Polyvinyl Chloride-Based Coordination Polymer as Membrane for Phenol Detection
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
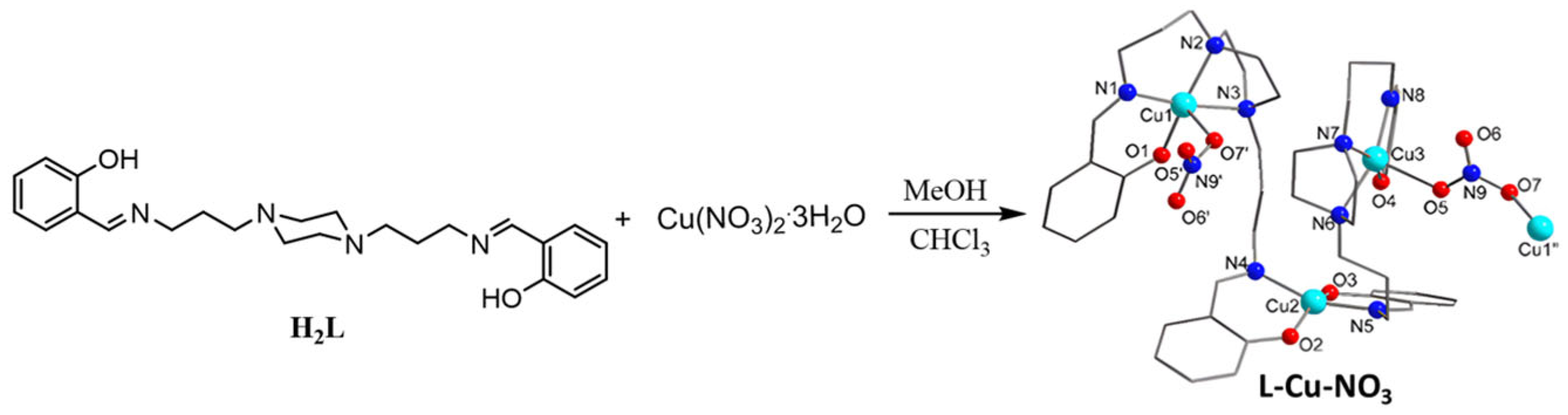
2.2. Synthesis of CPs
2.3. Membrane Preparation
2.4. Electrode Preparation
2.5. Instrumentation
2.6. Electrochemical Characterization
3. Results
3.1. Characterization
3.1.1. CP Characterization
3.1.2. Membrane Characterization
3.2. Electrochemical Investigation
3.3. Formatting of Mathematical Components
- = weight of wet membrane, after 24 h immersion (g);
- = weight of dry membrane, before immersion (g).
- Vt = total volume of membrane (cm3).
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hashim, H.S.; Yap, W.F.; Sheh Omar, N.A.; Muhamad Fauzi, N.I.; Wan, M.E.M. Recent advances of priority phenolic compounds detection using phenol oxidases-based electrochemical and optical sensors. Measurement 2021, 18, 117724. [Google Scholar] [CrossRef]
- El-Kosasy, A.M.; Riad, S.M.; Abd El-Fattah, L.E.; Abd El-Kader Ahmad, S. Novel poly (vinyl chloride) matrix membrane electrodes for the determination of phenolic pollutants in waste water. Water Res. 2003, 37, 1769–1770. [Google Scholar] [CrossRef]
- Chen, K.; Zhang, Z.-L.; Liang, Y.-M.; Liu, W.A. Graphene-Based Electrochemical Sensor for Rapid Determination of Phenols in Water. Sensors 2013, 13, 6204–6216. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Zhou, D.; Liu, X.; Zheng, X. Electrochemical determination of phenol using CTAB-functionalized montmorillonite electrode. Environ. Technol. 2009, 30, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Ojala, M.; Ketola, R.A.; Virkki, V.; Sorsa, H.; Kotiaho, T. Determination of phenolic compounds in water using membrane inlet mass spectrometry. Talanta 1997, 44, 1253–1259. [Google Scholar] [CrossRef] [PubMed]
- Faisal, M.; Alam, M.M.; Ahmed, J.; Asiri, A.M.; Jalalah, M.; Alruwai, R.S.; Rahman, M.M.; Harraz, F.A. Sensitive Electrochemical Detection of 4-Nitrophenol with PEDOT:PSS Modified Pt NPs-Embedded PPy-CB@ZnO Nanocomposites. Biosensors 2022, 12, 990. [Google Scholar] [CrossRef]
- Sergeyeva, T.A.; Slinchenko, O.A.; Gorbach, L.A.; Matyushov, V.F.; Brovko, O.O.; Piletsky, S.A.; Sergeeva, L.M.; Elska, G.V. Catalytic molecularly imprinted polymer membranes: Development of the biomimetic sensor for phenols detection. Anal. Chim. Acta 2010, 659, 274–279. [Google Scholar] [CrossRef]
- Hamid, A.A.; Alam, J.; Shukla, A.K.; Ali, F.A.A.; Alhoshan, M. Sustainable removal of phenol from wastewater using a biopolymer hydrogel adsorbent comprising crosslinked chitosan and κ-carrageenan. Int. J. Biol. Macromol. 2023, 251, 126340. [Google Scholar] [CrossRef]
- Zhang, X.; Miao, S.; Zhou, J.; Zhou, T.; Gan, T.; Tang, Y. Paper-based molecularly imprinted electrochemical sensor integrated with Pt single atom decorated porous hollow carbon polyhedrons for enhanced phenolic pollutants monitoring in wastewaters. Sens. Actuators B Chem. 2025, 445, 138592. [Google Scholar] [CrossRef]
- Padilha, L.F.; Borges, C.P. PVC membranes prepared via non-solvent induced phase separation process. Braz. J. Chem. Eng. 2019, 36, 497–509. [Google Scholar] [CrossRef]
- Mulyasuryani, A.; Mustaghfiroh, A.M. Development of Potentiometric Phenol Sensors by Nata de Coco Membrane on Screen-Printed Carbon Electrode. J. Anal. Methods Chem. 2019, 2019, 4608135. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M. Selective capturing of phenolic derivative by a binary metal oxide microcubes for its detection. Sci. Rep. 2019, 9, 19234. [Google Scholar] [CrossRef] [PubMed]
- Siontorou, C.G.; Georgopoulos, K.N. A Ready-to-Use Metal-Supported Bilayer Lipid Membrane Biosensor for the Detection of Phenol in Water. Membranes 2021, 11, 871. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Balkhoyor, H.B.; Asiri, A.M. Phenolic sensor development based on chromium oxide-decorated carbon nanotubes for environmental safety. J. Environ. Manag. 2017, 188, 228–237. [Google Scholar] [CrossRef]
- Devic, T.; Serre, C. High valence 3p and transition metal based MOFs. Chem. Soc. Rev. 2014, 43, 6097–6115. [Google Scholar] [CrossRef]
- Zhang, C.; Mu, Y.; Zhang, W.; Zhao, S.; Wang, Y. PVC-based hybrid membranes containing metal-organic frameworks for Li+/Mg2+ separation. J. Membr. Sci. 2020, 596, 117724. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, Z.; Xin, R.; Li, D.; Dong, X.; Kushwaha, A.; Parvez, M.K.; Al-Dosari, M.S.; Kumar, A.; Huang, Y. Electrochemical sensing properties of cobalt-based coordination polymers for trace l-tryptophan in milk. Dalton Trans. 2025, 54, 6472–6485. [Google Scholar] [CrossRef]
- Teng, J.; Gao, R.; Cai, Q.; Ma, Y.; Wu, H. Bifunctional electrochemical sensor for L-ascorbic acid and hydrazine based on a two-dimensional cobalt(II) coordination polymer. Electrochim. Acta 2025, 542, 147507. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, W.; Yao, W.; Kang, L.; Gao, E.; Fedin, V.P. An electrochemical sensor based on carbon composites derived from bisbenzimidazole biphenyl coordination polymers for dihydroxybenzene isomers detection. Microchim. Acta 2023, 191, 20. [Google Scholar] [CrossRef] [PubMed]
- Buta, I.; Ardelean, A.; Lönnecke, P.; Novitchi, G.; Hey-Hawkins, E.; Andruh, M.; Costisor, O. Structural and magnetic properties of three one-dimensional nitrato-, azido- and phenoxido-bridged copper(II) coordination polymers. Polyhedron 2020, 190, 114766. [Google Scholar] [CrossRef]
- Narayanan, R.M.; Gowri, S.; Krishna, M.M. On Line Surface Roughness Measurement Using Image Processing and Machine Vision. In Proceedings of the World Congress on Engineering, London, UK, 2–4 July 2007; pp. 645–647. [Google Scholar]
- Lever, A.B.P. Inorganic Electronic Spectroscopy, 2nd ed.; Elsevier: New York, NY, USA, 1968; pp. 359–361. [Google Scholar]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds, 4th ed.; John Wiley & Sons, Inc. Publication: New York, NY, USA, 1986; pp. 225–233. [Google Scholar]
- Novio, F.; Ruiz-Molina, D. Hydrophobic coordination polymer nanoparticles and application for oil–water separation. RSC Adv. 2014, 4, 15293–15296. [Google Scholar] [CrossRef]
- Vatanpour, V.; Boroujeni, N.I.; Pasaoglu, M.E.; Mahmodi, G.; Mohammadikish, M.; Kazemi-Andalib, F.; Koyuncu, I. Novel infinite coordination polymer (ICP) modified thin-film polyamide nanocomposite membranes for simultaneous enhancement of antifouling and chlorine-resistance performance. J. Membr. Sci. 2022, 647, 120305. [Google Scholar] [CrossRef]
- Díaz, D.R.; Carmona, F.J.; Palacio, L.; Ochoa, N.A.; Hernández, A.; Prádanos, P. Impedance spectroscopy and membrane potential analysis of microfiltration membranes. The influence of surface fractality. Chem. Eng. Sci. 2018, 178, 27–38. [Google Scholar] [CrossRef]
- Arfin, T.; Rangari, S.N. Graphene oxide–ZnO nanocomposite modified electrode for the detection of phenol. Anal. Methods 2018, 10, 347–358. [Google Scholar] [CrossRef]
- Niu, K.; Gao, J.; Wu, L.; Lu, X.; Chen, J. Nitrogen-doped graphdiyne present Nitrogen-doped graphdiyne as a robust electrochemical biosensing platform for ultrasensitive detection of environmental pollutants. Anal. Chem. 2021, 93, 8656–8662. [Google Scholar] [CrossRef]







| Membrane Notation | PVC, Weight % | DOS Weight % | THF Weight % | CP Weight % | Thickness, μm | Ra, μm |
|---|---|---|---|---|---|---|
| M1 | 4.9 | 8.9 | 86.2 | 0.00 | 139 ± 6.3 | 4.91 ± 0.83 |
| M2 | 4.9 | 8.9 | 86 | 0.2 | 140 ± 8.9 | 5.41 ± 0.98 |
| M3 | 4.9 | 8.9 | 85.8 | 0.4 | 144 ± 9.7 | 5.93 ± 0.11 |
| M4 | 4.8 | 8.8 | 85.7 | 0.6 | 142 ± 6.1 | 5.83 ± 1.02 |
| M5 | 4.8 | 8.8 | 85.5 | 0.8 | 150 ± 9.1 | 6.57 ± 0.24 |
| Membrane Notation | Water Absorption, W % | Porosity % | Water Contact Angle, ° |
|---|---|---|---|
| M1 | 2.0865 | 5.8567 | 73.4 ± 2.8 |
| M2 | 3.5622 | 8.0984 | 72.48 ± 6.9 |
| M3 | 2.7712 | 9.6506 | 71.58 ± 3.7 |
| M4 | 3.4800 | 14.6389 | 70.85 ± 2.4 |
| M5 | 4.0143 | 17.79 | 70.78 ± 1.1 |
| Cu-M5 | Chi-Sqr | Rs, Ω·cm2 | CPE1-T, F/cm2/sφ−1 | CPE1-P, (φ) | R1, Ω·cm2 | CPE2-T, F/cm2/sφ−1 | CPE2-P, (φ) | R2, Ω·cm2 |
|---|---|---|---|---|---|---|---|---|
| 10−10 M | 0.010 | 20 | 5.71 × 10−7 | 0.57 | 3.81 × 105 | 1.65 × 10−10 | 0.90 | 6.84 × 105 |
| 10−9 M | 0.009 | 20 | 4.93 × 10−7 | 0.60 | 3.88 × 105 | 1.39 × 10−10 | 0.91 | 9.04 × 105 |
| 10−8 M | 0.009 | 20 | 4.99 × 10−7 | 0.60 | 3.78 × 105 | 1.28 × 10−10 | 0.91 | 1.04 × 106 |
| 10−7 M | 0.009 | 20 | 5.12 × 10−7 | 0.59 | 3.23 × 105 | 1.18 × 10−10 | 0.92 | 1.12 × 106 |
| 10−6 M | 0.009 | 20 | 5.36 × 10−7 | 0.60 | 2.70 × 105 | 1.10 × 10−10 | 0.92 | 1.16 × 106 |
| 10−5 M | 0.009 | 20 | 4.57 × 10−7 | 0.61 | 2.20 × 105 | 1.03 × 10−10 | 0.93 | 1.20 × 106 |
| 10−4 M | 0.011 | 20 | 4.32 × 10−7 | 0.62 | 1.76 × 105 | 1.03 × 10−10 | 0.93 | 1.20 × 106 |
| 10−3 M | 0.012 | 20 | 2.68 × 10−7 | 0.65 | 1.59 × 105 | 1.02 × 10−10 | 0.93 | 1.17 × 106 |
| 10−2 M | 0.020 | 20 | 6.17 × 10−8 | 0.74 | 1.48 × 105 | 9.76 × 10−11 | 0.93 | 1.10 × 106 |
| Cu Electrode | M5 | M4 | M3 | M2 | M1 |
|---|---|---|---|---|---|
| Intercept | 54,466.5658 ± 10,995.9128 | 154,478.7434 ± 2400.3356 | 198,108.1789 ± 3001.6758 | 189,405.1247 ± 5460.7023 | 144,760.8708 ± 4074.7654 |
| Slope | −27,605.4245 ± 1729.0162 | −15,033.9254 ± 440.3213 | −7032.4564 ± 485.3213 | −5692.7496 ± 944.0604 | −8578.7323 ± 746.8570 |
| Residual Sum of Squares (RSS) | 799.5200 | 172.3429 | 104.0752 | 631.4166 | 123.5292 |
| Pearson’s r | −0.9922 | −0.9982 | −0.9906 | −0.9491 | −0.9851 |
| R-Square (COD) | 0.9845 | 0.9965 | 0.9813 | 0.9009 | 0.9705 |
| Adj. R-Square | 0.9806 | 0.9957 | 0.9766 | 0.8761 | 0.9632 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cornea, A.-M.; Tara-Lunga Mihali, M.; Buta, I.; Maranescu, V.; Visa, A.; Bora, A.; Plesu, N. Polyvinyl Chloride-Based Coordination Polymer as Membrane for Phenol Detection. Processes 2026, 14, 435. https://doi.org/10.3390/pr14030435
Cornea A-M, Tara-Lunga Mihali M, Buta I, Maranescu V, Visa A, Bora A, Plesu N. Polyvinyl Chloride-Based Coordination Polymer as Membrane for Phenol Detection. Processes. 2026; 14(3):435. https://doi.org/10.3390/pr14030435
Chicago/Turabian StyleCornea, Anemona-Mariana, Milica Tara-Lunga Mihali, Ildiko Buta, Valentin Maranescu, Aurelia Visa, Alina Bora, and Nicoleta Plesu. 2026. "Polyvinyl Chloride-Based Coordination Polymer as Membrane for Phenol Detection" Processes 14, no. 3: 435. https://doi.org/10.3390/pr14030435
APA StyleCornea, A.-M., Tara-Lunga Mihali, M., Buta, I., Maranescu, V., Visa, A., Bora, A., & Plesu, N. (2026). Polyvinyl Chloride-Based Coordination Polymer as Membrane for Phenol Detection. Processes, 14(3), 435. https://doi.org/10.3390/pr14030435








