Elevated Bioactivity of Ruta graveolens against Cancer Cells and Microbes Using Seaweeds
Abstract
1. Introduction
2. Results
2.1. Morphological Responses to SWE
2.2. Gas Exchange and SWE Mixture
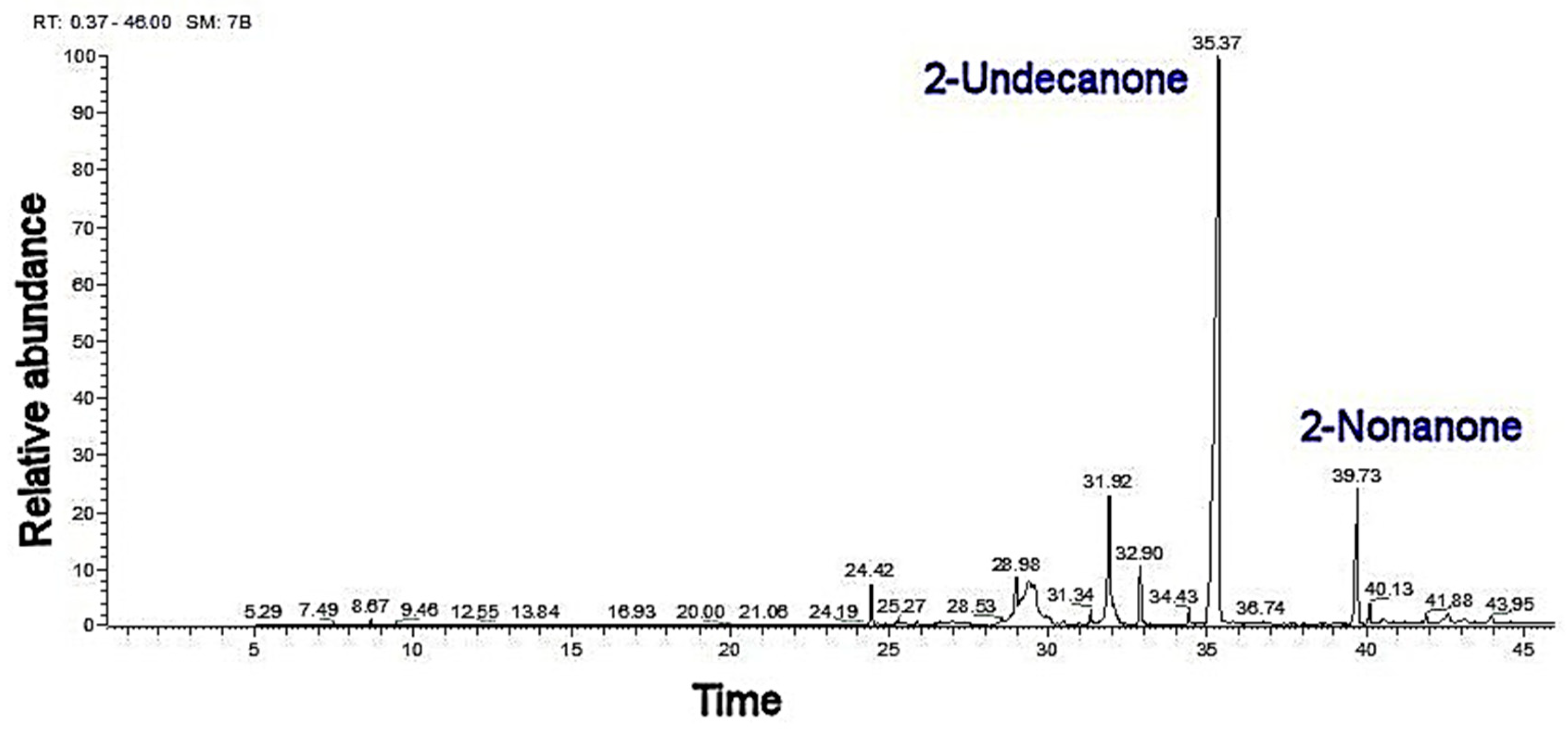
2.3. Essential Oil and SWE Mixture
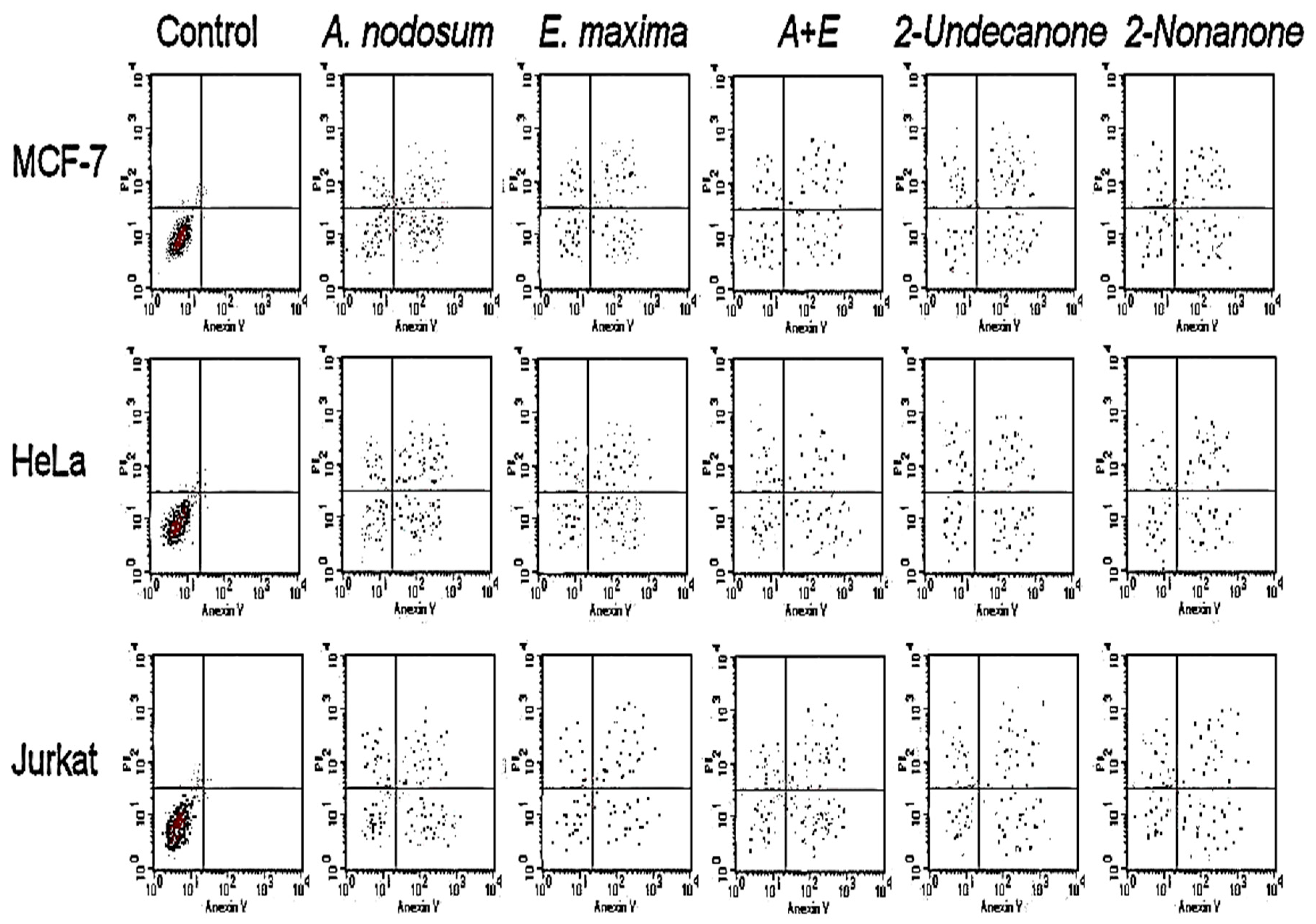
2.4. Antiproliferative Activities and Apoptotic Assay
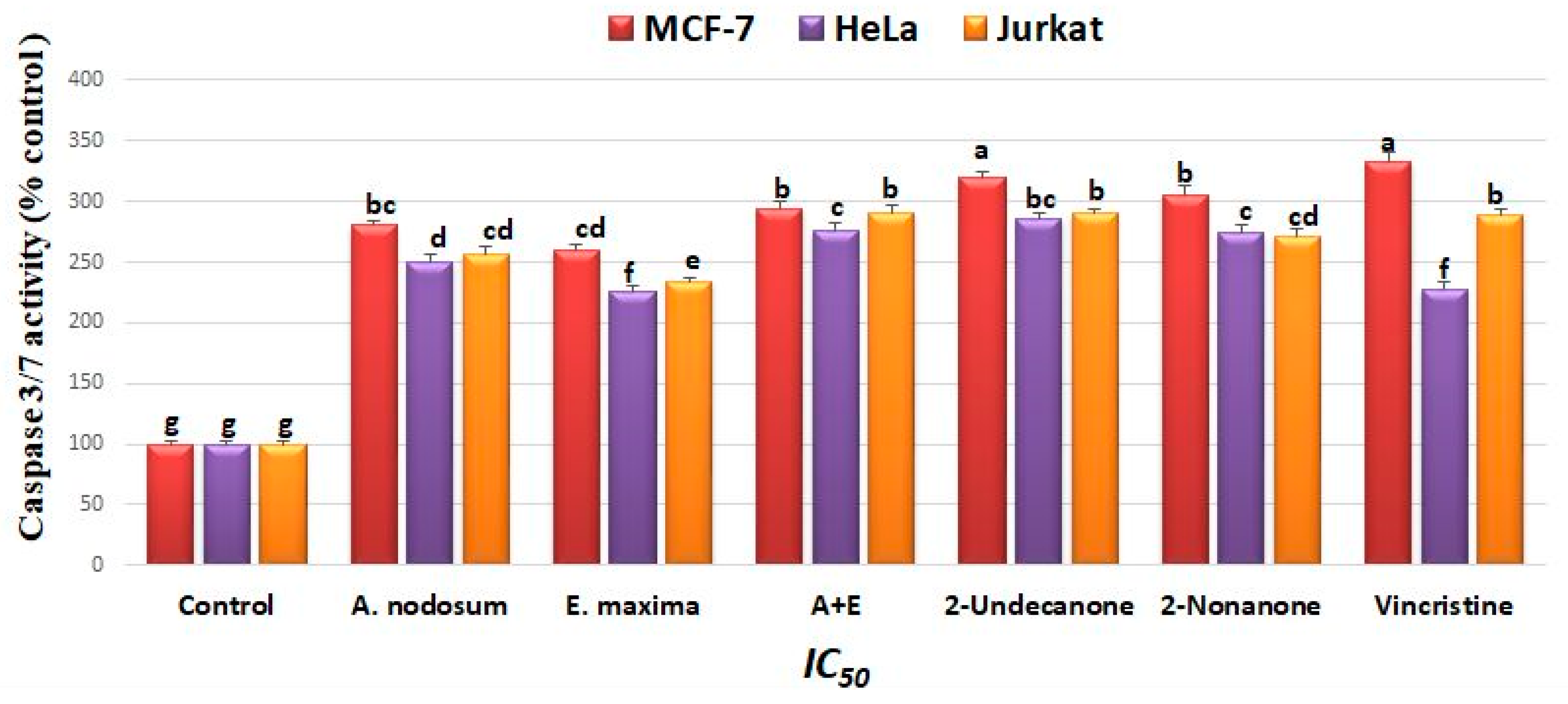
2.5. Caspase-3/7 Activity Study
2.6. EO and Antibacterial Activities
2.7. EO and Antifungal Activities
3. Discussion
4. Materials and Methods
4.1. Plant Material, Treatments, and Microorganisms
4.2. Measurements
4.3. Essential Oil Isolation, Gas Chromatography/Mass Spectrometry (GC-MS), and Analyses of Seaweeds
4.4. Antiproliferative Activity and Apoptotic Cell Population
4.5. Caspase-Glo 3/7 Assay
4.6. Antibacterial Activities
4.7. Antifungal Activities
4.8. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ueng, Y.-F.; Chen, C.-C.; Huang, Y.-L.; Lee, I.J.; Yun, C.-H.; Chen, Y.-H.; Huang, C.-C. Effects of aqueous extract of Ruta graveolens and its ingredients on cytochrome P450, uridine diphosphate (UDP)-glucuronosyltransferase, and reduced nicotinamide adenine dinucleotide (phosphate) (NAD(P)H)-quinone oxidoreductase in mice. J. Food Drug Anal. 2015, 23, 516–528. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, F.G.E.; Mendes, F.R.D.S.; Assunção, J.C.D.C.; Maria Pinheiro Santiago, G.; Aislania Xavier Bezerra, M.; Barbosa, F.G.; Mafezoli, J.; Rodrigues Rocha, R. Seasonal variation, larvicidal and nematicidal activities of the lef essential oil of Ruta graveolens L. J. Essent. Oil Res. 2014, 26, 204–209. [Google Scholar] [CrossRef]
- Pollio, A.; De Natale, A.; Appetiti, E.; Aliotta, G.; Touwaide, A. Continuity and change in the Mediterranean medical tradition: Ruta spp. (rutaceae) in Hippocratic medicine and present practices. J. Ethnopharmacol. 2008, 116, 469–482. [Google Scholar] [CrossRef] [PubMed]
- Dob, T.; Dahmane, D.; Gauriat-Desrdy, B.; Daligault, V. Volatile Constituents of the Essential Oil of Ruta chalepensis L. subsp. Angustifolia (Pers.) P. Cout. J. Essent. Oil Res. 2008, 20, 306–309. [Google Scholar] [CrossRef]
- Mejri, J.; Abderrabba, M.; Mejri, M. Chemical composition of the essential oil of Ruta chalepensis L: Influence of drying, hydro-distillation duration and plant parts. Ind. Crop. Prod. 2010, 32, 671–673. [Google Scholar] [CrossRef]
- Amdouni, T.; Ben Abdallah, S.; Msilini, N.; Merck, F.; Chebbi, M.; Lachaal, M.; Karray-Bouraoui, N.; Ouerghi, Z.; Fernandez, X. Effect of salt stress on the antimicrobial activity of Ruta chalepensis essential oils. Acta Physiol. Plant. 2016, 38, 147. [Google Scholar] [CrossRef]
- Reddy, D.N.; Al-Rajab, A.J. Chemical composition, antibacterial and antifungal activities of Ruta graveolens L. volatile oils. Cogent Chem. 2016, 2, 1220055. [Google Scholar] [CrossRef]
- Yaacob, K.B.; Abdullah, C.M.; Joulain, D. Essential Oil of Ruta graveolens L. J. Essent. Oil Res. 1989, 1, 203–207. [Google Scholar] [CrossRef]
- Al-Ghamdi, A.A.; Elansary, H.O. Synergetic effects of 5-aminolevulinic acid and Ascophyllum nodosum seaweed extracts on Asparagus phenolics and stress related genes under saline irrigation. Plant Physiol. Bioch. 2018, 129, 273–284. [Google Scholar] [CrossRef]
- Elansary, H.O. Green roof Petunia, Ageratum, and Mentha responses to water stress, seaweeds, and trinexapac-ethyl treatments. Acta Physiol. Plant. 2017, 39, 145. [Google Scholar] [CrossRef]
- Elansary, H.O.; Yessoufou, K.; Shokralla, S.; Mahmoud, E.A.; Skaicka-Wozniak, K. Enhancing mint and basil oil composition and antibacterial activity using seaweed extracts. Ind. Crops Prod. 2016, 92, 50–56. [Google Scholar] [CrossRef]
- Cheong, K.-L.; Qiu, H.-M.; Du, H.; Liu, Y.; Khan, M.B. Oligosaccharides Derived from Red Seaweed: Production, Properties, and Potential Health and Cosmetic Applications. Molecules 2018, 23, 2451. [Google Scholar] [CrossRef] [PubMed]
- Meng, S.; Wang, R.; Zhang, M.; Meng, X.; Liu, H.; Wang, L. Insights into the Fouling Propensities of Natural Derived Alginate Blocks during the Microfiltration Process. Processes 2019, 7, 858. [Google Scholar] [CrossRef]
- Elansary, H.O.; Skalicka-Woźniake, K.; King, I.W. Enhancing stress growth traits as well as phytochemical and antioxidant contents of Spiraea and Pittosporum under seaweed extract treatments. Plant Physiol. Biochem. 2016, 105, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Elansary, H.O.; Yessoufou, K.; Abdel-Hamid, A.M.E.; El-Esawi, M.A.; Ali, H.M.; Elshikh, M.S. Seaweed Extracts Enhance Salam Turfgrass Performance during Prolonged Irrigation Intervals and Saline Shock. Front. Plant Sci. 2017, 8, 830. [Google Scholar] [CrossRef] [PubMed]
- Ertani, A.; Francioso, O.; Tinti, A.; Schiavon, M.; Pizzeghello, D.; Nardi, S. Evaluation of Seaweed Extracts from Laminaria and Ascophyllum nodosum spp. as Biostimulants in Zea mays L. Using a Combination of Chemical, Biochemical and Morphological Approaches. Front. Plant Sci. 2018, 9, 428. [Google Scholar] [CrossRef] [PubMed]
- Kocira, S.; Szparaga, A.; Kuboń, M.; Czerwińska, E.; Piskier, T. Morphological and Biochemical Responses of Glycine max (L.) Merr. to the Use of Seaweed Extract. Agronomy 2019, 9, 93. [Google Scholar] [CrossRef]
- Rouphael, Y.; Giordano, M.; Cardarelli, M.; Cozzolino, E.; Mori, M.; Kyriacou, C.M.; Bonini, P.; Colla, G. Plant- and Seaweed-Based Extracts Increase Yield but Differentially Modulate Nutritional Quality of Greenhouse Spinach through Biostimulant Action. Agronomy 2018, 8, 126. [Google Scholar] [CrossRef]
- Zaorsky, N.G.; Churilla, T.M.; Egleston, B.L.; Fisher, S.G.; Ridge, J.A.; Horwitz, E.M.; Meyer, J.E. Causes of death among cancer patients. Ann. Oncol. 2017, 28, 400–407. [Google Scholar] [CrossRef]
- Sharma, K.; Mahato, N.; Lee, Y.R. Systematic study on active compounds as antibacterial and antibiofilm agent in aging onions. J. Food Drug Anal. 2018, 26, 518–528. [Google Scholar] [CrossRef]
- Wang, S.; Zheng, Y.; Xiang, F.; Li, S.; Yang, G. Antifungal activity of Momordica charantia seed extracts toward the pathogenic fungus Fusarium solani L. J. Food Drug Anal. 2016, 24, 881–887. [Google Scholar] [CrossRef] [PubMed]
- Van Duin, D.; Paterson, D.L. Multidrug-Resistant Bacteria in the Community: Trends and Lessons Learned. Infect. Dis. Clin. 2016, 30, 377–390. [Google Scholar] [CrossRef] [PubMed]
- Arioli, T.; Mattner, S.W.; Winberg, P.C. Applications of seaweed extracts in Australian agriculture: Past, present and future. J. Appl. Phycol. 2015, 27, 2007–2015. [Google Scholar] [CrossRef] [PubMed]
- Dias, C.S.; Araujo, L.; Alves Chaves, J.A.; DaMatta, F.M.; Rodrigues, F.A. Water relation, leaf gas exchange and chlorophyll a fluorescence imaging of soybean leaves infected with Colletotrichum truncatum. Plant Physiol. Biochem. 2018, 127, 119–128. [Google Scholar] [CrossRef]
- Blunden, G.; Jenkins, T.; Liu, Y.-W. Enhanced leaf chlorophyll levels in plants treated with seaweed extract. J. Appl. Phycol. 1996, 8, 535–543. [Google Scholar] [CrossRef]
- Whapham, C.A.; Blunden, G.; Jenkins, T.; Hankins, S.D. Significance of Betaines in the Increased Chlorophyll Content of Plants Treated with Seaweed Extract. J. Appl. Phycol. 1993, 5, 231–234. [Google Scholar] [CrossRef]
- Attia, E.Z.; Abd El-Baky, R.M.; Desoukey, S.Y.; El Hakeem Mohamed, M.A.; Bishr, M.M.; Kamel, M.S. Chemical composition and antimicrobial activities of essential oils of Ruta graveolens plants treated with salicylic acid under drought stress conditions. Future J. Pharm. Sci. 2018, 4, 254–264. [Google Scholar] [CrossRef]
- Fadlalla, K.; Watson, A.; Yehualaeshet, T.; Turner, T.; Samuel, T. Ruta graveolens extract induces DNA damage pathways and blocks Akt activation to inhibit cancer cell proliferation and survival. Anticancer Res. 2011, 31, 233–241. [Google Scholar]
- Schelz, Z.; Ocsovszki, I.; Bozsity, N.; Hohmann, J.; Zupko, I. Antiproliferative Effects of Various Furanoacridones Isolated from Ruta graveolens on Human Breast Cancer Cell Lines. Anticancer Res. 2016, 36, 2751–2758. [Google Scholar]
- Alvarado-Sansininea, J.J.; Sanchez-Sanchez, L.; Lopez-Munoz, H.; Escobar, M.L.; Flores-Guzman, F.; Tavera-Hernandez, R.; Jimenez-Estrada, M. Quercetagetin and Patuletin: Antiproliferative, Necrotic and Apoptotic Activity in Tumor Cell Lines. Molecules 2018, 23, 2579. [Google Scholar] [CrossRef]
- Elansary, H.O.; Szopa, A.; Kubica, P.; Al-Mana, F.A.; Mahmoud, E.A.; El-Abedin, T.K.A.Z.; Mattar, M.A.; Ekiert, H. Phenolic Compounds of Catalpa speciosa, Taxus cuspidata, and Magnolia acuminata have Antioxidant and Anticancer Activity. Molecules 2019, 24, 412. [Google Scholar] [CrossRef] [PubMed]
- Bell, R.A.V.; Megeney, L.A. Evolution of caspase-mediated cell death and differentiation: Twins separated at birth. Cell Death Differ. 2017, 24, 1359–1368. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.S.; Joshi, N.; Padalia, R.C.; Singh, V.R.; Goswami, P.; Kumar, A.; Iqbal, H.; Verma, R.K.; Chanda, D.; Chauhan, A.; et al. Chemical Composition and Allelopathic, Antibacterial, Antifungal, and Antiacetylcholinesterase Activity of Fish-mint (Houttuynia cordataThunb.) from India. Chem. Biodivers. 2017, 14, e1700189. [Google Scholar] [CrossRef] [PubMed]
- Viljoen, A.M.; Moolia, A.; van Vuuren, S.F.; van Zyl, R.L.; Başer, K.H.C.; Demirci, B.; Özek, T.; Trinder-Smith, T.H. The Biological Activity and Essential Oil Composition of 17 Agathosma (Rutaceae) Species. J. Essent. Oil Res. 2006, 18 (Suppl. 1), 2–16. [Google Scholar] [CrossRef]
- França Orlanda, J.F.; Nascimento, A.R. Chemical composition and antibacterial activity of Ruta graveolens L. (Rutaceae) volatile oils, from São Luís, Maranhão, Brazil. S. Afr. J. Bot. 2015, 99, 103–106. [Google Scholar] [CrossRef]
- El-Esawi, M.A.; Elansary, H.O.; El-Shanhorey, N.A.; Abdel-Hamid, A.M.E.; Ali, H.M.; Elshikh, M.S. Salicylic Acid-Regulated Antioxidant Mechanisms and Gene Expression Enhance Rosemary Performance under Saline Conditions. Front. Physiol. 2017, 8, 716. [Google Scholar] [CrossRef]
- Elansary, H.O.; Ashmawy, N.A. Essential Oils of Mint between Benefits and Hazards. J. Essent. Oil Bear Plants 2013, 16, 429–438. [Google Scholar] [CrossRef]
- Elansary, H.O.; Mahmoud, E.A. Basil cultivar identification using chemotyping still favored over genotyping using core barcodes and possible resources of antioxidants. J. Essent. Oil Res. 2015, 27, 82–87. [Google Scholar] [CrossRef]
- Elansary, H.O.; Norrie, J.; Ali, H.M.; Salem, M.Z.M.; Mahmoud, E.A.; Yessoufou, K. Enhancement of Calibrachoa growth, secondary metabolites and bioactivity using seaweed extracts. BMC Complement. Altern. Med. 2016, 16, 341. [Google Scholar] [CrossRef]
- Parry, J.; Su, L.; Moore, J.; Cheng, Z.; Luther, M.; Rao, J.N.; Wang, J.Y.; Yu, L.L. Chemical compositions, antioxidant capacities, and antiproliferative activities of selected fruit seed flours. J. Agric. Food Chem. 2006, 54, 3773–3778. [Google Scholar] [CrossRef]
- Elansary, H.O.; Szopa, A.; Kubica, P.; Ekiert, H.; Mattar, M.A.; Al-Yafrasi, A.M.; El-Ansary, O.D.; Zin El-Abedin, K.T.; Yessoufou, K. Polyphenol Profile and Pharmaceutical Potential of Quercus spp. Bark Extracts. Plants 2019, 8, 486. [Google Scholar] [CrossRef] [PubMed]
- Elansary, H.O.; Salem, M.Z.M.; Ashmawy, N.A.; Yessoufou, K.; El-Settawy, A.A.A. In vitro antibacterial, antifungal and antioxidant activities of Eucalyptus spp. leaf extracts related to phenolic composition. Nat. Prod. Res. 2017, 31, 2927–2930. [Google Scholar] [CrossRef] [PubMed]
- Elansary, H.O.; Szopa, A.; Kubica, P.; Ekiert, H.; Ali, H.M.; Elshikh, M.S.; Abdel-Salam, E.M.; El-Esawi, M.; El-Ansary, D.O. Bioactivities of Traditional Medicinal Plants in Alexandria. Evid. Based Complement. Alternat. Med. 2018, 2018, 1463579. [Google Scholar] [CrossRef] [PubMed]
- Elansary, H.O.; Mahmoud, A.E.; El-Ansary, D.O.; Mattar, M.A. Effects of Water Stress and Modern Biostimulants on Growth and Quality Characteristics of Mint. Agronomy 2019, 10, 6. [Google Scholar] [CrossRef]
- Elansary, H.O.; Szopa , A.; Klimek-Szczykutowicz, M.; Jafernik, K.; Ekiert, H.; Mahmoud, E.A.; Barakat, A.A.; El-Ansary, D.O. Mammillaria Species—Polyphenols Studies and Anti-Cancer, Anti-Oxidant, and Anti-Bacterial Activities. Molecules 2019, 25, 131. [Google Scholar] [CrossRef]
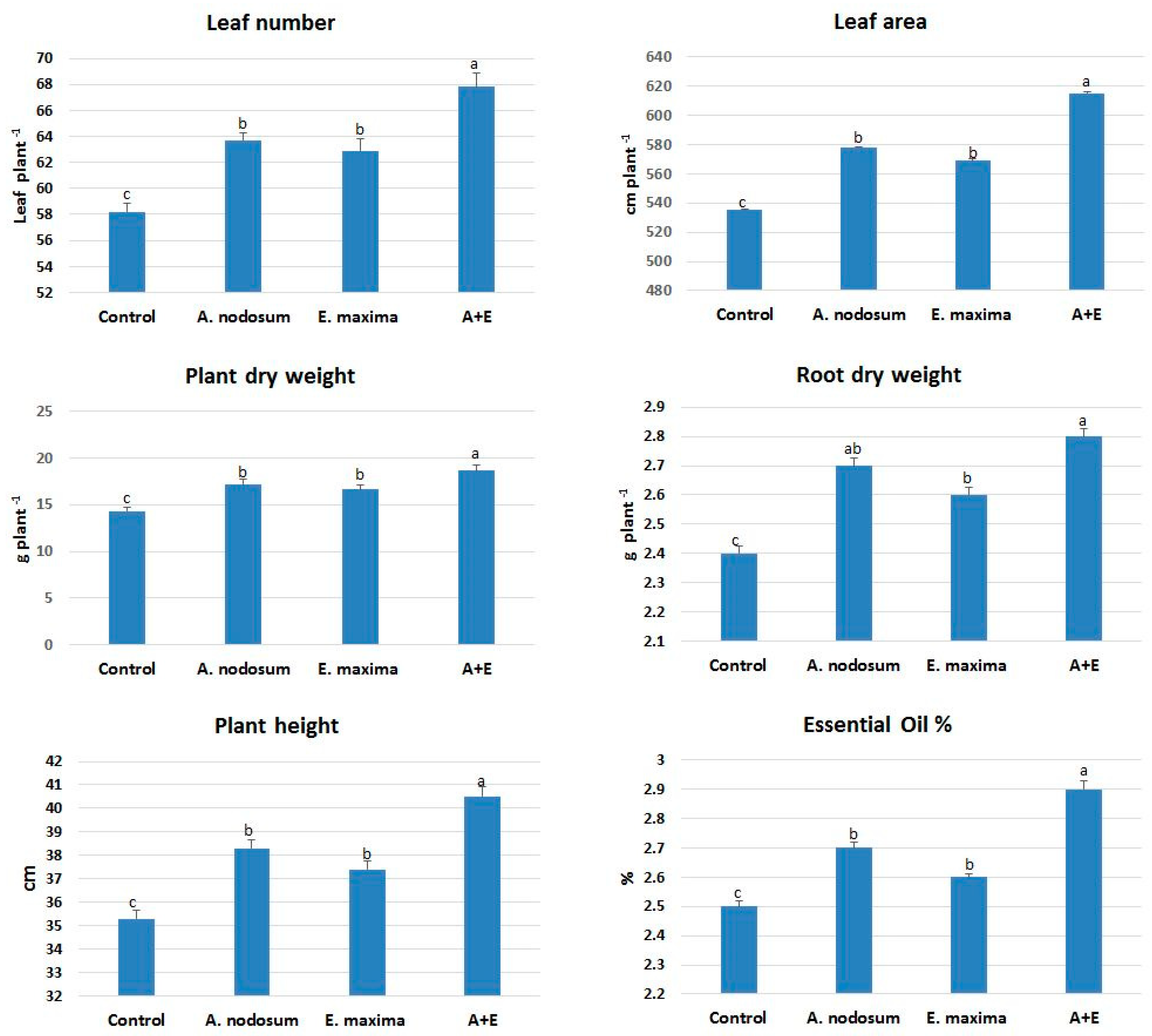
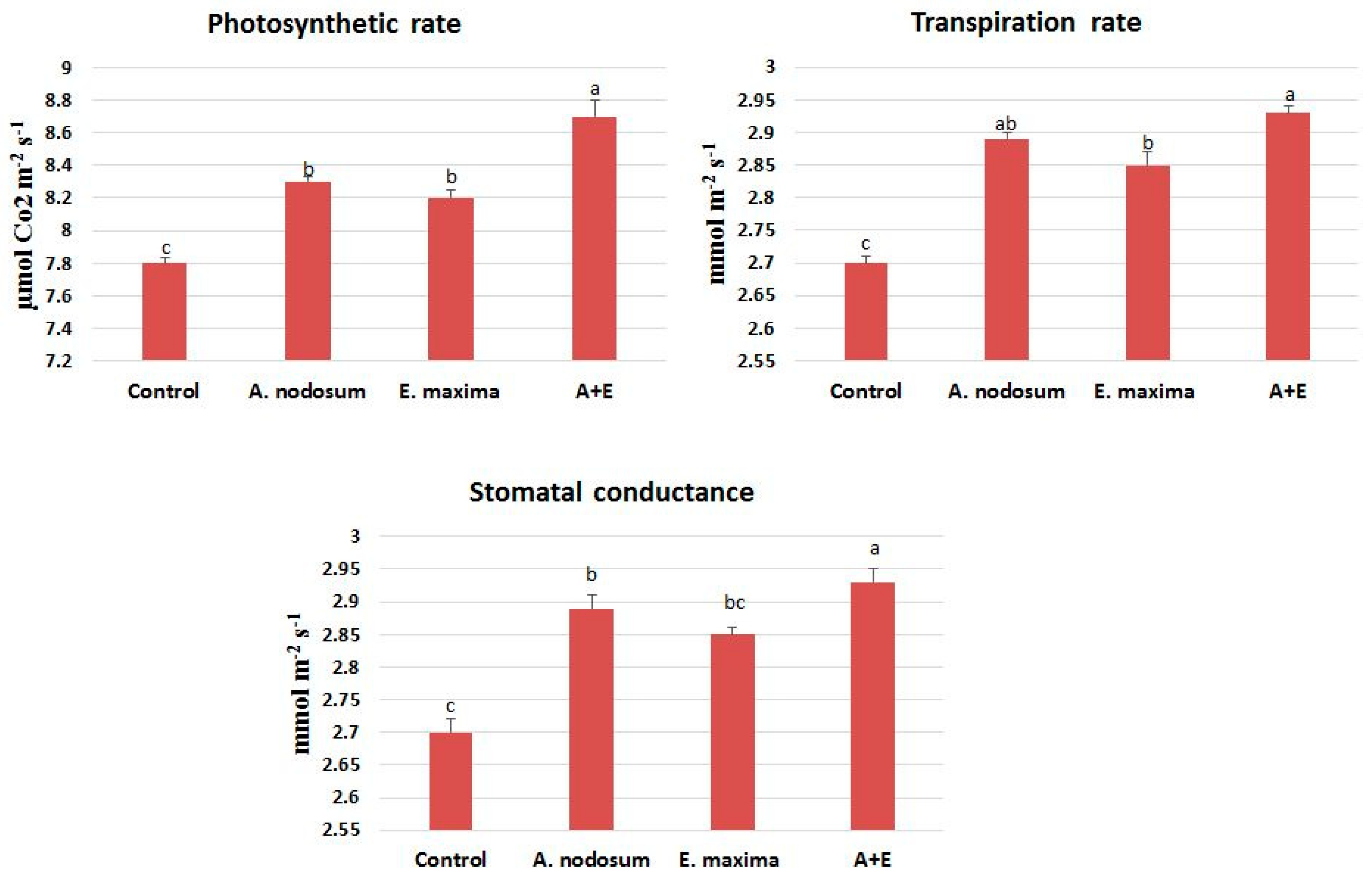
| Identified Compounds | Control | A. nodosum | E. maxima | A + E | |||
|---|---|---|---|---|---|---|---|
| RI | LRI | Standard Identification | |||||
| 1096 | 1096 | + | 2-nonanone | 17.01 ± 0.1 d | 19.11 ± 0.3 b | 18.21 ± 0.3 c | 18.09 ± 0.5 a |
| 1294 | 1294 | + | 2-undecanone | 49.66 ± 0.5 c | 54.38 ± 0.7 b | 53.1 ± 0.9 b | 61.9 ± 0.5 a |
| MCF-7 | HeLa | Jurkat | T24 | HT-29 | HEK-293 | |
|---|---|---|---|---|---|---|
| Control | 20.11 ± 0.9 | 6.5 ± 0.1 | 26.03 ± 0.1 | 95.3 ± 1.7 | 122.18 ± 2.3 | >200 |
| A. nodosum | 15.0 ± 0.3 | 5.24 ± 0.2 | 19.5 ± 0.8 | 83.85 ± 2.4 | 90.14 ± 3.8 | >200 |
| E. maxima | 16.7 ± 0.1 | 5.9 ± 0.3 | 20.46 ± 0.7 | 85.52 ± 3.1 | 101.17 ± 1.4 | >200 |
| A + E | 8.11 ± 0.7 | 4.3 ± 0.1 | 17.05 ± 0.3 | 82.2 ± 1.9 | 70.53 ± 2.1 | >200 |
| 2-undecanone | 4.21 ± 0.3 | 2.1 ± 0.1 | 8.32 ± 0.2 | 41.5 ± 0.7 | 33.26 ± 1.5 | >200 |
| 2-nonanone | 7.02 ± 0.2 | 3.2 ± 0.1 | 14.15 ± 0.1 | 64.1 ± 2.1 | 57.33 ± 2.2 | >200 |
| Vinblastine sulfate | ‒ | 2.4 ± 0.07 | 0.1 ± 0.03 | 61.44 ± 2.1 | 20.3 ± 0.8 | 50.3 ± 1.4 |
| Taxol | 0.08 ± 0.005 | ‒ | ‒ | ‒ | ‒ | ‒ |
| Bacteria | Control MIC MBC | A. nodosum MIC MBC | E. maxima MIC MBC | A + E MIC MBC | 2-Undecanone | 2-Nonanone | Streptomycin | Ampicillin |
|---|---|---|---|---|---|---|---|---|
| Bacillus cereus | 0.21 ± 0.01 | 0.13 ± 0.01 | 0.16 ± 0.01 | 0.07± 0.01 | 0.04± 0.01 | 0.06± 0.01 | 0.07 ± 0.02 | 0.09 ± 0.01 |
| 0.43 ± 0.03 | 0.29 ± 0.01 | 0.37± 0.01 | 0.16 ± 0.01 | 0.08 ± 0.01 | 0.13 ± 0.01 | 0.15 ± 0.02 | 0.18 ± 0.01 | |
| Dickeya solani | 0.42 ± 0.01 | 0.33± 0.01 | 0.38 ± 0.01 | 0.29 ± 0.01 | 0.16 ± 0.02 | 0.24 ± 0.02 | 0.22 ± 0.02 | 0.31 ± 0.01 |
| 0.93 ± 0.03 | 0.81 ± 0.03 | 0.85 ± 0.03 | 0.71 ± 0.02 | 0.32 ± 0.03 | 0.55 ± 0.01 | 0.43 ± 0.03 | 0.56 ± 0.01 | |
| Escherichia coli | 0.10 ± 0.01 | 0.80 ± 0.01 | 0.90 ± 0.02 | 0.70 ± 0.02 | 0.31 ± 0.03 | 0.54 ± 0.02 | 0.10 ± 0.01 | 0.20 ± 0.01 |
| 0.23 ± 0.01 | 0.20 ± 0.01 | 0.21 ± 0.03 | 0.16 ± 0.03 | 0.16 ± 0.01 | 0.12 ± 0.03 | 0.25 ± 0.01 | 0.40 ± 0.01 | |
| Listeria monocytogenes | 0.21 ± 0.01 | 0.19 ± 0.01 | 0.20± 0.01 | 0.17 ± 0.01 | 0.09 ± 0.01 | 0.14 ± 0.01 | 0.13 ± 0.01 | 0.16 ± 0.01 |
| 0.45 ± 0.03 | 0.39 ± 0.01 | 0.42 ± 0.01 | 0.31 ± 0.01 | 0.16 ± 0.03 | 0.24 ± 0.01 | 0.27 ± 0.01 | 0.30 ± 0.01 | |
| Micrococcus flavus | 0.21 ± 0.02 | 0.18 ± 0.01 | 0.19 ± 0.01 | 0.17 ± 0.01 | 0.09 ± 0.01 | 0.13 ± 0.01 | 0.11 ± 0.01 | 0.10 ± 0.01 |
| 0.47 ± 0.03 | 0.39 ± 0.01 | 0.43 ± 0.01 | 0.32 ± 0.01 | 0.16 ± 0.01 | 0.26 ± 0.01 | 0.20 ± 0.01 | 0.20± 0.01 | |
| Pectobacterium atrosepticum | 0.31 ± 0.01 | 0.23 ± 0.01 | 0.26 ± 0.02 | 0.20 ± 0.00 | 0.10 ± 0.01 | 0.16 ± 0.00 | 0.08 ± 0.01 | 0.26± 0.03 |
| 0.71 ± 0.03 | 0.45 ± 0.01 | 0.49 ± 0.01 | 0.40 ± 0.01 | 0.20 ± 0.01 | 0.31 ± 0.01 | 0.16 ± 0.01 | 0.52 ± 0.03 | |
| P. c. subsp. carotovorum | 0.11 ± 0.01 | 0.09 ± 0.01 | 0.10 ± 0.01 | 0.70 ± 0.01 | 0.33 ± 0.01 | 0.53 ± 0.01 | 0.09 ± 0.01 | 0.21± 0.01 |
| 0.25 ± 0.01 | 0.18 ± 0.00 | 0.23 ± 0.01 | 0.15 ± 0.01 | 0.07 ± 0.01 | 0.12 ± 0.01 | 0.20 ± 0.01 | 0.45 ± 0.03 | |
| Pseudomonas aeruginosa | 0.35 ± 0.01 | 0.28 ± 0.01 | 0.33± 0.01 | 0.25 ± 0.01 | 0.12 ± 0.01 | 0.21 ± 0.01 | 0.07 ± 0.02 | 0.11 ± 0.01 |
| 0.73 ± 0.01 | 0.54 ± 0.03 | 0.63 ± 0.01 | 0.53 ± 0.03 | 0.26 ± 0.01 | 0.42 ± 0.03 | 0.15 ± 0.01 | 0.24 ± 0.01 | |
| Staphylococcus aureus | 0.10 ± 0.00 | 0.08 ± 0.01 | 0.09 ± 0.01 | 0.06 ± 0.01 | 0.03 ± 0.00 | 0.05 ± 0.01 | 0.23 ± 0.02 | 0.10 ± 0.01 |
| 0.21 ± 0.01 | 0.19 ± 0.03 | 0.21 ± 0.01 | 0.13 ± 0.03 | 0.06 ± 0.01 | 0.10 ± 0.03 | 0.43 ± 0.01 | 0.21 ± 0.03 |
| Fungi | Control MIC MFC | A. nodosum MIC MFC | E. maxima MIC MFC | A + E MIC MFC | 2-Undecanone | 2-Nonanone | FLZ MIC MFC | KTZ MIC MFC |
|---|---|---|---|---|---|---|---|---|
| Aspergillus flavus | 0.24 ± 0.01 | 0.19 ± 0.02 | 0.21 ± 0.01 | 0.17 ± 0.01 | 0.8 ± 0.01 | 0.14 ± 0.01 | 0.11 ± 0.01 | 0.20 ± 0.01 |
| 0.54 ± 0.01 | 0.47 ± 0.03 | 0.49 ± 0.01 | 0.43 ± 0.01 | 0.21 ± 0.01 | 0.37 ± 0.03 | 0.23 ± 0.03 | 0.42 ± 0.01 | |
| Aspergillus ochraceus | 0.22 ± 0.01 | 0.18 ± 0.01 | 0.20 ± 0.03 | 0.18 ± 0.01 | 0.09 ± 0.01 | 0.15 ± 0.01 | 0.19 ± 0.01 | 0.21 ± 0.01 |
| 0.51 ± 0.02 | 0.45 ± 0.01 | 0.49 ± 0.01 | 0.32 ± 0.01 | 0.15 ± 0.01 | 0.26 ± 0.01 | 0.33 ± 0.01 | 0.40 ± 0.01 | |
| Aspergillus niger | 0.21 ± 0.01 | 0.18 ± 0.03 | 0.19 ± 0.01 | 0.17 ± 0.01 | 0.08 ± 0.01 | 0.13 ± 0.01 | 0.14 ± 0.01 | 0.10 ± 0.01 |
| 0.48 ± 0.03 | 0.39 ± 0.01 | 0.42 ± 0.03 | 0.34 ± 0.03 | 0.16 ± 0.01 | 0.26 ± 0.02 | 0.29 ± 0.01 | 0.20 ± 0.01 | |
| Penicillium funiculosum | 0.31 ± 0.01 | 0.26 ± 0.02 | 0.29 ± 0.01 | 0.24 ± 0.01 | 0.22 ± 0.03 | 0.20 ± 0.01 | 0.13 ± 0.01 | 2.10 ± 0.07 |
| 0.63 ± 0.03 | 0.58 ± 0.03 | 0.59 ± 0.09 | 0.55 ± 0.07 | 0.26 ± 0.02 | 0.41 ± 0.03 | 0.25 ± 0.01 | 3.58 ± 0.05 | |
| Penicillium ochrochloron | 0.15 ± 0.01 | 0.12 ± 0.01 | 0.13 ± 0.01 | 0.12 ± 0.01 | 0.06 ± 0.01 | 0.10 ± 0.01 | 0.20 ± 0.01 | 0.17 ± 0.01 |
| 0.35 ± 0.01 | 0.25 ± 0.03 | 0.27 ± 0.03 | 0.22 ± 0.01 | 0.10 ± 0.01 | 0.17 ± 0.01 | 0.31 ± 0.01 | 0.31 ± 0.01 | |
| Candida albicans | 0.32 ± 0.13 | 0.28 ± 0.03 | 0.29 ± 0.11 | 0.26 ± 0.02 | 0.12 ± 0.01 | 0.21 ± 0.01 | 0.10 ± 0.01 | 0.20 ± 0.01 |
| 0.73 ± 0.11 | 0.62 ± 0.05 | 0.67 ± 0.14 | 0.61 ± 0.06 | 0.30 ± 0.01 | 0.52 ± 0.02 | 0.20 ± 0.01 | 0.41 ± 0.01 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahmoud, E.A.; Elansary, H.O.; El-Ansary, D.O.; Al-Mana, F.A. Elevated Bioactivity of Ruta graveolens against Cancer Cells and Microbes Using Seaweeds. Processes 2020, 8, 75. https://doi.org/10.3390/pr8010075
Mahmoud EA, Elansary HO, El-Ansary DO, Al-Mana FA. Elevated Bioactivity of Ruta graveolens against Cancer Cells and Microbes Using Seaweeds. Processes. 2020; 8(1):75. https://doi.org/10.3390/pr8010075
Chicago/Turabian StyleMahmoud, Eman A., Hosam O. Elansary, Diaa O. El-Ansary, and Fahed A. Al-Mana. 2020. "Elevated Bioactivity of Ruta graveolens against Cancer Cells and Microbes Using Seaweeds" Processes 8, no. 1: 75. https://doi.org/10.3390/pr8010075
APA StyleMahmoud, E. A., Elansary, H. O., El-Ansary, D. O., & Al-Mana, F. A. (2020). Elevated Bioactivity of Ruta graveolens against Cancer Cells and Microbes Using Seaweeds. Processes, 8(1), 75. https://doi.org/10.3390/pr8010075






