Physical State and Mass Transport of Metals in Liquid Cadmium Cathodes: A Review
Abstract
1. Introduction
2. Mass Transfer Processes and Mechanisms of Metals at Liquid Cd Cathodes
- (1)
- Interfacial Electrochemical Reduction: Metal ions dissolved in the molten salt electrolyte gain electrons at the cathode/electrolyte interface and are electrochemically reduced to neutral metal atoms.
- (2)
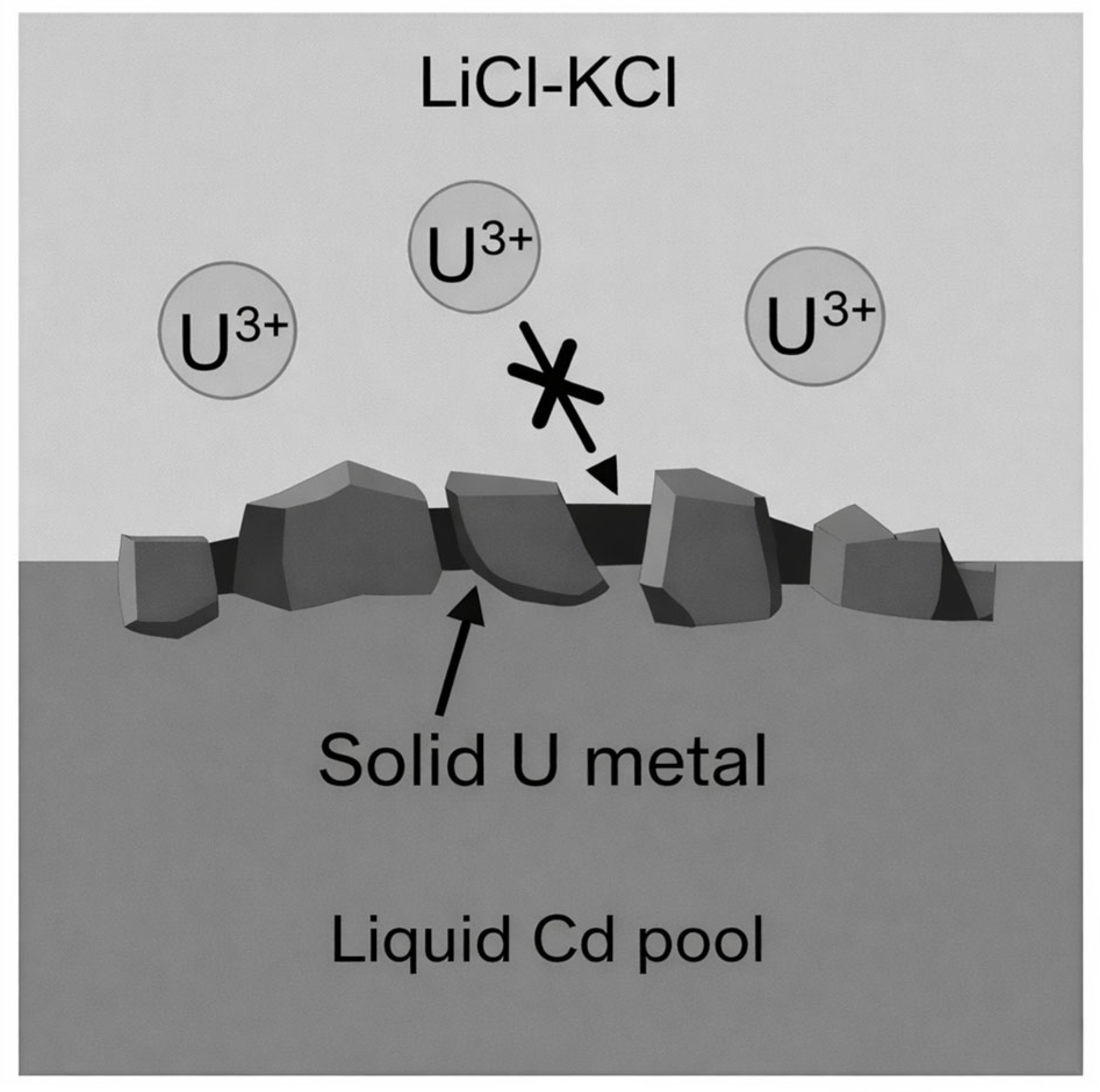
- Dissolution or Intermetallic Compound Formation: The newly formed metal atoms subsequently cross the interface and dissolve into the liquid cadmium phase, resulting in an increase in local concentration near the interface. Under certain conditions, nuclei may form and grow on the cathode surface, potentially resulting in the formation of a solid layer composed of intermetallic compounds or elemental metal.
- (3)
- Bulk diffusion and growth of deposited phases: The concentration gradient between the interface and the bulk liquid cathode serves as the primary driving force for mass transfer. Metal atoms migrate into the cathode interior through liquid-phase diffusion. Although natural or forced convection may influence macroscopic mass transfer, atomic diffusion generally remains the rate-controlling step. As the metal concentration increases and approaches the saturation solubility, intermetallic compounds may form with Cd, or the metal may precipitate as a solid phase. In some cases, a solid deposit layer may develop on the electrode surface, which can hinder further mass transport.
2.1. Dissolution and Precipitation Mechanisms of Metals in Liquid Cd
| Element | Category | Solubility in Liquid Cd (at%) | Major Intermetallic Compounds | References |
|---|---|---|---|---|
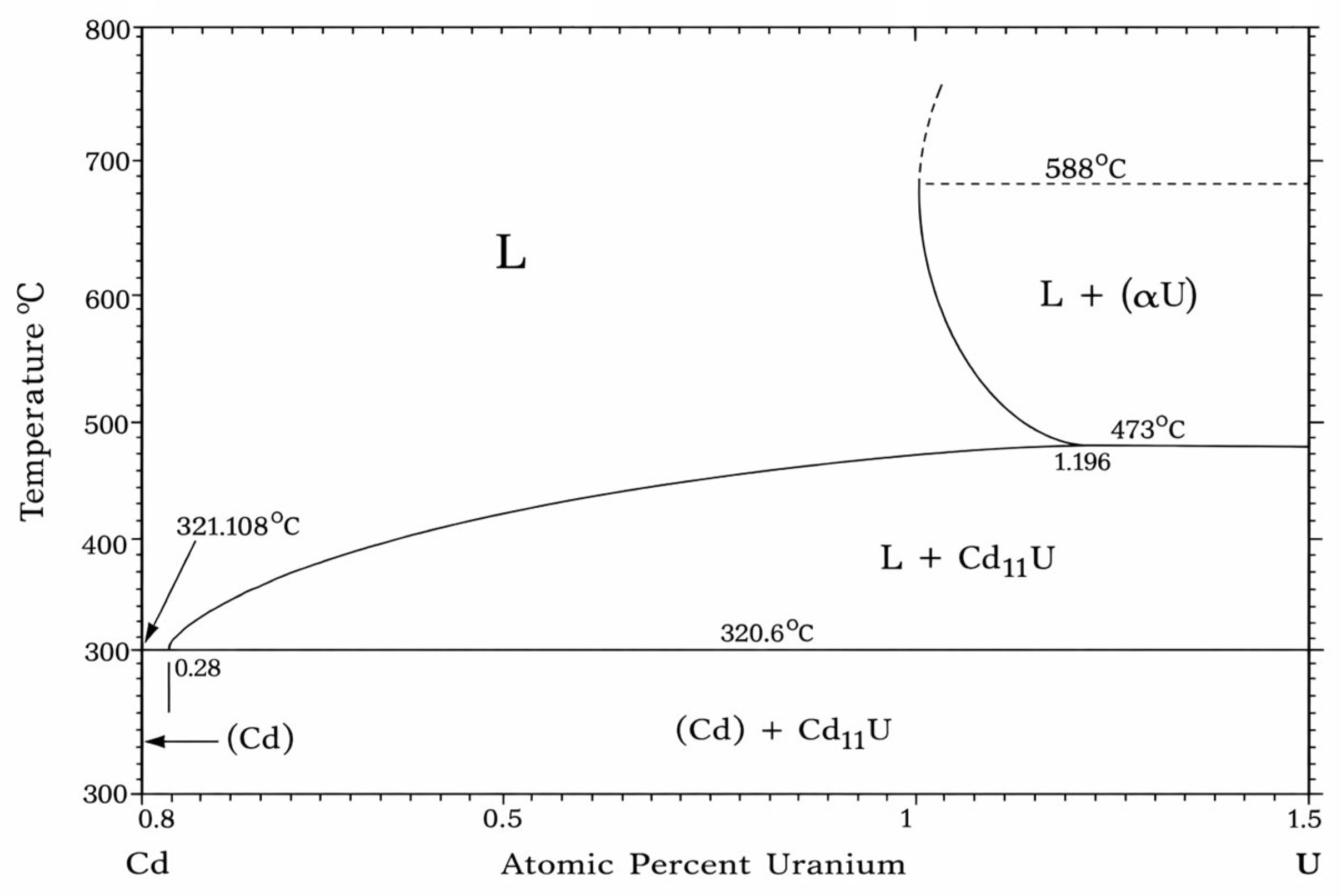
| U | Actinide | 1.11 | UCd11 | [16,36] |
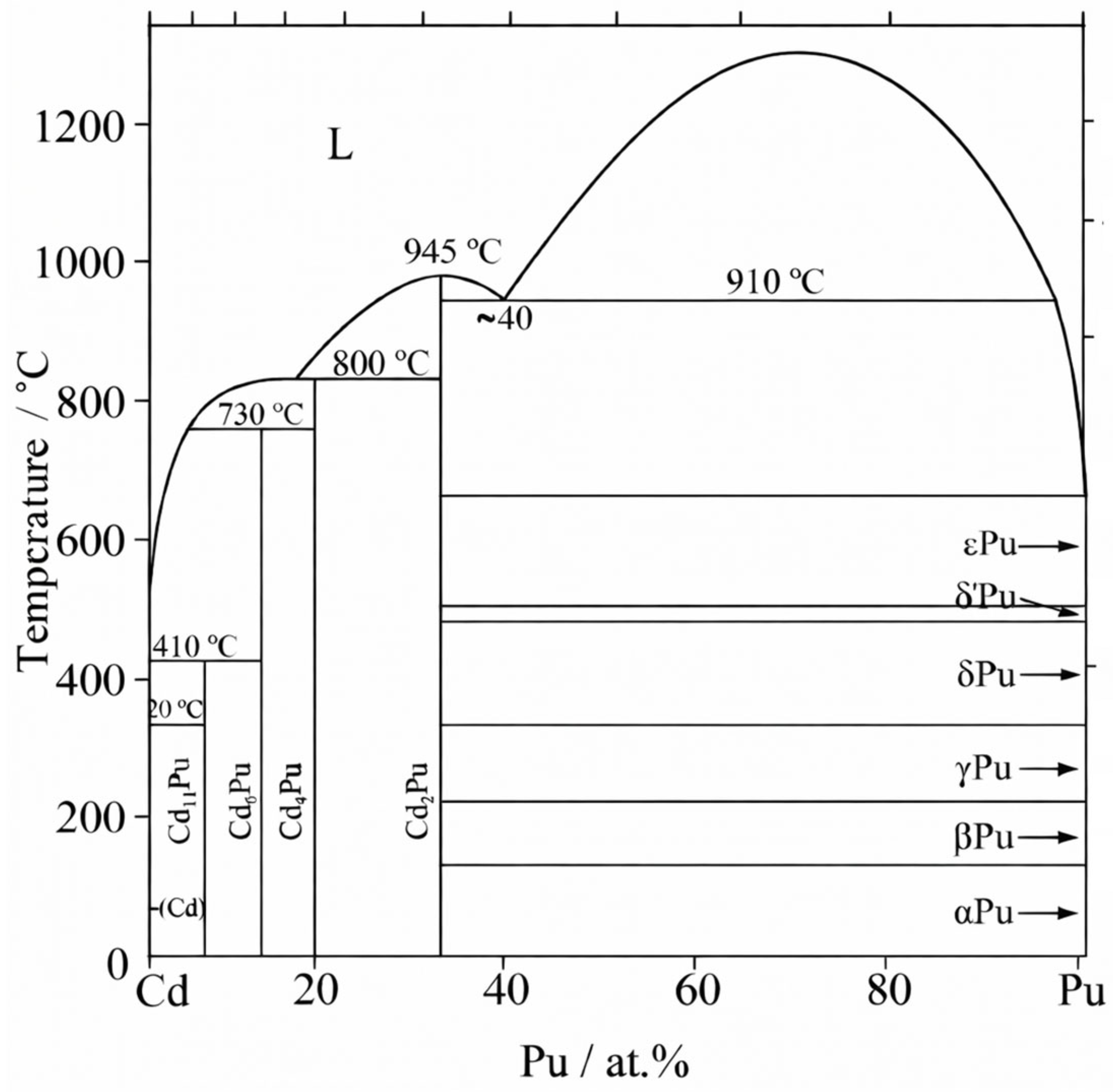
| Pu | Actinide | 1.86 | PuCd11, PuCd6; | [37,38] |
| Np | Actinide | 2.20 | NpCd11, NpCd6 | [39] |
| Am | Actinide | 2.00 | AmCd6 | [40,41] |
| La | Lanthanide | 0.32 | LaCd11 | [42] |
| Ce | Lanthanide | 0.60 | CeCd11, CeCd6 | [42] |
| Nd | Lanthanide | 1.46 | NdCd11, NdCd6 | [43] |
| Sm | Lanthanide | 2.23 | SmCd11, SmCd6 | [44] |
| Zn | Post-transition | Forms a continuous liquid alloy | No fixed-stoichiometry intermetallic compounds | [45] |
| In | Post-transition | Forms a continuous liquid alloy | Described by liquid/solid solution phases | [46] |
2.1.1. Alloy Systems Exhibiting Intermetallic Compound Formation or Base-Metal Precipitation at Saturation
2.1.2. Highly Miscible Liquid Alloy Systems
2.1.3. Immiscible Systems
2.2. Mass Transport Behavior of Metals in Liquid Cd
2.3. Multicomponent Diffusion in Liquid Cd
3. Experimental Study on Mass Transport in Liquid Cd Cathode
3.1. Capillary Method
3.2. Electrochemical Methods
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kim, H.; Boysen, D.A.; Newhouse, J.M.; Spatocco, B.L.; Chung, B.; Burke, P.J.; Bradwell, D.J.; Jiang, K.; Tomaszowska, A.A.; Wang, K. Liquid metal batteries: Past, present, and future. Chem. Rev. 2013, 113, 2075–2099. [Google Scholar] [PubMed]
- Li, H.; Yin, H.; Wang, K.; Cheng, S.; Jiang, K.; Sadoway, D.R. Liquid metal electrodes for energy storage batteries. Adv. Energy Mater. 2016, 6, 1600483. [Google Scholar] [CrossRef]
- Lin, R.; He, H.; Tang, H.; Ye, G. Progress and development of dry reprocessing technology of spent fuel in China. At. Energy Sci. Technol. 2020, 54, 115–125. [Google Scholar]
- Wang, J.; Wu, P.; Zhou, W.; Chen, Y.; Hou, L.; Ma, J. Research progress in pyrochemical reprocessing of spent nuclear fuel. Transit. Met. Chem. 2025, 50, 813–827. [Google Scholar] [CrossRef]
- Schmieder, H.; Galla, U. Electrochemical processes for nuclear fuel reprocessing. J. Appl. Electrochem. 2000, 30, 201–207. [Google Scholar] [CrossRef]
- Paviet-Hartmann, P.; Benedict, B.; Lineberry, M.J. Nuclear fuel reprocessing. In Nuclear Engineering Handbook; CRC Press: Boca Raton, FL, USA, 2009; pp. 333–384. [Google Scholar]
- Fredrickson, G.L.; Yoo, T.-S. Nuclear Fuels and Reprocessing Technologies: A US Perspective; U.S. Department of Energy Office of Scientific and Technical Information: Oak Ridge, TN, USA, 2021. [Google Scholar]
- Natarajan, R. Reprocessing of spent nuclear fuel in India: Present challenges and future programme. Prog. Nucl. Energy 2017, 101, 118–132. [Google Scholar] [CrossRef]
- Mirza, M.; Abdulaziz, R.; Maskell, W.C.; Wilcock, S.; Jones, A.H.; Woodall, S.; Jackson, A.; Shearing, P.R.; Brett, D.J. Electrochemical processing in molten salts—A nuclear perspective. Energy Environ. Sci. 2023, 16, 952–982. [Google Scholar]
- Fredrickson, G.L.; Yoo, T.-S. Engineering Scale Pyroprocessing Activities in the United States; Idaho National Laboratory (INL): Idaho Falls, ID, USA, 2023. [Google Scholar]
- Vaden, D.; Li, S.; Westphal, B.; Davies, K.; Johnson, T.; Pace, D. Engineering-scale liquid cadmium cathode experiments. Nucl. Technol. 2008, 162, 124–128. [Google Scholar] [CrossRef]
- Solbrig, C.; Westphal, B.; Johnson, T.; Li, S.; Marsden, K.; Goff, K. Pyroprocessing Progress at Idaho National Laboratory; Idaho National Laboratory (INL): Idaho Falls, ID, USA, 2007. [Google Scholar]
- Koyama, T.; Iizuka, M.; Shoji, Y.; Fujita, R.; Tanaka, H.; Kobayashi, T.; Tokiwai, M. An experimental study of molten salt electrorefining of uranium using solid iron cathode and liquid cadmium cathode for development of pyrometallurgical reprocessing. J. Nucl. Sci. Technol. 1997, 34, 384–393. [Google Scholar] [CrossRef]
- Kato, T.; Inoue, T.; Iwai, T.; Arai, Y. Separation behaviors of actinides from rare-earths in molten salt electrorefining using saturated liquid cadmium cathode. J. Nucl. Mater. 2006, 357, 105–114. [Google Scholar] [CrossRef]
- Iizuka, M.; Uozumi, K.; Inoue, T.; Iwai, T.; Shirai, O.; Arai, Y. Behavior of plutonium and americium at liquid cadmium cathode in molten LiCl–KCl electrolyte. J. Nucl. Mater. 2001, 299, 32–42. [Google Scholar]
- Uozumi, K.; Iizuka, M.; Kato, T.; Inoue, T.; Shirai, O.; Iwai, T.; Arai, Y. Electrochemical behaviors of uranium and plutonium at simultaneous recoveries into liquid cadmium cathodes. J. Nucl. Mater. 2004, 325, 34–43. [Google Scholar] [CrossRef]
- Kwon, S.; Kim, J.; Ahn, D.; Lee, H.; Ahn, H. A study on the evaporation of cadmium for the recovery of actinides from a liquid cathode. J. Radioanal. Nucl. Chem. 2010, 284, 143–149. [Google Scholar] [CrossRef]
- Wu, B.; Zhou, X.; Ding, X.; Çubuk, A.B.; Wang, Y.; Guo, S.; Lin, P. Electrochemical Drawdown of U3+ and Ce3+ in Molten LiCl-KCl Using a Liquid Cadmium Cathode. J. Electrochem. Soc. 2024, 171, 112504. [Google Scholar]
- Cussler, E.L. Diffusion: Mass Transfer in Fluid Systems; Cambridge University Press: Cambridge, UK, 2009. [Google Scholar]
- Koter, S.; Szczepański, P.; Mateescu, M.; Nechifor, G.; Badalau, L.; Koter, I. Modeling of the cadmium transport through a bulk liquid membrane. Sep. Purif. Technol. 2013, 107, 135–143. [Google Scholar] [CrossRef]
- Protopapas, P.; Andersen, H.C.; Parlee, N. Theory of transport in liquid metals. I. Calculation of self-diffusion coefficients. J. Chem. Phys. 1973, 59, 15–25. [Google Scholar] [CrossRef]
- Vrentas, J.S.; Vrentas, C.M. Diffusion and Mass Transfer; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Quattrini, F.; Galceran, J.; David, C.; Puy, J.; Alberti, G.; Rey-Castro, C. Dynamics of trace metal sorption by an ion-exchange chelating resin described by a mixed intraparticle/film diffusion transport model. The Cd/Chelex case. Chem. Eng. J. 2017, 317, 810–820. [Google Scholar] [CrossRef]
- Larsen, R. Dissolution of uranium metal and its alloys. Anal. Chem. 1959, 31, 545–549. [Google Scholar] [CrossRef]
- Ye, Y.; Fan, B.; Qin, Z.; Tang, X.; Feng, Y.; Lv, M.; Miao, S.; Li, H.; Chen, Y.; Chen, F. Electrochemical removal and recovery of uranium: Effects of operation conditions, mechanisms, and implications. J. Hazard. Mater. 2022, 432, 128723. [Google Scholar] [CrossRef]
- Hege, N.; Jackson, J.; Shafer, J. Fundamental uranium electrochemistry and spectroscopy in molten salt systems. J. Electrochem. Soc. 2023, 170, 016503. [Google Scholar] [CrossRef]
- Kindlimann, L.; Greene, N. Dissolution Kinetics of Nuclear Fuels 1. Uranium. Corrosion 1967, 23, 29–38. [Google Scholar] [CrossRef]
- Qin, A.; Wang, R.; Wang, Y.; Wang, J.; Liu, S.; Cheng, K.; Du, Y.; Sundman, B. Thermodynamic assessment of the Cd–X (X = Sn, Mn, Fe) systems. Calphad 2014, 47, 83–91. [Google Scholar] [CrossRef]
- Halimi, A.; Ferah, M. Thermodynamic description of the systems Cd-Te, Hg-Te and Cd-Hg-Te using the model of associated liquid solution. Int. J. Microstruct. Mater. Prop. 2008, 3, 77–85. [Google Scholar]
- Gokcen, N.A. Statistical Thermodynamics of Alloys; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Arzpeyma, G. Application of Engel-Brewer and Miedema Semi-Empirical Models Combined with Entropy Models to Thermodynamic Modeling of Binary Systems. Master’s Thesis, Concordia University, Montreal, QC, Canada, 2011. [Google Scholar]
- Adhikari, D.; Singh, B.; Jha, I. Energetics of Cd-based binary liquid alloys. J. Non-Cryst. Solids 2012, 358, 1362–1367. [Google Scholar]
- Erdem, M.; Tümen, F. A study on dissolution properties of the sludges from Cr (VI) reduction–precipitation processes. J. Environ. Sci. Health Part A 2004, 39, 253–267. [Google Scholar]
- Sulonen, M. Discontinuous mode of dissolution of a β phase precipitate into α Cu-Cd solid solutions. Acta Metall. 1960, 8, 669–676. [Google Scholar] [CrossRef]
- Samitsu, S.; Araki, J.; Kataoka, T.; Ito, K. New solvent for polyrotaxane. II. Dissolution behavior of polyrotaxane in ionic liquids and preparation of ionic liquid-containing slide-ring gels. J. Polym. Sci. Part B Polym. Phys. 2006, 44, 1985–1994. [Google Scholar] [CrossRef]
- Ahluwalia, R.K.; Hua, T.Q. Electrotransport of uranium from a liquid cadmium anode to a solid cathode. Nucl. Technol. 2002, 140, 41–50. [Google Scholar] [CrossRef]
- Kurata, M.; Sakamura, Y. Thermodynamic assessment of systems of actinide or rare earth with Cd. J. Phase Equilibria 2001, 22, 232–240. [Google Scholar] [CrossRef]
- Kato, T.; Iizuka, M.; Inoue, T.; Iwai, T.; Arai, Y. Distillation of cadmium from uranium–plutonium–cadmium alloy. J. Nucl. Mater. 2005, 340, 259–265. [Google Scholar]
- Shirai, O.; Uozumi, K.; Iwai, T.; Arai, Y. Electrode reaction of the Np3+/Np couple at liquid Cd and Bi electrodes in LiCl–KCl eutectic melts. J. Appl. Electrochem. 2004, 34, 323–330. [Google Scholar] [CrossRef]
- Sakamura, Y.; Shirai, O.; Iwai, T.; Suzuki, Y. Distribution behavior of plutonium and americium in LiCl–KCl eutectic/liquid cadmium systems. J. Alloys Compd. 2001, 321, 76–83. [Google Scholar]
- Zhang, J.; Lahti, E.A.; Zhou, W. Thermodynamic properties of actinides and rare earth fission products in liquid cadmium. J. Radioanal. Nucl. Chem. 2015, 303, 1637–1648. [Google Scholar] [CrossRef]
- Sakata, M.; Kurata, M.; Hijikata, T.; Inoue, T. Equilibrium distribution of rare earth elements between molten KCl-LiCl eutectic salt and liquid cadmium. J. Nucl. Mater. 1991, 185, 56–65. [Google Scholar]
- Vandarkuzhali, S.; Chandra, M.; Ghosh, S.; Samanta, N.; Nedumaran, S.; Reddy, B.P.; Nagarajan, K. Investigation on the electrochemical behavior of neodymium chloride at W, Al and Cd electrodes in molten LiCl-KCl eutectic. Electrochim. Acta 2014, 145, 86–98. [Google Scholar]
- Skołyszewska-Kühberger, B.; Reichmann, T.L.; Ipser, H. Phase equilibria in the neodymium–cadmium binary system. J. Alloys Compd. 2014, 606, 242–248. [Google Scholar][Green Version]
- Soon-Don, C. A thermodynamic analysis of the Cd-Zn system and calculation of the phase diagram. Calphad 1990, 14, 307–310. [Google Scholar] [CrossRef]
- Dutkiewicz, J.; Zabdyr, L.; Zakulski, W.; Moser, Z.; Salawa, J.; Horrocks, P.; Hayes, F.; Rand, M.H. Cd-In (cadmium-indium). J. Phase Equilibria 1992, 13, 261–269. [Google Scholar] [CrossRef]
- Christian, J. The Theory of Transformations in Metals and Alloys; Pergamon Press: Oxford, UK, 2002. [Google Scholar]
- Mehrer, H. Diffusion in Solids: Fundamentals, Methods, Materials, Diffusion-Controlled Processes; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Turek, I.; Drchal, V.; Kudrnovský, J.; Sob, M.; Weinberger, P. Electronic Structure of Disordered Alloys, Surfaces and Interfaces; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Martin, R.M. Electronic Structure: Basic Theory and Practical Methods; Cambridge University Press: Cambridge, UK, 2020. [Google Scholar]
- Ferro, R.; Saccone, A. Intermetallic Chemistry; Elsevier: Amsterdam, The Netherlands, 2008; Volume 13. [Google Scholar]
- Mishra, R.; Dawar, R. Synthesis, Properties and Applications of Intermetallic Phases. In Handbook on Synthesis Strategies for Advanced Materials: Volume-III: Materials Specific Synthesis Strategies; Springer: Berlin/Heidelberg, Germany, 2021; pp. 741–784. [Google Scholar]
- Ikeuba, A.I.; Njoku, C.N.; Ekerenam, O.O.; Njoku, D.I.; Udoh, I.I.; Daniel, E.F.; Uzoma, P.C.; Etim, I.-I.N.; Okonkwo, B.O. A review of the electrochemical and galvanic corrosion behavior of important intermetallic compounds in the context of aluminum alloys. RSC Adv. 2024, 14, 31921–31953. [Google Scholar] [CrossRef]
- Martin, J.W.; Doherty, R.D.; Cantor, B. Stability of Microstructure in Metallic Systems; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Faizan, M. Experimental Study and Modeling of Metal Dissolution and Intermetallic Compound Growth During Soldering. Ph.D. Thesis, University of Akron, Akron, OH, USA, 2007. [Google Scholar]
- Ghosh, S.; Ganesan, R.; Sridharan, R.; Gnanasekaran, T. Measurement of chemical activities of rare earths (RE: Ce, Pr, Sm and Eu) in cadmium alloy. J. Nucl. Mater. 2015, 467, 280–285. [Google Scholar] [CrossRef]
- Liu, J.; Huang, X.; Du, Q.; Ouyang, L.; Xiao, J.; Li, Y. Critical systematic investigation of the Cd–Ce system: Phase stability and Gibbs energies of formation and equilibria via thermodynamic description. Int. J. Mater. Res. 2023, 114, 1101–1110. [Google Scholar] [CrossRef]
- Gschneidner, K.; Calderwood, F. The Cd− Nd (Cadmium-Neodymium) system. J. Phase Equilibria 1988, 9, 128–130. [Google Scholar] [CrossRef]
- Qureshi, A.M.; Naz, S.; Athar, M.; Ali, M.; Rauf, A.; Sharif, A.; Ahmed, E.; Ashiq, M.N. Manganese doped Sm–Cd nanoalloys–their synthesis, characterisation and evaluation of electrical properties. J. Alloys Compd. 2016, 662, 593–597. [Google Scholar] [CrossRef]
- Kurata, M.; Uozumi, K.; Kato, T.; Iizuka, M. Thermodynamic evaluation of liquid Cd cathode containing U and Pu. J. Nucl. Sci. Technol. 2009, 46, 1070–1075. [Google Scholar] [CrossRef]
- Roy, J.; Grantham, L.; Grimmett, D.; Fusselman, S.; Krueger, C.; Storvick, T.; Inoue, T.; Sakamura, Y.; Takahashi, N. Thermodynamic properties of U, Np, Pu, and Am in molten LiCl-KCl eutectic and liquid cadmium. J. Electrochem. Soc. 1996, 143, 2487. [Google Scholar] [CrossRef]
- Fredrickson, G.L.; Yoo, T.-S. Liquid cadmium cathode performance model based on the equilibrium behaviors of U and Pu in molten LiCl–KCl/Cd system at 500 °C. J. Nucl. Mater. 2020, 528, 151883. [Google Scholar] [CrossRef]
- Iizuka, M.; Uozumi, K.; Inoue, T.; Iwai, T.; Shirai, O.; Arai, Y. Development of plutonium recovery process by molten salt electrorefining with liquid cadmium cathode. In Proceedings of the 6th Information Exchange Meeting on Actinide and Fission Product P&T, Madrid, Spain, 11–13 December 2000. [Google Scholar]
- Huang, Y.; Lu, Y.; He, Q.; Wang, Y.; Fang, W.; Liu, X.; Jia, J.; Wang, C. Thermodynamic assessment of the Be–Pu and Cd–Pu systems. Calphad 2016, 52, 120–124. [Google Scholar] [CrossRef]
- Paek, S.; Kim, S.-H.; Yoon, D.-S.; Lee, H.; Ahn, D.-H. Performance of the mesh-type liquid cadmium cathode structure for the electrodeposition of uranium from the molten salt. Radiochim. Acta 2010, 98, 779–783. [Google Scholar] [CrossRef]
- Battezzati, L.; Greer, A.L. The viscosity of liquid metals and alloys. Acta Metall. 1989, 37, 1791–1802. [Google Scholar] [CrossRef]
- Koirala, R.; Singh, B.; Jha, I.; Adhikari, D. Thermodynamic, structural and surface properties of liquid Cd–Zn alloys. J. Mol. Liq. 2013, 179, 60–66. [Google Scholar] [CrossRef]
- Moser, Z.; Fecht, H. Heat Capacity Measurements of Liquid Cd-Ga Alloys. Int. J. Mater. Res. 1986, 77, 377–379. [Google Scholar] [CrossRef]
- Chauhan, P.; Sharma, R.; Nehra, S.; Sharma, H.S.; Kumar, D. Advanced Materials for nickel-cadmium batteries. In Advanced Materials for Batteries; CRC Press: Boca Raton, FL, USA, 2024; pp. 317–335. [Google Scholar]
- Azza, H.; Selhaoui, N.; Bouirden, L. Thermodynamic Description of the Cadmium-Nickel System. IJARPS 2018, 5, 17–23. [Google Scholar]
- Han, J.-H.; Oda, T. Chemical states of 3d transition metal impurities in a liquid lead–bismuth eutectic analyzed using first principles calculations. Phys. Chem. Chem. Phys. 2017, 19, 9945–9956. [Google Scholar] [CrossRef]
- Daeneke, T.; Khoshmanesh, K.; Mahmood, N.; De Castro, I.A.; Esrafilzadeh, D.; Barrow, S.J.; Dickey, M.D.; Kalantar-Zadeh, K. Liquid metals: Fundamentals and applications in chemistry. Chem. Soc. Rev. 2018, 47, 4073–4111. [Google Scholar] [CrossRef]
- Kleppa, O. Thermodynamic analysis of binary liquid alloys of group II B metals—II The alloys of cadmium with gallium, indium, tin, thallium, lead and bismuth. Acta Metall. 1958, 6, 233–242. [Google Scholar] [CrossRef]
- Johnson, I.; Yonco, R.M. Thermodynamics of cadmium-and zinc-rich alloys in the Cd−La, Cd−Ce, Cd−Pr, Zn−La, Zn−Ce and Zn−Pr systems. Metall. Trans. 1970, 1, 905–910. [Google Scholar] [CrossRef]
- Hafner, J. Structure and thermodynamics of liquid metals and alloys. Phys. Rev. A 1977, 16, 351. [Google Scholar] [CrossRef]
- Chasanov, M.; Hunt, P.; Johnson, I.; Feder, H. Solubility of 3-d transition metals in liquid cadmium. Trans. Met. Soc. AIME 1962, 22, 4784949. [Google Scholar]
- Kerridge, D. The solubility of metals in liquid metals. J. Nucl. Energy Part B React. Technol. 1961, 1, 215–220. [Google Scholar] [CrossRef]
- Crawley, A. Densities and viscosities of some liquid alloys of zinc and cadmium. Metall. Trans. 1972, 3, 971–975. [Google Scholar] [CrossRef]
- Shunk, F.; Nash, P. The Cd− Ni (Cadmium-Nickel) system. J. Phase Equilibria 1987, 8, 122–124. [Google Scholar] [CrossRef]
- Pugachev, V.; Zakharov, Y.A.; Valnyukova, A.; Popova, A.; Khitsova, L.; Yashnik, S.; Ismagilov, Z. Phase transformations in Cd–Ni nanostructured system at elevated temperatures. Russ. Chem. Bull. 2019, 68, 17–23. [Google Scholar] [CrossRef]
- Jia, Y.; Chang, S.; Du, X.; Guo, S. Corrosion performance of commercial alloys and refractory metals in conditions for electrorefining of spent nuclear fuels. Crystals 2023, 13, 817. [Google Scholar] [CrossRef]
- Personnettaz, P. Simulations of Mass Transport in Liquid Metal Electrodes. Ph.D. Thesis, Technische Universität Dresden, Dresden, Germany, 2022. [Google Scholar]
- Monchoux, J.-P. Mass transport driving forces under electric current in the liquid Sn-Zn system. Scr. Mater. 2025, 255, 116349. [Google Scholar]
- Kumar, S.; Kumar, P.; Pratap, R. A model for electromigration induced flow in liquid metals. J. Phys. D Appl. Phys. 2017, 50, 39LT02. [Google Scholar]
- Shaw, R.E.; Verhoeven, J. Convection effects during electrotransport of liquid metals. Metall. Trans. 1973, 4, 2349–2355. [Google Scholar] [CrossRef][Green Version]
- Fick, A. On liquid diffusion. J. Membr. Sci. 1995, 100, 33–38. [Google Scholar] [CrossRef]
- Serin, B.; Ellickson, R. Determination of diffusion coefficients. J. Chem. Phys. 1941, 9, 742–747. [Google Scholar] [CrossRef]
- Guenneau, S.; Puvirajesinghe, T. Fick’s second law transformed: One path to cloaking in mass diffusion. J. R. Soc. Interface 2013, 10, 20130106. [Google Scholar] [CrossRef]
- Darken, L.S. Diffusion, mobility and their interrelation through free energy in binary metallic systems. Trans. AIME 1948, 175, 184–201. [Google Scholar]
- Huang, Y.; Chen, H.; Li, D.; Zhou, R.; Zhang, B. The relationship between inter-diffusion and self-diffusion of different liquid metals studied by molecular dynamics simulations. Phys. B Condens. Matter 2022, 638, 413844. [Google Scholar] [CrossRef]
- Krishna, R.; Wesselingh, J.A. The Maxwell-Stefan approach to mass transfer. Chem. Eng. Sci. 1997, 52, 861–911. [Google Scholar] [CrossRef]
- Van den Broeke, L.; Krishna, R. Experimental verification of the Maxwell-Stefan theory for micropore diffusion. Chem. Eng. Sci. 1995, 50, 2507–2522. [Google Scholar] [CrossRef]
- Rudd, R.E.; Cabot, W.H.; Caspersen, K.J.; Greenough, J.A.; Richards, D.F.; Streitz, F.H.; Miller, P.L. Self-diffusivity and interdiffusivity of molten aluminum-copper alloys under pressure, derived from molecular dynamics. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2012, 85, 031202. [Google Scholar] [CrossRef] [PubMed]
- Li, S.X.; Herrmann, S.D.; Simpson, M.F. Experimental Investigations into U/TRU Recovery Using a Liquid Cadmium Cathode and Salt Containing High Rare Earth Concentrations; Idaho National Laboratory (INL): Idaho Falls, ID, USA, 2009. [Google Scholar]
- Jung, C.-Y.; Jeong, H.; Yeon, J.-W.; Bae, S.-E. Direct electrochemical separation of uranium and lanthanides (Nd, Ce, La) from LiCl-KCl molten salt by bismuth and cadmium liquid metal electrodes. Sep. Purif. Technol. 2025, 380, 135382. [Google Scholar] [CrossRef]
- Ju, Y.-Y.; Zhang, Q.-M.; Gong, Z.-Z.; Ji, G.-F. Molecular dynamics simulation of self-diffusion coefficients for liquid metals. Chin. Phys. B 2013, 22, 083101. [Google Scholar] [CrossRef]
- Lü, Y.; Cheng, H.; Chen, M. A molecular dynamics examination of the relationship between self-diffusion and viscosity in liquid metals. J. Chem. Phys. 2012, 136, 214505. [Google Scholar] [CrossRef]
- Gao, Y.; Takahashi, M.; Cavallotti, C.; Raos, G. Molecular dynamics simulation of metallic impurity diffusion in liquid lead-bismuth eutectic (LBE). J. Nucl. Mater. 2018, 501, 253–260. [Google Scholar] [CrossRef]
- Shiinoki, M.; Hirata, A.; Suzuki, S. Molecular Dynamics Simulation of Diffusion Behavior in Liquid Sn and Pb. Metall. Mater. Trans. B 2024, 55, 278–286. [Google Scholar] [CrossRef]
- Shiinoki, M.; Hashimoto, N.; Fukuda, H.; Ando, Y.; Suzuki, S. Self-diffusion measurements of liquid Sn using the shear cell technique and stable density layering. Metall. Mater. Trans. B 2018, 49, 3357–3366. [Google Scholar] [CrossRef]
- Suzuki, S.; Kraatz, K.-H.; Frohberg, G.; Roşu-Pflumm, R.; Müller-Vogt, G. Impurity diffusion measurements of Bi in liquid Sn using stable density layering and the shear cell technique. J. Non-Cryst. Solids 2007, 353, 3300–3304. [Google Scholar] [CrossRef]
- Yamada, N.; Suzuki, S.; Suzuki, K.; Tanaka, A.; Morita, R.; Che, C.; Frohberg, G. Investigation of the influence of different solute on Impurity Diffusion in Liquid Sn using the Shear cell technique. Int. J. Microgravity Sci. Appl. 2018, 35, 350402. [Google Scholar]
- Suzuki, S.; Kraatz, K.-H.; Frohberg, G. Diffusion experiments in liquid Sn-Bi and Al-Ni systems with a stable density layering using the FOTON shear cell under 1g conditions. Microgravity-Sci. Technol. 2005, 16, 120–126. [Google Scholar] [CrossRef]
- Verhoeven, J.; Gibson, E.; Griffith, R. Measurement of liquid metal diffusion coefficients from steady-state solidification experiments. Metall. Trans. B 1975, 6, 475–480. [Google Scholar] [CrossRef]
- Porth, C.; Cahoon, J. Interdiffusion of Bi in liquid Sn. J. Phase Equilibria Diffus. 2010, 31, 149–156. [Google Scholar] [CrossRef]
- Nowok, J.W. Analysis of atomic diffusion in liquid metals at melting temperatures in capillary-like media. Acta Metall. Mater. 1994, 42, 4025–4028. [Google Scholar] [CrossRef]
- Kargl, F.; Engelhardt, M.; Yang, F.; Weis, H.; Schmakat, P.; Schillinger, B.; Griesche, A.; Meyer, A. In situ studies of mass transport in liquid alloys by means of neutron radiography. J. Phys. Condens. Matter 2011, 23, 254201. [Google Scholar] [CrossRef]
- Egelstaff, P.A. Neutron scattering studies of liquid diffusion. Adv. Phys. 1962, 11, 203–232. [Google Scholar] [CrossRef]
- Springer, T.; Lechner, R.E. Diffusion studies of solids by quasielastic neutron scattering. In Diffusion in Condensed Matter: Methods, Materials, Models; Springer: Berlin/Heidelberg, Germany, 2005; pp. 93–164. [Google Scholar]
- Venkatesan, T.; Edelson, D.; Brown, W. Pulsed ion beam technique for measuring diffusion coefficient of a slow diffusant in polymers. Appl. Phys. Lett. 1983, 43, 364–366. [Google Scholar] [CrossRef]
- Rej, D.; Davis, H.; Olson, J.; Remnev, G.; Zakoutaev, A.; Ryzhkov, V.; Struts, V.; Isakov, I.; Shulov, V.; Nochevnaya, N. Materials processing with intense pulsed ion beams. J. Vac. Sci. Technol. A Vac. Surf. Film. 1997, 15, 1089–1097. [Google Scholar] [CrossRef]
- Barriga, S.A. An Electrochemical Investigation of the Chemical Diffusivity in Liquid Metal Alloys. Ph.D. Thesis, Massachusetts Institute of Technology, Cambridge, MA, USA, 2013. [Google Scholar]
- McCue, I.; Karma, A.; Erlebacher, J. Pattern formation during electrochemical and liquid metal dealloying. Mrs Bull. 2018, 43, 27–34. [Google Scholar] [CrossRef]
- Wen, C.; Ho, C.; Boukamp, B.; Raistrick, I.; Weppner, W.; Huggins, R. Use of electrochemical methods to determine chemical-diffusion coefficients in alloys: Application to ‘LiAI’. Int. Met. Rev. 1981, 26, 253–268. [Google Scholar] [CrossRef]
- Anderson, J.S.; Saddington, K. S 80. The use of radioactive isotopes in the study of the diffusion of ions in solution. J. Chem. Soc. (Resumed) 1949, S381–S386. [Google Scholar] [CrossRef]
- Geng, Y.; Zhu, C.; Zhang, B. A sliding cell technique for diffusion measurements in liquid metals. AIP Adv. 2014, 4, 037102. [Google Scholar] [CrossRef]
- Krishnamurthi, V.; Parker, C.J.; Nguyen, C.K.; Vaillant, P.H.; Hocking, R.K.; Haas, B.; Christofferson, A.J.; Russo, S.P.; Chiang, K.; Elbourne, A. A toolbox for investigating liquid metal systems. Cell Rep. Phys. Sci. 2024, 5, 101820. [Google Scholar] [CrossRef]
- Zhmud, B.; Tiberg, F.; Hallstensson, K. Dynamics of capillary rise. J. Colloid Interface Sci. 2000, 228, 263–269. [Google Scholar] [CrossRef]
- Chang, I.; Cai, Q. From simple binary to complex multicomponent eutectic alloys. Prog. Mater. Sci. 2022, 123, 100779. [Google Scholar] [CrossRef]
- Shu, J.; Lu, Y.; Wang, E.; Li, X.; Tang, S.-Y.; Zhao, S.; Zhou, X.; Sun, L.; Li, W.; Zhang, S. Particle-based porous materials for the rapid and spontaneous diffusion of liquid metals. ACS Appl. Mater. Interfaces 2020, 12, 11163–11170. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Jin, T.; Kou, J.; Zou, S.; Xiao, J.; Meng, Q. Lucas–Washburn equation-based modeling of capillary-driven flow in porous systems. Langmuir 2021, 37, 1623–1636. [Google Scholar] [CrossRef]
- Porth, C.B. An Investigation of the Concentration Dependence of the Interdiffusion Coefficient in the Binary Liquid Aluminum-copper System. Ph.D. Thesis, University of Manitoba, Winnipeg, MB, Canada, 2016. [Google Scholar]
- Mirshamsi, M. Diffusion Phenomena in the Liquid Cadmium-Lead Binary System. Ph.D. Thesis, The University of Oklahoma, Norman, OK, USA, 1964. [Google Scholar]
- Trimble, L.; Pieper, W.; Canfield, F. Diffusion in liquid mixtures of cadmium and lead at 350 °C. Metall. Trans. 1971, 2, 1669–1673. [Google Scholar] [CrossRef]
- Niwa, K.; Kado, S.; Ohno, T. Diffusion of silver in molten copper and copper-silver alloy. Jpn. Inst. Met. Mater. 1962, 26, 726–730. [Google Scholar][Green Version]
- Porth, C.B. Interdiffusion of Bismuth and Zinc in Liquid Tin & the Effect of Capillary Diameter on the Diffusion Coefficient. Master’s Thesis, University of Manitoba, Winnipeg, MB, Canada, 2008. [Google Scholar]
- Robert, J. DüTusion Molten Metals Metailoids. Master’s Thesis, Queen’s University, Kingston, ON, Canada, 1999. [Google Scholar]
- Luk, C.; Nanis, L.; Litt, M. Improved Capillary Method for Diffusivity Measurements. Ind. Eng. Chem. Fundam. 1975, 14, 92–95. [Google Scholar] [CrossRef]
- Kurtuldu, G.; Jarry, P.; Rappaz, M. Influence of icosahedral short range order on diffusion in liquids: A study on Al-Zn-Cr alloys. Acta Mater. 2016, 115, 423–433. [Google Scholar] [CrossRef]
- Shirakata, K.; Yamamoto, T.; Takeda, T.; Unesaki, H. An improved cell calculation method for liquid-metal fast breeder reactor blanket analysis. Nucl. Sci. Eng. 1988, 98, 118–127. [Google Scholar] [CrossRef]
- Delgado, H.E.; Brown, G.H.; Bartels, D.M.; Rumbach, P.; Go, D.B. The scaling of kinetic and transport behaviors in the solution-phase chemistry of a plasma–liquid interface. J. Appl. Phys. 2021, 129, 083303. [Google Scholar] [CrossRef]
- Sanders, P.; Aziz, M. Self-diffusivity of liquid silicon measured by pulsed laser melting. J. Appl. Phys. 1999, 86, 4258–4261. [Google Scholar] [CrossRef]
- Heshmatpour, B.; Stevenson, D. An electrochemical study of the solubility and diffusivity of oxygen in the respective liquid metals indium, gallium, antimony and bismuth. J. Electroanal. Chem. Interfacial Electrochem. 1981, 130, 47–55. [Google Scholar]
- Chou, H.; Chow, T.; Tsay, S.; Chen, H. Diffusivity of oxygen in liquid Sn and Ba0.35Cu0.65 alloys. J. Electrochem. Soc. 1995, 142, 1814. [Google Scholar] [CrossRef]
- Ganesan, R.; Gnanasekaran, T.; Srinivasa, R.S. Diffusivity, activity and solubility of oxygen in liquid lead and lead–bismuth eutectic alloy by electrochemical methods. J. Nucl. Mater. 2006, 349, 133–149. [Google Scholar] [CrossRef]
- Sears, B.; Anderson, T.; Narayanan, R.; Fripp, A. The detection of solutal convection during electrochemical measurement of the oxygen diffusivity in liquid tin. Metall. Trans. B 1993, 24, 91–100. [Google Scholar] [CrossRef]
- Murakami, T.; Koyama, T. Application of electrochemical method to measure diffusion coefficient in liquid metal. J. Electrochem. Soc. 2011, 158, F147. [Google Scholar] [CrossRef]
- Novoselova, A.; Smolenski, V.; Mushnikov, P. Electrochemistry of uranium on liquid cadmium electrode in LiCl-KCl eutectic. J. Electrochem. Soc. 2024, 171, 042504. [Google Scholar] [CrossRef]
- Murakami, T.; Sakamura, Y.; Akiyama, N.; Kitawaki, S.; Nakayoshi, A.; Koyama, T. Electrochemical measurement of diffusion coefficient of actinides and rare earths in liquid Cd. Procedia Chem. 2012, 7, 798–803. [Google Scholar] [CrossRef]
- Yoon, D.; Phongikaroon, S.; Zhang, J. Electrochemical and thermodynamic properties of CeCl3 on liquid cadmium cathode (LCC) in LiCl-KCl eutectic salt. J. Electrochem. Soc. 2016, 163, E97–E103. [Google Scholar] [CrossRef]
- Weppner, W.; Huggins, R.A. Determination of the kinetic parameters of mixed-conducting electrodes and application to the system Li3Sb. J. Electrochem. Soc. 1977, 124, 1569. [Google Scholar] [CrossRef]
- Kim, J.; Park, S.; Hwang, S.; Yoon, W.-S. Principles and applications of galvanostatic intermittent titration technique for lithium-ion batteries. J. Electrochem. Sci. Technol. 2022, 13, 19–31. [Google Scholar] [CrossRef]
- Wang, P.; Leong, A.; Zhang, J. Electrochemical Methods for Fundamental Data Measurements in Molten Salts: Wang, Leong, and Zhang. JOM 2023, 75, 3687–3698. [Google Scholar] [CrossRef]
- Williams, T.; Shum, R.; Rappleye, D. Concentration measurements in molten chloride salts using electrochemical methods. J. Electrochem. Soc. 2021, 168, 123510. [Google Scholar] [CrossRef]
- Yin, T.; Liu, Y.; Yang, D.; Yan, Y.; Wang, G.; Chai, Z.; Shi, W. Thermodynamics and kinetics properties of lanthanides (La, Ce, Pr, Nd) on liquid bismuth electrode in LiCl-KCl molten salt. J. Electrochem. Soc. 2020, 167, 122507. [Google Scholar] [CrossRef]
- Mandzyuk, V.; Ivanichok, N.Y.; Solomoskyi, R. Li-ion Diffusion in Porous Carbon Electrode Materials by GITT Method. J. Nano-Electron. Phys. 2024, 16, 02024-1–02024-4. [Google Scholar] [CrossRef]
- Schied, T.; Nickol, A.; Heubner, C.; Schneider, M.; Michaelis, A.; Bobeth, M.; Cuniberti, G. Determining the diffusion coefficient of lithium insertion cathodes from GITT measurements: Theoretical analysis for low temperatures. ChemPhysChem 2021, 22, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Julien, C.; Mauger, A. Diffusion. In Rechargeable Lithium Metal Batteries: Science and Technology; Springer: Berlin/Heidelberg, Germany, 2024; pp. 1–24. [Google Scholar]
- Nickol, A.; Schied, T.; Heubner, C.; Schneider, M.; Michaelis, A.; Bobeth, M.; Cuniberti, G. GITT analysis of lithium insertion cathodes for determining the lithium diffusion coefficient at low temperature: Challenges and pitfalls. J. Electrochem. Soc. 2020, 167, 090546. [Google Scholar] [CrossRef]
- Horner, J.S.; Whang, G.; Ashby, D.S.; Kolesnichenko, I.V.; Lambert, T.N.; Dunn, B.S.; Talin, A.A.; Roberts, S.A. Electrochemical modeling of GITT measurements for improved solid-state diffusion coefficient evaluation. ACS Appl. Energy Mater. 2021, 4, 11460–11469. [Google Scholar] [CrossRef]
- Ivanishchev, A.; Ivanishcheva, I. Ion transport in lithium electrochemical systems: Problems and solutions. Russ. J. Electrochem. 2020, 56, 907–928. [Google Scholar] [CrossRef]
- Nowak, A.P.; Rutecki, P.; Szkoda, M.; Trzciński, K. Determination of sodium ion diffusion coefficient in tin sulfide@ carbon anode material using GITT and EIS techniques. Energies 2024, 17, 3233. [Google Scholar] [CrossRef]
- Liu, K.; Gao, Y.; Fang, Z.; Zhou, X.; Ma, Y.; Wu, H.; Kang, M.; Wang, B. Determination of diffusion coefficients of uranium in liquid gallium by GITT. J. Electroanal. Chem. 2020, 879, 114711. [Google Scholar] [CrossRef]
- Fawcett, N.; Caton, R.D. Analysis of errors in the capillary method for determining diffusion coefficients. Anal. Chem. 1976, 48, 228–229. [Google Scholar] [CrossRef]
- Zoski, C.G. Handbook of Electrochemistry; Elsevier: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Zhou, W.; Shen, Z.; Yin, X.; Zhang, Y.; Zhao, M. Effects of pressure on the eutectic or eutectoid temperatures of the CdPb, CdSn, PbSn and CdPbSn systems. J. Less Common Met. 1988, 143, 59–69. [Google Scholar] [CrossRef]
- Hesson, J.; Burris, L. Uranium diffusivity in liquid cadmium. Trans. AIME 1963, 227, 571–581. [Google Scholar]
- Carey, A.E.; Wheatcraft, S.W.; Glass, R.J.; O’Rourke, J.P. Non-Fickian ionic diffusion across high-concentration gradients. Water Resour. Res. 1995, 31, 2213–2218. [Google Scholar] [CrossRef]
- Goesele, U.; Laveant, P.; Scholz, R.; Engler, N.; Werner, P. Diffusion engineering by carbon in silicon. Mater. Res. Soc. Symp. 2000, 610, 711. [Google Scholar]
- Shampine, L. Concentration-dependent diffusion. Q. Appl. Math. 1973, 30, 441–452. [Google Scholar] [CrossRef][Green Version]





| System | Diffusion Type | Temperature (K) | Diffusion Coefficient D (m2·s−1) | References |
|---|---|---|---|---|
| Cu → liquid Al | Solute diffusion | >933 K (liquid Al region) | D = 1.05 (±0.15) ×10−7 exp[−(23.8 ± 1.3)/RT] | [122] |
| Cd–Pb liquid alloy | Chemical/mutual diffusion | 628–758 K | (1.7–3.2) × 10−9 | [123] |
| Cd–Pb liquid alloy | Chemical/mutual diffusion | 623 K | (0.2–2.0) × 10−9 | [124] |
| Ag → liquid Bi | Solute diffusion | 656–872 K | D = (8.19 ± 0.79) × 10−8 exp[−(4100 ± 60)/RT] −1 | [125] |
| Ag → liquid Cd | Solute diffusion | 637–814 K | D = (5.68 ± 0.90) × 10−8 exp[−(4380 ± 90)/RT] cm2·s−1 | [125] |
| Bi → liquid Sn | Interdiffusion | 773–873 K | D = (11.56 ± 2.39) × 10−8 exp[−(2.29 ± 0.74)/RT] | [126] |
| Zn → liquid Sn | Interdiffusion | 773–873 K | D = (1.34 ± 0.83) × 10−8 exp[−(6.86 ± 3.17)/RT] | [126] |
| Ag → liquid Pb | Solute diffusion | 583.15 K | 5.80 × 10−9 | [127] |
| Sb → liquid Pb | Solute diffusion | 583.15 K | 4.60 × 10−9 | [127] |
| Au → liquid Pb | Solute diffusion | 593.15 K | 2.45 × 10−9 | [127] |
| U | Pu | La | Ce | Pr | Nd | Gd | Y | Sc | |
|---|---|---|---|---|---|---|---|---|---|
| D × 106 (cm2 s−1) for 738–743 K | 17 (738 K) | 11 (723 K) | 1.9 (723 K) | 2.9 (723 K) | 3.2 (723 K) | 2.7 (743 K) | 3.2 (723 K) | 6.8 (743 K) | 6.5 (737 K) |
| D × 106 (cm2 s−1) for 773 K | 19.9±0.7 | 14 | 2.5 | 4.8 | 3.8 | 3.4 | 3.9 | 8.2 | 7.7 |
| D × 106 (cm2 s−1) for 823 K | 23 | 16 | 3.4 | / | 4.3 | 3.9 | 4.3 | 9.3 | 8.0 |
| References | [138,139] | [139] | [137] | [140] | [137] | [137] | [137] | [137] | [137] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, Y.; Jia, Y.; Xiao, Y.; Yao, B.; He, H. Physical State and Mass Transport of Metals in Liquid Cadmium Cathodes: A Review. Processes 2026, 14, 953. https://doi.org/10.3390/pr14060953
Wang Y, Jia Y, Xiao Y, Yao B, He H. Physical State and Mass Transport of Metals in Liquid Cadmium Cathodes: A Review. Processes. 2026; 14(6):953. https://doi.org/10.3390/pr14060953
Chicago/Turabian StyleWang, Yilin, Yanhong Jia, Yiqun Xiao, Benlin Yao, and Hui He. 2026. "Physical State and Mass Transport of Metals in Liquid Cadmium Cathodes: A Review" Processes 14, no. 6: 953. https://doi.org/10.3390/pr14060953
APA StyleWang, Y., Jia, Y., Xiao, Y., Yao, B., & He, H. (2026). Physical State and Mass Transport of Metals in Liquid Cadmium Cathodes: A Review. Processes, 14(6), 953. https://doi.org/10.3390/pr14060953






