Evaluation of Three Traditional Curing Methods Applied in Mexican Lead-Glazed Ceramics: Detection, Concentration, and Leaching of Lead to Food
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Sample Analysis
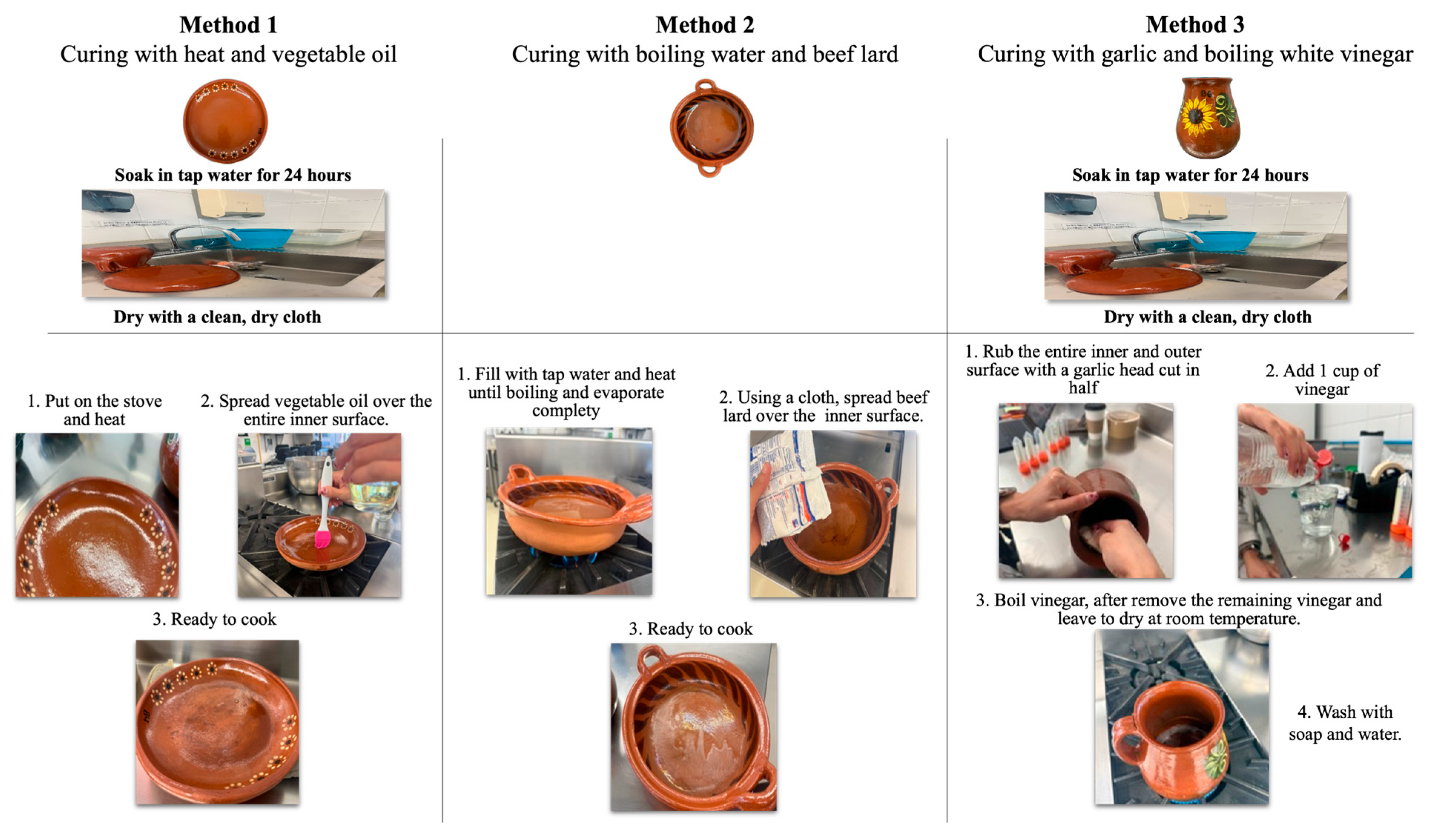
2.2.1. Method 1: Curing with Heat and Vegetable Oil
2.2.2. Method 2: Curing with Boiling Water and Beef Lard
2.2.3. Method 3: Curing with Garlic and Boiling White Vinegar
2.3. Detection and Quantification of Pb by Validated Qualitative and Quantitative Methods
2.3.1. Sodium Rhodizonate—Qualitative Method for Colorimetric Pb Detection
2.3.2. LumetallixTM—Qualitative Method for Colorimetric Pb Detection
2.3.3. X-Ray Fluorescence (XRF)—Quantitative Method for Pb Concentrations
2.4. Pb Leaching from Cured LGC to Food Simulation by ICP-MS
2.5. Statistical Analysis
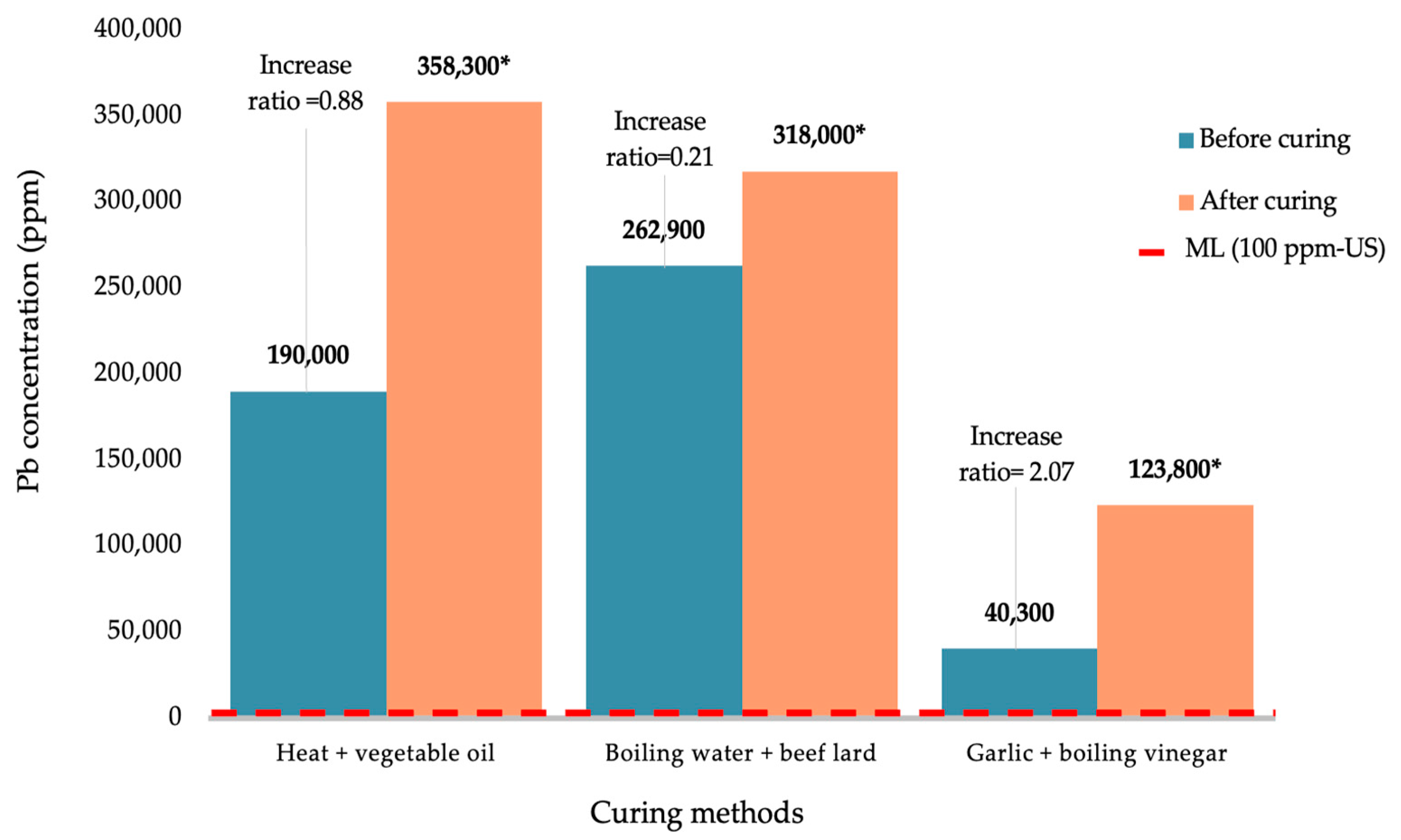
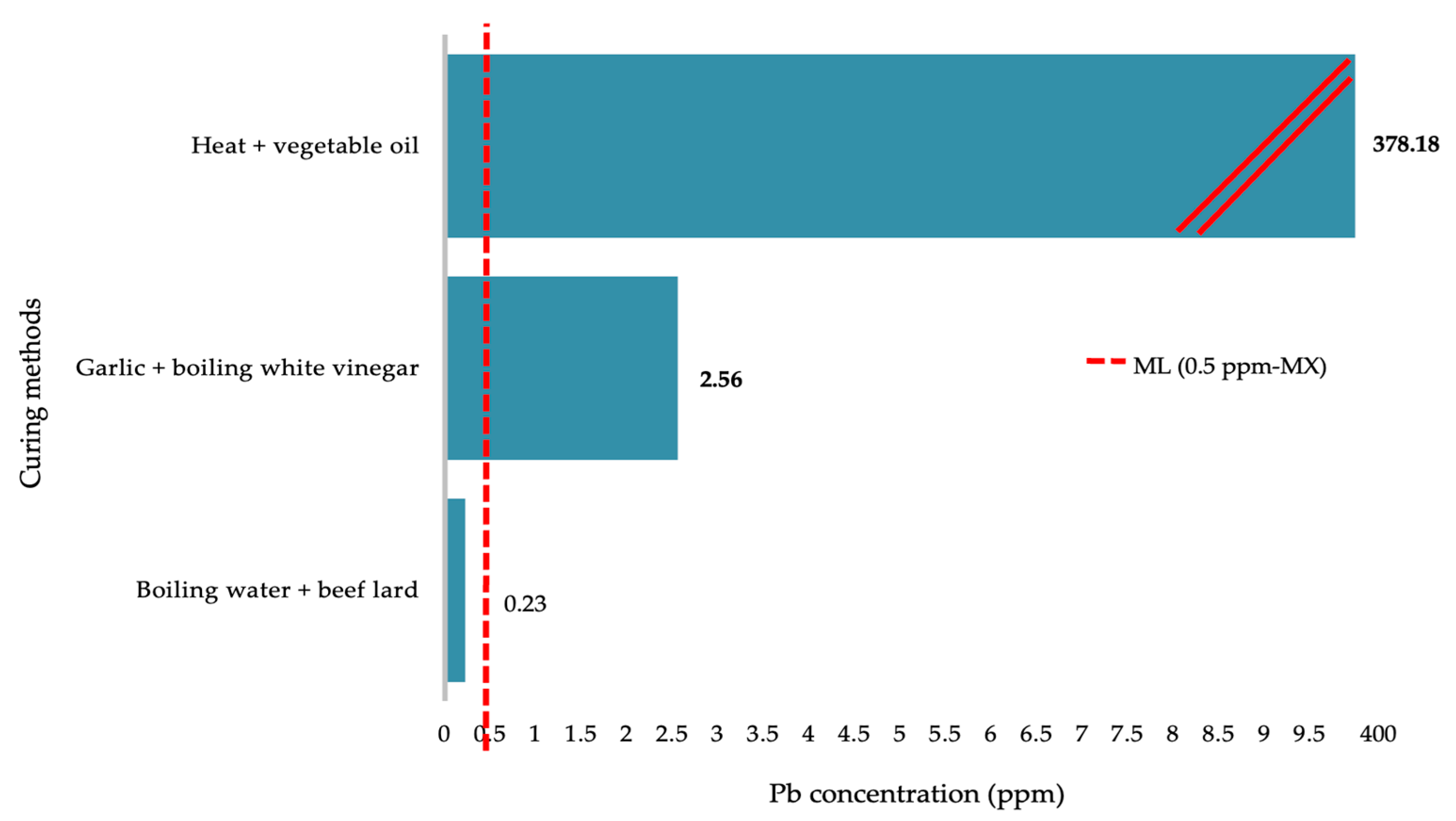
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAS | Atomic Absorption Spectrometry |
| FAO | Food and Agriculture Organization of the United Nations |
| ICP-MS | Inductively Coupled Plasma Mass Spectrometry |
| JECFA | Joint FAO/WHO Expert Committee on Food Additive |
| LGC | Lead-Glazed Ceramic |
| LOD | Limit of Detection |
| LOQ | Limit of Quantification |
| ML | Maximum Limits |
| Pb | Lead |
| PbO | Lead Oxide |
| PROFECO | Procuraduría Federal del Consumidor |
| CPSC | The U.S. Consumer Product Safety Commission |
| WHO | World Health Organization |
| XRF | X-Ray Fluorescence |
References
- Blacksmith Institute; Fondo Nacional para el Fomento de las Artesanías. Informe 2010: Uso de Plomo En la Alfarería En México: Historia, Aplicación, Alternativas, Riesgos a la Salud; Fomentar la Actividad Artesanal en El País (USA and MEX); Blacksmith Institute: New York, NY, USA, 2010. [Google Scholar]
- Wani, A.L.; Ara, A.; Usmani, J.A. Lead toxicity: A review. Interdiscip. Toxicol. 2015, 8, 55–64. [Google Scholar] [CrossRef]
- Bhasin, T.; Lamture, Y.; Kumar, M.; Dhamecha, R. Unveiling the Health Ramifications of Lead Poisoning: A Narrative Review. Cureus 2023, 15, e46727. [Google Scholar] [CrossRef]
- Santana, A.B.; Spelta, L.E.W.; Sobalvarro, J.V.M.; Podestá, M.H.M.C.; Garcia, R.C.T.; Dos Reis, T.M.; Torres, L.H. Gestational lead exposure and its effects on fetal/infant development—A systematic review. Reprod. Toxicol. 2023, 117, 108342. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.S. Neurotoxicity and Outcomes from Developmental Lead Exposure: Persistent or Permanent? Environ. Health Perspect. 2023, 131, 85002. [Google Scholar] [CrossRef]
- Hoffman-Pennesi, D.; Winfield, S.; Gavelek, A.; Santillana Farakos, S.M.; Spungen, J. Infants’ and young children’s dietary exposures to lead and cadmium: FDA total diet study 2018–2020. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2024, 41, 1454–1479. [Google Scholar] [CrossRef]
- Figueroa, J.L.; Rodríguez-Atristain, A.; Bautista-Arredondo, L.F.; Treviño, C.L.; Martínez, M.R.; Valdivia, B.T.; Avalos-Alvarez, S.V.; Sánchez-Morales, J.E.; Fuller, R.; Bautista-Arredondo, S.; et al. Loss of cognitive function in Mexican children due to lead exposure and the associated economic costs. Environ. Res. 2024, 263 Pt 1, 120013. [Google Scholar] [CrossRef]
- Lanphear, B.P.; Rauch, S.; Auinger, P.; Allen, R.W.; Hornung, R.W. Low-level lead exposure and mortality in US adults: A population-based cohort study. Lancet Public Health 2018, 3, e177–e184. [Google Scholar] [CrossRef] [PubMed]
- Reuben, A.; Caspi, A.; Belsky, D.W.; Broadbent, J.; Harrington, H.; Sugden, K.; Houts, R.M.; Ramrakha, S.; Poulton, R.; Moffitt, T.E. Association of Childhood Blood Lead Levels with Cognitive Function and Socioeconomic Status at Age 38 Years and with IQ Change and Socioeconomic Mobility Between Childhood and Adulthood. JAMA 2017, 317, 1244–1251. [Google Scholar] [CrossRef]
- Abadin, H.; Ashizawa, A.; Stevens, Y.W.; Llados, F.; Diamond, G.; Sage, G.; Citra, M.; Quinones, A.; Bosch, S.J.; Swarts, S.G. Toxicological Profile for Lead; Agency for Toxic Substances and Disease Registry (US): Atlanta, GA, USA, 2007.
- Tellez-Rojo, M.M.; Bautista-Arredondo, L.F.; Trejo-Valdivia, B.; Tamayo-Ortiz, M.; Estrada-Sánchez, D.; Kraiem, R.; Pantic, I.; Mercado-García, A.; Romero-Martínez, M.; Shamah-Levy, T.; et al. Análisis de la distribución nacional de intoxicación por plomo en niños de 1 a 4 años. Implicaciones parala política pública en México [Analysis of the national distribution of lead poisoning in 1–4-year-old children.Implications for Mexican public policy]. Salud Publica México 2020, 62, 627–636. (In Spanish) [Google Scholar] [CrossRef] [PubMed]
- Cendejas_Huerta, S.; Díaz_González, A. El Problema De La Toxicidad En La Loza Artesanal Mexicana. Salud Pública México 1974, 16, 83–88. Available online: https://saludpublica.mx/index.php/spm/article/view/1635/1618 (accessed on 25 August 2025).
- Viniegra, G.; Escobar_M, R. El Riesgo Del Saturnismo Por La cerámica folklórica Mexicana (Estudio preliminar). Salud Pública México 1966, 8, 69–77. [Google Scholar]
- Ortiz-Ortiz, E.; García-Nieto, E.; Juárez-Santacruz, L.; Gómez-Camarillo, M.A.; García-Gallegos, E.; Limón-Huitrón, P. Lead exposure: Pottery impact in Tlaxcala, Mexico. Rev. Int. Contam. Ambient. 2017, 33, 57–64. [Google Scholar] [CrossRef]
- Vega-Franco, L.; Alvear, G.; Meza-Camacho, C. La cerámica vidriada como factor de riesgo de exposición al plomo. Salud Publica México 1994, 36, 148–153. [Google Scholar]
- Téllez-Rojo, M.M.; Bautista-Arredondo, L.F.; Trejo-Valdivia, B.; Cantoral, A.; Estrada-Sánchez, D.; Kraiem, R.; Pantic, I.; Rosa-Parra, A.; Gómez-Acosta, L.M.; Romero-Martínez, M.; et al. Reporte nacional de niveles de plomo en sangre y uso de barro vidriado en población infantil vulnerable [National report of blood lead levels and lead-glazed ceramics use in vulnerable children]. Salud Publica México 2019, 61, 787–797. (In Spanish) [Google Scholar] [CrossRef]
- Bolle, F.; Fekete, V.; Demont, M.; Boutakhrit, K.; Petit, D.; Brian, W.; Feraille, G.; Van Loco, J. Lead migration from ceramicware in contact with foodstuff: Effect of glaze, temperature, pH and food simulant. J. Food Sci. Eng. 2012, 2, 301–313. [Google Scholar] [CrossRef]
- NORMA Oficial Mexicana NOM-231-SSA1-2016; Artículos de Alfarería Vidriada, Cerámica Vidriada, Porcelana y Artículos de Vidrio-Límites Máximos Permisibles de Plomo y Cadmio Solubles-Método de Ensayo. Diario Oficial de la Federación; Secretaría de Salud: Ciudad de México, Mexico, 2015.
- Peralta, N.; Cantoral, A.; Téllez-Rojo, M.M.; Trejo-Valdivia, B.; Estrada-Sánchez, D.; Richardson-L, V.; Caravanos, J.; Fuller, R. Lead levels in a potter’s population and its association with the use of different glazes: Cross-sectional evaluation of the approved pottery program. Front. Toxicol. 2022, 4, 799633. [Google Scholar] [CrossRef]
- Torres-Sánchez, L.; López-Carrillo, L.; Ríos, C. Eliminación del plomo por curado casero. Salud Publica México 1999, 41 (Suppl. S2), s106–s108. [Google Scholar] [CrossRef]
- Pure Earth. Lead in Consumers Goods: A 25-Country Analysis of Lead (Pb) Levels in 5000+ Products and Foods. 2023. Available online: https://www.pureearth.org/wp-content/uploads/2023/11/Pure-Earth-RMS-Final-Report.pdf (accessed on 12 May 2025).
- Martínez Maldonado, I.; Enríquez Martínez, Á. Revista Del Consumidor. Cura tus ollas de Barro. 2021, p. 98. Available online: https://www.gob.mx/cms/uploads/attachment/file/644065/HAZLO_TU_MISMO_CURA_TUS_OLLAS_DE_BARRO.pdf (accessed on 11 May 2025).
- Helmbrecht, L.; van Dongen, S.W.; van der Weijden, A.; van Campenhout, C.T.; Noorduin, W.L. Direct Environmental Lead Detection by Photoluminescent Perovskite Formation with Nanogram Sensitivity. Environ. Sci. Technol. 2023, 57, 20494–20500. [Google Scholar] [CrossRef] [PubMed]
- Tighe, M.; Knaub, C.; Sisk, M.; Ngai, M.; Lieberman, M.; Peaslee, G.; Beidinger, H. Validation of a screening kit to identify environmental lead hazards. Environ. Res. 2020, 181, 108892. [Google Scholar] [CrossRef] [PubMed]
- Scharman, E.J.; Krenzelok, E.P. A sodium rodizonate lead testing kit for home use-valid for paint and soil samples? J. Toxicol. Clin. Toxicol. 1996, 34, 699–702. [Google Scholar] [CrossRef]
- Pure Earth. ¿Cómo Detectar la Presencia de Plomo en Cazuelas, Ollas, Platos y Jarros de Barro Esmaltado? pp. 12–23. Available online: https://www.barroaprobado.org/documentos/ManualPruebas.pdf (accessed on 11 May 2025).
- Van der Kroef, I.; Nash, E.; Helmbrecht, L. The Assessment of Lead Detection Methods in Tamil Nadu India and Beyond. 2024, pp. 1–11. Available online: https://www.pureearth.org/wp-content/uploads/2024/07/White-Paper-Lead-Detection-Methods_final-version.pdf (accessed on 11 May 2025).
- The LumetallixTM. Instant Lead Detection Kit—Lumetallix. Available online: https://lumetallixusa.com/pages/faq-frequently-asked-questions?srsltid=AfmBOoq13dbfb6fTGUU3-9UAcL8CUlaEzTmB4nlwekYY3-4XXWjIIiJo (accessed on 11 May 2025).
- Binkhorst, G. Metal Cookware Leaching Test Protocol. 2024. Available online: https://www.pureearth.org/wp-content/uploads/2024/12/Metal-Cookware-Leachate-Test-Protocol.pdf (accessed on 11 May 2025).
- Thermo Scientific. Thermo Scientific Niton XL3t GOLDD Series XRF Analyzers for Consumer Goods. 2013. Available online: https://tools.thermofisher.com/content/sfs/brochures/Niton-XL3t-GOLDD-Consumer-Goods-LODs-2013Feb05.pdf (accessed on 11 May 2025).
- Wills, J.D.; Jarvis, K.E.; Williams, J.G. Feasibility of on-line ion exchange interfaced to an inductively coupled plasma mass spectrometer (ICP-MS). Sci. Total Environ. 1993, 135, 137–143. [Google Scholar] [CrossRef]
- Paniagua de Gudiel, D.R.; Héctor Aguilar, J. Presencia de plomo soluble en la cerámica vidriada típica de Guatemala. Rev. Científica 1983, 1, 36–40. [Google Scholar] [CrossRef]
- Paucar-Menacho, L.M.; Salvador-Reyes, R.; Guillén-Sánchez, J.; Capa-Robles, J.; Moreno-Rojo, C. Estudio comparativo de las características físico-químicas del aceite de sacha inchi (Plukenetia volubilis L.), aceite de oliva (Olea europaea) y aceite crudo de pescado. Sci. Agropecu. 2015, 6, 279–290. [Google Scholar] [CrossRef]
- Rasmussen, K.L.; Milner, G.R.; Delbey, T.; Jensen, L.K.I.; Witte, F.; Rehren, T.; Kjaer, U.; Grinder-Hansen, P. Release of lead from Renaissance lead-glazed ceramics from southern Denmark and northern Germany: Implications from acetic acid etching experiments. Herit. Sci. 2022, 10, 63. [Google Scholar] [CrossRef]
- Seth, T.D.; Sircar, S.; Hasan, M.Z. Studies on lead extraction from glazed pottery under different conditions. Bull. Environ. Contam. Toxicol. 1973, 10, 51–56. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations; World Health Organization. Standard for Named Vegetable Oils. 2025. Available online: https://www.fao.org/fao-who-codexalimentarius/sh-proxy/tr/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FStandards%252FCXS%2B210-1999%252FCXS_210e.pdf (accessed on 29 June 2025).
- Ichu, C.B.; Nwakanma, H.O. Comparative Study of the Physicochemical Characterization and Quality of Edible Vegetable Oils. Int. J. Res. Inf. Sci. Appl. Tech. (IJRISAT) 2019, 3, 1–9. [Google Scholar] [CrossRef]
- Suljagić, J.; Suljkanović, M.; Bjelić, E.; Kovačević, A. Selective separation and removal of lead(ii) ion from multicomponent system through BLMs supported by fatty acids. Technol. Acta Sci./Prof. J. Chem. Technol. 2022, 15, 47–51. [Google Scholar] [CrossRef]
- Yohannes, L.; Feleke, H.; Melaku, M.S.; Amare, D.E. Analysis of heavy metals and minerals in edible vegetable oils produced and marketed in Gondar City, Northwest Ethiopia. BMC Public Health 2024, 24, 2204. [Google Scholar] [CrossRef]
- Roca Cruz, A.; Cabrera Vique, C.; Lorenzo Tovar, M.; López Martínez, M.C. Contenido de plomo y cadmio en aceites de girasol. Grasas Y Aceites 2001, 52, 229–234. [Google Scholar] [CrossRef]
- Nowacka, M.; Rybak, K.; Wiktor, A.; Mika, A.; Boruszewski, P.; Woch, J.; Przybysz, K.; Witrowa-Rajchert, D. The quality and safety of food contact materials—Paper and cardboard coated with paraffin emulsion. Food Control 2018, 93, 183–190. [Google Scholar] [CrossRef]
- Izzo, F.C.; Kratter, M.; Nevin, A.; Zendri, E. A Critical Review on the Analysis of Metal Soaps in Oil Paintings. ChemistryOpen 2021, 10, 904–921. [Google Scholar] [CrossRef] [PubMed]
- Raviolo, A.; Farré, A.S.; Schroh, N.T. Students’ understanding of molar concentration. Chem. Educ. Res. Pract. 2021, 22, 486–497. [Google Scholar] [CrossRef]
- Artesanos y Trabajadores en la Elaboración de Productos de Cerámica, Vidrio, Azulejo y Similares: Salarios, Diversidad, Industrias e Informalidad Laboral|Data México. Available online: https://www.economia.gob.mx/datamexico/es/profile/occupation/artesanos-y-trabajadores-en-la-elaboracion-de-productos-de-ceramica-vidrio-azulejo-y-similares# (accessed on 21 May 2025).
- Estadísticas a Propósito del día Internacional del Artesano. Available online: https://inegi.org.mx/contenidos/saladeprensa/aproposito/2023/EAP_ART23.pdf (accessed on 21 May 2025).
- Estrada-Sánchez, D.; Ericson, B.; Juárez-Pérez, C.A.; Aguilar-Madrid, G.; Hernández, L.; Gualtero, S.; Caravanos, J. Intelligence quotient loss in Mexican pottery artisan’s children. Rev. Médica Del Inst. Mex. Del Seguro Soc. 2017, 55, 292–299. [Google Scholar]
- California Department of Public Health. Lead in Traditional Ceramic Dishware. Available online: www.cdph.ca.gov/programs/ (accessed on 12 May 2025).
- Questions and Answers on Lead-Glazed Traditional Pottery|FDA. Available online: https://www.fda.gov/food/environmental-contaminants-food/questions-and-answers-lead-glazed-traditional-pottery (accessed on 12 May 2025).
- Cómo Curar Utensilios de Barro y Proteger tu Salud: Recomendaciones de Pure Earth México—Pure Earth México. Available online: https://barroaprobado.org/como-curar-utensilios-de-barro-y-proteger-tu-salud-recomendaciones-de-pure-earth-mexico/ (accessed on 12 May 2025).
| Piece | Shape | Model | Volume (L) | Picture |
|---|---|---|---|---|
| Piece 1 | Flat | Dish | __ |  |
| Piece 2 | Flat | Pot | 2 |  |
| Piece 3 | Round | Jar | 1.85 |  |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Hernández, M.F.; Betanzos-Robledo, L.; Mariscal-Moreno, R.M.; Valverde-Arámbula, F.A.; Chuck-Hernández, C.; Peralta-Delgado, N.; Fuller, R.; Cantoral, A. Evaluation of Three Traditional Curing Methods Applied in Mexican Lead-Glazed Ceramics: Detection, Concentration, and Leaching of Lead to Food. Processes 2025, 13, 2766. https://doi.org/10.3390/pr13092766
Rodríguez-Hernández MF, Betanzos-Robledo L, Mariscal-Moreno RM, Valverde-Arámbula FA, Chuck-Hernández C, Peralta-Delgado N, Fuller R, Cantoral A. Evaluation of Three Traditional Curing Methods Applied in Mexican Lead-Glazed Ceramics: Detection, Concentration, and Leaching of Lead to Food. Processes. 2025; 13(9):2766. https://doi.org/10.3390/pr13092766
Chicago/Turabian StyleRodríguez-Hernández, Maria F., Larissa Betanzos-Robledo, Rosa María Mariscal-Moreno, Francisco A. Valverde-Arámbula, Cristina Chuck-Hernández, Netzy Peralta-Delgado, Richard Fuller, and Alejandra Cantoral. 2025. "Evaluation of Three Traditional Curing Methods Applied in Mexican Lead-Glazed Ceramics: Detection, Concentration, and Leaching of Lead to Food" Processes 13, no. 9: 2766. https://doi.org/10.3390/pr13092766
APA StyleRodríguez-Hernández, M. F., Betanzos-Robledo, L., Mariscal-Moreno, R. M., Valverde-Arámbula, F. A., Chuck-Hernández, C., Peralta-Delgado, N., Fuller, R., & Cantoral, A. (2025). Evaluation of Three Traditional Curing Methods Applied in Mexican Lead-Glazed Ceramics: Detection, Concentration, and Leaching of Lead to Food. Processes, 13(9), 2766. https://doi.org/10.3390/pr13092766









