Design and Validation of a Cyclic Strain Bioreactor to Condition Spatially-Selective Scaffolds in Dual Strain Regimes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bioreactor System


2.2. Mechanical Characterization
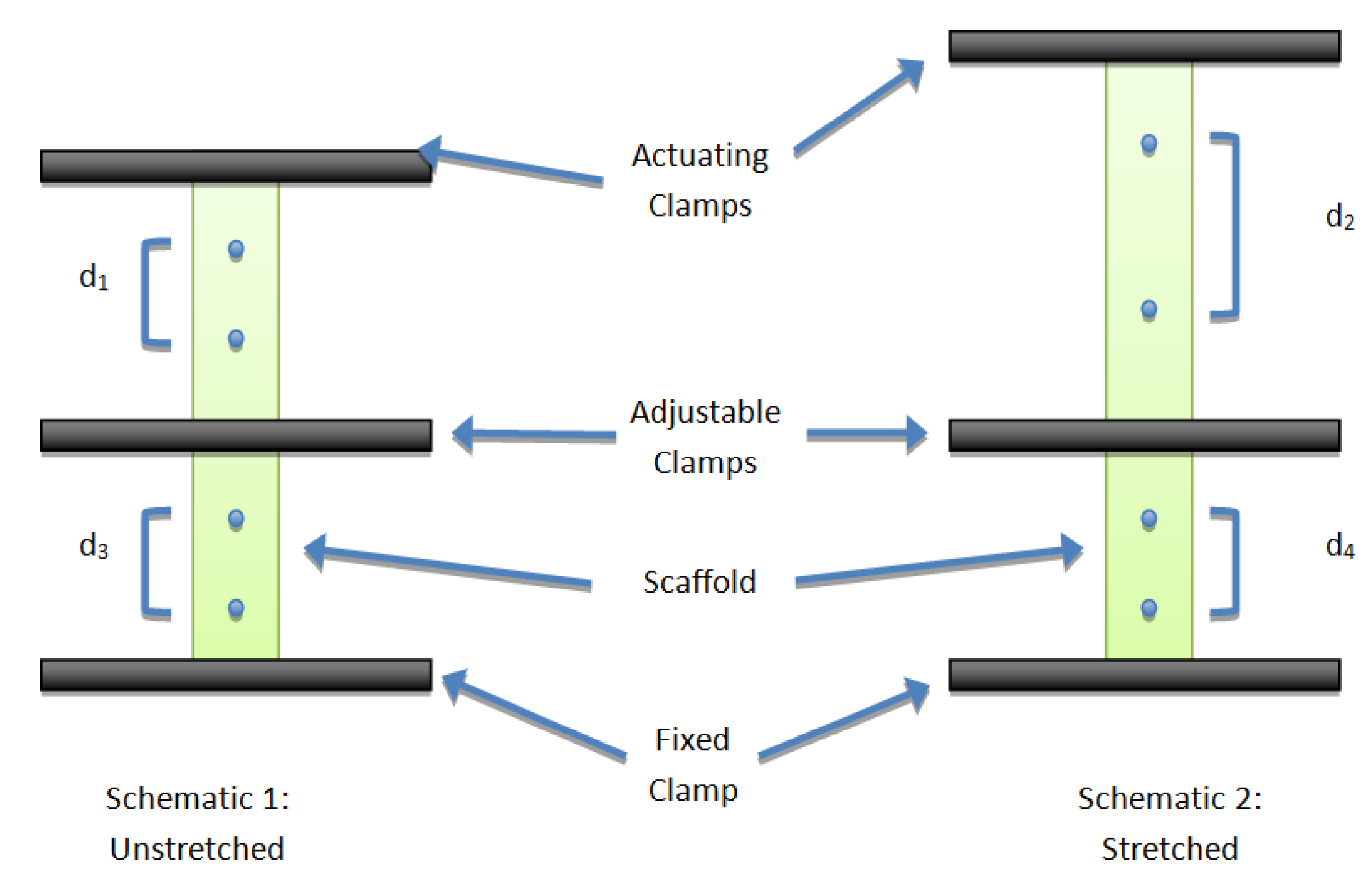
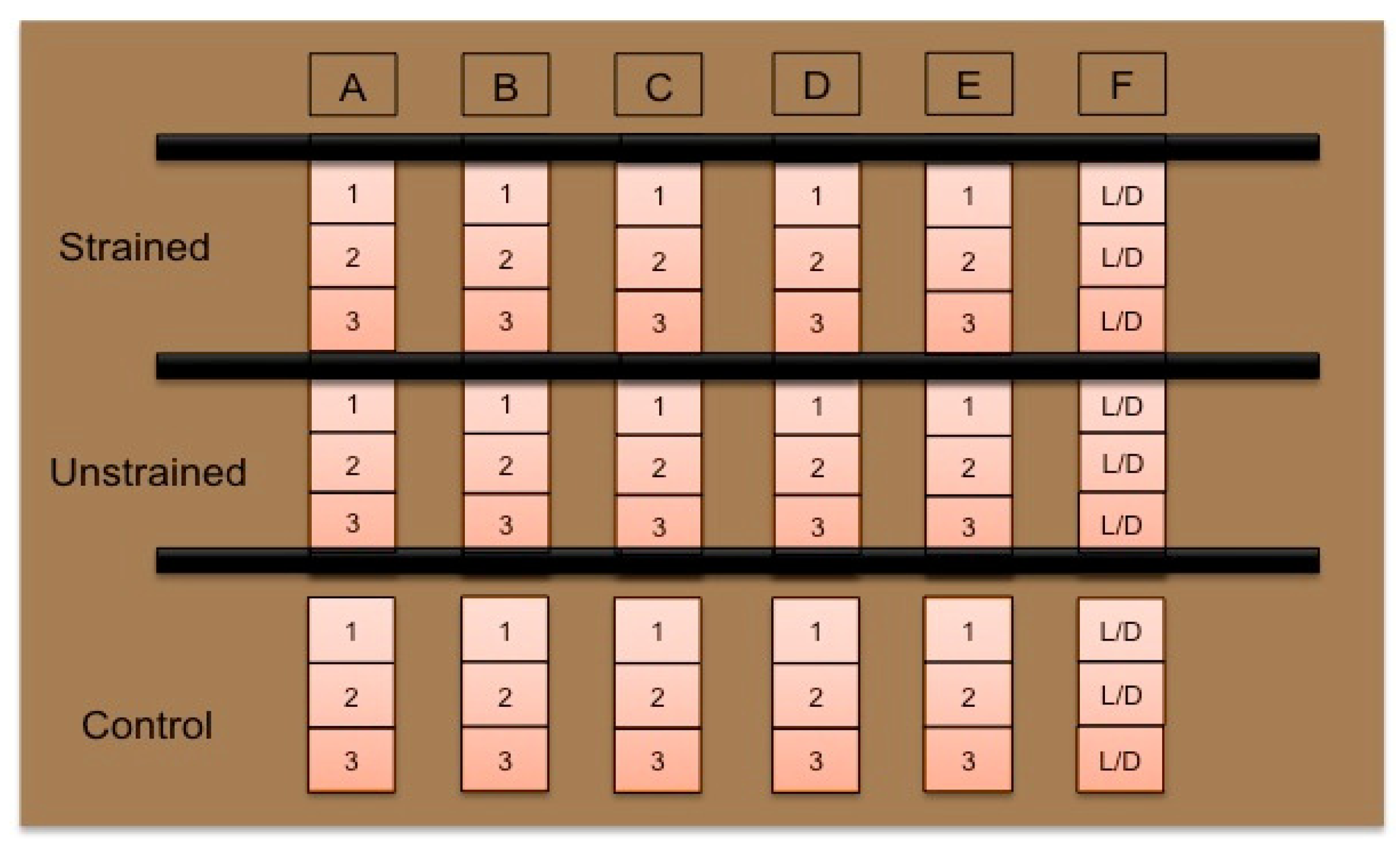
2.3. Cellular Characterization
| Group | Location | Stretch Schedule |
|---|---|---|
| Control | TCP Petri Dish | No stretch |
| Unstretched | Bioreactor | No stretch |
| Stretched | Bioreactor | 5%, 0.5 Hz, 1 h daily, 14 days |
2.4. Statistical Analysis
3. Results
3.1. Mechanical Characterization
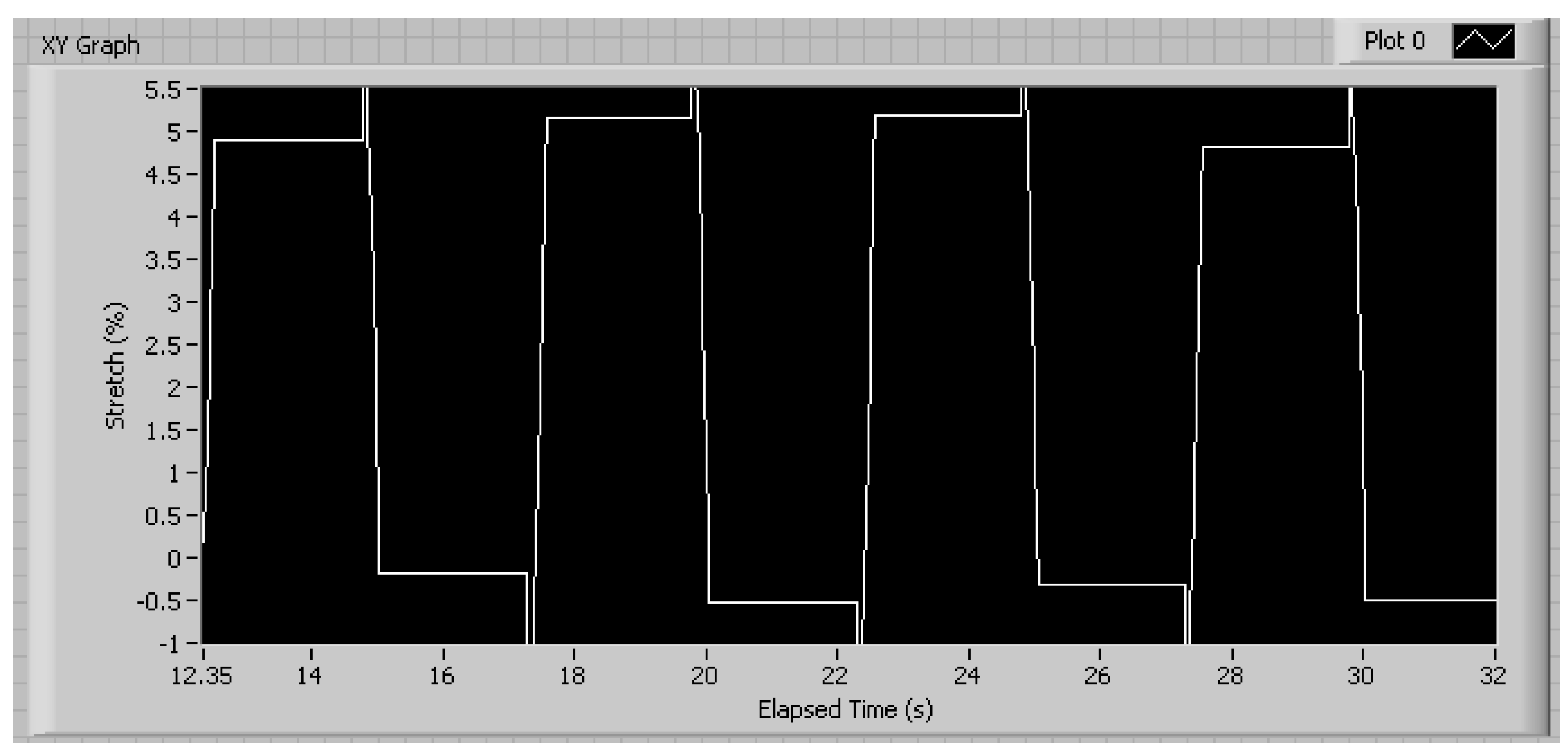
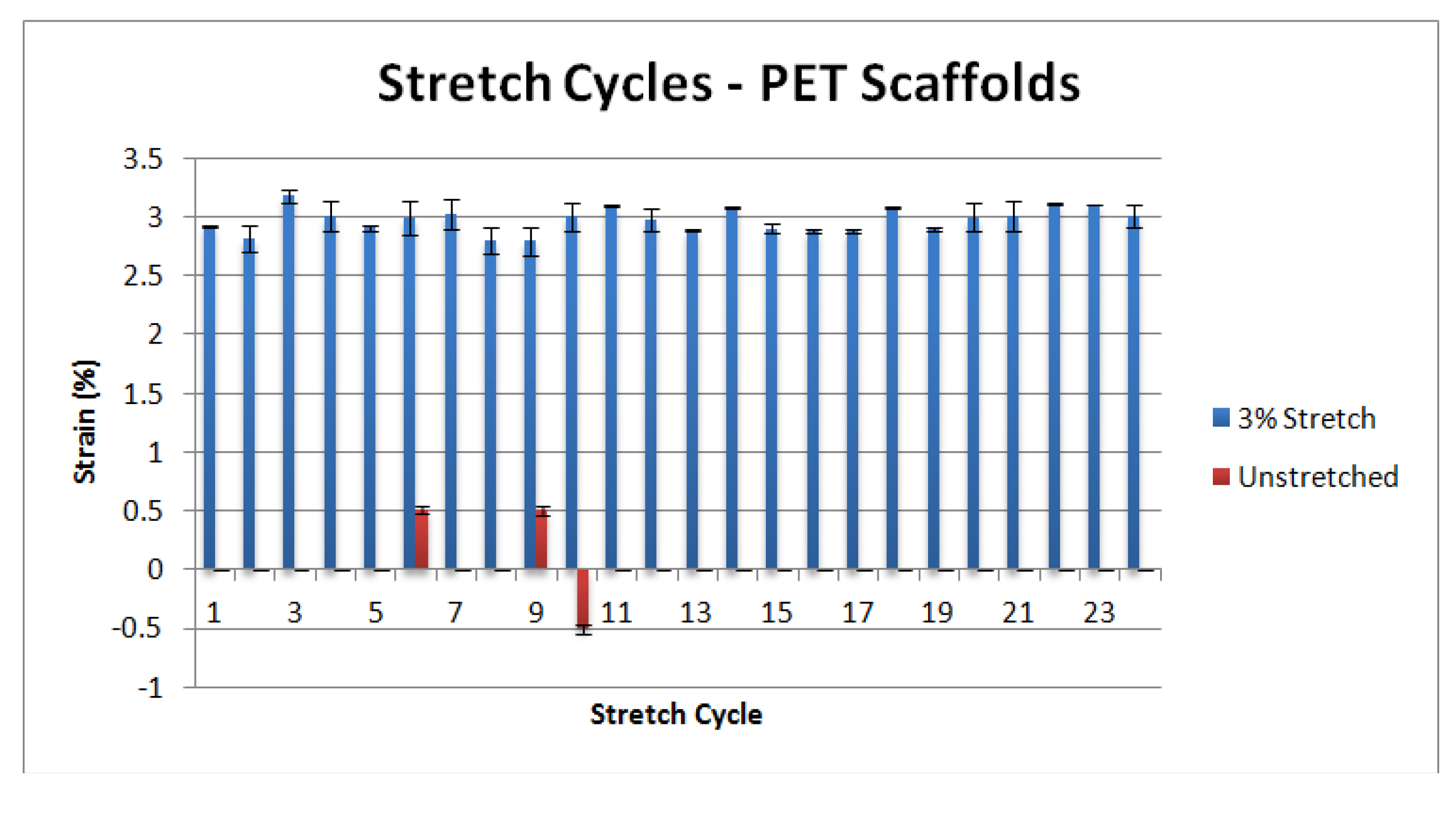
3.2. Cellular Characterization
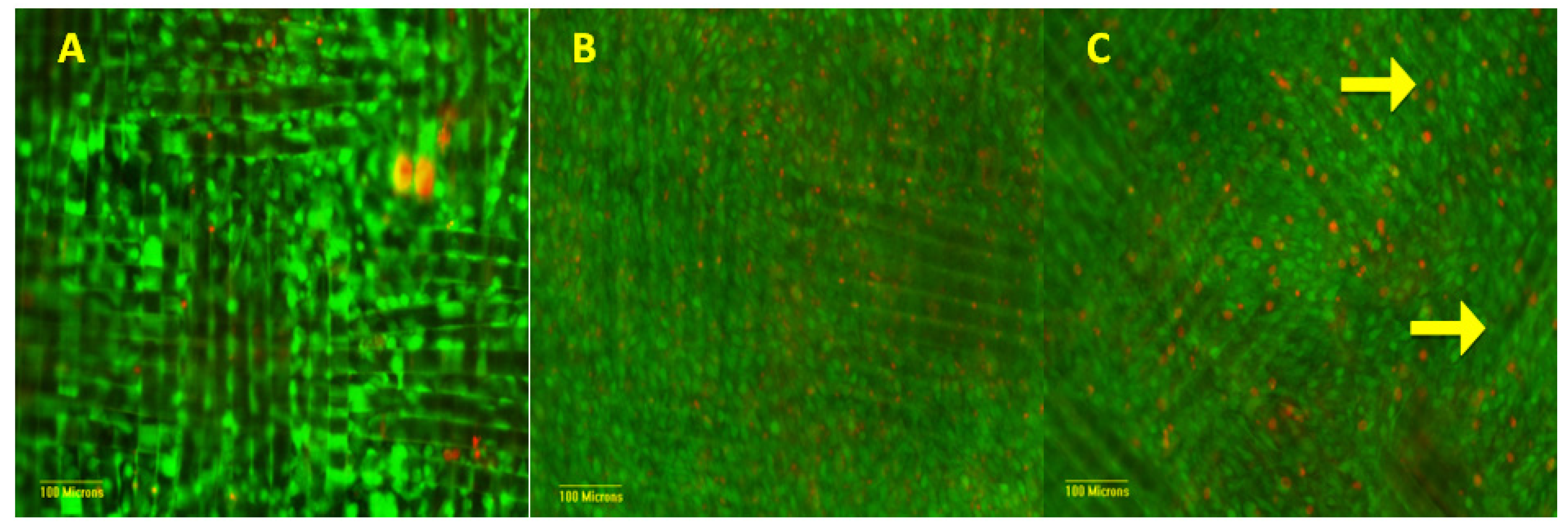
3.3. Matrix Characterization
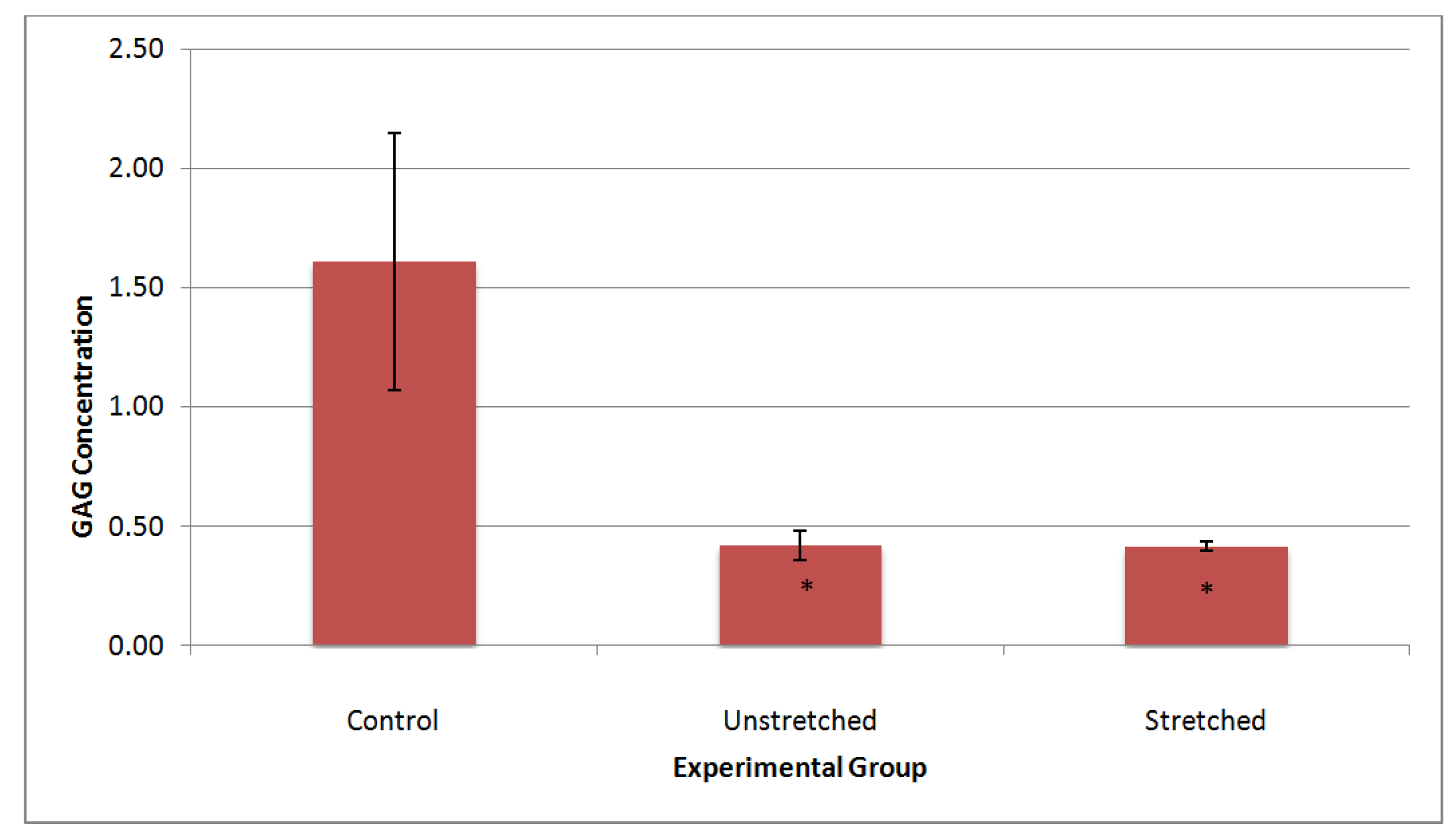
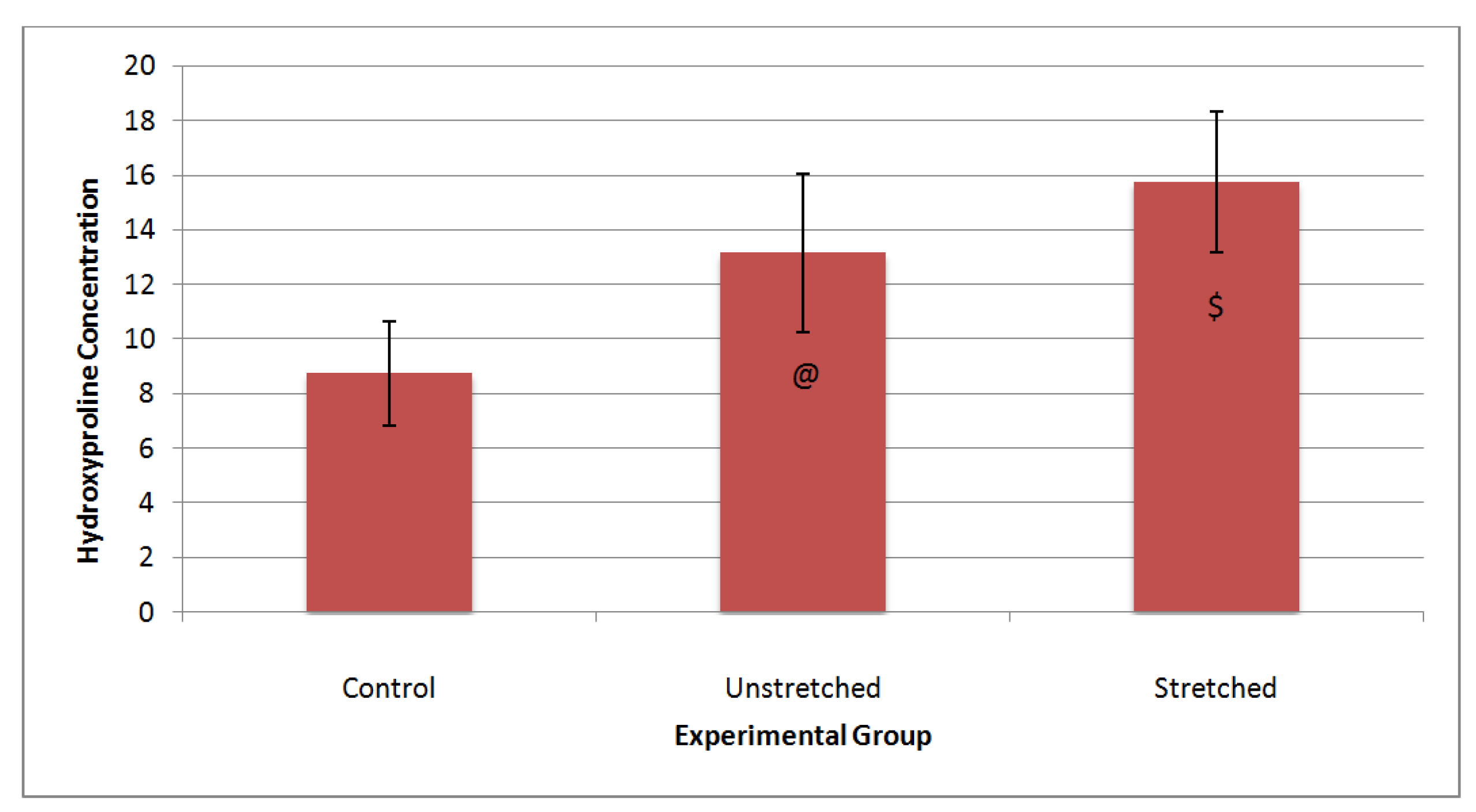
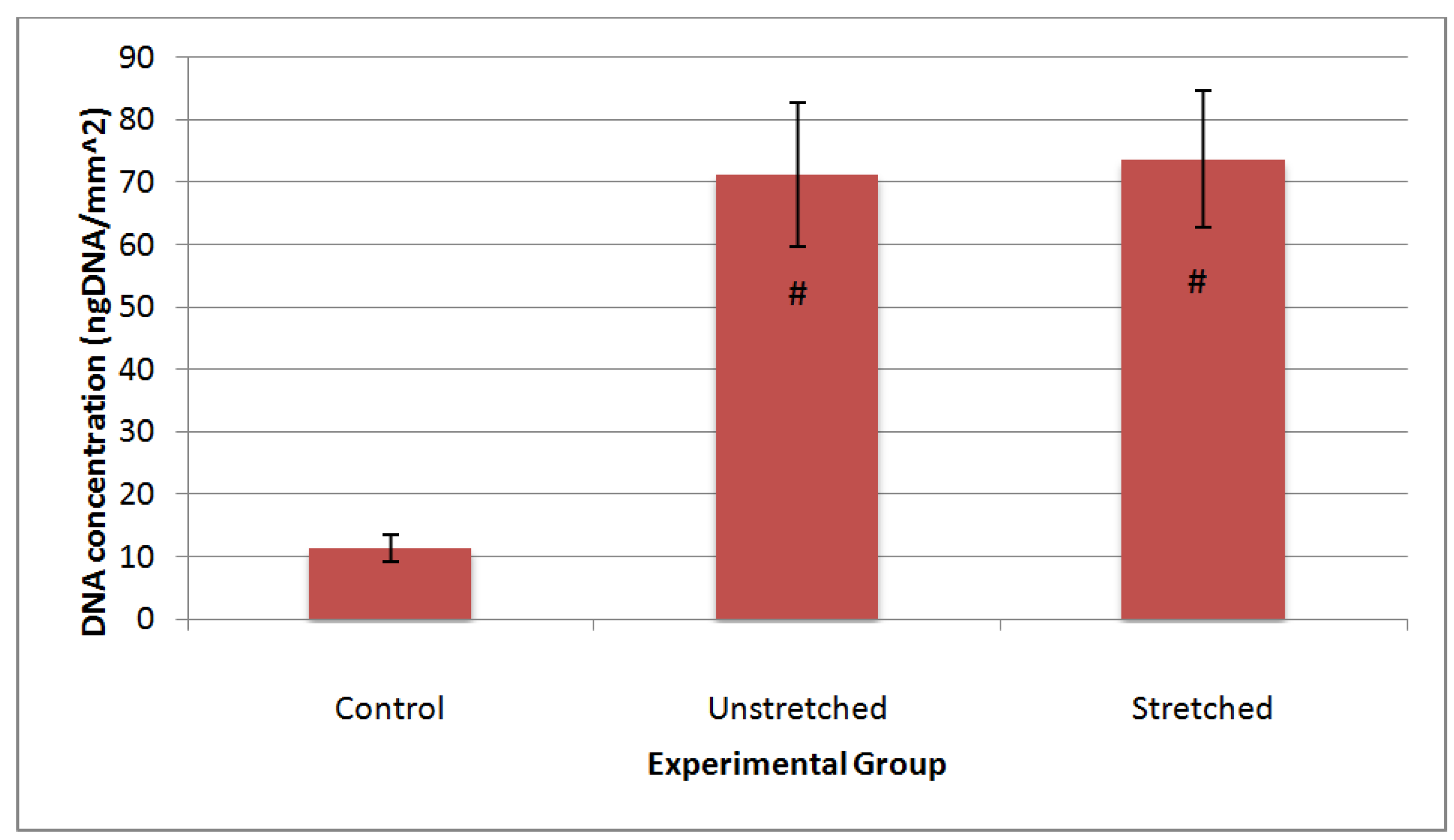
4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Caliari, S.R.; Harley, B.A.C. The effect of anisotropic collagen-GAG scaffolds and growth factor supplementation on tendon cell recruitment, alignment, and metabolic activity. Biomaterials 2011, 32, 5330–5340. [Google Scholar] [CrossRef]
- Caliari, S.R.; Ramirez, M.A.; Harley, B.A.C. The development of collagen-GAG scaffold-membrane composites for tendon tissue engineering. Biomaterials 2011, 32, 8990–8998. [Google Scholar] [CrossRef]
- Sahoo, S.; Toh, S.L.; Goh, J.C.H. A bFGF-releasing silk/PLGA-based biohybrid scaffold for ligament/tendon tissue engineering using mesenchymal progenitor cells. Biomaterials 2010, 31, 2990–2998. [Google Scholar] [CrossRef]
- Whitlock, P.W.; Smith, T.L.; Poehling, G.G.; Shilt, J.S.; van Dyke, M. A naturally derived, cytocompatible, and architecturally optimized scaffold for tendon and ligament regeneration. Biomaterials 2007, 28, 4321–4329. [Google Scholar] [CrossRef]
- Deng, D.; Liu, W.; Xu, F.; Yang, Y.; Zhou, G.; Zhang, W.J.; Cui, L.; Cao, Y. Engineering human neo-tendon tissue in vitro with human dermal fibroblasts under static mechanical strain. Biomaterials 2009, 30, 6724–6730. [Google Scholar] [CrossRef]
- Garvin, J.; Qi, J.; Maloney, M.; Banes, A.J. Novel system for engineering bioartificial tendons and application of mechanical load. Tissue Eng. 2003, 9, 967–979. [Google Scholar] [CrossRef]
- Joshi, S.D.; Webb, K. Variation of cyclic strain parameters regulates development of elastic modulus in fibroblast/substrate constructs. J. Orthop. Res. 2008, 26, 1105–1113. [Google Scholar] [CrossRef]
- Butler, D.L.; Hunter, S.A.; Chokalingam, K.; Cordray, M.J.; Shearn, J.; Juncosa-Melvin, N.; Nirmalanandhan, S.; Jain, A. Using functional tissue engineering and bioreactors to mechanically stimulate tissue-engineered constructs. Tissue Eng. 2009, 15, 741–749. [Google Scholar] [CrossRef]
- Riboh, J.; Chong, A.K.S.; Pham, H.; Longaker, M.; Jacobs, C.; Chang, J. Optimization of flexor tendon tissue engineering with a cyclic strain bioreactor. J. Hand Surg. 2008, 33A, 1388–1396. [Google Scholar]
- Wolchok, J.C.; Brokopp, C.; Underwood, C.J.; Tresco, P.A. The effect of bioreactor induced vibrational stimulation on extracellular matrix production from human derived fibroblasts. Biomaterials 2009, 30, 327–335. [Google Scholar] [CrossRef]
- Abousleiman, R.I.; Reyes, Y.; McFetridge, P.; Sikavitsas, V. Tendon tissue engineering using cell-seeded umbilical veins cultured in a mechanical stimulator. Tissue Eng. 2009, 15, 787–795. [Google Scholar] [CrossRef]
- Korin, N.; Bransky, A.; Dinnar, U.; Levenberg, S. A parametric study of human fibroblasts culture in a microchannel bioreactor. Lab Chi 2007, 7, 611–617. [Google Scholar] [CrossRef]
- Titze, I.R.; Hitchcock, R.W.; Broadhead, K.; Webb, K.; Li, W.; Gray, S.D.; Tresco, P.A. Design and validation of a bioreactor for engineering vocal fold tissues under combined tensile and vibrational stresses. J. Biomech. 2004, 37, 1521–1529. [Google Scholar] [CrossRef]
- Wang, N.E.; Shan, J.; Choi, R.; Oh, S.; Kepler, C.K.; Chen, F.H.; Lu, H.H. Role of osteoblast-fibroblast interactions in the formation of the ligament-to-bone interface. J. Orthop. Res. 2007, 25, 1609–1620. [Google Scholar] [CrossRef]
- Screen, H.R.C.; Shelton, J.C.; Bader, D.L.; Lee, D.A. Cyclic tensile strain upregulates collagen synthesis in isolated tendon fascicles. Biochem. Biophys. Res. Commun. 2005, 336, 424–429. [Google Scholar] [CrossRef]
- Geesin, J.C.; Darr, D.; Kaufman, R.; Murad, S.; Pinnell, S.R. Ascorbic acid specifically increases type I and type III procollagen messenger RNA levels in human skin fibroblasts. J. Invest. Dermatol. 1988, 90, 420–424. [Google Scholar]
- Redaelli, A.; Vesentini, S.; Soncini, M.; Vena, P.; Mantero, S.; Montevecchi, F.M. Possible role of decorin glycosaminoglycans in fibril to fibril force transfer in relative mature tendons—A computational study from molecular to microstructural level. J. Biomech. 2003, 36, 1555–1569. [Google Scholar] [CrossRef]
- Stegemann, H.; Stalder, K. Determination of hydroxyproline. Clin. Chim. Acta 1967, 18, 267–273. [Google Scholar] [CrossRef]
- Lechat, C.; Bunsell, A.R.; Davies, P. Tensile and creep behaviour of polyethylene terephthalate and polyethylene naphthalate fibres. J. Mater. Sci. 2011, 46, 528–533. [Google Scholar] [CrossRef]
- Blair, B.; Ricci, A.J.L.; Casar, R.S. The effect of mechanical strain on rat tendon fibroblasts grown on a three-dimensional scaffold. In Proceedings of Transactions of the Annual Meeting—Orthopaedic Research Society, New Orleans, LA, USA, 21–24 February 1994.
- Dan, L.; Chua, C.K.; Leong, K.F. Fibroblast response to interstitial flow: A state-of-the-art review. Biotechnol. Bioeng. 2010, 107, 1–10. [Google Scholar] [CrossRef]
- Jarman-Smith, M.; Bodamyali, T.; Stevens, C.; Howell, J.A.; Horrocks, M.; Chaudhuri, J.B. Human fibroblast culture on a crosslinked dermal porcine collagen matrix. Biochem. Eng. J. 2004, 20, 217–222. [Google Scholar] [CrossRef]
- Park, J.Y.; Yoo, S.J.; Patel, L.; Lee, S.H.; Lee, S.H. Cell morphological response to low shear stress in a two-dimensional culture microsystem with magnitudes comparable to interstitial shear stress. Biorheology 2010, 47, 165–178. [Google Scholar]
- Wang, H.J.; Bertrand-De Haas, M.; Riesle, J.; Lamme, E.; van Blitterswijk, C.A. Tissue engineering of dermal substitutes based on porous PEGT/PBT copolymer scaffolds: Comparison of culture conditions. J. Mater. Sci. 2003, 14, 235–240. [Google Scholar]
- GanjiBakhsh, M.; Nejati, V.; Delirezh, N.; Asadi, M.; Gholami, K. Mixture of fibroblast, epithelial and endothelial cells conditioned media induce monocyte-derived dendritic cell maturation. Cell. Immunol. 2001, 272, 18–24. [Google Scholar]
- Lee, S.R.; Kim, M.O.; Kim, S.H.; Kim, B.S.; Yoo, D.H.; Par, Y.S.; Park, Y.B.; Ha, J.H.; Ryoo, Z.Y. Effect of conditioned medium of mouse embryonic fibroblasts produced form EC-SOD transgenic mice in nuclear maturation of canine oocytes in vitro. Anim. Reprod. Sci. 2007, 99, 106–116. [Google Scholar] [CrossRef]
- Soma, S.; Matsumoto, S.; Takano-Yamamoto, T. Enhancement by Conditioned medium of stretched calvarial bone cells of the osteoclast-like cell formation induced by parathyroid hormone in mouse bone marrow cultures. Arch. Oral Biol. 1997, 42, 205–211. [Google Scholar] [CrossRef]
- Owan, I.; Burr, D.B.; Turner, C.H.; Qiu, J.Y.; Tu, Y.; Onyia, J.E.; Duncan, R.L. Mechanotransduction in bone: Osteoblasts are more responsive to fluid forces than mechanical strain. Amer. J. Physiol.-Cell Physiol. 1997, 273, C810–C815. [Google Scholar]
- Van Dyke, W.S.; Sun, X.; Richard, A.B.; Nauman, E.A.; Akkus, O. Novel mechanical bioreactor for concomitant fluid shear stress and substrate strain. J. Biomech. 2012, 45, 1323–1327. [Google Scholar] [CrossRef]
- You, J.; Yellowley, C.E.; Donahue, H.J.; Zhang, Y.; Chen, Q.; Jacobs, C.R. Substrate deformation levels associated with routine physical activity are less stimulatory to bone cells relative to loading-induced oscillatory fluid flow. J. Biomech. Eng. 2000, 122, 387–393. [Google Scholar] [CrossRef]
- Rust, P.A.; Eckersley, R. Twenty questions on tendon injuries in the hand. Curr. Orthop. 2008, 22, 17–24. [Google Scholar] [CrossRef]
- Sommer, F.; Kobuch, K.; Brandl, F.; Wild, B.; Framme, C.; Weiser, B.; Tessmar, J.; Gabel, V.P.; Blunk, T.; Goepferich, A. Ascorbic acid modulates proliferation and extracellular matrix accumulation of hyalocytes. Tissue Eng. 2007, 13, 1281–1289. [Google Scholar] [CrossRef]
- Webb, K.; Hitchcock, R.W.; Smeal, R.M.; Li, W.; Gray, S.D.; Tresco, P.A. Cyclic strain increases fibroblast proliferation, matrix accumulation, and elastic modulus of fibroblast-seeded polyurethane constructs. J. Biomech. 2006, 39, 136–144. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Goodhart, J.M.; Cooper, J.O.; Smith, R.A.; Williams, J.L.; Haggard, W.O.; Bumgardner, J.D. Design and Validation of a Cyclic Strain Bioreactor to Condition Spatially-Selective Scaffolds in Dual Strain Regimes. Processes 2014, 2, 345-360. https://doi.org/10.3390/pr2020345
Goodhart JM, Cooper JO, Smith RA, Williams JL, Haggard WO, Bumgardner JD. Design and Validation of a Cyclic Strain Bioreactor to Condition Spatially-Selective Scaffolds in Dual Strain Regimes. Processes. 2014; 2(2):345-360. https://doi.org/10.3390/pr2020345
Chicago/Turabian StyleGoodhart, J. Matthew, Jared O. Cooper, Richard A. Smith, John L. Williams, Warren O. Haggard, and Joel D. Bumgardner. 2014. "Design and Validation of a Cyclic Strain Bioreactor to Condition Spatially-Selective Scaffolds in Dual Strain Regimes" Processes 2, no. 2: 345-360. https://doi.org/10.3390/pr2020345
APA StyleGoodhart, J. M., Cooper, J. O., Smith, R. A., Williams, J. L., Haggard, W. O., & Bumgardner, J. D. (2014). Design and Validation of a Cyclic Strain Bioreactor to Condition Spatially-Selective Scaffolds in Dual Strain Regimes. Processes, 2(2), 345-360. https://doi.org/10.3390/pr2020345





