Metal Oxide Electrode-Based Treatment of Industrial Dyes with Assessment of Performance and Oxidation Efficiency
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Chemical Specifications
2.2. Equipment and Instrumentation
2.3. Synthesis, Characterization, and Electrochemical Evaluation
2.4. UV–Vis Spectrophotometric Quantification of Color Removal and Hydrogen Peroxide
2.5. Chemical Oxygen Demand (COD) Determination
2.6. Hydroxyl Radical Quantification Using the RNO Test
3. Assessment of Experimental Findings
3.1. Electrode Properties and Oxidative Treatment Performance
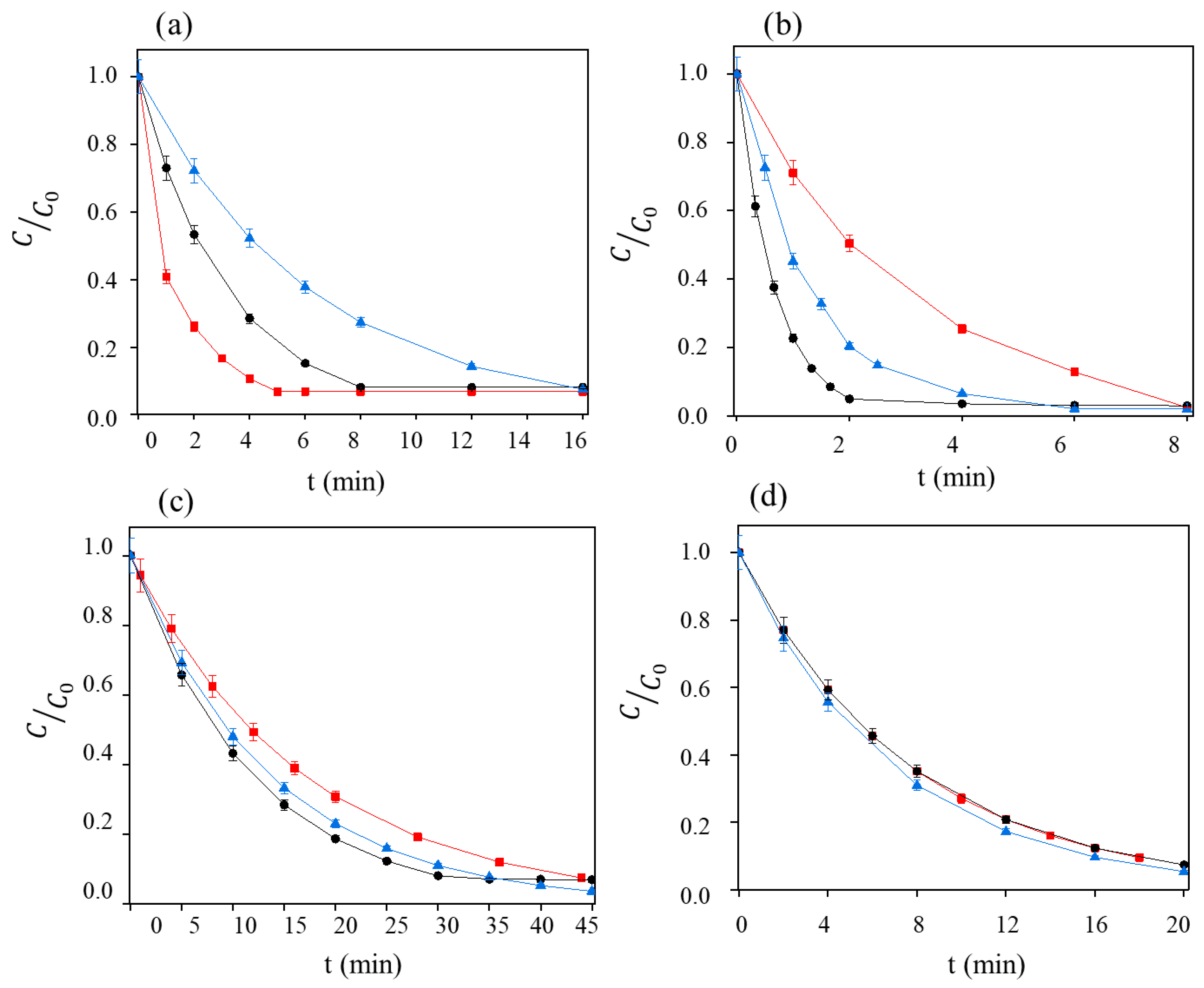
3.2. The Role of Current Density
3.3. Impact of Initial Dye Concentration on Electrochemical Performance
3.4. Impact of the Chosen Electrochemical Oxidation Process
3.5. Determination of the Chemical Oxygen Demand (COD)
3.6. Effect on Oxidant Activity
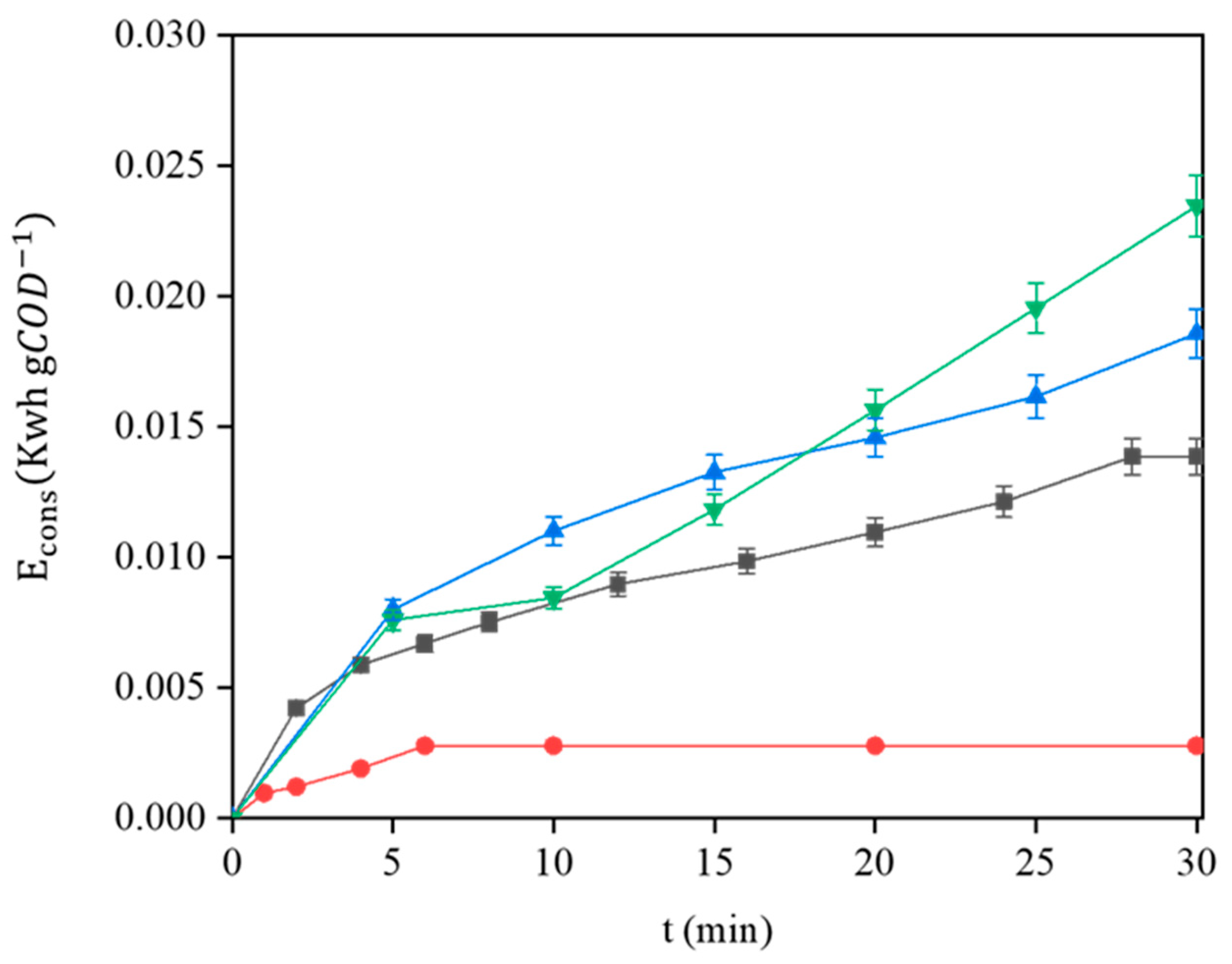
3.7. Energetic Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bravo-Yumi, N.; Pacheco-Álvarez, M.; Bandala, E.R.; Brillas, E.; Peralta-Hernández, J.M. Studying the influence of different parameters on the electrochemical oxidation of tannery dyes using a Ti/IrO2–SnO2–Sb2O5 anode. Chem. Eng. Process. 2022, 181, 109173. [Google Scholar] [CrossRef]
- Chan-Bacab, M.J.; Sanmartín, P.; Camacho-Chab, J.C.; Palomo-Ascanio, K.B.; Huitz-Quimé, H.E.; Ortega-Morales, B.O. Characterization and dyeing potential of colorant-bearing plants of the Mayan area in Yucatan Peninsula, Mexico. J. Clean. Prod. 2015, 91, 191–200. [Google Scholar] [CrossRef]
- Villaseñor-Basulto, D.L.; Picos-Benítez, A.; Pacheco-Álvarez, M.; Pérez, T.; Bandala, E.R.; Peralta-Hernández, J.M. Tannery wastewater treatment using combined electrocoagulation and electro-Fenton processes. J. Environ. Chem. Eng. 2022, 10, 107290. [Google Scholar] [CrossRef]
- Hansen, É.; de Aquim, P.M.; Gutterres, M. Environmental assessment of water, chemicals and effluents in leather post-tanning process: A review. Environ. Impact Assess. Rev. 2021, 89, 106597. [Google Scholar] [CrossRef]
- Singh, K.; Kumari, M.; Prasad, K.S. Tannery effluents: Current practices, environmental consequences, human health risks, and treatment options. Clean Soil Air Water 2022, 50, 2200303. [Google Scholar] [CrossRef]
- Tadesse, G.L. Impacts of tannery effluent on environments and human health: A review article. Adv. Life Sci. Technol. 2017, 54, 58–67. [Google Scholar]
- Mohd, I.; Ahamed, I.; Lichtfouse, E. Water Pollution Remediation: Organic Pollutants; Springer: Cham, Switzerland, 2019. [Google Scholar]
- Zhao, J.; Wu, Q.; Tang, Y.; Zhou, J.; Guo, H. Tannery wastewater treatment: Conventional and promising processes, an updated 20-year review. J. Leather Sci. Eng. 2022, 4, 10. [Google Scholar] [CrossRef]
- Aguilar, Z.G.; Brillas, E.; Salazar, M.; Nava, J.L.; Sirés, I. Evidence of Fenton-like reaction with active chlorine during the electrocatalytic oxidation of Acid Yellow 36 azo dye with Ir–Sn–Sb oxide anode in the presence of iron ion. Appl. Catal. B 2017, 206, 44–52. [Google Scholar] [CrossRef]
- Deng, F.; Brillas, E. Advances in the decontamination of wastewaters with synthetic organic dyes by electrochemical Fenton-based processes. Sep. Purif. Technol. 2023, 316, 123764. [Google Scholar] [CrossRef]
- Pacheco-Álvarez, M.; Fuentes-Ramírez, R.; Brillas, E.; Peralta-Hernández, J.M. Assessing the electrochemical degradation of Reactive Orange 84 with Ti/IrO2–SnO2–Sb2O5 anode using electrochemical oxidation, electro-Fenton, and photoelectro-Fenton under UVA irradiation. Chemosphere 2023, 339, 139666. [Google Scholar] [CrossRef]
- Behrouzeh, M.; Mehdi, M.; Danesh, E. Application of photo-Fenton, electro-Fenton, and photo-electro-Fenton processes for the treatment of DMSO and DMAC wastewaters. Arab. J. Chem. 2022, 15, 104229. [Google Scholar] [CrossRef]
- Zhou, Q.; Chen, H.; Liu, G.; Wang, X. Occurrence, sustainable treatment technologies, potential sources, and future prospects of emerging pollutants in aquatic environments: A review. Front. Environ. Sci. 2024, 12, 1455377. [Google Scholar] [CrossRef]
- Nozari-Asbemarz, M.; Amiri, M.; Imanzadeh, H. Mixed metal oxides as efficient electrocatalysts for water oxidation. Int. J. Hydrogen Energy 2021, 47, 5250–5259. [Google Scholar] [CrossRef]
- Ribeiro, J.Y.C.; Santos, G.O.S.; Dória, A.R.; Requena, I.; Lanza, M.R.V.; Salazar-Banda, G.R.; Eguiluz, K.I.B.; Lobato, J.; Rodrigo, M.A. Platinum-Modified Mixed Metal Oxide Electrodes for Efficient Chloralkaline-Based Energy Storage. Catalysts 2024, 14, 152. [Google Scholar] [CrossRef]
- Ganzoury, M.A.; Ghasemian, S.; Zhang, N.; Yagar, M.; de Lannoy, C.F. Mixed metal oxide anodes used for the electrochemical degradation of a real mixed industrial wastewater. Chemosphere 2022, 286, 131600. [Google Scholar] [CrossRef]
- Alves, L.; Chantelle, L.; Garcia, M.; Menezes, L.; Alves, F.M.C. The influence of synthesis methods and experimental conditions on the photocatalytic properties of SnO2: A review. Catalysts 2022, 12, 428. [Google Scholar] [CrossRef]
- Luna-Trujillo, M.; Palma-Goyes, R.; Vazquez-Arenas, J.; Manzo-Robledo, A. Formation of active chlorine species involving the higher oxide MOx+1 on active Ti/RuO2–IrO2 anodes: A DEMS analysis. J. Electroanal. Chem. 2020, 878, 114661. [Google Scholar] [CrossRef]
- Oakton, E.; Lebedev, D.; Povia, M.; Abbott, D.F.; Fabbri, E.; Fedorov, A.; Nachtegaal, M.; Copéret, C.; Schmidt, T.J. IrO2–TiO2: A high-surface-area, active, and stable electrocatalyst for the oxygen evolution reaction. ACS Catal. 2017, 7, 2346–2352. [Google Scholar] [CrossRef]
- Moradi, M.; Vasseghian, Y.; Khataee, A.; Kobya, M.; Arabzade, H.; Dragoi, E.N. Service life and stability of electrodes applied in electrochemical advanced oxidation processes: A comprehensive review. J. Ind. Eng. Chem. 2020, 87, 18–39. [Google Scholar] [CrossRef]
- Trasatti, S. Electrocatalysis: Understanding the success of DSA®. Electrochim. Acta 2000, 45, 2377–2385. [Google Scholar] [CrossRef]
- Titchou, F.E.; El Ghachtouli, N.; El Hourch, A.; Zaitan, H. Removal of Orange G Dye from Water by Heterogeneous Electro-Fenton-Based Processes Using Ti4O7 Anode and Iron-Based Catalysts. Emergent Mater. 2025, 8, 112–126. [Google Scholar] [CrossRef]
- Zheng, T.-H.; Zhang, Z.-Z.; Liu, Y.; Zou, L.-H. Recent Progress in Catalytically Driven Advanced Oxidation Processes for Wastewater Treatment. Catalysts 2025, 15, 761. [Google Scholar] [CrossRef]
- Najafzadeh, A.A.; Ayati, B. Improvement of Electro-Fenton Process Using Heterogeneous Fe-MIL-88B Nanocatalyst for Dye Removal. Sci. Rep. 2024, 14, 74655. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Venegas, L.A.; Cortés-Muñoz, J.; Arancibia-Miranda, N.; Maturana-Rojas, J.; Rubilar, O. Kinetics of Decolorization of Reactive Textile Dye via Heterogeneous Photocatalysis Using Titanium Dioxide. Water 2024, 16, 633. [Google Scholar] [CrossRef]
- Al-Jabri, H.; Al-Musharfi, M.; Al-Khatri, H.; Al-Amri, I. Heterogeneous Electro-Fenton Process Using a Novel Catalytic Electrode for the Degradation of Direct Dye from Aqueous Solutions. Appl. Water Sci. 2025, 15, 239. [Google Scholar] [CrossRef]
- Agadyekar, V.G.; Kakodkar, E.; Barretto, D.A.; Barni, R.; Riccardi, C.; Joshi, N.G. Concurrent Removal of Benzene, Toluene, and P-Nitrophenol from Water Using Dielectric Barrier Discharge Plasma. Clean. Eng. Technol. 2025, 27, 101042. [Google Scholar] [CrossRef]
- Khourshidi, A.; Ajam, F.; Rabieian, M.; Taghavijeloudar, M. Efficient Degradation of p-Nitrophenol from Water by Enhancing Dielectric Barrier Discharge (DBD) Plasma through Ozone Circulation: Optimization, Kinetics and Mechanism. Chemosphere 2024, 362, 142749. [Google Scholar] [CrossRef]
- Rodríguez, F.A.; Rivero, E.P.; González, I. Adapted Pechini method to prepare DSA type electrodes of RuO2–ZrO2 doped with Sb2O5 over titanium plates. MethodsX 2018, 5, 1613–1617. [Google Scholar] [CrossRef]
- Bravo-Yumi, N.; Espinoza-Montero, P.; Picos-Benítez, A.; Navarro-Mendoza, R.; Brillas, E.; Peralta-Hernández, J.M. Synthesis and characterization of Sb2O5-doped Ti/SnO2–IrO2 anodes toward efficient degradation of tannery dyes by in situ generated oxidizing species. Electrochim. Acta 2020, 358, 136904. [Google Scholar] [CrossRef]
- Masood, Z.; Muhammad, H.; Tahiri, I.A. Comparison of Different Electrochemical Methodologies for Electrode Reactions: A Case Study of Paracetamol. Electrochem 2024, 5, 57–69. [Google Scholar] [CrossRef]
- León, M.I.; Aguilar, Z.G.; Nava, J.L. Electrochemical combustion of indigo at ternary oxide coated titanium anodes. J. Electrochem. Sci. Eng. 2014, 4, 247–258. [Google Scholar] [CrossRef]
- El-Desoky, H.S.; Ghoneim, M.M.; El-Sheikh, R.; Zidan, N.M. Oxidation of Levafix CA reactive azo-dyes in industrial wastewater of textile dyeing by electro-generated Fenton’s reagent. J. Hazard. Mater. 2010, 175, 858–865. [Google Scholar] [CrossRef] [PubMed]
- Mansour, D.; Alblawi, E.; Alsukaibi, A.K.D.; Al Shammari, B. Removal of Congo Red Dye by Electrochemical Advanced Oxidation Process: Optimization, Degradation Pathways, and Mineralization. Sustain. Water Resour. Manag. 2024, 10, 41. [Google Scholar] [CrossRef]
- Cornejo, O.M.; Ortiz, M.; Aguilar, Z.G.; Nava, J.L. Degradation of Acid Violet 19 textile dye by electro-peroxone in a laboratory flow plant. Chemosphere 2021, 271, 129804. [Google Scholar] [CrossRef]
- Pérez, T.; Coria, G.; Sirés, I.; Nava, J.L.; Uribe, A.R. Electrosynthesis of hydrogen peroxide in a filter-press flow cell using graphite felt as air-diffusion cathode. J. Electroanal. Chem. 2018, 812, 54–58. [Google Scholar] [CrossRef]
- Baird, R.; Eaton, A.D.; Rice, E.W.; Bridgewater, L.; American Public Health Association; American Water Works Association; Water Environment Federation (Eds.) Method 5220, D. Chemical Oxygen Demand (Closed reflux, Colorimetric Method). In Standard Methods for the Examination of Water Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017. [Google Scholar]
- García-López, M.; Oturan, N.; Brillas, E. Hydroxyl radicals in anodic oxidation systems: Generation, identification and quantification. Water Res. 2022, 217, 118425. [Google Scholar] [CrossRef]
- Aguilar, Z.G.; Coreño, O.; Salazar, M.; Sirés, I.; Brillas, E.; Nava, J.L. Ti|Ir–Sn–Sb oxide anode: Service life and role of the acid sites content during water oxidation to hydroxyl radicals. J. Electroanal. Chem. 2018, 820, 82–88. [Google Scholar] [CrossRef]
- Dória, A.R.; Silva, R.S.; Júnior, P.H.O.; dos Santos, E.A.; Mattedi, S.; Hammer, P.; Salazar-Banda, G.R.; Eguiluz, K.I. Influence of the RuO2 layer thickness on the physical and electrochemical properties of anodes synthesized by the ionic liquid method. Electrochim. Acta 2020, 354, 136625. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, C.; Shao, D.; Xu, H.; Rao, Y.; Tan, G.; Yan, W. Magnetically assembled electrodes based on Pt, RuO2–IrO2–TiO2 and Sb–SnO2 for electrochemical oxidation of wastewater featured by fluctuant Cl− concentration. J. Hazard. Mater. 2021, 421, 126803. [Google Scholar] [CrossRef]
- Bartošová, A.; Blinová, L.; Sirotiak, M.; Michalíková, A. Usage of FTIR-ATR as non-destructive analysis of selected toxic dyes. Res. Pap. Fac. Mater. Sci. Technol. Trnava 2017, 25, 103–111. [Google Scholar] [CrossRef]
- Bechtold, T.; Aguiló-Aguayo, N.; Pham, T. Cyclic voltammetry of C.I. Disperse Orange 62 in an aqueous electrolyte. Materials 2023, 16, 6901. [Google Scholar] [CrossRef] [PubMed]
- Muff, J.; Bennedsen, L.R.; Søgaard, E.G. Study of electrochemical bleaching of p-nitrosodimethylaniline and its role as hydroxyl radical probe compound. J. Appl. Electrochem. 2011, 41, 599–607. [Google Scholar] [CrossRef]
- Martínez-Hincapié, R.; Wegner, J.; Anwar, M.U.; Raza-Khan, A.; Franzka, S.; Kleszczynski, S.; Čolić, V. The determination of the electrochemically active surface area and its effects on the electrocatalytic properties of structured nickel electrodes produced by additive manufacturing. Electrochim. Acta 2024, 476, 143663. [Google Scholar] [CrossRef]
- Zeng, C.; Tsui, L.S.; Lam, F.L.Y.; Wu, T.; Yip, A.C.K. Revisiting the crucial roles of oxygen vacancies in photo/electro-catalytic degradation of aqueous organic pollutants. Appl. Catal. A Gen. 2024, 694, 120930. [Google Scholar] [CrossRef]
- Brillas, E.; Sirés, I.; Oturan, M.A. Electro-Fenton process and related electrochemical technologies based on Fenton’s reaction chemistry. Chem. Rev. 2009, 109, 6570–6631. [Google Scholar] [CrossRef]
- Sirés, I.; Brillas, E.; Oturan, M.A.; Rodrigo, M.A.; Panizza, M. Electrochemical advanced oxidation processes: Today and tomorrow. Environ. Sci. Pollut. Res. 2014, 21, 8336–8367. [Google Scholar] [CrossRef]
- Barcenas-Grangeno, C.; Pacheco-Álvarez, M.O.A.; Brillas, E.; Sandoval, M.A.; Peralta-Hernández, J.M. Kinetic insights and process selection for electrochemical remediation of industrial dye effluents using mixed electrode systems. Processes 2025, 13, 3439. [Google Scholar] [CrossRef]
- Abilaji, S.; Narenkumar, J.; Das, B.; Rajakrishnan, R.; Sathishkumar, K.; Rajamohan, R.; Rajasekar, A. Electrochemical Oxidation of Azo Dyes Degradation by RuO2–IrO2–TiO2 Electrode with Biodegradation Aeromonas hydrophila. Chemosphere 2023, 345, 140516. [Google Scholar] [CrossRef]
- Lima, A.P.; dos Santos, W.T.; Nossol, E.; Richter, E.M.; Munoz, R.A. Critical evaluation of voltammetric techniques for antioxidant capacity and activity: Presence of alumina on glassy-carbon electrodes alters the results. Electrochim. Acta 2020, 358, 136925. [Google Scholar] [CrossRef]
- Wang, L.; Shao, Y.; Zhao, Z.; Chen, S.; Shao, X. Optimized utilization studies of dredging sediment for making water treatment ceramsite based on an extreme vertex design. J. Water Process Eng. 2020, 38, 101603. [Google Scholar] [CrossRef]
- Sun, N.; Wang, Z.; Sun, B.; Li, Y.; Guo, Z. Purification of primary aluminum liquid through supergravity-induced filtration. Chem. Eng. Process. 2022, 182, 109199. [Google Scholar] [CrossRef]
- Song, K.; Gao, Y.; Yang, Y.; Guo, B.Q.; Wang, Y.Z. Performance of simultaneous carbon and nitrogen removal of high-salinity wastewater in heterotrophic nitrification-aerobic denitrification mode. J. Environ. Chem. Eng. 2023, 11, 109682. [Google Scholar] [CrossRef]
- Ouagued, I.; Cretin, M.; Petit, E.; Lesage, G.; Djafer, A.; Ouagued, A.; Lacour, S. Screening refractory dye degradation by different advanced oxidation processes: Influence of operational parameters including initial concentration and kinetics. Molecules 2025, 30, 712. [Google Scholar] [CrossRef] [PubMed]
- García-Segura, S.; Brillas, E. Applied photoelectrocatalysis on the degradation of organic pollutants in wastewaters. J. Photochem. Photobiol. C 2017, 31, 1–35. [Google Scholar] [CrossRef]
- Bi, H.; Zhao, X.; Ma, K.; Wang, Y.; An, L.; Yang, Q.; Liu, J. Role of molecular structure in the electrochemical degradation of coordination structure dye effluents. J. Environ. Chem. Eng. 2025, 13, 117969. [Google Scholar] [CrossRef]
- Moreira, F.C.; Boaventura, R.A.R.; Brillas, E.; Vilar, V.J.P. Electrochemical advanced oxidation processes: A review on their application to synthetic and real wastewaters. Appl. Catal. B Environ. 2017, 202, 217–261. [Google Scholar] [CrossRef]
- Qiao, Q.; Singh, S.; Lo, S.-L.; Jin, J.; Yu, Y.; Wang, L. Effect of current density and pH on the electrochemically generated active chloro species for the rapid mineralization of p-substituted phenol. Chemosphere 2021, 275, 129848. [Google Scholar] [CrossRef]
- Wei, J.; Liu, Y.; Wu, X. Electrochemical advanced oxidation processes using PbO2 anode and H2O2 electrosynthesis cathode for wastewater treatment. Process Saf. Environ. Prot. 2023, 169, 109634. [Google Scholar] [CrossRef]
- Körbahti, B.K.; Turan, K.M. Evaluation of energy consumption in electrochemical oxidation of Acid Violet 7 textile dye using Pt/Ir electrodes. J. Turk. Chem. Soc. Sect. A Chem. 2016, 3, 75–92. [Google Scholar] [CrossRef][Green Version]


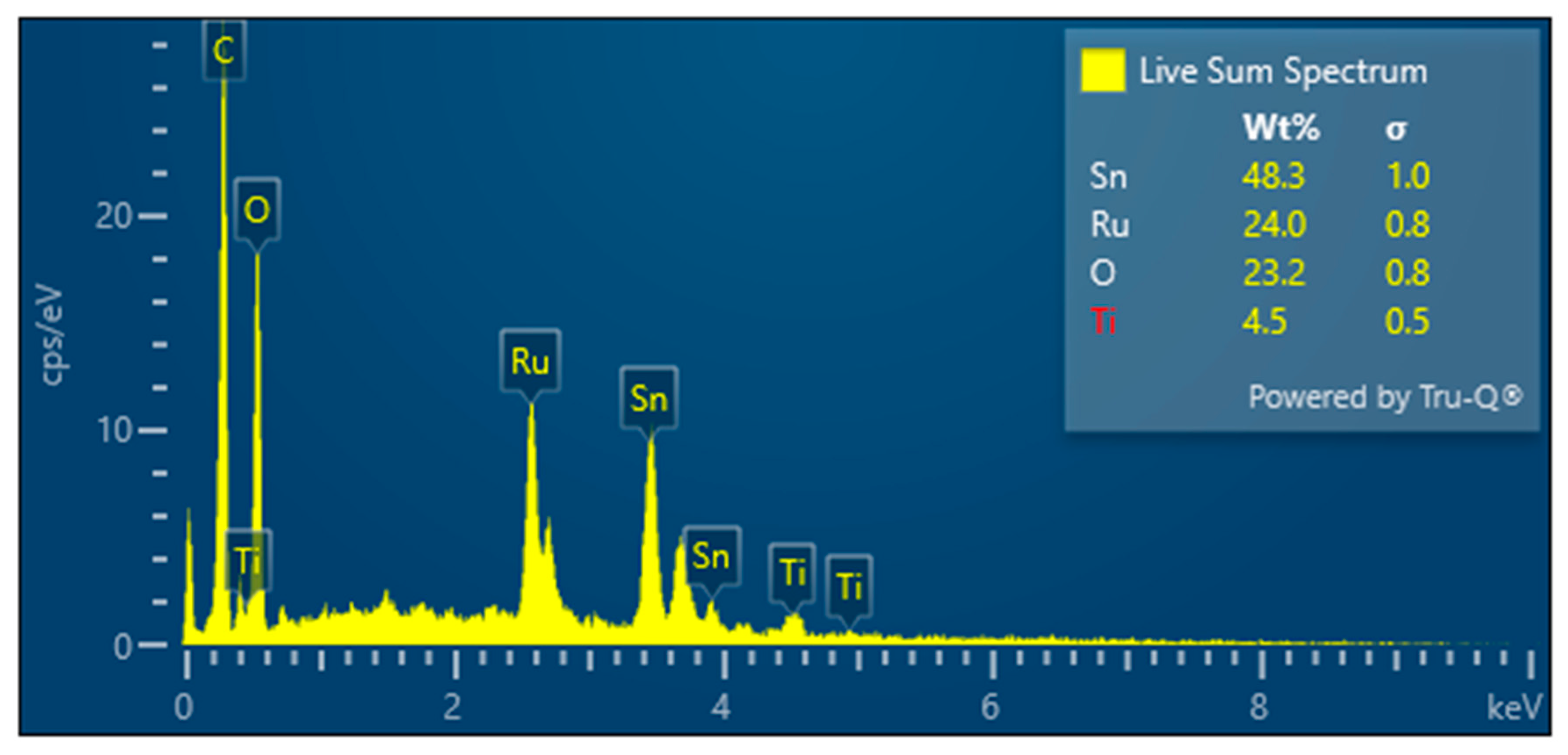
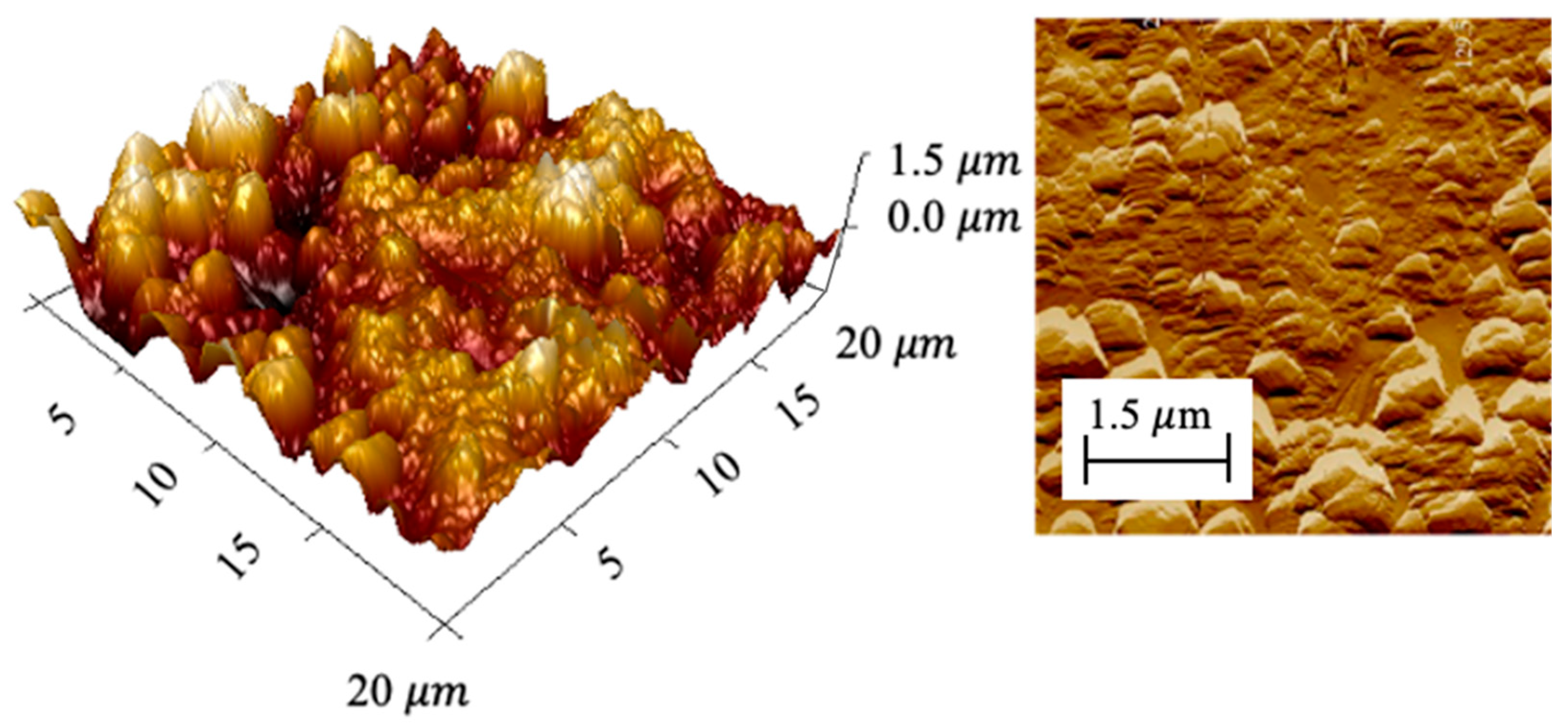









| Electrode | Ethylene Glycol | Citric Acid | Ru | Sn | Sb |
|---|---|---|---|---|---|
| Electrode | 3.2 mM | 0.024 mM | 0.041 mM | 0.041 mM | 0.0010 mM |
| Dye | Peak Absorbance (nm) |
|---|---|
| Brown KK | 410.019 |
| Brown NT | 449.272 |
| Brown 5VR | 378.445 |
| Brown mixture | 414.64 |
| Scan Rate (mV s−1) | Ipa (mA) | Ipc (mA) | |Ipc|/Ipa (mA) | Epa (V) | Epc (V) | Epa − Epc (V) |
|---|---|---|---|---|---|---|
| 20 | 79 | −33 | 0.41 | 0.58 | 0.09 | 0.49 |
| 50 | 103 | −58 | 0.56 | 0.65 | 0.03 | 0.62 |
| 75 | 126 | −80 | 0.63 | 0.60 | 0.03 | 0.57 |
| 100 | 138 | −92 | 0.66 | 0.56 | 0.05 | 0.51 |
| 125 | 145 | −101 | 0.69 | 0.66 | 0.03 | 0.63 |
| 150 | 178 | −138 | 0.77 | 0.57 | 0.03 | 0.54 |
| Number | Vibrational Assignment | Brown 5VR (cm−1) | Brown NT (cm−1) | Brown KK (cm−1) |
|---|---|---|---|---|
| 1 | νas(SO3−)/ν(C–O) | 1042–1105 | 1033–1112 | 1036–1181 |
| 2 | δ(N–H) aromatic amine | 1323 | 1316 | 1332 |
| 3 | ν(N=N) azo group | 1491 | 1589 | 1549 |
| 4 | ν(C=C) aromatic ring | 1598 | — | 1457 |
| 5 | ν(C–H) aliphatic | — | 2921 | 2920 |
| 6 | ν(O–H)/ν(N–H) stretching | 3415 | 3443 | 3450 |
| Anode Material | Process | Dye | Current Density (mA cm−2) | % Removal | Ref. |
|---|---|---|---|---|---|
| Ti/IrO2–SnO2–Sb2O5 | EOx | Brown DR (azo dye) | 50 | ~6 min (color >86%) | [51] |
| Ti/IrO2–SnO2–Sb2O5 | EOx | Mixed azo dyes | 50 | ~20 min (color), ~60 min (COD) | [52] |
| Ti/IrO2–SnO2–Sb2O5 | EOx | Mixed azo dyes | 50 | ~92% COD removal (60 min) | [53] |
| Ti/IrO2–SnO2–Sb2O5 | EOx/EF/PEF | Reactive Orange 84 | 25–100 | ~91% COD removal (60 min) | [54] |
| RuO2–SnO2–Sb2O5/Ti (this work) | EOx | Brown NT | 20–50 | ~95% COD removal (20 min) | This work |
| RuO2–SnO2–Sb2O5/Ti (this work) | EOx | Brown KK | 20–50 | ~99% COD removal (6 min) | This work |
| RuO2–SnO2–Sb2O5/Ti (this work) | EOx | Brown 5VR | 20–50 | ~97% COD removal (60 min) | This work |
| RuO2–SnO2–Sb2O5/Ti (this work) | EOx | Mixed dye brown | 20–50 | ~99% COD removal (60 min) | This work |
| Dye/System | Process | C0 (mg L−1) | j (mA cm−2) | kdis (min−1) | R2 | Regime |
|---|---|---|---|---|---|---|
| Brown NT | EOx | 100 | 50 | 0.45 ± 0.02 | 0.992 | Kinetic |
| Brown NT | EOx | 200 | 50 | 0.29 ± 0.01 | 0.987 | Mixed/MT |
| Brown NT | EOx | 100 | 35 | 0.31 ± 0.02 * | 0.985 | Kinetic |
| Brown KK | EF | 100 | 50 | 1.48 ± 0.08 | 0.995 | Kinetic |
| Brown 5VR | EF | 100 | 50 | 0.084 ± 0.005 | 0.981 | Mixed |
| Mixture | PEF | 100 | 50 | 0.14 ± 0.01 | 0.989 | Kinetic–Transport |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Partida-Joya, D.K.; Ornelas-Soto, N.; Medina-Ramírez, I.E.; Rodríguez, O.; Feria-Reyes, R.; Peralta-Hernández, J.M. Metal Oxide Electrode-Based Treatment of Industrial Dyes with Assessment of Performance and Oxidation Efficiency. Processes 2026, 14, 987. https://doi.org/10.3390/pr14060987
Partida-Joya DK, Ornelas-Soto N, Medina-Ramírez IE, Rodríguez O, Feria-Reyes R, Peralta-Hernández JM. Metal Oxide Electrode-Based Treatment of Industrial Dyes with Assessment of Performance and Oxidation Efficiency. Processes. 2026; 14(6):987. https://doi.org/10.3390/pr14060987
Chicago/Turabian StylePartida-Joya, D. Kiabeth, Nancy Ornelas-Soto, Iliana E. Medina-Ramírez, Oscar Rodríguez, Rossy Feria-Reyes, and Juan M. Peralta-Hernández. 2026. "Metal Oxide Electrode-Based Treatment of Industrial Dyes with Assessment of Performance and Oxidation Efficiency" Processes 14, no. 6: 987. https://doi.org/10.3390/pr14060987
APA StylePartida-Joya, D. K., Ornelas-Soto, N., Medina-Ramírez, I. E., Rodríguez, O., Feria-Reyes, R., & Peralta-Hernández, J. M. (2026). Metal Oxide Electrode-Based Treatment of Industrial Dyes with Assessment of Performance and Oxidation Efficiency. Processes, 14(6), 987. https://doi.org/10.3390/pr14060987









