Effect-Directed Extraction of Grape Pomace: Optimizing Antioxidant and Antibrowning Efficacy
Abstract
1. Introduction
2. Materials and Methods
2.1. Grape Pomace Material
2.2. General Extraction Procedure
2.3. General TLC Spotting and Development
2.4. TLC Scavenging and Inhibition Assays
2.4.1. TLC-ABTS Assay
2.4.2. TLC-DPPH Assay
2.4.3. TLC-Tyrosinase Assay
2.4.4. TLC-Laccase Assay
2.5. Extraction Optimization and Validation
2.6. Anti-Browning Effect Application on Apple Slices
2.7. Bioassay-Guided Purification and Spectral Characterization
2.8. Statistical Analysis
3. Results and Discussion
3.1. Selection of GP Samples Based on Antioxidant and Tyrosinase Inhibitory Effects on TLC
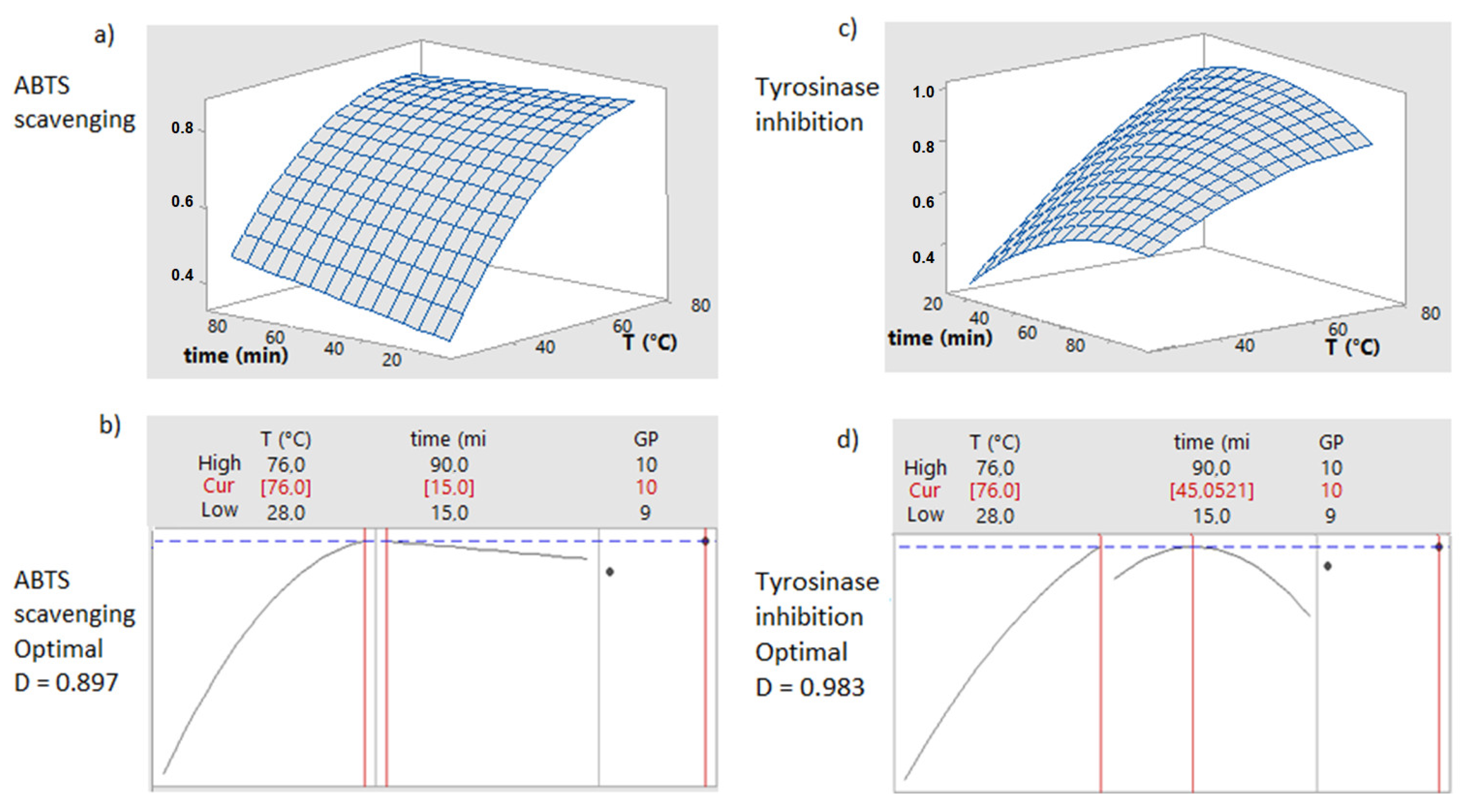
3.2. Optimization of Time and Temperature Using GP9 and GP10 Hydroalcoholic Extractions
3.3. Validation of Response Surface Values
3.4. Additional DPPH Radical Scavenging and Laccase Enzyme Inhibition Bioactivities on TLC
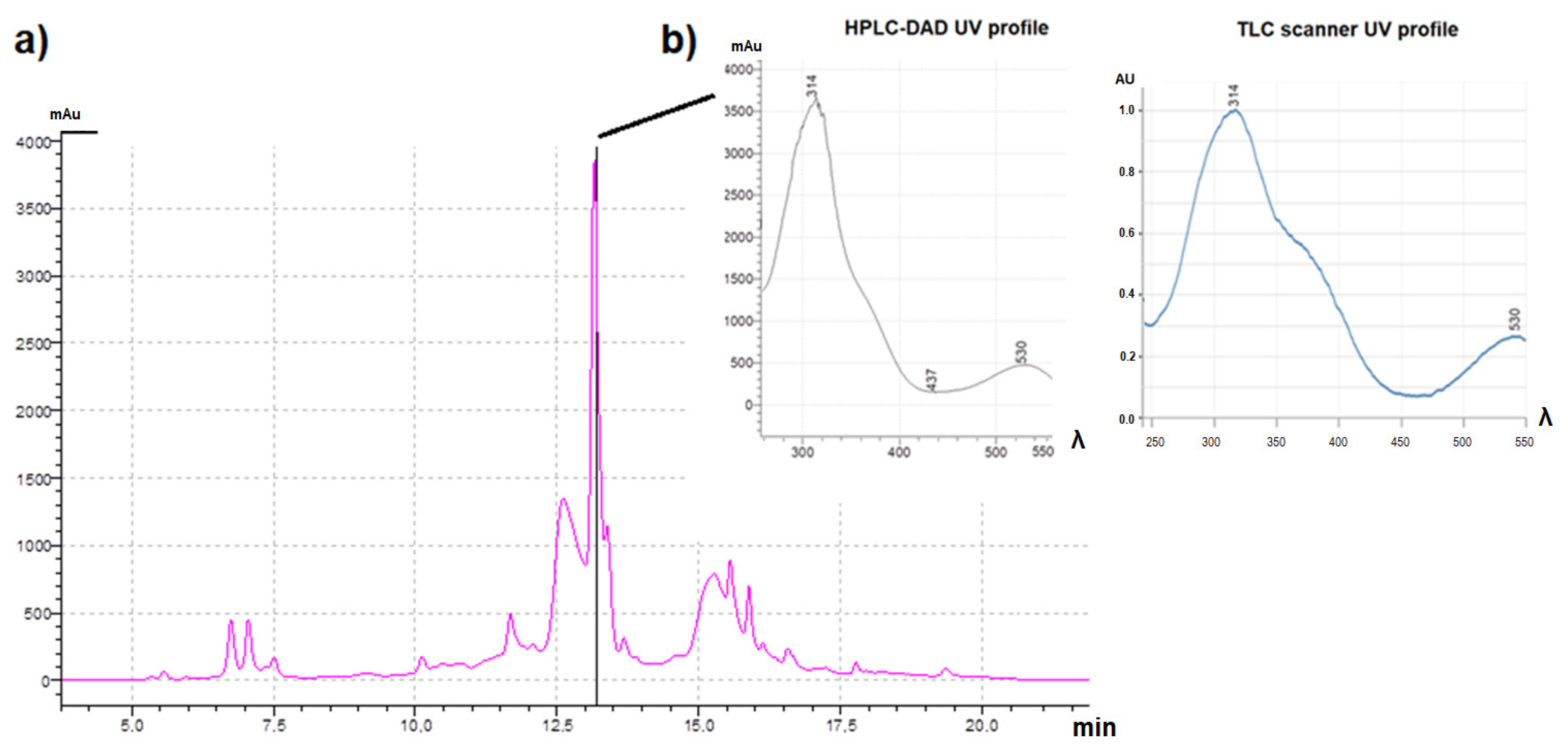
3.5. Effect-Directed Fractionation and HRMS/MS Structural Elucidation
3.6. Anti-Browning Effect on Apples of the Optimised Extracts
3.7. Mechanistic Insights and Comparison with Commercial Inhibitors
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ordoñez-Cano, A.J.; Ramírez-Esparza, U.; Alvarado-González, M.; Baeza-Jiménez, R.; Espinoza-Hicks, J.C.; Prado-Barragán, L.A.; Buenrostro-Figueroa, J.J. Valorization of Pistachio Green Hull: Advances in Extraction and Characterization of Phenolic Compounds. Processes 2025, 13, 3761. [Google Scholar] [CrossRef]
- Gerardi, C.; D’amico, L.; Migoni, D.; Santino, A.; Salomone, A.; Carluccio, M.A.; Giovinazzo, G. Strategies for Reuse of Skins Separated From Grape Pomace as Ingredient of Functional Beverages. Front. Bioeng. Biotechnol. 2020, 8, 645. [Google Scholar] [CrossRef]
- Almanza-Oliveros, A.; Bautista-Hernández, I.; Castro-López, C.; Aguilar-Zárate, P.; Meza-Carranco, Z.; Rojas, R.; Michel, M.R.; Martínez-Ávila, G.C.G. Grape Pomace—Advances in Its Bioactivity, Health Benefits, and Food Applications. Foods 2024, 13, 580. [Google Scholar] [CrossRef]
- Castellanos-Gallo, L.; Ballinas-Casarrubias, L.; Espinoza-Hicks, J.C.; Hernández-Ochoa, L.R.; Muñoz-Castellanos, L.N.; Zermeño-Ortega, M.R.; Borrego-Loya, A.; Salas, E. Grape Pomace Valorization by Extraction of Phenolic Polymeric Pigments: A Review. Processes 2022, 10, 469. [Google Scholar] [CrossRef]
- Singh, B.; Suri, K.; Shevkani, K.; Kaur, A.; Kaur, A.; Singh, N. Enzymatic Browning of Fruit and Vegetables: A Review. In Enzymes in Food Technology: Improvements and Innovations; Kuddus, M., Ed.; Springer: Singapore, 2018; pp. 63–78. ISBN 978-981-13-1933-4. [Google Scholar]
- Soares, C.; Moreira, M.M.; Ramos, S.; Ramalhosa, M.J.; Correia, M.; Svarc-Gajić, J.; Delerue-Matos, C.; Barroso, M.F. A Critical Assessment of Extraction Methodologies for the Valorization of Agricultural Wastes: Polyphenolic Profile and Bioactivity. Processes 2023, 11, 1767. [Google Scholar] [CrossRef]
- Galante, M.; Brassesco, M.E.; Maragoni Santos, C.; Beres, C.; Fai, A.E.C.; Cabezudo, I. Grape Pomace as a Natural Source of Antimicrobial Agents for Food Preservation. Front. Nutr. 2025, 12, 1650450. [Google Scholar] [CrossRef] [PubMed]
- Artem, V.; Negreanu–Pirjol, T.; Ranca, A.; Ciobanu, C.; Bratu, M.M.; Popoviciu, D.R.; Moldovan, L.; Vasile, M.; Negreanu-Pirjol, B.-S. Total Phenolic Content Correlated with Antioxidant Activity of Some Grape Pomace Biomass Hydroalcoholic Extracts, White and Red Varieties. UPB Sci. Bull. Ser. B 2021, 83, 61–72. [Google Scholar]
- Ferri, M.; Rondini, G.; Calabretta, M.M.; Michelini, E.; Vallini, V.; Fava, F.; Roda, A.; Minnucci, G.; Tassoni, A. White Grape Pomace Extracts, Obtained by a Sequential Enzymatic plus Ethanol-Based Extraction, Exert Antioxidant, Anti-Tyrosinase and Anti-Inflammatory Activities. New Biotechnol. 2017, 39, 51–58. [Google Scholar] [CrossRef]
- Li, Z.; Smith, K.H.; Stevens, G.W. The Use of Environmentally Sustainable Bio-Derived Solvents in Solvent Extraction Applications—A Review. Chin. J. Chem. Eng. 2016, 24, 215–220. [Google Scholar] [CrossRef]
- Mangiapelo, L.; Blasi, F.; Ianni, F.; Suvieri, C.; Sardella, R.; Volpi, C.; Cossignani, L. Optimization of a Simple Analytical Workflow to Characterize the Phenolic Fraction from Grape Pomace. Food Bioprocess Technol. 2024, 17, 1942–1957. [Google Scholar] [CrossRef]
- Vaou, N.; Stavropoulou, E.; Voidarou, C.; Tsigalou, C.; Bezirtzoglou, E. Towards Advances in Medicinal Plant Antimicrobial Activity: A Review Study on Challenges and Future Perspectives. Microorganisms 2021, 9, 2041. [Google Scholar] [CrossRef]
- Choma, I.M.; Grzelak, E.M. Bioautography Detection in Thin-Layer Chromatography. J. Chromatogr. A 2011, 1218, 2684–2691. [Google Scholar] [CrossRef]
- Cabezudo, I.; Salazar, M.O.; Ramallo, I.A.; Furlan, R.L. Effect-Directed Analysis in Food by Thin-Layer Chromatography Assays. Food Chem. 2022, 390, 132937. [Google Scholar] [CrossRef]
- Cabezudo, I.; Ayelen Ramallo, I.; Alonso, V.L.; Furlan, R.L.E. Effect Directed Synthesis of a New Tyrosinase Inhibitor with Anti-Browning Activity. Food Chem. 2021, 341, 128232. [Google Scholar] [CrossRef]
- García, P.; Furlan, R.L.E. Multiresponse Optimisation Applied to the Development of a TLC Autography for the Detection of Tyrosinase Inhibitors. Phytochem. Anal. 2015, 26, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Danciu, V.; Hosu, A.; Cimpoiu, C. Comparative Evaluation of Antioxidant Activity Using 1,1-Diphenyl-2-Picrylhydrazyl and 2,2′-Azino-Bis(3-Ethylbenzothiazoline-6-Sulphonic Acid) Methods. JPC-J. Planar Chromatogr.-Mod. TLC 2016, 29, 306–309. [Google Scholar] [CrossRef]
- Cabezudo, I.; Galetto, C.S.; Romanini, D.; Furlán, R.L.E.; Meini, M.R. Production of Gallic Acid and Relevant Enzymes by Aspergillus Niger and Aspergillus Oryzae in Solid-State Fermentation of Soybean Hull and Grape Pomace. Biomass Convers. Biorefinery 2022, 13, 14939–14947. [Google Scholar] [CrossRef]
- Yu, J.; Ahmedna, M. Functional Components of Grape Pomace: Their Composition, Biological Properties and Potential Applications. Int. J. Food Sci. Technol. 2013, 48, 221–237. [Google Scholar] [CrossRef]
- Cabezudo, I.; Furlan, R.L.E. Thin Layer Chromatography Assay to Detect Laccase Inhibitors. Food Chem. 2024, 460, 140583. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant Activity Applying an Improved ABTS Radical Cation Decolorization Assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Zampini, I.C.; Ordoñez, R.M.; Isla, M.I. Autographic Assay for the Rapid Detection of Antioxidant Capacity of Liquid and Semi-Solid Pharmaceutical Formulations Using ABTS•+ Immobilized by Gel Entrapment. AAPS PharmSciTech 2010, 11, 1159–1163. [Google Scholar] [CrossRef]
- Wu, J.-J.; Cheng, K.-W.; Li, E.T.S.; Wang, M.; Ye, W.-C. Antibrowning Activity of MRPs in Enzyme and Fresh-Cut Apple Slice Models. Food Chem. 2008, 109, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Lante, A.; Tinello, F.; Nicoletto, M. UV-A Light Treatment for Controlling Enzymatic Browning of Fresh-Cut Fruits. Innov. Food Sci. Emerg. Technol. 2016, 34, 141–147. [Google Scholar] [CrossRef]
- Moon, K.M.; Kwon, E.-B.; Lee, B.; Kim, C.Y. Recent Trends in Controlling the Enzymatic Browning of Fruit and Vegetable Products. Molecules 2020, 25, 2754. [Google Scholar] [CrossRef] [PubMed]
- Moutinho, J.; Gouvinhas, I.; Domínguez-Perles, R.; Barros, A. Optimization of the Extraction Methodology of Grape Pomace Polyphenols for Food Applications. Molecules 2023, 28, 3885. [Google Scholar] [CrossRef]
- Pop, A.; Fizeșan, I.; Vlase, L.; Rusu, M.E.; Cherfan, J.; Babota, M.; Gheldiu, A.-M.; Tomuta, I.; Popa, D.-S. Enhanced Recovery of Phenolic and Tocopherolic Compounds from Walnut (Juglans regia L.) Male Flowers Based on Process Optimization of Ultrasonic Assisted-Extraction: Phytochemical Profile and Biological Activities. Antioxidants 2021, 10, 607. [Google Scholar] [CrossRef]
- Chaudhary, D.; Chong, F.; Neupane, T.; Choi, J.; Jee, J.-G. New Inhibitors of Laccase and Tyrosinase by Examination of Cross-Inhibition between Copper-Containing Enzymes. Int. J. Mol. Sci. 2021, 22, 13661. [Google Scholar] [CrossRef]
- Oliveira, J.; Alhinho da Silva, M.; Teixeira, N.; De Freitas, V.; Salas, E. Screening of Anthocyanins and Anthocyanin-Derived Pigments in Red Wine Grape Pomace Using LC-DAD/MS and MALDI-TOF Techniques. J. Agric. Food Chem. 2015, 63, 7636–7644. [Google Scholar] [CrossRef]
- Flamini, R. Mass Spectrometry in Grape and Wine Chemistry. Part I: Polyphenols. Mass Spectrom. Rev. 2003, 22, 218–250. [Google Scholar] [CrossRef]
- Sharafan, M.; Malinowska, M.A.; Ekiert, H.; Kwaśniak, B.; Sikora, E.; Szopa, A. Vitis vinifera (Vine Grape) as a Valuable Cosmetic Raw Material. Pharmaceutics 2023, 15, 1372. [Google Scholar] [CrossRef]
- Perez-Gago, M.B.; Serra, M.; Alonso, M.; Mateos, M.; del Río, M.A. Effect of Whey Protein- and Hydroxypropyl Methylcellulose-Based Edible Composite Coatings on Color Change of Fresh-Cut Apples. Postharvest Biol. Technol. 2005, 36, 77–85. [Google Scholar] [CrossRef]
- Fia, G.; Gori, C.; Bucalossi, G.; Borghini, F.; Zanoni, B. A Naturally Occurring Antioxidant Complex from Unripe Grapes: The Case of Sangiovese (v. Vitis vinifera). Antioxidants 2018, 7, 27. [Google Scholar] [CrossRef] [PubMed]
- Fang, T.; Chen, J.; Lin, Q.; Zhong, Y.; Duan, Y.; Bi, J. Phenolic Profiling Reveals the Metabolite Basis of Flesh Colour and Fresh-Cut Browning in Apple Fruit. Int. J. Food Sci. Technol. 2022, 57, 2257–2266. [Google Scholar] [CrossRef]
- Shen, M.; Liu, K.; Liang, Y.; Liu, G.; Sang, J.; Li, C. Extraction Optimization and Purification of Anthocyanins from Lycium Ruthenicum Murr. and Evaluation of Tyrosinase Inhibitory Activity of the Anthocyanins. J. Food Sci. 2020, 85, 696–706. [Google Scholar] [CrossRef]
- Pathak, S.S.; Sonawane, A.; Srinivas, A.; Pradhan, R.C. Application of Image Analysis for Detecting the Browning of Unripe Banana Slices. ACS Food Sci. Technol. 2021, 1, 1507–1513. [Google Scholar] [CrossRef]
- Sultan, M.Z.; Farouk, K.A.; Elbagoury, M.M.; Yahia, E.M. Trends in Biochemical, Anatomical Mechanisms and Molecular Aspects in Enzymatic Browning of Apples: A Review. Eur. Food Res. Technol. 2025, 251, 3305–3326. [Google Scholar] [CrossRef]





| Sample ID | Cultivar | Region |
|---|---|---|
| GP1 | Malbec | Valle de Uco, Mendoza |
| GP2 | Cabernet Sauvignon | Valle de Uco, Mendoza |
| GP3 | Malbec | Valle de Uco, Mendoza |
| GP4 | Malbec | Gualtallari, Mendoza |
| GP5 | Malbec | Tunuyán, Mendoza |
| GP6 | Malbec | San Rafael, Mendoza |
| GP7 | Malbec | San Rafael, Mendoza |
| GP8 | Marselan | Victoria, Entre Ríos |
| GP9 | Malbec | San Rafael, Mendoza |
| GP10 | Malbec | San Rafael, Mendoza |
| GP11 | Cabernet Sauvignon | San Rafael, Mendoza |
| GP12 | Cabernet Sauvignon | San Rafael, Mendoza |
| (a) ABTS Scavenging Optimisation | ||||
|---|---|---|---|---|
| Trial | GP | T (°C) | Time (min) | ABTS Total Area |
| 1 | 9 | 28 | 15 | 0.05 |
| 3 | 9 | 28 | 90 | 0.27 |
| 4 | 9 | 50 | 15 | 0.61 |
| 6 | 9 | 50 | 90 | 0.59 |
| 7 | 9 | 76 | 15 | 0.71 |
| 9 | 9 | 76 | 90 | 0.90 |
| 10 | 10 | 28 | 15 | 0.39 |
| 12 | 10 | 28 | 90 | 0.46 |
| 13 | 10 | 50 | 15 | 0.56 |
| 15 | 10 | 50 | 90 | 0.80 |
| 16 | 10 | 76 | 15 | 0.95 |
| 18 | 10 | 76 | 90 | 0.75 |
| (b) Tyrosinase Inhibition Optimisation | ||||
| Trial No. | GP | T (°C) | Time (min) | Tirosinase inh. (Peak Area) |
| 1 | 9 | 28 | 15 | 0.11 |
| 2 | 9 | 28 | 45 | 0.33 |
| 3 | 9 | 28 | 90 | 0.44 |
| 4 | 9 | 50 | 15 | 0.37 |
| 5 | 9 | 50 | 45 | 0.70 |
| 6 | 9 | 50 | 90 | 0.81 |
| 7 | 9 | 76 | 15 | 0.81 |
| 8 | 9 | 76 | 45 | 0.96 |
| 9 | 9 | 76 | 90 | 0.85 |
| 10 | 10 | 28 | 15 | 0.30 |
| 11 | 10 | 28 | 45 | 0.48 |
| 12 | 10 | 28 | 90 | 0.52 |
| 13 | 10 | 50 | 15 | 0.52 |
| 14 | 10 | 50 | 45 | 0.74 |
| 15 | 10 | 50 | 90 | 0.85 |
| 16 | 10 | 76 | 15 | 0.99 |
| 17 | 10 | 76 | 45 | 0.96 |
| 18 | 10 | 76 | 90 | 0.78 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cabezudo, I.; Campero, M.; Escalante, A.M.; Furlan, R.L.E. Effect-Directed Extraction of Grape Pomace: Optimizing Antioxidant and Antibrowning Efficacy. Processes 2026, 14, 925. https://doi.org/10.3390/pr14060925
Cabezudo I, Campero M, Escalante AM, Furlan RLE. Effect-Directed Extraction of Grape Pomace: Optimizing Antioxidant and Antibrowning Efficacy. Processes. 2026; 14(6):925. https://doi.org/10.3390/pr14060925
Chicago/Turabian StyleCabezudo, Ignacio, Maximiliano Campero, Andrea M. Escalante, and Ricardo L. E. Furlan. 2026. "Effect-Directed Extraction of Grape Pomace: Optimizing Antioxidant and Antibrowning Efficacy" Processes 14, no. 6: 925. https://doi.org/10.3390/pr14060925
APA StyleCabezudo, I., Campero, M., Escalante, A. M., & Furlan, R. L. E. (2026). Effect-Directed Extraction of Grape Pomace: Optimizing Antioxidant and Antibrowning Efficacy. Processes, 14(6), 925. https://doi.org/10.3390/pr14060925







