Sustainable Foam-like Carbon as a Flexible Radar Absorbing Material
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Sustainable Porous Carbon Material
2.2. Physicochemical Characterization Methods
2.3. Preparation of Composites Samples
2.4. Electromagnetic Characterization of Sustainable Composites
2.5. Mechanical Properties of Composite Samples
3. Results and Discussion
3.1. Physicochemical Properties
3.2. Mechanical Properties of SPC Composites
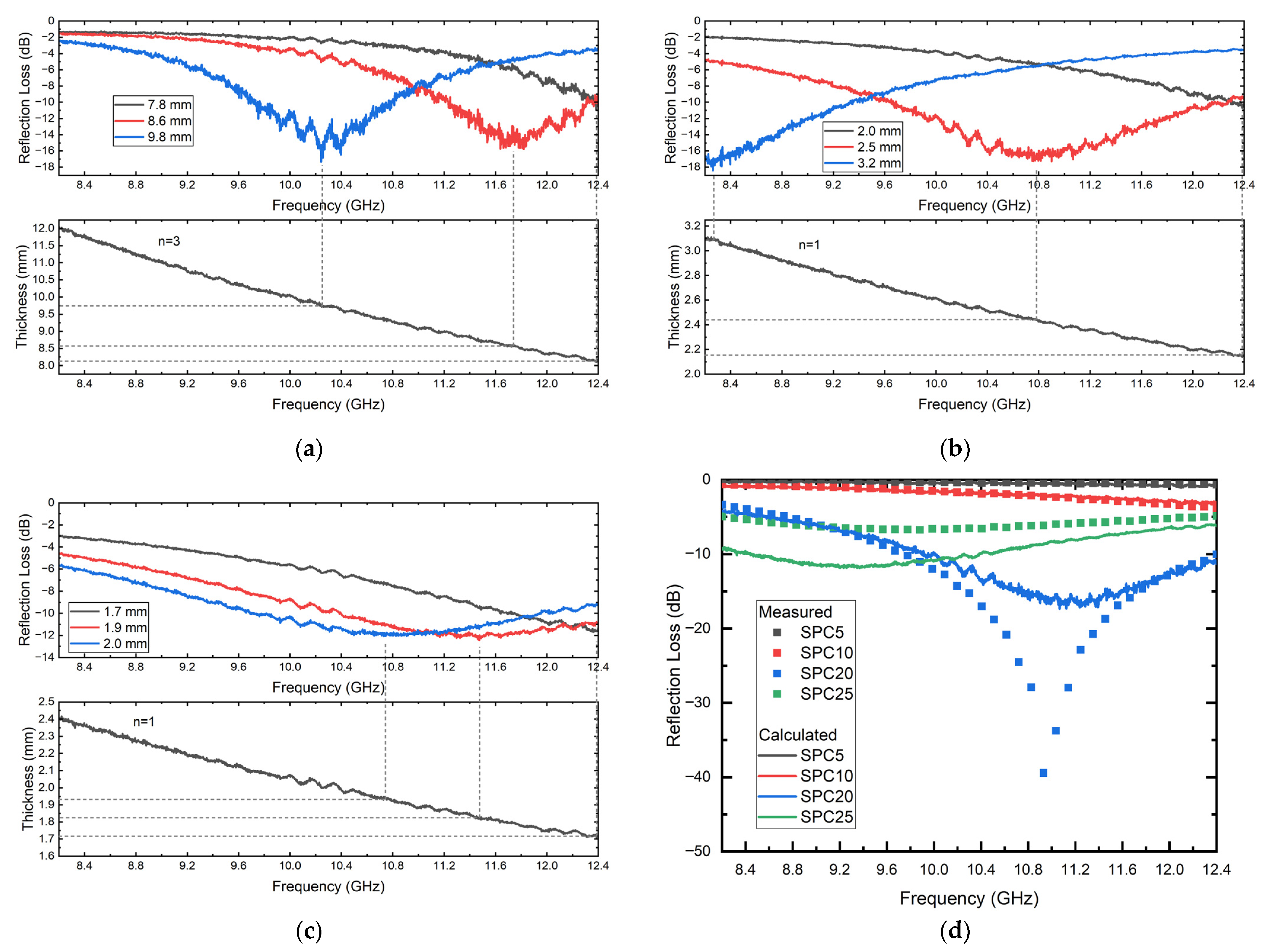
3.3. Electromagnetic Properties of SPC Composites
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| RAM | Radar absorbing materials |
| BET | Brunauer–Emmett–Teller theory |
| EAB | Effective Absorption Bandwidth |
References
- Shukla, V. Review of Electromagnetic Interference Shielding Materials Fabricated by Iron Ingredients. Nanoscale Adv. 2019, 1, 1640–1671. [Google Scholar] [CrossRef]
- Wilson, R.; George, G.; Kuruvilla, J. Chapter 1-An Introduction to Materials for Potential EMI Shielding Applications: Status and Future. In Materials for Potential EMI Shielding Applications Processing, Properties and Current Trends; Wilson, R., George, G., Kuruvilla, J., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2020; pp. 1–8. ISBN 978-0-12-817590-3. [Google Scholar]
- Chahal, R.K.; Kumar, N.; Batra, S. Trust Management in Social Internet of Things: A Taxonomy, Open Issues, and Challenges. Comput. Commun. 2020, 150, 13–46. [Google Scholar] [CrossRef]
- Nord, J.H.; Koohang, A.; Paliszkiewicz, J. The Internet of Things: Review and Theoretical Framework. Expert Syst. Appl. 2019, 133, 97–108. [Google Scholar] [CrossRef]
- Kumar, A.; Gupta, M. A Review on Activities of Fifth Generation Mobile Communication System. Alex. Eng. J. 2018, 57, 1125–1135. [Google Scholar] [CrossRef]
- Skondras, E.; Michalas, A.; Vergados, D.D. Mobility Management on 5G Vehicular Cloud Computing Systems. Veh. Commun. 2019, 16, 15–44. [Google Scholar] [CrossRef]
- ICNIRP (International Commission for Non-Ionizing Radiation Protection) Standing Committee on Epidemiology; Ahlbom, A.; Green, A.; Kheifets, L.; Savitz, D.; Swerdlow, A. Epidemiology of Health Effects of Radiofrequency Exposure. Env. Health Perspect. 2004, 112, 1741–1754. [Google Scholar] [CrossRef]
- Ferna, M.; Silva, F. Mobile Phones Electromagnetic Interference in Medical Environments: A Review. In Proceedings of the 2010 IEEE International Symposium on Electromagnetic Compatibility, Fort Lauderdale, FL, USA, 25–30 July 2010; IEEE: New York NY, USA; pp. 311–316.
- Abate, J.C.; Marinoff, I.I.; Arnal, N.; Machuca, M.; Papa-Gobbi, R.; Vecchio, L.; Rumbo, M.; Stringa, P.; Lausada, N.R. Effect of Multiorgan Abdominal Ischemic Preconditioning on Experimental Kidney Transplantation. Acta Cir. Bras. 2025, 40, e400225. [Google Scholar] [CrossRef]
- Xie, S.; Ji, Z.; Zhu, L.; Zhang, J.; Cao, Y.; Chen, J.; Liu, R.; Wang, J. Recent Progress in Electromagnetic Wave Absorption Building Materials. J. Build. Eng. 2020, 27, 100963. [Google Scholar] [CrossRef]
- Wanasinghe, D.; Aslani, F. A Review on Recent Advancement of Electromagnetic Interference Shielding Novel Metallic Materials and Processes. Compos. Part B Eng. 2019, 176, 107207. [Google Scholar] [CrossRef]
- Cheng, J.; Li, C.; Xiong, Y.; Zhang, H.; Raza, H.; Ullah, S.; Wu, J.; Zheng, G.; Cao, Q.; Zhang, D.; et al. Recent Advances in Design Strategies and Multifunctionality of Flexible Electromagnetic Interference Shielding Materials. Nano-Micro Lett. 2022, 14, 80. [Google Scholar] [CrossRef]
- Chung, D.D.L. Materials for Electromagnetic Interference Shielding. Mater. Chem. Phys. 2020, 255, 123587. [Google Scholar] [CrossRef]
- Lima, U.R.; Nasar, M.C.; Nasar, R.S.; Rezende, M.C.; Araújo, J.H.; Oliveira, J.F. Synthesis of NiCuZn Ferrite Nanoparticles and Microwave Absorption Characterization. Mater. Sci. Eng. B 2008, 151, 238–242. [Google Scholar] [CrossRef]
- Rybicki, T.; Stempien, Z.; Karbownik, I. EMI Shielding and Absorption of Electroconductive Textiles with PANI and PPy Conductive Polymers and Numerical Model Approach. Energies 2021, 14, 7746. [Google Scholar] [CrossRef]
- Yang, J.; Wei, R.; Zhang, C.; Liu, Y.; Zhu, H. Porous and Compressible Silicone Rubber Composites Integrated with Progressive Magneto-Electric Networks for Electromagnetic Interference Shielding. J. Mater. Sci. Technol. 2026, 254, 117–125. [Google Scholar] [CrossRef]
- Wan, Y.; Zhu, P.; Yu, S.; Sun, R.; Wong, C.; Liao, W. Anticorrosive, Ultralight, and Flexible Carbon-Wrapped Metallic Nanowire Hybrid Sponges for Highly Efficient Electromagnetic Interference Shielding. Small 2018, 14, 1800534. [Google Scholar] [CrossRef]
- Li, Y.; Feng, Z.; Huang, L.; Essa, K.; Bilotti, E.; Zhang, H.; Peijs, T.; Hao, L. Additive Manufacturing High Performance Graphene-Based Composites: A Review. Compos. Part A Appl. Sci. Manuf. 2019, 124, 105483. [Google Scholar] [CrossRef]
- Xie, H.; Zhou, Y.; Ren, Z.; Wei, X.; Tao, S.; Yang, C. Enhancement of Electromagnetic Interference Shielding and Heat-Resistance Properties of Silver-Coated Carbonyl Iron Powders Composite Material. J. Magn. Magn. Mater. 2020, 499, 166244. [Google Scholar] [CrossRef]
- Joseph, N.; Thomas Sebastian, M. Electromagnetic Interference Shielding Nature of PVDF-Carbonyl Iron Composites. Mater. Lett. 2013, 90, 64–67. [Google Scholar] [CrossRef]
- Im, J.S.; Kim, J.G.; Lee, Y.-S. Fluorination Effects of Carbon Black Additives for Electrical Properties and EMI Shielding Efficiency by Improved Dispersion and Adhesion. Carbon 2009, 47, 2640–2647. [Google Scholar] [CrossRef]
- Xie, S.; Ji, Z.; Yang, Y.; Hou, G.; Wang, J. Electromagnetic Wave Absorption Enhancement of Carbon Black/Gypsum Based Composites Filled with Expanded Perlite. Compos. Part B Eng. 2016, 106, 10–19. [Google Scholar] [CrossRef]
- Jung, M.; Lee, Y.; Hong, S.-G.; Moon, J. Carbon Nanotubes (CNTs) in Ultra-High Performance Concrete (UHPC): Dispersion, Mechanical Properties, and Electromagnetic Interference (EMI) Shielding Effectiveness (SE). Cem. Concr. Res. 2020, 131, 106017. [Google Scholar] [CrossRef]
- Shao, R.; Wang, F.; Yang, S.; Jin, J.; Li, G. Preparation of Hollow Porous Carbon Nanofibers and Their Performance and Mechanism of Broadband Microwave Absorption. Materials 2022, 15, 7273. [Google Scholar] [CrossRef]
- Chen, S.; Cui, C.; Yan, J.; Lin, C.; Jiang, S.; Tang, H.; Guo, R. Lightweight Carbon Fiber Aerogel@hollow Carbon/Co3O4 Microsphere for Broadband Electromagnetic Wave Absorption in X and Ku Bands. Carbon 2024, 230, 119617. [Google Scholar] [CrossRef]
- Medeiros, N.C.F.L.; Amaral-Labat, G.; De Medeiros, L.I.; Boss, A.F.N.; Da Silva Fonseca, B.C.; De Castro Munhoz, M.G.; Lenz E Silva, G.F.B.; Baldan, M.R.; Braghiroli, F.L. Optimizing Activation Temperature of Sustainable Porous Materials Derived from Forestry Residues: Applications in Radar-Absorbing Technologies. JRM 2025, 13, 1021–1042. [Google Scholar] [CrossRef]
- Boss, A.F.N.; Munhoz, M.G.C.; Amaral-Labat, G.; Lima, R.G.A.; Medeiros, L.I.; Medeiros, N.C.F.L.; Fonseca, B.C.S.; Braghiroli, F.L.; Silva, G.F.B.L.E. Why Sustainable Porous Carbon Should Be Further Explored as Radar-Absorbing Material? A Comparative Study with Different Nanostructured Carbons. JRM 2024, 12, 1639–1659. [Google Scholar] [CrossRef]
- Boss, A.F.; Braghiroli, F.L.; Amaral-Labat, G.; Souza, A.A.; Baldan, M.R.; Bouafif, H.; Koubaa, A.; Lenz E Silva, G.F. Dielectric Characterization of White Birch–Activated Biochar Composites: A Sustainable Alternative to Radar-Absorbing Materials. J. Compos. Mater. 2020, 54, 1233–1244. [Google Scholar] [CrossRef]
- Amaral-Labat, G.; Da Silva, E.L.; Cuña, A.; Malfatti, C.F.; Marcuzzo, J.S.; Baldan, M.R.; Celzard, A.; Fierro, V.; Lenz E Silva, G.F.B. A Sustainable Carbon Material from Kraft Black Liquor as Nickel-Based Electrocatalyst Support for Ethanol Electro-Oxidation. Waste Biomass Valor. 2021, 12, 2507–2519. [Google Scholar] [CrossRef]
- BioResources. Preparation and Characterization of Organic Aerogels from a Lignin–Resourcinol–Formaldehyde Copolymer. Available online: https://bioresources.cnr.ncsu.edu/ (accessed on 30 January 2026).
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Singh, P.; Babbar, V.K.; Razdan, A.; Puri, R.K.; Goel, T.C. Complex Permittivity, Permeability, and X-Band Microwave Absorption of CaCoTi Ferrite Composites. J. Appl. Phys. 2000, 87, 4362–4366. [Google Scholar] [CrossRef]
- Kim, S.S.; Jo, S.B.; Gueon, K.I.; Choi, K.K.; Kim, J.M.; Churn, K.S. Complex Permeability and Permittivity and Microwave Absorption of Ferrite-Rubber Composite at X-Band Frequencies. IEEE Trans. Magn. 1991, 27, 5462–5464. [Google Scholar] [CrossRef]
- Hidd, C.; Morales, G.; Monsalves, N.; Vidal, G. Toxic Effects of Liquors Generated During Kraft Pulp Production Process on Aerobic Biomass and Growth of Selenastrum Capricornutum. Sustainability 2025, 17, 4494. [Google Scholar] [CrossRef]
- Amaral-Labat, G.; Munhoz, M.G.C.; Fonseca, B.C.D.S.; Boss, A.F.N.; De Almeida-Mattos, P.; Braghiroli, F.L.; Bouafif, H.; Koubaa, A.; Lenz E Silva, G.F.B.; Baldan, M.R. Xerogel-like Materials from Sustainable Sources: Properties and Electrochemical Performances. Energies 2021, 14, 7977. [Google Scholar] [CrossRef]
- Pola, L.; Collado, S.; Oulego, P.; Díaz, M. Kraft Black Liquor as a Renewable Source of Value-Added Chemicals. Chem. Eng. J. 2022, 448, 137728. [Google Scholar] [CrossRef]
- Davis, T.W.; Mahmud, R.; McNaul, S.; Jamieson, M.; Lewis, E. A Comparative Analysis of Waste-as-a-Feedstock Accounting Methods in Life Cycle Assessments. Hydrogen 2025, 6, 74. [Google Scholar] [CrossRef]
- Füchsl, S.; Huber, J.; Fröhling, M.; Röder, H. Balancing the Green Carbon Cycle—Biogenic Carbon within Life Cycle Assessment. Int. J. Life Cycle Assess. 2025, 30, 2300–2313. [Google Scholar] [CrossRef]
- Albert, D.F.; Andrews, G.R.; Mendenhall, R.S.; Bruno, J.W. Supercritical Methanol Drying as a Convenient Route to Phenolic–Furfural Aerogels. J. Non-Cryst. Solids 2001, 296, 1–9. [Google Scholar] [CrossRef]
- Braghiroli, F.L.; Amaral-Labat, G.; Boss, A.F.N.; Lacoste, C.; Pizzi, A. Tannin Gels and Their Carbon Derivatives: A Review. Biomolecules 2019, 9, 587. [Google Scholar] [CrossRef] [PubMed]
- Amaral-Labat, G.; Grishechko, L.I.; Fierro, V.; Kuznetsov, B.N.; Pizzi, A.; Celzard, A. Tannin-Based Xerogels with Distinctive Porous Structures. Biomass Bioenergy 2013, 56, 437–445. [Google Scholar] [CrossRef]
- Maj, I.; Niesporek, K.; Płaza, P.; Maier, J.; Łój, P. Biomass Ash: A Review of Chemical Compositions and Management Trends. Sustainability 2025, 17, 4925. [Google Scholar] [CrossRef]
- Portes, R.C.; Baldan, M.R.; Amaral-Labat, G. From Waste to Resource: Transforming Kraft Black Liquor into Sustainable Porous Carbon Fillers for Radome Applications. Chem. Eng. J. 2024, 497, 155007. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of Gases, with Special Reference to the Evaluation of Surface Area and Pore Size Distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Amaral-Labat, G.; Szczurek, A.; Fierro, V.; Pizzi, A.; Celzard, A. Systematic Studies of Tannin–Formaldehyde Aerogels: Preparation and Properties. Sci. Technol. Adv. Mater. 2013, 14, 015001. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Shishkin, A.; Koppel, T.; Gupta, N. A Review of Porous Lightweight Composite Materials for Electromagnetic Interference Shielding. Compos. Part B Eng. 2018, 149, 188–197. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, Y.; Li, Z.; Yang, Z.; Shen, C.; He, C. Effect of Pore Morphology on the Dielectric Properties of Porous Carbons for Microwave Absorption Applications. J. Phys. Chem. C 2014, 118, 26027–26032. [Google Scholar] [CrossRef]
- Delgado-Plana, P.; Bueno-Rodríguez, S.; Pérez-Villarejo, L.; Eliche-Quesada, D. Synthesis of Solid Sodium Silicate from Waste Glass and Utilization on One-Part Alkali-Activated Materials Based on Spent Oil Filtering Earth. Env. Sci. Pollut. Res. Int. 2025, 32, 27763–27785. [Google Scholar] [CrossRef]
- Gupta, S. CO2 Assisted Geo-Polymerization: A Win-Win Pragmatic Approach for the Synthesis of Soda Ash Leading to Reversal of the Climate Clock. RSC Sustain. 2024, 2, 3782–3787. [Google Scholar] [CrossRef]
- Petrus, H.T.B.M.; Olvianas, M.; Shafiyurrahman, M.F.; Pratama, I.G.A.A.N.; Jenie, S.N.A.; Astuti, W.; Nurpratama, M.I.; Ekaputri, J.J.; Anggara, F. Circular Economy of Coal Fly Ash and Silica Geothermal for Green Geopolymer: Characteristic and Kinetic Study. Gels 2022, 8, 233. [Google Scholar] [CrossRef] [PubMed]
- Sadezky, A.; Muckenhuber, H.; Grothe, H.; Niessner, R.; Pöschl, U. Raman Microspectroscopy of Soot and Related Carbonaceous Materials: Spectral Analysis and Structural Information. Carbon 2005, 43, 1731–1742. [Google Scholar] [CrossRef]
- McDonald-Wharry, J.; Manley-Harris, M.; Pickering, K. Carbonisation of Biomass-Derived Chars and the Thermal Reduction of a Graphene Oxide Sample Studied Using Raman Spectroscopy. Carbon 2013, 59, 383–405. [Google Scholar] [CrossRef]
- Kgwadibane, T.; Morad, R.; Momodu, D.Y.; Hlongwa, N.W.; Fuku, X.; Madito, M.J. Heteroatom-Doped Electrochemically Exfoliated Graphene Thin Films: A Raman Spectroscopy and Density Functional Theory Study. Diam. Relat. Mater. 2025, 160, 113058. [Google Scholar] [CrossRef]
- Liu, X.; Yuan, L.; Zhong, M.; Ni, S.; Yang, F.; Fu, Z.; Xu, X.; Wang, C.; Tang, Y. Enhanced Capacitive Performance by Improving the Graphitized Structure in Carbon Aerogel Microspheres. RSC Adv. 2020, 10, 22242–22249. [Google Scholar] [CrossRef] [PubMed]
- Casallas Caicedo, F.M.; Vera López, E.; Agarwal, A.; Drozd, V.; Durygin, A.; Franco Hernandez, A.; Wang, C. Synthesis of Graphene Oxide from Graphite by Ball Milling. Diam. Relat. Mater. 2020, 109, 108064. [Google Scholar] [CrossRef]
- Ilić, M.; Haegel, F.-H.; Lolić, A.; Nedić, Z.; Tosti, T.; Ignjatović, I.S.; Linden, A.; Jablonowski, N.D.; Hartmann, H. Surface Functional Groups and Degree of Carbonization of Selected Chars from Different Processes and Feedstock. PLoS ONE 2022, 17, e0277365. [Google Scholar] [CrossRef]
- Long, S.-Y.; Qin, Y.; Liu, J.-L.; Xian, X.-Q.; Zhou, L.-Q.; Lv, W.-D.; Tang, P.-D.; Wang, Q.-Y.; Du, Q.-S. Study on the Lignin-Derived Sp2–Sp3 Hybrid Hard Carbon Materials and the Feasibility for Industrial Production. Sci. Rep. 2024, 14, 5091. [Google Scholar] [CrossRef]
- Yi, W.; Wang, Y.; Wang, G.; Tao, X. Investigation of Carbon Black/Silicone Elastomer/Dimethylsilicone Oil Composites for Flexible Strain Sensors. Polym. Test. 2012, 31, 677–684. [Google Scholar] [CrossRef]
- Yoshimura, K.; Nakano, K.; Hishikawa, Y. Flexible Tactile Sensor Materials Based on Carbon Microcoil/Silicone-Rubber Porous Composites. Compos. Sci. Technol. 2016, 123, 241–249. [Google Scholar] [CrossRef]
- Fu, Y.-F.; Yi, F.-L.; Liu, J.-R.; Li, Y.-Q.; Wang, Z.-Y.; Yang, G.; Huang, P.; Hu, N.; Fu, S.-Y. Super Soft but Strong E-Skin Based on Carbon Fiber/Carbon Black/Silicone Composite: Truly Mimicking Tactile Sensing and Mechanical Behavior of Human Skin. Compos. Sci. Technol. 2020, 186, 107910. [Google Scholar] [CrossRef]
- Liu, Y.-F.; Huang, P.; Li, Y.-Q.; Liu, Q.; Tao, J.-K.; Xiong, D.-J.; Hu, N.; Yan, C.; Wang, H.; Fu, S.-Y. A Biomimetic Multifunctional Electronic Hair Sensor. J. Mater. Chem. A 2019, 7, 1889–1896. [Google Scholar] [CrossRef]
- Quan, B.; Liang, X.; Ji, G.; Cheng, Y.; Liu, W.; Ma, J.; Zhang, Y.; Li, D.; Xu, G. Dielectric Polarization in Electromagnetic Wave Absorption: Review and Perspective. J. Alloys Compd. 2017, 728, 1065–1075. [Google Scholar] [CrossRef]
- Tamura, H. Microwave Dielectric Losses Caused by Lattice Defects. J. Eur. Ceram. Soc. 2006, 26, 1775–1780. [Google Scholar] [CrossRef]
- Fang, Y.; Zhang, Y.; Wang, W.; Wang, S.; Hou, X.; Huang, Y.; Ye, W.; Yang, R.; Zhao, R.; Xue, W.; et al. Modulation of Electromagnetic Wave Absorption via Porosity in Pechini-Derived Carbon Guided by a Random Network Model. Nanoscale 2023, 15, 5855–5864. [Google Scholar] [CrossRef]
- Qin, F.; Peng, M.; Estevez, D.; Brosseau, C. Electromagnetic Composites: From Effective Medium Theories to Metamaterials. J. Appl. Phys. 2022, 132, 101101. [Google Scholar] [CrossRef]
- Bakry, M.; Klinkenbusch, L. Using The Kramers-Kronig Transforms to Retrieve the Conductivity from the Effective Complex Permittivity. Adv. Radio Sci. 2018, 16, 23–28. [Google Scholar] [CrossRef]
- Chung, D.D.L.; Xi, X. Factors That Govern the Electric Permittivity of Carbon Materials in the Graphite Allotrope Family. Carbon 2021, 184, 245–252. [Google Scholar] [CrossRef]
- Zeng, X.; Cheng, X.; Yu, R.; Stucky, G.D. Electromagnetic Microwave Absorption Theory and Recent Achievements in Microwave Absorbers. Carbon 2020, 168, 606–623. [Google Scholar] [CrossRef]
- Wang, T.; Chen, G.; Zhu, J.; Gong, H.; Zhang, L.; Wu, H. Deep Understanding of Impedance Matching and Quarter Wavelength Theory in Electromagnetic Wave Absorption. J. Colloid Interface Sci. 2021, 595, 1–5. [Google Scholar] [CrossRef]
- Panwar, R.; Agarwala, V.; Singh, D. A Cost Effective Solution for Development of Broadband Radar Absorbing Material Using Electronic Waste. Ceram. Int. 2015, 41, 2923–2930. [Google Scholar] [CrossRef]
- Abbasi, M.I.; Ismail, M.Y. Reflection Loss and Bandwidth Performance of X-band Infinite Reflectarrays: Simulations and Measurements. Micro Opt. Tech Lett. 2011, 53, 77–80. [Google Scholar] [CrossRef]
- Negi, P.; Chhantyal, A.K.; Dixit, A.K.; Kumar, S.; Kumar, A. Activated Carbon Derived from Mango Leaves as an Enhanced Microwave Absorbing Material. Sustain. Mater. Technol. 2021, 27, e00244. [Google Scholar] [CrossRef]
- Pan, F.; Liu, Z.; Deng, B.; Dong, Y.; Zhu, X.; Huang, C.; Shi, Z.; Lu, W. Magnetic Fe3S4 LTMCs Micro-Flowers@ Wax Gourd Aerogel-Derived Carbon Hybrids as Efficient and Sustainable Electromagnetic Absorber. Carbon 2021, 179, 554–565. [Google Scholar] [CrossRef]
- Gu, W.; Xia, A.; Jin, C.; Zhang, H.; Li, H.; Ji, G. An Ultralight, Eco-Friendly 3D Porous Carbon Aerogel Derived from Cotton Nano-Cellulose for Infrared Stealth and Microwave Absorption. Carbon 2024, 229, 119565. [Google Scholar] [CrossRef]










| Sample | SBET (m2·g−1) | ρb (g·mL−1) | ρs (g·mL−1) | ϕ (%) | VHg (mL·g−1) | Atotal (m2·g−1) |
|---|---|---|---|---|---|---|
| SPC | 2.9 | 0.80 | 1.83 | 56 | 0.71 | 44 |
| Biosource | Chemical Activator | Material | Filler Loading (wt.%) | Matrix | Thickness (mm) | RL (dB) | Frequency (GHz) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Mango leaves | KOH | Activated Carbon | 20 | Paraffin | 2.5 | ~−21 | ~10 | [72] |
| Wax gourd | - | Aerogel | 20 | Paraffin | 0.5–5.0 | * | - | [73] |
| White birch biomass | KOH/CO2 | Activated Biochar | 2 | Silicone rubber | 5.85 | −8 | 10.56 | [28] |
| Cotton | Nano-cellulose aerogel | 30 | Paraffin | 2.00 | −19.8 | ~10 | [74] | |
| End-of-life wood panels | CO2 | Activated Carbon | 15 | Silicone rubber | 8.4 | −37.2 | 11.3 | [26] |
| Black wattle bark | Carbon | 20 | Silicone rubber | 7.2 | −13.34 | 12.32 | [27] | |
| Black liquor | - | Foam-like carbon | 20 | Silicone rubber | 2.43 | −42.3 | 10.97 | This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Flórez-Vergara, D.E.; Lopes, B.H.K.; Boss, A.F.N.; Lenz e Silva, G.F.B.; Amaral-Labat, G.; Baldan, M.R. Sustainable Foam-like Carbon as a Flexible Radar Absorbing Material. Processes 2026, 14, 1082. https://doi.org/10.3390/pr14071082
Flórez-Vergara DE, Lopes BHK, Boss AFN, Lenz e Silva GFB, Amaral-Labat G, Baldan MR. Sustainable Foam-like Carbon as a Flexible Radar Absorbing Material. Processes. 2026; 14(7):1082. https://doi.org/10.3390/pr14071082
Chicago/Turabian StyleFlórez-Vergara, D. E., B. H. K. Lopes, A. F. N. Boss, G. F. B. Lenz e Silva, G. Amaral-Labat, and M. R. Baldan. 2026. "Sustainable Foam-like Carbon as a Flexible Radar Absorbing Material" Processes 14, no. 7: 1082. https://doi.org/10.3390/pr14071082
APA StyleFlórez-Vergara, D. E., Lopes, B. H. K., Boss, A. F. N., Lenz e Silva, G. F. B., Amaral-Labat, G., & Baldan, M. R. (2026). Sustainable Foam-like Carbon as a Flexible Radar Absorbing Material. Processes, 14(7), 1082. https://doi.org/10.3390/pr14071082







