Characterizing the Suitability of Granular Fe0 for the Water Treatment Industry
Abstract
1. Introduction
2. Materials and Methods
2.1. Solutions
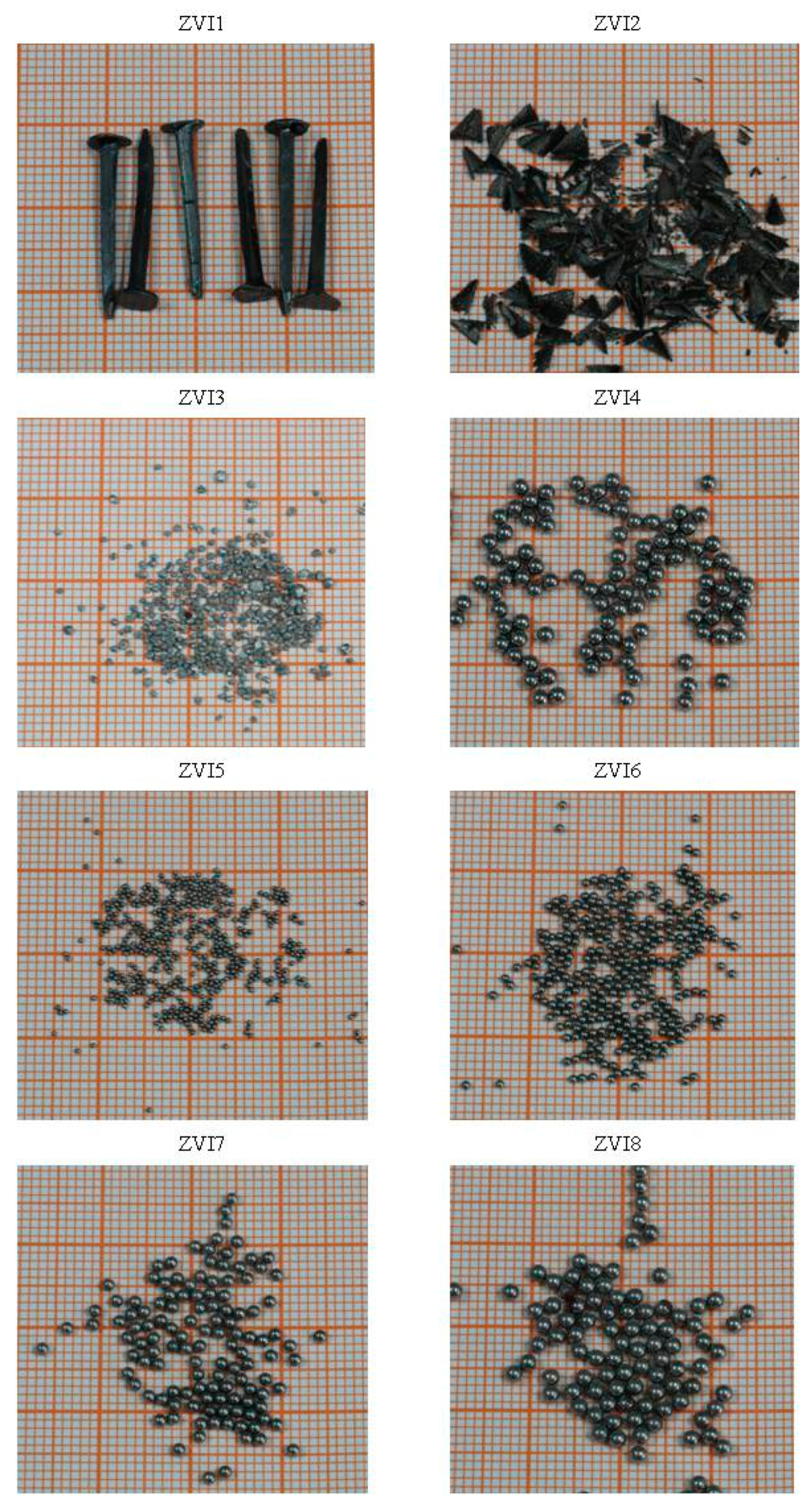
2.2. Fe0 Materials
2.3. Experimental Procedure
2.4. Analytical Method
2.5. Expression of Results
3. Results and Discussion
3.1. Suitability of the Experimental Protocol
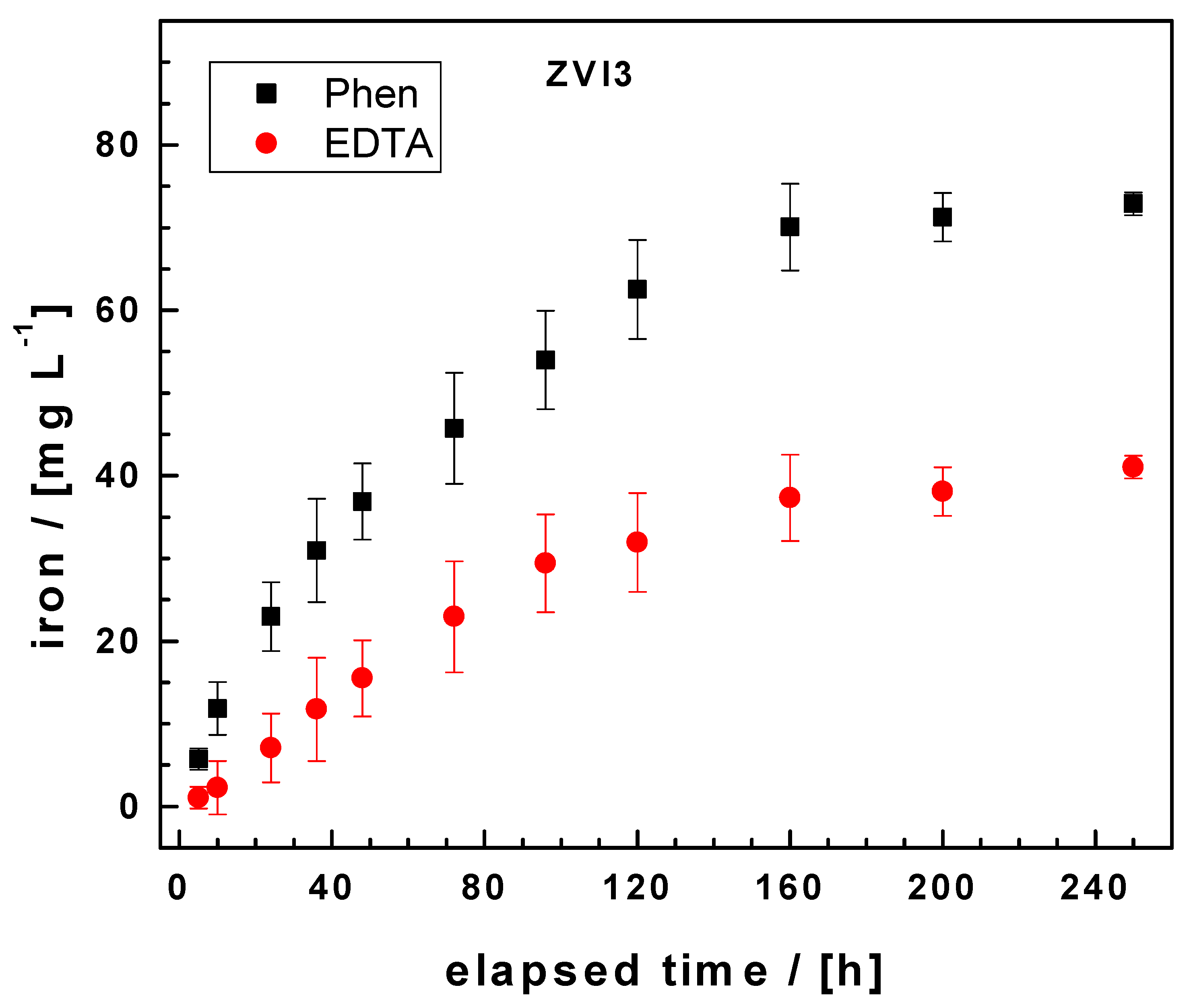
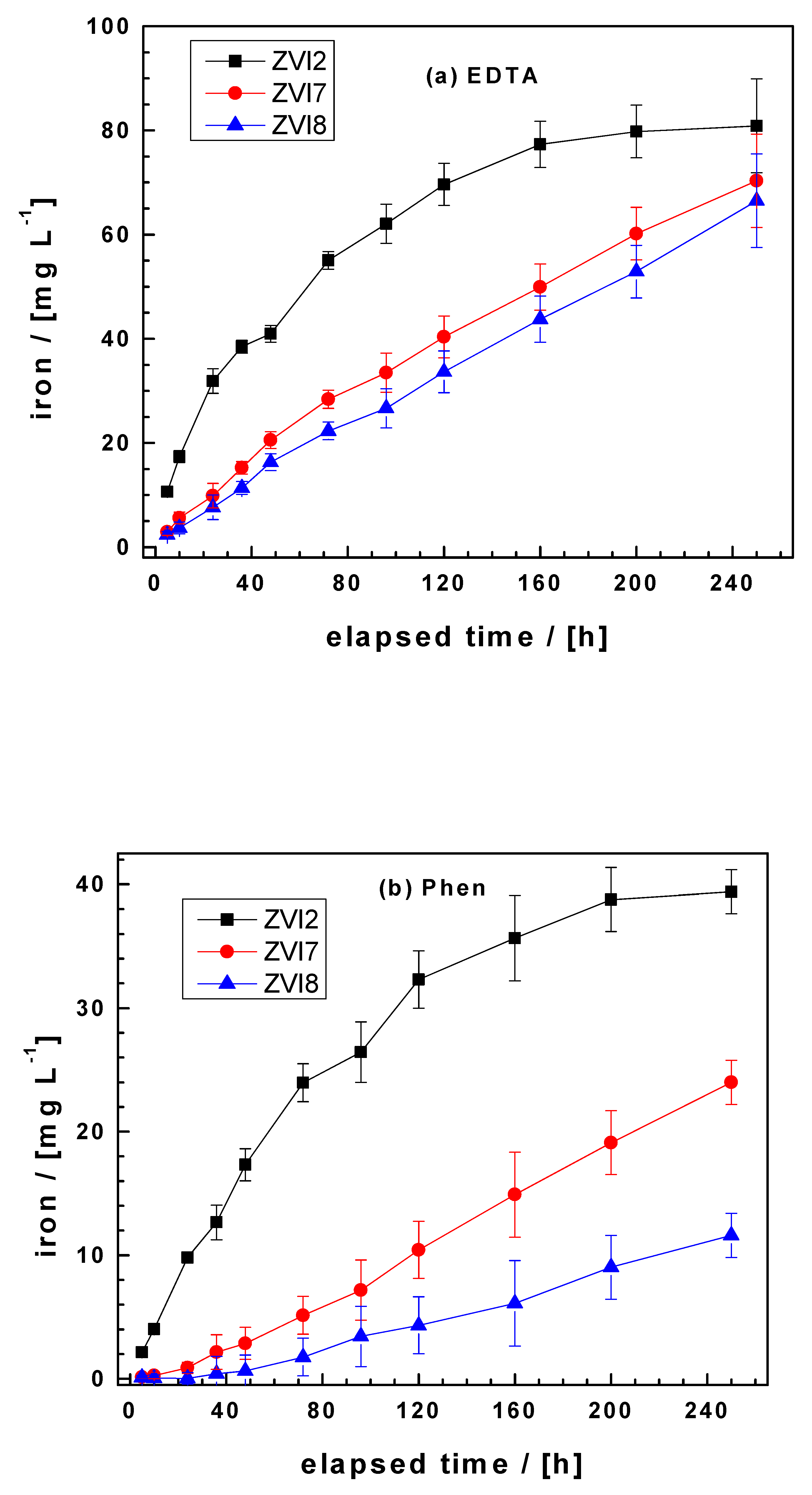
3.2. The EDTA Test
3.3. The Phen Test
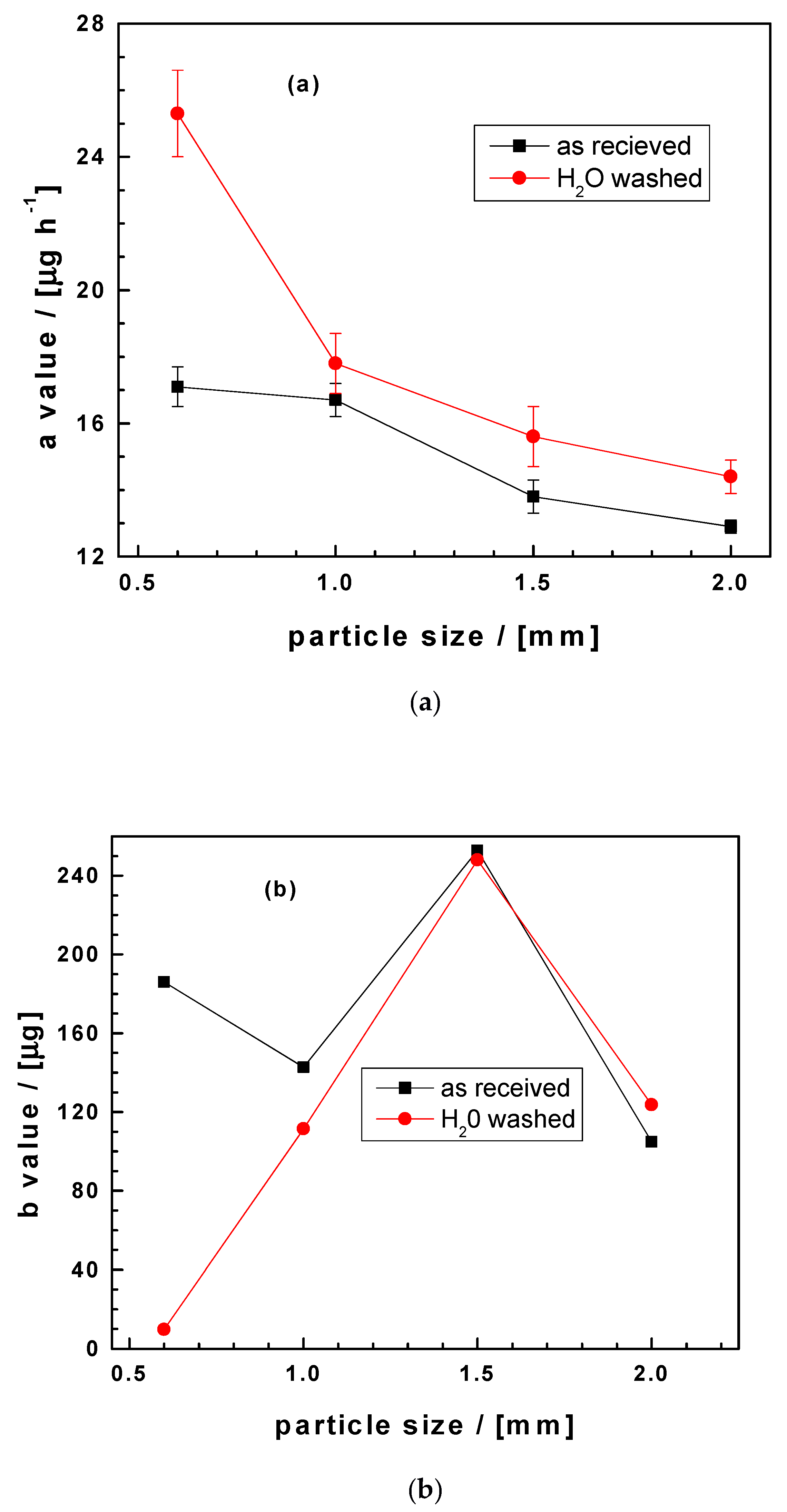
3.4. Method Validation
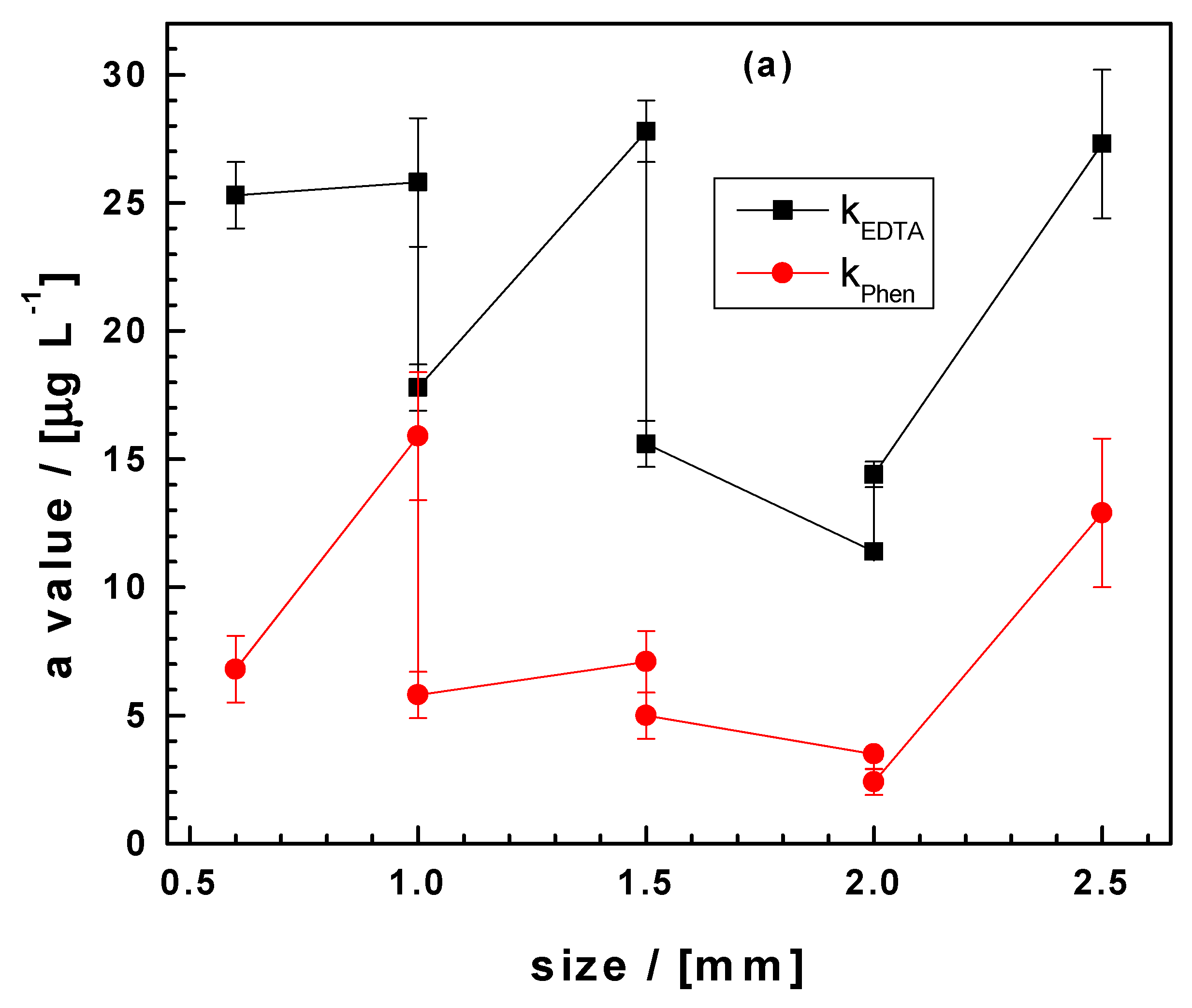
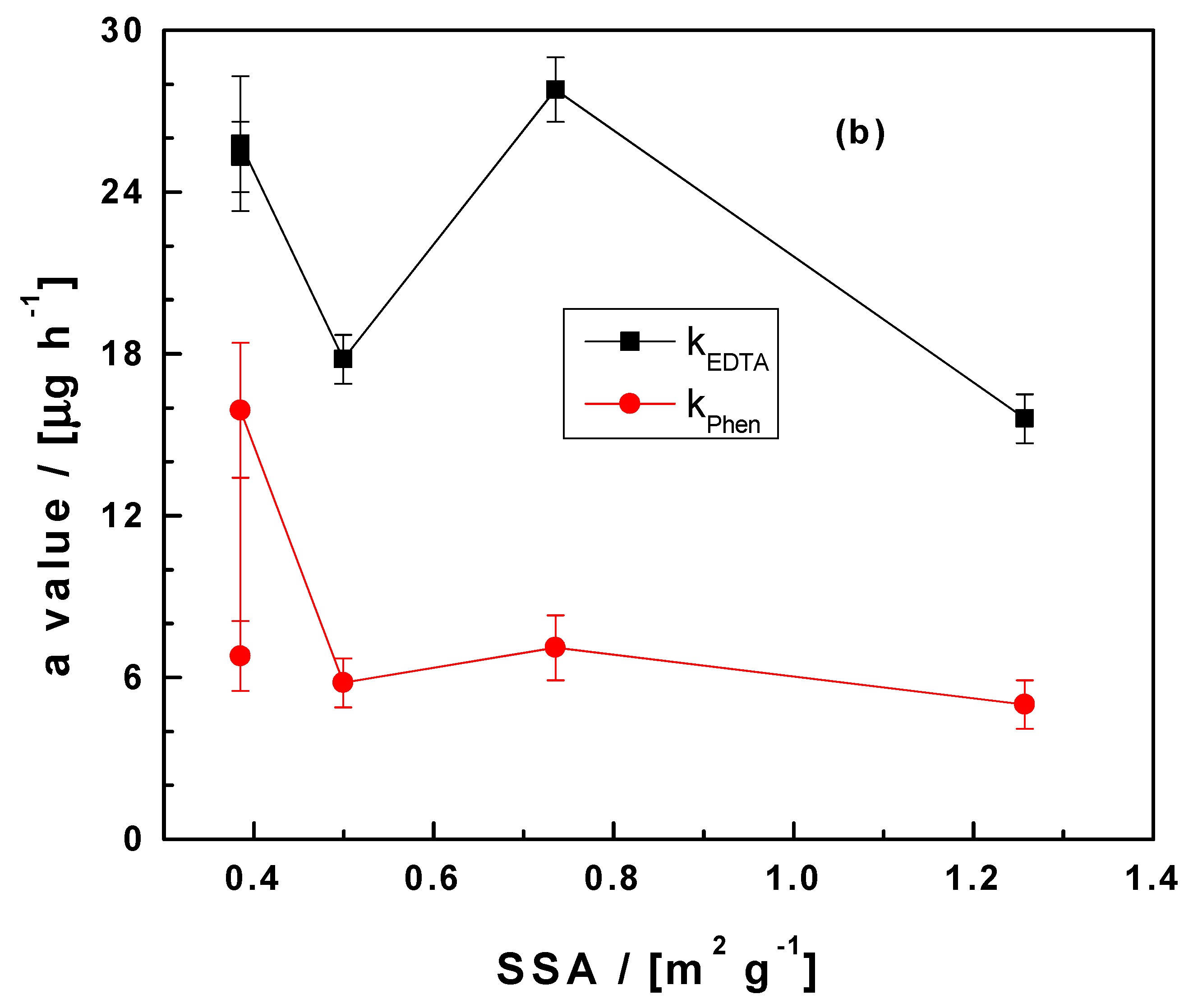
3.5. Significance of the Results
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O´Hannesin, S.F.; Gillham, R.W. Long-term performance of an in situ “iron wall” for remediation of VOCs. Ground Water 1998, 36, 164–170. [Google Scholar] [CrossRef]
- Henderson, A.D.; Demond, A.H. Long-term performance of zero-valent iron permeable reactive barriers: A critical review. Environ. Eng. Sci. 2007, 24, 401–423. [Google Scholar] [CrossRef]
- Li, X.-Q.; Zhang, W.-X. Sequestration of metal cations with zerovalent iron nanoparticles: A study with high resolution X-ray photoelectron spectroscopy (HR-XPS). J. Phys. Chem. C 2007, 111, 6939–6946. [Google Scholar] [CrossRef]
- Guan, X.; Sun, Y.; Qin, H.; Li, J.; Lo, I.M.C.; He, D.; Dong, H. The limitations of applying zero-valent iron technology in contaminants sequestration and the corresponding countermeasures: The development in zero-valent iron technology in the last two decades (1994–2014). Water Res. 2015, 75, 224–248. [Google Scholar] [CrossRef] [PubMed]
- Noubactep, C. Metallic iron for environmental remediation: A review of reviews. Water Res. 2015, 85, 114–123. [Google Scholar] [CrossRef]
- Li, J.; Dou, X.; Qin, H.; Sun, Y.; Yin, D.; Guan, X. Characterization methods of zerovalent iron for water treatment and remediation. Water Res. 2019, 148, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Pyae, H.A.; Aye, W.W.; Yossapol, C. Investigation and characterization of iron powders for zero-valent iron (Fe0) in synchrotron radiations. Int. J. Eng. Adv. Technol. 2019, 8, 181–193. [Google Scholar]
- Devonshire, E. The purification of water by means of metallic iron. J. Frankl. Inst. 1890, 129, 449–461. [Google Scholar] [CrossRef]
- Ghauch, A. Iron-based metallic systems: An excellent choice for sustainable water treatment. Freib. Online Geosci. 2015, 32, 1–80. [Google Scholar]
- Banerji, T.; Chaudhari, S. A cost-effective technology for arsenic removal: Case study of zerovalent iron-based IIT Bombay arsenic filter in West Bengal. In Water and Sanitation in the New Millennium; Nath, K., Sharma, V., Eds.; Springer: New Delhi, India, 2017. [Google Scholar]
- Gheju, M. Progress in understanding the mechanism of CrVI Removal in Fe0-based filtration systems. Water 2018, 10, 651. [Google Scholar] [CrossRef]
- Vollprecht, D.; Krois, L.-M.; Sedlazeck, K.P.; Müller, P.; Mischitz, R.; Olbrich, T.; Pomberger, R. Removal of critical metals from waste water by zero-valent iron. J. Clean. Prod. 2018, 208, 1409–1420. [Google Scholar] [CrossRef]
- Hu, R.; Gwenzi, W.; Sipowo-Tala, V.R.; Noubactep, C. Water treatment using metallic iron: A tutorial review. Processes 2019, 7, 622. [Google Scholar] [CrossRef]
- Hu, R.; Ndé-Tchoupé, A.I.; Lufingo, M.; Xiao, M.; Nassi, A.; Noubactep, C.; Njau, K.N. The impact of selected pre-treatment procedures on iron dissolution from metallic iron specimens used in water treatment. Sustainability 2019, 11, 671. [Google Scholar] [CrossRef]
- Nanseu-Njiki, C.P.; Gwenzi, W.; Pengou, M.; Rahman, M.A.; Noubactep, C. Fe0/H2O filtration systems for decentralized safe drinking water: Where to from here? Water 2019, 11, 429. [Google Scholar] [CrossRef]
- Btatkeu-K, B.D.; Miyajima, K.; Noubactep, C.; Caré, S. Testing the suitability of metallic iron for environmental remediation: Discoloration of methylene blue in column studies. Chem. Eng. J. 2013, 215–216, 959–968. [Google Scholar] [CrossRef]
- Birke, V.; Schuett, C.; Burmeier, H.; Friedrich, H.-J. Impact of trace elements and impurities in technical zero-valent iron brands on reductive dechlorination of chlorinated ethenes in groundwater. In Permeable Reactive Barrier Sustainable Groundwater Remediation; Naidu, R., Birke, V., Eds.; CRC Press: Boca Raton, FL, USA, 2015; pp. 87–98. ISBN 978-1-4822-2447-4. [Google Scholar]
- Miyajima, K.; Noubactep, C. Characterizing the impact of sand addition on the efficiency of granular iron for water treatment. Chem. Eng. J. 2015, 262, 891–896. [Google Scholar] [CrossRef]
- Kim, H.; Yang, H.; Kim, J. Standardization of the reducing power of zerovalent iron using iodine. Environ. Lett. 2014, 49, 514–523. [Google Scholar]
- Li, S.; Ding, Y.; Wang, W.; Lei, H. A facile method for determining the Fe(0) content and reactivity of zero valent iron. Anal. Methods 2016, 8, 1239–1248. [Google Scholar] [CrossRef]
- Gillham, R.W.; O’Hannesin, S.F. Enhanced degradation of halogenated aliphatics by zero-valent iron. Ground Water 1994, 32, 958–967. [Google Scholar] [CrossRef]
- Matheson, L.J.; Tratnyek, P.G. Reductive dehalogenation of chlorinated methanes by iron metal. Environ. Sci. Technol. 1994, 28, 2045–2053. [Google Scholar] [CrossRef]
- Lauderdale, R.A.; Emmons, A.H. A method for decontaminating small volumes of radioactive water. J. Am. Water Works Assoc. 1951, 43, 327–331. [Google Scholar] [CrossRef]
- Gould, J.P. The kinetics of hexavalent chromium reduction by metallic iron. Water Res. 1982, 16, 871–877. [Google Scholar] [CrossRef]
- Khudenko, B.M. Feasibility evaluation of a novel method for destruction of organics. Water Sci. Technol. 1991, 23, 1873–1881. [Google Scholar] [CrossRef]
- James, B.R.; Rabenhorst, M.C.; Frigon, G.A. Phosphorus sorption by peat and sand amended with iron oxides or steel wool. Water Environ. Res. 1992, 64, 699–705. [Google Scholar] [CrossRef]
- Bojic, A.; Purenovic, M.; Kocic, B.; Perovic, J.; Ursic-Jankovic, J.; Bojic, D. The inactivation of Escherichia coli by microalloyed aluminium based composite. Facta Universitatis 2001, 2, 115–124. [Google Scholar]
- Bojic, A.; Purenovic, M.; Bojic, D. Removal of chromium(VI) from water by micro-alloyed aluminium based composite in flow conditions. Water SA 2004, 30, 353–359. [Google Scholar] [CrossRef]
- Mwakabona, H.T.; Ndé-Tchoupé, A.I.; Njau, K.N.; Noubactep, C.; Wydra, K.D. Metallic iron for safe drinking water provision: Considering a lost knowledge. Water Res. 2017, 117, 127–142. [Google Scholar] [CrossRef]
- Xi, Y.; Luo, Y.; Zou, J.; Li, J.; Liao, T.; Zhang, L.; Wang, C.; Li, X.; Lin, G. Kinetics of arsenic removal in waste acid by the combination of CuSO4 and zero-valent iron. Processes 2019, 7, 401. [Google Scholar] [CrossRef]
- Miehr, R.; Tratnyek, G.P.; Bandstra, Z.J.; Scherer, M.M.; Alowitz, J.M.; Bylaska, J.E. Diversity of contaminant reduction reactions by zerovalent iron: Role of the reductate. Environ. Sci. Technol. 2004, 38, 139–147. [Google Scholar] [CrossRef]
- Xu, J.; Wang, Y.; Weng, C.; Bai, W.; Jiao, Y.; Kaegi, R.; Lowry, G.V. Reactivity, selectivity, and long-Term performance of sulfidized nanoscale zerovalent iron with different properties. Environ. Sci. Technol. 2019, 53, 5936–5945. [Google Scholar] [CrossRef]
- Scott, T.B.; Popescu, I.C.; Crane, R.A.; Noubactep, C. Nano-scale metallic iron for the treatment of solutions containing multiple inorganic contaminants. J. Hazard. Mater. 2011, 186, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Lowry, G.V. Progress towards standardized and validated characterizations for measuring physicochemical properties of manufactured nanomaterials relevant to nano health and safety risks. NanoImpact 2018, 9, 14–30. [Google Scholar] [CrossRef]
- Hildebrant, B. Characterizing the reactivity of commercial steel wool for water treatment. Freib. Online Geosci. 2018, 53, 1–60. [Google Scholar]
- Lufingo, M. Investigation of Metallic Iron for Water Defluoridation. Master’s Thesis, NM-AIST, Arusha, Tanzania, 2019. [Google Scholar]
- Naseri, E.; Ndé-Tchoupé, A.I.; Mwakabona, H.T.; Nanseu-Njiki, C.P.; Noubactep, C.; Njau, K.N.; Wydra, K.D. Making Fe0-Based filters a universal solution for safe drinkingwater provision. Sustainability 2017, 9, 1224. [Google Scholar] [CrossRef]
- Weber, E.J. Iron-mediated reductive transformations: Investigation of reaction mechanism. Environ. Sci. Technol. 1996, 30, 716–719. [Google Scholar] [CrossRef]
- Lufingo, M.; Ndé-Tchoupé, A.I.; Hu, R.; Njau, K.N.; Noubactep, C. A novel and facile method to characterize the suitability of metallic iron for water treatment. Water 2019. submitted. [Google Scholar]
- Noubactep, C.; Meinrath, G.; Dietrich, P.; Sauter, M.; Merkel, B. Testing the suitability of zerovalent iron materials for reactive Walls. Environ. Chem. 2005, 2, 71–76. [Google Scholar] [CrossRef]
- Pierce, E.M.; Wellman, D.M.; Lodge, A.M.; Rodriguez, E.A. Experimental determination of the dissolution kinetics of zero-valent iron in the presence of organic complexants. Environ. Chem. 2007, 4, 260–270. [Google Scholar] [CrossRef]
- Noubactep, C.; Licha, T.; Scott, T.B.; Fall, M.; Sauter, M. Exploring the influence of operational parameters on the reactivity of elemental iron materials. J. Hazard. Mater. 2009, 172, 943–951. [Google Scholar] [CrossRef] [PubMed]
- Noubactep, C. Characterizing the reactivity of metallic iron in Fe0/EDTA/H2O systems with column experiments. Chem. Eng. J. 2010, 162, 656–661. [Google Scholar] [CrossRef]
- Martell, A.E.; Motekaitis, R.J.; Chen, D.; Hancock, R.D.; McManus, D. Selection of new Fe(lll)/Fe(ll) chelating agents as catalysts for the oxidation of hydrogen sulfide to sulfur by air. Can. J. Chem. 1996, 74, 1872–1879. [Google Scholar] [CrossRef]
- Ibanez, J.G.; Gonzalez, I.; Cardenas, M.A. The effect of complex formation upon the redox potentials of metallic ions: Cyclic voltammetry experiments. J. Chem. Educ. 1998, 65, 173–175. [Google Scholar] [CrossRef]
- Wen, Y.H.; Zhang, H.M.; Qian, P.; Zhou, H.T.; Zhao, P.; Yi, B.L.; Yang, Y.S. Studies on iron (Fe3+/Fe2+)-complex/bromine (Br2/Br−) Redox flow cell in sodium acetate solution. J. Electrochem. Soc. 2006, 153, A929–A934. [Google Scholar] [CrossRef]
- Rizvi, M.A. Complexation modulated redox behavior of transition metal systems. Russ. J. Gen. Chem. 2015, 85, 959–973. [Google Scholar] [CrossRef]
- Reardon, J.E. Anaerobic corrosion of granular iron: Measurement and interpretation of hydrogen evolution rates. Environ. Sci. Technol. 1995, 29, 2936–2945. [Google Scholar] [CrossRef]
- Reardon, E.J. Zerovalent irons: Styles of corrosion and inorganic control on hydrogen pressure buildup. Environ. Sci. Tchnol. 2005, 39, 7311–7317. [Google Scholar] [CrossRef]
- Macé, C.; Desrocher, S.; Gheorghiu, F.; Kane, A.; Pupeza, M.; Cernik, M.; Kvapil, P.; Venkatakrishnan, R.; Zhang, W.-X. Nanotechnology and groundwater remediation: A step forward in technology understanding. Remed. J. 2006, 16, 23–33. [Google Scholar] [CrossRef]
- Johnson, T.L.; Scherer, M.M.; Tratnyek, P.G. Kinetics of halogenated organic compound degradation by iron metal. Environ. Sci. Technol. 1996, 30, 2634–2640. [Google Scholar] [CrossRef]
- Sikora, E.; Macdonald, D.D. The passivity of iron in the presence of ethylenediaminetetraacetic acid I. General electrochemical behavior. J. Electrochem. Soc. 2000, 147, 4087–4092. [Google Scholar] [CrossRef]
- Nesic, S. Key issues related to modelling of internal corrosion of oil and gas pipelines—A review. Corros. Sci. 2007, 49, 4308–4338. [Google Scholar] [CrossRef]
- Lazzari, L. General aspects of corrosion, Chapter 9.1. In Encyclopedia of Hydrocarbons; Istituto Enciclopedia Italiana: Rome, Italy, 2008; Volume V. [Google Scholar]
- Kosmulski, M. Isoelectric points and points of zero charge of metal (hydr)oxides: 50 years after Parks’ review. Adv. Colloid Interface Sci. 2016, 238, 1–61. [Google Scholar] [CrossRef]
- Noubactep, C. An analysis of the evolution of reactive species in Fe0/H2O systems. J. Hazard. Mater. 2009, 168, 1626–1631. [Google Scholar] [CrossRef] [PubMed]
- Bedia, G.; Peñas-Garzón, M.; Gómez-Avilés, A.; Rodriguez, J.J.; Belver, C. A review on the synthesis and characterization of biomass-derived carbons for adsorption of emerging contaminants from water. J. Carbon Res. 2018, 4, 63. [Google Scholar] [CrossRef]
| Test | Anno | Fe0 | Rationale | Comments |
|---|---|---|---|---|
| (-) | (g) | |||
| H2 evolution | 1995 | > 500 | Fe0 corrosion by H2O | too large Fe0 amounts |
| EDTA test | 2005 | 0.1 | Fe0 corrosion by H2O | disturbed by dissolved O2 |
| Tri-iodide (I3-) | 2014 | 0.2 | Fe0 corrosion by iodine | past efforts ignored |
| FeIII | 2016 | Fe0 corrosion by FeIII | I3- test considered | |
| Phen test | 2019 | 0.1 | Fe0 corrosion by H2O | very simple to operate |
| Code | Shape | Size | Color | SSA | Fe | Supplier |
|---|---|---|---|---|---|---|
| (mm) | (m2/g) | (%) | ||||
| ZVI1 | nail | 1.5 / 22 | black | n.s. | n.s. | Pinnacle hardware store |
| ZVI2 | scrap | 0.05–5.0 | black | n.s. | n.s. | Shanghai Institute of Fine Technology |
| ZVI3 | irregular | 0.50–1.5 | grey | n.s. | n.s. | Tongda alloy material factory |
| ZVI4 | spherical | 2.0 | grey | 0.39 | 99.99 | Tongda alloy material factory |
| ZVI5 | spherical | 0.6 | grey | 1.26 | 99.99 | Tongda alloy material factory |
| ZVI6 | spherical | 1.0 | grey | 0.74 | 99.99 | Tongda alloy material factory |
| ZVI7 | spherical | 1.5 | grey | 0.50 | 99.99 | Tongda alloy material factory |
| ZVI8 | spherical | 2.0 | grey | 0.39 | 99.99 | Tongda alloy material factory |
| Fe0 | a | Δa | b | Δb | n | R2 |
|---|---|---|---|---|---|---|
| (μg/h) | (μg/h) | (μg) | (μg) | (-) | (-) | |
| EDTA | ||||||
| ZVI1 | 27.8 | 1.2 | 88.2 | 60.1 | 7 | 0.990 |
| ZVI2 | 27.3 | 2.9 | 697.1 | 149.5 | 7 | 0.936 |
| ZVI3 | 25.8 | 2.5 | 413.7 | 127.6 | 7 | 0.947 |
| ZVI4 | 11.4 | 0.2 | 136.8 | 29.9 | 11 | 0.994 |
| ZVI5 | 17.1 | 0.6 | 186.0 | 60.4 | 7 | 0.989 |
| ZVI6 | 16.7 | 0.5 | 142.7 | 44.1 | 9 | 0.992 |
| ZVI7 | 13.8 | 0.5 | 252.9 | 61.0 | 11 | 0.987 |
| ZVI8 | 12.9 | 0.2 | 104.9 | 24.2 | 11 | 0.990 |
| EDTA, H2O washed | ||||||
| ZVI5 | 25.3 | 1.3 | 9.8 | 67.4 | 7 | 0.984 |
| ZVI6 | 17.8 | 0.9 | 111.4 | 70.2 | 9 | 0.981 |
| ZVI7 | 15.6 | 0.9 | 248.0 | 114.6 | 11 | 0.965 |
| ZVI8 | 14.4 | 0.5 | 123.6 | 61.1 | 11 | 0.990 |
| Phen | ||||||
| ZVI1 | 7.1 | 0.1 | −72.4 | 14.0 | 11 | 0.997 |
| ZVI2 | 12.9 | 0.9 | 138.8 | 55.3 | 8 | 0.970 |
| ZVI3 | 15.9 | 0.3 | −17.5 | 19.5 | 7 | 0.997 |
| ZVI4 | 3.5 | 0.2 | −70.6 | 21.1 | 11 | 0.976 |
| ZVI5 | 6.8 | 0.3 | −37.6 | 30.6 | 11 | 0.986 |
| ZVI6 | 5.8 | 0.2 | −64.5 | 18.2 | 11 | 0.993 |
| ZVI7 | 5.0 | 0.1 | −70.8 | 15.2 | 11 | 0.994 |
| ZVI8 | 2.4 | 0.1 | −55.1 | 14.0 | 11 | 0.978 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, R.; Cui, X.; Xiao, M.; Qiu, P.; Lufingo, M.; Gwenzi, W.; Noubactep, C. Characterizing the Suitability of Granular Fe0 for the Water Treatment Industry. Processes 2019, 7, 652. https://doi.org/10.3390/pr7100652
Hu R, Cui X, Xiao M, Qiu P, Lufingo M, Gwenzi W, Noubactep C. Characterizing the Suitability of Granular Fe0 for the Water Treatment Industry. Processes. 2019; 7(10):652. https://doi.org/10.3390/pr7100652
Chicago/Turabian StyleHu, Rui, Xuesong Cui, Minhui Xiao, Pengxiang Qiu, Mesia Lufingo, Willis Gwenzi, and Chicgoua Noubactep. 2019. "Characterizing the Suitability of Granular Fe0 for the Water Treatment Industry" Processes 7, no. 10: 652. https://doi.org/10.3390/pr7100652
APA StyleHu, R., Cui, X., Xiao, M., Qiu, P., Lufingo, M., Gwenzi, W., & Noubactep, C. (2019). Characterizing the Suitability of Granular Fe0 for the Water Treatment Industry. Processes, 7(10), 652. https://doi.org/10.3390/pr7100652








