Inhibition of Key Enzymes Linked to Obesity and Cytotoxic Activities of Whole Plant Extracts of Vernonia mesplilfolia Less
Abstract
1. Introduction
2. Results
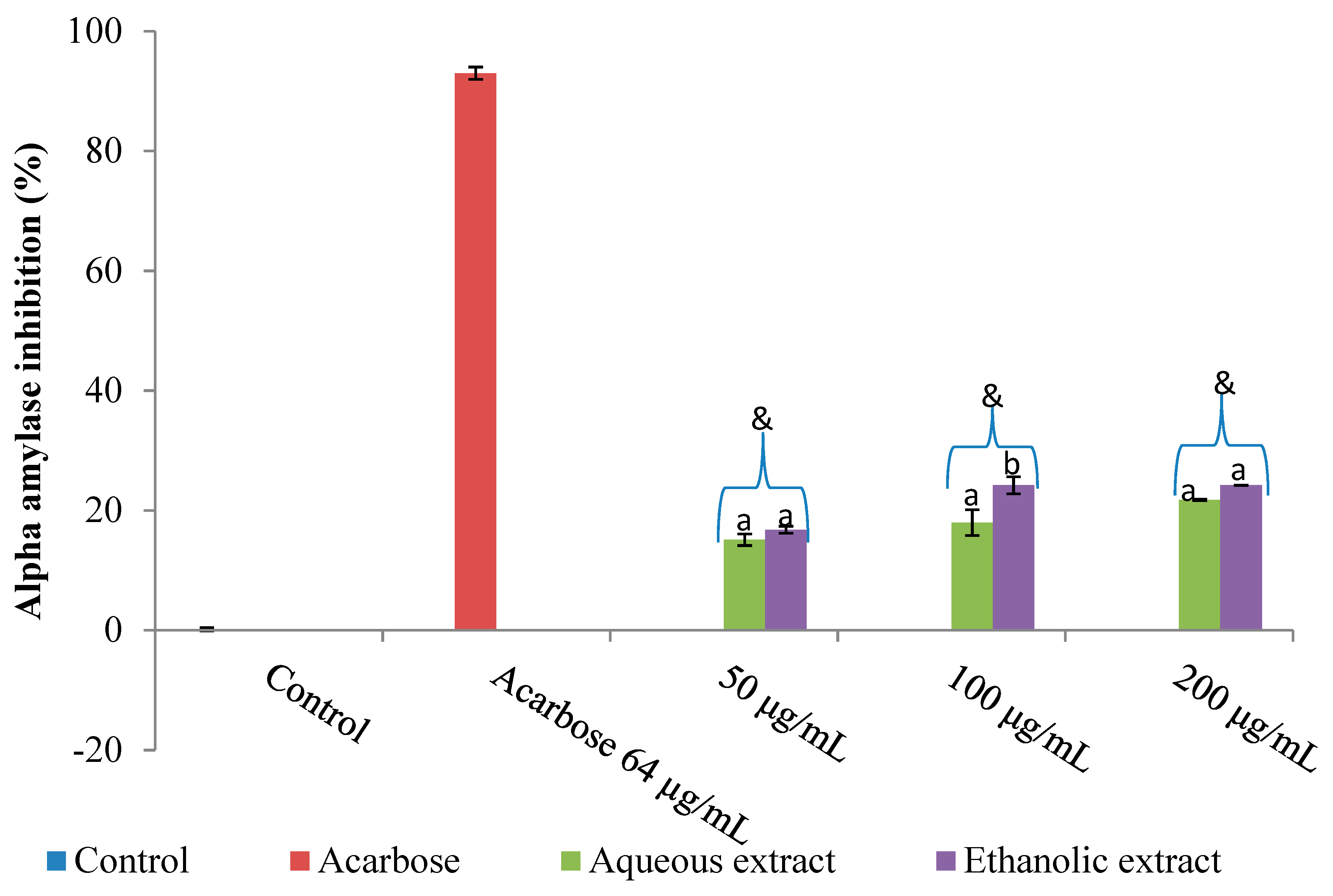
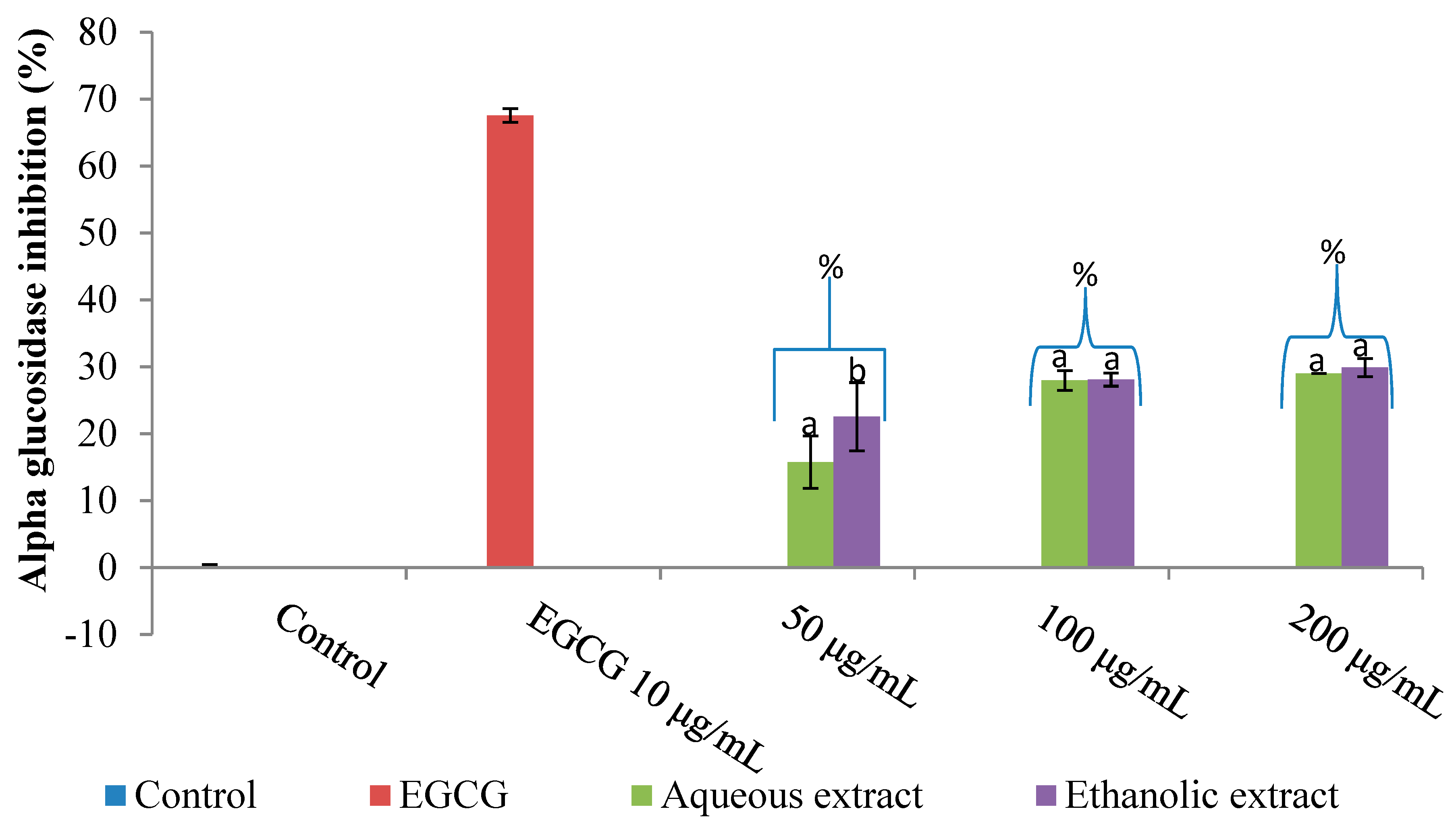
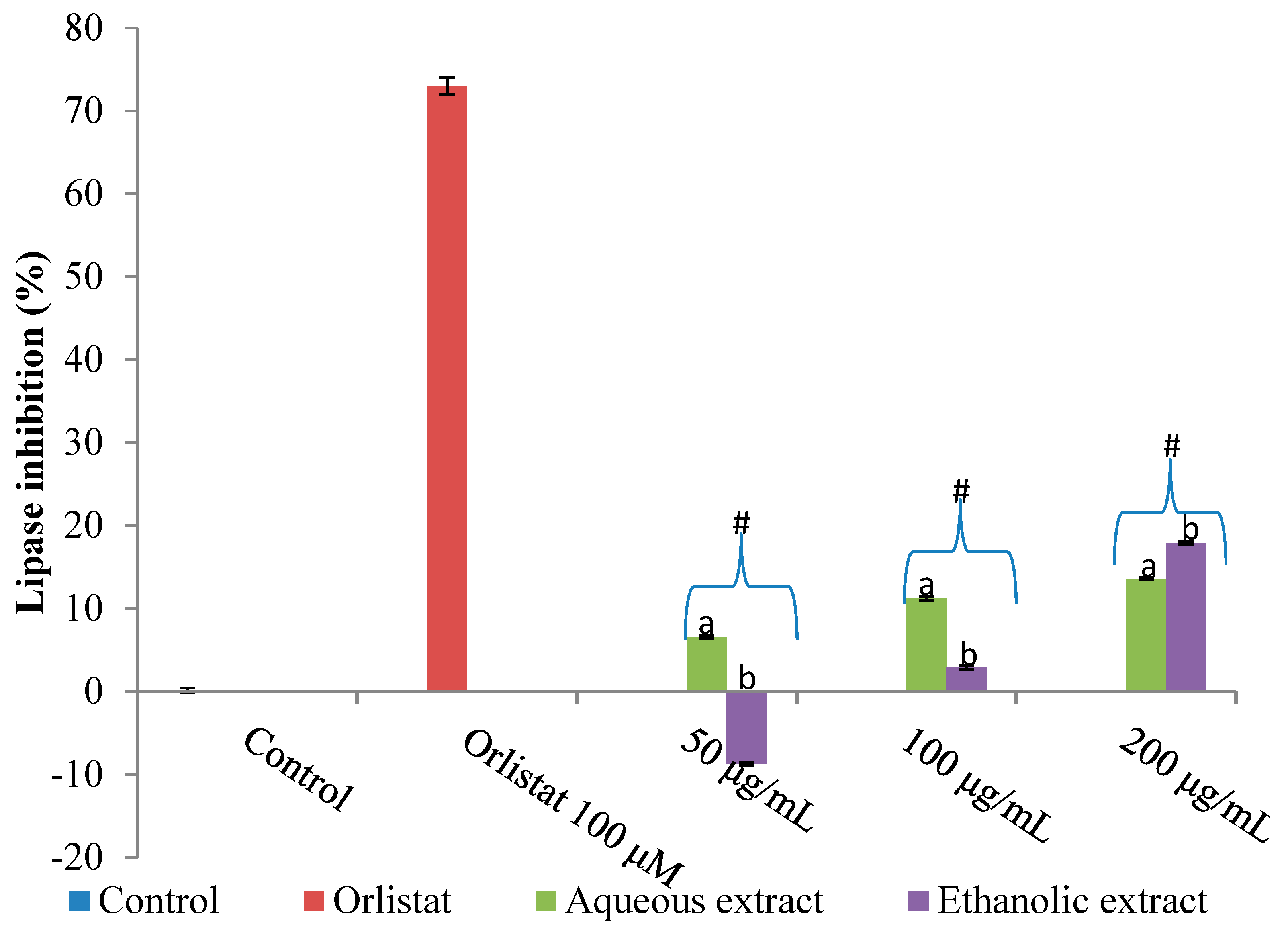
2.1. Inhibition of Amylase, Glucosidase and Lipase by V. mespilifolia
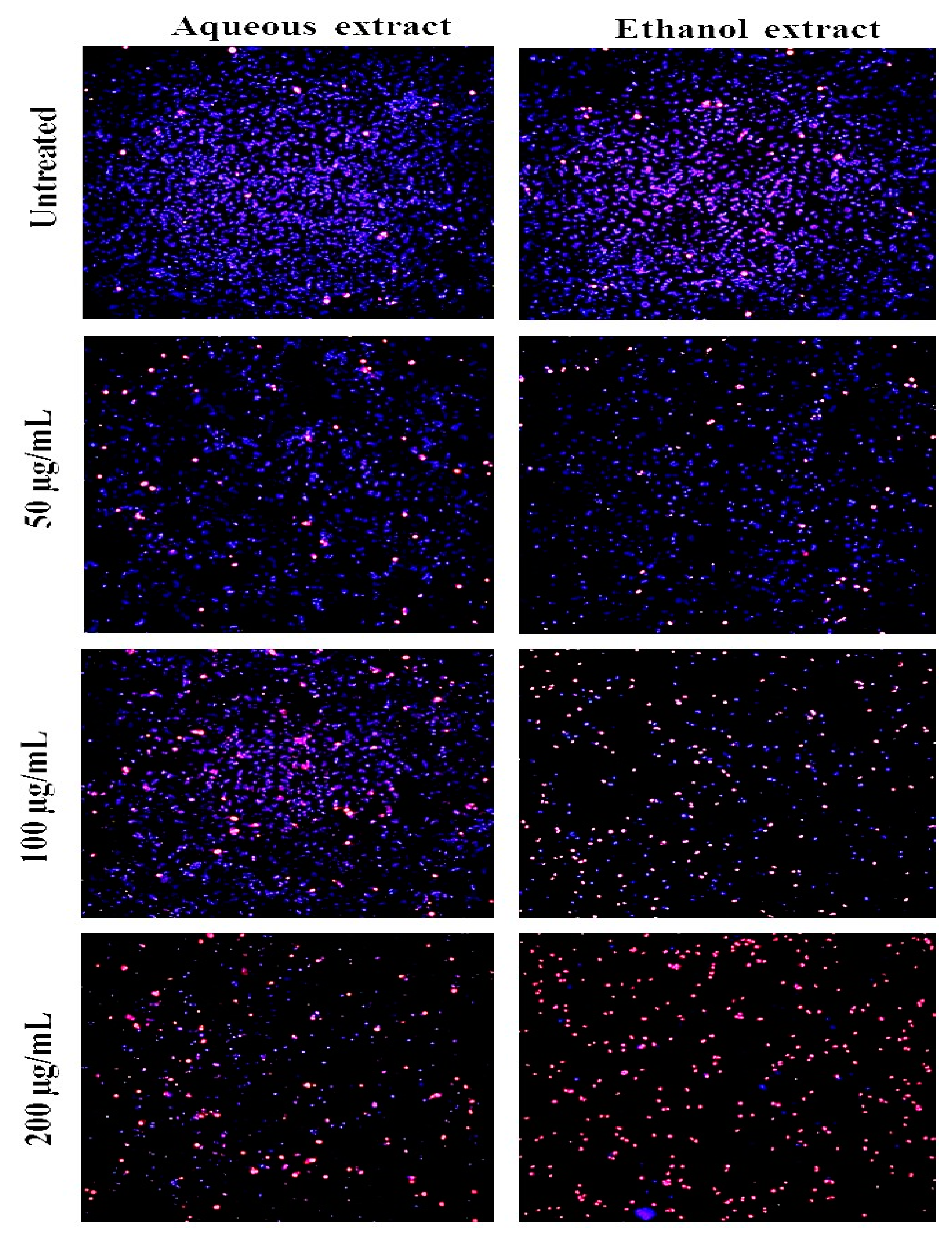
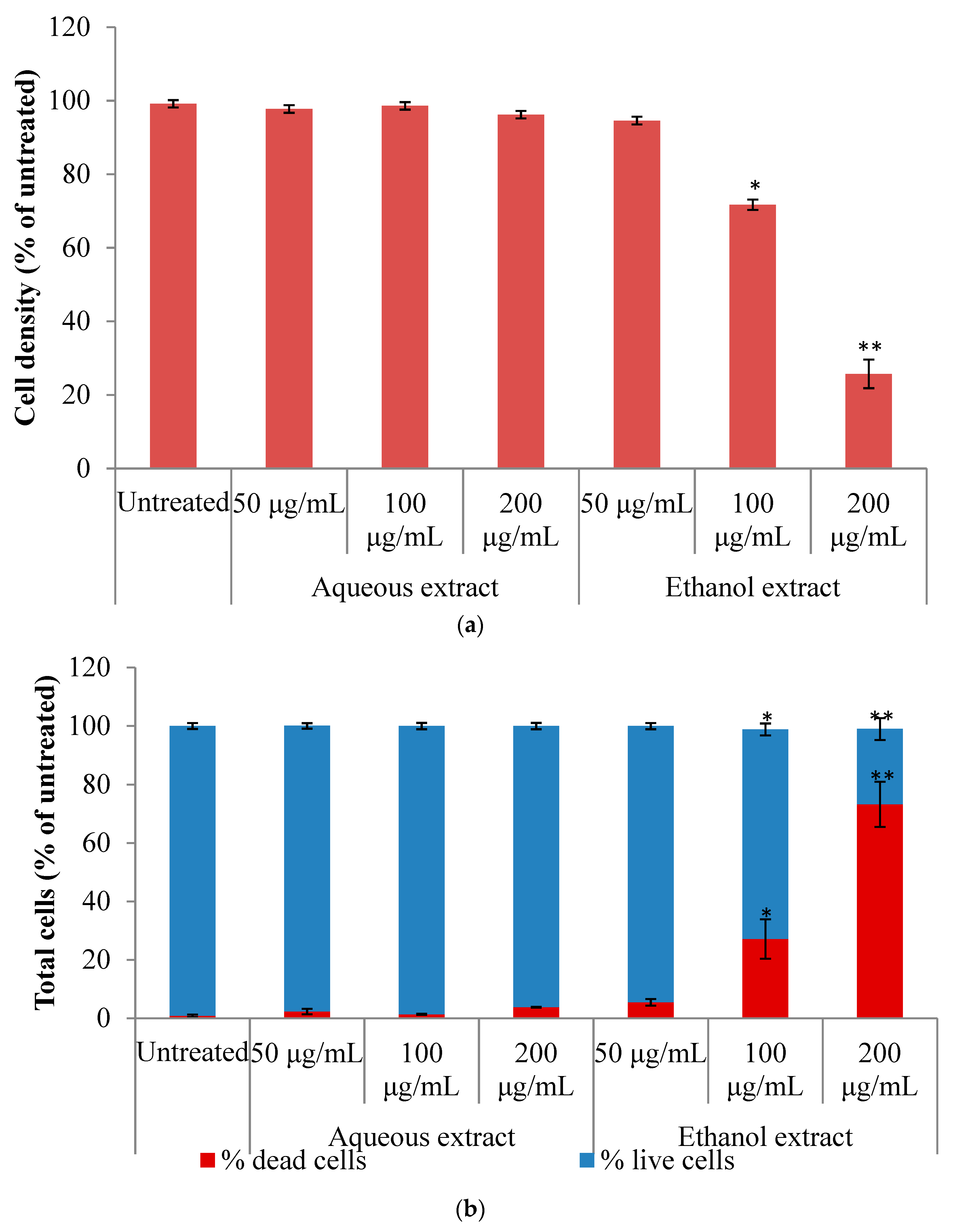
2.2. Cytotoxicity Activity
2.3. Discussion
3. Experimental Procedure
3.1. Chemicals, Reagents and Equipment
3.2. Plant Materials
3.3. Extraction Procedure
3.4. Inhibitory Potential of Plant Extracts
3.5. Alpha-Glucosidase
3.6. Pancreatic Lipase
4. Cell Cytotoxicity Assay Using the Hoechst/PI Staining
4.1. Cell Line
4.2. Data Evaluation
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mohamed, G.A.; Ibrahim, S.R.M.; Elkhayat, E.S.; El Dine, R.S. Natural anti-obesity agents. Bull. Fac. Pharm. Cairo Univ. 2014. [Google Scholar] [CrossRef]
- Global Burden of Disease Global Burden of Disease Study 2015 (GBD 2015) Obesity and Overweight Prevalence 1980–2015|GHDx. Available online: http://ghdx.healthdata.org (accessed on 7 November 2018).
- Chooi, Y.C.; Ding, C.; Magkos, F. The epidemiology of obesity. Metabolism 2019. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; Shimomura, I. Increased oxidative stress in obesity: Implications for metabolic syndrome, diabetes, hypertension, dyslipidemia, atherosclerosis, and cancer. Obes. Res. Clin. Pract. 2013, 7, e330–e341. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Kim, K.; Chung, Y.; Shong, M.; Park, S. Anti-obesity Agents: A Focused Review on the Structural Classification of Therapeutic Entities. Curr. Top. Med. Chem. 2009, 9, 466–481. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; McPherson, K.; Marsh, T.; Gortmaker, S.L.; Brown, M. Health and economic burden of the projected obesity trends in the USA and the UK. Lancet 2011, 378, 815–825. [Google Scholar] [CrossRef]
- Finkelstein, E.A.; Khavjou, O.A.; Thompson, H.; Trogdon, J.G.; Pan, L.; Sherry, B.; Dietz, W. Obesity and severe obesity forecasts through 2030. Am. J. Prev. Med. 2012, 42, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Yanovski, S.Z.; Yanovski, J.A. Long-term drug treatment for obesity: A systematic and clinical review. J. Am. Med. Assoc. 2014, 311, 74–86. [Google Scholar] [CrossRef]
- Kang, J.G.; Park, C.Y. Anti-obesity drugs: A review about their effects and safety. Diabetes Metab. J. 2012, 36, 13–25. [Google Scholar] [CrossRef]
- Unuofin, J.O.; Otunola, G.A.; Afolayan, A.J. In Vitro α-amylase, α-glucosidase, lipase inhibitory and cytotoxic activities of tuber extracts of Kedrostis africana (L.) Cogn. Heliyon 2018, 4, e00810. [Google Scholar] [CrossRef]
- Hatware, K.V.; Sharma, S.; Patil, K.; Shete, M.; Karri, S.; Gupta, G. Evidence for gastroprotective, anti-inflammatory and antioxidant potential of methanolic extract of Cordia dichotoma leaves on indomethacin and stress-induced gastric lesions in Wistar rats. Biomed. Pharmacother. 2018, 103, 317–325. [Google Scholar] [CrossRef]
- Fennell, C.W.; Lindsey, K.L.; McGaw, L.J.; Sparg, S.G.; Stafford, G.I.; Elgorashi, E.E.; Grace, O.M.; Van Staden, J. Assessing African medicinal plants for efficacy and safety: Pharmacological screening and toxicology. J. Ethnopharmacol. 2004, 94, 205–217. [Google Scholar] [CrossRef] [PubMed]
- Raimondo, D.; Van Staden, L.; Foden, W.; Victor, J.E.; Helme, N.A.; Turner, R.C.; Kamundi, D.A.; Manyama, P.A. Red List of South African Plants. Strelitzia 2009, 95, 186. [Google Scholar]
- Robinson, H.; Funk, V.A. Gymnanthemum koekemoerae (Compositae, Vernonieae), a new species from South Africa. PhytoKeys 2014. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.S. Zulu Medicinal Plants: An Inventory by A. Hutchings with A. H. Scott, G. Lewis, and A. B. Cunningham (University of Zululand). University of Natal Press, Pietermaritzburg. 1996. xiv + 450 pp. 21 × 29.5 cm. $133.00. ISBN 0-86980-893-1. J. Nat. Prod. 1997, 60, 955. [Google Scholar] [CrossRef]
- Dold, A.P.; Cocks, M.L. Traditional veterinary medicine in the Alice district of the Eastern Cape Province, South Africa. S. Afr. J. Sci. 2001, 97, 375–379. [Google Scholar]
- Afolayan, A.J.; Mbaebie, B.O. Ethnobotanical study of medicinal plants used as anti-obesity remedies in Nkonkobe Municipality of South Africa. Pharmacogn. J. 2010, 2, 368–373. [Google Scholar] [CrossRef]
- Unuofin, J.O.; Otunola, G.A.; Afolayan, A.J. Essential Oil Composition, Nutrient and Anti-Nutrient Analysis of Vernonia mespilifolia Less. Res. J. Bot. 2017, 12, 38–45. [Google Scholar]
- McGaw, L.J.; Lall, N.; Meyer, J.J.M.; Eloff, J.N. The potential of South African plants against Mycobacterium infections. J. Ethnopharmacol. 2008, 119, 482–500. [Google Scholar] [CrossRef]
- Mokoka, T.A.; Xolani, P.K.; Zimmermann, S.; Hata, Y.; Adams, M.; Kaiser, M.; Moodley, N.; Maharaj, V.; Koorbanally, N.A.; Hamburger, M.; et al. Antiprotozoal screening of 60 South African plants, and the identification of the antitrypanosomal germacranolides schkuhrin I and II. Planta Med. 2013, 79, 1380–1384. [Google Scholar] [CrossRef]
- Unuofin, J.O.; Otunola, G.A.; Afolayan, A.J. Toxicity Evaluation of Vernonia mespilifolia Less (A South Africa Medicinal Plant) Using Brine Shrimp. J. Pharmacol. Toxicol. 2017, 12, 103–110. [Google Scholar]
- Unuofin, J.O.; Otunola, G.A.; Afolayan, A.J. Polyphenolic Content, Antioxidant and Antimicrobial Activities of Vernonia mespilifolia Less. Used in Folk Medicine in the Eastern Cape Province, South Africa. J. Evid.-Based Integr. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Akah, P.A.; Ekekwe, R.K. Ethnopharmacology of some Asteraceae family used in Nigerian traditional medicine. Fitoterapia 1995, 66, 351–355. [Google Scholar]
- Erasto, P.; Van De Venter, M.; Roux, S.; Grierson, D.S.; Afolayan, A.J. Effect of leaf extracts of Vernonia amygdalina on glucose utilization in changliver, C2C12 muscle and 3T3-L1 cells. Pharm. Biol. 2009, 47, 175–181. [Google Scholar] [CrossRef]
- Fatima, S.S.; Rajasekhar, M.D.; Kumar, K.V.; Kumar, M.T.; Babu, K.R.; Rao, C.A. Antidiabetic and antihyperlipidemic activity of ethyl acetate:isopropanol (1:1) fraction of Vernonia anthelmintica seeds in streptozotocin induced diabetic rats. Food Chem. Toxicol. 2010, 48, 495–501. [Google Scholar] [CrossRef]
- Nafiu, M.O.; Hamid, A.A.; Muritala, H.F.; Adeyemi, S.B. Chapter 7—Preparation, standardization, and quality control of medicinal plants in Africa. In Medicinal Spices and Vegetables from Africa; Kuete, V., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 171–204. [Google Scholar]
- Marrelli, M.; Loizzo, M.R.; Nicoletti, M.; Menichini, F.; Conforti, F. Inhibition of Key Enzymes Linked to Obesity by Preparations from Mediterranean Dietary Plants: Effects on α-Amylase and Pancreatic Lipase Activities. Plant Foods Hum. Nutr. 2013, 68, 340–346. [Google Scholar] [CrossRef]
- Balfour, J.A.; McTavish, D. Acarbose. Drugs 1993, 46, 1025–1054. [Google Scholar] [CrossRef]
- Ademiluyi, A.O.; Oboh, G. Aqueous extracts of roselle (Hibiscus sabdariffa Linn) varieties inhibits α-amylase and α-glucosidase activities In Vitro. J. Med. Food 2013, 16, 88–93. [Google Scholar] [CrossRef]
- Wagner, A.E.; Piegholdt, S.; Rabe, D.; Baenas, N.; Schloesser, A.; Eggersdorfer, M.; Stocker, A.; Rimbach, G. Epigallocatechin gallate affects glucose metabolism and increases fitness and lifespan in Drosophila melanogaster. Oncotarget 2015, 6, 30568. [Google Scholar] [CrossRef]
- Yan, W.; Shao, Y.; Liu, B. A Novel Synthesis Method of Lipophilic EGCG Palmitate and evaluation for Its alpha amylase and alpha glucosidase inhibtory potential. J. Food Sci. Technol. 2019. [Google Scholar] [CrossRef]
- Podsȩdek, A.; Majewska, I.; Redzynia, M.; Sosnowska, D.; Koziołkiewicz, M. In Vitro inhibitory effect on digestive enzymes and antioxidant potential of commonly consumed fruits. J. Agric. Food Chem. 2014, 62, 4610–4617. [Google Scholar] [CrossRef]
- Kajaria, D.; Tiwari, S.; Ranjana, J.; Tripathi, Y. In-Vitro α amylase and glycosidase inhibitory effect of ethanolic extract of antiasthmatic drug—Shirishadi. J. Adv. Pharm. Technol. Res. 2013, 4, 206. [Google Scholar] [CrossRef] [PubMed]
- Nagmoti, D.M.; Juvekar, A. In Vitro inhibitory effects of Pithecellobium dulce (Roxb.) Benth. seeds on intestinal α-glucosidase and pancreatic α-amylase. J. Biochem. Technol. 2013, 4, 616–621. [Google Scholar]
- Birari, R.B.; Bhutani, K.K. Pancreatic lipase inhibitors from natural sources: Unexplored potential. Drug Discov. Today 2007, 12, 879–889. [Google Scholar] [CrossRef] [PubMed]
- Golay, A.; Ybarra, J. Link between obesity and type 2 diabetes. Best Pract. Res. Clin. Endocrinol. Metab. 2005, 19, 649–663. [Google Scholar] [CrossRef]
- Moreno, D.A.; Ripoll, C.; Ilic, N.; Poulev, A.; Aubin, C.; Raskin, I. Inhibition of lipid metabolic enzymes using Mangifera indica extracts. J. Food Agric. Environ. 2006, 4, 21. [Google Scholar]
- Sugiyama, H.; Akazome, Y.; Shoji, T.; Yamaguchi, A.; Yasue, M.; Kanda, T.; Ohtake, Y. Oligomeric procyanidins in apple polyphenol are main active components for inhibition of pancreatic lipase and triglyceride absorption. J. Agric. Food Chem. 2007, 55, 4604–4609. [Google Scholar] [CrossRef]
- Tan, Y.; Chang, S.K.C.; Zhang, Y. Comparison of α-amylase, α-glucosidase and lipase inhibitory activity of the phenolic substances in two black legumes of different genera. Food Chem. 2017, 214, 259–268. [Google Scholar] [CrossRef]
- Kuete, V.; Seo, E.J.; Krusche, B.; Oswald, M.; Wiench, B.; Schröder, S.; Greten, H.J.; Lee, I.S.; Efferth, T. Cytotoxicity and pharmacogenomics of medicinal plants from traditional Korean medicine. Evid.-Based Complement. Altern. Med. 2013. [Google Scholar] [CrossRef]
- McGaw, L.J.; van der Merwe, D.; Eloff, J.N. In Vitro anthelmintic, antibacterial and cytotoxic effects of extracts from plants used in South African ethnoveterinary medicine. Vet. J. 2007, 173, 366–372. [Google Scholar] [CrossRef]
- Nchu, F.; Githiori, J.B.; McGaw, L.J.; Eloff, J.N. Anthelmintic and cytotoxic activities of extracts of Markhamia obtusifolia Sprague (Bignoniaceae). Vet. Parasitol. 2011, 183, 184–188. [Google Scholar] [CrossRef]
- Kudumela, R.G.; McGaw, L.J.; Masoko, P. Antibacterial interactions, anti-inflammatory and cytotoxic effects of four medicinal plant species. BMC Complement. Altern. Med. 2018, 18, 199. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Castro, A.J.; Domínguez, F.; Ruiz-Padilla, A.J.; Campos-Xolalpa, N.; Zapata-Morales, J.R.; Carranza-Alvarez, C.; Maldonado-Miranda, J.J. Medicinal Plants from North and Central America and the Caribbean Considered Toxic for Humans: The Other Side of the Coin. Evid.-Based Complement. Altern. Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Shai, L.J.; Masoko, P.; Mokgotho, M.P.; Magano, S.R.; Mogale, A.M.; Boaduo, N.; Eloff, J.N. Yeast alpha glucosidase inhibitory and antioxidant activities of six medicinal plants collected in Phalaborwa, South Africa. S. Afr. J. Bot. 2010, 76, 465–470. [Google Scholar] [CrossRef]
- Sun, J.; Zhang, F.; Yang, M.; Zhang, J.; Chen, L.; Zhan, R.; Li, L.; Chen, Y. Isolation of α-glucosidase inhibitors including a new flavonol glycoside from Dendrobium devonianum. Nat. Prod. Res. 2014, 28, 1900–1905. [Google Scholar] [CrossRef]
- Kim, Y.S.; Lee, Y.M.; Kim, H.; Kim, J.; Jang, D.S.; Kim, J.H.; Kim, J.S. Anti-obesity effect of Morus bombycis root extract: Anti-lipase activity and lipolytic effect. J. Ethnopharmacol. 2010, 130, 621–624. [Google Scholar] [CrossRef]
| Name of Extract | IC50 Values (μg/mL) | ||
|---|---|---|---|
| α-Amylase | α-Glucosidase | Lipase | |
| Aqueous | 847.49 ± 1.69 | 450.88 ± 2.84 | 1028.18 ± 3.27 |
| Ethanolic | 781.72 ± 2.58 | 637.35 ± 1.89 | 331.16 ± 1.85 |
| Name of Extract | Cytotoxicity IC50 (μg/mL) |
|---|---|
| Aqueous | >200 |
| Ethanolic | 149.12 ± 3.82 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Unuofin, J.O.; Otunola, G.A.; Afolayan, A.J. Inhibition of Key Enzymes Linked to Obesity and Cytotoxic Activities of Whole Plant Extracts of Vernonia mesplilfolia Less. Processes 2019, 7, 841. https://doi.org/10.3390/pr7110841
Unuofin JO, Otunola GA, Afolayan AJ. Inhibition of Key Enzymes Linked to Obesity and Cytotoxic Activities of Whole Plant Extracts of Vernonia mesplilfolia Less. Processes. 2019; 7(11):841. https://doi.org/10.3390/pr7110841
Chicago/Turabian StyleUnuofin, Jeremiah Oshiomame, Gloria Aderonke Otunola, and Anthony Jide Afolayan. 2019. "Inhibition of Key Enzymes Linked to Obesity and Cytotoxic Activities of Whole Plant Extracts of Vernonia mesplilfolia Less" Processes 7, no. 11: 841. https://doi.org/10.3390/pr7110841
APA StyleUnuofin, J. O., Otunola, G. A., & Afolayan, A. J. (2019). Inhibition of Key Enzymes Linked to Obesity and Cytotoxic Activities of Whole Plant Extracts of Vernonia mesplilfolia Less. Processes, 7(11), 841. https://doi.org/10.3390/pr7110841





