Recent Advances on Carbon Molecular Sieve Membranes (CMSMs) and Reactors
Abstract
:1. Introduction
2. Carbon Molecular Sieve Membranes
3. Composite Carbon Molecular Sieve Membranes (c-CMSMs)
4. Supported Carbon Molecular Sieve Membranes
5. Supported CMSMs Prepared by One Dip-Dry-Carbonization Cycle
6. Carbon Molecular Sieve Membrane Modules
7. Carbon Molecular Sieve Membrane for Water Separation
8. Carbon Molecular Sieve Membrane Reactors
8.1. CMSM Reactors for Production of Hydrogen
8.2. CMSM Reactors for Water Gas Shift Reaction
8.3. CMSM Reactors for Methanol Steam Reforming (MSR)
8.4. CMSM Reactors for Ethanol Steam Reforming (ESR)
8.5. CMSM Reactors for Dehydrogenation Reactions
9. Conclusions and Future Trends
Supplementary Files
Supplementary File 1Acknowledgments

Disclaimer
Conflicts of Interest
References
- Sholl, D.S.; Lively, R.P. Seven chemical separations to change the world. Nature 2016, 532, 435–437. [Google Scholar] [CrossRef] [PubMed]
- Industrial Technologies Program. Energy Efficiency and Renewable Energy. U.S. Departament of Energy. Available online: https://www1.eere.energy.gov/manufacturing/industries technologies (accessed on 4 May 2015).
- Robeson, L.M. The upper bound revisited. J. Membr. Sci. 2008, 320, 390–400. [Google Scholar] [CrossRef]
- Robeson, L.M. Correlation of separation factor versus permeability for polymeric membranes. J. Membr. Sci. 1991, 62, 165–185. [Google Scholar] [CrossRef]
- Henry, R.; Tayakout-Fayolle, M.; Afanasiev, P.; Lorentz, C.; Lapisardi, G.; Pirngruber, G. Vacuum gas oil hydrocracking performance of bifunctional Mo/Y zeolite catalysts in a semi-batch reactor. Catal. Today 2014, 220–222, 159–167. [Google Scholar] [CrossRef]
- Nakhaei, A.; Housaindokht, M.R. Study of activity, products selectivity and physico-chemical properties of bifunctional Fe/HZSM-5 Fischer-Tropsch catalyst: Effect of catalyst shaping. J. Nat. Gas Sci. Eng. 2013, 14, 29–33. [Google Scholar] [CrossRef]
- Sartipi, S.; Parashar, K.; Valero-Romero, M.J.; Santos, V.P.; van der Linden, B.; Makkee, M.; Kapteijn, F.; Gascon, J. Hierarchical H-ZSM-5-supported cobalt for the direct synthesis of gasoline-range hydrocarbons from syngas: Advantages, limitations, and mechanistic insight. J. Catal. 2013, 305, 179–190. [Google Scholar] [CrossRef]
- Koros, W.J.; Ma, Y.H.; Shimidzu, T. Terminology for membranes and membrane processes (IUPAC Recommendations 1996). Pure Appl. Chem. 1996, 68, 1479–1489. [Google Scholar] [CrossRef]
- Zaman, J.; Chakma, A. Inorganic membrane reactors. J. Membr. Sci. 1994, 92, 1–28. [Google Scholar] [CrossRef]
- Saufi, S.M.; Ismail, A.F. Fabrication of carbon membranes for gas separation—A review. Carbon 2004, 42, 241–259. [Google Scholar] [CrossRef]
- Barbosa-Coutinho, E.; Salim, V.M.M.; Piacsek Borges, C. Preparation of carbon hollow fiber membranes by pyrolysis of polyetherimide. Carbon 2003, 41, 1707–1714. [Google Scholar] [CrossRef]
- Foley, H.C. Carbogenic molecular sieves: Synthesis, properties and applications. Microporous Mater. 1995, 4, 407–433. [Google Scholar] [CrossRef]
- Sá, S.; Sousa, J.M.; Mendes, A. Steam reforming of methanol over a CuO/ZnO/Al2O3 catalyst part II: A carbon membrane reactor. Chem. Eng. Sci. 2011, 66, 5523–5530. [Google Scholar] [CrossRef]
- Koresh, J.; Soffer, A. Molecular sieve carbon permselective membrane. Part I. Presentation of a new device for gas mixture separation. Sep. Sci. Technol. 1983, 18, 723–734. [Google Scholar] [CrossRef]
- Salleh, W.N.W.; Ismail, A.F. Carbon membranes for gas separation processes: Recent progress and future perspective. J. Membr. Sci. Res. 2015, 1, 2–15. [Google Scholar]
- Briceño, K.; Basile, A.; Tong, J.; Haraya, K. Carbon-based membranes for membrane reactors. In Handbook of Membrane Reactors, 1st ed.; Basile, A., Ed.; Woodhead Publishing Limited: Cambridge, UK, 2013; pp. 370–400. [Google Scholar]
- Xu, L.; Rungta, M.; Brayden, M.K.; Martinez, M.V.; Stears, B.A.; Barbay, G.A.; Koros, W.J. Olefins-selective asymmetric carbon molecular sieve hollow fiber membranes for hybrid membrane-distillation processes for olefin/paraffin separations. J. Membr. Sci. 2012, 423–424, 314–323. [Google Scholar] [CrossRef]
- Hayashi, J.; Mizuta, H.; Yamamoto, M.; Kusakabe, K.; Morooka, S.; Suh, S.H. Separation of ethane/ethylene and propane/propylene systems with a carbonized BPDA−pp‘ODA polyimide membrane. Ind. Eng. Chem. Res. 1996, 35, 4176–4181. [Google Scholar] [CrossRef]
- Okamoto, K.; Kawamura, S.; Yoshino, M.; Kita, H.; Hirayama, Y.; Tanihara, N.; Kusuki, Y. Olefin/Paraffin Separation through carbonized membranes derived from an asymmetric polyimide hollow fiber membrane. Ind. Eng. Chem. Res. 1999, 38, 4424–4432. [Google Scholar] [CrossRef]
- Salleh, W.N.W.; Ismail, A.F.; Matsuura, T.; Abdullah, M.S. Precursor selection and process conditions in the preparation of carbon membrane for gas separation: A Review. Sep. Purif. Rev. 2011, 40, 261–311. [Google Scholar] [CrossRef]
- Tin, P.S.; Chung, T.S.; Hill, A.J. Advanced fabrication of carbon molecular sieve membranes by nonsolvent pretreatment of precursor polymers. Ind. Eng. Chem. Res. 2004, 43, 6476–6483. [Google Scholar] [CrossRef]
- Tseng, H.H.; Chang, S.H.; Wey, M.Y. A carbon gutter layer-modified α-Al2O3 substrate for PPO membrane fabrication and CO2 separation. J. Membr. Sci. 2014, 454, 51–61. [Google Scholar] [CrossRef]
- Baker, R.W. Future directions of membrane gas separation technology. Ind. Eng. Chem. Res. 2002, 41, 1393–1411. [Google Scholar] [CrossRef]
- Ning, X.; Koros, W.J. Carbon molecular sieve membranes derived from Matrimid® polyimide for nitrogen/methane separation. Carbon 2014, 66, 511–522. [Google Scholar] [CrossRef]
- Hosseini, S.S.; Omidkhah, M.R.; Moghaddam, A.Z.; Pirouzfar, V.; Krantz, W.B.; Tan, N.R. Enhancing the properties and gas separation performance of PBI-polyimides blend carbon molecular sieve membranes via optimization of the pyrolysis process. Sep. Purif. Technol. 2014, 122, 278–289. [Google Scholar] [CrossRef]
- Singh, R.; Koros, W.J. Carbon molecular sieve membrane performance tuning by dual temperature secondary oxygen doping (DTSOD). J. Membr. Sci. 2013, 427, 472–478. [Google Scholar] [CrossRef]
- Xu, L.; Rungta, M.; Koros, W.J. Matrimid® derived carbon molecular sieve hollow fiber membranes for ethylene/ethane separation. J. Membr. Sci. 2011, 380, 138–147. [Google Scholar] [CrossRef]
- Li, N.; Fane, A.; Winston, W.S.; Matsuura, T. Advanced Membrane Technology and Applications; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011. [Google Scholar]
- Ismail, F.A.; Rana, D.; Matsuura, T.; Foley, H. Carbon-Based Membranes for Separation Processes; Springer: New York, NY, USA, 2011. [Google Scholar]
- Gilron, J.; Soffer, A. Knudsen diffusion in microporous carbon membranes with molecular sieving character. J. Membr. Sci. 2002, 209, 339–352. [Google Scholar] [CrossRef]
- Cheng, L.H.; Fu, Y.J.; Liao, K.S.; Chen, J.T.; Hu, C.C.; Hung, W.S.; Lee, K.R.; Lai, J.Y. A high-permeance supported carbon molecular sieve membrane fabricated by plasma-enhanced chemical vapor deposition followed by carbonization for CO2 capture. J. Membr. Sci. 2014, 460, 1–8. [Google Scholar] [CrossRef]
- Geiszler, V.C.; Koros, W.J. Effects of polyimide pyrolysis conditions on carbon molecular sieve membrane properties. Ind. Eng. Chem. Res. 1996, 35, 2999–3003. [Google Scholar] [CrossRef]
- Centeno, T.; Vilas, J.; Fuertes, A. Effects of phenolic resin pyrolysis conditions on carbon membrane performance for gas separation. J. Membr. Sci. 2004, 228, 45–54. [Google Scholar] [CrossRef]
- Barsema, J.; Klijnstra, S.; Balster, J.; Vandervegt, N.; Koops, G.; Wessling, M. Intermediate polymer to carbon gas separation membranes based on Matrimid PI. J. Membr. Sci. 2004, 238, 93–102. [Google Scholar] [CrossRef]
- Han, S.H.; Kim, G.W.; Jung, C.H.; Lee, Y.M. Control of pore characteristics in carbon molecular sieve membranes (CMSM) using organic/inorganic hybrid materials. Desalination 2008, 233, 88–95. [Google Scholar] [CrossRef]
- Chen, J.; Loo, L.S.; Wang, K.; Do, D.D. The structural characterization of a CMS membrane using Ar sorption and permeation. J. Membr. Sci. 2009, 335, 1–4. [Google Scholar] [CrossRef]
- Llosa Tanco, M.A.; Pacheco Tanaka, D.A.; Rodrigues, S.C.; Texeira, M.; Mendes, A. Composite-alumina-carbon molecular sieve membranes prepared from novolac resin and boehmite. Part I: Preparation, characterization and gas permeation studies. Int. J. Hydrog. Energy 2015, 40, 5653–5663. [Google Scholar] [CrossRef]
- Williams, P.; Koros, W. Gas Separation by Carbon Membranes. In Advanced Membrane Technology and Applications; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008. [Google Scholar]
- Fuertes, A.B. Effect of air oxidation on gas separation properties of adsorption-selective carbon membranes. Carbon 2001, 39, 697–706. [Google Scholar] [CrossRef]
- Teixeira, M.; Campo, M.C.; Pacheco Tanaka, D.A.; Llosa Tanco, M.A.; Magen, C.; Mendes, A. Composite phenolic resin-based carbon molecular sieve membranes for gas separation. Carbon 2011, 49, 4348–4358. [Google Scholar] [CrossRef]
- Zhang, B.; Li, L.; Wang, C.; Pang, J.; Zhang, S.; Jian, X.; Wang, T.; Koros, W.J. Effect of membrane-casting parameters on the microstructure and gas permeation of carbon membranes. RSC Adv. 2015, 5, 60345–60353. [Google Scholar] [CrossRef]
- Ma, X.; Swaidan, R.; Teng, B.; Tan, H.; Salinas, O.; Litwiller, E.; Han, Y.; Pinnau, I. Carbon molecular sieve gas separation membranes based on an intrinsically microporous polyimide precursor. Carbon 2013, 62, 88–96. [Google Scholar] [CrossRef]
- Salinas, O.; Ma, X.; Litwiller, E.; Pinnau, I. High-performance carbon molecular sieve membranes for ethylene/ethane separation derived from an intrinsically microporous polyimide. J. Memb. Sci. 2016, 500, 115–123. [Google Scholar] [CrossRef]
- Yin, X.; Wang, J.; Chu, N.; Yang, J.; Lu, J.; Zhang, Y.; Yin, D. Zeolite L/carbon nanocomposite membranes on the porous alumina tubes and their gas separation properties. J. Membr. Sci. 2010, 348, 181–189. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, T.; Qiu, J.; Cao, Y. A novel carbon/ZSM-5 nanocomposite membrane with high performance for oxygen/nitrogen separation. Chem. Commun. 2006, 1230–1232. [Google Scholar] [CrossRef] [PubMed]
- Foley, H.C.; Rajagopalan, R.; Merritt, A.R. Carbon Nanocomposite Membranes and Methods for Their Fabrication. U.S. Patent 7,708,810 B2, 4 May 2010. [Google Scholar]
- Barsema, J.; Balster, J.; Jordan, V.; van der Vegt, N.F.; Wessling, M. Functionalized carbon molecular sieve membranes containing Ag-nanoclusters. J. Membr. Sci. 2003, 219, 47–57. [Google Scholar] [CrossRef]
- Barsema, J.N.; van der Vegt, N.F.; Koops, G.H.; Wessling, M. Ag-functionalized carbon molecular-sieve membranes based on polyelectrolyte/polyimide blend precursors. Adv. Funct. Mater. 2005, 15, 69–75. [Google Scholar] [CrossRef]
- Xiao, Y.; Chng, M.L.; Chung, T.S.; Toriida, M.; Tamai, S.; Chen, H.; Jean, Y.C.J. Asymmetric structure and enhanced gas separation performance induced by in situ growth of silver nanoparticles in carbon membranes. Carbon 2010, 48, 408–416. [Google Scholar] [CrossRef]
- Teixeira, M.; Rodrigues, S.; Campo, M.; Pacheco Tanaka, D.A.; Llosa Tanco, M.A.; Madeira, L.; Sousa, J.; Mendes, A. Boehmite-phenolic resin carbon molecular sieve membranes-Permeation and adsorption studies. Chem. Eng. Res. Des. 2014, 92, 2668–2680. [Google Scholar] [CrossRef]
- Llosa Tanco, M.A.; Pacheco Tanaka, D.A.; Mendes, A. Composite-alumina-carbon molecular sieve membranes prepared from novolac resin and boehmite. Part II: Effect of the carbonization temperature on the gas permeation properties. Int. J. Hydrog. Energy 2014, 40, 3485–3496. [Google Scholar] [CrossRef]
- Teixeira, M.; Campo, M.; Pacheco Tanaka, D.A.; Llosa Tanco, M.A.; Magen, C.; Mendes, A. Carbon-Al2O3-Ag composite molecular sieve membranes for gas separation. Chem. Eng. Res. Des. 2012, 90, 2338–2345. [Google Scholar] [CrossRef]
- Li, L.; Wang, C.; Wang, N.; Cao, Y.; Wang, T. The preparation and gas separation properties of zeolite/carbon hybrid membranes. J. Mater. Sci. 2015, 50, 2561–2570. [Google Scholar] [CrossRef]
- Zhang, B.; Jiang, Y.; Wu, Y.; Lu, Y.; Zhao, D.; Wang, T. Fabrication of zeolite hybrid supported carbon membranes with high hydrogen permselective performance. J. Inorg. Mater. 2016, 31, 257–262. [Google Scholar] [CrossRef]
- Zhao, X.; Li, W.; Huang, Z.; Liu, S. Synthesis of nickel-incorporated larch-based carbon membranes with controllable porous structure for gas separation. J. Nanopart. Res. 2015, 17, 433. [Google Scholar] [CrossRef]
- De Vos, R.; Maier, W.; Verweij, H. Hydrophobic silica membrane for gas separation. J. Membr. Sci. 1999, 158, 277–288. [Google Scholar] [CrossRef]
- Duke, M.; Diniz da Costa, J.; Lu, G.; Petch, M.; Gray, P. Carbonised template molecular sieve silica membranes in fuel processing systems: Permeation, hydrostability and regeneration. J. Membr. Sci. 2004, 241, 325–333. [Google Scholar] [CrossRef]
- Kim, S.J.; Park, Y.I.; Nam, S.E.; Park, H.; Lee, P.S. Separations of F-gases from nitrogen through thin carbon membranes. Sep. Purif. Technol. 2016, 158, 108–114. [Google Scholar] [CrossRef]
- Sazali, N.; Salleh, W.N.W.; Nordin, N.A.; Ismail, A.F. Matrimid-based carbon tubular membrane: Effect of carbonization environment. J. Ind. Eng. Chem. 2015, 32, 167–171. [Google Scholar] [CrossRef]
- Lee, P.S.; Kim, D.; Nam, S.E.; Bhave, R.R. Carbon molecular sieve membranes on porous composite tubular supports for high performance gas separations. Microporous Mesoporous Mater. 2016, 224, 332–338. [Google Scholar] [CrossRef]
- Favvas, E.P.; Romanos, G.E.; Katsaros, F.K.; Stefanopoulos, K.L.; Papageorgiou, S.K.; Mitropoulos, A.C.; Kanellopoulos, N.K. Gas permeance properties of asymmetric carbon hollow fiber membranes at high feed pressures. J. Nat. Gas Sci. Eng. 2016, 31, 842–851. [Google Scholar] [CrossRef]
- Song, Y.; Wang, D.; Birkett, G.; Martens, W.; Duke, M.; Smart, S.; Diniz da Costa, J. Mixed matrix carbon molecular sieve and alumina (CMS-Al2O3) membranes. Sci. Rep. 2016, 6, 30703. [Google Scholar] [CrossRef] [PubMed]
- Hennis, J.; Tripodi, M. Composite hollow fiber membranes for gas separation, the resistance model approach. J. Membr. Sci. 1981, 8, 233–246. [Google Scholar] [CrossRef]
- Kaltenborn, N.; Muller, S.; Richter, H.; Voigt, I. Structure of nanoporous carbon membranes for gas separation. In Proceedings of the 5th International Zeolite Membrane Meeting, Loutraki, Greece, 23–26 May 2010.
- Voss, H.; Joerg, F.; Therre, W.; Kaemnitz, K. Process for Producing Carbon Membranes. U.S. Patent 8,608,837 B2, 17 December 2013. [Google Scholar] [CrossRef]
- Voss, H.; Therre, J.; Kaltenborn, N.; Richter, H.; Voigt, I. Process for Producing Carbon Membranes. U.S. Patent 8,608,828 B2, 17 December 2013. [Google Scholar]
- Reger-Wagner, N. Carbon Membranes. Available online: http://www.ikts.fraunhofer.de/en/departments/environmental_process_engineering/nanoporous_membranes/carbon-based_membranes/carbon_membranes.html (accessed on 7 July 2016).
- Campo, M.; Magalhães, F.; Mendes, A. Carbon molecular sieve membranes from cellophane paper. J. Membr. Sci. 2010, 1–2, 180–188. [Google Scholar] [CrossRef]
- Tseng, H.H.; Itta, A. Modification of carbon molecular sieve membrane structure by self-assisted deposition carbon segment for gas separation. J. Membr. Sci. 2012, 389, 223–233. [Google Scholar] [CrossRef]
- Hosseini, S.; Chung, T. Carbon membranes from blends of PBI and polyimides for N2/CH4 and CO2/CH4 separation and hydrogen purification. J. Membr. Sci. 2009, 328, 174–185. [Google Scholar] [CrossRef]
- Zhou, W.; Yoshino, M.; Kita, H.; Okamoto, K. Preparation and gas permeation properties of carbon molecular sieve membranes based on sulfonated phenolic resin. J. Membr. Sci. 2003, 217, 55–67. [Google Scholar] [CrossRef]
- Pacheco Tanaka, D.A.; Llosa Tanco, M.A.; Okazaki, J.; Wakui, Y.; Mizukami, F.; Suzuki, T. Preparation of “pore-filled” type Pd-YSZ-γ-Al2O3 composite membrane supported on α-Al2O3 tube for hydrogen separation. J. Membr. Sci. 2008, 320, 436–441. [Google Scholar] [CrossRef]
- Low, B.; Xiao, Y.; Chung, T.; Liu, Y. Simultaneous occurrence of chemical grafting, cross.linking, and etching on the surface of polyimide membranes and their impact on H2/CO2 separation. Macromolecules 2008, 41, 1297–1309. [Google Scholar] [CrossRef]
- Bux, H.; Liang, F.; Li, Y.; Cravillon, J.; Wiebcke, M.; Caro, J. Zeolitic imidazolate framework membrane with molecular sieving properties by microwave-assisted solvothermal synthesis. J. Am. Chem. Soc. 2009, 131, 16000–16001. [Google Scholar] [CrossRef] [PubMed]
- Mc Carthy, M.; Varela-Guerrero, V.; Barnett, G.; Jeong, H. Synthesis of zeolitic imidazolate framework films and membranes with controlled microstructures. Langmuir 2010, 26, 14636–14641. [Google Scholar] [CrossRef] [PubMed]
- Tseng, H.H.; Wang, C.T.; Zhuang, G.L.; Uchytil, P.; Reznickova, J.; Setnickova, K. Enhanced H2/CH4 and H2/CO2 separation by carbon molecular sieve membrane coated on titania modified alumina support: Effects of TiO2 intermediate layer preparation variables on interfacial adhesion. J. Membr. Sci. 2016, 510, 391–404. [Google Scholar] [CrossRef]
- Ismail, N.; Salleh, W.; Sazali, N.; Ismail, A. The effect of polymer composition on CO2/CH4 separation of supported carbon membrane. Chem. Eng. Trans. 2015, 45, 1465–1470. [Google Scholar] [CrossRef]
- Kruse, N.; Schießer, Y.; Kämnitz, S.; Richter, H.; Voigt, I.; Braun, G.; Repke, J.U. Carbon membrane gas separation of binary CO2 mixtures at high pressure. Sep. Purif. Technol. 2016, 164, 132–137. [Google Scholar] [CrossRef]
- Kusuki, Y.; Shimazaki, H.; Tanihara, N.; Nakanishi, S.; Yoshinaga, T. Gas permeation properties and characterization of asymmetric carbon membranes prepared by pyrolyzing asymmetric polyimide hollow fiber membrane. J. Membr. Sci. 1997, 134, 245–253. [Google Scholar] [CrossRef]
- Tanihara, N.; Shimazaki, H.; Hirayama, Y.; Nakanishi, S.; Yoshinaga, T.; Kusuki, Y. Gas permeation properties of asymmetric carbon hollow fiber membranes prepared from asymmetric polyimide hollow fiber. J. Membr. Sci. 1999, 160, 179–186. [Google Scholar] [CrossRef]
- Zhou, W.; Watari, T.; Kita, H.; Okamoto, K. Gas permeation properties of flexible pyrolytic membranes from sulfonated polyimides. Chem. Lett. 2002, 31, 534–535. [Google Scholar] [CrossRef]
- Islam, M.; Zhou, W.; Honda, T.; Tanaka, K.; Kita, H.; Okamoto, K. Preparation and gas separation performance of flexible pyrolytic membranes by low-temperature pyrolysis of sulfonated polyimides. J. Membr. Sci. 2005, 261, 17–26. [Google Scholar] [CrossRef]
- Acharya, M.; Raich, B.A.; Foley, H.C.; Harold, M.P.; Lerou, J.J. Metal-supported carbogenic molecular sieve membranes: Synthesis and applications. Ind. Eng. Chem. Res. 1997, 36, 2924–2930. [Google Scholar] [CrossRef]
- Merritt, A.; Rajagopalan, R.; Foley, H.C. High performance nanoporous carbon membranes for air separation. Carbon 2007, 45, 1267–1278. [Google Scholar] [CrossRef]
- Yoshimune, M.; Haraya, K. CO2/CH4 mixed gas separation using carbon hollow fiber membranes. Energy Procedia 2013, 37, 1109–1116. [Google Scholar] [CrossRef]
- Lagorsse, S.; Magalhães, F.; Mendes, A. Carbon molecular sieve membranes: Sorption, kinetic and structural characterization. J. Membr. Sci. 2004, 241, 275–287. [Google Scholar] [CrossRef]
- Lagorsse, S.; Leite, A.; Magalhães, F.; Bischofberger, N.; Rathenow, J.; Mendes, A. Novel carbon molecular sieve honeycomb membrane module: Configuration and membrane characterization. Carbon 2005, 43, 809–819. [Google Scholar] [CrossRef]
- Richter, H.; Voigt, I.; Kaltenborn, N.; Kamnitz, S.; Voss, H.; Terre, J.; Kuhn, J. H2-selective mole sieving carbon membranes. In Proceedings of the 12th ICIM, Enschede, The Netherlands, 10–13 July 2012.
- Parsley, D.; Ciora, R.; Flowers, D.; Laukaitaus, J.; Chen, A.; Liu, P.; Yu, J.; Sahimi, M.; Bonsu, A.; Tsotsis, T.T. Field evaluation of carbon molecular sieve membranes for the separation and purification of hydrogen from coal- and biomass-derived syngas. J. Membr. Sci. 2014, 450, 81–92. [Google Scholar] [CrossRef]
- Media and Process Technology Inc. Available online: http://www.mediaandprocess.com/index.html (accessed on 8 January 2016).
- Kita, H. Gas and vapor separation membranes based on carbon membranes. In Materials Science of Membranes for Gas and Vapor Separation; Yampolskii, Y., Pinnau, I., Freeman, B., Eds.; John Wiley & Sons Ltd.: West Sussex, England, UK, 2006; pp. 337–354. [Google Scholar]
- Dong, Y.; Nakao, M.; Nishiyama, N.; Egashira, Y.; Ueyama, K. Gas permeation and pervaporation of water/alcohols through the microporous carbon membranes prepared from resorcinol/formaldehyde/quaternary ammonium compounds. Sep. Purif. Technol. 2010, 73, 2–7. [Google Scholar] [CrossRef]
- Tanaka, S.; Yasuda, T.; Katayama, Y.; Miyake, Y. Pervaporation dehydration performance of microporous carbon membranes prepared from resorcinol/formaldehyde polymer. J. Membr. Sci. 2011, 379, 52–59. [Google Scholar] [CrossRef]
- Yoshimune, M.; Mizoguchi, K.; Haraya, K. Alcohol dehydration by pervaporation using a carbon hollow fiber membrane derived from sulfonated poly(phenylene oxide). J. Membr. Sci. 2013, 425–426, 149–155. [Google Scholar] [CrossRef]
- Richter, H.; Kamnitz, S.; Gunther, C.; Weyd, M.; Jager, B.; Richter, J.; Voigt, I. High temperature water separation with nanoporous inorganic membranes. In Proceedings of the 13th International Conference on Inorganic Membranes, Brisbane, Australia, 6–9 July 2014.
- Tin, P.; Yi, H.; Chin, R.; Chung, T. Carbon molecular sieve membranes for biofuel separation. Carbon 2011, 49, 369–375. [Google Scholar] [CrossRef]
- Liao, K.; Fu, Y.; Hu, C.; Chen, J.; Lin, D.; Lee, K.; Tung, K.; Jean, Y.; Lai, J. Microstructure of carbon molecular sieve membranes and their application to separation of aqueous bioethanol. Carbon 2012, 50, 4220–4227. [Google Scholar] [CrossRef]
- Elma, M.; Wang, D.; Yacou, C.; Diniz da Costa, J. Interlayer-free P123 carbonised template silica membranes for desalination with reduced salt concentration polarisation. J. Membr. Sci. 2015, 475, 376–383. [Google Scholar] [CrossRef]
- Gallucci, F.; Fernandez, E.; Corengia, P.; van Sint Annaland, M. Recent advances on membranes and membrane reactors for hydrogen production. Chem. Eng. Sci. 2013, 92, 40–66. [Google Scholar] [CrossRef]
- Itoh, N.; Haraya, K. A carbon membrane reactor. Catal. Today. 2000, 56, 103–111. [Google Scholar] [CrossRef]
- Bi, Y.; Xu, H.; Li, W.; Goldbach, A. Water-gas shift reaction in a Pd membrane reactor over Pt/Ce0.6Zr0.4O2 catalyst. Int. J. Hydrog. Energy 2009, 34, 2965–2971. [Google Scholar] [CrossRef]
- Brunetti, A.; Barbieri, G.; Drioli, E. Upgrading of a syngas mixture for pure hydrogen production in a Pd-Ag membrane reactor. Chem. Eng. Sci. 2009, 64, 3448–3454. [Google Scholar] [CrossRef]
- Kim, S.; Xu, Z.; Reddy, G.; Smirniotis, P.; Dong, J. Effect of pressure on high-temperature water gas shift reaction in microporous zeolite membrane reactor. Ind. Eng. Chem. Res. 2012, 51, 1364–1375. [Google Scholar] [CrossRef]
- Harale, A.; Hwang, H.; Liu, P.; Sahimi, M.; Tsotsis, T.T. Experimental studies of a hybrid adsorbent-membrane reactor (HAMR) system for hydrogen production. Chem. Eng. Sci. 2007, 62, 4126–4137. [Google Scholar] [CrossRef]
- Liu, P.; Sahimi, M.; Tsotsis, T.T. Process intensification in hydrogen production from coal and biomass via the use of membrane-based reactive separations. Curr. Opin. Chem. Eng. 2012, 1, 342–351. [Google Scholar] [CrossRef]
- Zhang, B.; Zhao, D.; Shen, G.; Yu, Z.; Wu, Y.; Wang, T. Fabrication of phenolic resin-based carbon membranes for intensification of hydrogen production reaction from methanol. J. Shenyang Univ. Technol. 2014, 36, 503–508. [Google Scholar]
- Zhang, B.; Zhao, D.; Wu, Y.; Liu, H.; Wang, T.; Qiu, J. Fabrication and application of catalytic carbon membranes for hydrogen production from methanol steam reforming. Ind. Eng. Chem. Res. 2015, 54, 623–632. [Google Scholar] [CrossRef]
- Sá, S.; Silva, H.; Sousa, J.; Mendes, A. Hydrogen production by methanol steam reforming in a membrane reactor: Palladium vs carbon molecular sieve membranes. J. Membr. Sci. 2009, 339, 160–170. [Google Scholar] [CrossRef]
- Gimeno, M.; Wu, Z.; Soler, J.; Herguido, J.; Li, K.; Menéndez, M. Combination of a Two-Zone Fluidized Bed Reactor with a Pd hollow fibre membrane for catalytic alkane dehydrogenation. Chem. Eng. J. 2009, 155, 298–303. [Google Scholar] [CrossRef]
- Matsumura, Y.; Tong, J. Methane steam reforming in hydrogen-permeable membrane reactor for pure hydrogen production. Top. Catal. 2008, 51, 123–132. [Google Scholar] [CrossRef]
- Battersby, S.; Duke, M.; Liu, S.; Rudolph, V.; da Costa, J.C.D. Metal doped silica membrane reactor: Operational effects of reaction and permeation for the water gas shift reaction. J. Membr. Sci. 2008, 316, 46–52. [Google Scholar] [CrossRef]
- Harale, A.; Hwang, H.; Liu, P.; Sahimi, M.; Tsotsis, T.T. Design aspects of the cyclic hybrid adsorbent-membrane reactor (HAMR) system for hydrogen production. Chem. Eng. Sci. 2010, 65, 427–435. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, H.; Zhu, Y.; Zhu, S. Methanol steam reforming to hydrogen in a carbon membrane reactor system. Ind. Eng. Chem. Res. 2006, 45, 7997–8001. [Google Scholar] [CrossRef]
- Sá, S.; Sousa, J.; Mendes, A. Steam reforming of methanol over a CuO/ZnO/Al2O3 catalyst, part I: Kinetic modelling. Chem. Eng. Sci. 2011, 66, 4913–4921. [Google Scholar] [CrossRef]
- Briceño, K.; Iulianelli, A.; Montané, D.; Garcia-Valls, R.; Basile, A. Carbon molecular sieve membranes supported on non-modified ceramic tubes for hydrogen separation in membrane reactors. Int. J. Hydrog. Energy 2012, 37, 13536–13544. [Google Scholar] [CrossRef]
- Wollbrink, A.; Volgmann, K.; Koch, J.; Kanthasamy, K.; Tegenkamp, C.; Li, Y.; Richter, H.; Kämnitz, S.; Steinbach, F.; Feldhoff, A.; et al. Amorphous, turbostratic and crystalline carbon membranes with hydrogen selectivity. Carbon 2016, 106, 93–105. [Google Scholar] [CrossRef]
- Hirota, Y.; Ishikado, A.; Uchida, Y.; Egashira, Y.; Nishiyama, N. Pore size control of microporous carbon membranes by post-synthesis activation and their use in a membrane reactor for dehydrogenation of methylcyclohexane. J. Membr. Sci. 2013, 440, 134–139. [Google Scholar] [CrossRef]

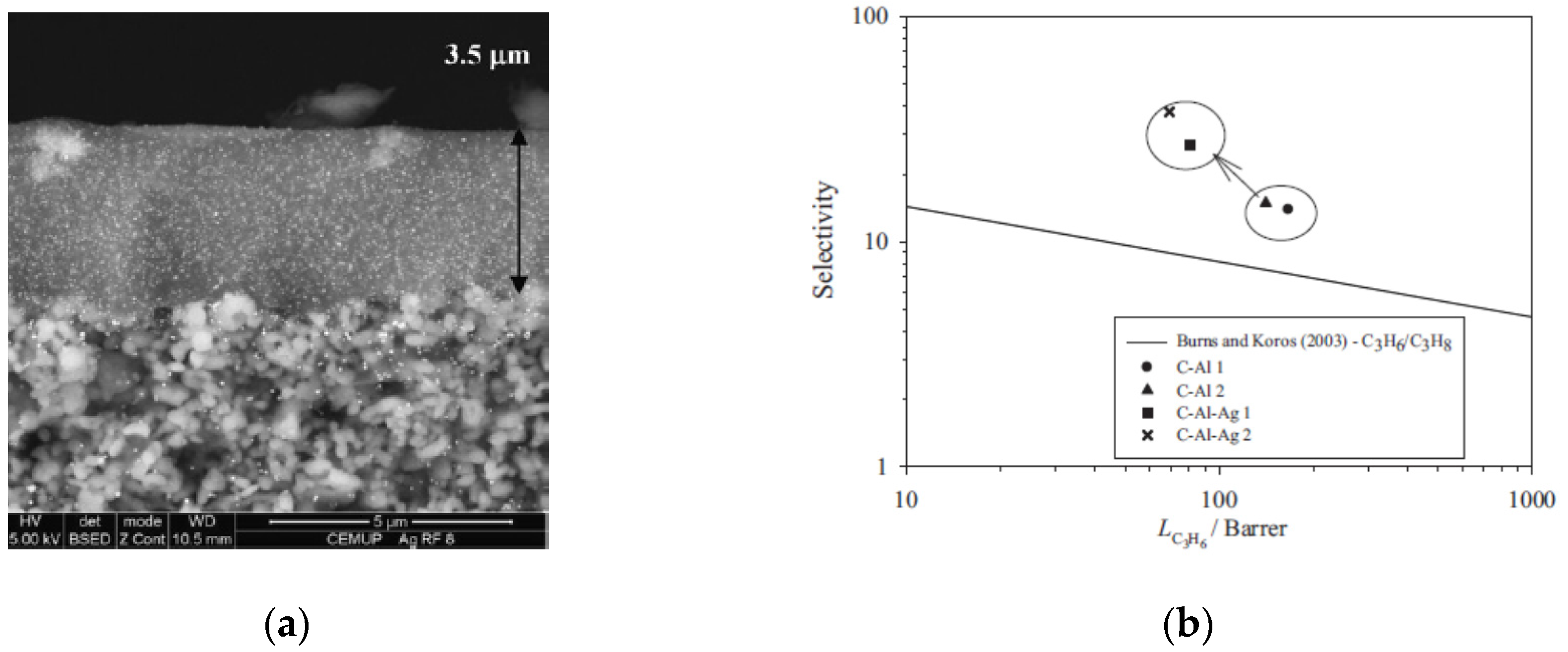
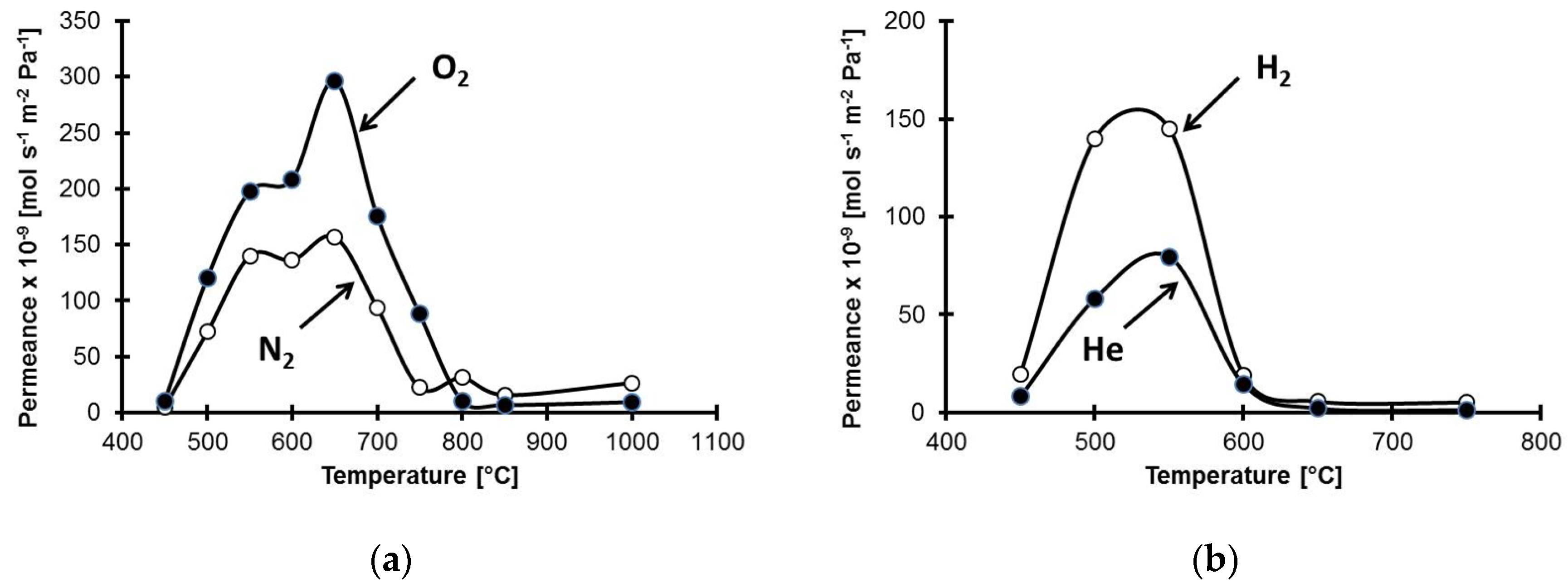
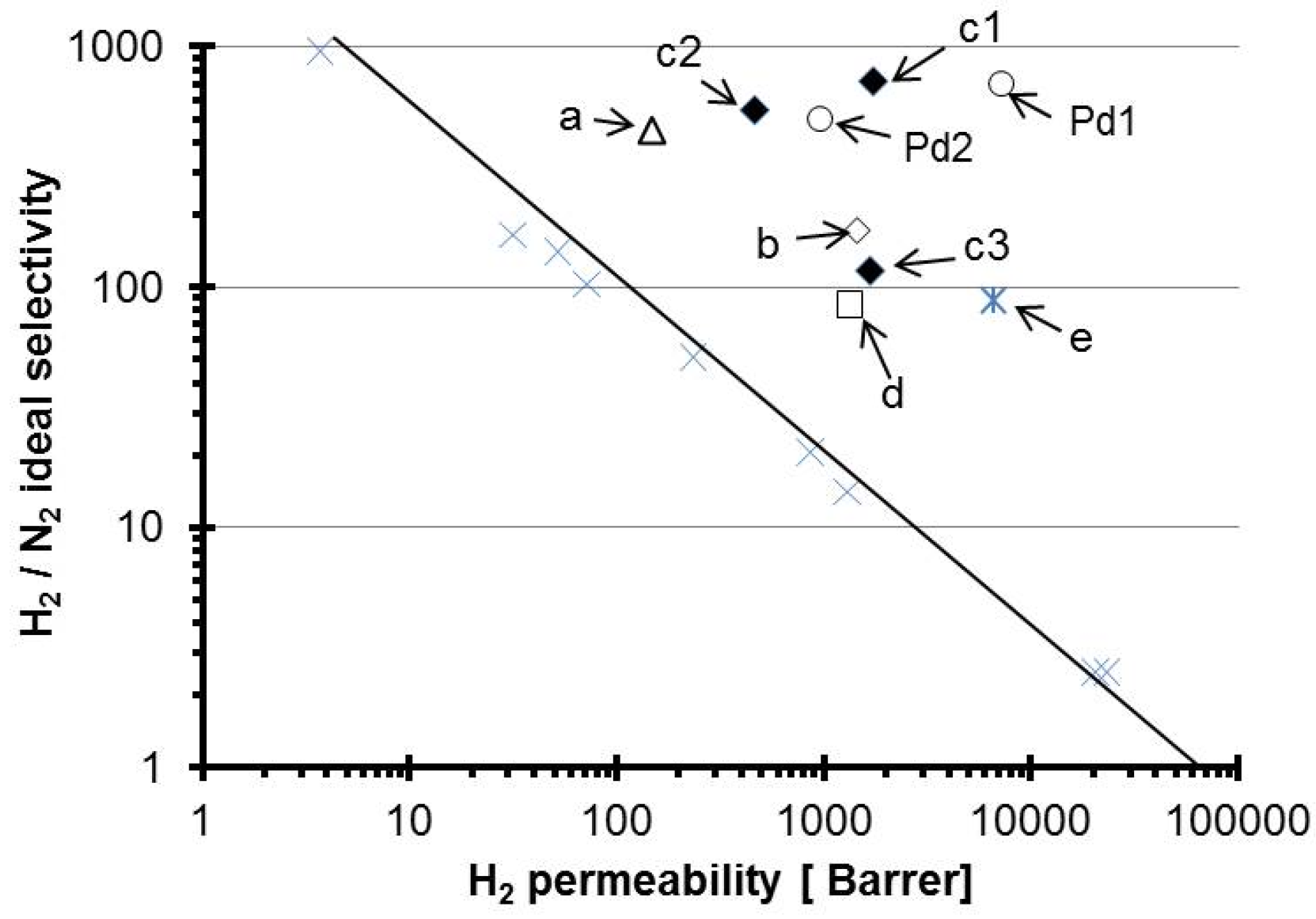
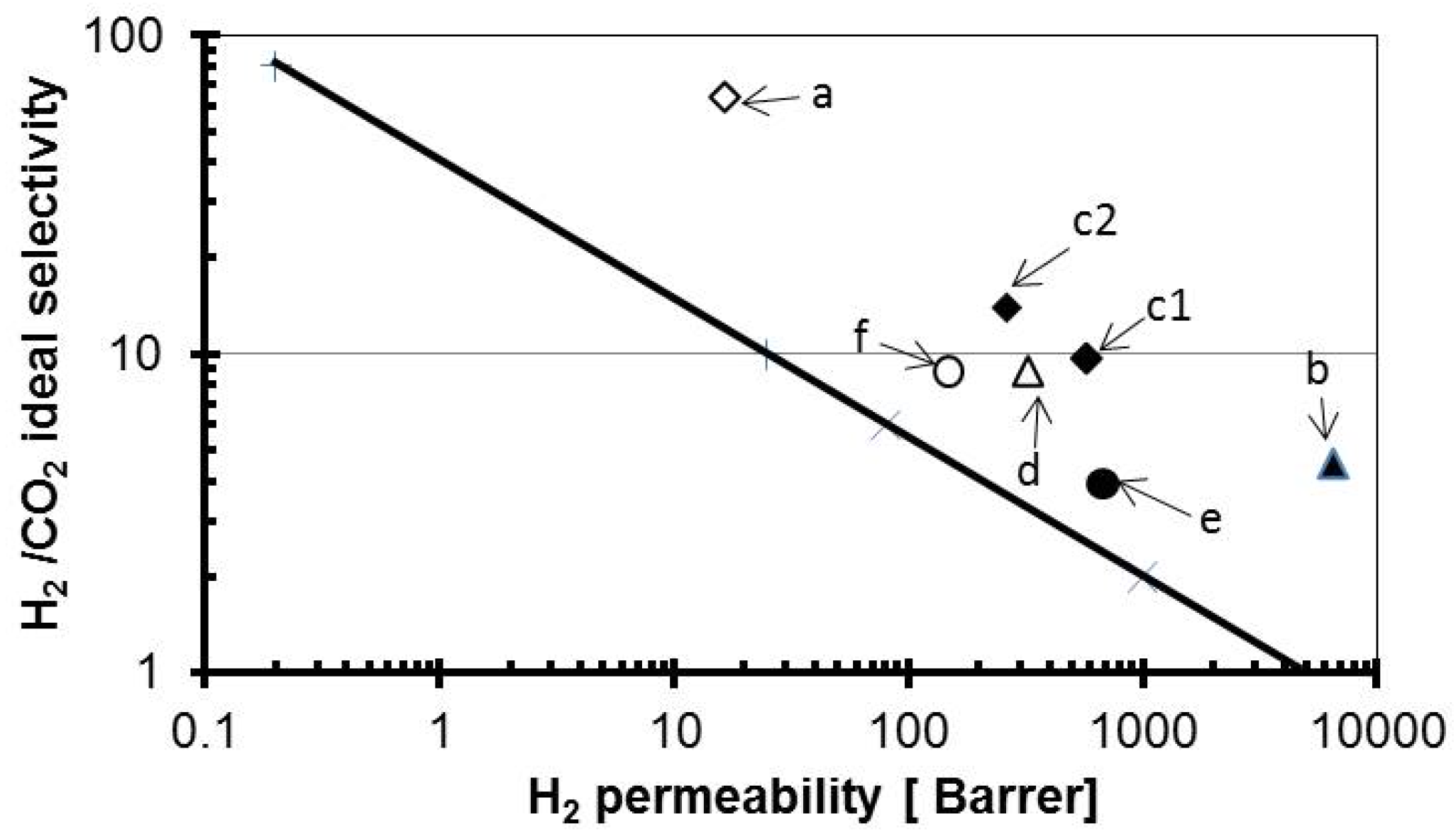

| Membrane c/Alumina (wt.%) | Permeability (Barrer) | ||||||
|---|---|---|---|---|---|---|---|
| N2 | O2 | He | H2 | CO2 | C3H8 | C3H6 | |
| 7.3 | 32.7 | 153 | 971 | 2047 | 1148 | 1.96 | 36.2 |
| 4.4 | 58.2 | 256 | 1024 | - | 1499 | 28 | 420 |
| 3.7 | 83 | 284 | 1200 | - | 2017 | 78.5 | 776 |
| Temperature K | mol·m−2·s−1·Pa−1 × 10−9 | Ideal Permselectivity | |||||
|---|---|---|---|---|---|---|---|
| N2 | O2 | H2 | He | O2/N2 | H2/N2 | He/N2 | |
| 723.15 | 0.4 | 1.0 | 19.7 | 8.2 | 2.5 | 49 | 20.5 |
| 773.15 | 1.2 | 8.5 | 140.0 | 58.2 | 7.1 | 117 | 48.5 |
| 823.15 | 0.2 | 3.0 | 145.0 | 79.5 | 15 | 725 | 398 |
| 873.15 | n.d. | n.d | 18.9 | 14.4 | -- | -- | -- |
| 923.15 | n.d | n.d | 5.6 | 2.1 | -- | -- | -- |
| 1023.15 | n.d | n.d | 5.1 | 1.2 | -- | -- | -- |
| Precursor | Configuration | Catalyst | Reaction | Temperature K | Ref. |
|---|---|---|---|---|---|
| not disclosed USC and MPT *1 | Tubular | Cu/ZnO/Al2O3 | Water gas shift | < 573.15 | [104] [112] |
| Novolac MPT *2 | Module (13–86 tubes) | Cu/ZnO/Al2O3 | Syngas treatment | - | [89] |
| Novolac + PEG | Tubular | Cu/ZnO/Al2O3 | Methanol steam reforming | ~523.15 | [113] |
| Phenolic resin | Supported | Nanosize Cu | Methanol steam reforming | - | [107] |
| Cellulose CM Ltda *3 | Hollow fibers | Cu/ZnO/Al2O3 | Methanol steam reforming | 473.15 | [13] [108] |
| Matrimid 5218 | Asymmetric tube | - | Methanol steam reforming | - | [115] |
| Carbon layer: Amorphous, turbostratic and crystalline | Supported | - | Ethanol steam reforming | - | [116] |
| FFA *4 NGK *5 | Tubular Supported | Pt/Al2O3 | Dehydrogenation of methyl-cyclohexane | - | [117] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Llosa Tanco, M.A.; Pacheco Tanaka, D.A. Recent Advances on Carbon Molecular Sieve Membranes (CMSMs) and Reactors. Processes 2016, 4, 29. https://doi.org/10.3390/pr4030029
Llosa Tanco MA, Pacheco Tanaka DA. Recent Advances on Carbon Molecular Sieve Membranes (CMSMs) and Reactors. Processes. 2016; 4(3):29. https://doi.org/10.3390/pr4030029
Chicago/Turabian StyleLlosa Tanco, Margot A., and David A. Pacheco Tanaka. 2016. "Recent Advances on Carbon Molecular Sieve Membranes (CMSMs) and Reactors" Processes 4, no. 3: 29. https://doi.org/10.3390/pr4030029
APA StyleLlosa Tanco, M. A., & Pacheco Tanaka, D. A. (2016). Recent Advances on Carbon Molecular Sieve Membranes (CMSMs) and Reactors. Processes, 4(3), 29. https://doi.org/10.3390/pr4030029








