1. Introduction
Global warming and fossil fuels scarcity necessitate the transformation of traditional processes towards more sustainable and efficient processes for both energy conversion and chemical transformations. A boost in efficiency for reactive systems can be achieved via the so-called Process Intensification (PI), which is defined as “any chemical engineering development that leads to a substantially smaller, cleaner, safer, and more energy efficient technology” [
1]. The need for more efficient processes, including further flexible engineering designs and, at the same time, increasing the safety and environmental impact of these processes, is pushing the industry to carry out novel research in this field. The chemistry and related sectors have already recognized the benefits of PI and estimate a potential for energy saving of about 1000 kilo tonnes of oil equivalent (ktoe) per year using these processes.
Nowadays, the main activities regarding PI are related to the development of novel technologies with high efficiency as well as accomplishing a reduction in CO
2 emissions to the atmosphere, thus achieving a large positive impact as compared to benchmark technologies [
2]. Within the chemical engineering framework, the development of more active catalysts, more efficient contactors, heat exchangers or gas separation units are of interest as these technologies are involved in most chemical processes. The technology of membrane reactors has been recently developed and has demonstrated a high degree of process intensification where the combination of a membrane based separation and a catalytic chemical reaction take place in one unit [
3,
4,
5,
6,
7,
8,
9]. Membranes can be used in membrane reactors for either selective gas separation from the reaction environment or selective gas feeding into the reaction system [
3]. In equilibrium limited reaction systems such as in fuel reforming or dehydrogenations, the selective separation of a product of the reaction (
i.e., H
2), leads to a fuel conversion beyond the value of the thermodynamic equilibrium, thus achieving higher fuel conversions and direct product separation (a separation unit downstream is not required). The other possible configuration is selective gas addition via the membrane. For instance, in autothermal reforming where oxygen is fed together with the fuel, the dosing of oxygen alongside the membrane instead of direct co-feeding leads to an enhancement of the selectivity of products. In general, almost every catalytic industrial process can potentially be enhanced by the introduction of catalytic membranes and membrane reactors instead of the conventional reactors. According to SusChem (European Technology Platform for Sustainable Chemistry, Strategic Research Agenda 2005) more than 80% of the processes in the chemical industry worth approximately €1,500 billion, depend on catalytic technologies, and one of the shorter-term (5–10 years) objectives of this Platform is to “integrate reactor-catalyst-separation design: integration and intensification of processes require the development of new catalytic concepts which break down the current barriers (for example, low flux in catalytic membranes)” [
10].
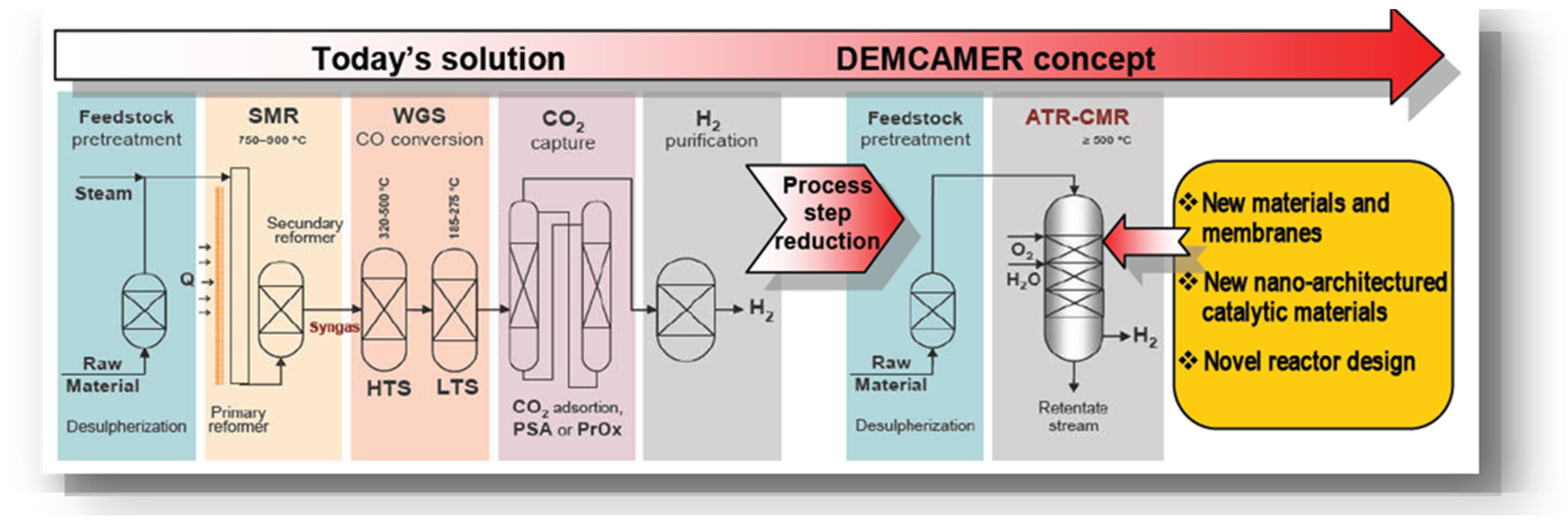
In this work, an overview is presented of the DEMCAMER project, which proposes a solution for many industrial processes that can benefit from membrane reactors: the increase in production rate while maintaining the same product quality as well as reducing both production costs and environmental impact. The objective here is to carry out this action through the implementation of a novel process intensification approach consisting of the combination of reaction and separation in a “Catalytic Membrane Reactor” single unit as is depicted in
Figure 1.
The aim of the DEMCAMER project was to develop innovative multifunctional Catalytic Membrane Reactors (CMR) based on new nano-architectured catalysts and selective membrane materials to improve their performance, cost effectiveness (i.e., reducing the number of steps), and sustainability (lower environmental impact and use of new raw materials) over four selected chemical processes ((Autothermal Reforming (ATR), Fischer-Tropsch (FTS), Water Gas Shift (WGS), and Oxidative Coupling of Methane (OCM)) for pure hydrogen, liquid hydrocarbons, and ethylene production.
The DEMCAMER work plan consisted of activities related to the whole product chain: i.e., development of materials/components (membranes, supports, seals, catalyst...) through integration/validation at lab-scale, until development/validation of four semi-industrial pilot scale CMRs prototypes. Additionally, assessment of environmental, health and safety issues—in relation to the new intensified chemical processes—were also carried out.
For a maximum impact on the European industry this research, covering the complete value chain of catalytic membrane reactors, was carried out with a multidisciplinary and complementary team having the right expertise, including top level European Research Institutes and Universities (8 RES) working together with representative top industries (4 SME and 6 IND) in different sectors (from raw materials to petrochemical end-users).
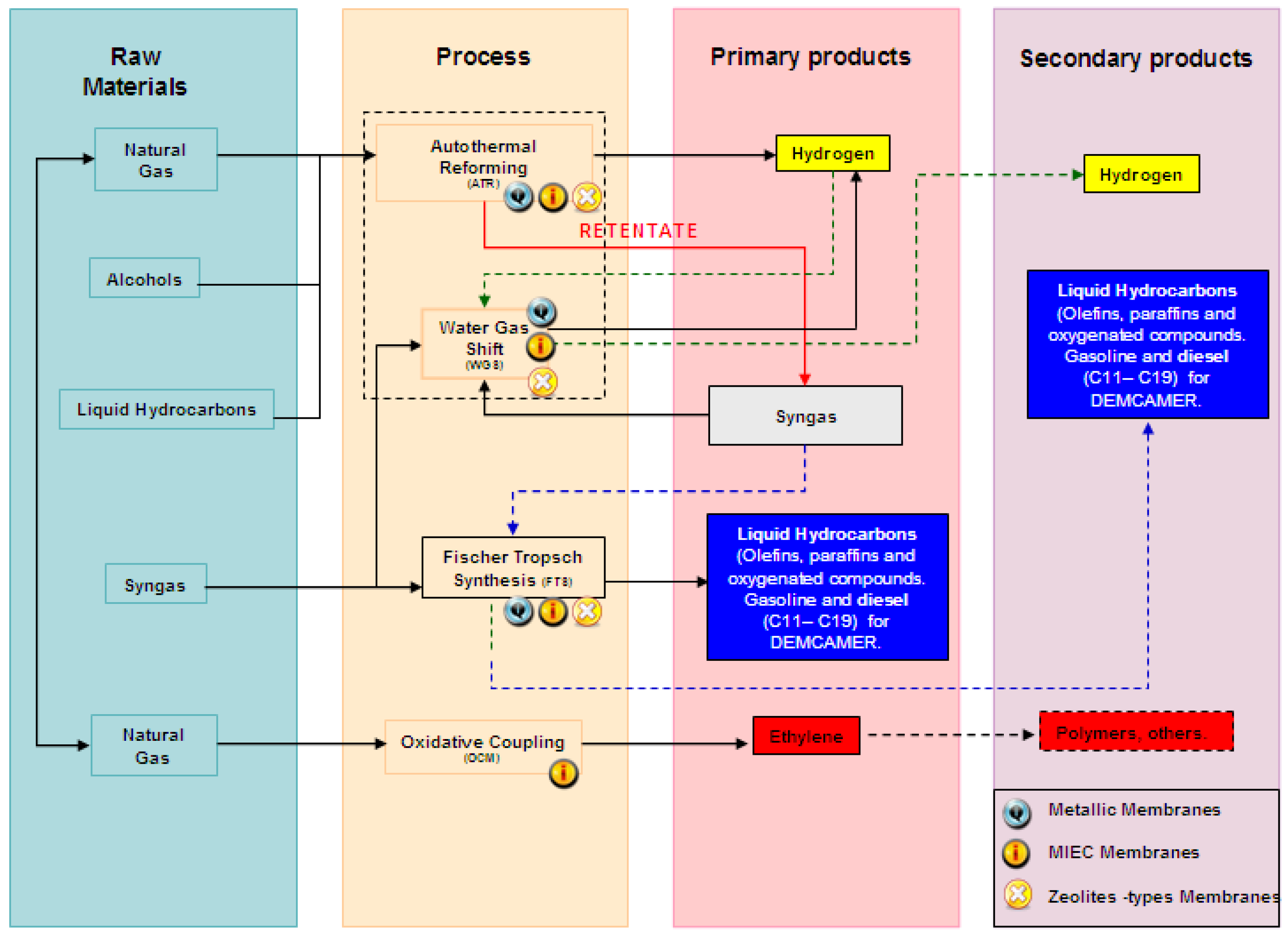
Figure 2 and
Table 1 summarize the four chemical processes addressed by DEMCAMER including the raw materials and products per each targeted reaction. Membranes for the different processes are also indicated in the flow sheet. Both the flow sheet and the table do not take into account previous stages of pre-treatment for the raw materials as this has not been addressed in DEMCAMER.
The raw materials for the ATR process could cover a wide range: natural gas, SNG (Synthetic Natural Gas produced from coal, biomass, petroleum coke, or solid waste), alcohols (methanol and ethanol), dimethyl ether (DME), liquid hydrocarbons derived from fossil fuels (gasoline, diesel, naphtha, etc.), biogas, biofuels (biodiesel, bioethanol, etc.). DEMCAMER only addressed natural gas (mainly CH4) as raw material. On the other hand, Syngas could be the raw material for the WGS and the FTS processes. Syngas could be derived from dry, steam, partial oxidation and autothermal reforming processes using natural gas, coal or biomass. Finally, natural gas, SNG or Biomass could be the raw materials in the OCM process but natural gas was the raw material considered in DEMCAMER.
In this project the targeted primary products were H2, liquid hydrocarbons (gasoline/diesel fractions (C11–C20), and ethylene. The definition and identification of the specific industrial requirements for the intensification of each targeted reaction was addressed at the beginning of the project.
The DEMCAMER Project was funded under FP7 Cooperation Specific Programme and Nanotechnologies, Materials and Processes NMP Theme. The Project started on July 1 2011 and it has run for 48 months. In the following, the main results in terms of catalysts, membranes, and membrane reactors are highlighted.
3. Results and Discussion
In this section the main outcome obtained from the DEMCAMER project is presented. All processes investigated in the DEMCAMER project involved the development and study of different catalysts for the process, membranes, lab-scale facilities, etc. Due to this fact, this section is divided into sub-sections containing the main results obtained in each process.
3.1. Autothermal Reforming of Methane
The whole process was designed starting from the development of the catalyst for the process, which should show good activity under the operating conditions in membrane reactors. In parallel, MIEC membranes were prepared and the stability and performance assessed. Subsequently, the catalyst and membranes were combined for the demonstration of the concept at lab scale and, finally to pilot plant scale.
The developed final generation ATR catalyst achieved the required targets proposed in the project related to activity, selectivity, and stability. The final composition of this catalyst is NiPd/10Ce
0.5Zr0
.5O
2/Al
2O
3, which shows a good performance as summarized in
Table 2, where the target objective and the developed catalyst for ATR are compared. The interaction of Ni and Pd makes a structure suitable for the ATR process [
15]. The effect of the support and loading were compared in terms of both catalytic activity under reacting conditions and characterization, giving as results the developed catalyst abovementioned prepared by sequential impregnation of Ni and Pd, which gives better results as compared to co-impregnation methods.
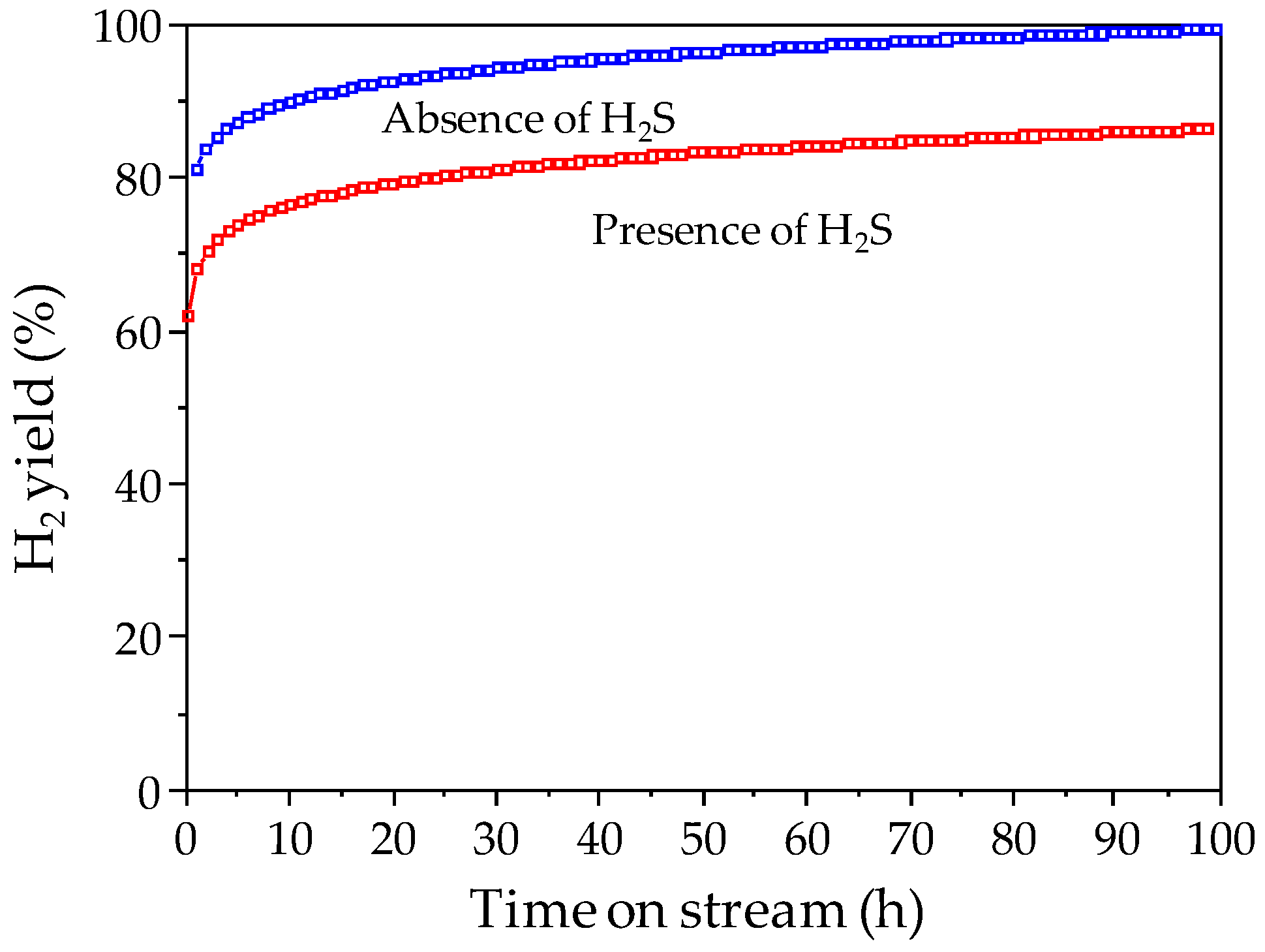
The ATR catalyst shows also stable performance (methane conversion >90%, hydrogen yield >80%) and resistance to deactivation by sulfur and carbon deposition during 100 h of operation under reaction feed containing 50 ppm of H
2S (
Figure 16). Characterization of used catalysts showed excellent resistance to deactivation by carbon formation (no carbonaceous deposits on used catalyst).
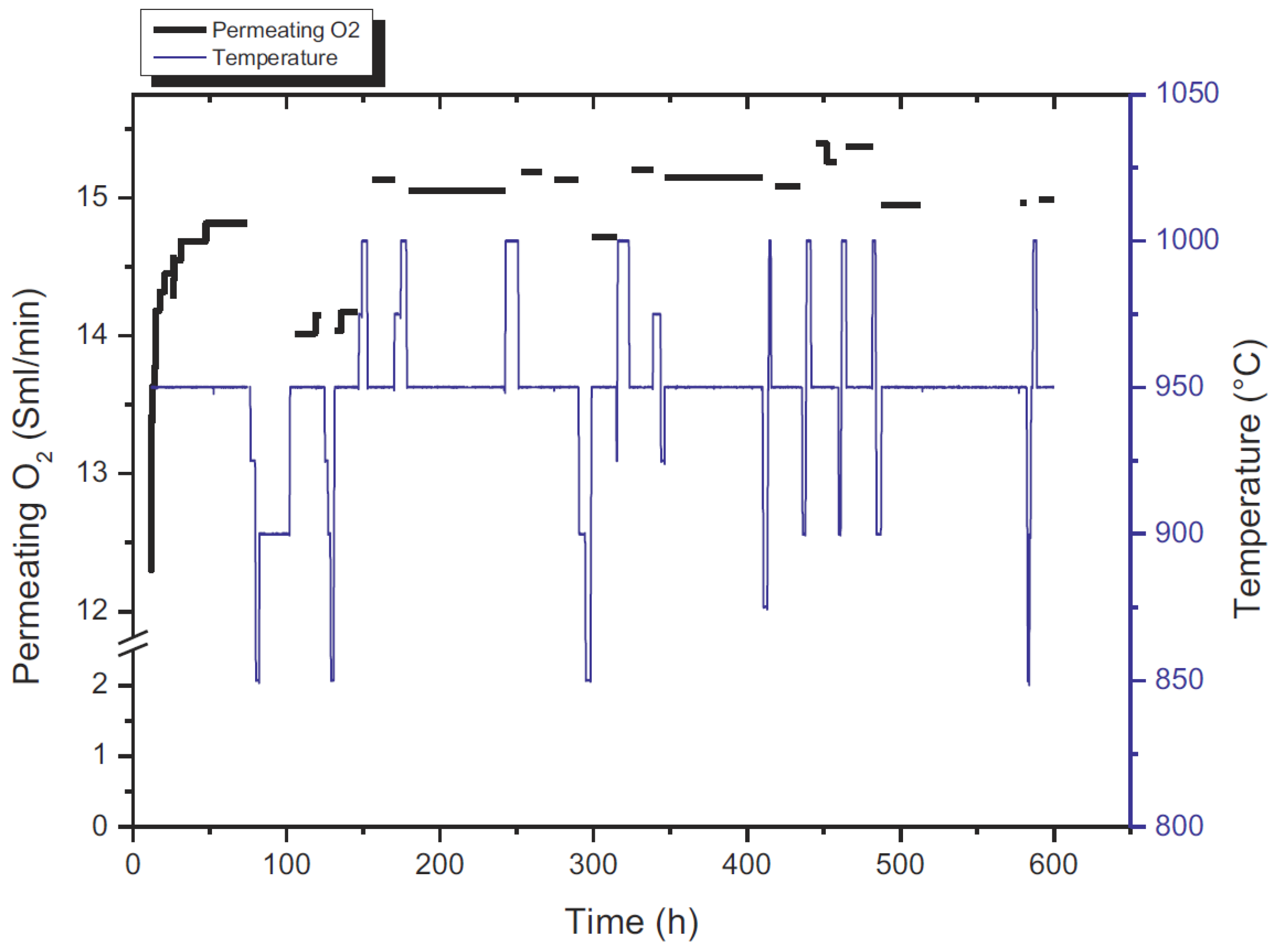
Once the catalysts was designed and tested under reacting conditions, the stability of MIEC membranes was also investigated for selective feeding of O
2 in the reactor. The BCFZ membrane showed high fluxes and good stability for more than 1000 h as reported in
Figure 17, where it can be observed that after a first activation period of around 50 h (due to adjustments in the lattice structure of the MIEC) the membrane flux remained very stable even after repeated cooling down and heating up of the system.
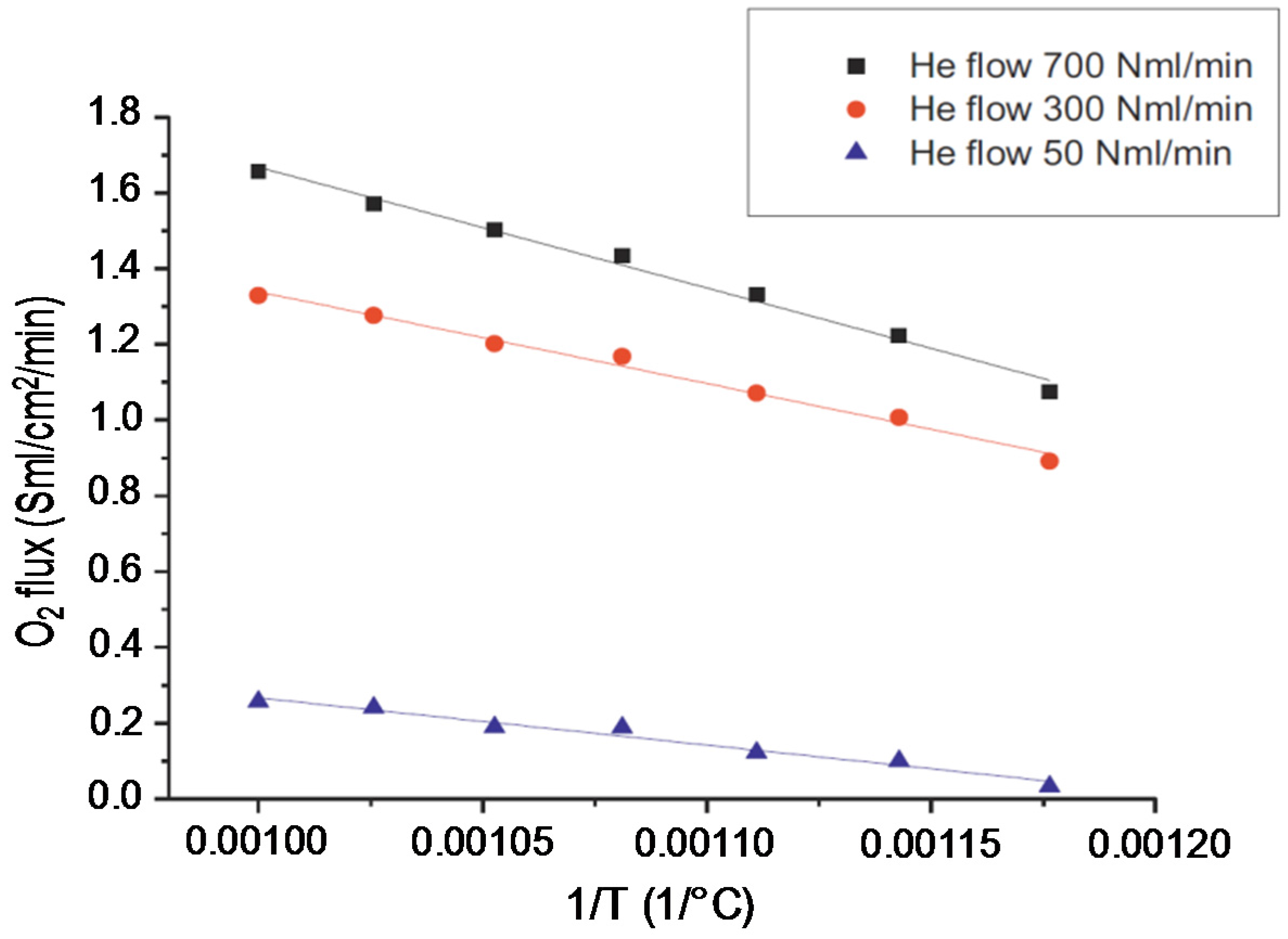
The observed Arrhenius plot for different He flowrates in the permeate side as a function of temperature (
Figure 18) shows a constant slope, which implies that the activation energy is not a function of the sweep gas flow rate, and thus represents the activation energy of the perovskite itself.
This effect was also observed by other researches using the same type of MIEC membranes [
41]. The same membrane was prepared using different thicknesses and non-linearity was observed in terms of O
2 permeation with increasing membrane thickness. This suggests that bulk diffusion of O
2 through the membrane and surface exchange kinetics have a strong effect on the limitation in O
2 permeation.
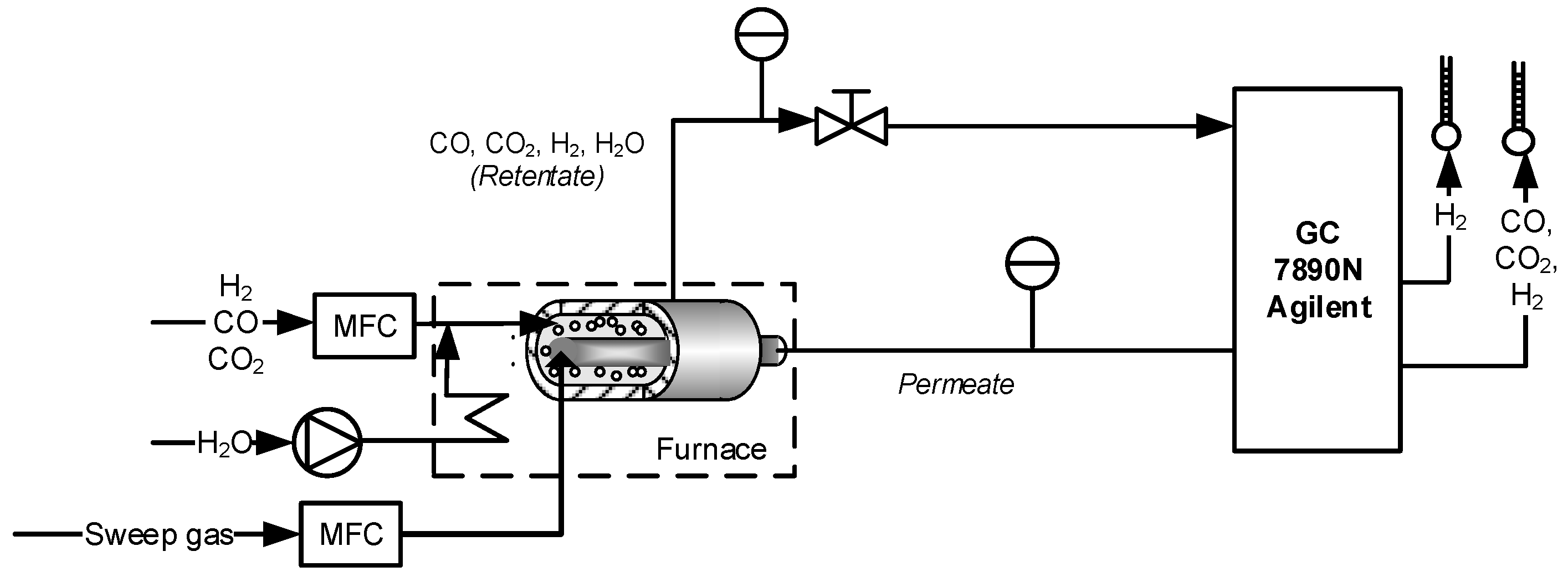
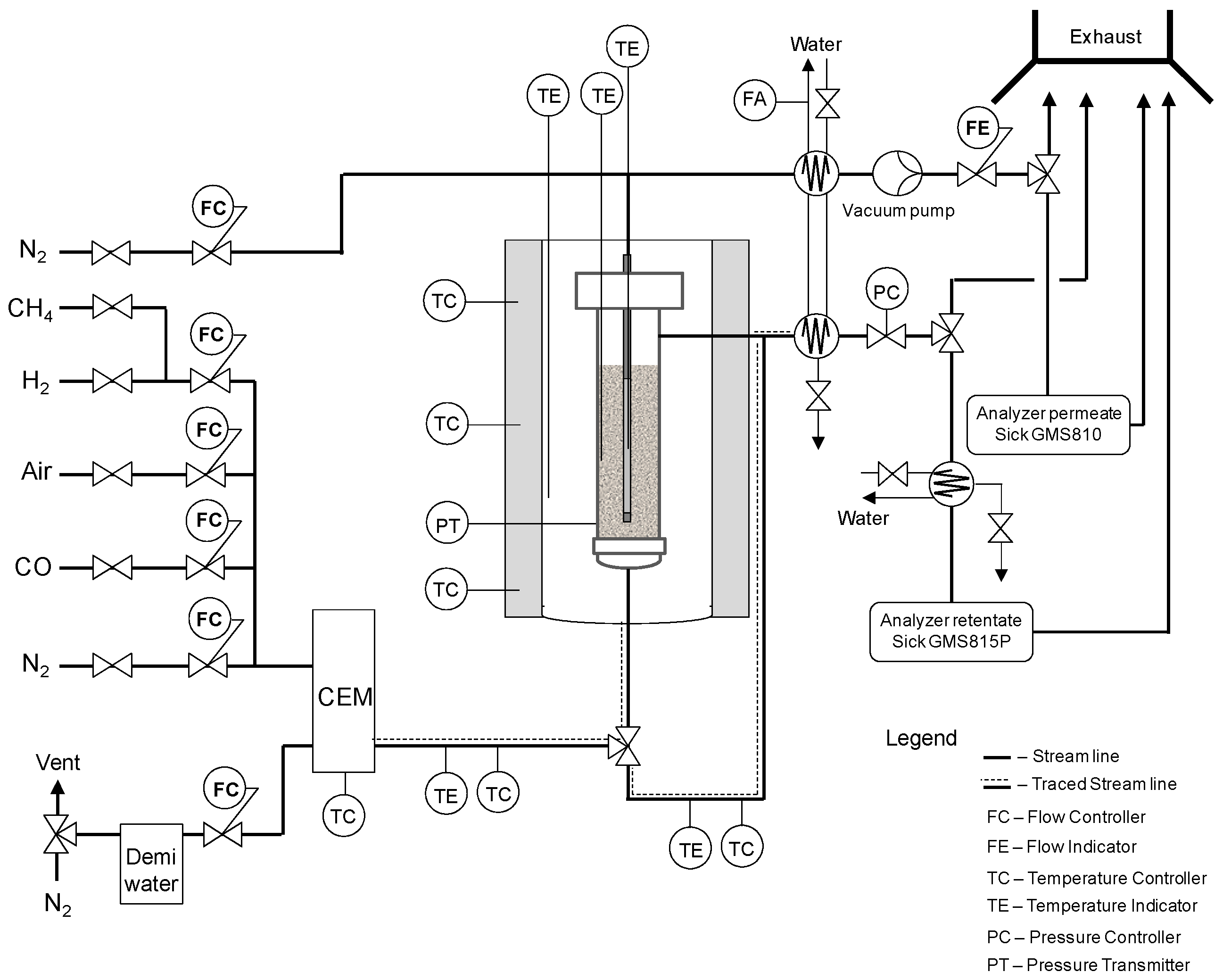
Once the catalyst and membrane were developed and investigated, when both were combined in the lab-scale reactor described in
Section 2.3.1, the membranes were not able to withstand the high temperature process and they showed leakages from the sealing points. Therefore it was not possible to test the ATR process at lab scale.
The last step for the ATR process was the demonstration at prototype scale. The facility constructed at HyGear showed leakage from each bundle equipped with membranes. Due to this fact, it was not possible to feed the oxygen required for the autothermal operating condition. Under this situation the prototype reactor was operated and tested as a natural gas steam reformer. During the testing (ca. one week) the system was stable and allowed the production of up to 3.3 Nm3 of H2 per Nm3 of natural gas feed. The novel developed catalyst exhibited good activity and stability: test results showed almost complete conversion (>99%) and hydrogen productivity higher than 90%.
3.2. Water Gas Shift
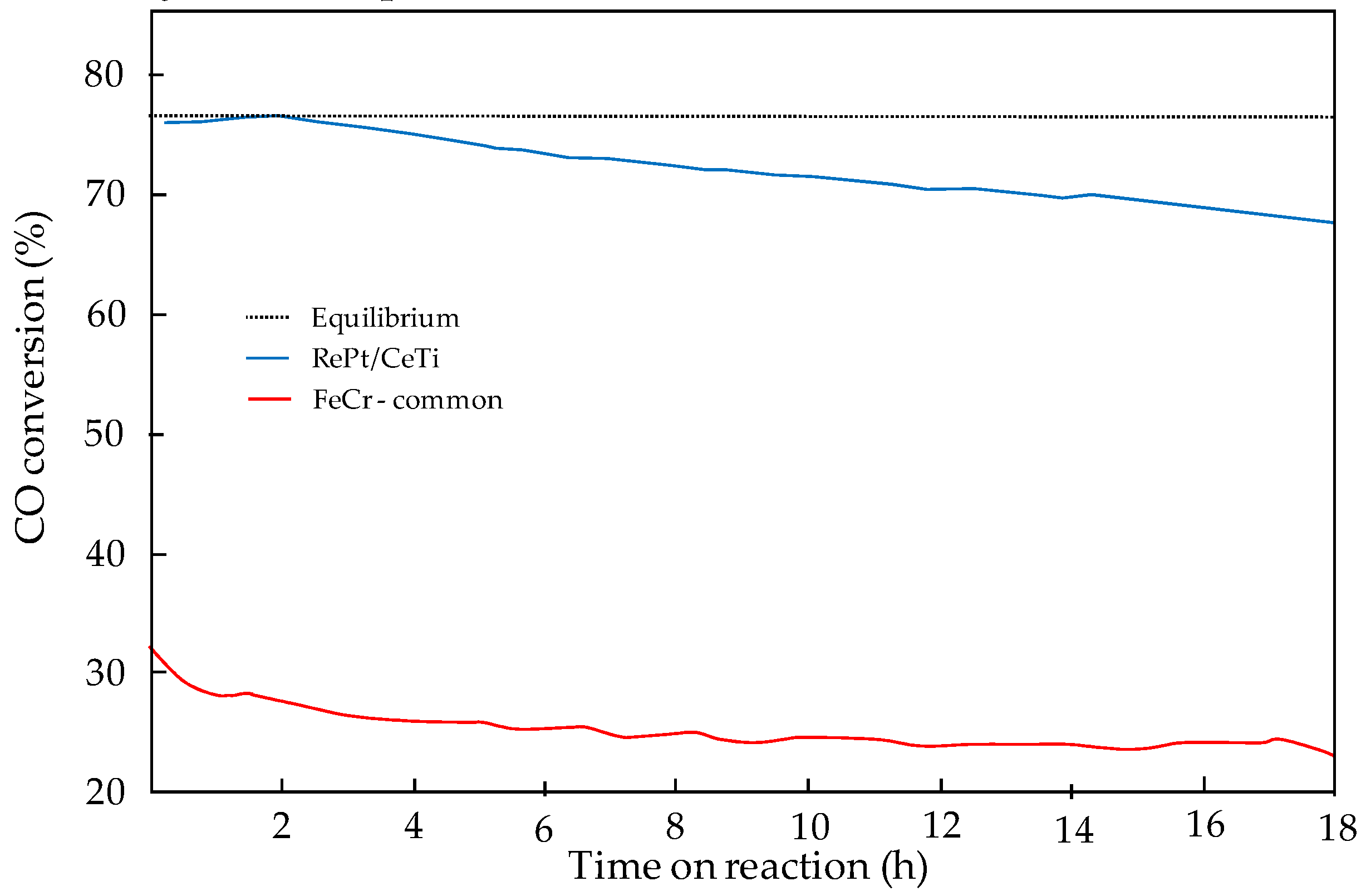
The procedure followed for the development of the WGS process using the DEMCAMER technology was similar to the line-time presented for ATR. The WGS catalyst developed showed very high WGS activity and stability, demonstrating an important improvement with respect to the state-of-the art FeCr HT WGS commercial catalyst (
Figure 19).
Characterization of the catalyst demonstrated that the addition of Re to the Pt-based catalysts implies a modification in the intrinsic WGS activity and also provides high stability over time. The addition of Re is also considered to be responsible for preventing Pt sintering, which would imply a strong decrease in the stability over time observed during the catalytic screening tests. All these characteristics improve the behavior of the catalyst under WGS reacting conditions and, therefore, it was selected as the catalyst for the process.
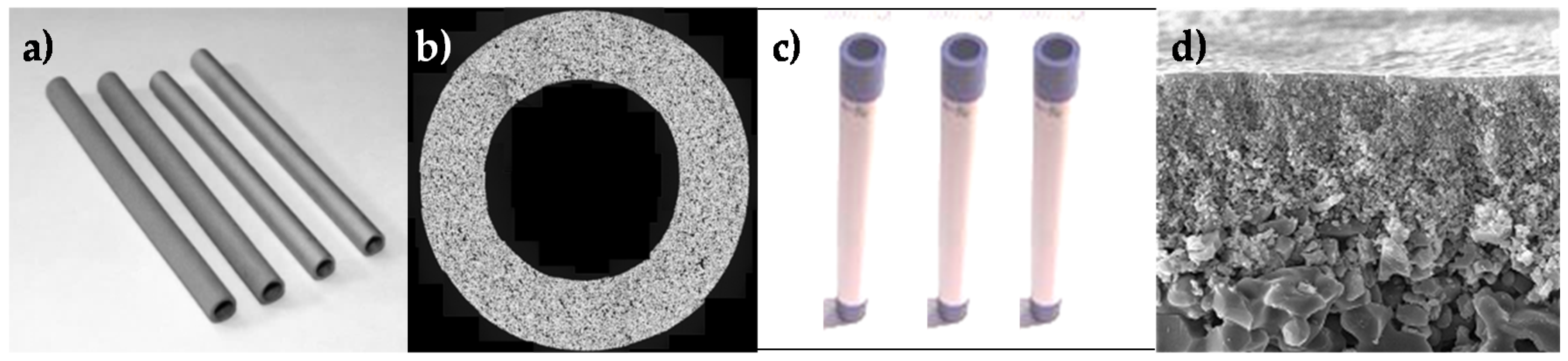
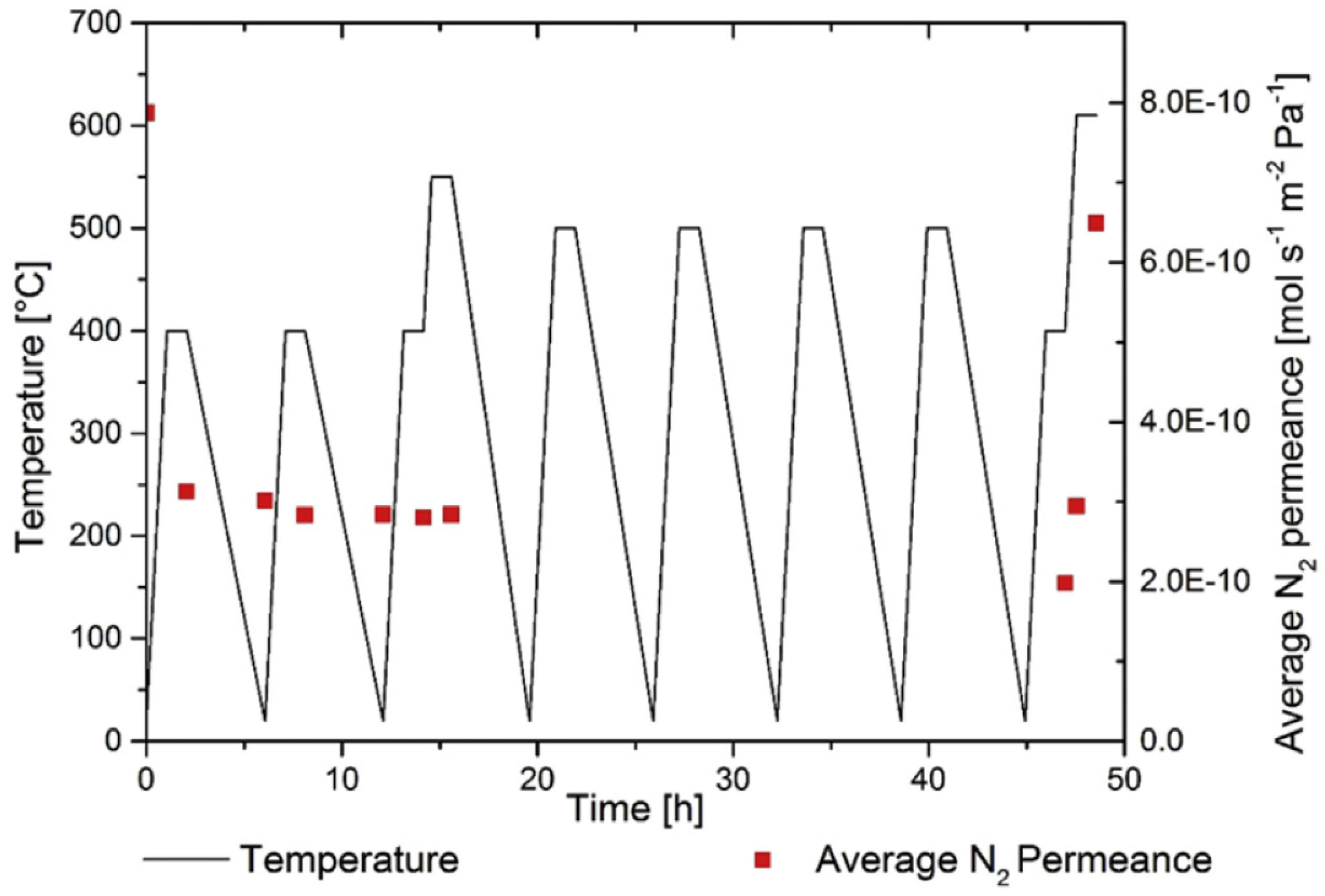
The stability and sealing of the ceramic supported Pd-based membranes developed for this process were first investigated. The new sealing method consisted in using graphite gaskets and Swagelok connectors. In this way, the difference in thermal expansion of the ceramic tube and the Swagelok piece is absorbed by the gasket with an intermediate thermal expansion. Thermal cycles for the membrane proved the stability of the sealing method as depicted in
Figure 20. Furthermore, single gas permeation tests showed that the Pd-based membrane gave one of the highest permeances ever reported in the literature. A table with the comparison of different Pd-based membranes investigated in the literature can be found in Fernandez
et al. [
38].
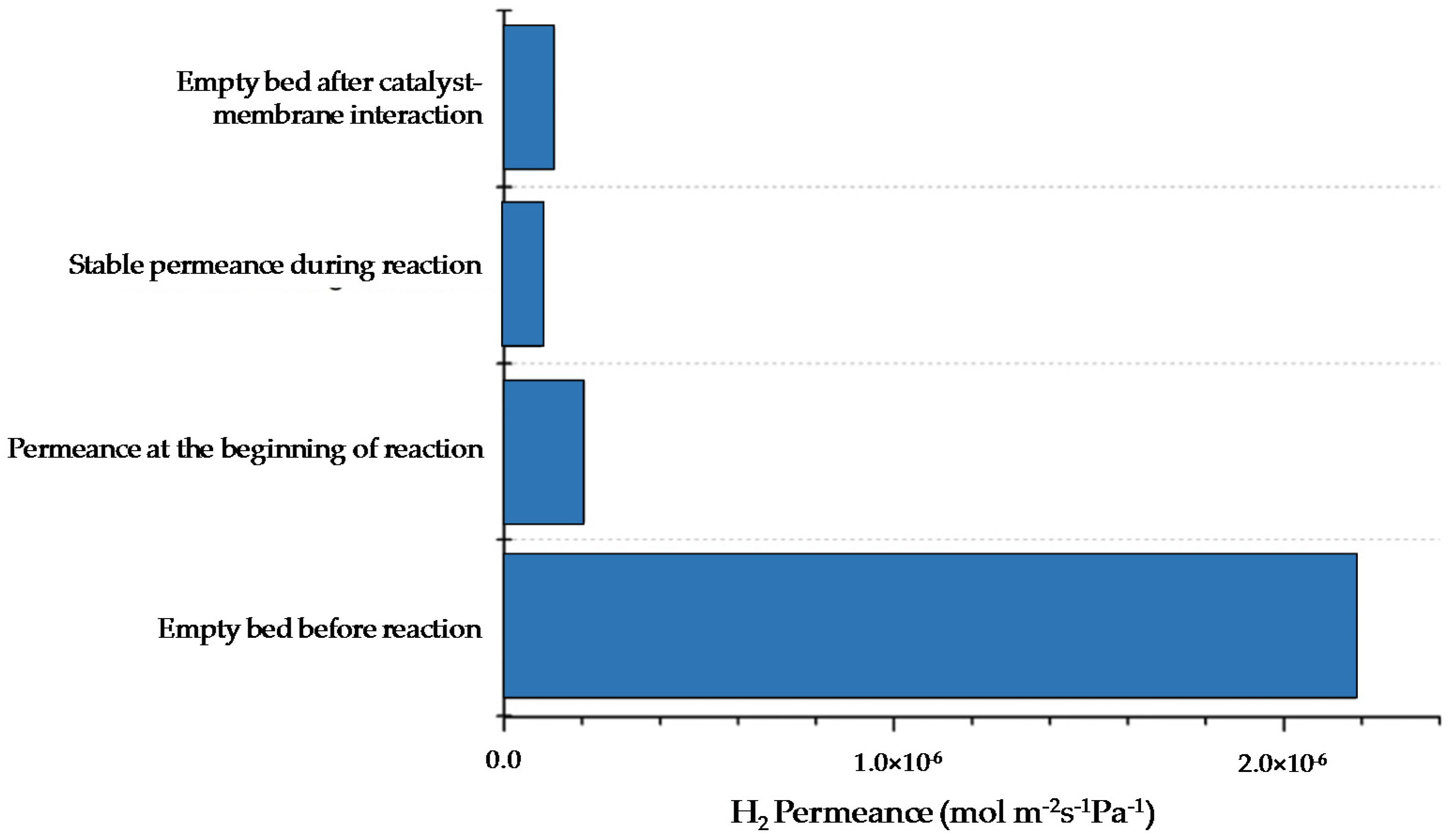
First, lab-scale tests were carried out both in PBMR and FBMR, using the catalyst and membranes (thickness 3.6 μm) developed in the project. Even though the catalyst exhibited superior performance in a traditional reactor configuration, in both cases, the combination resulted in a significant drop (98%) in membrane permeance, probably owing to interactions between membrane and catalysts as depicted in
Figure 21.
To overcome this drawback, a commercial catalyst was used with another membrane developed in DEMCAMER with the same characteristics of that previously mentioned and no significant drop in performance was observed. In addition, the combination between a Pd-Ag commercial membrane (100 μm thickness) from REB Research & Consulting and Goodfellow and the DEMCAMER catalyst did not show any drop in the membrane permeance. The overall performance of the membrane reactor resulted in being among the best in the literature in terms of both CO conversion and hydrogen recovery (96% CO conversion and 84% H2 recovery at 400 °C, 4 bar and a gas hourly space velocity of 2500 h−1), when the Pd-Ag membrane developed in DEMCAMER and commercial catalyst were used.
In addition, very extensive validation tests of fluidized bed membrane reactors were achieved. Both commercial and the developed Pd-based membranes were tested in single tube and multi-tube configurations and with different catalysts. In fluidization conditions, the performance of the WGS-MR process was assessed for more than 800 h where the system was able to achieve flux and selectivities well above the initial targets. Best results show CO levels in the permeate side at levels of 5–7 ppm, which means that the produced hydrogen (recovered up to 60% in these tests) could be directly used in fuel cells. Additionally the catalyst did not react with nor stick to the membrane surface. These results fully demonstrate the WGS reactor concept at lab-scale conditions.
The testing and validation of the WGS-MR prototype was performed afterwards. The upgrading, by the WGS reaction, of a syngas mixture in a packed bed membrane reactor integrated downstream of a reformer was proven on a pilot scale. The results at prototype level showed 20% of hydrogen recovery using only 63% of the design membrane area. In this system the hydrogen purity grade achieved was 2.5. The system, tested for ca. 1000 h at several operating conditions showed stable performance. During the testing time, an increasing trend of membrane performance was overall observed. From the initial value, less than 10%, a maximum H2 recovery of 20% was observed. Overall, the increase in WGS feed temperature promoted the H2 permeation.
3.3. Fischer Tropsch
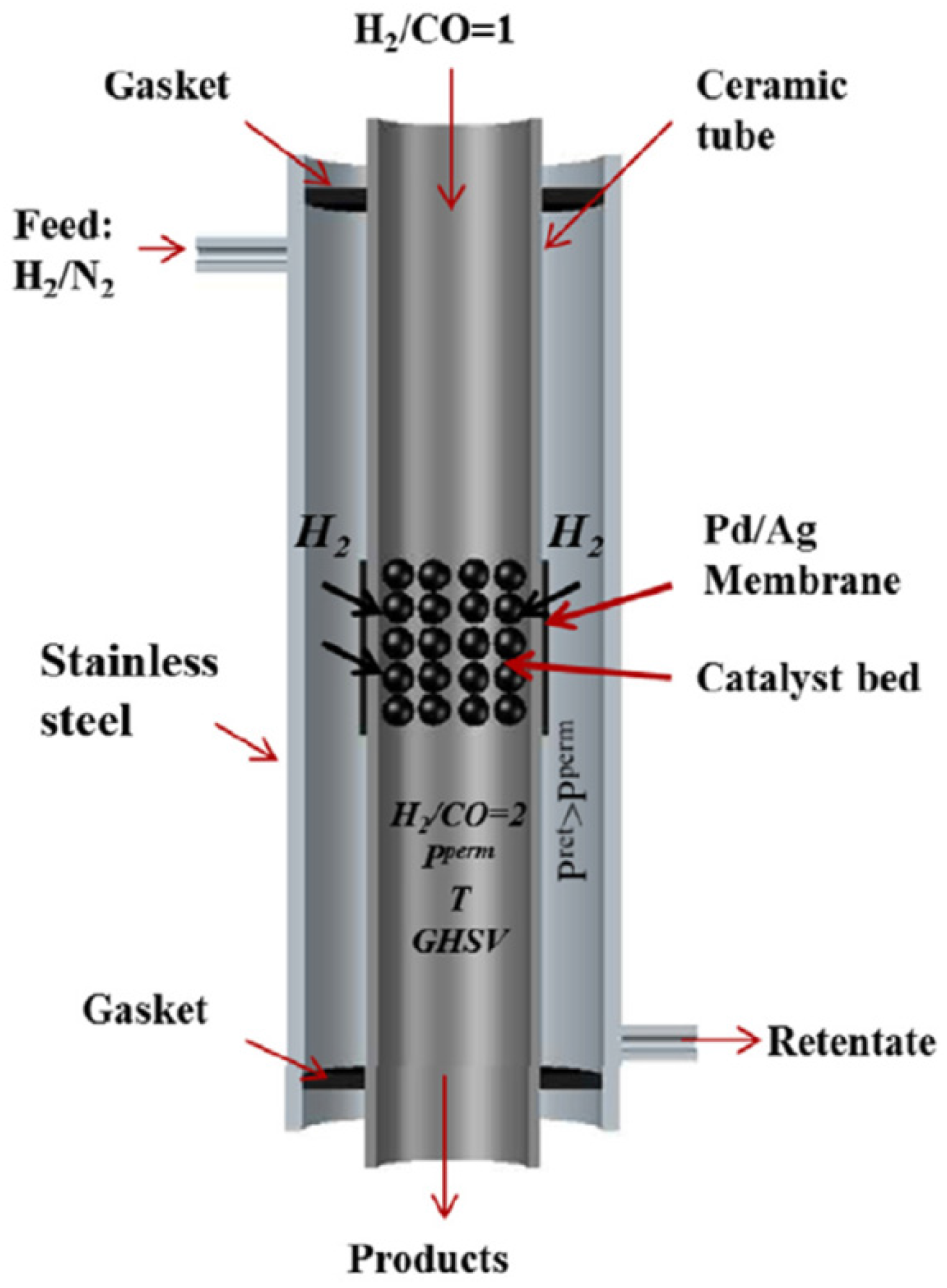
While for the WGS process, ceramic supported Pd-Ag membranes were used for selective H2 separation from the reaction system, similar Pd-Ag membranes were used in the FTS reaction for controlled feeding of H2 into the catalytic bed in order to enhance the selectivity to the products.
The catalyst developed for the DEMCAMER project (B-Ru/TiO
2) achieved high values of activity, selectivity, and stability in compliance with the initial targets (
Table 3) proposed. It should be mentioned that hydrocarbon productivity with the final FTS catalyst formulation was higher than that reported for the benchmark Co-based and also identified in the industrial requirements for the intensification of targeted FTS reaction. The stability of the B-Ru/TiO
2 FTS catalyst achieves a CO conversion above 60% maintained for up to 80 h on stream, hence the stability under reacting conditions was also demonstrated.
This catalyst was used for the demonstration of the reactor concept at lab-scale. Before this, the performance of the Pd-Ag membrane used in this process has been evaluated. In this case the membrane was prepared by standard electroless platting. However, the normal operating conditions for these types of membranes is always above 300 °C in order to avoid “embrittlement” of the Pd-layer resulting in a decrease in the ideal selectivity. However, FTS process takes place at lower temperatures and this effect might be present in the system. Due to this fact, more Ag is added in the metallic layer, which increases the resistance to this defect at lower temperatures. In this case the Ag was added into the membrane by PVD techniques.
Another problem associated with the FTS reaction, and related to the use of selective Pd-Ag membranes for H
2 separation (feeding for this process), is the presence of CO in the bed. CO is a poisoning gas for the membranes since it becomes adsorbed in the matrix of the Pd layer, therefore reducing the surface area available for permeation. Moreover, the CO poisoning effect is even more important at lower temperatures, which is actually the case of interest.
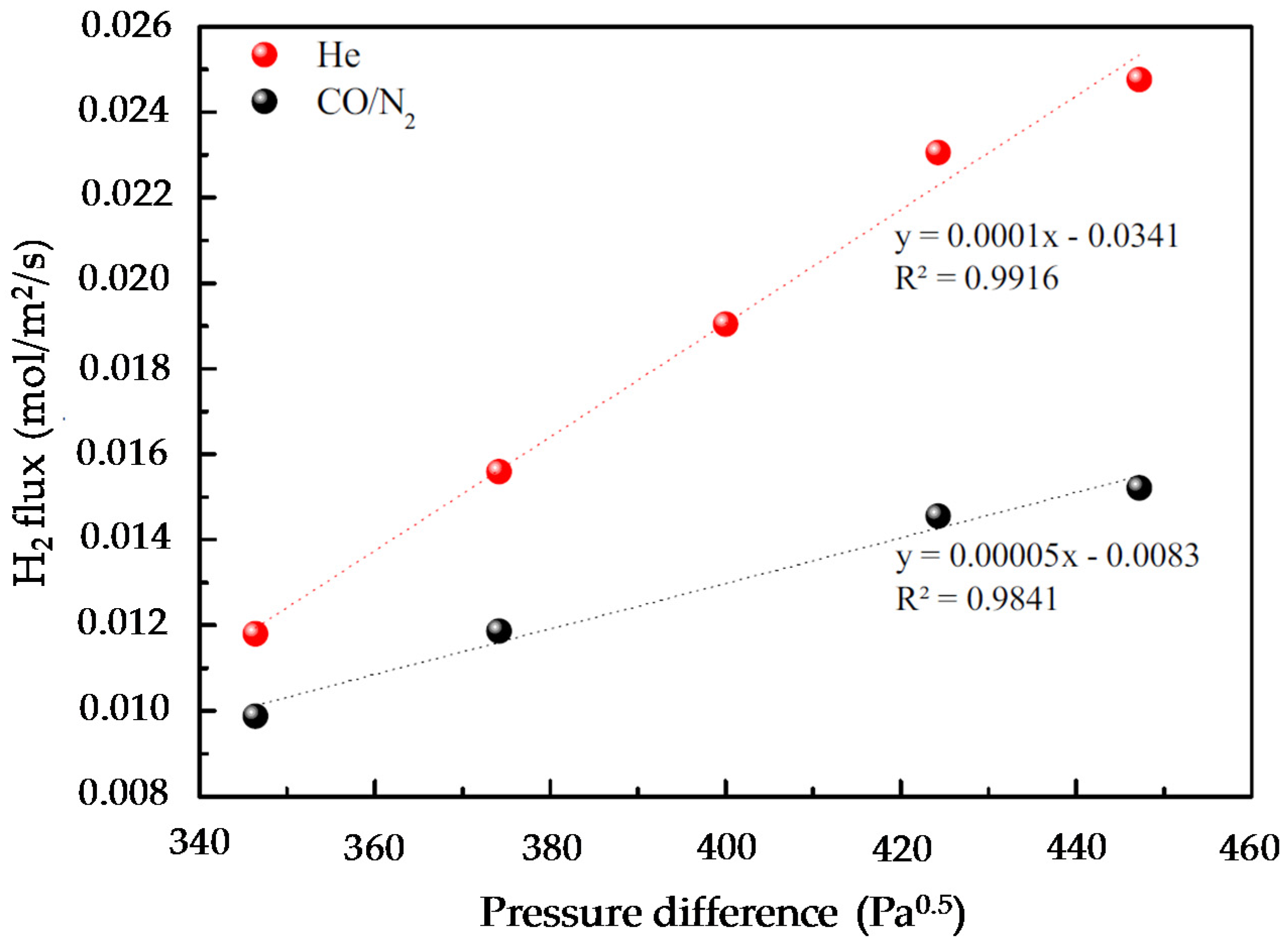
Figure 22 shows the effect of the gas fed into the system on the amount of H
2 permeated through the membrane. As observed, for the same H
2 partial pressures at both sides of the membrane, the presence of CO clearly effects H
2 permeation in the system. Once having studied the effect of CO on the membrane, lab-scale experiments were carried out in the setup described in
Section 2.3.3. In this case, two different configurations were compared (packed bed reactor with and without membrane).
The best conditions in terms of selectivity for the desired products were obtained when poor H
2 syngas (H
2/CO = 1) was fed into the reaction chamber from the inside of the membrane, while the H
2 needed to reach the right FTS stoichiometry (H
2/CO = 2) was fed (and properly distributed) into the reaction chamber through the Pd-based membranes by flowing H
2/He at the outer side of the membrane.
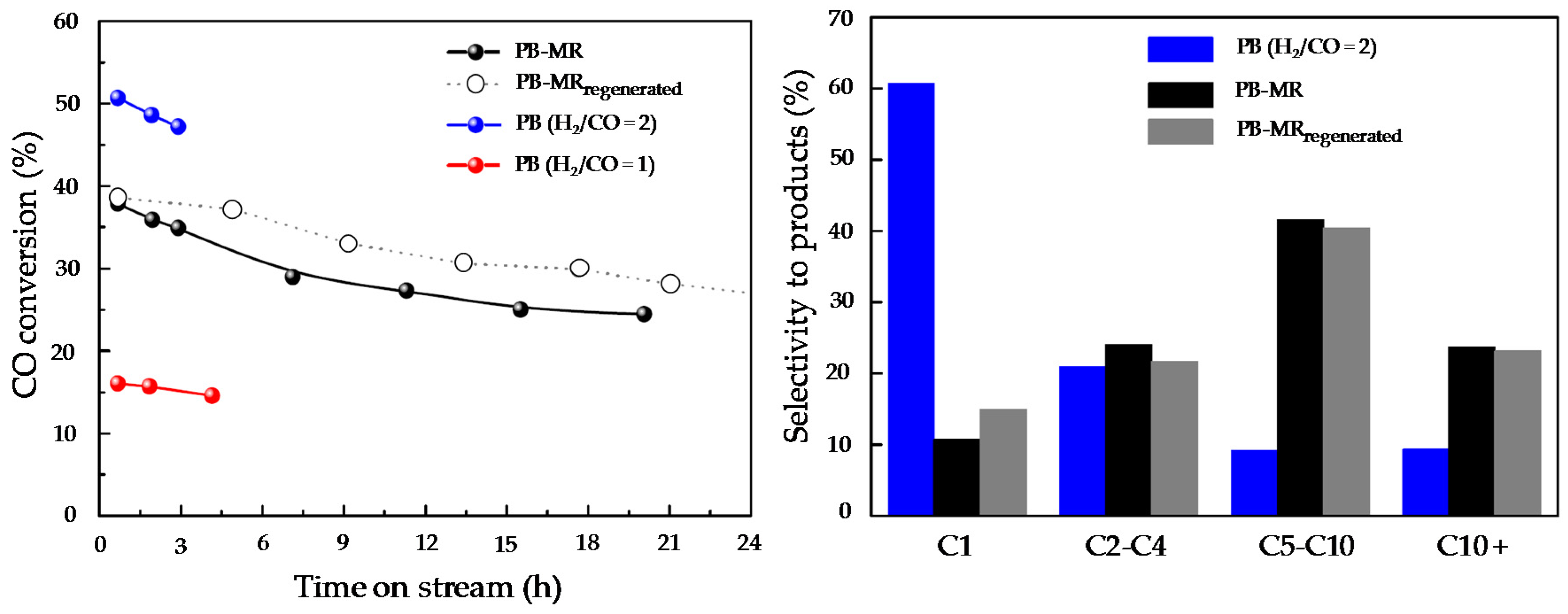
Figure 23 compares CO conversion for the different concepts investigated. It is observed that the PB-MR configuration gets an intermediate CO conversion (37.9%) compared to the packed bed system with the stoichiometric H
2/CO ratio of two (50.7%) and much higher than the H
2/CO ratio of one (14.1%). Positively, product selectivity is remarkably affected by the “more adequate” H
2-distribution attained in the PB-MR and the selectivity towards the targeted high-molecular hydrocarbons increases by a factor of four. In addition, a significant decrease of CH
4 production (from 60% to 15%) is also observed when compared to PB. Under the studied experimental conditions, membranes tend to deactivate after a few hours on stream (probably due to the formation of waxes) but their performance can be recovered by
in-situ thermal treatments under inert atmosphere as explained in the literature [
24].
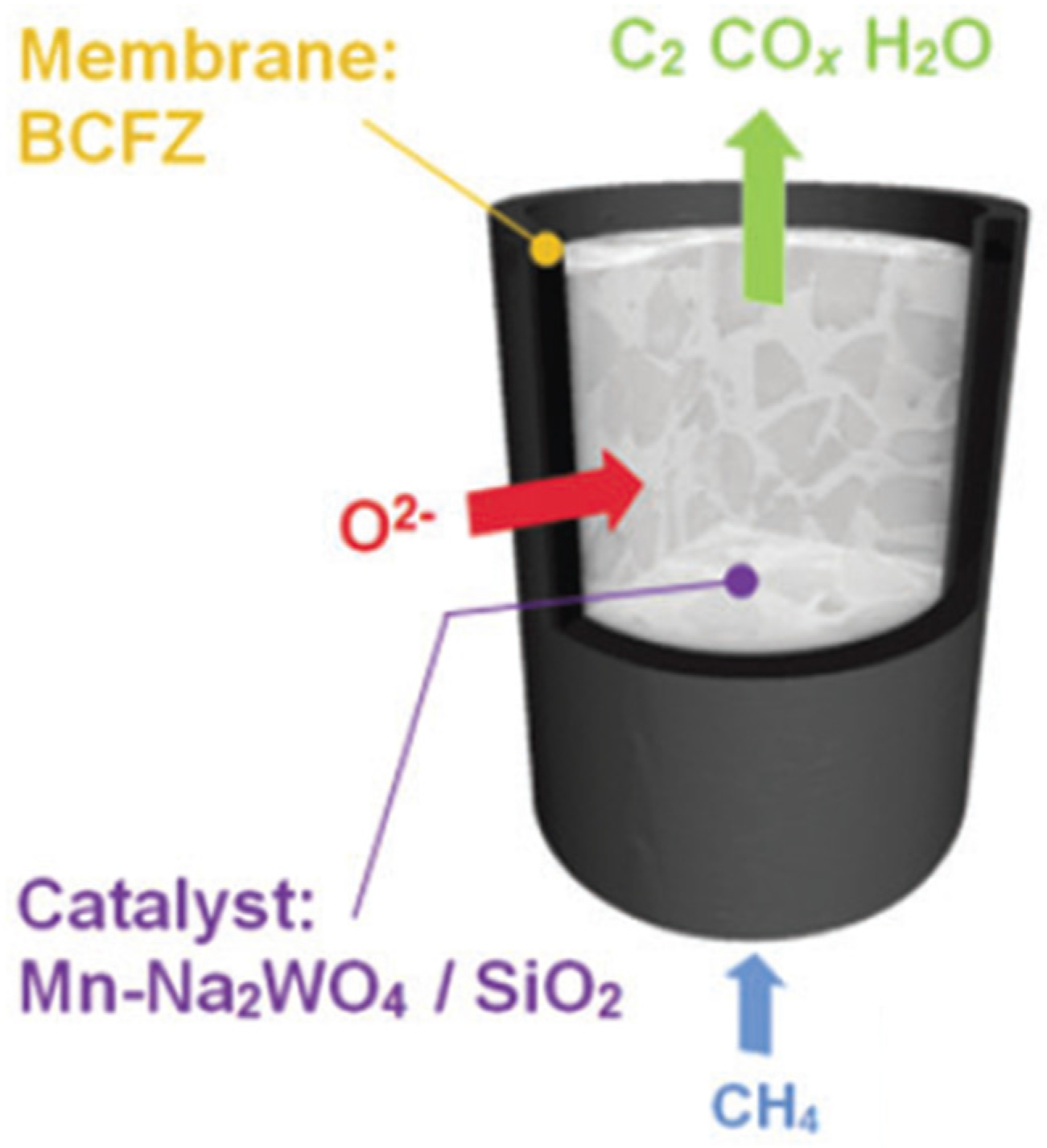
3.4. Oxidative Coupling of Methane
The same MIEC membranes prepared and tested for the ATR process were used during the OCM process. Therefore this section is focused on the catalyst activity, lab scale reaction, and final prototype reaction, but not on the membranes themselves as they have already been presented and discussed in
Section 3.2.
The developed OCM catalyst for the project achieved a high activity/selectivity (CH4 conversion = 55%, C2 yield = 22%), which is close to the best results reported in the state-of-the-art literature.
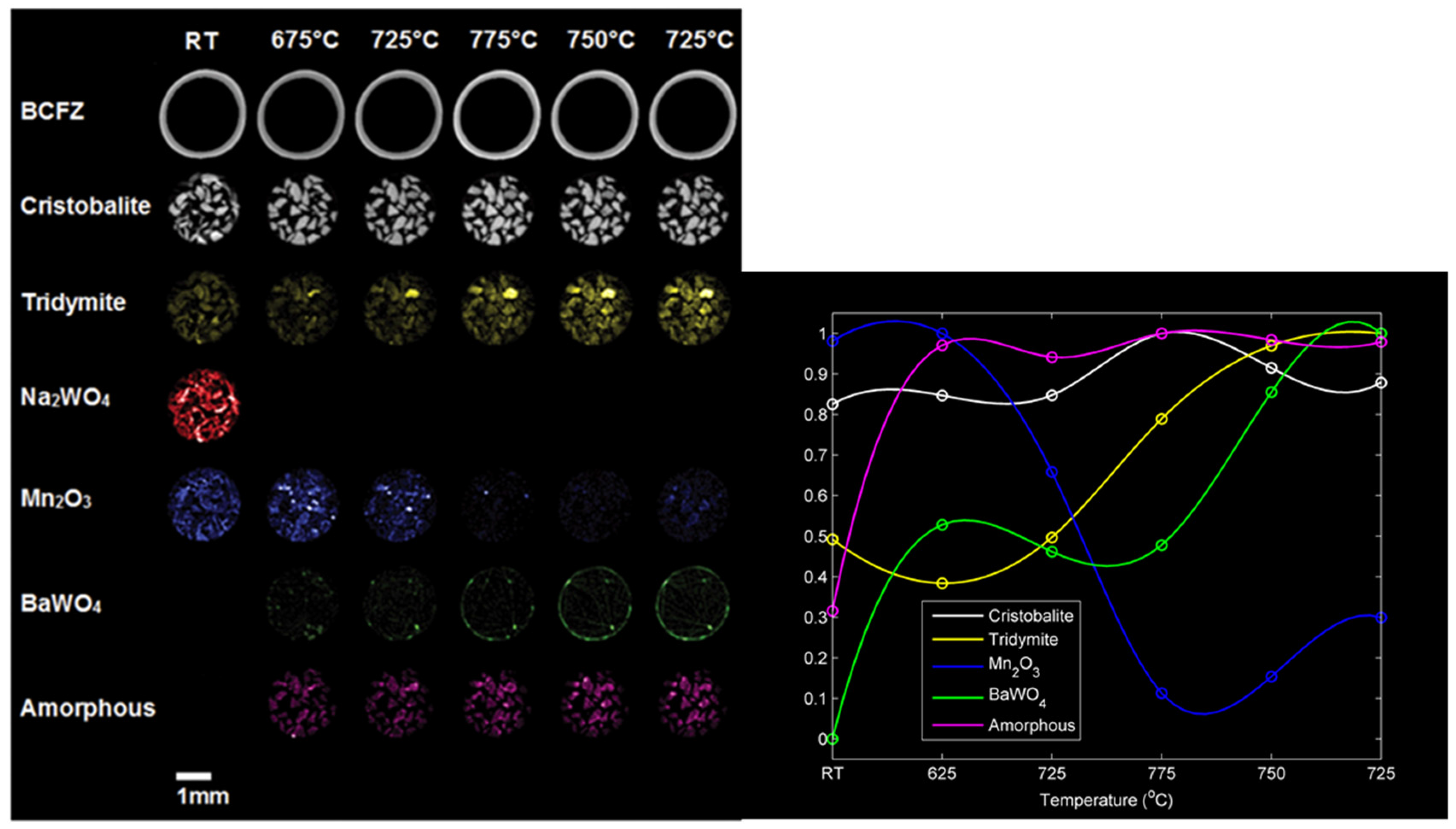
Lab scale reaction was carried out by placing the developed catalyst inside the MIEC membrane and selective feeding of oxygen from the retentate side. Lab scale experiments were performed at different temperatures and the phase maps of five identified crystalline phases and one amorphous phase of the catalyst (also the BCFZ reflection) were monitored in the X-Ray diffraction computed tomography (XRD-CT) equipment (
Figure 24). At room temperature, Mn
2O
3 and Na
2WO
4 are clearly present as active phases in the catalyst (also the support SiO
2 (cristobalite and tridymite) can be clearly identified). However, once the reaction takes place at 675 °C, it is observed that the Na
2WO
4 phase disappears, which corresponds to the formation of an amorphous phase. Simultaneously, the formation of a new crystalline phase near the membrane and identified as BaWO
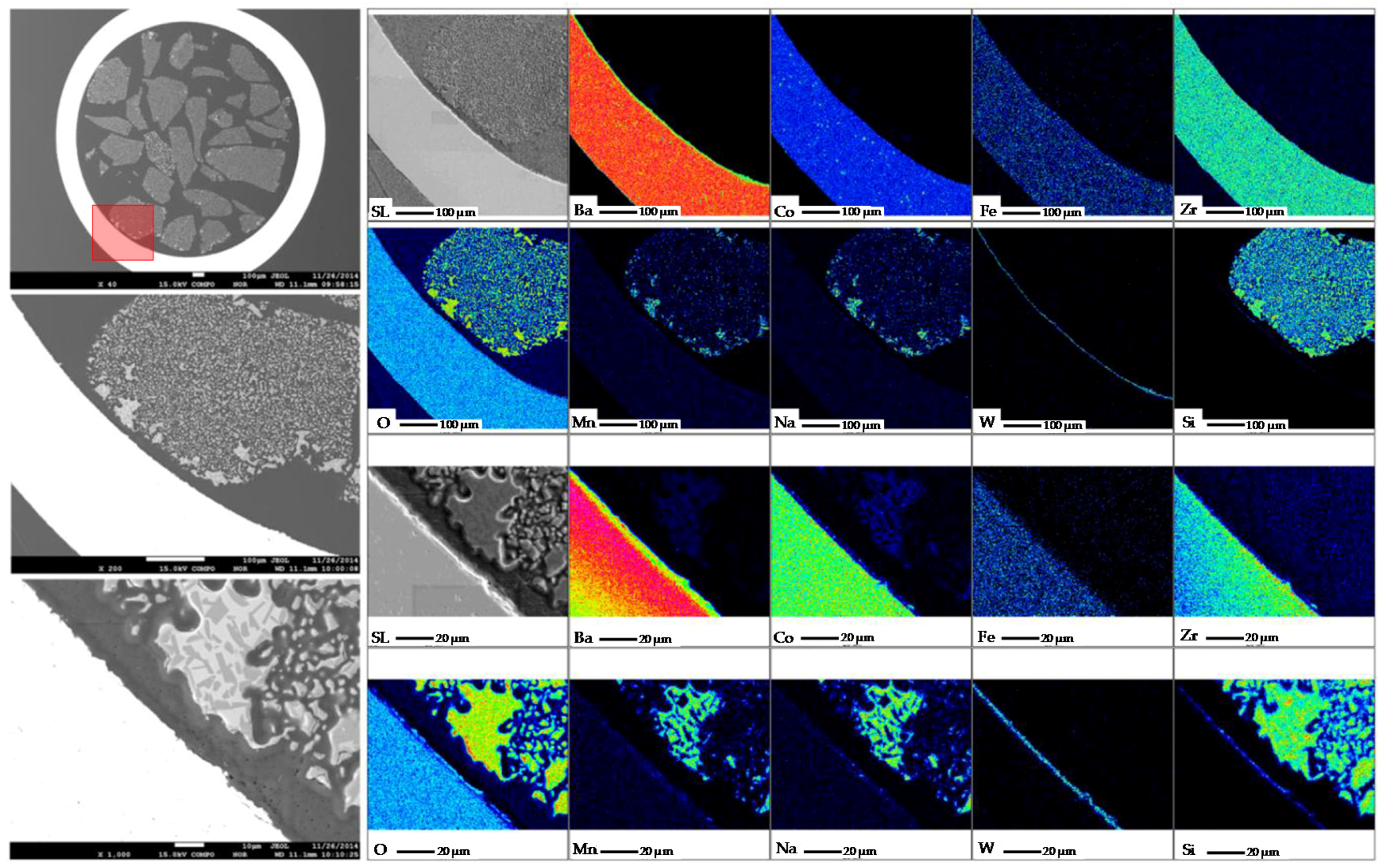
4 is observed, which implies a migration of the active phases in the catalysts to a new reorganization that occurs at high temperatures. This effect can be better observed after OCM reaction via Scanning Electron Microscopy coupled with Wavelength Dispersive X-ray Spectrometry (SEM–WDS) characterization in the surroundings of the BCFZ membrane. In
Figure 25, where qualitative elemental analysis is depicted at two different magnifications, the migration of W from the catalyst to the surface of the membrane to form the BaWO
4 is observed, which supports the findings observed in the XRD-CT reaction monitoring.
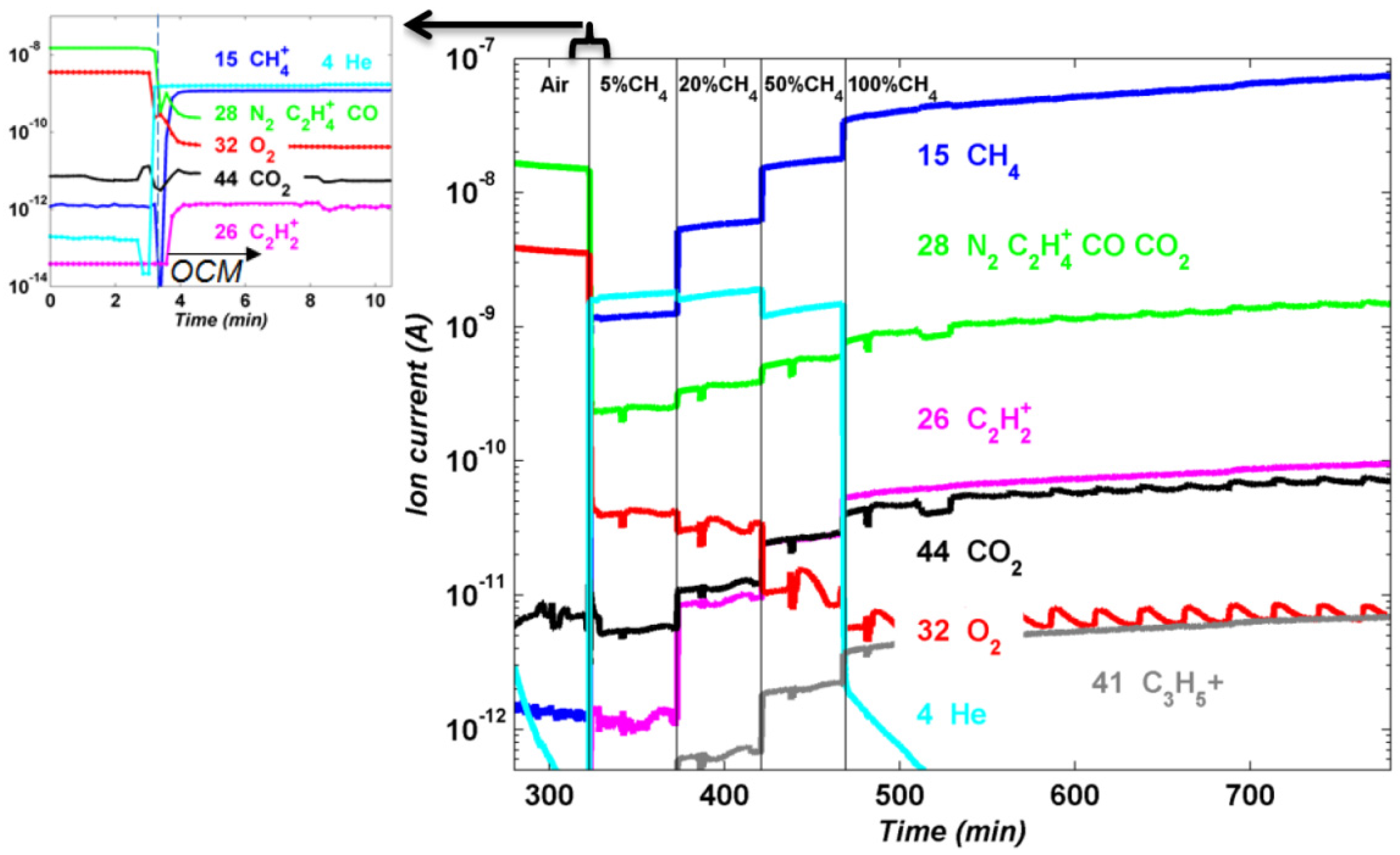
The outflow gases from the OCM lab-scale reactor were analyzed with mass spectrometry. In this case, different scenarios are presented in
Figure 26 corresponding to different gas mixtures of CH
4 diluted in He). It can be observed that the presence of C
2+ (pink trend in
Figure 26) is only observed once the reaction starts (
i.e., when diluted CH
4 is fed into the reactor) and it is increased with increasing fuel concentration at the inlet of the reactor. What becomes more interesting is that, even though there is migration of W towards the membrane, higher hydrocarbons molecules as compared to CH
4 are formed during the reaction (C
2 and C
3 fractions).
Moving towards the prototype reactor, the MIEC membranes were not able to withstand the mechanical strength required for the coupling to the reactor and at this stage the technology for the OCM reaction failed to deliver results beyond the laboratory scale. In order to demonstrate the OCM-MR concept, significant improvements are needed on the robustness of the MIEC membranes before any industrial application under safe conditions could be imagined.
4. Conclusions
The DEMCAMER project aimed at the development of novel membrane reactors to provide important process intensification for key processes in the chemical engineering field. The industrial processes studied in this project have some drawbacks (viz. global efficiency, CO2 emissions, and volume of the process) that can be solved or reduced by using more compact systems that integrate reaction and separation in the same unit. For all the processes studied in this project (ATR, WGS, FTS and OCM), the streamline followed was similar. First was the development of active catalysts for the experimental conditions present in membrane reactors, which differ from those in benchmark technologies. In parallel, improved membranes for H2 and O2 permeation were investigated. In general, stability over time and mechanical resistance were the two main characteristics aimed for in this project. Subsequently, the interaction between the catalyst and the membranes, and the performance of the reactor concept were proven at lab-scale when combining both at reactive conditions. Finally, and following Hazard and Operability (HAZOP) procedures, all membrane reactors were scaled up to prototype scale.
The different catalysts developed for the processes showed catalytic activities well above the DEMCAMER targets. The Ni-Pd catalyst developed for the ATR reaction has improved activities and selectivity as compared to state-of-the-art catalysts due to the synergistic interaction Ni-Pd and the optimized oxygen storage capacity on the cerium support. The catalyst for WGS (Pt-Re over Ce-Ti mixed oxide) has shown high stability over time with high CO conversions. However, interaction of the Ti with the membrane was observed, resulting in the underperformance of the membranes used for the WGS reaction. For the FTS process, a B-Ru/TiO2 catalyst was developed, which showed good stability and resistance to sulfur poisoning and carbon formation. The Mn-Na-W/SiO2 catalyst for OCM also showed high CH4 conversions with good selectivity to C2+ with results close to state of the art catalysts. The catalyst shows an optimal active phase exposition through XRD-CT analysis, whereas the surface phase is modified in the presence of membranes as a consequence of W migration from the catalyst to the membrane surface. However, W migration did not affect the conversion of CH4 to higher hydrocarbons.
Two different types of membranes were improved within the project: MIEC membranes for selective oxygen separation from air and ceramic supported Pd-based membranes for selective H2 separation. Stability of both membranes was confirmed through single gas permeation tests at operating temperatures over many hours without any decrease in the membrane performance. Furthermore, new sealing methods were tested and proposed in order to fix the membranes inside the membrane reactors. Permeances and selectivities obtained with the different membranes prepared in the project are among the best ever reported in the literature and well above the targets for the DEMCAMER project. For the different processes investigated, the preferred configuration is the packed bed membrane reactor due to higher simplicity in the configuration. In addition, stability was also assessed in the fluidized bed membrane reactor configuration for the WGS reaction.
All the different processes were investigated at lab-scale and only the OCM process failed due to the lack in mechanical strength of the membranes when connected to the reactor. Possible interactions between catalysts and membranes were observed for the WGS and OCM processes. This effect was remarkable for Pd-based membranes, where the permeance of H2 dropped significantly and membrane reactor performance was affected. However, during the OCM reaction using MIEC membranes the interaction between tungsten (W) and the surface of the membrane did not alter the selectivities to products. At the final stage, all processes were considered for prototype validation. However the prepared MIEC membranes were not able to handle the assemblage to the reactor and the demonstration failed for OCM. The ATR reactor was tested with an external heat source because damages in the MIEC membranes did not allow the oxygen permeation at prototype scale to be tested. In the case of FTS prototype demonstration, the reaction system did not progress to the desired products. The WGS prototype system was fully demonstrated using syngas from a reformer reactor installed upstream of the WGS reactor. In this case, a pure H2 stream (grade 2.5) was obtained and reaction was maintained for more than 40 days without any drop in the performance. At this stage, only the WGS concept achieved TLR level 5, whereas the other processes investigated in this project remained in TLR level 4.
According to the results obtained in the DEMCAMER project, only WGS can be considered beyond prototype scale for industrial application. However, many efforts should still be carried out to improve the standard sealing methods and poisoning of the active layer by CO and sulfur.