Environmental Sustainability Assessment of Typical Cathode Materials of Lithium-Ion Battery Based on Three LCA Approaches
Abstract
1. Introduction
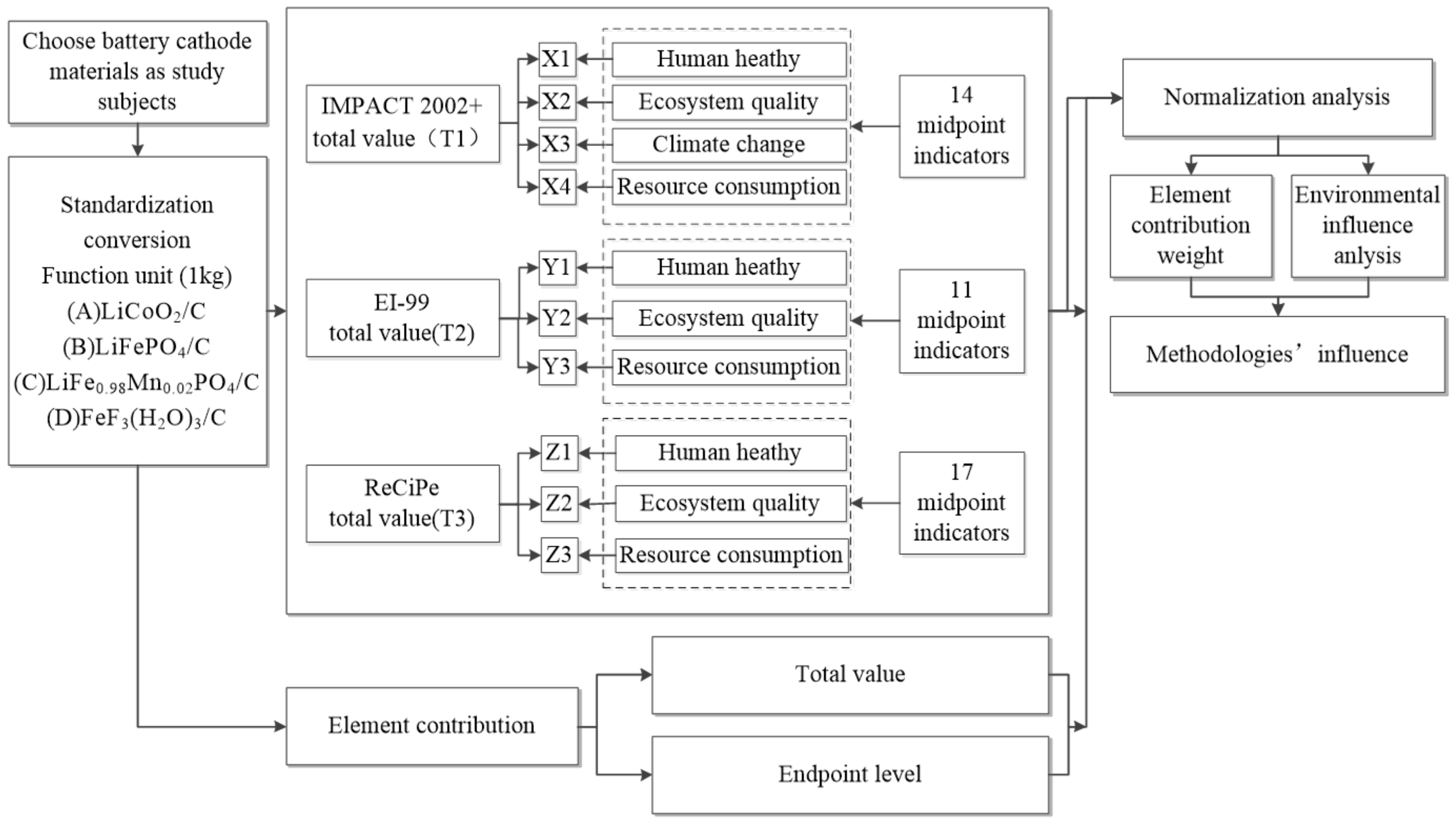
2. Materials and Methods
2.1. LCAs and Cathode Materials
2.2. Experimental Designation
2.2.1. Scope and Function Unit
2.2.2. Experimental Devices
2.2.3. Experimental Process
- i.
- we calculate the total value of the four cathode materials by the entropy method, so that it satisfies a unit (Pt).
- ii.
- we calculate the values of all endpoint indicators in the unit (Pt) and calculate their contribution to the total value.
- iii.
- we calculate the values of all midpoint indicators and give the normalized values of all midpoint indicators in this paper. Material B has been a major market cathode in recent years, especially in the field of electric vehicles. We choose all values of midpoint indicators of material B as baseline 1.
- iv.
- we calculate the contribution ratio of elements at the endpoint and the total level.
3. Results and Discussion
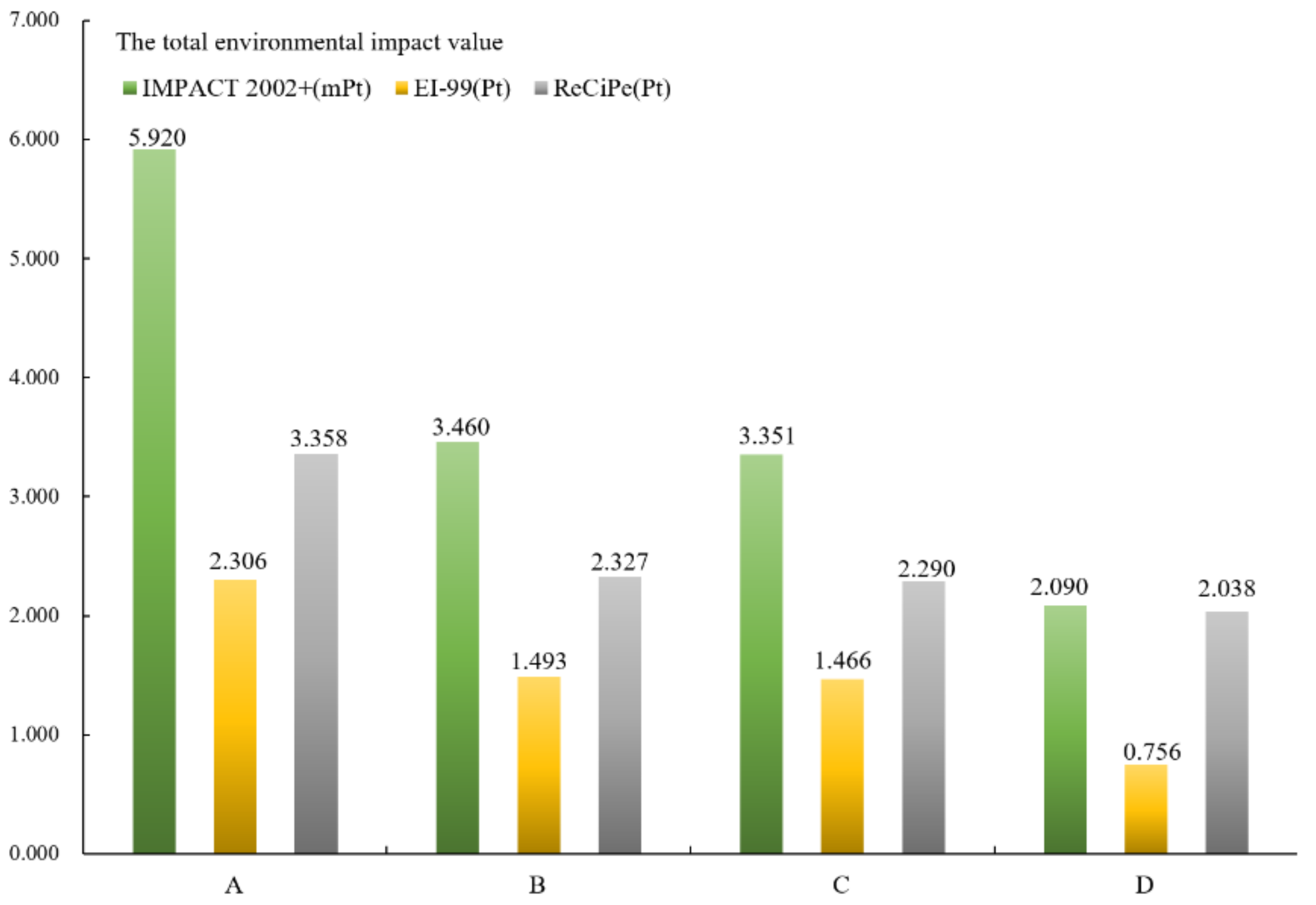
3.1. The Total Value of the Four Cathodes’ Environmental Impact
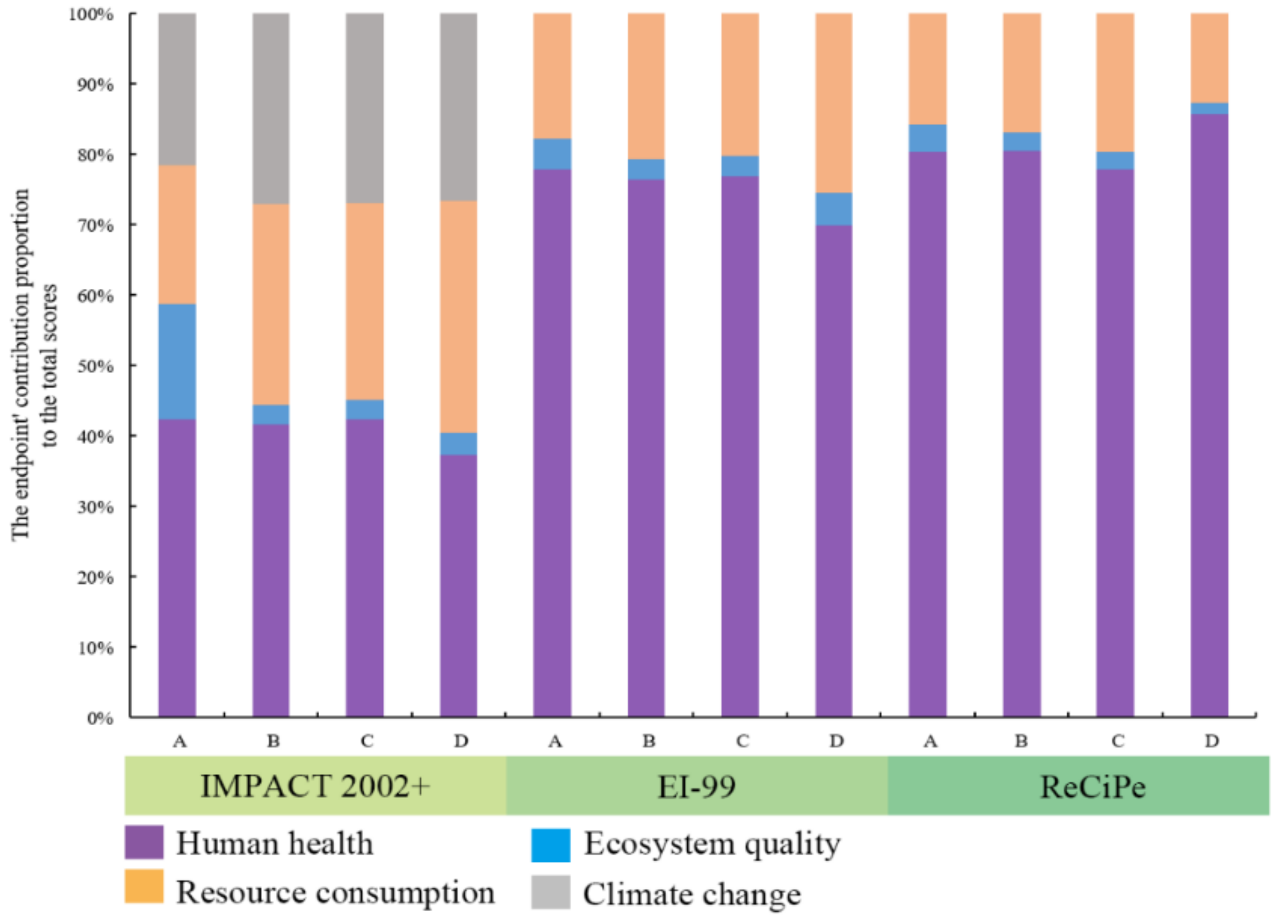
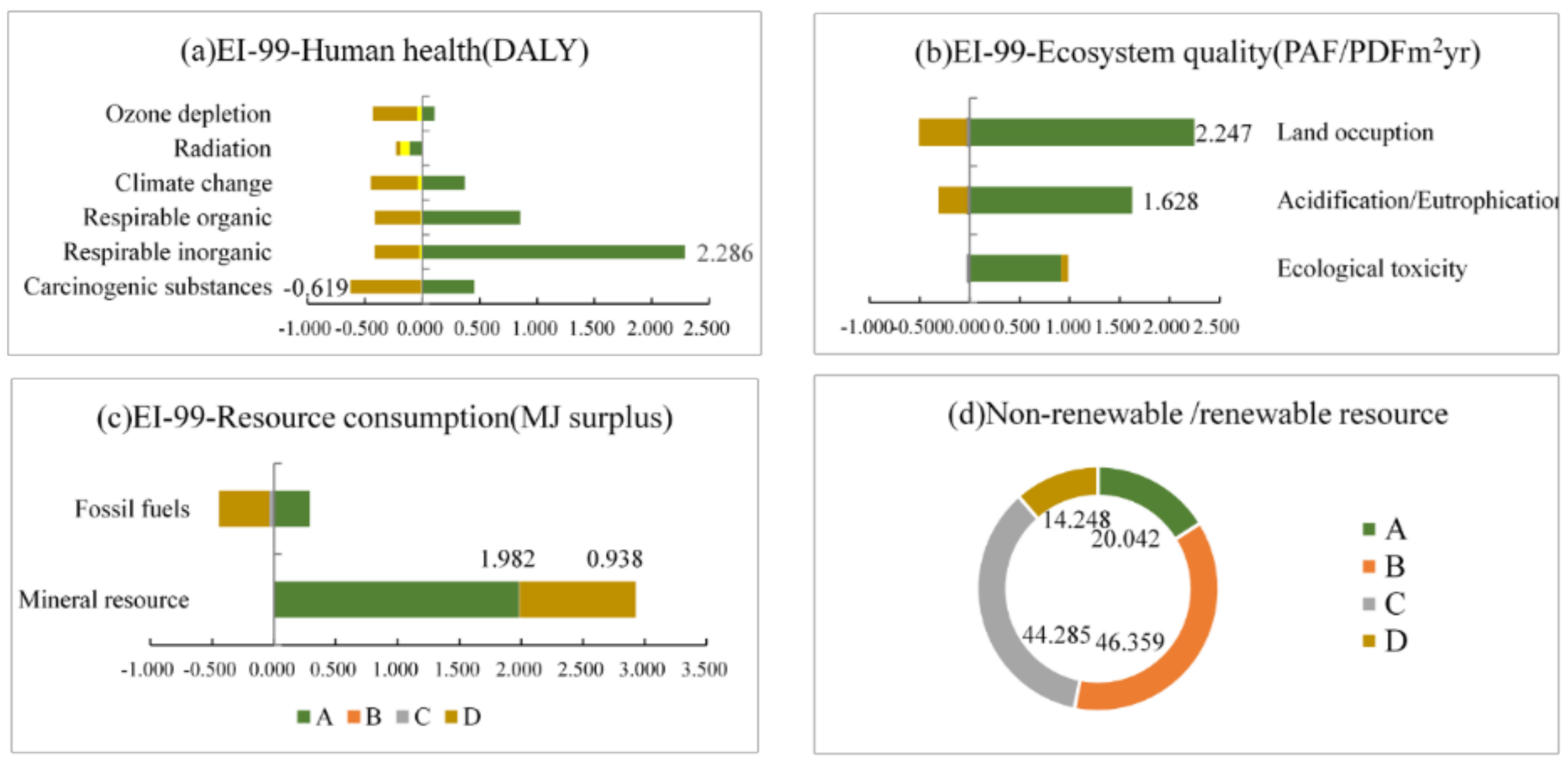
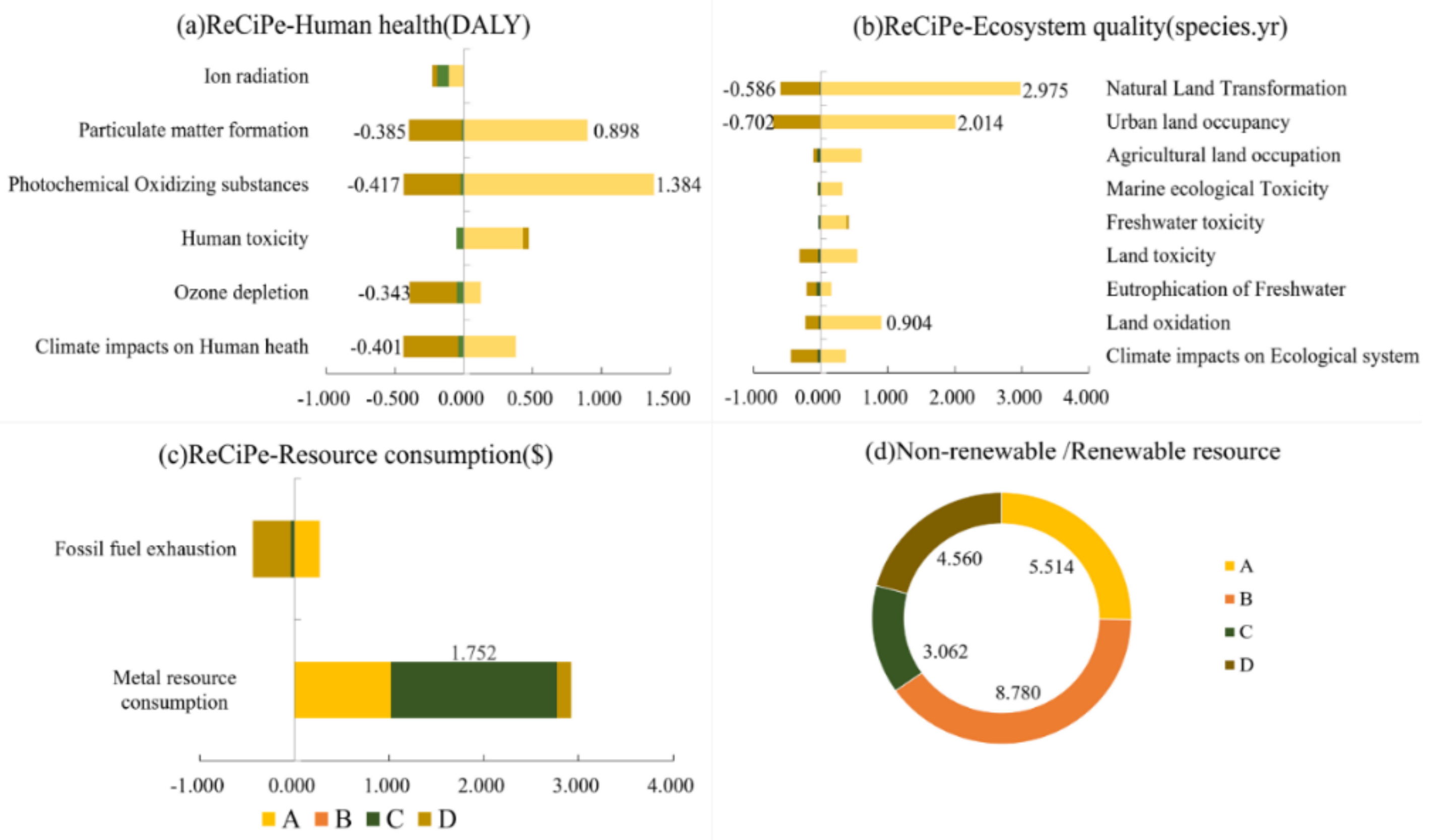
3.2. Endpoint Level
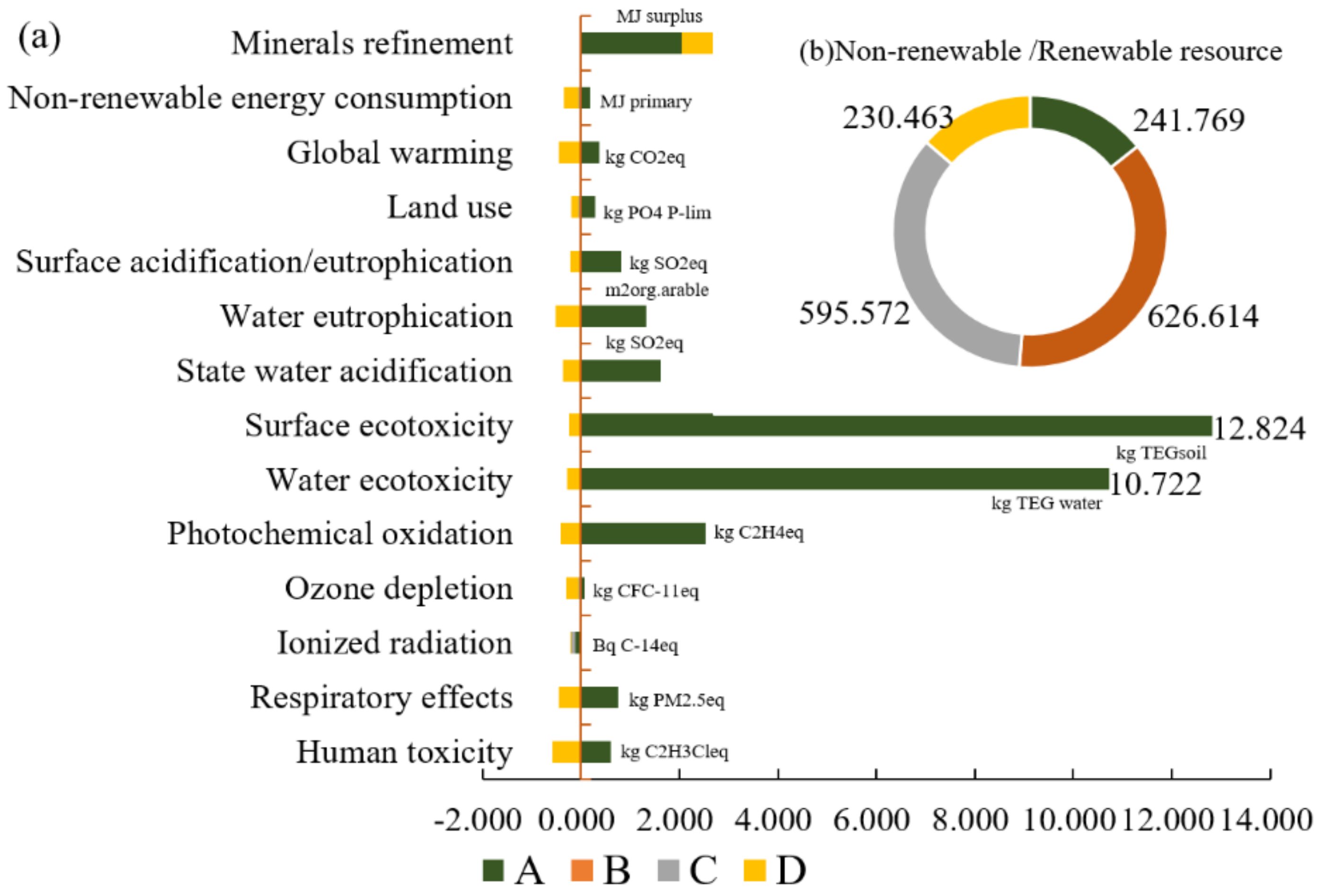
3.3. Midpoint Level
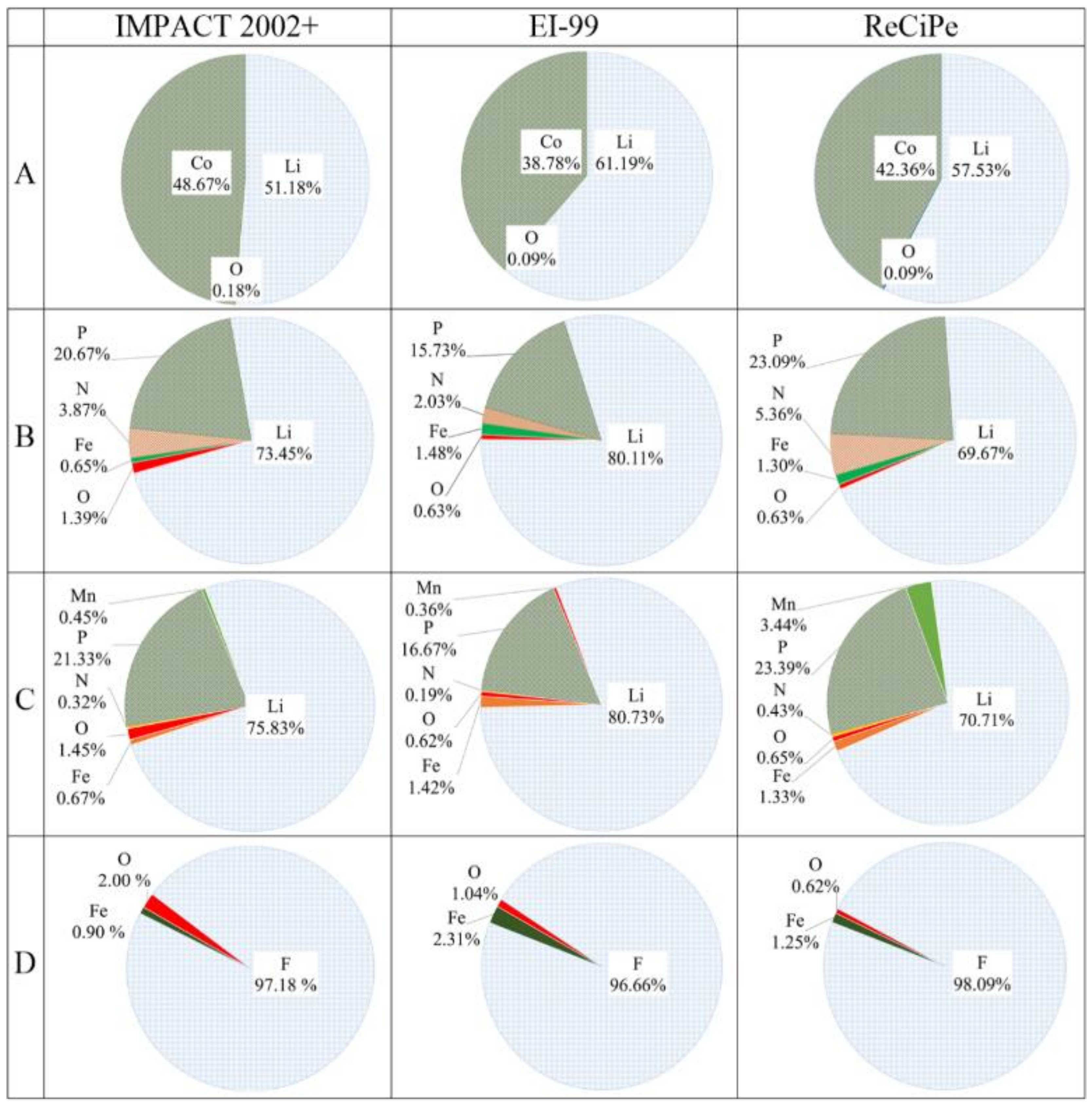
3.4. The Element Contribution to Environmental Sustainability
3.4.1. The Element Contribution in Endpoint Level
3.4.2. The Element Contribution to the Total Value
3.5. The Methodologies’ Influence to Environmental Sustainability Assessment
- (1)
- Different LCAs have little impact on the environmental sustainability assessment of the total, endpoint, and element levels of the four cathodes. The environmental sustainability of FeF3(H2O)3/C is better than LiFe0.98Mn0.02PO4/C, which is better than LiFePO4/C and LiCoO2/C.
- (2)
- The environmental sustainability of the four cathode types is mainly affected by human health. Lithium is the largest contributor to the environmental load of the first three market cathodes. However, for FeF3(H2O)3/C, fluorine is the largest contributor to its environmental load.
- (3)
- At the midpoint level, the four cathodes show different environmental sustainability in concrete indicators due to the different LCAs we chose. The mineral resource consumption of FeF3(H2O)3/C, the metal resource consumption of LiFe0.98Mn0.02PO4/C, high non-renewable resource consumption of LiFePO4/C, and the toxicity and land issues ofLiCoO2/C have seriously affected their environmental sustainability. Furthermore, the cobalt in LiCoO2/C, because of its ecosystem quality, and the phosphorus in LiFePO4/C and LiFe0.98Mn0.02PO4/C also obviously impair the environmental sustainability at the midpoint level.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Swart, P.; Dewulf, J.; Biernaux, A. Resource demand for the production of different cathode materials for lithium ion batteries. J. Clean. Prod. 2014, 84, 391–399. [Google Scholar] [CrossRef]
- Zhu, X.; Lin, T.; Manning, E.; Zhang, Y.; Yu, M.; Zuo, B.; Wang, L. Recent advances on Fe- and Mn-based cathode materials for lithium and sodium ion batteries. J. Nanoparticle Res. 2018, 20, 160. [Google Scholar] [CrossRef]
- Chakraborty, S.; Banerjee, A.; Watcharatharapong, T.; Araujo, R.B.; Ahuja, R. Current computational trends in polyanionic cathode materials for Li and Na batteries. J. Phys. Condens. Matter: Inst. of Phys. J. 2018, 30, 283003. [Google Scholar] [CrossRef] [PubMed]
- Jeong, G.; Kim, Y.-U.; Kim, H.; Kim, Y.-J.; Sohn, H.-J. Prospective materials and applications for li secondary batteries. Energy Environ. Sci. 2011, 4, 1986. [Google Scholar] [CrossRef]
- Cerdas, F.; Titscher, P.; Bognar, N.; Schmuch, R.; Winter, M.; Kwade, A.; Herrmann, C. Exploring the effect of increased energy density on the environmental impacts of traction batteries: A comparison of energy optimized lithium-ion and lithium-sulfur batteries for mobility applications. Energies 2018, 11, 150. [Google Scholar] [CrossRef]
- Andersson, K.; Eide, M.H. The feasibility of including sustainability in lca for product development. J. Clean. Prod. 1998, 6, 289–298. [Google Scholar] [CrossRef]
- Peters, J.F.; Baumann, M.; Zimmermann, B.; Braun, J.; Weil, M. The environmental impact of li-ion batteries and the role of key parameters—A review. Renew. Sustain. Energy Rev. 2017, 67, 491–506. [Google Scholar] [CrossRef]
- Peters, J.F.; Weil, M. Providing a common base for life cycle assessments of Li-ion batteries. J. Clean. Prod. 2018, 171, 704–713. [Google Scholar] [CrossRef]
- Li, L.; Dunn, J.B.; Zhang, X.X.; Gaines, L.; Chen, R.J.; Wu, F.; Amine, K. Recovery of metals from spent lithium-ion batteries with organic acids as leaching reagents and environmental assessment. J. Power Sources 2013, 233, 180–189. [Google Scholar] [CrossRef]
- Yuan, L.X.; Wang, Z.H.; Zhang, W.X.; Hu, X.L.; Chen, J.T.; Huang, Y.H.; Goodenough, J.B. Development and challenges of lifepo4 cathode material for lithium-ion batteries. Energy Environ. Sci. 2011, 4, 269–284. [Google Scholar] [CrossRef]
- Lin, Y.; Zeng, B.; Lin, Y.; Li, X.; Zhao, G.; Zhou, T.; Lai, H.; Huang, Z. Electrochemical properties of carbon-coated lifepo4 and LiFe0.98Mn0.02PO4 cathode materials synthesized by solid-state reaction. Rare Met. 2012, 31, 145–149. [Google Scholar] [CrossRef]
- Zeng, L.; Gong, Q.; Liao, X.; He, L.; He, Y.; Ma, Z. Enhanced low-temperature performance of slight Mn-substituted lifepo4/c cathode for lithium ion batteries. Chin. Sci. Bull. 2011, 56, 1262–1266. [Google Scholar] [CrossRef]
- Togo, M.; Nakahira, A. Structure refinement of mn-substituted LiMnxFe1-xPO4. Mater. Sci. Appl. 2018, 09, 542–553. [Google Scholar]
- Wu, C.; Li, X.X.; Wu, F.; Bai, Y.; Chen, M.Z.; Zhong, Y. Composite FeF3•3H2O/C cathode material for lithium ion battery. Adv. Mater. Res. 2011, 391–392, 1090–1094. [Google Scholar] [CrossRef]
- Ludwig, J.; Nilges, T. Recent progress and developments in lithium cobalt phosphate chemistry- syntheses, polymorphism and properties. J. Power Sources 2018, 382, 101–115. [Google Scholar] [CrossRef]
- Pfleging, W. A review of laser electrode processing for development and manufacturing of lithium-ion batteries. Nanophotonics 2018, 7, 549–573. [Google Scholar] [CrossRef]
- Winslow, K.M.; Laux, S.J.; Townsend, T.G. A review on the growing concern and potential management strategies of waste lithium-ion batteries. Resour. Conserv. Recycl. 2018, 129, 263–277. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, Y.; Huang, K.; Chen, B.; Deng, W.; Yao, Y. Quantifying the environmental impact of a Li-rich high-capacity cathode material in electric vehicles via life cycle assessment. Environ. Sci. Pollut. Res. Int. 2017, 24, 1251–1260. [Google Scholar] [CrossRef]
- Wang, C.; Chen, B.; Yu, Y.; Wang, Y.; Zhang, W. Carbon footprint analysis of lithium ion secondary battery industry: Two case studies from China. J. Clean. Prod. 2017, 163, 241–251. [Google Scholar] [CrossRef]
- Liang, Y.; Su, J.; Xi, B.; Yu, Y.; Ji, D.; Sun, Y.; Cui, C.; Zhu, J. Life cycle assessment of lithium-ion batteries for greenhouse gas emissions. Resour. Conserv. Recycl. 2017, 117, 285–293. [Google Scholar] [CrossRef]
- Deng, Y.; Li, J.; Li, T.; Zhang, J.; Yang, F.; Yuan, C. Life cycle assessment of high capacity molybdenum disulfide lithium-ion battery for electric vehicles. Energy 2017, 123, 77–88. [Google Scholar] [CrossRef]
- Zackrisson, M.; Fransson, K.; Hildenbrand, J.; Lampic, G.; O’Dwyer, C. Life cycle assessment of lithium-air battery cells. J. Clean. Prod. 2016, 135, 299–311. [Google Scholar] [CrossRef]
- Gong, Y.; Yu, Y.; Huang, K.; Hu, J.; Li, C. Evaluation of lithium-ion batteries through the simultaneous consideration of environmental, economic and electrochemical performance indicators. J. Clean. Prod. 2018, 170, 915–923. [Google Scholar] [CrossRef]
- Xu, Y.J.; Huang, K.; Yu, Y.J.; Wang, X.M. Changes in water footprint of crop production in beijing from 1978 to 2012: A logarithmic mean divisia index decomposition analysis. J. Clean. Prod. 2015, 87, 180–187. [Google Scholar] [CrossRef]
- Jolliet, O.; Margni, M.; Charles, R.; Humbert, S.; Payet, J.; Rebitzer, G.; Rosenbaum, R. Impact 2002+: A new life cycle impact assessment methodology. Int. J. Life Cycle Assess. 2003, 8, 324–330. [Google Scholar] [CrossRef]
- Audenaert, A.; De Cleyn, S.H.; Buyle, M. Lca of low-energy flats using the eco-indicator 99 method: Impact of insulation materials. Energy Build. 2012, 47, 68–73. [Google Scholar] [CrossRef]
- Lamnatou, C.; Motte, F.; Notton, G.; Chemisana, D.; Cristofari, C. Building-integrated solar thermal system with/without phase change material: Life cycle assessment based on recipe, usetox and ecological footprint. J. Clean. Prod. 2018, 193, 672–683. [Google Scholar] [CrossRef]
- Henclik, A.; Bajdur, W.M. Application of selected methods of life cycle assessment to judgment of environmental hazard of production process of flocculant synthesized from waste phenol-formaldehyde resin. Rocz. Ochr. Sr. 2011, 13, 1809–1822. [Google Scholar]
- Lamnatou, C.; Baig, H.; Chemisana, D.; Mallick, T.K. Environmental assessment of a building-integrated linear dielectric-based concentrating photovoltaic according to multiple life-cycle indicators. J. Clean. Prod. 2016, 131, 773–784. [Google Scholar] [CrossRef]
- Cavalett, O.; Chagas, M.F.; Seabra, J.E.A.; Bonomi, A. Comparative lca of ethanol versus gasoline in Brazil using different lcia methods. Int. J. Life Cycle Assess. 2012, 18, 647–658. [Google Scholar] [CrossRef]
- Owsianiak, M.; Laurent, A.; Bjørn, A.; Hauschild, M.Z. Impact 2002+, recipe 2008 and ilcd’s recommended practice for characterization modelling in life cycle impact assessment: A case study-based comparison. Int. J. Life Cycle Assess. 2014, 19, 1007–1021. [Google Scholar] [CrossRef]
- Starostka-Patyk, M. New products design decision making support by simapro software on the base of defective products management. Procedia Comput. Sci. 2015, 65, 1066–1074. [Google Scholar] [CrossRef]
- Herrmann, I.T.; Moltesen, A. Does it matter which life cycle assessment (lca) tool you choose? A comparative assessment of simapro and gabi. J. Clean. Prod. 2015, 86, 163–169. [Google Scholar] [CrossRef]
- Wanger, T.C. The lithium future-resources, recycling, and the environment. Conserv. Lett. 2011, 4, 202–206. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, K.; Yu, Y.J.; Yang, B.B. Mapping of water footprint research: A bibliometric analysis during 2006–2015. J. Clean. Prod. 2017, 149, 70–79. [Google Scholar] [CrossRef]
- Koellner, T. Land use in product life cycles and its consequences for ecosystem quality. Int. J. Life Cycle Assess. 2002, 7, 130–130. [Google Scholar] [CrossRef]
- Klinglmair, M.; Sala, S.; Brandão, M. Assessing resource depletion in lca: A review of methods and methodological issues. Int. J. Life Cycle Assess. 2013, 19, 580–592. [Google Scholar] [CrossRef]
- Li, W.; Song, B.; Manthiram, A. High-voltage positive electrode materials for lithium-ion batteries. Chem. Soci. Rev. 2017, 46, 3006–3059. [Google Scholar] [CrossRef]
- Branco, H.; Castro, R.; Setas Lopes, A. Battery energy storage systems as a way to integrate renewable energy in small isolated power systems. Energy Sustain. Dev. 2018, 43, 90–99. [Google Scholar] [CrossRef]
- Li, Y.; Yang, J.; Song, J. Design structure model and renewable energy technology for rechargeable battery towards greener and more sustainable electric vehicle. Renew. Sustain. Energy Rev. 2017, 74, 19–25. [Google Scholar] [CrossRef]
- Peng, C.; Hamuyuni, J.; Wilson, B.P.; Lundstrom, M. Selective reductive leaching of cobalt and lithium from industrially crushed waste Li-ion batteries in sulfuric acid system. Waste Manag. 2018, 76, 582–590. [Google Scholar] [CrossRef] [PubMed]
- Xiang, W.; Zhong, Y.; Tang, Y.; Shen, H.; Wang, E.; Liu, H.; Zhong, B.; Guo, X. Improving the electrochemical kinetics of lithium manganese phosphate via co-substitution with iron and cobalt. J. Alloys Compd. 2015, 635, 180–187. [Google Scholar] [CrossRef]
- Di Lecce, D.; Brutti, S.; Panero, S.; Hassoun, J. A new sn-c/life0.1co0.9po4 full lithium-ion cell with ionic liquid-based electrolyte. Mater. Lett. 2015, 139, 329–332. [Google Scholar] [CrossRef]
- Yang, Y.; Meng, X.; Cao, H.; Lin, X.; Liu, C.; Sun, Y.; Zhang, Y.; Sun, Z. Selective recovery of lithium from spent lithium iron phosphate batteries: A sustainable process. Green Chem. 2018, 20, 3121–3133. [Google Scholar] [CrossRef]
| Endpoint | Indicator Value/Pt | |||
|---|---|---|---|---|
| A | B | C | D | |
| X1 | 2.510×10−03 | 1.443×10−03 | 1.423×10−03 | 0.779×10−03 |
| X2 | 0.966×10−03 | 0.093×10−03 | 0.090×10−03 | 0.068×10−03 |
| X3 | 1.278×10−03 | 0.937×10−03 | 0.900×10−03 | 0.555×10−03 |
| X4 | 1.166×10−03 | 0.988×10−03 | 0.938×10−03 | 0.688×10−03 |
| Y1 | 1.796 | 1.142 | 1.128 | 0.528 |
| Y2 | 0.101 | 0.042 | 0.041 | 0.035 |
| Y3 | 0.409 | 0.309 | 0.297 | 0.193 |
| Z1 | 2.697 | 1.875 | 1.784 | 1.747 |
| Z2 | 0.134 | 0.060 | 0.058 | 0.034 |
| Z3 | 0.527 | 0.393 | 0.449 | 0.258 |
| LCAs | Endpoint | Li | O | Co |
|---|---|---|---|---|
| IMPACT 2002+ (%) | X1 | 53.60 | 0.11 | 46.30 |
| X2 | 8.30 | 0.01 | 91.70 | |
| X3 | 64.70 | 0.26 | 35.10 | |
| X4 | 66.70 | 0.36 | 33.00 | |
| EI-99 (%) | Y1 | 62.60 | 0.05 | 37.40 |
| Y2 | 30.70 | 0.06 | 69.30 | |
| Y3 | 62.50 | 0.23 | 37.30 | |
| ReCiPe (%) | Z1 | 58.30 | 0.07 | 41.60 |
| Z2 | 40.50 | 0.09 | 59.40 | |
| Z3 | 57.90 | 0.20 | 41.90 |
| LCAs | Endpoint | Li | O | Fe | N | P |
|---|---|---|---|---|---|---|
| IMPACT 2002+ (%) | X1 | 78.20 | 0.91 | 1.14 | 2.28 | 17.5 |
| X2 | 72.40 | 0.28 | 3.09 | 3.31 | 20.9 | |
| X3 | 74.10 | 1.63 | 0.16 | 4.41 | 19.7 | |
| X4 | 66.00 | 1.96 | 0.16 | 5.73 | 26.2 | |
| EI-99 (%) | Y1 | 83.70 | 0.42 | 1.81 | 1.34 | 12.7 |
| Y2 | 60.50 | 0.81 | 0.40 | 3.18 | 35.1 | |
| Y3 | 69.50 | 1.40 | 0.40 | 4.44 | 24.3 | |
| ReCiPe (%) | Z1 | 70.40 | 0.49 | 0.34 | 5.64 | 23.2 |
| Z2 | 76.20 | 0.97 | 0.17 | 3.84 | 18.9 | |
| Z3 | 65.20 | 1.25 | 6.09 | 4.27 | 23.2 |
| LCAs | Endpoint | Li | Fe | O | N | P | Mn |
|---|---|---|---|---|---|---|---|
| IMPACT 2002+ (%) | X1 | 79.30 | 1.16 | 0.93 | 0.18 | 17.70 | 0.72 |
| X2 | 74.30 | 3.17 | 0.28 | 0.27 | 21.50 | 0.47 | |
| X3 | 77.00 | 0.17 | 1.71 | 0.36 | 20.50 | 0.23 | |
| X4 | 69.50 | 0.17 | 2.08 | 0.48 | 27.60 | 0.25 | |
| EI-99 (%) | Y1 | 83.60 | 1.72 | 0.40 | 0.14 | 13.80 | 0.34 |
| Y2 | 63.50 | 0.34 | 0.66 | 0.31 | 33.90 | 1.29 | |
| Y3 | 72.20 | 0.42 | 1.47 | 0.37 | 25.20 | 0.29 | |
| ReCiPe (%) | Z1 | 73.90 | 0.36 | 0.52 | 0.47 | 24.30 | 0.37 |
| Z2 | 78.50 | 0.18 | 1.00 | 0.31 | 19.50 | 0.53 | |
| Z3 | 57.00 | 5.33 | 1.10 | 0.30 | 20.30 | 16.00 |
| LCAs | Endpoint | Fe | O | F |
|---|---|---|---|---|
| IMPACT 2002+ (%) | X1 | 1.78 | 1.47 | 96.80 |
| X2 | 3.55 | 0.33 | 96.10 | |
| X3 | 0.23 | 2.39 | 97.40 | |
| X4 | 0.19 | 2.44 | 97.40 | |
| EI-99 (%) | Y1 | 3.08 | 0.73 | 96.20 |
| Y2 | 0.33 | 0.66 | 99.00 | |
| Y3 | 0.54 | 1.95 | 97.50 | |
| ReCiPe (%) | Z1 | 0.31 | 0.46 | 99.20 |
| Z2 | 0.25 | 1.47 | 98.30 | |
| Z3 | 7.81 | 1.64 | 90.50 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Wu, H.; Hu, Y.; Yu, Y.; Huang, K. Environmental Sustainability Assessment of Typical Cathode Materials of Lithium-Ion Battery Based on Three LCA Approaches. Processes 2019, 7, 83. https://doi.org/10.3390/pr7020083
Wang L, Wu H, Hu Y, Yu Y, Huang K. Environmental Sustainability Assessment of Typical Cathode Materials of Lithium-Ion Battery Based on Three LCA Approaches. Processes. 2019; 7(2):83. https://doi.org/10.3390/pr7020083
Chicago/Turabian StyleWang, Lei, Haohui Wu, Yuchen Hu, Yajuan Yu, and Kai Huang. 2019. "Environmental Sustainability Assessment of Typical Cathode Materials of Lithium-Ion Battery Based on Three LCA Approaches" Processes 7, no. 2: 83. https://doi.org/10.3390/pr7020083
APA StyleWang, L., Wu, H., Hu, Y., Yu, Y., & Huang, K. (2019). Environmental Sustainability Assessment of Typical Cathode Materials of Lithium-Ion Battery Based on Three LCA Approaches. Processes, 7(2), 83. https://doi.org/10.3390/pr7020083





