1. Introduction
Applications of biomass gasification have recently received heightened attention due to its versatile conversion of renewable solid fuels into synthesis gas for heat, power, and fuel production. Biomass gasification presents various merits over direct combustion, such as high efficiency, flexible fuel, and the potential for hydrogen-rich syngas toward further chemical synthesis and energetic applications. However, tar formation, low hydrogen yield, and complexity in the process still present obstacles toward its generalization in practice.
Catalytic enhancement has long been recognized as a viable method for improving biomass gasification performance, including tar cracking and reforming of hydrocarbons, with the adjustment of syngas composition toward higher hydrogen content [
1,
2,
3]. A wide range of catalysts has been investigated to enhance biomass gasification reactions, including natural minerals such as dolomite, metal-supported catalysts (particularly Ni-based systems), and iron-containing materials [
4,
5,
6]. Many experimental studies have been conducted but, due to differences in the reactor configuration, operating conditions, and biomass feedstock type, it remains difficult to compare directly catalytic performances between studies.
To these drawbacks, thermodynamic equilibrium modeling has been extensively utilized as a theoretical approach to understand the behavior of the biomass gasification process and determine the primary limitations of the composition of syngas under specific conditions [
7,
8,
9]. The primary interests of the equilibrium-based models are to obtain a qualitative perspective of the impacts of the temperature, gasifying agents, and the composition of the feedstock while avoiding the complications of the reactor [
10,
11,
12].
Several authors make use of equilibrium modeling to examine influence factors for steam gasification and found that the high temperature and high steam-to-biomass ratio favored hydrogen production and inhibited methane formation [
13,
14,
15]. However, the specific consideration of catalytic effects within gasification modeling via equilibrium approach has not gained much popularity so far. Specifically, differences between catalyst types are often not taken into account, with catalytic effects either not included at all or implicitly assumed within ideal equilibrium conditions.
Recent modeling developments have tried to overcome this information gap with the introduction of correction factors or modified equilibrium constraints to reflect catalytic enhancement in the overall system of biomass gasification reaction systems [
2,
16,
17]. These representations enable the evaluation of the theoretical impact of reforming and shift reaction catalysts, avoiding the need for detailed simulation of kinetic surfaces, which may be specific to individual catalysts and reactors [
18,
19,
20].
In this context, this research puts forward a thermodynamic equilibrium-based modeling approach aiming to evaluate, in a theoretical manner, the effect of catalytic activity on biomass steam gasification performance. The catalytic effect is introduced through an activity-based correction method and is examined on key reforming and water–gas shift reactions, making possible a comparative evaluation of a variety of typically cited catalyst types such as dolomite, Ni Olivine, and iron-based catalysts, among others.
The objectives of this work are to (i) develop a consistent equilibrium modeling framework for catalytic biomass gasification, (ii) quantify the influence of catalyst activity on syngas composition under steam gasification conditions, and (iii) provide a theoretical basis for catalyst screening and process optimization at the conceptual design level. The outcomes of this study aim to complement experimental research by clarifying fundamental thermodynamic trends and supporting the rational development of catalytic biomass gasification systems. While equilibrium-based models and catalytic gasification trends are well-established in the literature, most existing studies either assume ideal equilibrium without explicitly differentiating catalyst types or focus on detailed kinetic descriptions that are strongly reactor- and catalyst-specific. The novelty of the present work lies in the systematic incorporation of catalyst-dependent activity coefficients into a Gibbs free energy minimization framework, enabling a unified and comparative assessment of different catalyst classes under identical thermodynamic conditions. This approach provides a practical screening-level tool for catalyst selection and process conceptual design, rather than aiming at exact prediction of experimental outcomes.
Unlike conventional equilibrium-based gasification models that primarily report absolute syngas compositions or single-parameter trends, the present study introduces a catalyst activity-normalized equilibrium framework that enables systematic and internally consistent differentiation among catalyst classes within a unified thermodynamic structure. By embedding catalyst-dependent activity corrections directly into the Gibbs energy minimization formulation, the proposed approach allows the comparative assessment of reforming effectiveness across catalysts under identical operating conditions, thereby offering a novel methodological bridge between ideal equilibrium limits and catalyst oriented process screening.
2. Materials and Methods
2.1. Process Description and Modeling Framework
Biomass gasification represents a complex thermochemical conversion, involving drying, devolatilization, heterogeneous gas–solid reactions, and homogeneous gas-phase reactions. In the present work, biomass gasification is analyzed using a thermodynamic equilibrium-based modeling approach. This approach enables a systematic theoretical evaluation of the influence of catalytic enhancement on syngas composition under steam gasification conditions.
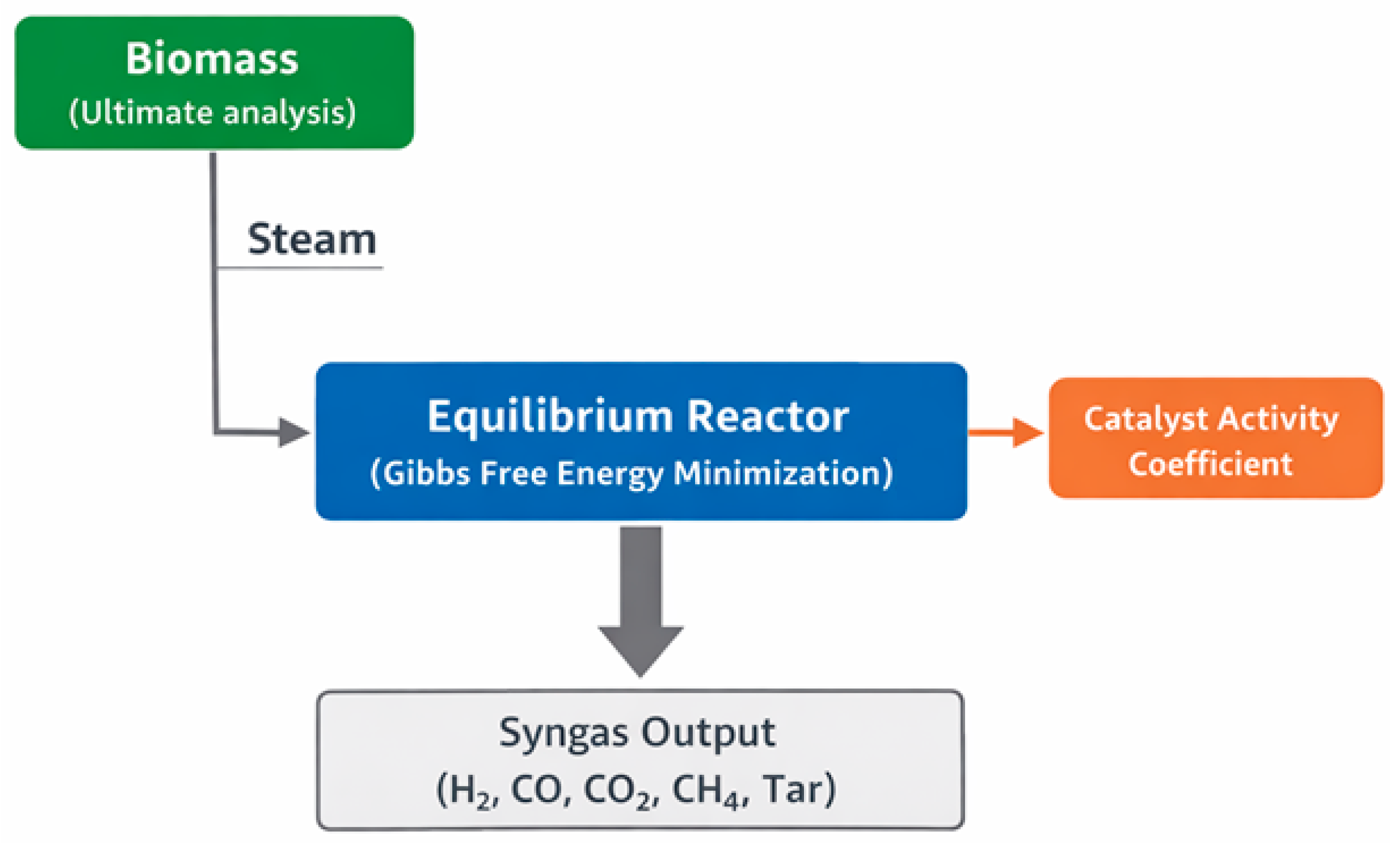
The overall modeling framework developed in this work is schematically illustrated in
Figure 1. The model represents an idealized gasification system in which the biomass, characterized by its elemental composition, is introduced together with steam into an equilibrium reactor. The reactor formulation is based on the minimization of the total Gibbs free energy of the reacting system, assuming thermodynamic equilibrium among the major gaseous species formed during gasification.
The catalytic effects are also implemented in the modeling framework using the activity-based correction approach. Instead of accounting for the surface kinetics associated with catalysts, this approach is based on the adjustment of the equilibrium tendency of specific gas-phase reactions. The approach targets tar reforming reactions, methane steam reforming reactions, and the water–gas shift reaction. This approach is analogous to other equilibrium-based catalytic gasification models reported in the literature [
1,
2,
16].
The model simulates the equilibrium molar composition of the producer gas, including H2, CO, CO2, CH4, and light hydrocarbons, as a function of the operating temperature, steam-to-biomass ratio, and catalyst activity. Solid phases such as char and ash are assumed to be inert and are therefore excluded from the equilibrium calculations.
It should be emphasized that the present model does not represent a true thermodynamic equilibrium in the strictest sense, in which catalyst presence would not alter the final equilibrium state. Instead, the adopted formulation constitutes a constrained or pseudo-equilibrium framework, in which catalyst-dependent activity coefficients are introduced to selectively modify the effective extent of specific reactions. This approach does not imply a change in fundamental thermodynamic equilibria but rather provides a heuristic representation of how catalytic enhancement may shift the apparent equilibrium composition under practical gasification conditions where full equilibrium is not attained.
2.2. Biomass Feedstock Characterization
Pine wood sawdust is selected as a representative biomass due to its particle homogeneity, low ash content, and widespread use in biomass gasification modeling studies [
21,
22].
In the present investigation, no experimental characterization of biomass is conducted. Instead, the physicochemical properties of biomass are adopted from the referenced experimental studies and standardized literature sources.
The proximate and ultimate analysis values used in the model are obtained from various literature sources in which standardized ASTM-based procedures have been followed, and these values are considered benchmark data applicable for simulating the biomass gasification process [
7]. The elemental composition from the ultimate analysis values is normalized to the dry basis and considered the input to the thermodynamic equilibrium model. It should be noted that proximate analysis data are reported on an as-received basis with moisture content listed separately, whereas the ultimate analysis used for model input is normalized on a dry basis.
Table 1 summarizes the biomass properties adopted in this study for modeling purposes.
2.3. Catalyst Representation and Functional Role in the Model
To examine the influence of catalytic enhancement on the performance of the biomass gasification process, three different catalysts are considered. The catalysts are selected as representative examples of those reported in the literature, including a natural mineral catalyst like dolomite, a catalyst supported by metal like Ni/olivine, and a composite iron-based catalyst [
2,
5,
6].
The selection of dolomite, Ni/olivine, and Fe-based catalysts is motivated by their representative roles in biomass gasification research rather than by their specific formulation. These catalyst types collectively represent three widely studied catalytic classes in biomass gasification: natural mineral catalysts (dolomite), metal-supported catalysts with high reforming activity (Ni-based systems), and transition metal-based catalysts (iron-based materials). Dolomite is commonly used as a low-cost reference catalyst with moderate tar cracking capability, Ni-based catalysts are well-known for their superior steam reforming and hydrogen production performance, and iron-based catalysts provide an intermediate level of activity with enhanced stability and water–gas shift contribution. This representative selection enables systematic and comparative assessment of catalytic effects across different catalyst classes within a unified thermodynamic framework.
Instead of simulating catalyst preparation and surface reaction kinetics, the influence of catalysts is incorporated via relative catalytic activity coefficients for specific chemical reactions in the equilibrium model. These coefficients account for the relative effectiveness of the various types of catalysts for tar reforming, methane steam reforming, and water–gas shifting.
The selected catalyst types, including their reported properties in the corresponding literature and functional representation in the model, are presented in
Table 2. The purpose of the activity-based correction is not to reproduce catalyst-specific kinetics, but rather to provide a relative thermodynamic ranking of catalyst effectiveness under identical operating conditions, which is particularly useful at early-stage process design and catalyst screening.
With respect to iron-based catalysts, their contribution to biomass gasification is not primarily associated with direct hydrocarbon steam reforming activity comparable to Ni-based systems (Ni0: zero oxidation state nickel). Instead, Fe2O3-based materials are known to promote tar conversion through redox-mediated cracking mechanisms and to enhance hydrogen production indirectly via their well-established activity toward the water–gas shift reaction. The Fe3+/Fe2+ redox cycle facilitates the breakdown of complex hydrocarbon structures and suppresses coke formation, while simultaneously favoring CO conversion to H2. As a result, iron-based catalysts typically exhibit an intermediate reforming effectiveness between natural mineral catalysts and highly active Ni-based systems, which is consistently reflected in the assigned activity coefficients used in the present equilibrium model.
The use of activity coefficients to represent the catalytic effects constitutes a deliberate modeling simplification. This approach does not aim to resolve detailed catalytic mechanisms, reaction pathways, transport limitations, or catalyst deactivation phenomena, which are known to require kinetic- or reactor-scale descriptions. Instead, the present formulation focuses on capturing the relative thermodynamic influence of different catalysts on the equilibrium composition under identical operating conditions. Such simplification is intentionally adopted to preserve model generality and computational efficiency, particularly for early-stage process analysis and catalyst screening purposes.
In contrast to conventional equilibrium models where catalytic effects are often implicitly assumed or qualitatively discussed, the present formulation explicitly enforces catalyst differentiation through reaction-specific activity normalization. This enables direct, parameter consistent comparisons of catalyst classes within a single equilibrium framework, which is not commonly addressed in existing equilibrium-based gasification studies.
The catalyst activity coefficients used in this study are dimensionless scaling parameters introduced to represent the relative effectiveness of different catalyst classes in promoting reforming and water–gas shift reactions within an equilibrium framework. These values are not derived from kinetic rate constants or surface reaction models but are assigned based on the consistent qualitative ranking of catalytic performance, as widely reported in the biomass gasification literature. Specifically, Ni-based catalysts are commonly identified as the most active systems for tar and methane reforming, mineral catalysts such as dolomite exhibit lower activity, and iron-based materials display intermediate reforming and shift activity. Accordingly, the activity coefficient of Ni/olivine is normalized to unity (α = 1.0), while relative values of 0.6 and 0.4 are assigned to Fe-based and dolomite catalysts, respectively, to preserve this established performance hierarchy under identical thermodynamic conditions.
2.4. Thermodynamic Modeling of Catalytic Biomass Gasification
A model of thermodynamic equilibrium in a state of steady-state, as based on thermodynamics, is used to simulate a catalytic biomass gasification process and to evaluate theoretically the influence of catalytic activity on syngas composition. The thermodynamic model based on the minimization of Gibbs free energy is an established practice that can be employed for simulating gas composition in a gasification system, including the influence of process parameters [
10,
11].
In the model, the chemical equilibrium assumption is made for the main gaseous species: H
2, CO, CO
2, CH
4, and H
2O. The solid char is assumed to be pure carbon. The biomass is modeled based on elemental composition using the composition from the ultimate analysis data provided in
Table 1. Tar is modeled as a pseudo-component with a corresponding empirical formula of C
6H
6; this is in compliance with conventional equilibrium modeling practices. In the present equilibrium framework, tar is represented as a single pseudo-component to enable tractable thermodynamic analysis of its overall reforming tendency. This simplification is intentionally adopted to support the comparative assessment of catalytic effects rather than the detailed resolution of individual tar species or reaction pathways.
The gasification process is described using the following principal reactions listed below:
Steam reforming of hydrocarbons:
Water–gas shift reaction:
Reaction equilibrium constants are calculated as a function of temperature using Gibbs free energy data. The model is implemented in MATLAB
® R2021a (The MathWorks Inc., Natick, MA, USA) and solved by minimizing the total Gibbs free energy of the system subject to elemental mass balance constraints [
23].
Catalytic enhancement is included using activity coefficients for specific reforming and shift reactions. The activity coefficients are higher in the case of Ni/olivine, intermediate for Fe-based catalysts, and lower for dolomite, following the trends in the literature [
5,
6].
2.5. Model Implementation in MATLAB
The thermodynamic equilibrium model is implemented in MATLAB using a Gibbs free energy minimization approach. The equilibrium composition of the gasification products is determined by minimizing the total Gibbs free energy of the reacting system subject to elemental mass balance constraints.
The total Gibbs free energy of the system is expressed as:
where
is the number of moles of species
i;
is the standard Gibbs free energy of formation of species
i;
R is the universal gas constant; and
T is the absolute temperature.
Elemental mass balance constraints for carbon, hydrogen, oxygen, and nitrogen are imposed as:
where
represents the number of atoms of element
j in species
i, and
denotes the total number of moles of element
j introduced into the system through biomass and gasifying agents.
Catalytic enhancement is incorporated through activity correction factors applied to selected reforming reactions. These factors modify the effective equilibrium tendency of methane steam reforming and tar reforming reactions without explicitly modeling surface kinetics:
where
is the catalyst activity coefficient and
is the thermodynamic equilibrium constant of the corresponding reaction.
In this formulation, the activity coefficient acts as a heuristic modifier of the equilibrium constant, representing the extent to which catalytic enhancement promotes an approach toward the thermodynamic equilibrium of selected reforming reactions, rather than altering the equilibrium state itself.
The resulting constrained nonlinear optimization problem is solved using MATLAB’s built-in numerical solvers. Convergence is achieved when the elemental balances are satisfied and the total Gibbs free energy reaches a minimum within a specified tolerance.
2.6. Operating Conditions and Model Input Parameters
The simulations have been conducted under operating conditions that are representative of laboratory scale and pilot scale biomass gasification systems reported in the literature. The temperature for the gasification process is fixed at 800 °C, which is a temperature that is always cited as being conducive for hydrogen production as well as the reforming of tar under steam gasification conditions [
7,
13]. The pressure for the process is held constant at atmospheric pressure for all the processes.
Steam is employed as the main gasifying medium, and the steam-to-biomass ratio is fixed at 0.6. The influence of the catalyst is accounted for through the dimensionless activity coefficient, rather than the catalyst loading itself; this is typical practice in equilibrium calculation approaches. A summary of the major input parameters to the model, as adopted in the present work, is given in
Table 3.
2.7. Model Assumptions and Input Parameters
The thermodynamic equilibrium model developed in this study is based on a set of assumptions used to enable computational efficiency, as well as the dominant physicochemical characteristics of the biomass gasification process. The assumptions are in line with other studies on equilibrium-based modeling of the gasification process [
11,
24,
25].
The reactor is assumed to be a steady-state, isothermal system, operating at atmospheric pressure. The gas is assumed to be an ideal gas mixture, and heat losses are neglected.
Biomass feedstock is considered homogeneous and is typically represented only in terms of its ultimate analysis, normalized on a dry, ash-free basis, while ash is assumed inert and does not take part in any chemical reaction.
Chemical equilibrium in all the major gasification reactions, i.e., char gasification, tar reforming, steam reforming, and water-gas shift, is assumed. The catalytic effect is accounted for through activity coefficients; these are applied to some of the reactions, facilitating comparison of different catalysts without considering the kinetics of reaction or mass transfer. Accordingly, catalyst activity coefficients should be interpreted as relative performance indicators rather than physically measurable kinetic parameters. Catalyst stability, deactivation, and long-term performance are not explicitly evaluated in this study, as such effects are inherently time-dependent and require kinetic or experimental descriptions beyond the scope of the present equilibrium-based modeling framework.
Accordingly, the model should be interpreted as a heuristic, screening-level pseudo-equilibrium tool rather than a representation of true thermodynamic equilibrium, with its primary purpose being the comparative assessment of catalyst classes under identical boundary conditions.
Uncertainty associated with biomass composition variability is not explicitly quantified in a statistical sense, as the present study is based on the reported benchmark compositions in the literature and is intended to enable screening-level thermodynamic analysis. However, the influence of key operating parameters and catalyst activity on syngas composition is systematically investigated through sensitivity analysis, which implicitly captures the robustness of predicted trends against compositional and operational variability commonly reported in biomass gasification studies.
2.8. Model Consistency and Literature-Based Benchmarking
Consistency of the developed thermodynamic equilibrium model is assured in terms of both internal consistency checks as well as comparisons of the predicted trends with the ranges found in the open literature. In this work, since the main aim is to theoretically explore the impact of catalyst activity, no attempt has been made towards direct experimental validation.
The predicted trends in syngas composition with respect to the temperature and steam-to-biomass ratio are compared to the reported results of biomass gasification systems under similar conditions [
7,
11,
25].
The model captures the commonly known thermodynamic trends, such as the increase of hydrogen yield and the decrease in methane concentration during gasification at high temperatures.
In addition, the impact of the catalyst activity coefficients with respect to tar reforming reactions and water–gas shift reactions are investigated and compared to the published experimental and modeling research [
4,
5,
24]. The consistency of the predicted trends compared to the reported literature confirms the developed framework as a suitable tool for catalyst performance evaluation.
While the consistency of the predicted trends with the literature data supports the validity of the developed framework for thermodynamic analysis and comparative catalyst assessment, the model assumes equilibrium conditions modified by catalyst-dependent activity corrections and therefore does not explicitly account for reaction kinetics, mass and heat transfer limitations, or catalyst aging and deactivation effects.
Due to the conceptual and screening-oriented nature of the present study, no direct quantitative validation against a specific experimental dataset has been conducted. Instead, the model consistency is assessed through systematic comparison of predicted syngas composition trends and parameter sensitivities with widely reported equilibrium, as well as the experimental ranges available in the literature for biomass steam gasification systems operating under comparable conditions.
2.9. Quantitative Literature-Based Validation
To provide a quantitative reference for evaluating the reliability and practical relevance of the proposed pseudo-equilibrium modeling framework, a limited validation exercise has been conducted using experimental data available in the open literature. Given the screening-level nature of the present model and its intended role as a thermodynamic trend analysis tool, the validation is designed to benchmark equilibrium-based predictions against representative experimental results reported under comparable operating conditions, rather than to achieve pointwise predictive accuracy.
Experimental data are selected from published biomass steam gasification studies conducted at atmospheric pressure and gasification temperatures close to 800 °C, using woody biomass feedstocks and steam as the primary gasifying agent. In particular, the experimental results reported by Lv et al. [
13] and Gil et al. [
14] have been identified as suitable benchmark cases, as they provide detailed dry syngas compositions under conditions comparable to those simulated in the present study.
Table 4 presents a quantitative comparison between the predicted syngas composition obtained from the equilibrium model (Ni-based catalyst activity coefficient α = 1.0) and the experimentally reported values from the selected literature sources. The comparison focuses on the major gas components (H
2, CO, CH
4), which are the primary indicators of reforming efficiency and gas quality in biomass steam gasification systems.
The results indicate that the equilibrium-based model systematically overpredicts hydrogen and carbon monoxide concentrations while underpredicting methane content relative to experimental observations. This behavior is consistent with the well-documented tendency of equilibrium models to overestimate the reforming extent due to the absence of kinetic, mass transfer, and residence time limitations. Importantly, the magnitude of deviation observed (typically within 10–30% for major components) is comparable to that reported in previous assessments of equilibrium gasification models and remains acceptable for screening-level analysis.
These results provide a quantitative reference for interpreting the predictions of the proposed model. While the model does not aim to reproduce exact experimental gas compositions, the validation confirms that it captures the correct order of magnitude and relative distribution of major syngas components under practical operating conditions. Accordingly, the proposed framework offers a reliable thermodynamic baseline for comparative catalyst screening and early-stage process evaluation, while recognizing that detailed reactor scale predictions require kinetic or non-equilibrium modeling approaches.
3. Results and Discussion
3.1. Model Output Structure and Evaluation Metrics
The thermodynamic equilibrium model established in this study calculated the equilibrium molar fractions of the primary composition gases in the syngas (H2, CO, CO2, CH4, and H2O) as a function of the process temperature, the steam-to-biomass ratio, and the catalyst activity coefficient. In order to make the results consistent in their interpretation, the report only considered the composition of the syngas as a dry and nitrogen-free basis mixture. The hydrogen yield and the H2/CO ratio of the gases, as well as the suppression of methane content in the gases, were used as the primary basis for the model’s efficiency with reference to other studies regarding the evaluation of the efficiency of the gasification process.
Figure 2 below shows how the general structure of the model outputs and the considered parametric dependencies look in this research. The choice of evaluation criteria facilitated direct comparison with the equilibrium trends and ranges commonly found in the literature [
7,
14,
26].
3.2. Effect of Gasification Temperature on Syngas Composition
Figure 3 illustrates the effect of gasification temperature on the composition of the products formed during the steam gasification process. The range considered for the temperature in the model varies from 700 to 900 °C, conducted at atmospheric pressure with a constant steam-to-biomass ratio.
It was observed that as the gasification temperature was increased, the hydrogen concentration showed an increasing trend. This was largely due to the endothermic nature of char gasification and steam reforming reactions, which were favored at higher temperatures. The high conversion rates of hydrocarbon compounds at higher temperatures also contributed towards the production of hydrogen.
Carbon monoxide concentration followed a moderate increase pattern with temperature, reflecting the dominance of the Boudouard reaction and steam–carbon reaction at high thermal levels. However, the concentration of methane decreased significantly with an increase in temperature, reflecting the suppression of methanation reaction and the enhancement of the reforming reaction of methane.
The trend in the carbon dioxide concentration showed a tendency to slightly decrease as the temperature increased. This trend was consistent with the thermodynamic shift in the water–gas reaction toward hydrogen and carbon monoxide formation at high temperatures. It should be noted that the monotonic increase in hydrogen concentration observed in
Figure 3 reflects the investigated temperature range (700–900 °C), within which endothermic reforming and water–gas shift reactions were thermodynamically favored under equilibrium assumptions. At substantially higher temperatures (>1000 °C), non-monotonic hydrogen behavior may have arose due to reverse reforming reactions and thermal dissociation effects, which were not explicitly resolved within the present equilibrium framework. Accordingly, the selected temperature range was intentionally limited to conditions commonly reported for practical biomass steam gasification, where equilibrium-based trends remained physically meaningful and experimentally relevant.
In conclusion, the overall outcome from the outcomes obtained in
Figure 3 shows the significant influence that gasification temperature played in the determination of syngas composition, thereby emphasizing the increases in hydrogen-rich syngas while inhibiting methane production. The results obtained were in accordance with the equilibrium-based modeling studies for the thermodynamic trends.
3.3. Effect of Steam-to-Biomass Ratio on Syngas Composition
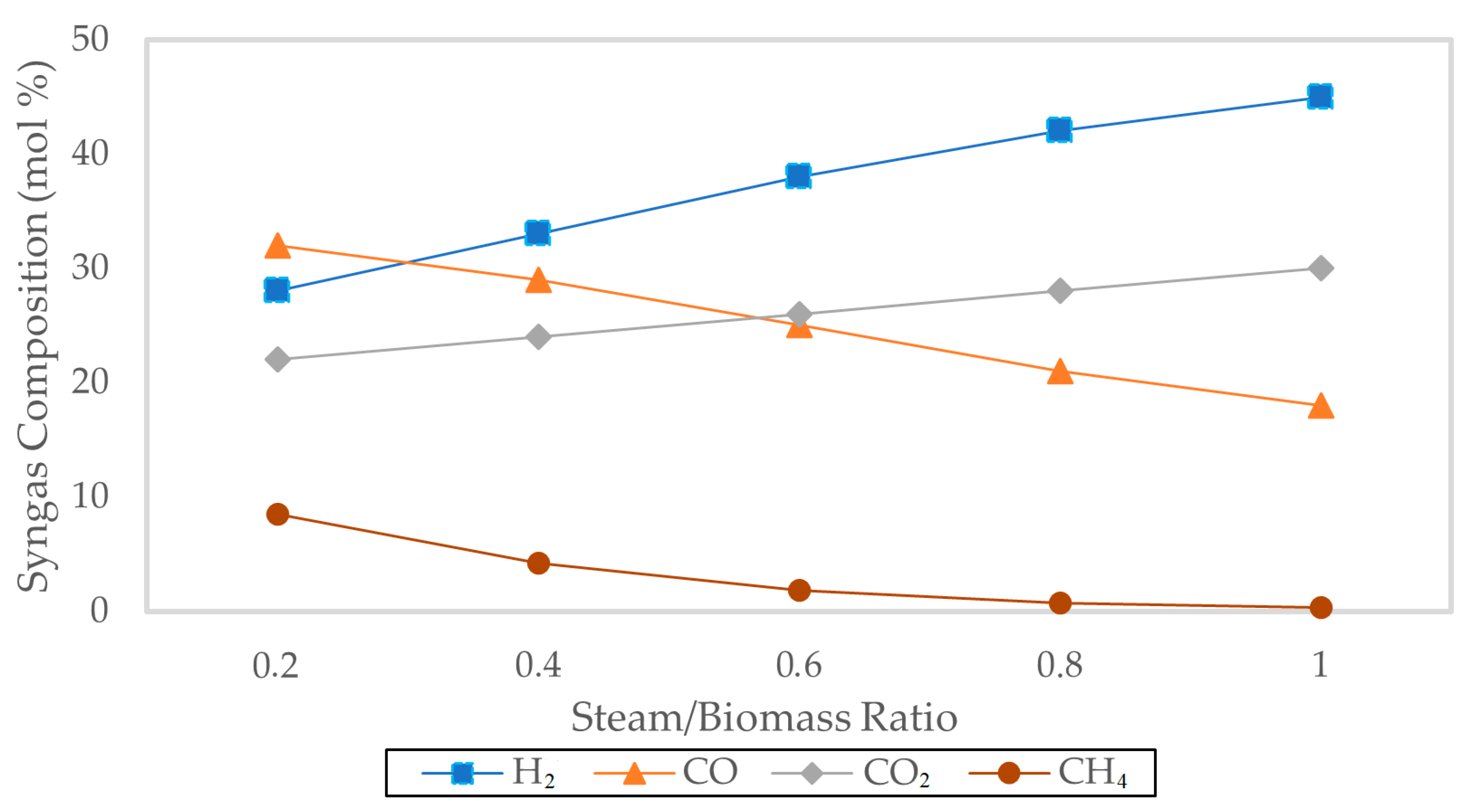
The effect of steam-to-biomass ratio (S/B) on composition is shown in
Figure 4, which illustrates an increase in hydrogen concentrations while carbon monoxide concentrations decreased with an increase in S/B ratio from 0.3 to 0.9.
This trend was mainly attributed to the catalysis of steam reforming and the water–gas shift reaction, in which steam was consumed to favor the production of hydrogen. However, high rates of steam addition led to a dilution effect and reduced thermal efficiency, a phenomenon also reported in previous modeling and experimental studies.
The model successfully reproduced the nonlinear dependence of hydrogen yield on steam availability, highlighting the existence of an optimal S/B range for maximizing hydrogen production under equilibrium conditions.
3.4. Effect of Catalyst Activity on Product Gas Distribution
Catalytic enhancement was formulated and incorporated in the model via a dimensionless activity coefficient for specific reforming and shift reactions.
Figure 5 depicts a comparison of the equilibrium composition for the syngas stream as calculated for low, intermediate, and high activity coefficients representative of dolomite, Fe-based, and Ni-based catalytic systems, respectively.
The rise in the catalytic activity was invariably followed by an increase in hydrogen content along with a decrease in methane content. The increase in the catalytic activity was due to the increased extent of tar reforming and methane steam reforming reactions, which were thermodynamically favored in the presence of very active catalysts, especially the nickel-based catalysts [
2,
5,
19].
The observed changes in the concentrations of carbon monoxide and carbon dioxide were an indication of the cumulative effects of increased reformer reaction kinetics, and the redistribution of the carbon-containing species in the water–gas shift equilibrium, as hydrogen production increases.
It should be clarified that the different equilibrium syngas compositions predicted in
Figure 5 do not represent multiple true thermodynamic equilibrium states for identical initial conditions. Instead, they reflect catalyst modified apparent equilibrium states resulting from the introduction of activity coefficients into selected reforming and shift reactions. In the absence of catalytic effects, a unique thermodynamic equilibrium composition would be obtained for a given set of boundary conditions. However, in the present framework, catalyst activity coefficients intentionally altered the effective equilibrium tendencies of specific reactions in order to represent the relative reforming performance of different catalyst classes. Accordingly, the model did not seek to identify a single “true” equilibrium composition, but rather to enable a consistent comparative assessment of how different catalysts shifted the apparent equilibrium behavior under otherwise identical operating conditions.
Although the present model did not include any explicit consideration for the reaction kinetics on catalyst surface, the predicted trends were indeed in agreement with several experimental studies reported on catalytic biomass gasification systems, in which Ni-based catalysts exhibited superior hydrogen yield and effective suppression of methane and tars compared to mineral-based catalysts [
4,
5,
6].
3.5. Effect of Equivalence Ratio on Syngas Composition
The equivalence ratio (ER) is an essential operating variable that controls the oxidation reaction during biomass gasification and thus, the composition and quality of the resultant syngas.
Figure 6 shows the effect of ER on the equilibrium syngas composition resulting from the catalytic steam gasification.
As the value of the equivalence ratio rose, there was a corresponding reduction in the concentration of hydrogen. This was a result of the increased oxidation reactions, since these reactions resulted in the consumption of combustible materials. The same was true for the trend in carbon monoxide concentrations, although this time the compound underwent a transformation into carbon dioxide.
Contrary to this, the concentration of carbon dioxide increased with an increase in ER levels, pointing to the significant rates of partial combustion at high ER levels. The concentration of methane decreased further at high ER levels due to the combined effects of high temperatures and oxidative cracking.
In summary, the results presented above demonstrated the necessity for a certain amount of oxidizing agents as far as sustaining an autothermal state was required, while too high a level of equivalence ratios decreased the concentration of both hydrogen and carbon monoxide in the syngas generated from the catalytic gasification process.
3.6. Methane Suppression and Tar Reforming Implications
Methane concentration can be regarded as an indirect measure of the efficiency of the reforming process in biomass gasification systems.
Figure 7 demonstrates the relationship, as predicted, between the catalyst activity coefficient and the temperature.
The model also predicted a strong suppression of methane at higher temperatures and higher catalytic activity levels, which was a sign that methane steam reforming and cracking reactions were thermodynamically favored processes [
2,
19]. Although tar was modeled as a pseudo-component, this decrease in methane concentration indicated a reinforcing effect, which agreed with experimentally observed effects on tar yield for catalytic gasification processes [
4,
5].
It should be emphasized that, within the present equilibrium-based framework, tar was represented as a single pseudo-component with an empirical formula of C
6H
6 in order to capture the overall thermodynamic tendency of tar reforming, rather than the detailed chemistry of real tar mixtures. Accordingly, the term “tar conversion efficiency” used in
Figure 7 does not refer to a kinetically defined conversion of individual tar species. Instead, it was defined as the relative reduction in the equilibrium molar fraction of the tar pseudo-component with increasing catalyst activity under otherwise identical operating conditions. This metric was introduced as a comparative thermodynamic indicator of tar reforming potential, enabling consistent assessment of the influence of catalyst activity within a screening-level pseudo-equilibrium model. While real gasification tars consist of a wide spectrum of polyaromatic hydrocarbons and oxygenated compounds, resolving such complexity would require detailed reaction networks and kinetic descriptions that were beyond the scope of the present study.
These results highlighted the importance of catalytic enhancement in improving gas quality and reducing downstream gas cleaning requirements.
3.7. Comparison with Literature Equilibrium Trends
The predicted syngas compositions and parametric trends obtained in this study were compared with representative ranges reported in the literature for biomass steam gasification under comparable operating conditions.
Table 5 shows a summary of comparisons for predictions and literature data.
It should be clarified that the literature ranges referenced in
Table 4 are not derived from a single unified dataset, but represent consolidated trends reported across multiple experimental and modeling studies on biomass steam gasification operating under broadly comparable conditions. These studies differed in reactor configuration, feedstock properties, catalyst formulation, and operating parameters, which precluded direct statistical aggregation or pointwise quantitative error analysis. Accordingly,
Table 4 is intended to provide a representative trend-level comparison with the established literature behavior, rather than a statistically rigorous meta-analysis. The cited references [
7,
11,
24,
25,
26] have collectively documented consistent directional sensitivities of syngas composition to temperature, steam-to-biomass ratio, pressure, and catalytic effects, which formed the qualitative benchmark against which the present model has been evaluated.
In this context, the qualitative sensitivity descriptors reported in
Table 4 (i.e., “weak”, “moderate”, and “strong”) were based on the relative magnitude of variation observed in key syngas components (H
2, CO, CH
4, and CO
2) in response to systematic parameter changes within the investigated ranges. These classifications reflected comparative trend intensities rather than statistically derived metrics, and were intended to support qualitative interpretation of thermodynamic sensitivities in a screening-level equilibrium framework.
It should be emphasized that the reduction in tar formation in this study was interpreted at a global thermodynamic level, primarily through the suppression of light hydrocarbons such as methane under enhanced reforming conditions. While more detailed approaches considering representative classes of tar compounds have been reported in the literature, such formulations typically required extended reaction networks and additional physicochemical assumptions. Within the scope of the present equilibrium-based screening study, the pseudo-component representation of tar provided a consistent and computationally efficient means of evaluating relative catalytic reforming performance.
The developed model has realistic potential in terms of accurately describing certain thermodynamic trends, such as increasing hydrogen content with the increase in temperature and the amount of steam added, the decrease in methane content, and the enhanced reforming characteristics in the presence of catalysts; however, certain absolute values might differ from those obtained in specific experimental setups, particularly owing to kinetic limitations [
7,
11,
26].
Unlike previous equilibrium modeling studies that reported absolute syngas compositions, the present work focused on the relative sensitivity of syngas composition to catalyst activity within a unified thermodynamic framework. This distinction allowed the present study to move beyond descriptive equilibrium predictions by systematically isolating the thermodynamic contribution of catalyst activity itself, independent of reactor-specific kinetic or transport constraints. By normalizing catalyst effects through activity coefficients, the model highlighted how catalyst selection influenced hydrogen enhancement and methane suppression trends independently of reactor-specific kinetic limitations. This comparative, thermodynamically consistent perspective could represent a key scientific contribution from the study.
Recent advances in biomass gasification modeling have increasingly relied on hybrid thermo-kinetic and non-equilibrium approaches to describe detailed reaction mechanisms, transport phenomena, and catalyst deactivation behavior. While such models can provide high fidelity predictions for specific reactor configurations, they often required extensive kinetic datasets and system specific calibration. In contrast, the equilibrium-based framework adopted in this study prioritized generality and comparability across catalyst types, offering a complementary perspective that was particularly suited for preliminary catalyst assessment and conceptual process design.
Accordingly, the validation strategy adopted in this work focused primarily on thermodynamic consistency and trend-level agreement with the established literature rather than pointwise quantitative error minimization against individual experimental datasets.
4. Conclusions
In this study, a modeling framework based on the concept of thermodynamic equilibrium has been established to extensively study the implications of catalytic enhancement in the process of biomass steam gasification. The model was programmed in MATLAB through the minimization of the Gibbs function to evaluate the corresponding composition of the resulting syngas as a function of key parameters, such as the gasification temperature, the steam-to-biomass ratio, the equivalence ratio, and the activity coefficient for the catalysts involved. This model highlighted the flexibility and efficiency of the catalytic enhancement factor approach without the necessity of selecting a kinetic model for the evaluation process.
The results of such modeling have confirmed that the gasification temperature and the steam-to-biomass ratio have a prominent effect on the composition of syngas produced. Increasing the gasification temperature favored the generation of hydrogen, while it inhibited the generation of methane due to the greater thermodynamic feasibility of reforming and cracking reactions at higher temperatures. In similar fashion, an increased value of the steam-to-biomass ratio favored the generation of hydrogen through enhanced levels of steam reforming reactions, albeit excess amounts of steam may have undesirable effects on overall process efficiency.
Catalytic activity was found to be of significant relevance in enhancing syngas quality, wherein hydrogen concentration was increased, and methane was comparably inhibited. High activity catalysts, as demonstrated through the Ni-based model catalysts, were found to have much higher reforming potential than the mineral- and iron-based catalysts. Although tar was treated as a pseudo-component, the decreased concentration of methane may be understood as an indicator of enhanced tar reforming, as commonly reported in the experimental catalytic gasification research. Similarly, the pseudo-component treatment of tar should be interpreted as a means of capturing overall reforming tendencies rather than detailed tar chemistry.
The influence of the equivalence ratio was found to be mainly linked to the intensity of oxidation. When increasing the equivalence ratio, the results indicated that concentrations of hydrogen and carbon monoxide decreased and that of carbon dioxide increased, indicating that partial combustion had occurred. These observations highlighted the importance of controlling the rate of input of oxidizers to arrive at balanced autothermal conditions without compromising the preservation of syngas quality.
Overall, the modeling framework identified for the development of the catalytic biomass gasification process exhibited potential as a useful tool for the analysis of the thermodynamic limits that were characteristic of the process; consequently, this could allow for valuable insights into catalyst selection while fostering future work to enhance the predictive capability of the presently identified model by considering reaction kinetics, catalyst deactivation, and transport phenomena.
From a scientific standpoint, a key contribution of this study was the demonstration that catalyst-dependent activity corrections can be consistently embedded into equilibrium gasification models to enable comparative catalyst screening, thereby bridging the gap between ideal thermodynamic limits and practical catalyst-oriented process design.
From a research perspective, the principal scientific contribution of this work could lie in demonstrating how catalyst-specific activity normalization can be rigorously embedded into equilibrium gasification models to enable structured comparative catalyst differentiation, an aspect that has not been explicitly resolved in most existing equilibrium-based studies. Accordingly, the proposed methodology should be viewed as a screening-level thermodynamic tool rather than a substitute for detailed thermo-kinetic reactor models. In this sense, the proposed framework can bridge ideal equilibrium limits and kinetically controlled catalytic behavior by offering a constrained thermodynamic perspective suitable for early-stage catalyst screening. Future extensions of the present framework may integrate catalyst deactivation and stability considerations through coupling with kinetic or time-resolved experimental data.