Comparative Characterization of Pumpkin Seed Protein Isolates Obtained by Alkaline, Ultrasound-Assisted, and Microwave-Assisted Extraction: Functionality, Particle Size, and Structural Integrity
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Reagents
2.2. Preparation of Defatted Pumpkin Seed Meal
2.3. Preparation of Pumpkin Seed Protein Isolate
2.4. Ultrasound-Assisted and Microwave-Assisted Extraction
2.5. Characterization of Pumpkin Seed Protein Isolates
2.5.1. Protein Solubility
2.5.2. Water Absorption Capacity and Fat Absorption Capacity
2.5.3. Emulsion Activity and Emulsion Stability
2.5.4. Foaming Capacity and Foaming Stability
2.5.5. Least Gelation Concentration
2.5.6. Particle Size Distribution and Zeta Potential
2.5.7. FTIR Spectroscopy and Secondary Structure Analysis
2.5.8. Scanning Electron Microscopy
2.5.9. Colorimetry
2.5.10. Statistical Analysis
3. Results and Discussion
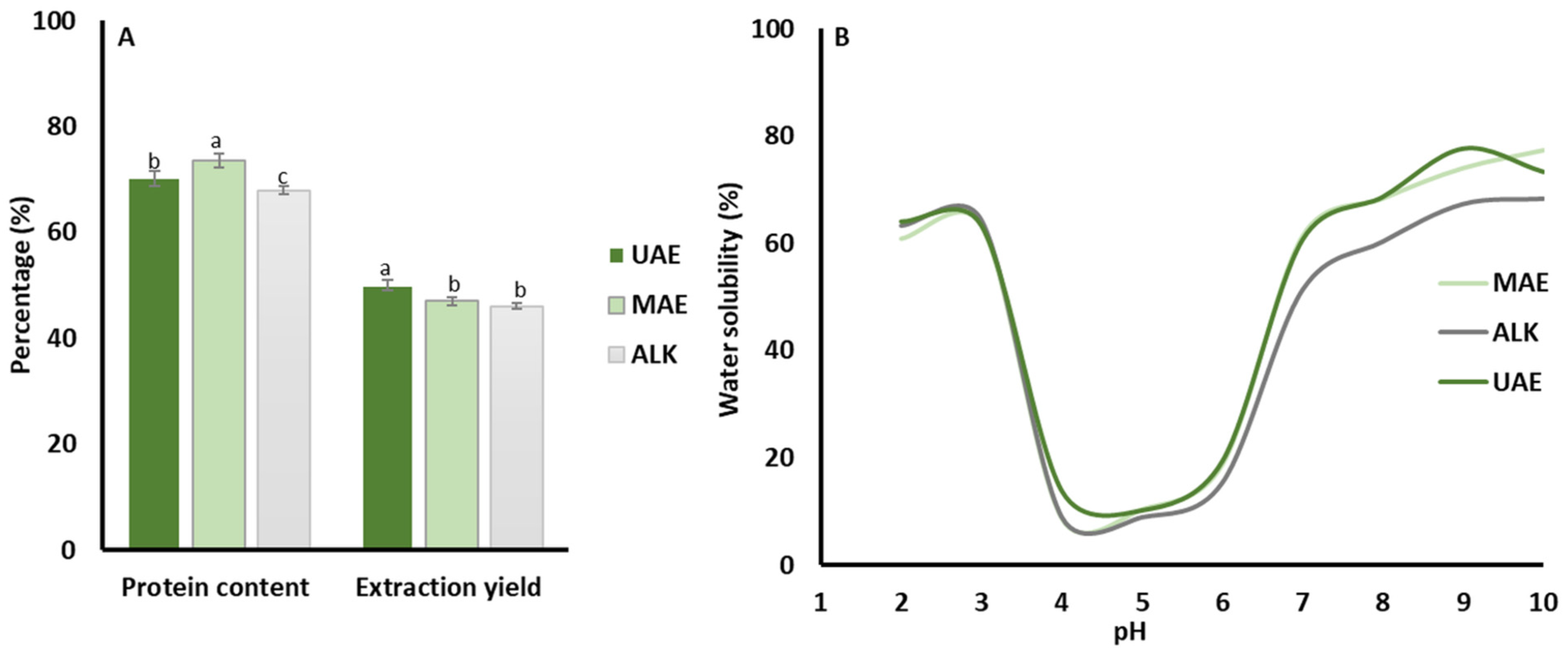
3.1. Protein Yield and Composition
3.2. pH-Dependent Solubility
3.3. Functional Properties
3.3.1. Water and Fat Absorption Capacities
3.3.2. Emulsifying Properties
3.3.3. Foaming Properties
3.3.4. Gelation Behavior
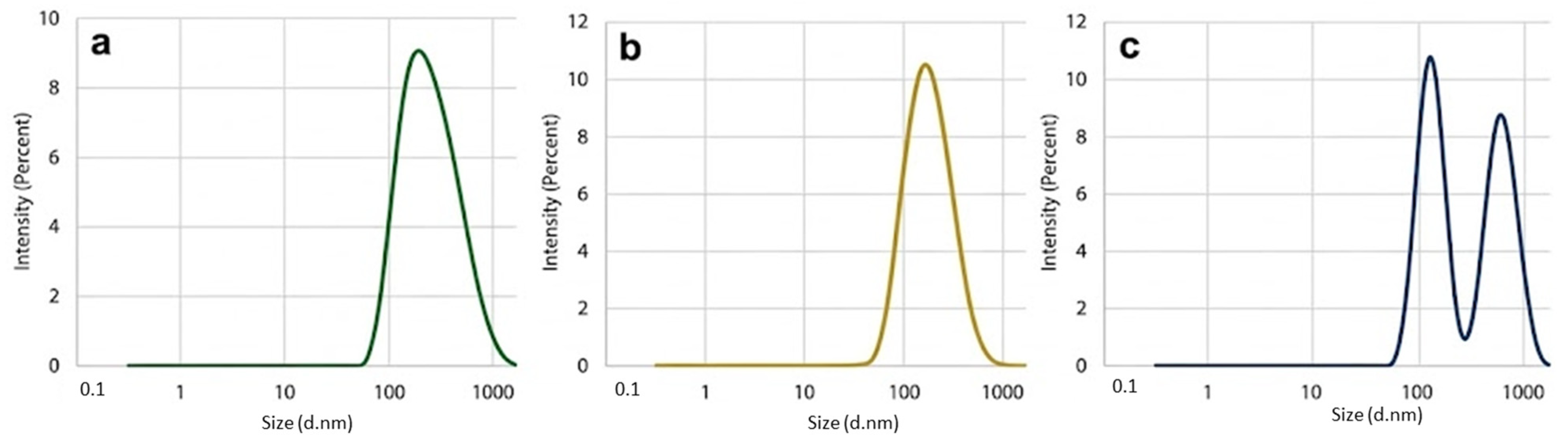
3.4. Colloidal Properties: Particle Size Distribution and Zeta Potential
3.5. Color Characteristics
3.6. FTIR Spectroscopy and Secondary Structure Analysis
3.7. Scanning Electron Microscopy
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations (FAO). The Future of Food and Agriculture: Trends and Challenges; Food and Agriculture Organization of the United Nations: Rome, Italy, 2014; Volume 4.
- Sengar, A.S.; Thirunavookarasu, N.; Choudhary, P.; Naik, M.; Surekha, A.; Sunil, C.; Rawson, A. Application of Power Ultrasound for Plant Protein Extraction, Modification and Allergen Reduction—A Review. Appl. Food Res. 2022, 2, 100219. [Google Scholar] [CrossRef]
- Amin, A.; Petersen, I.L.; Malmberg, C.; Orlien, V. Perspective on the Effect of Protein Extraction Method on the Antinutritional Factor (ANF) Content in Seeds. ACS Food Sci. Technol. 2022, 2, 604–612. [Google Scholar] [CrossRef]
- Negi, P.; Kalsi, R.; Bhasin, J.K.; Kashyap, P.; Thakur, A.; Goksen, G. Ultrasound-Driven Advancements in Food Waste Protein Extraction: Assessing Yield, Nutritional Impacts, Techno-Functionality, and Structural Modifications. Sustain. Chem. Pharm. 2024, 42, 101767. [Google Scholar] [CrossRef]
- Su, G.; Xie, Y.; Liu, R.; Cui, G.; Zhao, M.; Zhang, J. Effect of Transglutaminase on Taste Characteristics of Pea Protein Hydrolysates through Altering the Composition of Amino Acids and Peptides. Food Biosci. 2023, 56, 103261. [Google Scholar] [CrossRef]
- Chemat, F.; Vian, M.A.; Ravi, H.K.; Khadhraoui, B.; Hilali, S.; Perino, S.; Tixier, A.S.F. Review of Alternative Solvents for Green Extraction of Food and Natural Products: Panorama, Principles, Applications and Prospects. Molecules 2019, 24, 3007. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.L.; Yu, P.; Ge, X.L.; Bai, X.F.; Li, X.Q.; Fu, Q. Establishment of an Aqueous PEG 200-Based Deep Eutectic Solvent Extraction and Enrichment Method for Pumpkin (Cucurbita moschata) Seed Protein. Food Anal. Methods 2017, 10, 1669–1680. [Google Scholar] [CrossRef]
- Dotto, J.M.; Chacha, J.S. The Potential of Pumpkin Seeds as a Functional Food Ingredient: A Review: Biofunctional Ingredients of Pumpkin Seeds. Sci. Afr. 2020, 10, e00575. [Google Scholar]
- Syed, Q.A.; Akram, M.; Shukat, R. Nutritional and Therapeutic Importance of the Pumpkin Seeds. Biomed. J. Sci. Tech. Res. 2019, 21, 15798–15803. [Google Scholar] [CrossRef]
- Lemus-Mondaca, R.; Marin, J.; Rivas, J.; Sanhueza, L.; Soto, Y.; Vera, N.; Puente-Díaz, L. Pumpkin Seeds (Cucurbita maxima): A Review of Functional Attributes and by-Products. Rev. Chil. Nutr. 2019, 46, 783–791. [Google Scholar] [CrossRef]
- Sert, D.; Rohm, H.; Struck, S. Ultrasound-Assisted Extraction of Protein from Pumpkin Seed Press Cake: Impact on Protein Yield and Techno-Functionality. Foods 2022, 11, 4029. [Google Scholar] [CrossRef]
- Jafari, M.; Goli, S.A.H.; Rahimmalek, M. The Chemical Composition of the Seeds of Iranian Pumpkin Cultivars and Physicochemical Characteristics of the Oil Extract. Eur. J. Lipid Sci. Technol. 2012, 114, 161–167. [Google Scholar] [CrossRef]
- Lalnunthari, C.; Devi, L.M.; Amami, E.; Badwaik, L.S. Valorisation of Pumpkin Seeds and Peels into Biodegradable Packaging Films. Food Bioprod. Process. 2019, 118, 58–66. [Google Scholar] [CrossRef]
- Yang, C.; Wang, B.; Wang, J.; Xia, S.; Wu, Y. Effect of Pyrogallic Acid (1,2,3-Benzenetriol) Polyphenol-Protein Covalent Conjugation Reaction Degree on Structure and Antioxidant Properties of Pumpkin (Cucurbita sp.) Seed Protein Isolate. LWT 2019, 109, 443–449. [Google Scholar] [CrossRef]
- Das, M.; Devi, L.M.; Badwaik, L.S. Ultrasound-Assisted Extraction of Pumpkin Seeds Protein and Its Physicochemical and Functional Characterization. Appl. Food Res. 2022, 2, 100121. [Google Scholar] [CrossRef]
- Xu, X.; Liu, H.; Duan, S.; Liu, X.; Zhang, K.; Tu, J. A Novel Pumpkin Seeds Protein-Pea Starch Edible Film: Mechanical, Moisture Distribution, Surface Hydrophobicity, UV-Barrier Properties and Potential Application. Mater. Res. Express 2019, 6, 045304. [Google Scholar] [CrossRef]
- Hassan, B.; Ali, S.; Chatha, S.S.A.S.; Hussain, A.I.; Zia, K.M.; Akhtar, N. Recent Advances on Polysaccharides, Lipids and Protein Based Edible Films and Coatings: A Review. Int. J. Biol. Macromol. 2018, 109, 1095–1107. [Google Scholar] [CrossRef] [PubMed]
- Galus, S. Functional Properties of Soy Protein Isolate Edible Films as Affected by Rapeseed Oil Concentration. Food Hydrocoll. 2018, 85, 233–241. [Google Scholar] [CrossRef]
- Lalnunthari, C.; Devi, L.M.; Badwaik, L.S. Extraction of Protein and Pectin from Pumpkin Industry By-Products and Their Utilization for Developing Edible Film. J. Food Sci. Technol. 2020, 57, 1807–1816. [Google Scholar] [CrossRef]
- Kumar, Y.; Singh, L.; Sharanagat, V.S.; Patel, A.; Kumar, K. Effect of Microwave Treatment (Low Power and Varying Time) on Potato Starch: Microstructure, Thermo-Functional, Pasting and Rheological Properties. Int. J. Biol. Macromol. 2020, 155, 27–35. [Google Scholar] [CrossRef]
- Zhou, L.; Zhang, W.; Wang, J.; Zhang, R.; Zhang, J. Comparison of Oil-in-Water Emulsions Prepared by Ultrasound, High-Pressure Homogenization and High-Speed Homogenization. Ultrason. Sonochem. 2022, 82, 105885. [Google Scholar] [CrossRef]
- Prandi, B.; Di Massimo, M.; Tedeschi, T.; Rodríguez-Turienzo, L.; Rodríguez, Ó. Ultrasound and Microwave-Assisted Extraction of Proteins from Coffee Green Beans: Effects of Process Variables on the Protein Integrity. Food Bioprocess Technol. 2022, 15, 2712–2722. [Google Scholar] [CrossRef]
- Hui, D.; Liang, W.; Wang, R.; Feng, X.; Tang, X. A Review of Gelation of Plant Proteins and Their Influencing Factors. Sustain. Food Proteins 2025, 3, e70037. [Google Scholar] [CrossRef]
- Ochoa-Rivas, A.; Nava-Valdez, Y.; Serna-Saldívar, S.O.; Chuck-Hernández, C. Microwave and Ultrasound to Enhance Protein Extraction from Peanut Flour under Alkaline Conditions: Effects in Yield and Functional Properties of Protein Isolates. Food Bioprocess Technol. 2017, 10, 543–555. [Google Scholar] [CrossRef]
- Vinayashree, S.; Vasu, P. Biochemical, Nutritional and Functional Properties of Protein Isolate and Fractions from Pumpkin (Cucurbita moschata Var. Kashi Harit) Seeds. Food Chem. 2021, 340, 128177. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Helikh, A.; Duan, Z. Determining The Effect Of Ph-Shifting Treatment On The Solubility Of Pumpkin Seed Protein Isolate. East.-Eur. J. Enterp. Technol. 2021, 5, 29–34. [Google Scholar] [CrossRef]
- Bučko, S.; Katona, J.; Popović, L.; Vaštag, Ž.; Petrović, L.; Vučiniće-Vasić, M. Investigation on Solubility, Interfacial and Emulsifying Properties of Pumpkin (Cucurbita pepo) Seed Protein Isolate. LWT 2015, 64, 609–615. [Google Scholar] [CrossRef]
- Tas, O.; Sumnu, S.G.; Oztop, M.H. Effect of Extraction Methods and Preheat Treatments on the Functional Properties of Pumpkin Seed Protein Concentrate. ACS Food Sci. Technol. 2024, 5, 105–117. [Google Scholar] [CrossRef] [PubMed]
- AACC International. Approved Methods of Analysis, 11th ed.; Method 46-10.01. Crude Protein—Improved Kjeldahl Method (Archived); Cereals & Grains Association: St. Paul, MN, USA, 2010; Available online: https://www.cerealsgrains.org/resources/Methods/Pages/46Nitrogen.aspx (accessed on 12 March 2024).
- Wang, H.; Chen, K.; Cheng, J.; Jiang, L.; Yu, D.; Dai, Y.; Wang, L. Ultrasound-Assisted Three Phase Partitioning for Simultaneous Extraction of Oil, Protein and Polysaccharide from Pumpkin Seeds. LWT 2021, 151, 112200. [Google Scholar] [CrossRef]
- Bera, M.B.; Mukherjee, R.K. Solubility, Emulsifying, and Foaming Properties of Rice Bran Protein Concentrates. J. Food Sci. 1989, 54, 142–145. [Google Scholar] [CrossRef]
- Brishti, F.H.; Zarei, M.; Muhammad, S.K.S.; Ismail-Fitry, M.R.; Shukri, R.; Saari, N. Evaluation of the Functional Properties of Mung Bean Protein Isolate for Development of Textured Vegetable Protein. Int. Food Res. J. 2017, 24, 1595–1605. [Google Scholar]
- Lawal, O.S.; Adebowale, K.O.; Ogunsanwo, B.M.; Sosanwo, O.A.; Bankole, S.A. On the Functional Properties of Globulin and Albumin Protein Fractions and Flours of African Locust Bean (Parkia biglobossa). Food Chem. 2005, 92, 681–691. [Google Scholar] [CrossRef]
- Tang, J.; Cases, L.; Alves, S.; Sun, D.W.; Tiwari, B.K. Protein Extraction from Lupin (Lupinus angustifolius L.) Using Combined Ultrasound and Microwave Techniques: Impact on Protein Recovery, Structure, and Functional Properties. Ultrason. Sonochem. 2025, 115, 107232. [Google Scholar] [CrossRef]
- Náthia-Neves, G.; Getachew, A.T.; Santana, Á.L.; Jacobsen, C. Legume Proteins in Food Products: Extraction Techniques, Functional Properties, and Current Challenges. Foods 2025, 14, 1626. [Google Scholar] [CrossRef]
- Shreyas, H.K.; Adhikari, P.; Yasmeen Shaikh, A.E.; Arya, S.S. Hydrodynamic and Ultrasonic Cavitation Physically Modifies the Milk Protein Concentrates with Improved Functionality. Sustain. Food Technol. 2026, 4, 1004–1020. [Google Scholar] [CrossRef]
- Zakki, A.; Aryanti, N.; Hadiyanto, H. Functional and Structural Characteristic of Plant Protein Isolates as Emulsifier by Ultrasound-Assisted Extraction: A Review. Bioact. Carbohydr. Diet. Fibre 2024, 32, 100449. [Google Scholar] [CrossRef]
- Fematt-Flores, G.E.; Aguiló-Aguayo, I.; Marcos, B.; Camargo-Olivas, B.A.; Sánchez-Vega, R.; Soto-Caballero, M.C.; Salas-Salazar, N.A.; Flores-Córdova, M.A.; Rodríguez-Roque, M.J. Milk Protein-Based Edible Films: Influence on Mechanical, Hydrodynamic, Optical and Antioxidant Properties. Coatings 2022, 12, 196. [Google Scholar] [CrossRef]
- Ribeiro, F.P.; Naito, R.S.; dos Santos Galvão, B.; Júnior, W.F.; Toyonaga, K.K.; Branco, I.G.; Ferreira, S.; Malacrida, C.R. Innovative Optimized Protein Extraction from Pequi Almond Cake: A Comparison of Ultrasound-Assisted and Alkaline Extraction Methods. Food Chem. X 2025, 29, 102746. [Google Scholar] [CrossRef] [PubMed]
- Cui, B.; Zhang, Y.; Zhu, C.; Li, K.; Wang, Y.; Wang, Y.; Ma, W.; Bai, Y. Ultrasound-mediated fabrication of chickpea protein nanoparticles for stabilizing Pickering emulsions. Ultrason. Sonochem. 2025, 121, 107574. [Google Scholar] [CrossRef] [PubMed]
- Náthia-Neves, G.; Echers, S.G.; Getachew, A.T.; Jacobsen, C. Microwave-Assisted Extraction Drives Changes in the Proteomic Profile, Structural, and Functional Properties of Lupin Protein Isolates. Food Hydrocoll. 2026, 175, 112492. [Google Scholar] [CrossRef]
- Meurer, M.C.; de Souza, D.; Ferreira Marczak, L.D. Effects of Ultrasound on Technological Properties of Chickpea Cooking Water (Aquafaba). J. Food Eng. 2020, 265, 109688. [Google Scholar] [CrossRef]
- Ajayi, F.F.; Mudgil, P.; Maqsood, S. Molecular Structural Modification of Jack Bean Protein Using Thermo-Shearing/Ultrasound/Microwave Treatments for Improved Extractability, Functional and Gelling Properties: The Underlying Impacts of Matrix Pre-Treatment versus Alkaline-Assisted Extraction. Food Hydrocoll. 2024, 154, 110066. [Google Scholar] [CrossRef]
- Biswas, B.; Sit, N. Effect of Ultrasonication on Functional Properties of Tamarind Seed Protein Isolates. J. Food Sci. Technol. 2020, 57, 2070–2078. [Google Scholar] [CrossRef] [PubMed]
- Jahan, K.; Sultan, Z.; Younis, K.; Mir, S.S.; Yousuf, O. PH-Shift Extraction Followed by Microwave and Ultrasound Modified Functional Properties of Mustard Meal Protein. Biocatal. Agric. Biotechnol. 2024, 60, 103295. [Google Scholar] [CrossRef]
- Pochapski, D.J.; Carvalho Dos Santos, C.; Leite, G.W.; Pulcinelli, S.H.; Santilli, C.V. Zeta Potential and Colloidal Stability Predictions for Inorganic Nanoparticle Dispersions: Effects of Experimental Conditions and Electrokinetic Models on the Interpretation of Results. Langmuir 2021, 37, 13379–13389. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.; Roos, Y.H.; Miao, S. Plant Protein versus Dairy Proteins: A PH-Dependency Investigation on Their Structure and Functional Properties. Foods 2023, 12, 368. [Google Scholar] [CrossRef]
- Chen, Y.; Duan, Q.; Yu, L.; Xie, F. Thermomechanically Processed Chitosan:Gelatin Films Being Transparent, Mechanically Robust and Less Hygroscopic. Carbohydr. Polym. 2021, 272, 118522. [Google Scholar] [CrossRef]
- Malik, M.A.; Sharma, H.K.; Saini, C.S. High Intensity Ultrasound Treatment of Protein Isolate Extracted from Dephenolized Sunflower Meal: Effect on Physicochemical and Functional Properties. Ultrason. Sonochem. 2017, 39, 511–519. [Google Scholar] [CrossRef]
- Tang, S.; Li, Y.; Wang, X.; Zhou, L.; Liu, Z.; Jiang, L.; Guo, C.; Yi, J. Development of Soy-Based Meat Analogues via Wet Twin-Screw Extrusion: Enhancing Textural and Structural Properties Through Whole Yeast Powder Supplementation. Foods 2025, 14, 2479. [Google Scholar] [CrossRef]
- Loushigam, G.; Shanmugam, A. Modifications to Functional and Biological Properties of Proteins of Cowpea Pulse Crop by Ultrasound-Assisted Extraction. Ultrason. Sonochem. 2023, 97, 106448. [Google Scholar] [CrossRef]
- Rahman, M.M.; Lamsal, B.P. Ultrasound-Assisted Extraction and Modification of Plant-Based Proteins: Impact on Physicochemical, Functional, and Nutritional Properties. Compr. Rev. Food Sci. Food Saf. 2021, 20, 1457–1480. [Google Scholar] [CrossRef]
- Coutinho, G.S.M.; Prado, P.M.C.; Ribeiro, A.E.C.; Nickerson, M.T.; Caliari, M.; Júnior, M.S.S. Exploring Microwave-Assisted Extraction on Physicochemical and Functional Properties of Pigeon Pea Protein for Food Applications. J. Food Eng. 2025, 392, 112497. [Google Scholar] [CrossRef]
- Jahan, K.; Fatima, S.; Osama, K.; Younis, K.; Yousuf, O. Boosting Protein Yield from Mustard (Brassica juncea) Meal via Microwave-Assisted Extraction and Advanced Optimization Methods. Biomass Convers. Biorefin. 2023, 13, 16241–16251. [Google Scholar] [CrossRef]
- Scopelliti, P.E.; Borgonovo, A.; Indrieri, M.; Giorgetti, L.; Bongiorno, G.; Carbone, R.; Podestà, A.; Milani, P. The Effect of Surface Nanometre-Scale Morphology on Protein Adsorption. PLoS ONE 2010, 5, e11862. [Google Scholar] [CrossRef] [PubMed]
- Phuangjit, U.; Klinkesorn, U.; Tan, C.P.; Katekhong, W. Enhancing Silkworm Protein Yield, Extraction Efficiency, Structure, Functionality, and Antioxidant Activity Using Ultrasound-, Microwave-, and Freeze–Thaw-Assisted Methods. J. Sci. Food Agric. 2024, 104, 383–390. [Google Scholar] [CrossRef]




| Extraction Method | Zeta Potential (mV) | L* | a* | b* | LGC (%) |
|---|---|---|---|---|---|
| ALK | −29.00 ±0.23 a | 76.79 ± 0.87 b | −3.99 ± 0.08 a | 23.68 ± 1.63 b | 16.00 ± 0.00 a |
| UAE | −29.90 ± 1.11 a | 73.68 ± 0.84 c | −4.37 ± 0.15 b | 25.12 ± 1.70 a | 12.00 ± 0.00 c |
| MAE | −32.40 ± 1.27 b | 78.44 ± 1.32 a | −4.04 ± 0.04 a | 22.80 ± 1.20 c | 14.00 ± 0.00 b |
| Secondary Structure | ALK (%) | UAE (%) | MAE (%) |
|---|---|---|---|
| Alpha-Helix | 20.48 ± 0.85 b | 43.45 ± 0.92 a | 43.77 ± 1.05 a |
| Beta-Sheet | 50.79 ± 1.12 a | 35.05 ± 0.78 b | 30.18 ± 0.89 c |
| Beta-Turn | 19.56 ± 0.64 a | 13.81 ± 0.45 c | 17.10 ± 0.55 b |
| Aggregates | 9.17 ± 0.32 a | 7.69 ± 0.21 b | 8.89 ± 0.41 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zenasni, W.; Tekiner, I.H.; Abdelmoumen, H.; Nejjari, R.; Chergui, A.; Ennahli, S.; Ajal, E.A. Comparative Characterization of Pumpkin Seed Protein Isolates Obtained by Alkaline, Ultrasound-Assisted, and Microwave-Assisted Extraction: Functionality, Particle Size, and Structural Integrity. Processes 2026, 14, 1250. https://doi.org/10.3390/pr14081250
Zenasni W, Tekiner IH, Abdelmoumen H, Nejjari R, Chergui A, Ennahli S, Ajal EA. Comparative Characterization of Pumpkin Seed Protein Isolates Obtained by Alkaline, Ultrasound-Assisted, and Microwave-Assisted Extraction: Functionality, Particle Size, and Structural Integrity. Processes. 2026; 14(8):1250. https://doi.org/10.3390/pr14081250
Chicago/Turabian StyleZenasni, Walid, Ismail Hakkı Tekiner, Hanaa Abdelmoumen, Rachid Nejjari, Abdelhak Chergui, Said Ennahli, and El Amine Ajal. 2026. "Comparative Characterization of Pumpkin Seed Protein Isolates Obtained by Alkaline, Ultrasound-Assisted, and Microwave-Assisted Extraction: Functionality, Particle Size, and Structural Integrity" Processes 14, no. 8: 1250. https://doi.org/10.3390/pr14081250
APA StyleZenasni, W., Tekiner, I. H., Abdelmoumen, H., Nejjari, R., Chergui, A., Ennahli, S., & Ajal, E. A. (2026). Comparative Characterization of Pumpkin Seed Protein Isolates Obtained by Alkaline, Ultrasound-Assisted, and Microwave-Assisted Extraction: Functionality, Particle Size, and Structural Integrity. Processes, 14(8), 1250. https://doi.org/10.3390/pr14081250










