Geotechnical Properties of Effluent-Contaminated Cohesive Soils and Their Stabilization Using Industrial By-Products
Abstract
:1. Introduction
2. Materials and Methods
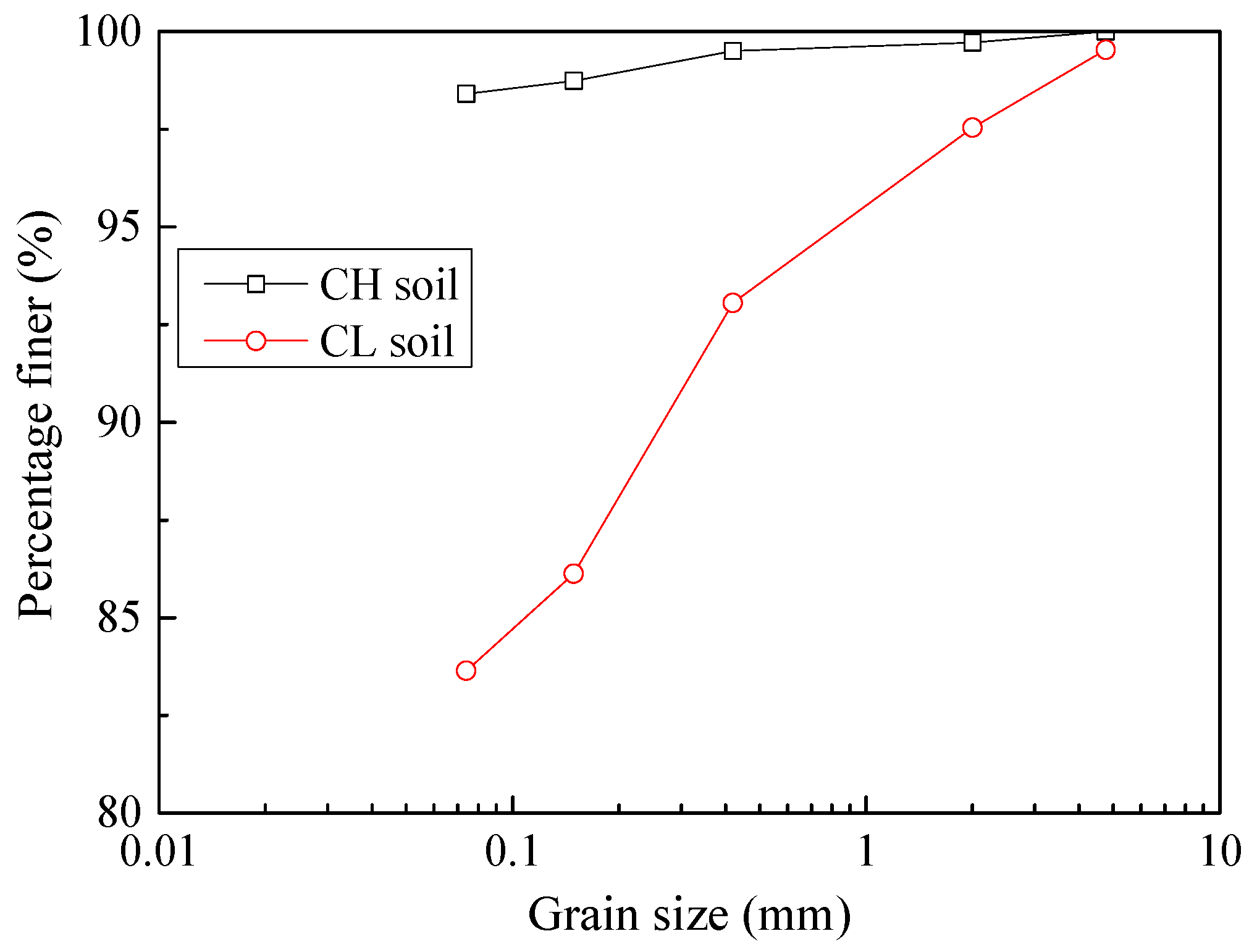
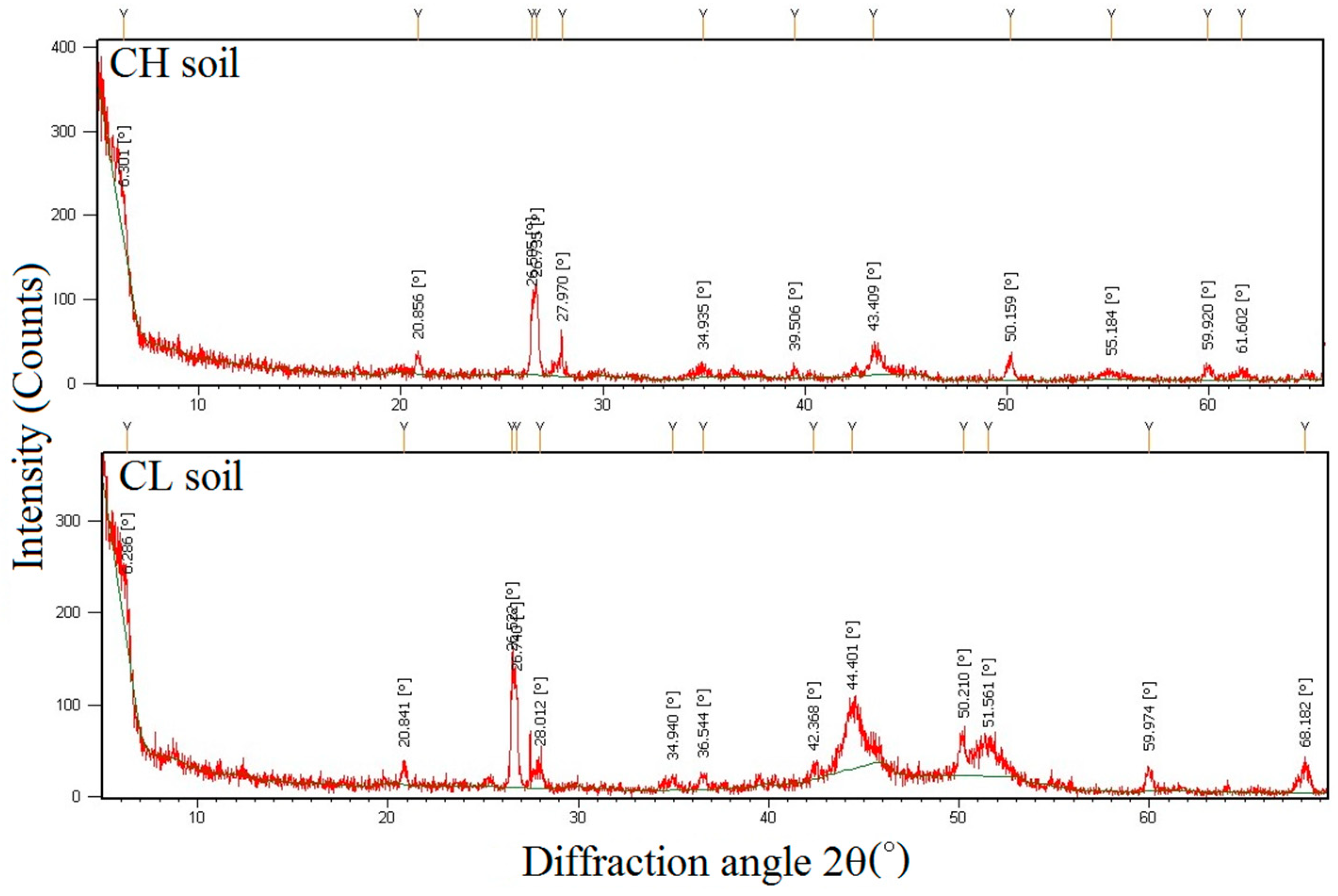
2.1. Soil Samples
2.2. Industrial Effluents
2.3. Industrial By-Products
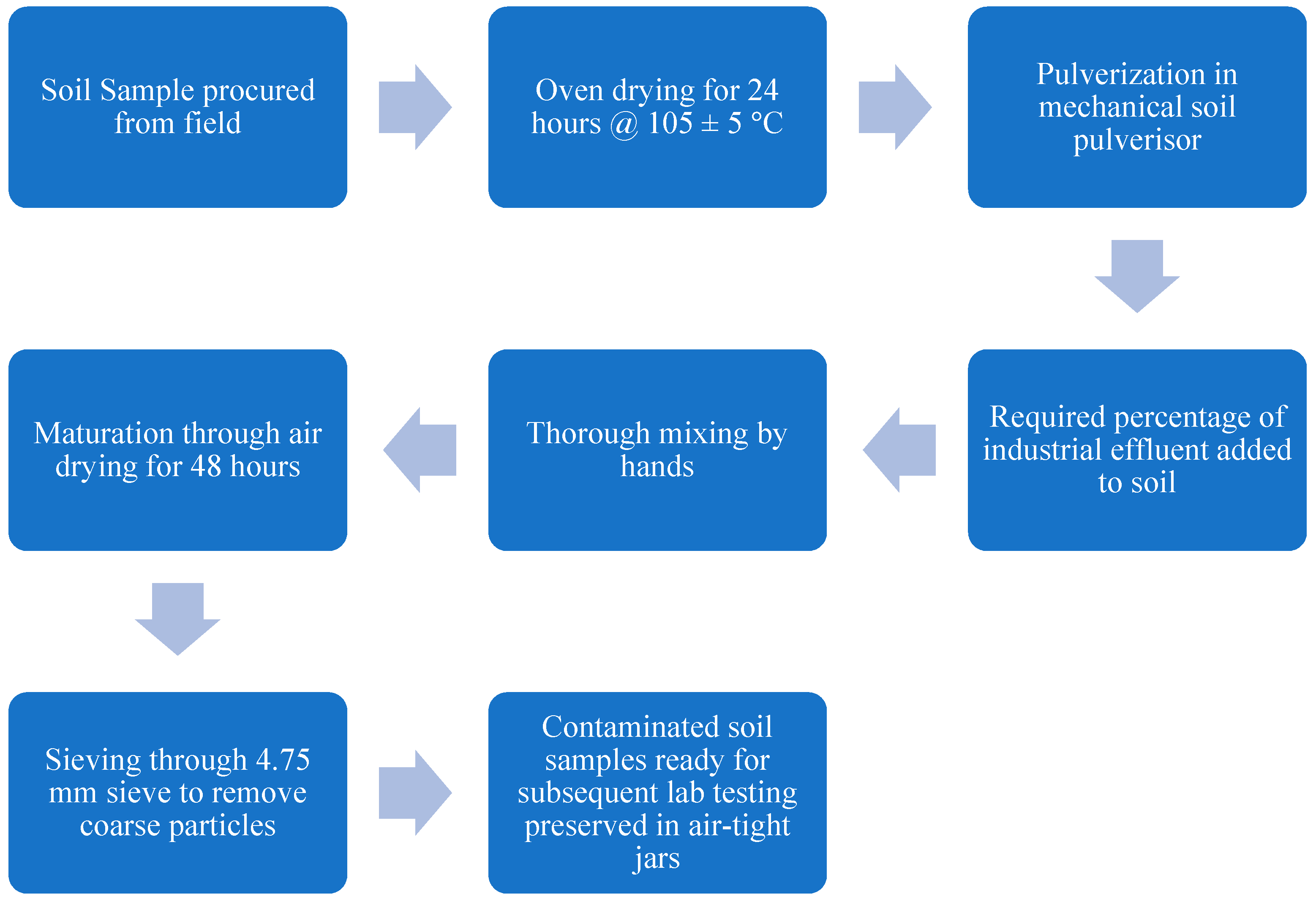
2.4. Testing Scheme
3. Results and Discussion
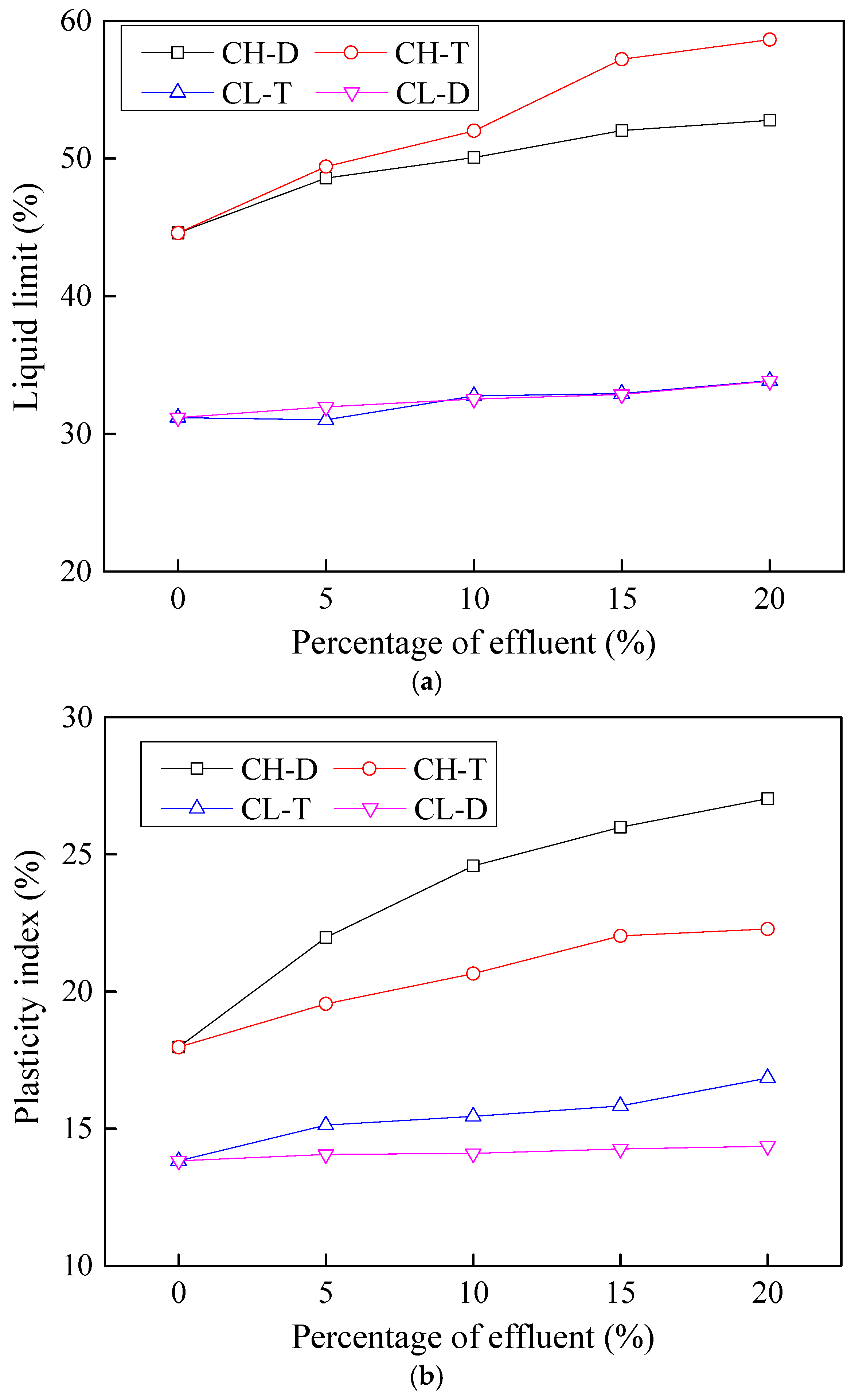
3.1. Effects of Contamination on Atterberg Limits
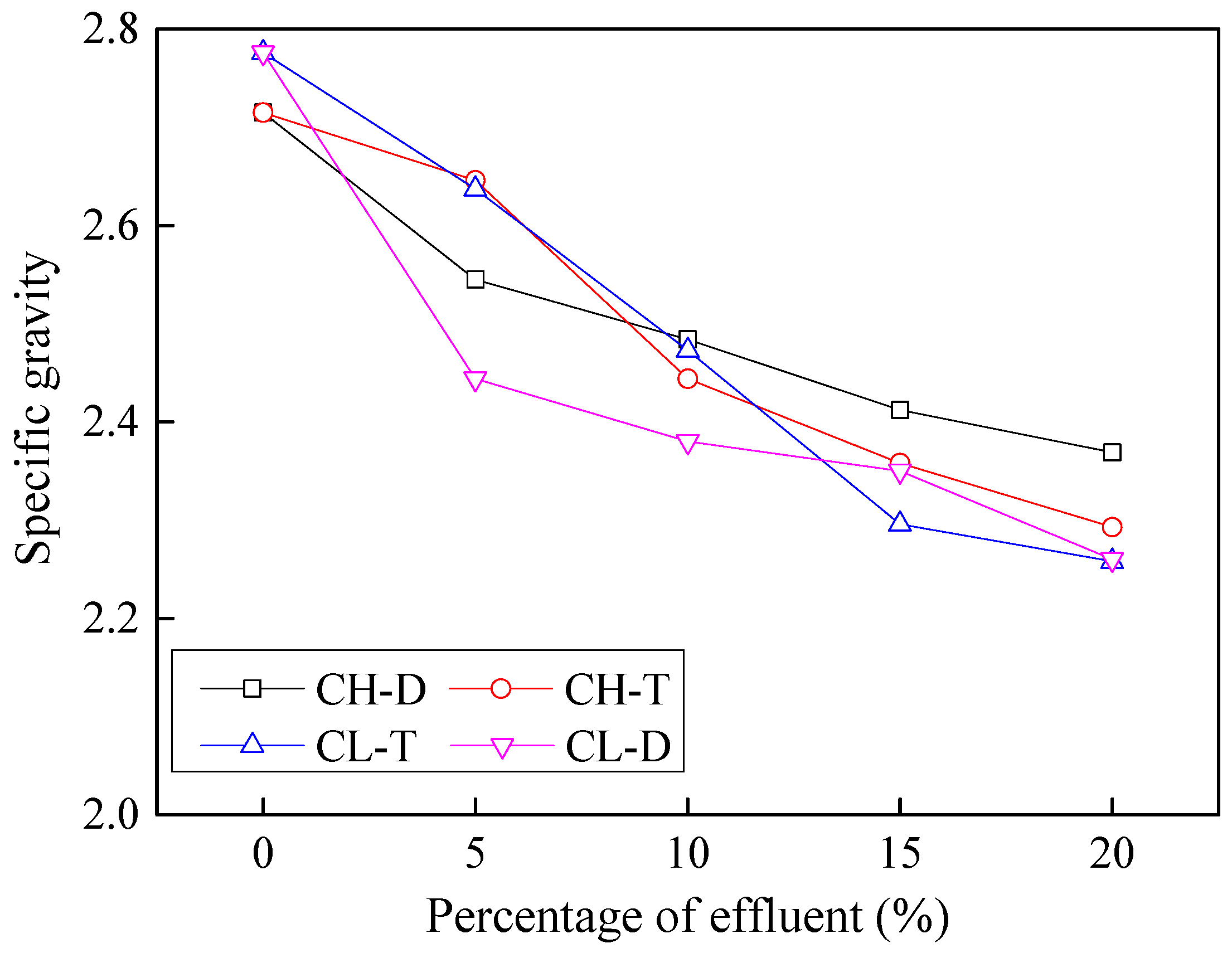
3.2. Effects of Contamination on Specific Gravity
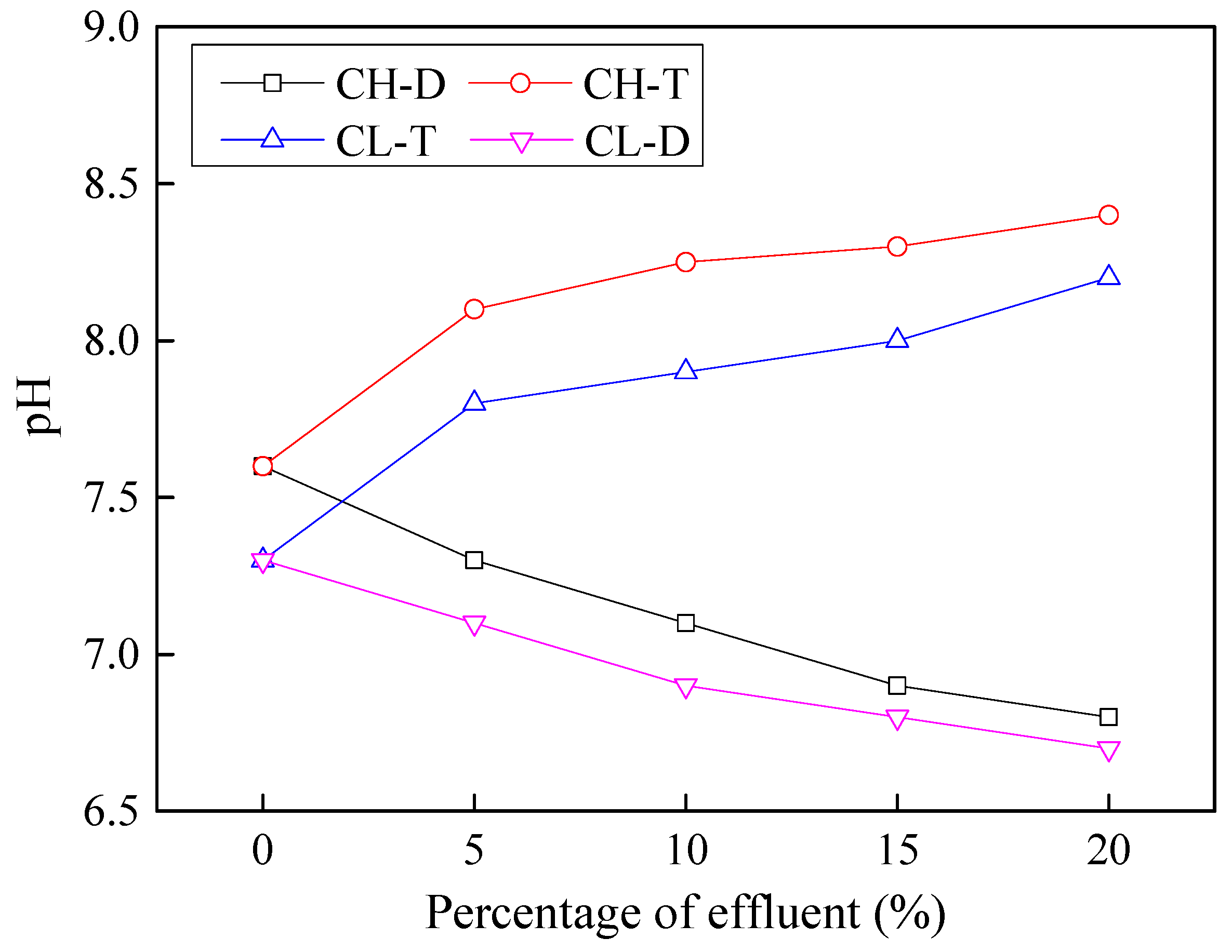
3.3. pH Value of Contaminated Soils
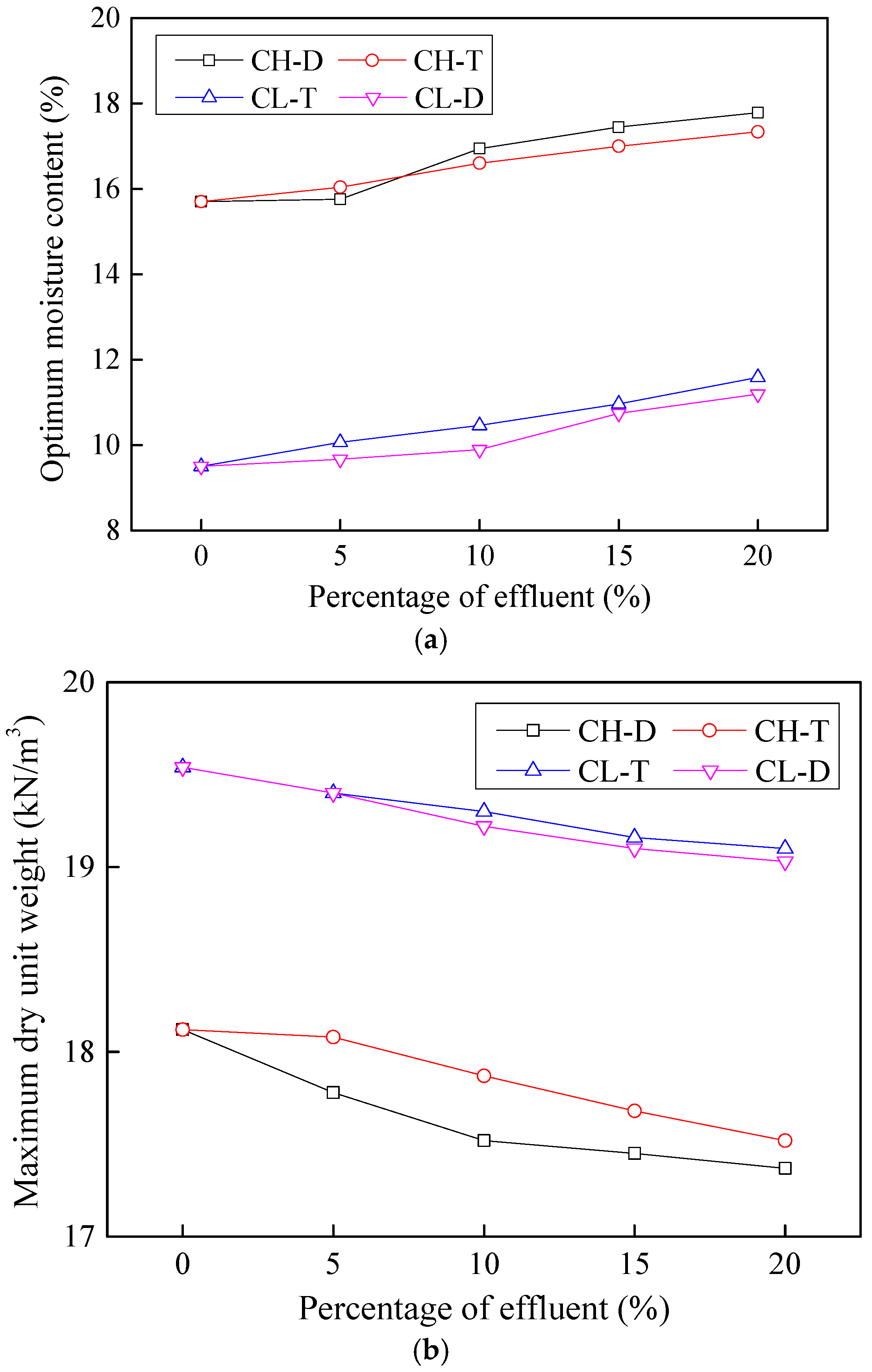
3.4. Effects on Compaction Characteristics
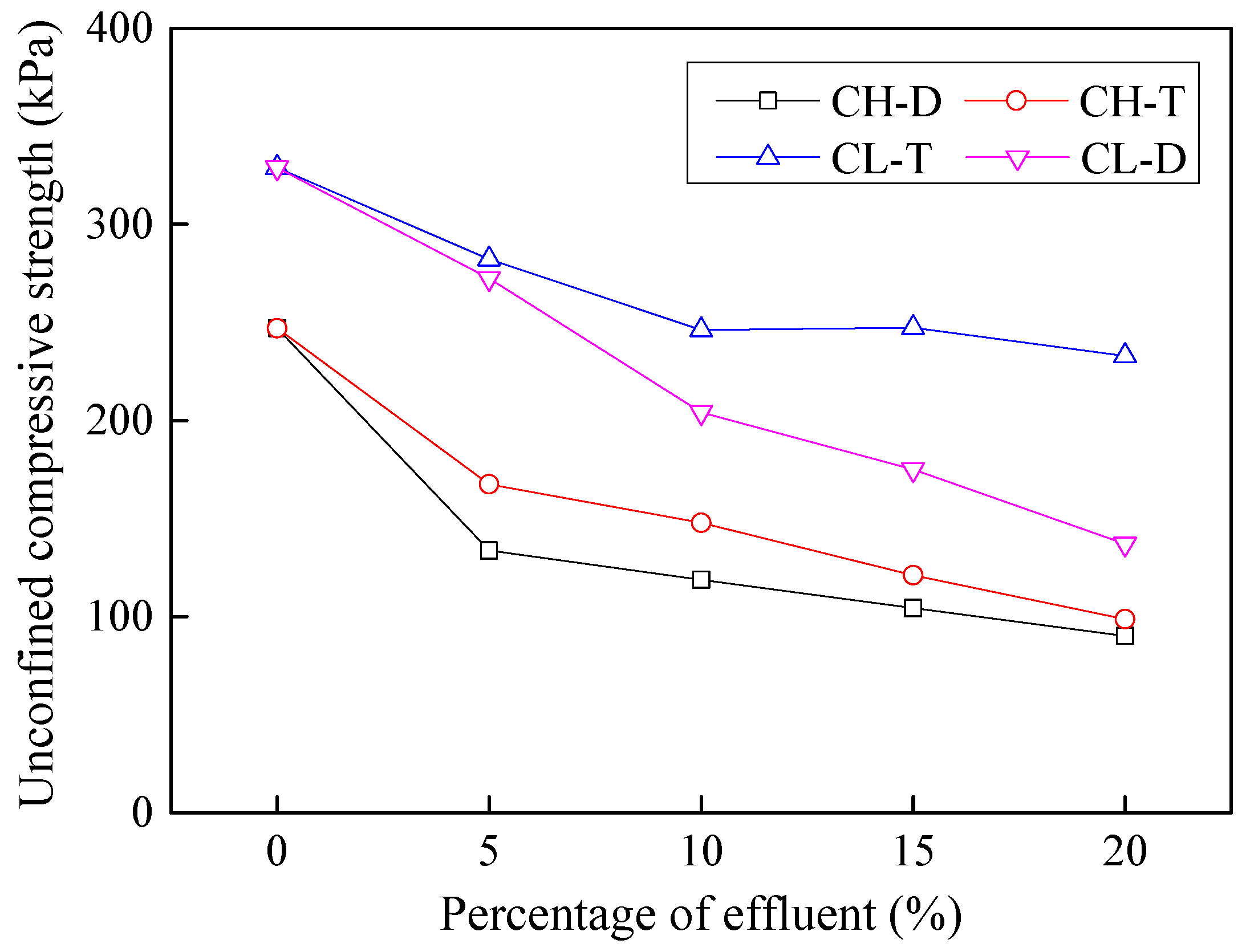
3.5. Effects of Contamination on Unconfined Compressive Strength
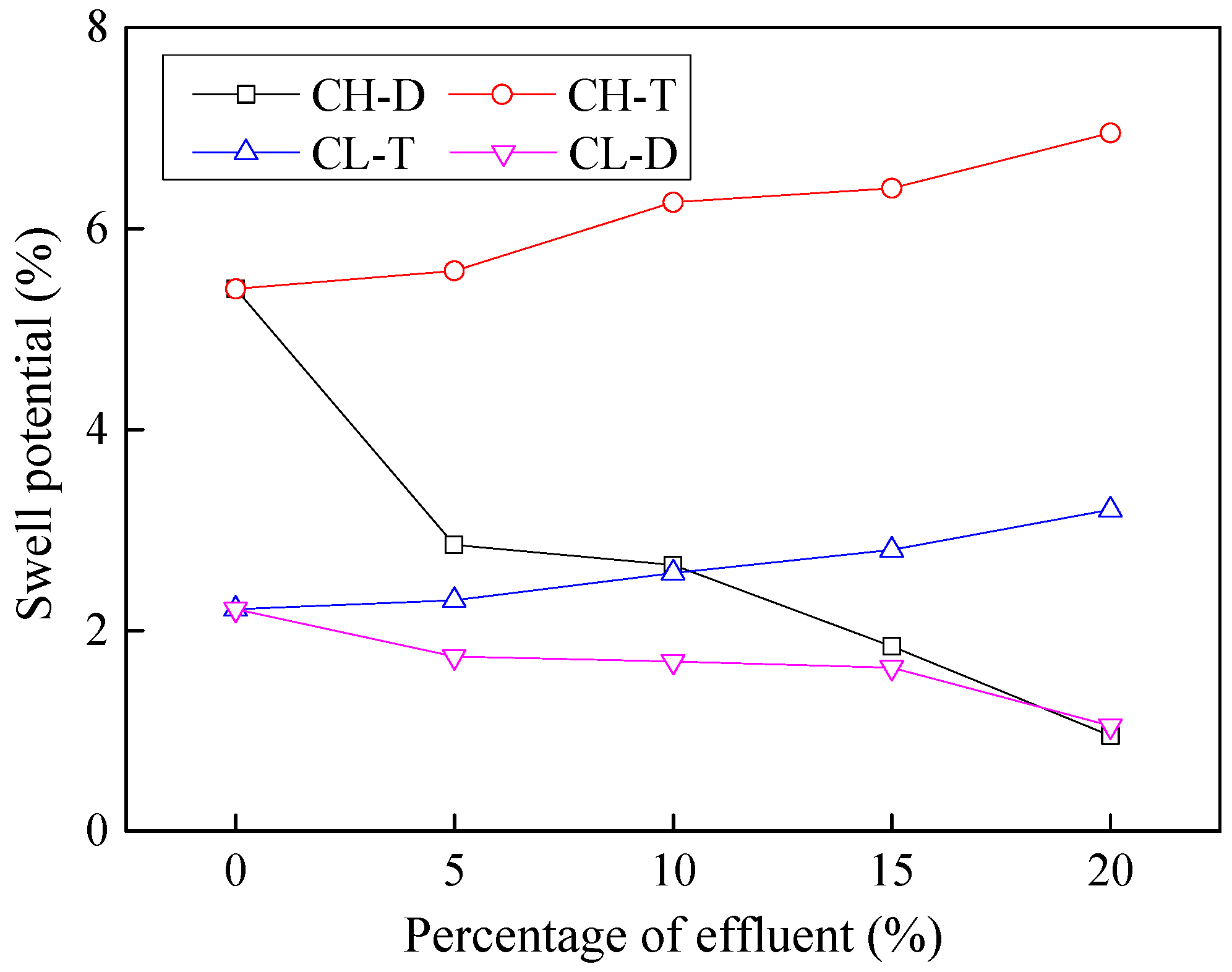
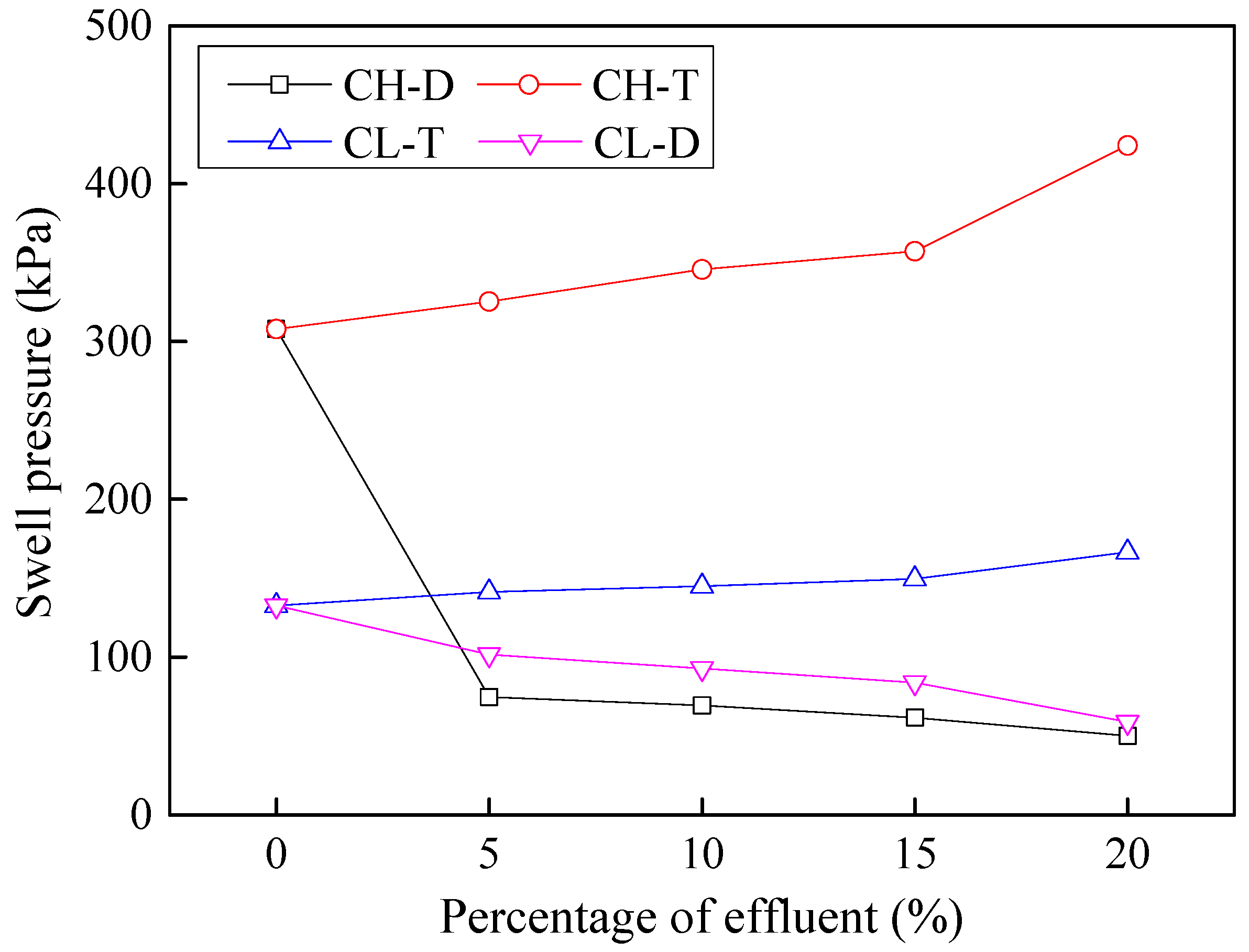
3.6. Effects of Contamination on Swelling Characteristics
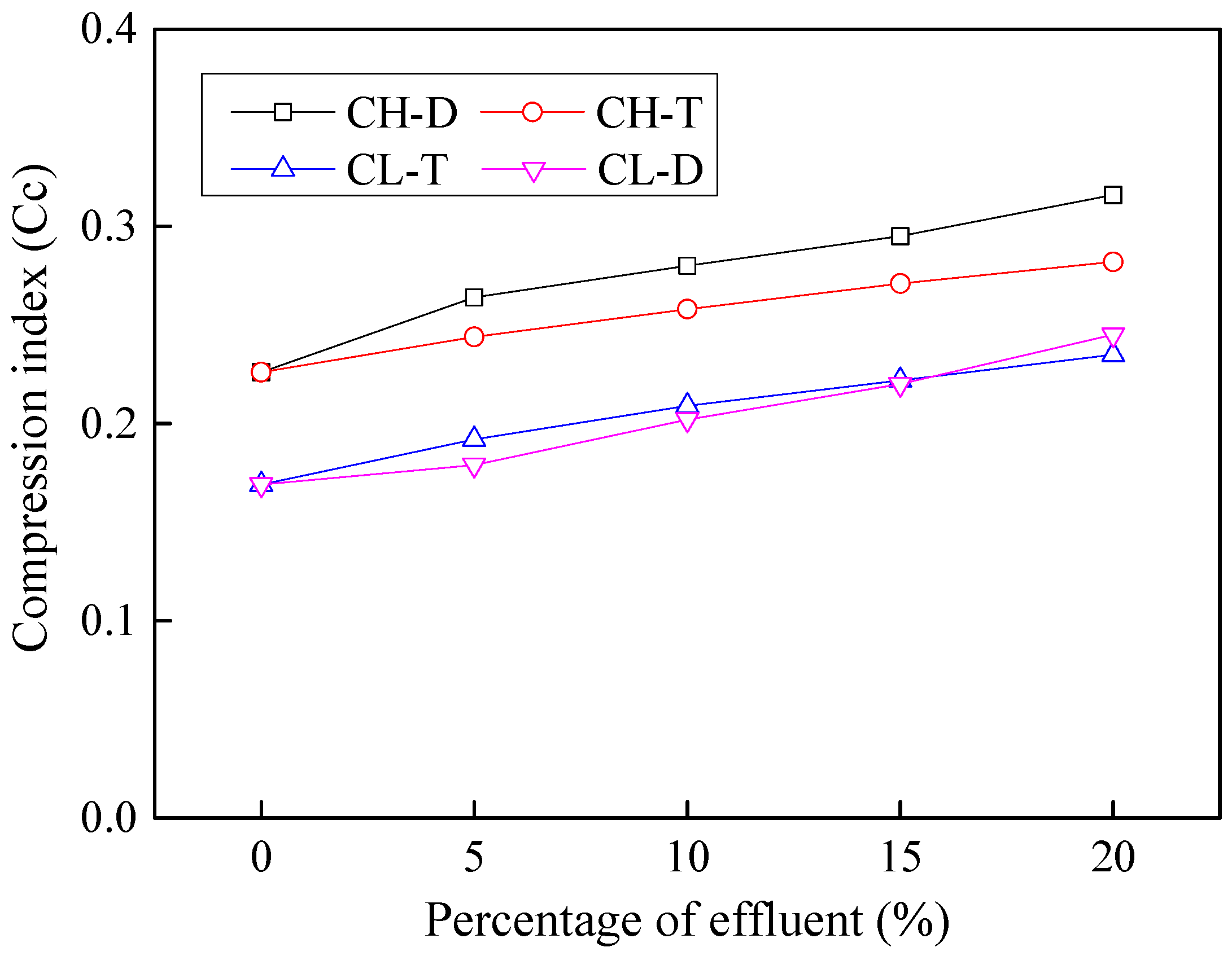
3.7. Effects of Contamination on Compression Index
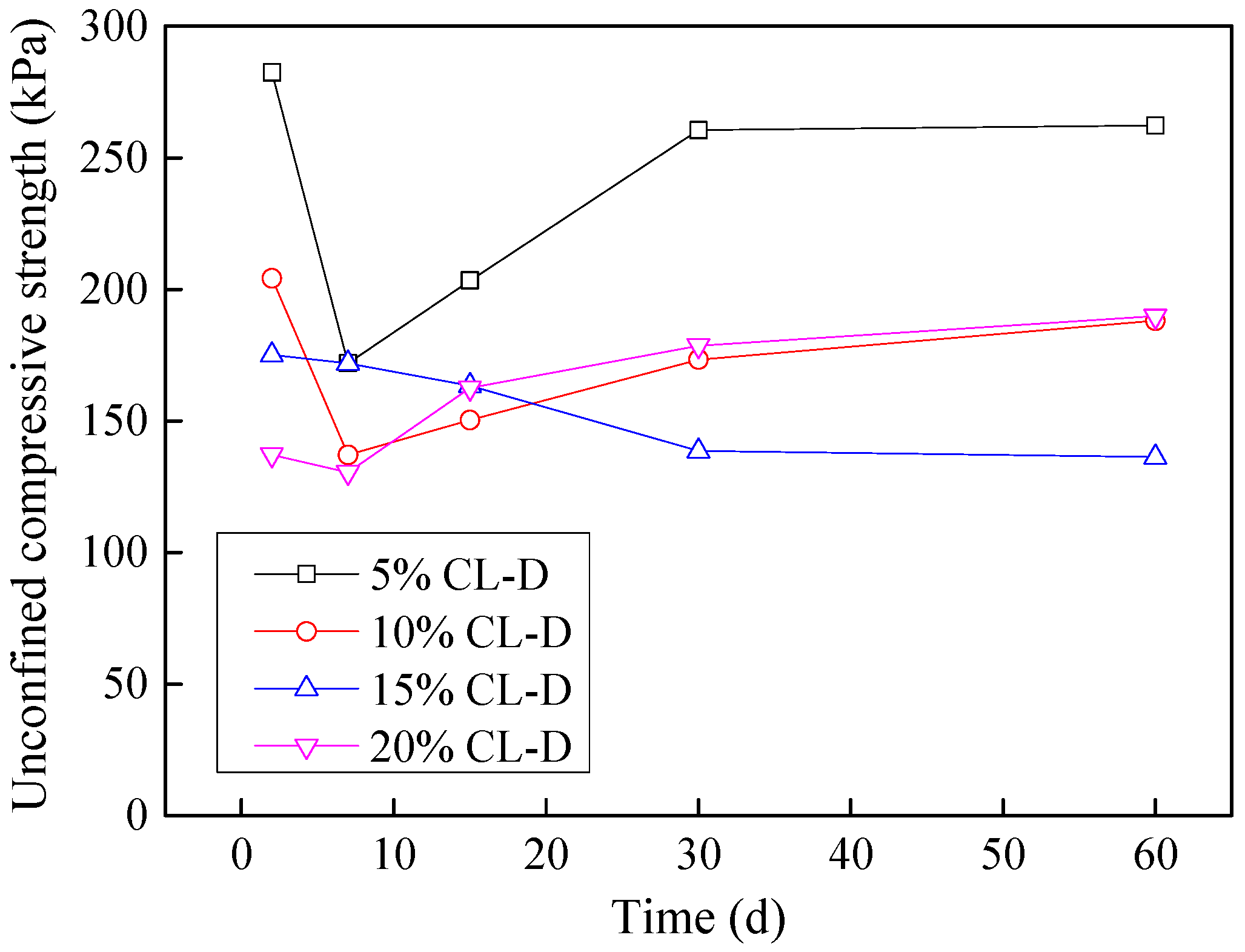
3.8. Time-Dependent Variation of Unconfined Compressive
3.8.1. Effects of Acidic Effluent
3.8.2. Effects of the Basic Effluent
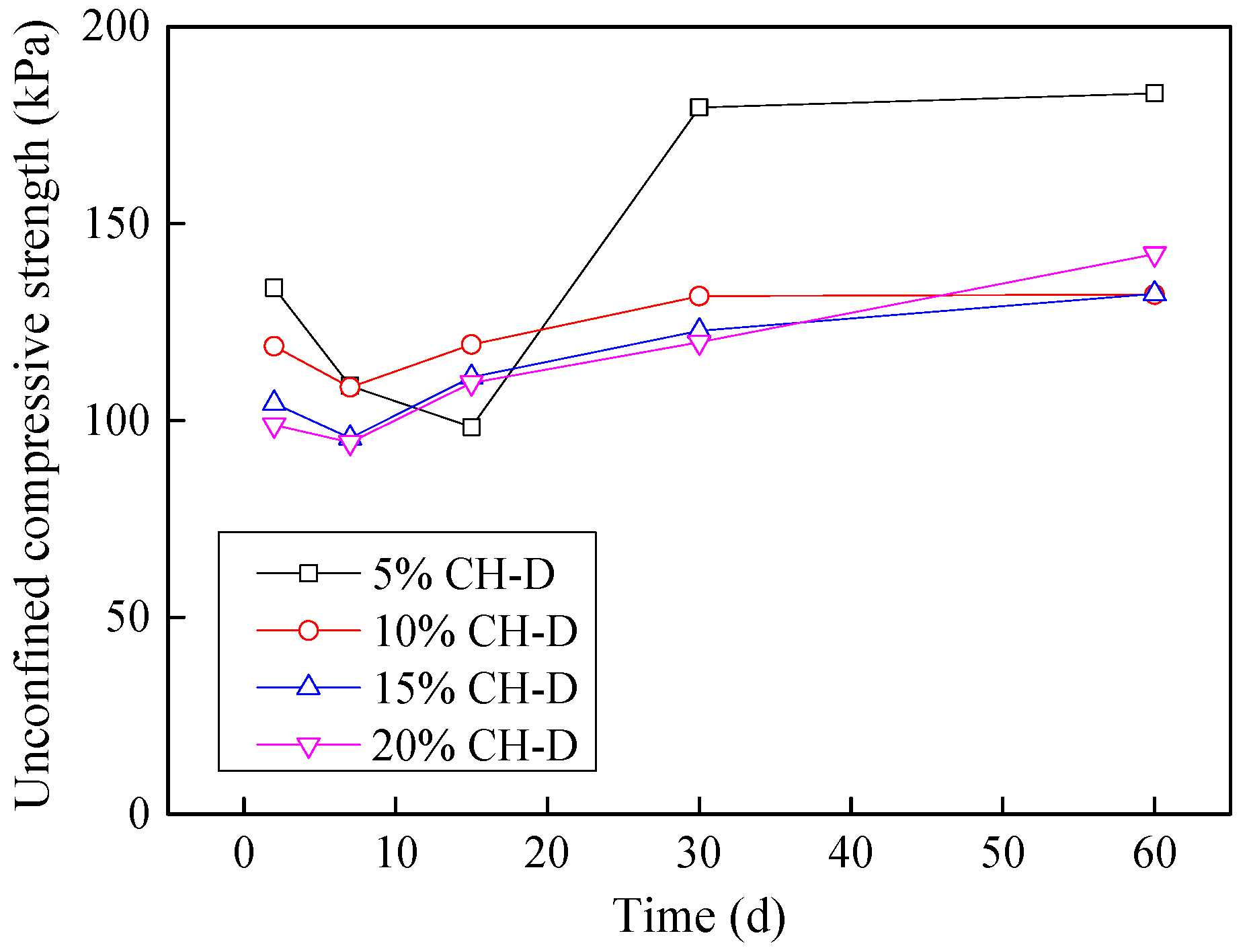
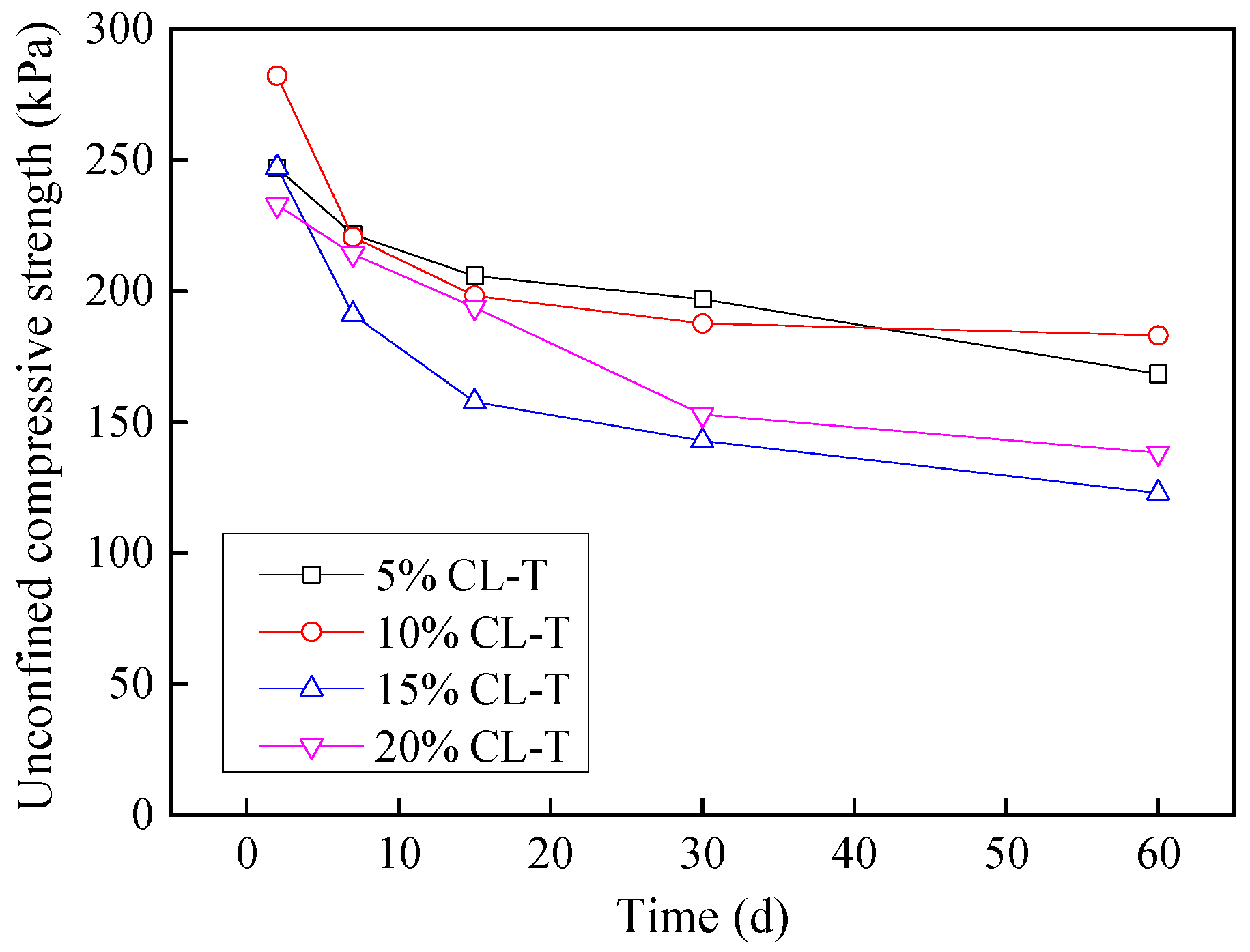
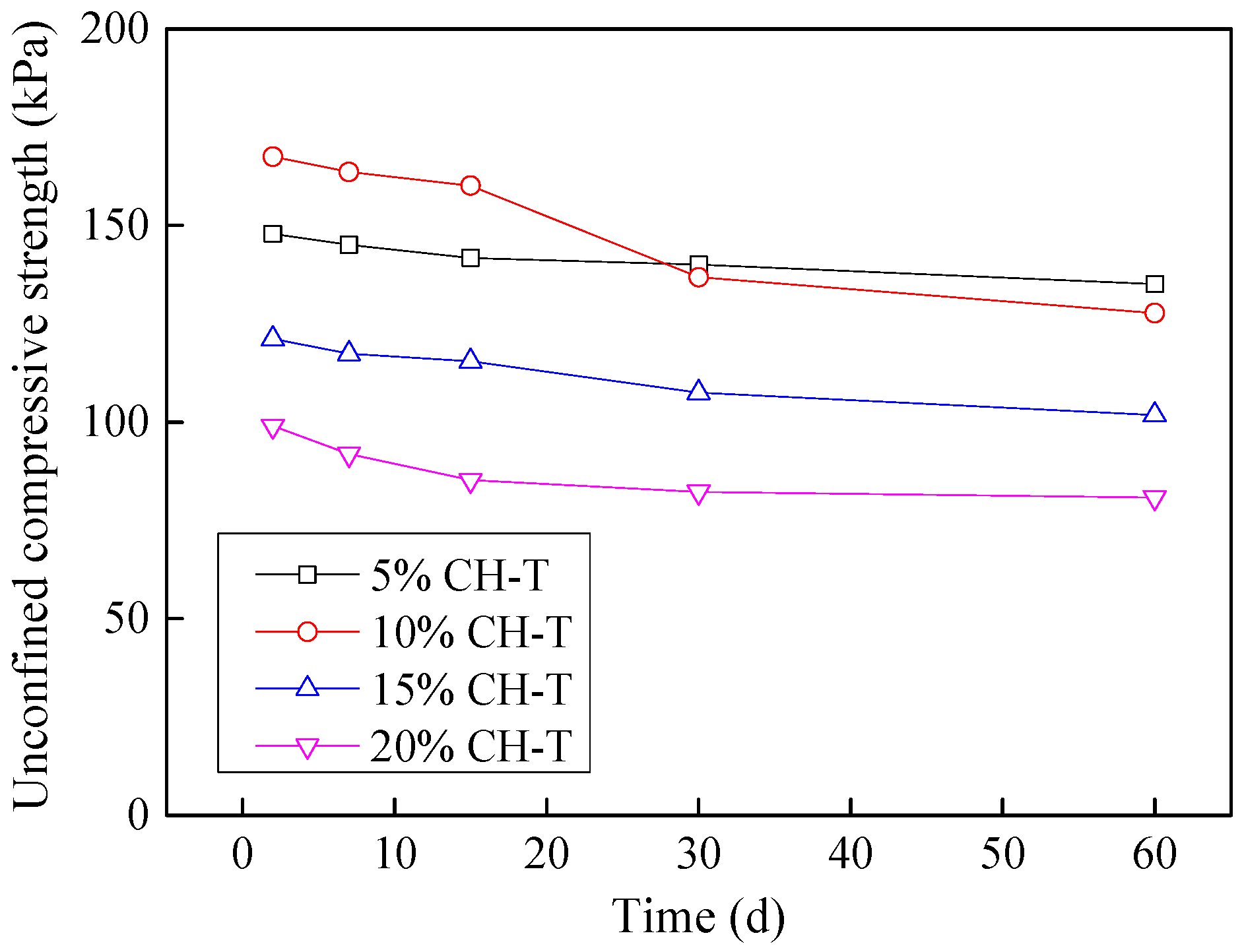
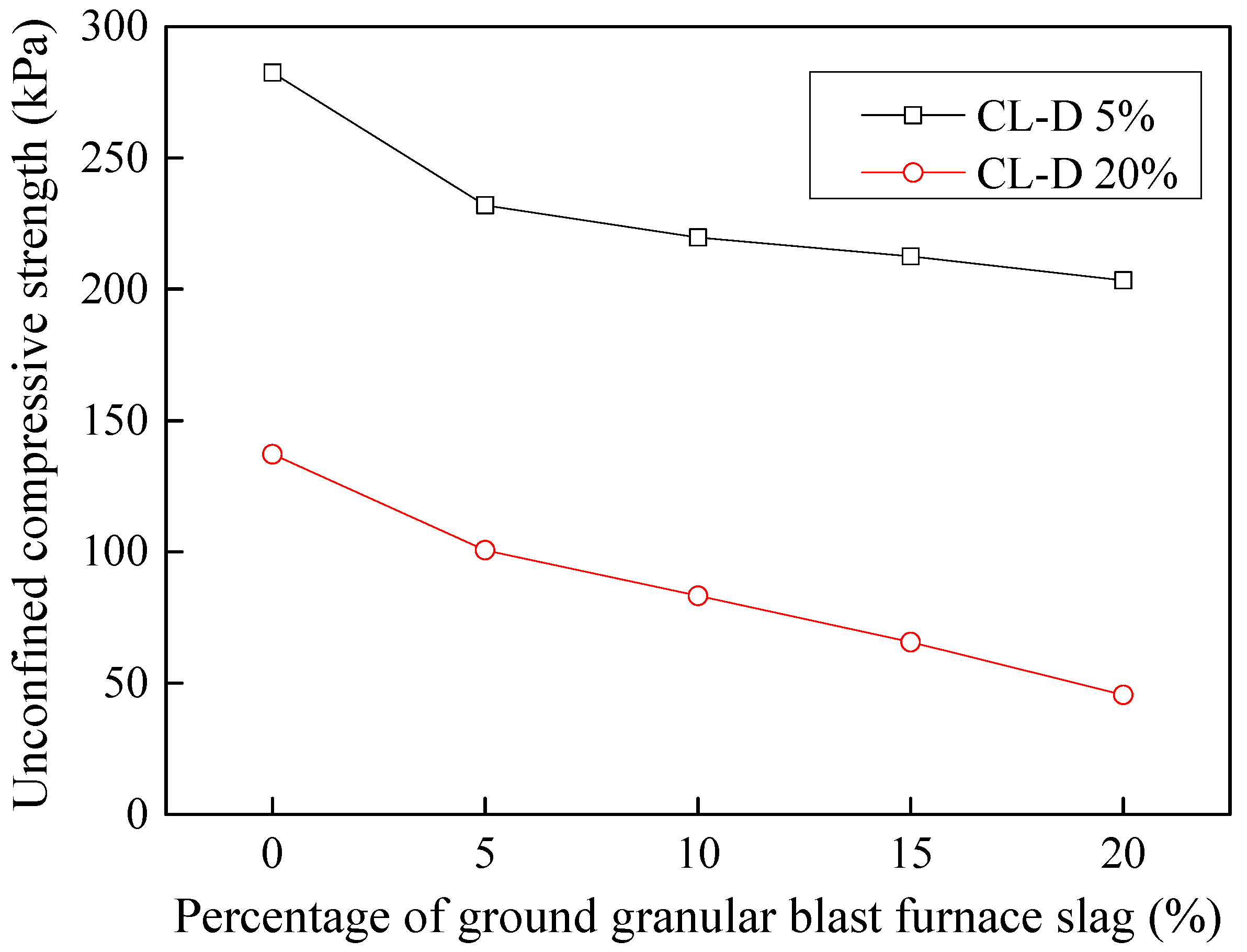
3.9. Effect of Industrial by-Products on Leachate Contaminated Soils
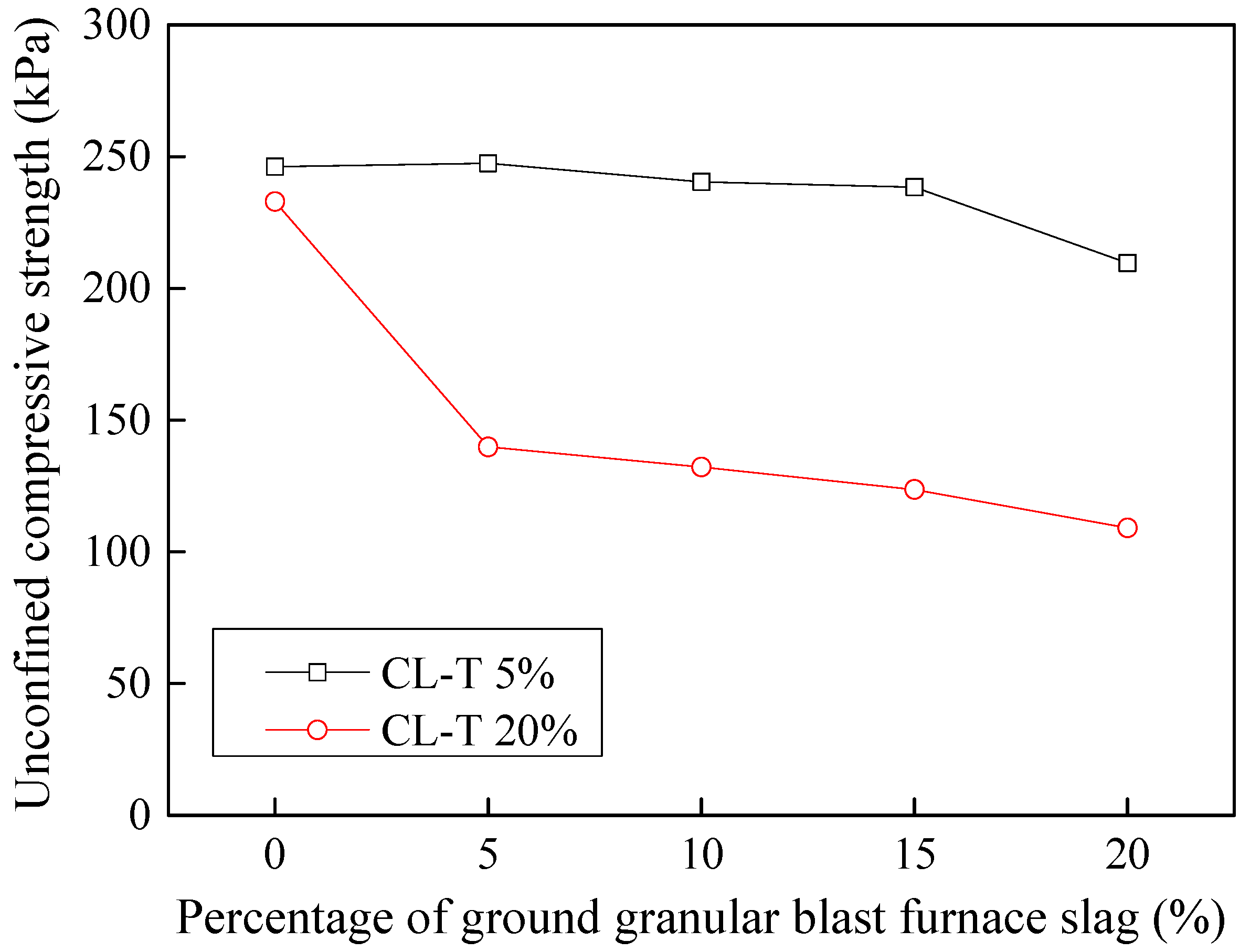
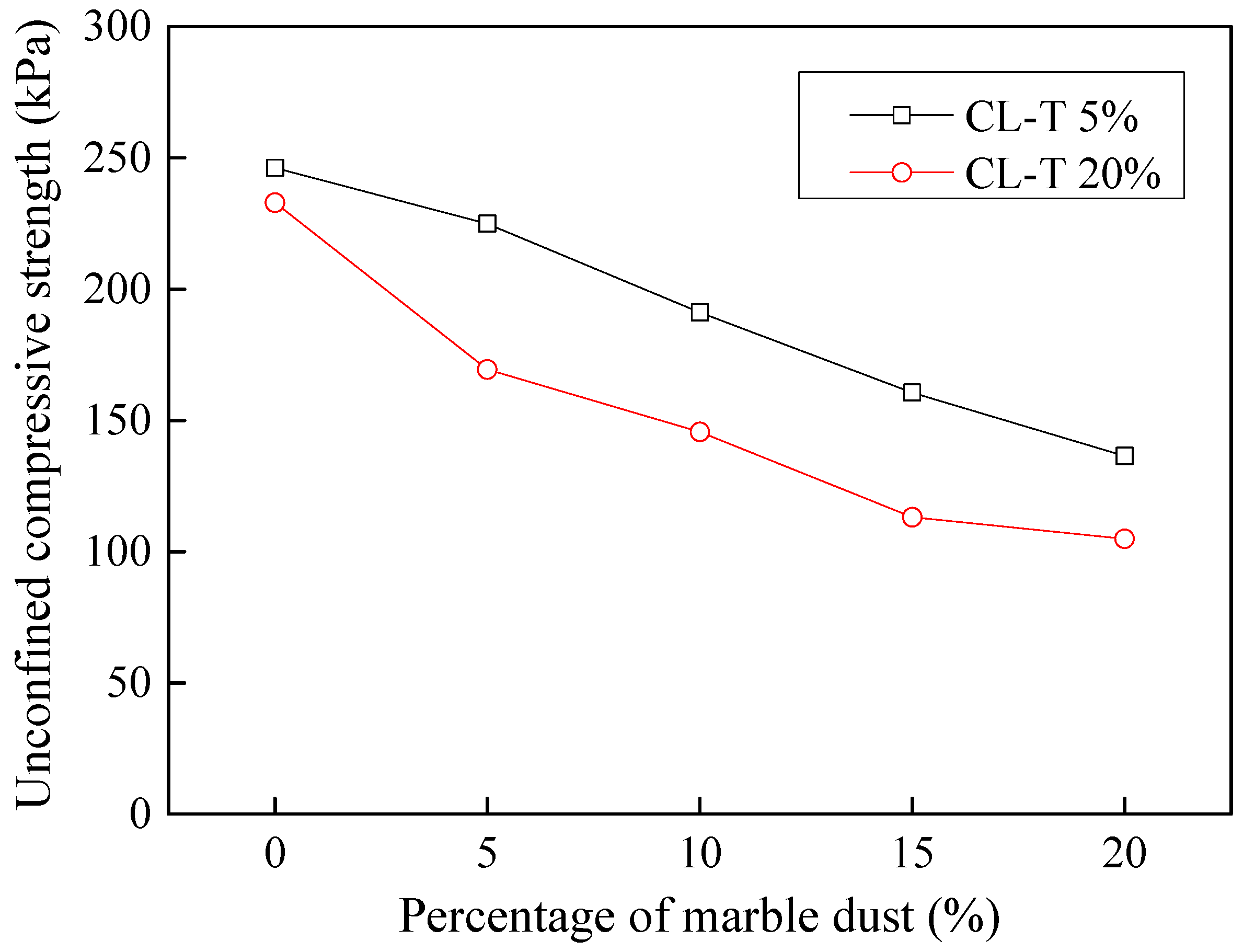
3.9.1. Stabilization of Acid-Affected CL Soil
3.9.2. Stabilization of Tannery-Affected CL Soil
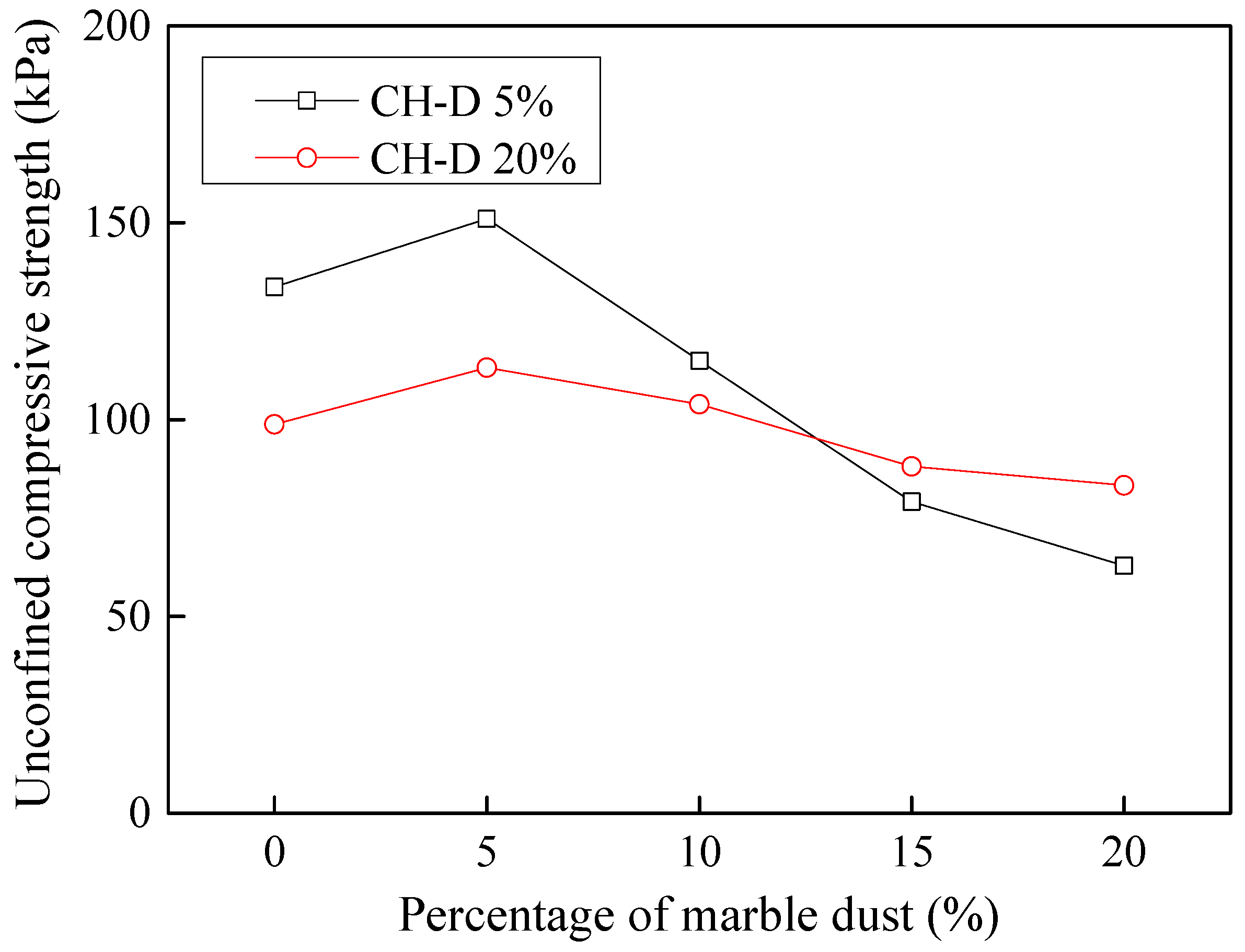
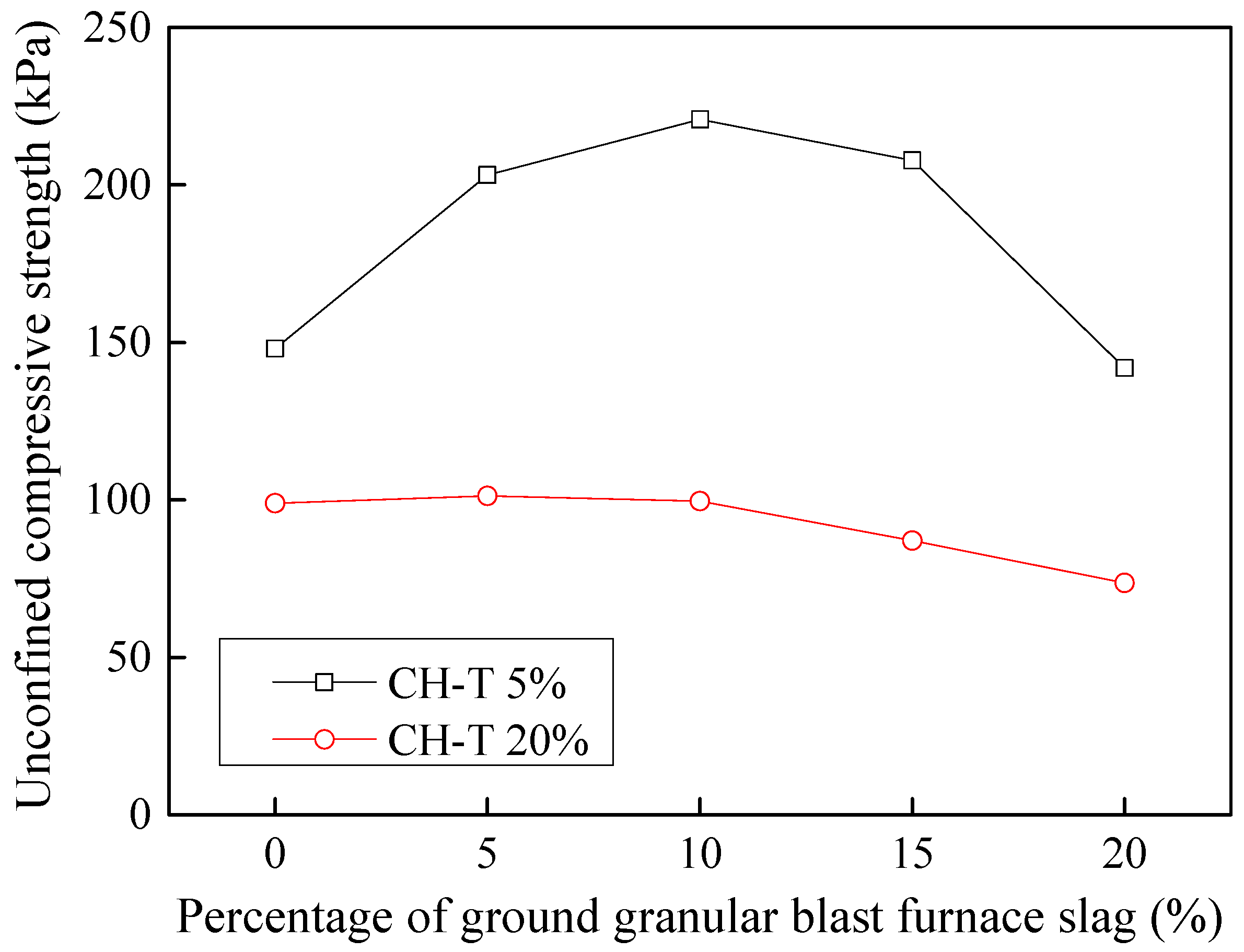
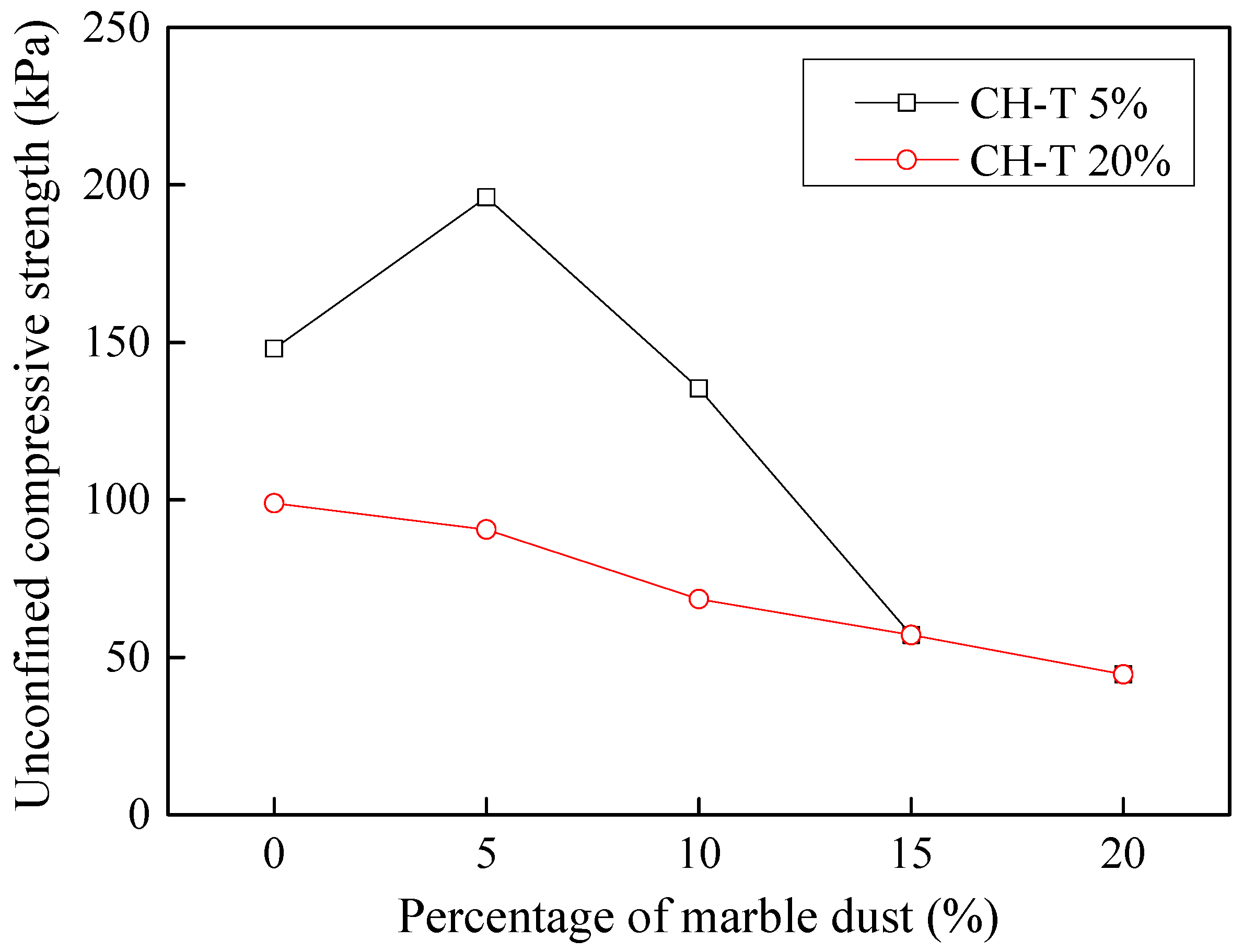
3.9.3. Stabilization of Dyeing-Affected CH Soil
3.9.4. Stabilization of Tannery-Affected CH Soil
4. Conclusions
- Effluents drastically reduce mechanical properties of soil, specifically cohesion. The unconfined compressive strength of effluent contaminate soils was observed to decrease by as much as 60% with 20% addition of industrial effluents. Possible disintegration of mineral particles such as Fe2O3, causing a decrease in soil density, can be attributed as the primary reason leading to such a reduction in soil strength.
- Both the liquid limit and the plasticity index of contaminated soils showed an increasing trend with contamination. This effect was however more pronounced for CH soils whose liquid limit and plasticity index increased by about 30% compared to around 8% increase for CL soils.
- Owing to the lower specific gravity of industrial effluents, the specific gravity of effluent contaminated soils decreaesed by 12% to 15% with 20% effluent contamination. The contaminated soils were thus found to have lower specific gravity, contained more fine particles, had a lower maximum dry density, and had a lower cohesion and friction angle.
- The behavior of basic effluent (tannery) is unique; it exhibits two distinct behaviors before and after optimum concentration. Prior to optimum concentration, changes in maximum dry density of soil may hypothetically occur because a reaction of the basic effluent’s constituents forms oxides of Fe present in the soil. This reaction forms FeO, which is lighter in weight and takes greater volume, thus leading to decreased density. Further increase in the basic effluent concentration post optimum concentration, trigger the remaining constituents to react to certain active masses of soils (likely not FeO), which fill the particle interspaces and increase the soil density.
- Contamination with industrial effluents was observed to cause around 30%–40% increase in compression index Cc of soil. An increase in Cc value suggests that the tendency of the soil to shrink or expand upon the intrusion of moisture is enhanced, which can lead to distress in structures constructed on such soils.
- The time dependent affect of effluents on cohesive soils was studied. The unconfined compressive strength of both the soils when contaminated with basic (tannery) effluent, kept on decreasing further with time. Both the soils when contaminated with acidic (dyeing) effluent however, show an initial decrease, followed by a gradual increase in compressive strength. Although both the soils start regaining strength with time, yet, in general, they fail to achieve unconfined compressive strength of virgin soil.
- Considering the deterioration in soil characeristics upon effluent contamination, second phase of this study focused on possible stabilization of effluent contaminated soils through addition of marble dust and ground granulated blast furnace slag (GGBFS). The main focus of this phase of research remained enhancement in the mechanical strength of soil, which remains among the most important characteristics for engineering use. Marble dust and GGBFS, which are typically considered as good stabilization agents for cohesive soils were found to possess negligible to low positive effect on the mechanical characteristics of effluent contaminated soils. This key finding of present research can hold significant practical value such that highlighting the need to devise special remediation materials for effluent contaminated soils.
Author Contributions
Funding
Conflicts of Interest
References
- Mitchell, J. Fundamentals of Soil Behavior; Wiley: New York, NY, USA, 1993. [Google Scholar]
- Gratchev, I.B.; Sassa, K. Cyclic behavior of fine-grained soils at different pH values. J. Geotech. Geoenviron. Eng. 2009, 135, 271–279. [Google Scholar] [CrossRef]
- Anson, R.W.W.; Hawkins, A.B. The effect of calcium ions in pore water on the residual shear strength of kaolinite and sodium montmorillonite. Geotechnique 1998, 48, 787–800. [Google Scholar] [CrossRef]
- Moore, R.; Brunsden, D. Physicochemical effects on the behavior of a coastal mudslide. Geotechnique 1998, 46, 259–278. [Google Scholar] [CrossRef]
- Tiwari, B.; Tuladhar, G.; Marui, H. Variation in residual shear strength of the soil with the salinity of pore fluid. J. Geotech. Geoenviron. Eng. 2005, 131, 1445–1456. [Google Scholar] [CrossRef]
- Ratnaweera, P.; Meegoda, J. Shear strength and stress–strain behavior of contaminated soils. Geotech. Test. J. 2006, 29, 133–140. [Google Scholar] [CrossRef]
- Gratchev, I.; Sassa, K.; Fukuoka, H. How reliable is the plasticity index for estimating the liquefaction potential of clayey sands? J. Geotech. Geoenviron. Eng. 2006, 132, 124–127. [Google Scholar] [CrossRef]
- Gratchev, I.; Sassa, K.; Osipov, V.; Fukuoka, H.; Wang, G. Undrained cyclic behavior of bentonite-sand mixtures and factors affecting it. Geotech. Geol. Eng. 2007, 25, 349–367. [Google Scholar] [CrossRef]
- Gratchev, I.; Towhata, I. Compressibility of soils containing kaolinite in acidic environments. KSCE J. Civ. Eng. 2016, 20, 623–630. [Google Scholar] [CrossRef]
- Sunil, B.; Nayak, S.; Shrihari, S. Effect of pH on the geotechnical properties of laterite. Eng. Geol. 2006, 85, 197–203. [Google Scholar] [CrossRef]
- Sunil, B.; Shrihari, S.; Nayak, S. Shear strength characteristics and chemical characteristics of leachate-contaminated lateritic soil. Eng. Geol. 2009, 106, 20–25. [Google Scholar] [CrossRef]
- Khan, M.I.; Irfan, M.; Aziz, M.; Khan, A.H. Geotechnical characteristics of effluent contaminated cohesive soils. J. Environ. Eng. Landsc. 2017, 25, 75–82. [Google Scholar] [CrossRef]
- Terzaghi, K.; Peck, R.; Mesri, G. Soil Mechanics in Engineering Practice; John Wiley & Sons: New York, NY, USA, 1948. [Google Scholar]
- Gibbs, H.J.; Bara, J.P. Stability problems of collapsing soil. J. Soil Mech. Found. 1967, 93, 577–594. [Google Scholar]
- Pandian, N.; Nagaraj, T.; Manoj, M. Re-examination of compaction characteristics of fie-grained soils. Geotechnique 1997, 47, 363–366. [Google Scholar] [CrossRef]
- Sridharan, A.; Nagaraj, H. Plastic limit and compaction characteristics of fine grained soils. Proc. Inst. Civ. Eng. Ground Improv. 2005, 9, 17–22. [Google Scholar] [CrossRef]
- Umesha, T.S.; Dinesh, S.V.; Sivapullaiah, P.V. Effects of acids on geotechnical properties of black cotton soil. Inter. J. Geol. 2012, 6, 69–76. [Google Scholar]
- Stalin, V.K.; Muthukumaran, K.; Kartikeyan, A. Effect of Liquid waste on the index and engineering behaviour of soils. In Proceedings of the Indian Geotechnical Conference: IGC-2000: The millennium conference, Mumbai, Indian, 13–15 December 2010; pp. 229–230. [Google Scholar]
- Anandarajah, A.; Zhao, D. Triaxial behavior of kaolinite in different pore fluids. J. Geotech. Geoenviron. Eng. 2000, 126, 148–156. [Google Scholar] [CrossRef]
- Van Olphen, H. An Introduction to Clay Colloid Chemistry, 2nd ed.; John Wiley & Sons: New York, NY, USA, 1977. [Google Scholar]
- Sridharan, A.; Rao, S.M.; Murthy, N.S. Liquid limit of kaolinitic soils. Geotechnique 1988, 38, 191–198. [Google Scholar] [CrossRef]
- Ural, N.; Karakurt, C.; Cömert, A.T. Influence of marble wastes on soil improvement and concrete production. J. Mater. Cycles Waste Manag. 2014, 16, 500–508. [Google Scholar] [CrossRef]
- Tozsin, G.; Arol, A.I.; Oztas, T.; Kalkan, E. Using marble wastes as a soil amendment for acidic soil neutralization. J. Environ. Manag. 2014, 133, 374–377. [Google Scholar] [CrossRef] [PubMed]
- Phummiphan, I.; Horpibulsuk, S.; Rachan, R.; Arulrajah, A.; Shen, S.L.; Chindaprasirt, P. High calcium fly ash geopolymer stabilized lateritic soil and granulated blast furnace slag blends as a pavement base material. J. Hazard. Mater. 2017, 341, 257. [Google Scholar] [CrossRef] [PubMed]
- Hasan, U.; Chegenizadeh, A.; Budihardjo, M.A.; Nikraz, H. Experimental evaluation of construction waste and ground granulated blast furnace slag as alternative soil stabilisers. Geotech. Geol. Eng. 2016, 34, 1707–1722. [Google Scholar] [CrossRef]
- Jaiswal, M.; Lai, B. Stabilization of clayey soil with garlic skin and rice husk ash for flexible pavement construction. Geotech. Test. J. 2017, 40, 20160227. [Google Scholar] [CrossRef]
- Mohanty, S.K.; Pradhan, P.K.; Mohanty, C.R. Consolidation and drainage characteristics of expansive soil stabilized with fly ash and dolochar. Geotech. Geol. Eng. 2016, 34, 1435–1451. [Google Scholar] [CrossRef]
- Phanikumar, B.R.; Shankar, M.U. Heave studies on fly ash-stabilised expansive clay liners. Geotech. Geol. Eng. 2017, 35, 111–120. [Google Scholar] [CrossRef]
- Chiou, J.S.; You, T.R.; Tsai, C.C.; Hwang, J.H. Performance of laterally loaded piles in improved coal ash deposit. Soils Found. 2017, 57, 872–881. [Google Scholar] [CrossRef]
- Rashid, A.S.A.; Latifi, N.; Meehan, C.L.; Manahiloh, K.N. Sustainable improvement of tropical residual soil using an environmentally friendly additive. Geotech. Geol. Eng. 2017, 35, 2613–2623. [Google Scholar] [CrossRef]




| Properties | CH | CL |
|---|---|---|
| Classification | High plastic clay | Low plastic clay |
| pH | 7.6 | 7.3 |
| Liquid limit | 54.6% | 31.18% |
| Plastic limit | 26.61% | 17.35% |
| Specific gravity | 2.71 | 2.78 |
| Max. dry density | 18.12 kN/m3 | 19.57 kN/m3 |
| Optimum moisture content | 15.7% | 9.5% |
| Swell potential | 5.4% | 2.2% |
| Swell pressure | 307.92 kPa | 132.58 kPa |
| Tests Performed | Tannery Effluent | Dyeing Effluent |
|---|---|---|
| Color | Dark greenish | Dark reddish |
| pH | 11.5 | 5.1 |
| Total solids (g/L) | 25.43 | 1.673 |
| Total dissolved solids (mg/L) | 19.78 | 910 |
| Total suspended solids (mg/L) | 603 | 330 |
| Suspended solids (g/L) | 6 | 1 |
| BOD (mg/L) | 4445 | 1980 |
| COD (mg/L) | 5000 | 2100 |
| Chlorides (mg/L) | 230 | 175 |
| Specific gravity | 1.012 | 1.007 |
| Sulphates (mg/L) | 281 | 198 |
| Nitrates (mg/L) | 34 | 28 |
| Material | Constituents | Percentage (%) |
|---|---|---|
| Marble dust | SiO2 | 6.2 |
| Fe2O3 | 0.8 | |
| CaO | 30.1 | |
| Al2O3 | 4.8 | |
| Ground granulated blast furnace slag | CaO | 30–50 |
| SiO2 | 28–38 | |
| Al2O3 | 8–24 | |
| MgO | 1–18 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Irfan, M.; Chen, Y.; Ali, M.; Abrar, M.; Qadri, A.; Bhutta, O. Geotechnical Properties of Effluent-Contaminated Cohesive Soils and Their Stabilization Using Industrial By-Products. Processes 2018, 6, 203. https://doi.org/10.3390/pr6100203
Irfan M, Chen Y, Ali M, Abrar M, Qadri A, Bhutta O. Geotechnical Properties of Effluent-Contaminated Cohesive Soils and Their Stabilization Using Industrial By-Products. Processes. 2018; 6(10):203. https://doi.org/10.3390/pr6100203
Chicago/Turabian StyleIrfan, Muhammad, Yulong Chen, Muhammad Ali, Muhammad Abrar, Ahmed Qadri, and Osama Bhutta. 2018. "Geotechnical Properties of Effluent-Contaminated Cohesive Soils and Their Stabilization Using Industrial By-Products" Processes 6, no. 10: 203. https://doi.org/10.3390/pr6100203
APA StyleIrfan, M., Chen, Y., Ali, M., Abrar, M., Qadri, A., & Bhutta, O. (2018). Geotechnical Properties of Effluent-Contaminated Cohesive Soils and Their Stabilization Using Industrial By-Products. Processes, 6(10), 203. https://doi.org/10.3390/pr6100203






