Physicochemical Characterization of Biochar Sorbents Produced at Different Temperatures from Malt Spent Rootlets
Abstract
1. Introduction
2. Materials and Methods
2.1. Material Production
2.2. Inorganic Properties
2.3. Carbon Nature
2.4. Texture and Sorption Properties
3. Results
3.1. Inorganic Properties
3.2. Carbon Nature
3.3. Texture and Sorption Properties
3.4. Chlorine Removal Ability
4. Discussion
5. Conclusions
- Malt spent rootlets can be successfully pyrolyzed at temperatures up to 900 °C to produce biochar with different textural and chemical properties.
- Based on the application and the desired characteristics of the material to be produced, one can pick the desired pyrolysis temperature and select simple analytical techniques that are presented in the present study to determine the suitability of each biochar.
- Increasing the pyrolysis temperature leads to a significant increase in the surface area and pore volume, mainly due to the development of micro and mesopores. Biochars pyrolyzed at 750–850 °C exhibit the highest specific surface area, making them the most promising sorbents.
- Sorption experiments using methylene blue and molasses number demonstrated that higher pyrolysis temperatures enhance both mesopore and macropore development, making biochar a diverse sorptive material suitable for different applications.
- Chlorine removal experiments revealed that biochar pyrolyzed at 850 °C has the highest chlorine removal capacity and the lowest half value length, confirming its efficiency. Higher pyrolysis temperatures and powdered forms increase the removal rate constants, indicating faster chlorine removal due to increased surface area and accessibility of active sites.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 1D | Distilled water |
| ATR-FTIR | Attenuated Total Reflectance-Fourier Transform Infra-Red |
| BET | Brunauer, Emmett, and Teller equation |
| EC | Electrical Conductivity |
| EDS | Energy Dispersive microanalyzer detector |
| HVL | Half Value Length |
| MSR | Malt Spent Rootlets |
| SEM | Scanning Electron Microscopy |
| XRD | X-ray Diffraction |
References
- Lehmann, J.; Joseph, S. (Eds.) Biochar for Environmental Management: Science, Technology and Implementation, 2nd ed.; Routledge: London, UK, 2015; 976p. [Google Scholar] [CrossRef]
- Mohan, D.; Sarswat, A.; Ok, Y.S.; Pittman, C.U., Jr. Organic and inorganic contaminants removal from water with biochar, a renewable, low cost and sustainable adsorbent—A critical review. Bioresour. Technol. 2014, 160, 191–202. [Google Scholar] [CrossRef]
- Sun, L.M.; McIntyre, S.R.; Iacomi, P.; Everden, K.; Williams, P.T.; Zong, S.; Liu, X.; Zhu, X.; Yang, Y.; Li, S.; et al. Biochar production, activation, and applications: A comprehensive technical review. Carbon Capture Sci. Technol. 2025, 16, 100421. [Google Scholar] [CrossRef]
- Chan, K.Y.; Van Zwieten, L.; Meszaros, I.; Downie, A.; Joseph, S. Agronomic values of greenwaste biochar as a soil amendment. Aust. J. Soil Res. 2007, 45, 629–634. [Google Scholar] [CrossRef]
- Yang, B.; Li, D.; Yuan, S.; Jin, L. Role of biochar from corn straw in influencing crack propagation and evaporation in sodic soils. Catena 2021, 204, 105457. [Google Scholar] [CrossRef]
- Liang, L.; Xi, F.; Tan, W.; Meng, X.; Hu, B.; Wang, J. Review of organic and inorganic pollutants removal by biochar and biochar-based composites. Biochar 2021, 3, 255–281. [Google Scholar] [CrossRef]
- Novak, J.M.; Lima, I.; Xing, B.; Gaskin, J.W.; Steiner, C.; Das, K.C.; Ahmedna, M.A.; Rehrah, D.; Watts, D.W.; Busscher, W.J.; et al. Characterization of designer biochar produced at different temperatures and their effects on a loamy sand. Ann. Environ. Sci. 2009, 3, 195. [Google Scholar] [CrossRef]
- Hossain, M.K.; Strezov, V.; Chan, K.Y.; Nelson, P.F. Agronomic properties of wastewater sludge biochar and bioavailability of metals in production of cherry tomato (Lycopersicon esculentum). Chemosphere 2010, 78, 1167. [Google Scholar] [CrossRef]
- Woolf, D.; Amonette, J.E.; Street-Perrott, F.A.; Lehmann, J.; Joseph, S. Sustainable biochar to mitigate global climate change. Nat. Commun. 2010, 1, 56. [Google Scholar] [CrossRef] [PubMed]
- Major, J.; Rondon, M.; Molina, D.; Riha, S.J.; Lehmann, J. Maize yield and nutrition during 4 years after biochar application to a Colombian savanna oxisol. Plant Soil 2010, 333, 117. [Google Scholar] [CrossRef]
- Cao, X.D.; Ma, L.Q.; Gao, B.; Harris, W. Dairy-manure derived biochar effectively sorbs lead and atrazine. Environ. Sci. Technol. 2009, 43, 3285. [Google Scholar] [CrossRef]
- Chen, X.C.; Chen, G.C.; Chen, L.G.; Chen, Y.X.; Lehmann, J.; McBride, M.B.; Hay, A.G. Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour. Technol. 2011, 102, 8877. [Google Scholar] [CrossRef]
- Paschalidou, P.; Pashalidis, I.; Manariotis, I.D.; Karapanagioti, H.K. Hyper sorption capacity of raw and oxidized biochars from various feedstocks for U(VI). J. Environ. Chem. Eng. 2020, 8, 103932. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef]
- Wang, S.; Shan, R.; Wang, Y.; Lu, L.; Yuan, H. Synthesis of calcium materials in biochar matrix as a highly stable catalyst for biodiesel production. Renew. Energy 2019, 130, 41–49. [Google Scholar] [CrossRef]
- Nikolopoulos, I.; Kordouli, E.; Mourgkogiannis, N.; Karapanagioti, H.K.; Lycourghiotis, A.; Kordulis, C. Valorization of pyrolyzed biomass residues for the transformation of waste Cooking Oil into Green Diesel. Catalysts 2023, 13, 1004. [Google Scholar] [CrossRef]
- Sathyabama, K.; Firdous, S. Effect of pyrolysis temperature on the physicochemical properties and structural characteristics of agricultural wastes-derived biochar. ACS Omega 2025, 10, 37013–37024. [Google Scholar] [CrossRef]
- Manariotis, I.D.; Fotopoulou, K.N.; Karapanagioti, H.K. Preparation and characterization of biochar sorbents produced from malt spent rootlets. Ind. Eng. Chem. Res. 2015, 54, 9577–9584. [Google Scholar] [CrossRef]
- Khater, E.S.; Bahnasawy, A.; Hamouda, R.; Ali, S.A.; Hassan, M. Biochar production under different pyrolysis temperatures with different types of agricultural wastes. Sci. Rep. 2024, 14, 2625. [Google Scholar] [CrossRef] [PubMed]
- Moya, R.; Tenorio, C.; Quesada-Kimzey, J.; Másis-Meléndez, F. Pyrogenic carbonaceous materials production of four tropical wood produced by slow pyrolysis at different temperatures: Charcoal and biochar properties. Energies 2024, 17, 1953. [Google Scholar] [CrossRef]
- Demirbas, A. Effects of temperature and particle size on bio-char yield from pyrolysis of agricultural residues. J. Anal. Appl. Pyrolysis 2004, 72, 243–248. [Google Scholar] [CrossRef]
- Rasa, K.; Viherä-Aarnio, A.; Rytkönen, P.; Hyväluoma, J.; Kaseva, J.; Suhonen, H.; Jyske, T. Quantitative analysis of feedstock structural properties can help to produce willow biochar with homogenous pore system. Ind. Crops Prod. 2021, 166, 113475. [Google Scholar] [CrossRef]
- Angin, D. Effect of pyrolysis temperature and heating rate on biochar obtained from pyrolysis of safflower seed press cake. Bioresour. Technol. 2013, 128, 593. [Google Scholar] [CrossRef]
- Fu, P.; Yi, W.; Bai, X.; Li, Z.; Hu, S.; Xiang, J. Effect of temperature on gas composition and char structural features of pyrolyzed agricultural residues. Bioresour. Technol. 2011, 102, 8211. [Google Scholar] [CrossRef]
- Al-Wabel, M.I.; Al-Omran, A.; El-Naggar, A.H.; Nadeem, M. Pyrolysis temperature induced changes in characteristics and chemical composition of biochar produced from conocarpus wastes. Bioresour. Technol. 2013, 131, 374. [Google Scholar] [CrossRef]
- Wang, S.; Gao, B.; Zimmerman, A.R.; Li, Y.; Ma, L.; Harris, W.G.; Migliaccio, K.W. Physicochemical and sorptive properties of biochars derived from woody and herbaceous biomass. Chemosphere 2015, 134, 257–262. [Google Scholar] [CrossRef]
- Uchimiya, M.; Wartelle, L.H.; Klasson, K.T.; Fortier, C.A.; Lima, I.M. Influence of pyrolysis temperature on biochar property and function as a heavy metal sorbent in soil. J. Agric. Food Chem. 2011, 59, 2501. [Google Scholar] [CrossRef] [PubMed]
- Mourgkogiannis, N.; Nikolopoulos, I.; Kordouli, E.; Lycourghiotis, A.; Kordulis, C.; Karapanagioti, H.K. The influence of biowaste type on the physicochemical and sorptive characteristics of corresponding biochar used as sustainable sorbent. Sustainability 2024, 16, 2889. [Google Scholar] [CrossRef]
- Đukanović, N.; Apostolović, T.; Anojčić, J.; Mutić, S.; Marjanović Srebro, T.; Kozma, G.; Deák, C.; Maletić, S.; Beljin, J. Comparative study of biochar from different biomass feedstocks: Toward sustainable resource utilization and environmental applications. Molecules 2026, 31, 37. [Google Scholar] [CrossRef]
- An, X.; Zhu, Z.; Luo, X.; Chen, C.; Liu, T.; Zou, L.; Li, S.; Liu, Y. Effects of raw materials and pyrolysis temperatures on physicochemical properties of biochars derived from hemp stalks. Plants 2025, 14, 2564. [Google Scholar] [CrossRef]
- Tripathi, M.; Sahu, J.N.; Ganesan, P. Effect of process parameters on production of biochar from biomass waste. Renew. Sustain. Energy Rev. 2016, 55, 467–481. [Google Scholar] [CrossRef]
- Neylon, E.; Arendt, E.K.; Lynch, K.M.; Zannini, E.; Bazzoli, P.; Monin, T.; Sahin, A.W. Rootlets, a Malting By-Product with Great Potential. Fermentation 2020, 6, 117. [Google Scholar] [CrossRef]
- Morais, E.G.; Silva, C.A.; Gao, S.; Melo, L.C.A.; Lago, B.C.; Teodoro, J.C.; Guilherme, L.R.G. Empirical correlation between electrical conductivity and nitrogen content in biochar as influenced by pyrolysis temperature. Nitrogen 2024, 5, 288–300. [Google Scholar] [CrossRef]
- Beljin, J.; Đukanović, N.; Anojčić, J.; Simetić, T.; Apostolović, T.; Mutić, S.; Maletić, S. Biochar in the remediation of organic pollutants in water: A review of polycyclic aromatic hydrocarbon and pesticide removal. Nanomaterials 2025, 15, 26. [Google Scholar] [CrossRef]
- Salama, A.-R.A.; El-Sahn, M.A.; Mesallam, A.S.; Shehata, A.M.E.-T. The chemical composition, the nutritive value and the functional properties of malt sprout and its components (Acrospires, Rootlets and Husks). J. Sci. Food Agric. 1979, 75, 50. [Google Scholar] [CrossRef]
- Bekatorou, A.; Kopsahelis, N.; Mallouchos, A.; Plessas, S.; Droushiotis, N.; Kanellaki, M.; Komaitis, M.; Koutinas, A.A.; Nigam, P. Biotechnological Exploitation of Brewery Solid Wastes; Current topics on Bioprocesses in Food Industry Volume III; Rao, L.V., Pandey, A., Larroche, C., Soccol, C.R., Dussap, C.G., Eds.; Asiatech Publishers Inc.: Delhi, India, 2010; pp. 193–209. [Google Scholar]
- Xu, R.K.; Xiao, S.C.; Yuan, J.H.; Zhao, A.Z. Adsorption of methyl violet from aqueous solutions by the biochars derived from crop residues. Bioresour. Technol. 2011, 102, 10293–10298. [Google Scholar] [CrossRef]
- Mukherjee, A.; Zimmerman, A.R.; Harris, W. Surface chemistry variations among a series of laboratory-produced biochars. Geoderma 2011, 163, 247–255. [Google Scholar] [CrossRef]
- EBC. European Biochar Certificate—Guidelines for a Sustainable Production of Biochar, Version 6.2E; European Biochar Foundation (EBC): Arbaz, Switzerland, 2012; Available online: http://www.european-biochar.org/en/download (accessed on 25 July 2025).
- Keiluweit, M.; Nico, P.S.; Johnson, M.G.; Kleber, M. Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environ. Sci. Technol. 2010, 44, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Santos, D.C.; Evaristo, R.B.; Dutra, R.C.; Suarez, P.A.; Silveira, E.A.; Ghesti, G.F. Advancing Biochar Applications: A Review of Production Processes, Analytical Methods, Decision Criteria, and Pathways for Scalability and Certification. Sustainability 2025, 17, 2685. [Google Scholar] [CrossRef]
- Tzachristas, A.; Manariotis, I.D.; Dailianis, S.; Karapanagioti, H.K. Decreasing Drinking Water Toxicity by Chlorine Removal Using Activated Carbons, Biomaterials and Biochars from Agro-industrial By-products. Water Air Soil Pollut. 2026, 237, 346. [Google Scholar] [CrossRef]
- Rambhatla, N.; Panicker, T.F.; Mishra, R.K.; Manjeshwar, S.K.; Sharma, A. Biomass pyrolysis for biochar production: Study of kinetics parameters and effect of temperature on biochar yield and its physicochemical properties. Results Eng. 2025, 25, 103679. [Google Scholar] [CrossRef]
- Sklivaniotis, L.N.; Economou, P.; Karapanagioti, H.K.; Manariotis, I.D. Chlorine Removal from Water by Biochar Derived from Various Food Waste Natural Mater. Environ. Proc. 2023, 10, 4. [Google Scholar] [CrossRef]
- Pantiora, D.; Karapanagioti, H.K.; Manariotis, I.D.; Lycourghiotis, A.; Kordulis, C. Evaluation of malt spent rootlets biochar as catalyst for biodiesel production. Geophys. Res. Abstr. 2014, 16, EGU2014–EGU6508. Available online: https://meetingorganizer.copernicus.org/EGU2014/EGU2014-6508.pdf (accessed on 8 January 2026).
- Tsouloufa, A.; Dailianis, S.; Karapanagioti, H.K.; Manariotis, I.D. Physicochemical and Toxicological Assay of Leachate from Malt Spent Rootlets Biochar. Bull. Environ. Contam. Toxicol. 2020, 104, 634–641. [Google Scholar] [CrossRef]
- Ntaflou, M.; Vakros, J. Transesterification activity of modified biochars from spent malt rootlets using triacetin. J. Clean. Prod. 2020, 259, 120931. [Google Scholar] [CrossRef]




| Characterization Method | Property Determined |
|---|---|
| Inorganic properties | |
| Moisture Content | Percentage of contained moisture |
| Ash Content | Percentage of inorganic constituents |
| Electrical Conductivity (EC) | Concentration and mobility of cations and anions in solution |
| pH Measurement | Determination of acidity/alkalinity |
| Carbon nature | |
| Total Carbon | Percentage of total carbon |
| Washing (Leaching Tests) | Easily extractable organic substances |
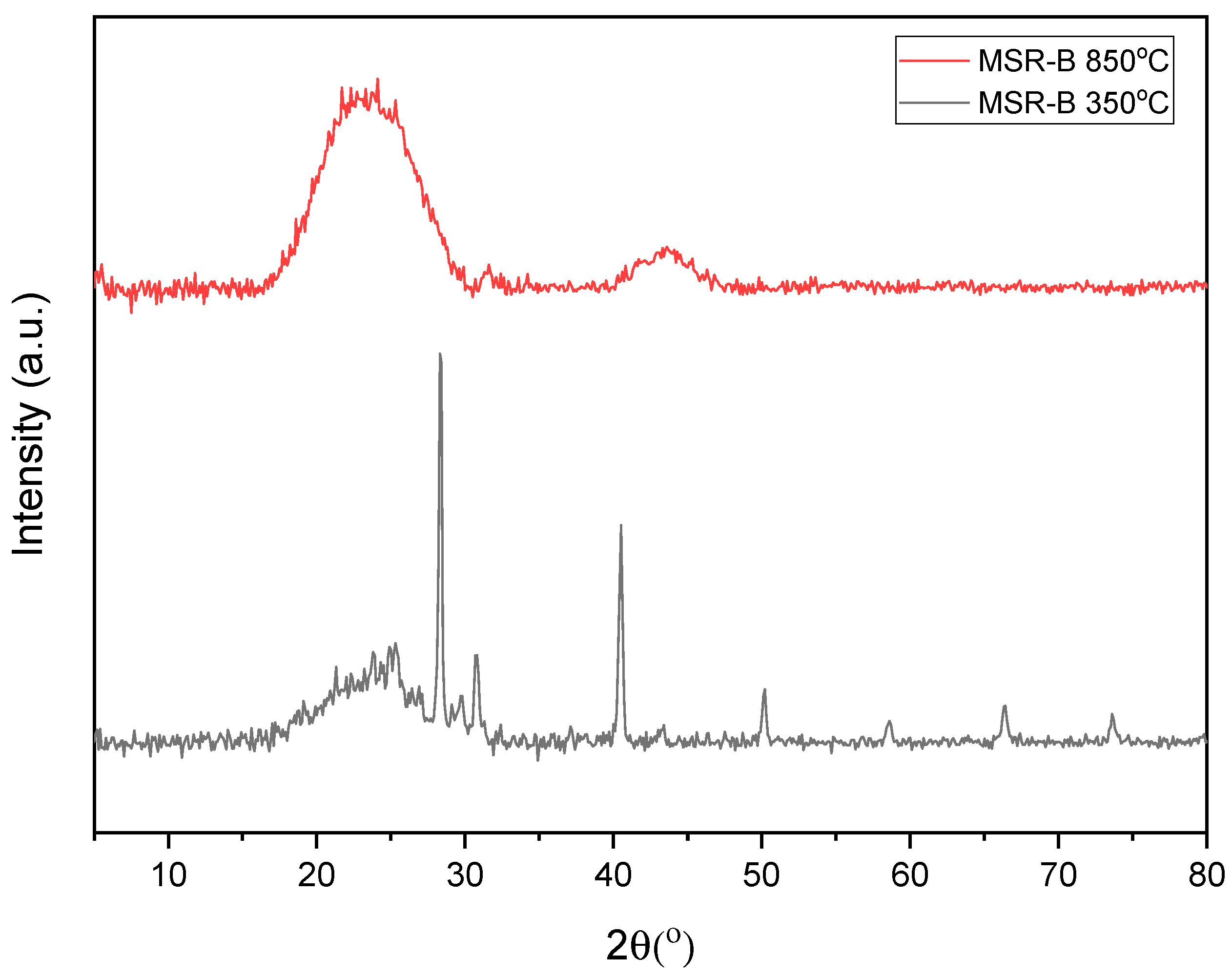
| XRD | Organic matter crystallinity |
| FTIR | Surface functional groups |
| Texture and sorption properties | |
| SEM | Surface texture |
| Surface Area and Porosity | Specific surface area |
| Methylene Blue Sorption | Sorption capacity of methylene blue |
| Molasses Number | Molasses concentration determination |
| Chlorine Removal Efficiency | Rate of chlorine removal in a column |
| Chlorine Removal Kinetics | Kinetics of chlorine removal in batch systems |
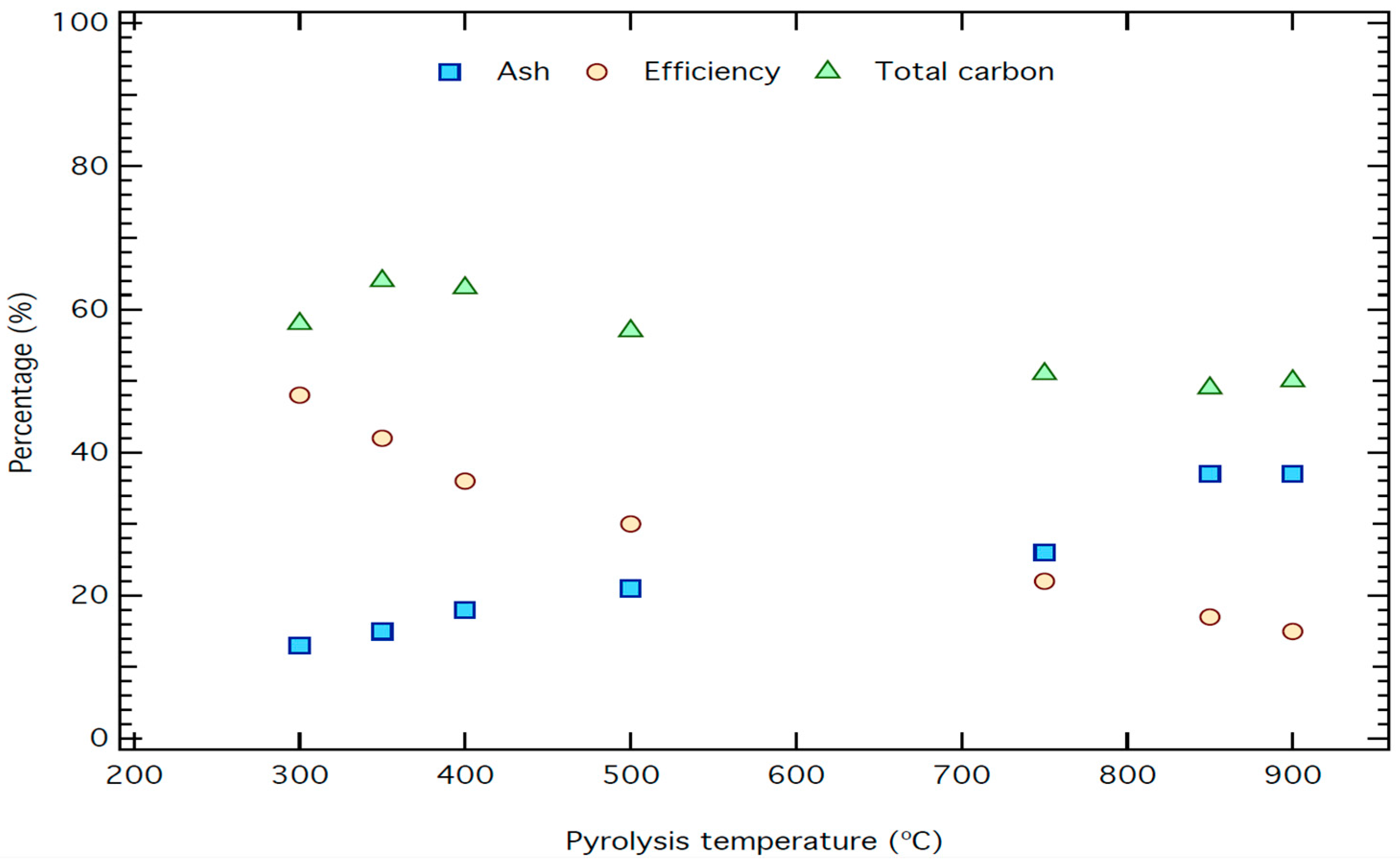
| T °C | Moisture (%) | Ash Content (%) | Efficiency (%) | Calculated Ash in the Raw Material (%) | Electrical Conductivity (μS/cm) | Total Carbon (%) |
|---|---|---|---|---|---|---|
| 300 | 6.1 | 13 | 48 | 6.2 | 800 | 58 |
| 350 | 6.1 | 15 | 42 | 6.3 | 900 | 64 |
| 400 | 6.9 | 18 | 36 | 6.5 | 1300 | 63 |
| 500 | 6.8 | 21 | 30 | 6.3 | 1100 | 57 |
| 750 | 9.4 | 26 | 22 | 6.2 | 1400 | 51 |
| 850 | 11 | 37 | 17 | 6.3 | 2600 | 49 |
| 900 | 12 | 37 | 15 | 5.6 | 2300 | 50 |
| T, °C | pH with 1D | pH with CaCl2 | pH with NaNO3 | |||
|---|---|---|---|---|---|---|
| After 1 h | After 1 day | After 1 h | After 1 day | After 1 h | After 1 day | |
| 300 | 7 | 7 | 7 | 7 | 7 | 7 |
| 350 | 7 | 7 | 7 | 7 | 7 | 7 |
| 400 | 7 | 7 | 7 | 7 | 7 | 7 |
| 500 | 9 | 9 | 9 | 9 | 8 | 8 |
| 750 | 10 | 10 | 10 | 10 | 8 | 8 |
| 850 | 10 | 10 | 10 | 10 | 9 | 9 |
| 900 | 10 | 10 | 10 | 10 | 9 | 9 |
| T, °C | ΒΕΤ (m2/g) | Pore Volume (cm3/g) | Average Pore Size (Å) | Methylene Blue | Molasses Number | ||
|---|---|---|---|---|---|---|---|
| Ce (mg/L) | qe (mg/g) | Ce (mg/L) | qe (mg/g) | ||||
| 300 | 0.50 ± 0.10 | 0.000 | 860 ± 9.2 | 12 | 79 | 600 | 3.8 |
| 350 | 0.73 | 0.008 | 911 | 9.5 | 90 | 570 | 9.2 |
| 400 | 3.4 | 0.016 ± 0.002 | 170 ± 15 | 5.7 | 110 | 580 | 7.7 |
| 500 | 5.5 | 0.002 | 750 | 8 | 96 | 530 | 18 |
| 750 | 167 | 0.15 ± 0.020 | 50 ± 2.1 | 13 | 130 | 530 | 18 |
| 850 | 230 | 0.18 ± 0.010 | 46 ± 1.7 | 9 | 150 | 440 | 35 |
| 900 | 246 | 0.16 ± 0.040 | 52 ± 7.1 | 3.4 | 190 | 320 | 58 |
| T °C | Chlorine Concentration in the Influent Solution (a) (mg/L) | Chlorine Concentration in the Effluent Solution (b) (mg/L) | HVL | 1st Order Rate Constant for the Granular Biochar (k) (1/min) | 1st Order Rate Constant for the Powder Biochar (k) (1/min) |
|---|---|---|---|---|---|
| 350 | 5 | 0.85 | 3.1 | 0.0006 | 0.0146 |
| 750 | 5 | 0.82 | 3.1 | 0.0008 | 0.0093 |
| 850 | 5 | 0.05 | 1.2 | 0.0009 | 0.0110 |
| 900 | 5 | 0.13 | 1.5 | 0.0010 | 0.0146 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tzachristas, A.; Natsi, P.D.; Politi, P.E.; Mourgkogiannis, N.; Manariotis, I.D.; Karapanagioti, H.K. Physicochemical Characterization of Biochar Sorbents Produced at Different Temperatures from Malt Spent Rootlets. Processes 2026, 14, 1012. https://doi.org/10.3390/pr14061012
Tzachristas A, Natsi PD, Politi PE, Mourgkogiannis N, Manariotis ID, Karapanagioti HK. Physicochemical Characterization of Biochar Sorbents Produced at Different Temperatures from Malt Spent Rootlets. Processes. 2026; 14(6):1012. https://doi.org/10.3390/pr14061012
Chicago/Turabian StyleTzachristas, Andreas, Panagiota D. Natsi, Panagiota E. Politi, Nikolaos Mourgkogiannis, Ioannis D. Manariotis, and Hrissi K. Karapanagioti. 2026. "Physicochemical Characterization of Biochar Sorbents Produced at Different Temperatures from Malt Spent Rootlets" Processes 14, no. 6: 1012. https://doi.org/10.3390/pr14061012
APA StyleTzachristas, A., Natsi, P. D., Politi, P. E., Mourgkogiannis, N., Manariotis, I. D., & Karapanagioti, H. K. (2026). Physicochemical Characterization of Biochar Sorbents Produced at Different Temperatures from Malt Spent Rootlets. Processes, 14(6), 1012. https://doi.org/10.3390/pr14061012









