colloid Interface Sci. 2019, 552, 659–670. [Google Scholar] [CrossRef] [PubMed]
- Braga, P.F.A.; Chaves, A.P.; Luz, A.B.; França, S.C.A. The use of dextrin in purification by flotation of molybdenite concentrates. Int. J. Miner. Process. 2014, 127, 23–27. [Google Scholar] [CrossRef]
- Lu, Z.; Liu, Q.; Xu, Z.; Zeng, H. Probing anisotropic surface properties of molybdenite by direct force measurements. Langmuir 2015, 31, 11409–11418. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Wang, J.; Huang, J.; Cui, X.; Wang, X.; Liu, Q.; Zhang, H.; Liu, Q.; Zeng, H. Anisotropic polymer adsorption on molybdenite basal and edge surfaces and interaction mechanism with air bubbles. Front. Chem. 2018, 6, 361. [Google Scholar] [CrossRef]
- Wang, X.; Liu, W.; Duan, H.; Wang, B.; Han, C.; Wei, D. The adsorption mechanism of calcium ion on quartz (101) surface: A DFT study. Powder Technol. 2018, 329, 158–166. [Google Scholar] [CrossRef]
- Hu, Y.; He, J.; Zhang, C.; Zhang, C.; Wang, L. Insights into the activation mechanism of calcium ions on the sericite surface: A combined experimental and computational study. Appl. Surf. Sci. 2017, 427, 162–168. [Google Scholar] [CrossRef]
- Feng, Q.; Wen, S.; Deng, J.; Zhao, W. DFT study on the interaction between hydrogen sulfide ions and cerussite (110) surface. Appl. Surf. Sci. 2017, 396, 920–925. [Google Scholar] [CrossRef]
- Kohn, W.; Sham, L.J. Softness and the fukui function in the electronic theory of metals and catalysis. Phys. Rev. A 1965, 140, 1133–1138. [Google Scholar] [CrossRef] [Green Version]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 8, 3865–3868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, C.; Min, F.; Liu, L.; Chen, J. The adsorption of CaOH+ on (001) basal and (010) edge surface of Na-montmorillonite: A DFT study. Surf. Interface Anal. 2017, 49, 267–277. [Google Scholar] [CrossRef]
- Flores-Álvarez, J.M.; Elizondo-Álvarez, M.A.; Dávila-Pulido, G.I.; Uribe-Salas, A. Electrochemical behavior of galena in the presence of calcium and sulfate ions. Miner. Eng. 2017, 111, 158–166. [Google Scholar] [CrossRef]
- Zhu, X.; Wei, H.; Hou, M.; Wang, Q.; You, X.; Li, L. Thermodynamic behavior and flotation kinetics of an ionic liquid microemulsion collector for coal flotation. Fuel 2020, 262, 116627. [Google Scholar] [CrossRef]
- Xing, Y.; Xu, X.; Gui, X.; Cao, Y.; Xu, M. Effect of kaolinite and montmorillonite on fine coal flotation. Fuel 2017, 195, 284–289. [Google Scholar] [CrossRef]
- Zhu, H.; Li, Y.; Lartey, C.; Li, W.; Qian, G. Flotation kinetics of molybdenite in common sulfate salt solutions. Miner. Eng. 2020, 148, 106182. [Google Scholar] [CrossRef]
- Lu, Z.; Ralston, J.; Liu, Q. Face or Edge? Control of molybdenite surface interactions with divalent cations. J. Phys. Chem. C 2019, 124, 372–381. [Google Scholar] [CrossRef]
- Wang, J.; Xie, L.; Lu, Q.; Wang, X.; Wang, J.; Zeng, H. Electrochemical investigation of the interactions of organic and inorganic depressants on basal and edge planes of molybdenite. J. Colloid Interface Sci. 2020, 570, 350–361. [Google Scholar] [CrossRef]
- Zanin, M.; Ametov, I.; Grano, S.; Zhou, L.; Skinner, W. A study of mechanisms affecting molybdenite recovery in a bulk copper/molybdenum flotation circuit. Int. J. Miner. Process. 2011, 93, 256–266. [Google Scholar] [CrossRef]
- Wan, H.; Yang, W.; He, T.; Yang, J.; Guo, L.; Peng, Y. The influence of Ca2+ and pH on the interaction between PAHs and molybdenite edges. Minerals 2017, 7, 104. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; He, C.; Yu, X.; Bai, Y.; Ye, L.; Wang, B.; Zhang, L. Net-like porous activated carbon materials from shrimp shell by solution-processed carbonization and H3PO4 activation for methylene blue adsorption. Powder Technol. 2018, 326, 181–189. [Google Scholar] [CrossRef]
- Meng, M.; Wang, Z.; Ma, L.; Zhang, M.; Wang, J.; Dai, X.; Yan, Y. Selective adsorption of methylparaben by submicrosized molecularly imprinted polymer: Batch and dynamic flow mode studies. Ind. Eng. Chem. Res. 2012, 51, 14915–14924. [Google Scholar] [CrossRef]
- Sarti, E.; Chenet, T.; Stevanin, C.; Costa, V.; Cavazzini, A.; Catani, M.; Martucci, A.; Precisvalle, A.; Beltrami, G.; Pasti, L. High-silica zeolites as sorbent media for adsorption and pre-concentration of pharmaceuticals in aqueous solutions. Molecules 2020, 25, 3331. [Google Scholar] [CrossRef] [PubMed]
- Rajahmundry, G.K.; Garlapati, C.; Kumar, P.S.; Alwi, R.S.; Vo, D.V.N. Statistical Analysis of Adsorption Isotherm Models and its Appropriate Selection. Chemosphere 2021, 276, 130176. [Google Scholar] [CrossRef]
- Hashem, A.; Sanousy, M.A.; Mohamed, L.A.; Okoye, P.U.; Hameed, B.H. Natural and low-cost p.turgidum for efficient adsorption of hg(ii) ions from contaminated solution: Isotherms and kinetics studies. J. Polym. Environ. 2021, 29, 304–312. [Google Scholar] [CrossRef]
- You, X.; He, M.; Cao, X.; Wang, P.; Wang, J.; Li, L. Molecular dynamics simulations of removal of nonylphenol pollutants by graphene oxide: Experimental study and modelling. Appl. Surf. Sci. 2019, 475, 621–626. [Google Scholar] [CrossRef]
- Hussain, A.; Muhammad, T. Effect of Cu concentration and dopant site on the band gap of MoS2: A DFT study. Comput. Condens. Matter 2020, 24, e00494. [Google Scholar] [CrossRef]
- Zhao, C.; Chen, J.; Wu, B.; Long, X. Density functional theory study on natural hydrophobicity of sulfide surfaces. Trans. Nonferrous Met. Soc. China 2014, 24, 491–498. [Google Scholar] [CrossRef]
- Tan, S.M.; Ambrosi, A.; Sofer, Z.K.; Huber, T.; Sedmidubsky, D.; Pumera, M. Pristine basal- and edge-plane-oriented molybdenite MoS2 exhibiting highly anisotropic properties. Chemistry 2015, 21, 7170–7178. [Google Scholar] [CrossRef] [PubMed]
- Hinnemann, B.; Moses, P.G.; Bonde, J.; Joergensen, K.P.; Nielsen, J.H.; Horvh, S.; Chorkendorff, I.; Noerskov, J.K. Biomimetic Hydrogen Evolution: MoS2 Nanoparticles as Catalyst for Hydrogen Evolution. J. Am. Chem. Soc. 2005, 127, 5308–5309. [Google Scholar] [CrossRef]
- Castellà-Ventura, M.; Moissette, A.; Kassab, E. DFT Study of Si/Al Ratio and Confinement Effects on the Energetics and Vibrational Properties of some Aza-Aromatic Molecules Adsorbed on H-ZSM-5 Zeolite. Computation 2020, 8, 81. [Google Scholar] [CrossRef]
- Garcia-Diaz, R.; Romero De La Cruz, M.T.; Ochoa Valiente, R.; Guerrero-Sanchez, J.; Hernández Cocoletzi, G. DFT study for OH radical formation on SrTiO3(001) surface and the effect of Bi. Appl. Surf. Sci. 2019, 487, 1394. [Google Scholar] [CrossRef]

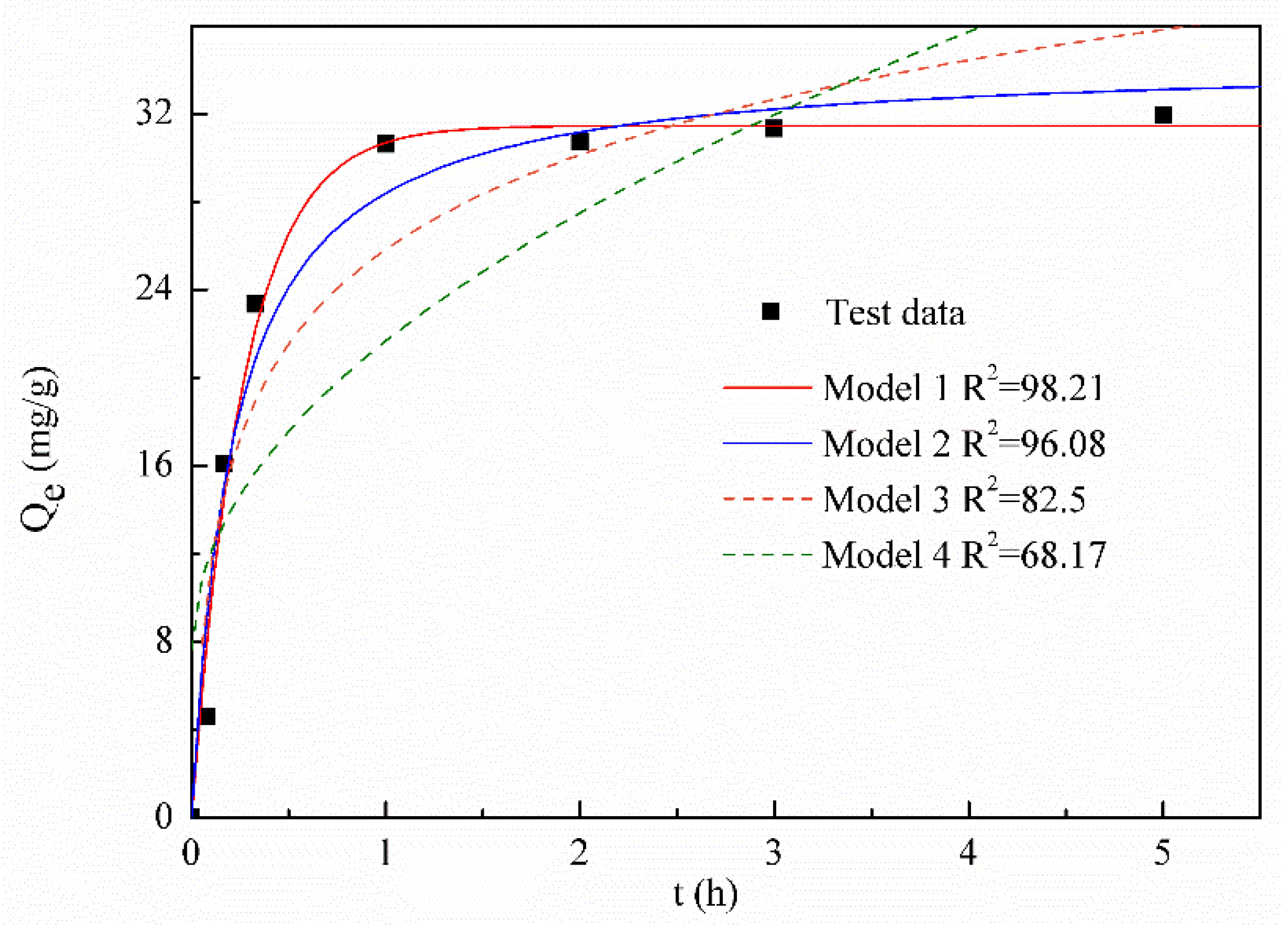

| Model | Formula | Notes |
|---|---|---|
| Pseudo-first-order (Model 1) | Widely used for describing adsorption process in liquid phase. | |
| Pseudo-second-order (Model 2) | Assume the adsorption is chemically affected. | |
| Elovich (Model 3) | Always used in adsorption study for mineral and soil. | |
| Intraparticle diffusion (Model 4) | Often used to analyze the rate-controlling process in the adsorption. |
| Langmuir | Freundlich | ||
|---|---|---|---|
| Qm | 102.46 | KF | 0.3952 |
| KL | 0.0034 | n | 1.0772 |
| R2 | 96.27 | R2 | 97.31 |
| Ionic Form | Surface | Adsorption Site | Adsorption Energy (kJ/mol) |
|---|---|---|---|
| Ca2+ | (001) | S-top | −123 |
| Mo-top | −229 | ||
| Hollow | −195 | ||
| S-bridge | −201 | ||
| (100) | S-(100)- Mo-top | −935 | |
| S-(100)- S-bridge | −905 | ||
| S-(100)- S-top | −927 | ||
| H | −835 | ||
| Mo-(100)- S-top | −890 | ||
| Mo-(100)- Mo-top | −770 | ||
| Mo-(100)- S-bridge | −917 | ||
| CaOH+ | (001) | S-top | 10 |
| Mo-top | −17 | ||
| Hollow | −7 | ||
| S-bridge | −7 | ||
| (100) | S-(100)- Mo-top | −917 | |
| S-(100)- S-bridge | −931 | ||
| S-(100)- S-top | −909 | ||
| H | −931 | ||
| Mo-(100)- S-top | −978 | ||
| Mo-(100)- Mo-top | −770 | ||
| Mo-(100)- Sbridge | −850 |
| Ionic Form-Surface | Adsorption Site | Bond | Populaiton | Bond Length(Å) |
|---|---|---|---|---|
| Ca2+-(001) | Mo-top | S1-Ca | 0.05 | 2.95 |
| S2-Ca | 0.10 | 2.73 | ||
| S3-Ca | 0.10 | 2.80 | ||
| Ca2+-(100) | S-(100)- Mo-top | S1-Ca | 0.09 | 2.54 |
| S2-Ca | 0.09 | 2.62 | ||
| S3-Ca | 0.08 | 2.42 | ||
| S4-Ca | 0.07 | 2.52 | ||
| CaOH+-(100) | Mo-(100)- S-top | S-Ca | 0.07 | 2.65 |
| O- Mo | 0.34 | 2.03 |
| Ionic -Surface | Adsorption Site | Atom | Hirshfeld Charge (e) | ||
|---|---|---|---|---|---|
| Before | After | Change | |||
| Ca2+-(001) | Mo-top | S1 | −0.11 | −0.14 | −0.03 |
| S2 | −0.11 | −0.13 | −0.02 | ||
| S3 | −0.11 | −0.14 | −0.03 | ||
| Ca | 1.99 | 0.71 | −1.28 | ||
| Mo | 0.22 | 0.24 | 0.02 | ||
| Ca2+-(100) | S-(100)- Mo-top | S1 | −0.18 | −0.14 | 0.04 |
| S2 | −0.18 | −0.14 | 0.04 | ||
| S3 | −0.18 | −0.15 | 0.03 | ||
| S4 | −0.18 | −0.14 | 0.04 | ||
| Ca | 1.99 | 0.26 | −1.73 | ||
| Mo | 0.28 | 0.32 | 0.04 | ||
| CaOH+-(100) | Mo-(100)- S-top | S | −0.12 | −0.10 | 0.02 |
| Ca | 1.19 | 0.59 | −0.60 | ||
| O | −0.44 | −0.33 | 0.11 | ||
| Mo | 0.24 | 0.36 | 0.12 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, L.; Cao, Y.; Li, L.; Zeng, Q. Adsorption Characteristics and Mechanism of Calcium Ions on Different Molybdenite Surfaces via Experiments and DFT Simulations. Separations 2021, 8, 107. https://doi.org/10.3390/separations8080107
Sun L, Cao Y, Li L, Zeng Q. Adsorption Characteristics and Mechanism of Calcium Ions on Different Molybdenite Surfaces via Experiments and DFT Simulations. Separations. 2021; 8(8):107. https://doi.org/10.3390/separations8080107
Chicago/Turabian StyleSun, Liqing, Yijun Cao, Lin Li, and Qingliang Zeng. 2021. "Adsorption Characteristics and Mechanism of Calcium Ions on Different Molybdenite Surfaces via Experiments and DFT Simulations" Separations 8, no. 8: 107. https://doi.org/10.3390/separations8080107




