Chitosan Composites Functionalized with Green-Synthesized Silver Nanoparticles from Manacá-da-Serra Flowers for the Disinfection of Industrial Wastewater
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Collection and Preparation of Manacá-da-Serra Flowers
2.3. Preparation of the Floral Extract
2.4. Characterization of the Floral Extract
2.4.1. Total Phenolic Content
2.4.2. Antioxidant Activity
2.5. Green Synthesis of AgNPs
Optimization and Influence of Experimental Parameters
2.6. Characterization of the Green AgNPs
2.6.1. UV-Vis Spectroscopy
2.6.2. Transmission Electron Microscopy (TEM) Analysis
2.6.3. Zeta Potential (ZP) Analysis
2.6.4. Evaluation of the Antimicrobial Activity of the Synthesized AgNPs
2.7. Incorporation of AgNPs in the Chitosan Matrix
2.8. Characterization of the Chitosan/AgNPs Composite
2.8.1. FEG-SEM and EDS Analysis
2.8.2. FTIR Analysis
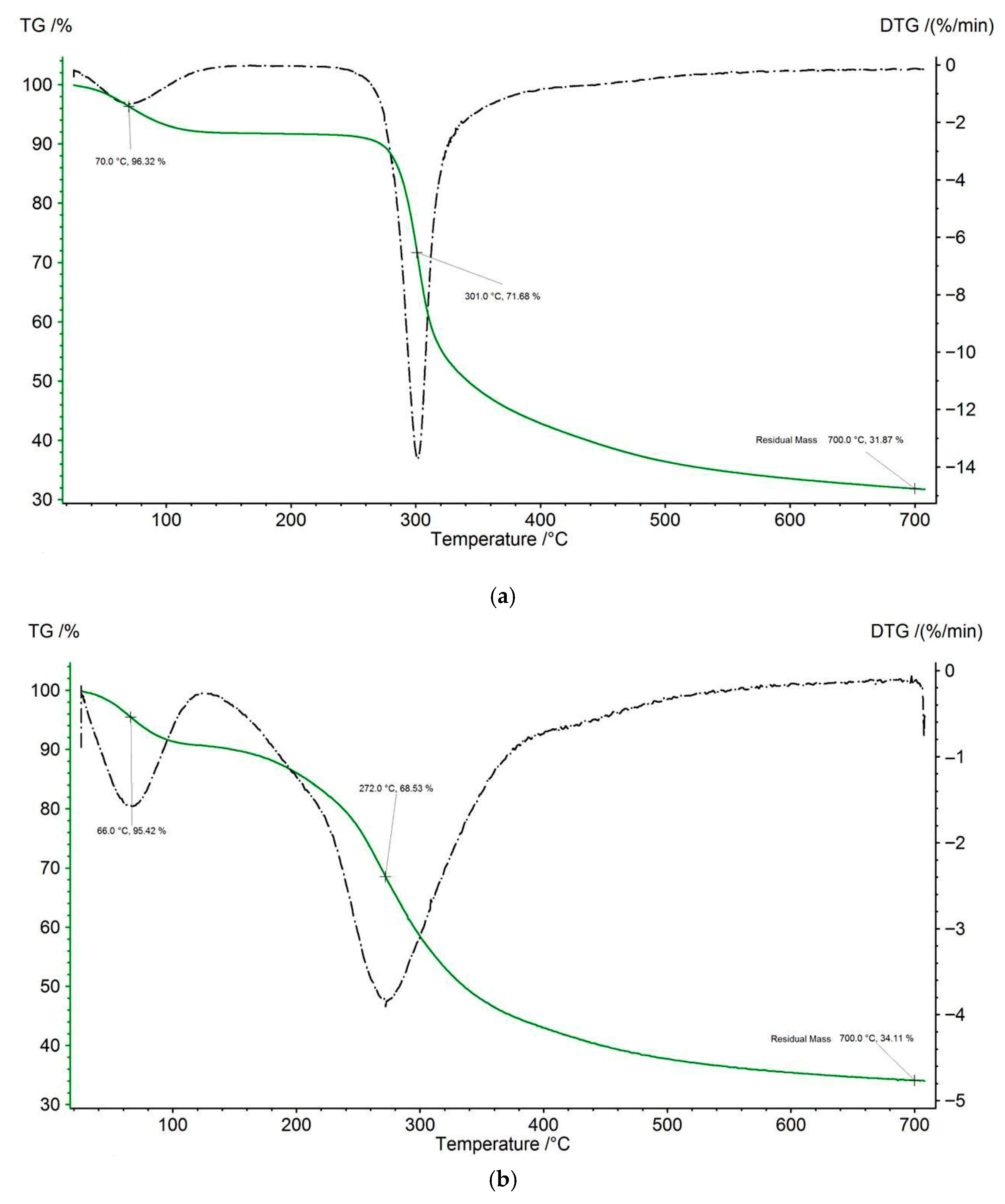
2.8.3. TGA Analysis
2.9. Evaluation of the Antimicrobial Activity of the Chitosan/AgNPs Composite
2.10. Industrial Wastewater Treatment
3. Results and Discussion
3.1. Characterization of the Floral Extract
3.1.1. Total Phenolic Content
3.1.2. Antioxidant Activity
3.2. Green Synthesis of AgNPs
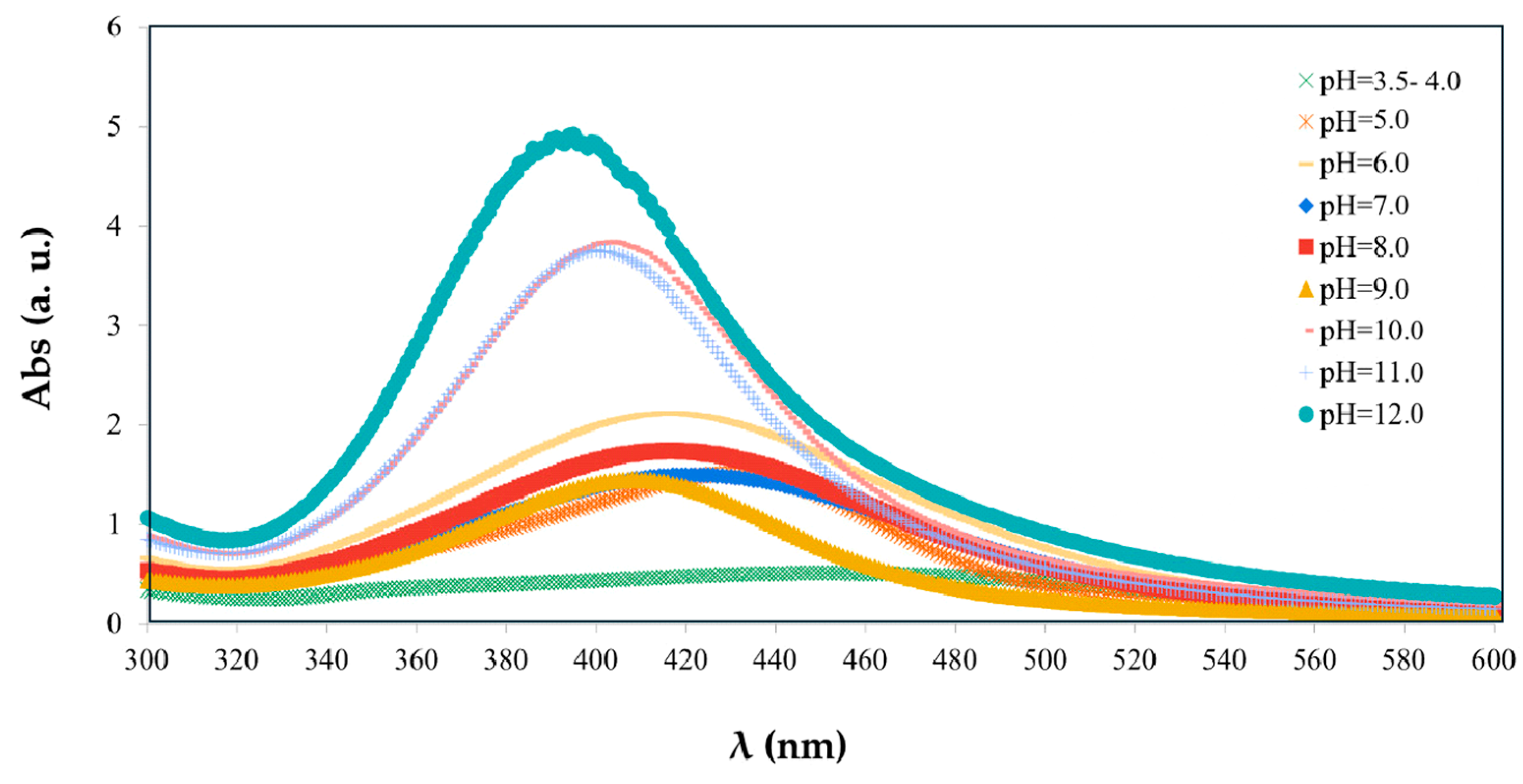
3.2.1. Optimization and Influence of Experimental Parameters
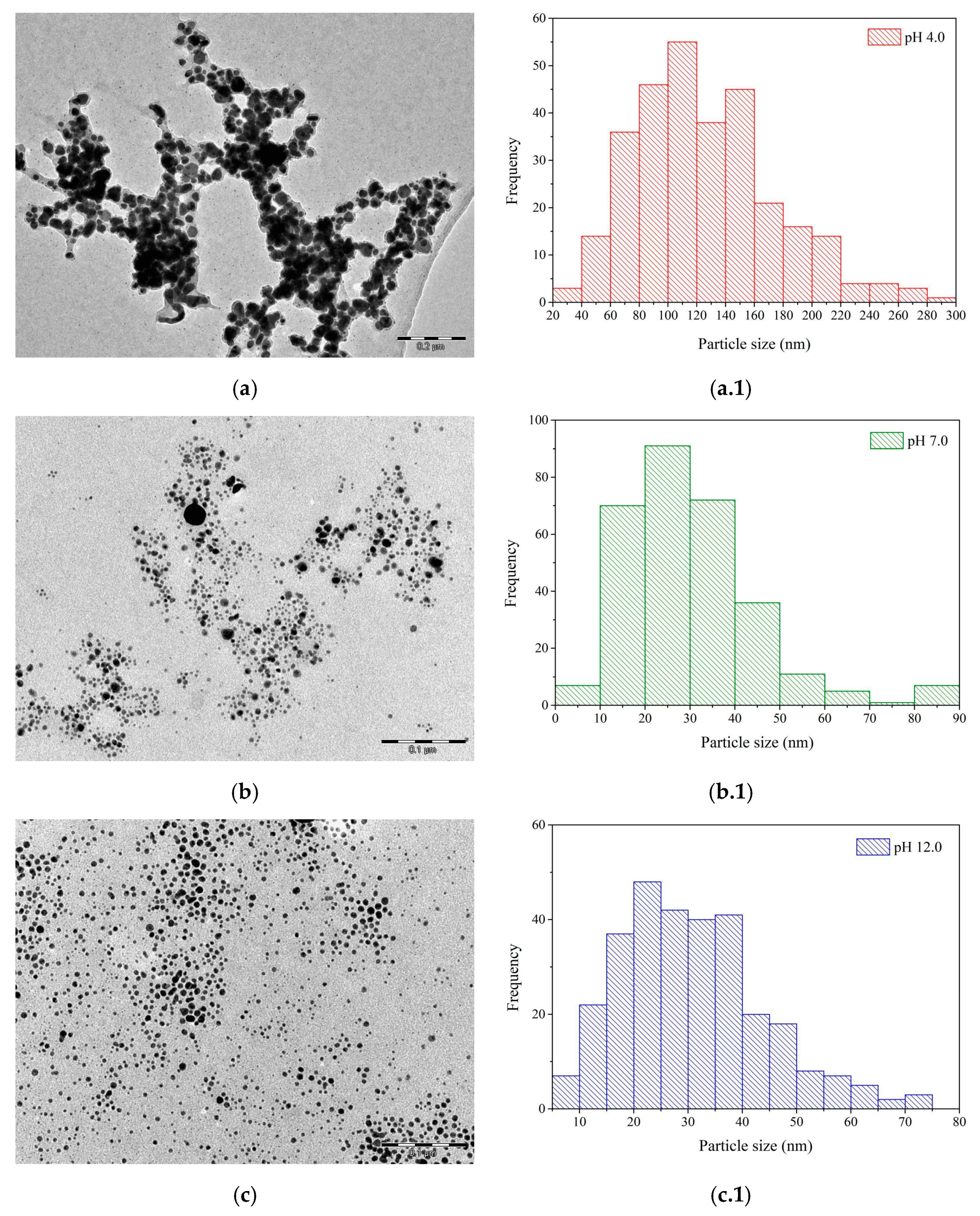
3.2.2. TEM Analysis
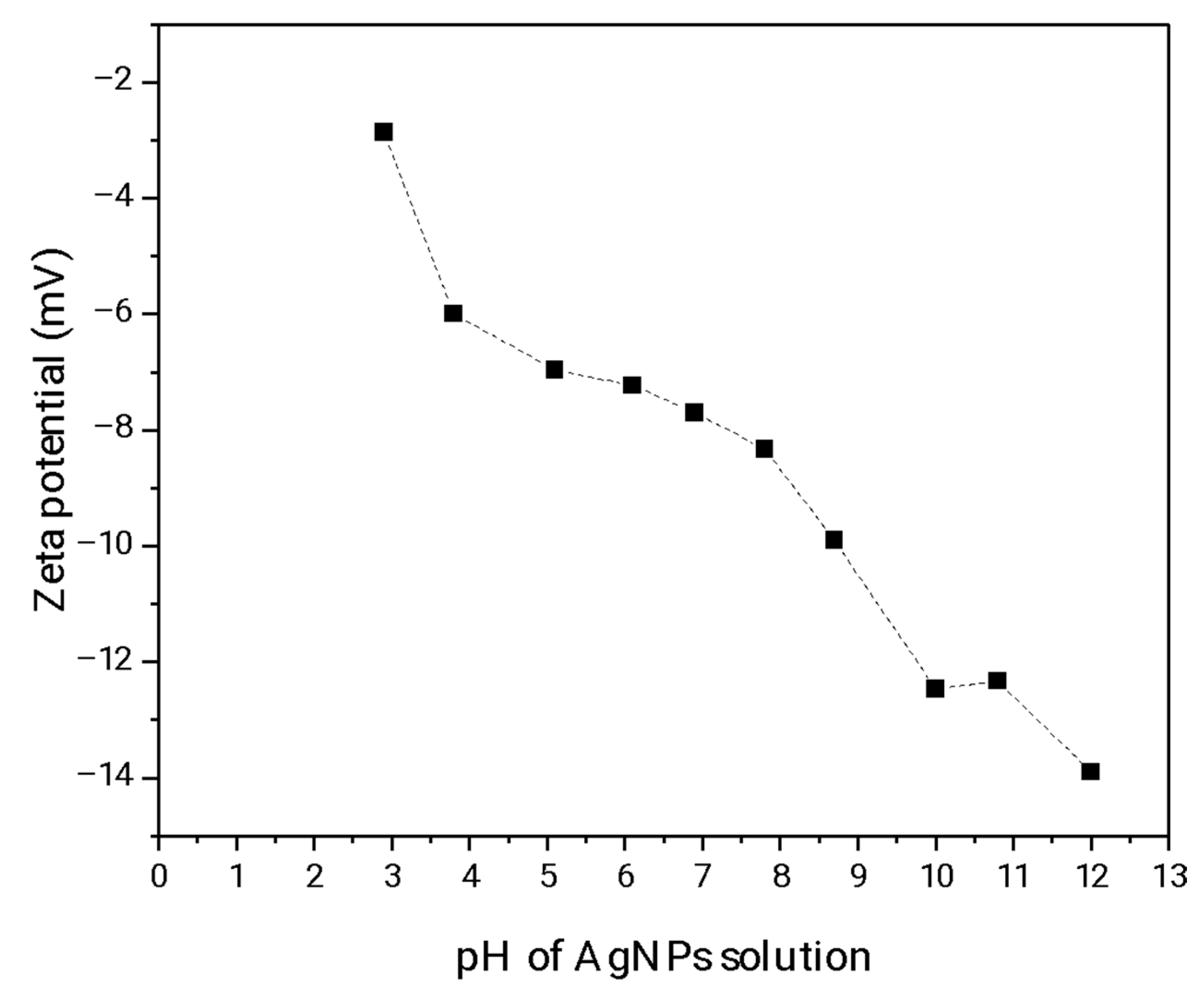
3.2.3. ZP Analysis
3.2.4. Evaluation of the Antimicrobial Activity of the Synthesized AgNPs
3.3. Characterization of the Chitosan/AgNPs Composite
3.3.1. FEG-SEM and EDS Analysis
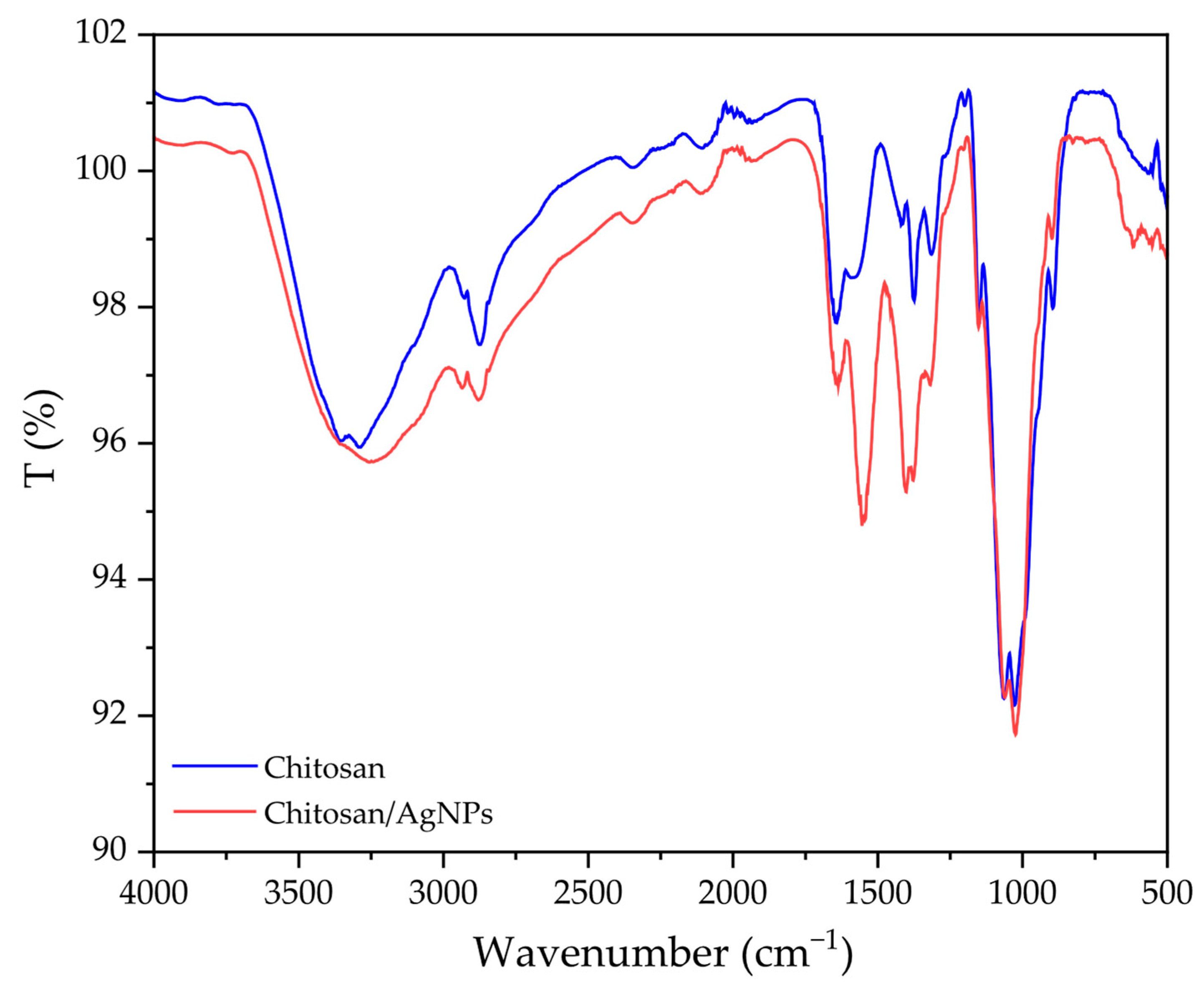
3.3.2. FTIR Analysis
3.3.3. TGA Analysis
3.3.4. Evaluation of the Antimicrobial Activity of the Chitosan/AgNPs Composite
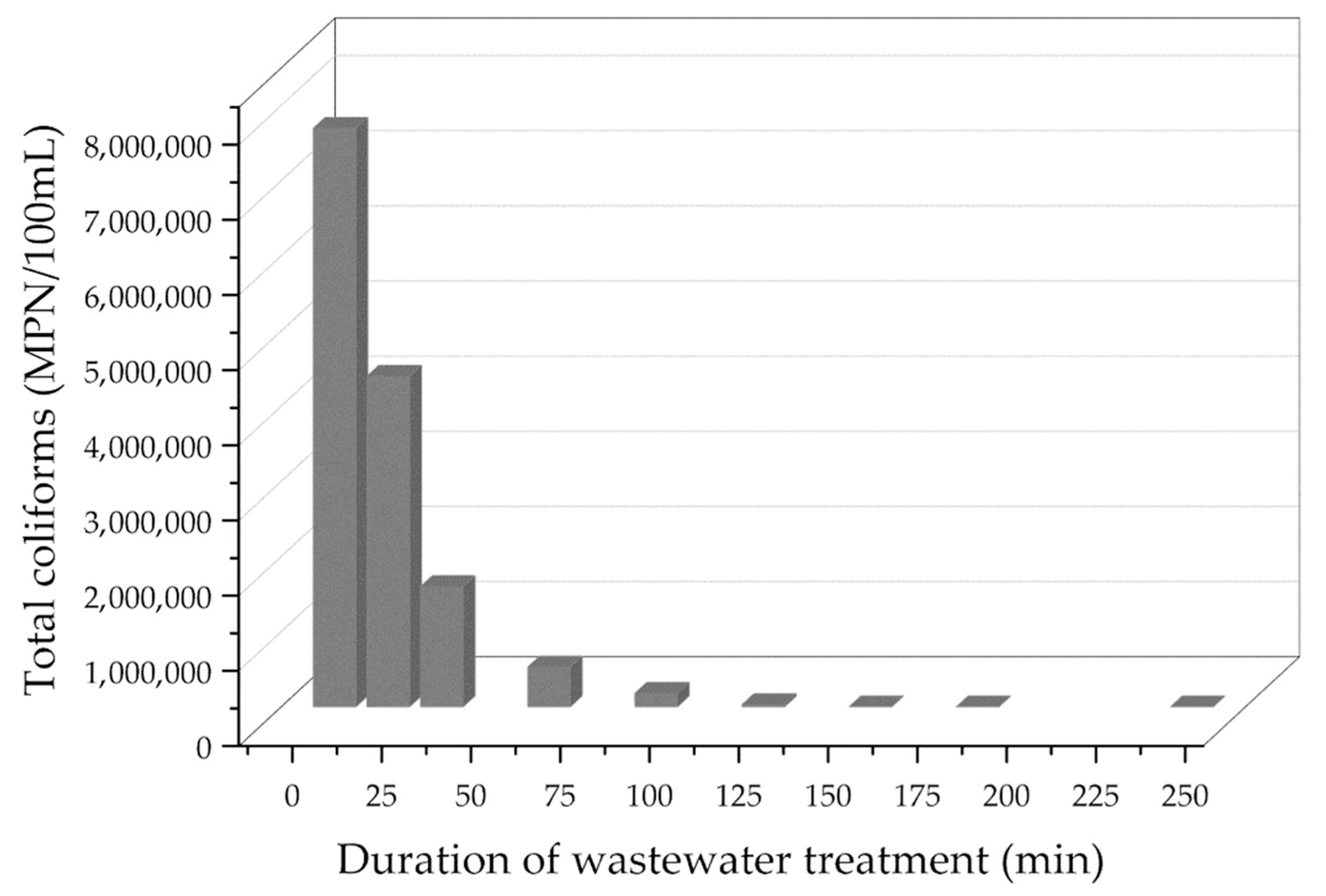
3.4. Industrial Wastewater Treatment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AgNPs | Silver Nanoparticles |
| ATR | Attenuated Total Reflection |
| BOD | Biochemical Oxygen Demand |
| CFU | Colony Forming Units |
| COD | Chemical Oxygen Demand |
| CLSI | Clinical and Laboratory Standards Institute |
| DNA | Deoxyribonucleic Acid |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| DTG | Derivative Thermogravimetry |
| EDS | Energy Dispersive Spectroscopy |
| FEG-SEM | Field Emission Gun Scanning Electron Microscopy |
| FTIR | Fourier Transform Infrared |
| GA | Gallic Acid |
| GAE | Gallic Acid Equivalent |
| ICP-OES | Inductively Coupled Plasma Optical Emission Spectroscopy |
| MIC | Minimum Inhibitory Concentration |
| MPN | Most Probable Number |
| NPs | Nanoparticles |
| PLA | Polylactic Acid |
| PVA | Polyvinyl Alcohol |
| PVP | Polyvinylpyrrolidone |
| SPR | Surface Plasmon Resonance |
| TDS | Total Dissolved Solids |
| TEM | Transmission Electron Microscopy |
| TGA | Thermogravimetric Analysis |
| UV-Vis | Ultraviolet-Visible Spectroscopy |
| WHO | World Health Organization |
References
- Bouaich, F.Z.; Maherzi, W.; El-Hajjaji, F.; Abriak, N.E.; Benzerzour, M.; Taleb, M.; Rais, Z. Reuse of treated wastewater in the manufacture of concrete: Major challenge of environmental preservation. Mater. Sustain. Dev. 2022, 29, 146–157. [Google Scholar] [CrossRef]
- Jhansi, S.C.; Mishra, S.K. Emerging technology in urban areas of developing countries for sustainable wastewater treatment and reuse. Consilience 2013, 10, 1–15. [Google Scholar] [CrossRef]
- ONU. World Water Development Report: Water for Prosperity and Peace; UNESCO: Paris, France, 2024; Available online: https://www.un.org/sustainabledevelopment/water-and-sanitation/ (accessed on 15 June 2025).
- Gelete, G.; Gokcekus, H.; Ozsahin, D.U.; Uzun, B.; Gichamo, T. Evaluating disinfection techniques of water treatment. Desalin. Water Treat. 2020, 177, 408–415. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Moulin, S.; Rozen-Rechels, D.; Stankovic, M. Traitement Des Eaux Usées [Report]; CERES-ERTI, 2013. Available online: https://ceres.ens.psl.eu/IMG/Traitement.pdf (accessed on 15 October 2024).
- Amin, M.M.; Hashemi, H.; Boyini, A.M. A review on wastewater disinfection. Int. J. Environ. Health Eng. 2013, 2, 22. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Abbà, A.; Benigna, I.; Sorlini, S.; Torretta, V. Overview of the main disinfection processes for wastewater and drinking water treatment plants. Sustainability 2017, 10, 86. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Parveen, T.; Umar, K.; Ibrahim, M.N.M. Role of nanomaterials in the treatment of wastewater: A review. Water 2020, 12, 495. [Google Scholar] [CrossRef]
- Kumar-Krishnan, S.; Prokhorov, E.; Hernández-Iturriaga, M.; Mota-Morales, J.D.; Vázquez-Lepe, M.; Kovalenko, Y.; Sánchez, I.C.; Luna-Bárcenas, G. Chitosan/silver nanocomposites: Synergistic antibacterial action of silver nanoparticles and silver ions. Eur. Polym. J. 2015, 67, 242–251. [Google Scholar] [CrossRef]
- Bruna, T.; Maldonado-Bravo, F.; Jara, P.; Caro, N. Silver nanoparticles and their antibacterial applications. Int. J. Mol. Sci. 2021, 22, 7202. [Google Scholar] [CrossRef]
- Saleh, T.A. Nanomaterials: Classification, properties, and environmental toxicities. Environ. Technol. Innov. 2020, 20, 101067. [Google Scholar] [CrossRef]
- Sun, D.; Turner, J.; Jiang, N.; Zhu, S.; Zhang, L.; Falzon, B.; McCoy, C.; Maguire, P.; Mariotti, D.; Sun, D. Atmospheric pressure microplasma for antibacterial silver nanoparticle/chitosan nanocomposites with tailored properties. Compos. Sci. Technol. 2020, 186, 107911. [Google Scholar] [CrossRef]
- Shukla, R.K.; Badiye, A.; Vajpayee, K.; Kapoor, N. Genotoxic potential of nanoparticles: Structural and functional modifications in DNA. Front. Genet. 2021, 12, 728250. [Google Scholar] [CrossRef]
- Galatage, S.T.; Hebalkar, A.S.; Dhobale, S.V.; Mali, O.R.; Kumbhar, P.S.; Nikade, S.V.; Killedar, S.G. Silver Nanoparticles: Properties, Synthesis, Characterization, Applications and Future Trends; IntechOpen: London, UK, 2021. [Google Scholar] [CrossRef]
- Ajith, P.; Murali, A.S.; Sreehari, H.; Vinod, B.S.; Anil, A.; Smitha, C.S. Green synthesis of silver nanoparticles using Calotropis gigantea extract and its applications in antimicrobial and larvicidal activity. Mater. Today Proc. 2019, 18, 4987–4991. [Google Scholar] [CrossRef]
- Chidambaram, S.M.; Sharita, K.; Maheswari, R.; Muzammil, M.S. Efficacy of green synthesis of silver nanoparticles using flowers of Calendula officinalis. Chem. Sci. Trans. 2014, 3, 773–777. [Google Scholar] [CrossRef]
- Surya, S.; Kumar, G.D.; Rajakumar, R. Green synthesis of silver nanoparticles from flower extract of Hibiscus rosa-sinensis and its antibacterial activity. Int. J. Innov. Res. Sci. Eng. Technol. 2016, 5, 5242–5247. Available online: https://www.ijirset.com/upload/2016/april/129_Green.pdf (accessed on 5 November 2025).
- Padalia, H.; Moteriya, P.; Chanda, S. Green synthesis of silver nanoparticles from marigold flower and its synergistic antimicrobial potential. Arab. J. Chem. 2015, 8, 732–741. [Google Scholar] [CrossRef]
- Jayaprakash, N.; Judith Vijaya, J.; Kaviyarasu, K.; Kombaiah, K.; Kennedy, L.J.; Ramalingam, R.J.; Munusamy, M.A.; Al-Lohedan, H.A. Green synthesis of Ag nanoparticles using tamarind fruit extract for antibacterial studies. J. Photochem. Photobiol. B Biol. 2017, 169, 178–185. [Google Scholar] [CrossRef]
- Alharbi, N.S.; Alsubhi, N.S.; Felimban, A.I. Green synthesis of silver nanoparticles using medicinal plants: Characterization and application. J. Radiat. Res. Appl. Sci. 2022, 15, 109–124. [Google Scholar] [CrossRef]
- Dawadi, S.; Katuwal, S.; Gupta, A.; Lamichhane, U.; Thapa, R.; Jaisi, S.; Lamicchane, G.; Bhattarai, D.P.; Parajuli, N. Current research on silver nanoparticles: Synthesis, characterization, and applications. J. Nanomater. 2021, 2021, 687290. [Google Scholar] [CrossRef]
- Velgosova, O.; Mačák, L.; Lisnichuk, M.; Vojtko, M. Synthesis and analysis of polymorphic silver nanoparticles and their incorporation into the polymer matrix. Polymers 2022, 14, 2666. [Google Scholar] [CrossRef]
- Ahmad, H. Celluloses as support materials for antibacterial agents: A review. Cellulose 2021, 28, 2715–2761. [Google Scholar] [CrossRef]
- Belattmania, B.; Rahou, A.; Smaili, A. Biosynthesis and characterization of silver nanoparticles using sodium alginate from the invasive macroalga Sargassum muticum. Bionanoscience 2018, 8, 617–623. [Google Scholar] [CrossRef]
- Bakshi, P.S.; Selvakumar, D.; Kadirvelu, K.; Kumar, N.S. Chitosan as an environment friendly biomaterial: A review on recent modifications and applications. Int. J. Biol. Macromol. 2020, 150, 1072–1083. [Google Scholar] [CrossRef]
- Khubiev, O.M.; Egorov, A.R.; Kirichuk, A.A.; Khrustalev, V.N.; Tskhovrebov, A.G.; Kritchenkov, A.S. Chitosan-based antibacterial films for biomedical and food applications. Int. J. Mol. Sci. 2023, 24, 10738. [Google Scholar] [CrossRef] [PubMed]
- Azmana, M.; Mahmood, S.; Hilles, A.R.; Rahman, A.; Arifin, M.A.B.; Ahmed, S. A review on chitosan and chitosan-based bionanocomposites: Promising material for combatting global issues and its applications. Int. J. Biol. Macromol. 2021, 185, 832–848. [Google Scholar] [CrossRef] [PubMed]
- Kadam, D.; Momin, B.; Palamthodi, S.; Lele, S.S. Physicochemical and functional properties of chitosan-based nanocomposite films incorporated with biogenic silver nanoparticles. Carbohydr. Polym. 2019, 211, 124–132. [Google Scholar] [CrossRef]
- Marin-Silva, D.A.; Romano, N.; Damonte, L.; Giannuzzi, L.; Pinotti, A. Hybrid materials based on chitosan functionalized with green synthesized copper nanoparticles: Physico-chemical and antimicrobial analysis. Int. J. Biol. Macromol. 2023, 242, 124898. [Google Scholar] [CrossRef]
- Lu, H.; Wang, J.; Stoller, M.; Wang, T.; Bao, Y.; Hao, H. An overview of nanomaterials for water and wastewater treatment. Adv. Mater. Sci. Eng. 2016, 2016, 4964828. [Google Scholar] [CrossRef]
- Chatterjee, P.; Ghangrekar, M.M.; Rao, S. Disinfection of secondary treated sewage using chitosan beads coated with ZnO-Ag nanoparticles to facilitate reuse of treated water. J. Chem. Technol. Biotechnol. 2017, 92, 2334–2341. [Google Scholar] [CrossRef]
- Olajire, A.A.; Bamigbade, L.A. Green synthesis of chitosan-based iron@silver nanocomposite as adsorbent for wastewater treatment. Water Resour. Ind. 2021, 26, 100158. [Google Scholar] [CrossRef]
- Sartori, P.; Delamare, A.P.L.; Machado, G.; Devine, D.M.; Crespo, J.S.; Giovanela, M. Synthesis and characterization of silver nanoparticles for the preparation of chitosan pellets and their application in industrial wastewater disinfection. Water 2023, 15, 190. [Google Scholar] [CrossRef]
- Vidinamo, F.; Fawzia, S.; Karim, M.A. Effect of drying methods and storage with agro-ecological conditions on phytochemicals and antioxidant activity of fruits: A review. Crit. Rev. Food Sci. Nutr. 2022, 62, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Mariadoss, A.V.A.; Ramachandran, V.; Shalini, V.; Agilan, B.; Franklin, J.H.; Sanjay, K.; Alaa, Y.G.; Tawfiq, M.A.-A.; Ernest, D. Green synthesis, characterization and antibacterial activity of silver nanoparticles by Malus domestica and its cytotoxic effect on (MCF-7) cell line. Microb. Pathog. 2019, 135, 103609. [Google Scholar] [CrossRef]
- Abbasi, Z.; Feizi, S.; Taghipour, E.; Ghadam, P. Green synthesis of silver nanoparticles using aqueous extract of dried Juglans regia green husk and examination of its biological properties. Green Process. Synth. 2017, 6, 477–485. [Google Scholar] [CrossRef]
- Mohammadi, F.; Yousefi, M.; Ghahremanzadeh, R. Green synthesis, characterization, and antimicrobial activity of silver nanoparticles (AgNPs) using leaves and stems extract of some plants. Adv. J. Chem. Sect. A 2019, 2, 266–275. [Google Scholar] [CrossRef]
- Ravichandran, V.; Vasanthi, S.; Shalini, S.; Shah, S.A.A.; Tripathy, M.; Paliwal, N. Green synthesis, characterization, antibacterial, antioxidant and photocatalytic activity of Parkia speciosa leaves extract mediated silver nanoparticles. Results Phys. 2019, 15, 102565. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Pires, J.S.; Torres, P.B.; dos Santos, D.Y.A.C.; Chow, F. Ensaio em Microplaca de Substâncias Redutoras Pelo Método do Folin-Ciocalteu Para Extratos de Algas; Instituto de Biociências, Universidade de São Paulo: São Paulo, Brazil, 2017; ISBN 978-85-85658-70-0. [Google Scholar]
- Yamaguchi, T.; Takamura, H.; Matoba, T.; Terao, J. HPLC method for evaluation of the free radical-scavenging activity of foods by using 1,1-diphenyl-2-picrylhydrazyl. Biosci. Biotechnol. Biochem. 1998, 62, 1201–1204. [Google Scholar] [CrossRef]
- Vanlalveni, C.; Lallianrawna, S.; Biswas, A.; Selvaraj, M.; Changmai, B.; Rokhum, S.L. Green synthesis of silver nanoparticles using plant extracts and their antimicrobial activities: A review of recent literature. RSC Adv. 2021, 11, 2804–2837. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 12th ed.; CLSI: Wayne, PA, USA, 2024; pp. 1–118. [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Disk Susceptibility Tests, 14th ed.; CLSI: Wayne, PA, USA, 2024; pp. 1–96. [Google Scholar]
- American Public Health Association (APHA); American Water Works Association (AWWA); Water Environment Federation (WEF). Standard Methods for the Examination of Water and Wastewater, 24th ed.; APHA/AWWA/WEF: Washington, DC, USA, 2022; pp. 1–1516. [Google Scholar]
- CONAMA. Resolução nº 430, de 13 de maio de 2011; Ministério do Meio Ambiente—Conselho Nacional do Meio Ambiente (Brasil): Brasília, Brazil, 2011; Available online: https://www.legisweb.com.br/legislacao/?id=114770 (accessed on 22 June 2025).
- Chowdhury, A.; Tuhin, M.N.; Ahmad, M.; Quader, M.F.B. From herb to dessert: Unveiling Swertia chirata’s impact on orange jelly dynamics. Health Dyn. 2024, 1, 178–186. [Google Scholar] [CrossRef]
- Alam, M. Analyses of biosynthesized silver nanoparticles produced from strawberry fruit pomace extracts in terms of biocompatibility, cytotoxicity, antioxidant ability, photodegradation, and in-silico studies. J. King Saud Univ. Sci. 2022, 34, 102327. [Google Scholar] [CrossRef]
- Lomelí-Rosales, D.A.; Zamudio-Ojeda, A.; Reyes-Maldonado, O.K.; López-Reyes, M.E.; Basulto-Padilla, G.C.; Lopez-Naranjo, E.J.; Zuñiga-Mayo, V.M.; Velázquez-Juárez, G. green synthesis of gold and silver nanoparticles using leaf extract of Capsicum chinense plant. Molecules 2022, 27, 1692. [Google Scholar] [CrossRef]
- Serna, D.M.O.; Martínez, J.H.I. Phenolics and polyphenols from Melastomataceae species. Molecules 2015, 20, 17818–17847. [Google Scholar] [CrossRef] [PubMed]
- Abbas, R.; Luo, J.; Qi, X.; Naz, A.; Khan, I.A.; Liu, H.; Yu, S.; Wei, J. Silver nanoparticles: Synthesis, structure, properties and applications. Nanomaterials 2024, 14, 1425. [Google Scholar] [CrossRef] [PubMed]
- Gontijo, L.A.P.; Raphael, E.; Santos Ferrari, D.P.; Ferrari, J.L.; Lyon, J.P.; Schiavon, M.A. pH effect on the synthesis of different size silver nanoparticles evaluated by DLS and their size-dependent antimicrobial activity. Rev. Mater. 2020, 25, e-12845. [Google Scholar] [CrossRef]
- Meshram, S.M.; Bonde, S.R.; Gupta, I.R.; Gade, A.K.; Rai, M.K. Green synthesis of silver nanoparticles using white sugar. IET Nanobiotechnol. 2013, 7, 28–32. [Google Scholar] [CrossRef]
- Anigol, L.B.; Charantimath, J.S.; Gurubasavaraj, P.M. Effect of Concentration and ph on the size of silver nanoparticles synthesized by green chemistry. Org. Med. Chem. Int. J. 2017, 3, 555622. [Google Scholar] [CrossRef]
- Chutrakulwong, F.; Thamaphat, K. Green synthesis of silver nanoparticles using durian rind extract and optical characteristics of surface plasmon resonance–based optical sensor for the detection of hydrogen peroxide. Green Process. Synth. 2023, 12, 20230070. [Google Scholar] [CrossRef]
- Linnert, T.; Mulvaney, P.; Henglein, A.; Weller, H. Long-lived nonmetallic silver clusters in aqueous solution: Preparation and photolysis. J. Am. Chem. Soc. 1990, 112, 4657–4664. [Google Scholar] [CrossRef]
- Ansari, M.A.; Kalam, A.; Al-Sehemi, A.G.; Alomary, M.N.; AlYahya, S.; Aziz, M.K.; Srivastava, S.; Alghamdi, S.; Akhtar, S.; Almalki, H.D. Counteraction of Biofilm Formation and Antimicrobial Potential of Terminalia catappa Functionalized Silver Nanoparticles Against Candida albicans and Multidrug-Resistant Gram-Negative and Gram-Positive Bacteria. Antibiotics 2021, 10, 725. [Google Scholar] [CrossRef]
- Salayová, A.; Bedlovičová, Z.; Daneu, N.; Baláž, M.; Lukáčová Bujňáková, Z.; Balážová, Ľ.; Tkáčiková, Ľ. Green synthesis of silver nanoparticles with antibacterial activity using various medicinal plant extracts: Morphology and antibacterial efficacy. Nanomaterials 2021, 11, 1005. [Google Scholar] [CrossRef]
- Reddy, N.V.; Li, H.; Hou, T.; Bethu, M.S.; Ren, Z.; Zhang, Z. Phytosynthesis of silver nanoparticles using Perilla frutescens leaf extract: Characterization and evaluation of antibacterial, antioxidant, and anticancer activities. Int. J. Nanomed. 2021, 16, 15–29. [Google Scholar] [CrossRef] [PubMed]
- Riaz, M.; Mutreja, V.; Sareen, S.; Ahmad, B.; Faheem, M.; Zahid, N.; Jabbour, G.; Park, J. Exceptional antibacterial and cytotoxic potency of monodisperse greener AgNPs prepared under optimized pH and temperature. Sci. Rep. 2021, 11, 2866. [Google Scholar] [CrossRef]
- Wypij, M.; Czarnecka, J.; Dahm, H.; Rai, M. Synthesis, Characterization and evaluation of antimicrobial and cytotoxic activities of biogenic silver nanoparticles synthesized from Streptomyces xinghaiensis OF1 strain. World J. Microbiol. Biotechnol. 2018, 34, 23. [Google Scholar] [CrossRef]
- Alahmad, A.; Al-Zereini, W.A.; Hijazin, T.J.; Al-Madanat, O.Y.; Alghoraibi, I.; Al-Qaralleh, O.; Al-Qaraleh, S.; Feldhoff, A.; Walter, J.-G.; Scheper, T. Green synthesis of silver nanoparticles using Hypericum perforatum L. aqueous extract with the evaluation of its antibacterial activity against clinical and food pathogens. Pharmaceutics 2022, 14, 1104. [Google Scholar] [CrossRef]
- Devanesan, S.; AlSalhi, M.S. Green synthesis of silver nanoparticles using the flower extract of Abelmoschus esculentus for cytotoxicity and antimicrobial studies. Int. J. Nanomed. 2021, 16, 3343–3356. [Google Scholar] [CrossRef]
- Abdullah Al Balushi, K.S.; Devi, G.; Al Gharibi, A.S.R.K.; Adeeb, M.A.S.; Al Hudaifi, A.S.M.; Al Shabibi, S.S.K. Extraction of bio polymers from crustacean shells and its application in refinery wastewater treatment. Walailak J. Sci. Technol. 2021, 18, 11543. [Google Scholar] [CrossRef]
- Grabska-Zielińska, S.; Sionkowska, A.; Olewnik-Kruszkowska, E.; Reczyńska, K.; Pamuła, E. Is Dialdehyde chitosan a good substance to modify physicochemical properties of biopolymeric materials? Int. J. Mol. Sci. 2021, 22, 3391. [Google Scholar] [CrossRef] [PubMed]
- Senthilkumar, P.; Yaswant, G.; Kavitha, S.; Chandramohan, E.; Kowsalya, G.; Vijay, R.; Sudhagar, B.; Kumar, D.S.R.S. Preparation and characterization of hybrid chitosan–silver nanoparticles (Chi-Ag NPs): A potential antibacterial agent. Int. J. Biol. Macromol. 2019, 141, 290–298. [Google Scholar] [CrossRef]
- Fernandes Queiroz, M.; Melo, K.R.T.; Sabry, D.A.; Sassaki, G.L.; Rocha, H.A.O. Does the use of chitosan contribute to oxalate kidney stone formation? Mar. Drugs 2015, 13, 141–158. [Google Scholar] [CrossRef] [PubMed]
- Panda, P.K.; Yang, J.; Chang, Y. Water-induced shape memory behavior of poly (vinyl alcohol) and p-coumaric acid-modified water-soluble chitosan blended membrane. Carbohydr. Polym. 2021, 257, 117633. [Google Scholar] [CrossRef] [PubMed]
- Elmehbad, N.Y.; Mohamed, N.A. Designing, Preparation and evaluation of the antimicrobial activity of biomaterials based on chitosan modified with silver nanoparticles. Int. J. Biol. Macromol. 2020, 151, 92–103. [Google Scholar] [CrossRef]
- An, J.; Luo, Q.; Yuan, X.; Wang, D.; Li, X. Preparation and characterization of silver–chitosan nanocomposite particles with antimicrobial activity. J. Appl. Polym. Sci. 2010, 120, 3180–3189. [Google Scholar] [CrossRef]
- Aranaz, I.; Navarro-García, F.; Morri, M.; Acosta, N.; Casettari, L.; Heras, A. Evaluation of Chitosan Salt Properties in the Production of AgNPs Materials with Antibacterial Activity. Int. J. Biol. Macromol. 2023, 235, 123849. [Google Scholar] [CrossRef]
- Chen, J.; Fan, L.; Yang, C.; Wang, S.; Zhang, M.; Xu, J.; Luo, S. Facile synthesis of Ag nanoparticles-loaded chitosan antibacterial nanocomposite and its application in polypropylene. Int. J. Biol. Macromol. 2020, 161, 1286–1295. [Google Scholar] [CrossRef]
- Raghavendra, G.M.; Jung, J.; Kim, D.; Seo, J. Microwave-assisted antibacterial chitosan-silver nanocomposite films. Int. J. Biol. Macromol. 2016, 84, 281–288. [Google Scholar] [CrossRef]
- Silhavy, T.J.; Kahne, D.; Walker, S. The bacterial cell envelope. Cold Spring Harb. Perspect. Biol. 2010, 2, a000414. [Google Scholar] [CrossRef]
- Pham, X.N.; Nguyen, H.T.; Pham, N.T. Green synthesis and antibacterial activity of HAp@Ag nanocomposite using Centella asiatica (L.) urban extract and eggshell. Int. J. Biomater. 2020, 2020, 841221. [Google Scholar] [CrossRef]
- Kaliaperumal, K.; Subramanian, K.; Thirunavukkarasu, R.; Varadharajan, R.K.; Binsuwaidan, R.; Alabdallah, N.M.; Alshammari, N.; Saeed, M.; Anbarasu, K.; Karunakaran, R. Antibacterial wound dressing with hydrogel from chitosan and polyvinyl alcohol from the red cabbage extract loaded with silver nanoparticles. Green Process. Synth. 2023, 12, 20230035. [Google Scholar] [CrossRef]
- Raota, C.S.; Cerbaro, A.F.; Salvador, M.; Delamare, A.P.L.; Echeverrigaray, S.; Crespo da Silva, J.; Silva, T.B.; Giovanela, M. Green synthesis of silver nanoparticles using an extract of Ives cultivar (Vitis labrusca) pomace: Characterization and application in wastewater disinfection. J. Environ. Chem. Eng. 2019, 7, 103383. [Google Scholar] [CrossRef]
- Zarpelon, F.; Galiotto, D.; Aguzzoli, C.; Carli, L.N.; Figueroa, C.A.; Baumvol, I.J.R.; Machado, G.; Crespo da Silva, J.; Giovanela, M. Removal of coliform bacteria from industrial wastewaters using polyelectrolytes/silver nanoparticles self-assembled thin films. Environ. Chem. Eng. 2016, 4, 137–146. [Google Scholar] [CrossRef]
- Lovatel, R.H.; Neves, R.M.; Oliveira, G.R.; Mauler, R.S.; Crespo, J.S.; Carli, L.N.; Giovanela, M. Disinfection of biologically treated industrial wastewater using montmorillonite/alginate/nanosilver hybrids. Water Process Eng. 2015, 7, 273–279. [Google Scholar] [CrossRef]








| Synthesis | Temperature (°C) | pH | [AgNO3] (mol/L) |
|---|---|---|---|
| 1 | 25 | 3.5–4.0 | 0.001 |
| 2 | 0.01 | ||
| 3 | 0.05 | ||
| 4 | 1.0 | ||
| 5 | 25 | 5.0 | 0.01 |
| 6 | 6.0 | ||
| 7 | 7.0 | ||
| 8 | 8.0 | ||
| 9 | 9.0 | ||
| 10 | 10.0 | ||
| 11 | 11.0 | ||
| 12 | 12.0 | ||
| 13 | 35 | 3.5–4.0 | 0.01 |
| 14 | 50 |
| Synthesis | Parameter | Variation | Main Observations |
|---|---|---|---|
| 1 | [AgNO3] (mol/L) | 0.001 | Color change from pink to yellowish-brown coloration. Characteristic SPR band at 410–420 nm detected using AgNO3 0.01 mol/L; absent at other concentrations. Salt concentration is a relevant parameter, but it must be optimized in conjunction with a suitable reagent ratio to be effective and to ensure synthesis efficiency |
| 2 | 0.01 | ||
| 3 | 0.05 | ||
| 4 | 1.0 | ||
| 5 | pH | 5.0 | Color change from pink to yellowish-brown coloration. Characteristic SPR band at 410–420 nm. Synthesis successful. pH is the most influential factor, favoring the formation of smaller and more uniform NPs, indicating a positive influence on the final product quality |
| 6 | 6.0 | ||
| 7 | 7.0 | ||
| 8 | 8.0 | ||
| 9 | 9.0 | ||
| 10 | 10.0 | ||
| 11 | 11.0 | ||
| 12 | 12.0 | ||
| 13 | Temperature (°C) | 35 | Color change from pink to yellowish-brown coloration. Characteristic SPR band at 410–420 nm and an additional band at 300–400 nm (silver oxides) at 35 and 50 °C. Synthesis not completely successful. Temperature, although relevant, requires simultaneous optimization with other parameters to ensure process selectivity and stability |
| 14 | 50 |
| Bacterial Strain | Average Diameter of Inhibition Zone (mm) |
|---|---|
| S. aureus | 11 ± 1 |
| E. coli | 9 ± 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jacquot, A.J.P.; de Souza, W.V.; Machado, G.; Roesch-Ely, M.; Crespo, J.d.S.; Bortoluz, J.; Giovanela, M. Chitosan Composites Functionalized with Green-Synthesized Silver Nanoparticles from Manacá-da-Serra Flowers for the Disinfection of Industrial Wastewater. Processes 2025, 13, 3622. https://doi.org/10.3390/pr13113622
Jacquot AJP, de Souza WV, Machado G, Roesch-Ely M, Crespo JdS, Bortoluz J, Giovanela M. Chitosan Composites Functionalized with Green-Synthesized Silver Nanoparticles from Manacá-da-Serra Flowers for the Disinfection of Industrial Wastewater. Processes. 2025; 13(11):3622. https://doi.org/10.3390/pr13113622
Chicago/Turabian StyleJacquot, Axel John Pascal, Wellington Vieira de Souza, Giovanna Machado, Mariana Roesch-Ely, Janaina da Silva Crespo, Jordana Bortoluz, and Marcelo Giovanela. 2025. "Chitosan Composites Functionalized with Green-Synthesized Silver Nanoparticles from Manacá-da-Serra Flowers for the Disinfection of Industrial Wastewater" Processes 13, no. 11: 3622. https://doi.org/10.3390/pr13113622
APA StyleJacquot, A. J. P., de Souza, W. V., Machado, G., Roesch-Ely, M., Crespo, J. d. S., Bortoluz, J., & Giovanela, M. (2025). Chitosan Composites Functionalized with Green-Synthesized Silver Nanoparticles from Manacá-da-Serra Flowers for the Disinfection of Industrial Wastewater. Processes, 13(11), 3622. https://doi.org/10.3390/pr13113622








