Phenolic Compounds, Fatty Acid Composition, and Antioxidant Activities of Some Flaxseed (Linum usitatissimum L.) Varieties: A Comprehensive Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Flaxseed Material and Chemicals
2.2. Extraction Procedure
2.3. Total Phenolic Content Analysis (TPC)
2.4. Total Flavonoid Content (TFC) Analysis
2.5. Determination of Cu2+ Reduction Capacity
2.6. Determination of Fe2+-Chelating Activity
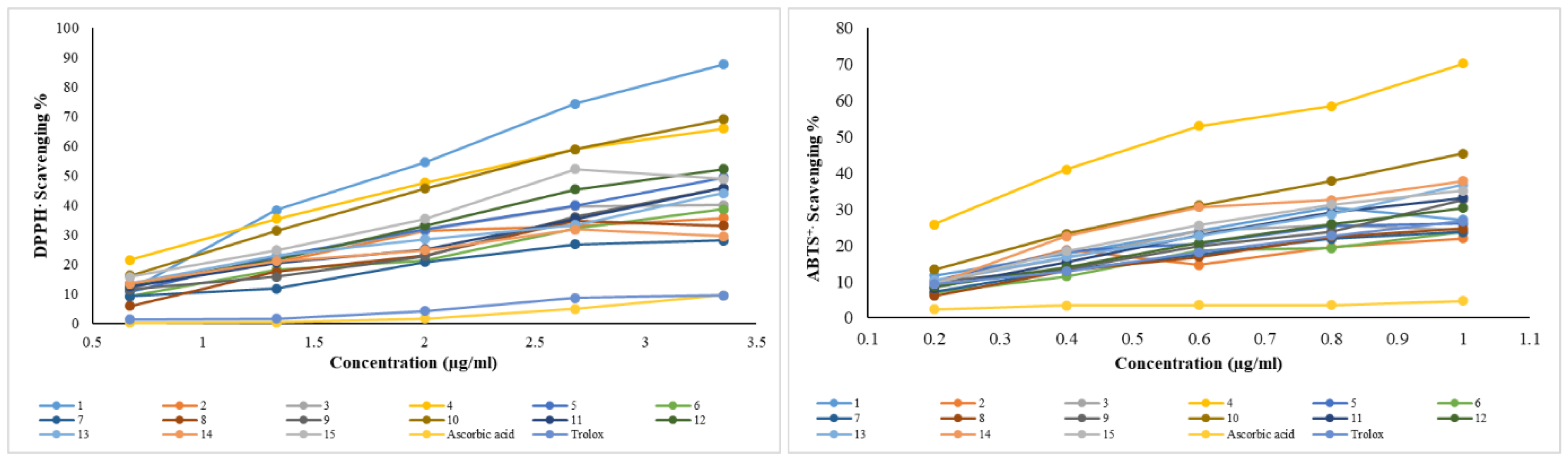
2.7. 1,1-diphenyl-2-picryl Hydrazyl (DPPH·) Radical Scavenging Activity
2.8. 2,2′-azino-bis [3-ethylbenzothiazoline-6-sulphonic Acid] (ABTS+) Radical Scavenging Activity
2.9. Analyzes of Fatty Acid and Phenolic Content
2.9.1. Extraction of Flaxseed Oils
2.9.2. Yield of Flaxseed Oil
2.10. Gas Chromatography Flame Ionization Detector (GC–FID) Analysis for of Fatty Acid Composition
2.11. High-Performance Liquid Chromatography (HPLC) Analysis for Phenolic Composition
2.12. Statistical Analysis
3. Results
3.1. Total Phenolic/Flavonoid Content and DPPH/ABTS+ Radical Scavenging Activities of Flaxseed Extracts
3.2. Fatty Acid Composition in Flaxseed Extract and Correlation Analysis
3.3. Phenolic Compounds Profile in Flaxseed Extract and Correlation Analysis
4. Discussion
4.1. Total Phenolic and Flavonoid Content
4.2. Individual Phenolic Compounds
4.3. Antioxidant Activities
4.4. Oil Yield and Content as Well as Individual Fatty Acids
4.5. Highlights and Limitations of the Present Study
5. Conclusions
Supplementary Materials
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zare, S.; Mirlohi, A.; Sabzalian, M.R.; Saeidi, G.; Koçak, M.Z.; Hano, C. Water Stress and Seed Color Interacting to Impact Seed and Oil Yield, Protein, Mucilage, and Secoisolariciresinol Diglucoside Content in Cultivated Flax (Linum usitatissimum L.). Plants 2023, 12, 1632. [Google Scholar] [CrossRef] [PubMed]
- Özcan, M.M.; Uslu, N. Investigation of changes in some chemical properties, bioactive compounds, antioxidant activity, phenolic and fatty acid profiles of flaxseed and oils. J. Food Process. Preserv. 2022, 46, e17091. [Google Scholar] [CrossRef]
- Goudenhooft, C.; Bourmaud, A.; Baley, C. Conventional or greenhouse cultivation of flax: What influence on the number and quality of flax fibers? Ind. Crops Prod. 2018, 123, 111–117. [Google Scholar] [CrossRef]
- Koçak, M.Z.; Göre, M.; Kurt, O. The effect of different salinity levels on germination development of some flax (Linum usitatissimum L.). Varieties TURJAF 2022, 10, 657–662. [Google Scholar]
- Moawia, R.M.; Nasef, M.M.; Mohamed, N.H.; Ripin, A.; Farag, H. Production of biodiesel from cottonseed oil over aminated flax fibres catalyst: Kinetic and thermodynamic behaviour and biodiesel properties. Adv. Chem. Eng. Sci. 2019, 9, 281–298. [Google Scholar] [CrossRef]
- Pisupati, A.; Willaert, L.; Goethals, F.; Uyttendaele, W.; Park, C.H. Variety and growing condition effect on the yield and tensile strength of flax fibers. Ind. Crops Prod. 2021, 170, 113736. [Google Scholar] [CrossRef]
- Parikh, M.; Pierce, G.N. Dietary flaxseed: What we know and don’t know about its effects on cardiovascular disease. Can. J. Physiol. Pharmacol. 2019, 97, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Buckner, A.L.; Buckner, C.A.; Montaut, S.; Lafrenie, R.M. Treatment with flaxseed oil induces apoptosis in cultured malignant cells. Heliyon 2019, 5, e02251. [Google Scholar] [CrossRef] [PubMed]
- Barre, D.E.; Mizier-Barre, K.A.; Griscti, O.; Hafez, K. High dose flaxseed oil supplementation may affect fasting blood serum glucose management in human type 2 diabetics. J. Oleo Sci. 2008, 57, 269–273. [Google Scholar] [CrossRef][Green Version]
- Wang, S.; Zhang, Z.S.; Zhang, T.F.; Xue, D.W. Extraction and characterization of flaxseed oil obtained with subcritical n-butane. J. Oleo Sci. 2020, 69, 1011–1020. [Google Scholar] [CrossRef]
- Akl, E.M.; Mohamed, S.S.; Hashem, A.I.; Taha, F.S. Biological activities of phenolic compounds extracted from flaxseed meal. Bull. Natl. Res. Cent. 2020, 44, 27. [Google Scholar] [CrossRef]
- Wang, H.; Qiu, C.; Abbasi, A.M.; Chen, G.; You, L.; Li, T.; Liu, R.H. Effect of germination on vitamin C, phenolic compounds and antioxidant activity in flaxseed (Linum usitatissimum L.). Int. J. Food Sci. Technol. 2015, 50, 2545–2553. [Google Scholar] [CrossRef]
- Beema, N.; Mukkamula, N.; Mothuku, S.; Thumu, R.; Azmeera, T.; Biman, K.K. Comparative analysis of physico-chemical properties and fatty acid composition of linseed (Linum usitatissimum L.) oils of Indian accessions. J. Appl. Biol. Biotechnol. 2022, 11, 80–87. [Google Scholar] [CrossRef]
- Güller, U.; Güller, P.; Çiftci, M. Radical Scavenging and Antiacetylcholinesterase Activities of Ethanolic Extracts of Carob, Clove, and Linden. Altern. Ther. Health Med. 2021, 27, 33–37. [Google Scholar]
- Cirak, C.; Radusiene, J. Factors affecting the variation of bioactive compounds in Hypericum species. Biol. Futur. 2019, 70, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventŏs, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu Reagent. Meth. Enzymol. 1999, 299, 152–178. [Google Scholar]
- Park, Y.K.; Koo, M.H.; Ikegaki, M.; Contado, J.L. Comparison of the flavonoid aglycone contents of Apis mellifera propolis from various regions of Brazil. Arq Bio. Tech. 1997, 40, 97–106. [Google Scholar]
- Köksal, E.; Gulcin, I. Antioxidant activity of cauliflower (Brassica oleracea L.). Turk. J. Agric. For. 2008, 32, 65–78. [Google Scholar]
- Dinis, T.C.P.; Madeira, V.M.C.; Almeida, L.M. Action of phenolic derivates (acetoaminophen, salycilate, and 5-aminosalycilate) as inhibitors of membrane lipid peroxidation and as peroxyl radical scavengers. Arch. Biochem. Biophys. 1994, 315, 161–169. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- AOCS. Official and Tentative Methods of the American Oil Chemists’ Society; American Oil Chemists’ Society: Urbana, IL, USA, 1997; Volume 35. [Google Scholar]
- Mubiru, E.; Shrestha, K.; Papastergiadis, A.; De Meulenaer, B. Improved gas chromatography-flame ionization detector analytical method for the analysis of epoxy fatty acids. J. Chromatogr. A 2013, 1318, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Corbin, C.; Fidel, T.; Leclerc, E.A.; Barakzoy, E.; Sagot, N.; Falguiéres, A.; Hano, C. Development and validation of an efficient ultrasound assisted extraction of phenolic compounds from flax (Linum usitatissimum L.) seeds. Ultrason. Sonochem. 2015, 26, 176–185. [Google Scholar] [CrossRef]
- Czemplik, M.; Korzun-Chłopicka, U.; Szatkowski, M.; Działo, M.; Szopa, J.; Kulma, A. Optimization of phenolic compounds extraction from flax shives and their effect on human fibroblasts. eCAM 2017, 3526392. [Google Scholar] [CrossRef] [PubMed]
- Punia, J.; Deen, M.K. Comparative evaluation of phenols, flavonoids and antioxidant activity of flax seed from two locations. Asian J. Chem. 2016, 28, 2038. [Google Scholar] [CrossRef]
- Mirończuk-Chodakowska, I.; Witkowska, A.M.; Zujko, M.E. Endogenous non-enzymatic antioxidants in the human body. Adv. Med. Sci. 2018, 63, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Belinskaia, D.A.; Voronina, P.A.; Shmurak, V.I.; Vovk, M.A.; Batalova, A.A.; Jenkins, R.O.; Goncharov, N.V. The universal soldier: Enzymatic and non-enzymatic antioxidant functions of serum albumin. Antioxidants 2020, 9, 966. [Google Scholar] [CrossRef] [PubMed]
- Bourgaud, F.; Gravot, A.; Milesi, S.; Gontier, E. Production of plant secondary metabolites: A historical perspective. Plant Sci. 2001, 161, 839–851. [Google Scholar] [CrossRef]
- Yang, L.; Wen, K.S.; Ruan, X.; Zhao, Y.X.; Wei, F.; Wang, Q. Response of plant secondary metabolites to environmental factors. Molecules 2018, 23, 762. [Google Scholar] [CrossRef] [PubMed]
- Cetinkaya, H.; Kulak, M.; Karaman, M.; Karaman, H.S.; Kocer, F. Flavonoid accumulation behavior in response to the abiotic stress: Can a uniform mechanism be illustrated for all plants. Flavonoids-Biosynth. Hum. Health 2017, 151–165. [Google Scholar] [CrossRef]
- Alu’datt, M.H.; Rababah, T.; Ereifej, K.; Alli, I. Distribution, antioxidant and characterization of phenolic compounds in soybeans, flaxseed and olives. Food Chem. 2013, 139, 93–99. [Google Scholar] [CrossRef]
- Oomah, B.D.; Kenaschuk, E.O.; Mazza, G. Phenolic acids in flaxseed. J. Agric. Food Chem. 1995, 43, 2016–2019. [Google Scholar] [CrossRef]
- Herchi, W.; Sakouhi, F.; Arráez-Román, D.; Segura-Carretero, A.; Boukhchina, S.; Kallel, H.; Fernández-Gutierrez, A. Changes in the content of phenolic compounds in flaxseed oil during development. J. Am. Oil Chem. Soc. 2011, 88, 1135–1142. [Google Scholar] [CrossRef]
- Noreen, S.; Taufail, T.; Ain, H.B.U.; Khalid, W.; Hanif, A.; Ali, B.; Abeed, A.H. Assessment of Antioxidant Activities of Flaxseed (Linum usitatisimum L.) and Fennel Seed (Foeniculum vulgare Mill.) Extracts. Pol. J. Environ. Stud. 2024, 33, 1–8. [Google Scholar] [CrossRef]
- Tasneem, R.; Khan, H.M.S.; Rasool, F.; Khan, K.U.R.; Umair, M.; Esatbeyoglu, T.; Korma, S.A. Development of Phytocosmeceutical microemulgel containing flaxseed extract and its in vitro and in vivo characterization. Pharmaceutics 2022, 14, 1656. [Google Scholar] [CrossRef] [PubMed]
- Anwar, F.; Przybylski, R. Effect of solvents extraction on total phenolics and antioxidant activity of extracts from flaxseed (Linum usitatissimum L.). Acta Sci. Pol. Technol. Aliment. 2012, 11, 293–302. [Google Scholar]
- Chen, G.; Wang, H.; Zhang, X.; Yang, S.T. Nutraceuticals and functional foods in the management of hyperlipidemia. Crit. Rev. Food Sci. Nutr. 2014, 54, 1180–1201. [Google Scholar] [CrossRef] [PubMed]
- Noreen, S.; Tufail, T.; Bader, U.L.; Ain, H.; Ali, A.; Aadil, R.M.; Nemat, A.; Manzoor, M.F. Antioxidant activity and phytochemical analysis of fennel seeds and flaxseed. Food Sci. Nutr. 2023, 11, 1309–1317. [Google Scholar] [CrossRef]
- Kumar, S.; Abedin, M.M.; Singh, A.K.; Das, S. Role of phenolic compounds in plant-defensive mechanisms. J. Plant Growth Regul. 2020, 1, 517–532. [Google Scholar]
- Bertoni, C.; Abodi, M.; D’Oria, V.; Milani, G.P.; Agostoni, C.; Mazzocchi, A. Alpha-Linolenic Acid and Cardiovascular Events: A Narrative Review. Int. J. Mol. Sci. 2023, 24, 14319. [Google Scholar] [CrossRef]
- Waszkowiak, K.; Mikołajczak, B.; Gliszczyńska-Świgło, A.; Niedźwiedzińska, K. Effect of thermal pre-treatment on the phenolic and protein profiles and oil oxidation dynamics of golden flaxseeds. Int. J. Food Sci. Technol. 2020, 55, 1272–1280. [Google Scholar] [CrossRef]
- Gai, F.; Janiak, M.A.; Sulewska, K.; Peiretti, P.G.; Karamać, M. Phenolic compound profile and antioxidant capacity of flax (Linum usitatissimum L.) harvested at different growth stages. Molecules 2023, 28, 1807. [Google Scholar] [PubMed]
- Rocchetti, G.; Gregorio, R.P.; Lorenzo, J.M.; Barba, F.J.; Oliveira, P.G.; Prieto, M.A.; Lucini, L. Functional implications of bound phenolic compounds and phenolics–food interaction: A review. Compr. Rev. Food Sci. 2022, 21, 811–842. [Google Scholar] [CrossRef] [PubMed]
- Qiu, C.; Wang, H.; Guo, Y.; Long, S.; Wang, Y.; Abbasi, A.M.; Jarvis, D.I. Comparison of fatty acid composition, phytochemical profile and antioxidant activity in four flax (Linum usitatissimum L.) varieties. Oil Crop Sci. 2020, 5, 136–141. [Google Scholar] [CrossRef]
- Sokoła-Wysoczańska, E.; Wysoczański, T.; Wagner, J.; Czyż, K.; Bodkowski, R.; Lochyński, S.; Patkowska-Sokoła, B. Polyunsaturated fatty acids and their potential therapeutic role in cardiovascular system disorders—A review. Nutrients 2018, 10, 1561. [Google Scholar] [CrossRef]
- Rodriguez-Leyva, D.; Bassett, C.M.; McCullough, R.; Pierce, G.N. The cardiovascular effects of flaxseed and its omega-3 fatty acid, alpha-linolenic acid. Can. J. Cardiol. 2010, 26, 489–496. [Google Scholar] [CrossRef]

| Sample | Variety | Origin Country | Place of Supply | Breeding Season | Flower Color | Seed Color |
|---|---|---|---|---|---|---|
| M.Z.K.7601 | Karakız | Türkiye | Agricultural institute of Türkiye | Summer | White | Brown |
| M.Z.K.7602 | BonnyDoon | Avustralya | Agricultural institute of Türkiye | Summer | White | Brown |
| M.Z.K.7603 | Norman | Canada | Agricultural institute of Türkiye | Spring | Blue | Brown |
| M.Z.K.7604 | McGregor | United States of North Dakota | Agricultural institute of Türkiye | Summer | Blue | Brown |
| M.Z.K.7605 | Dillman | United States of North Dakota | Agricultural institute of Türkiye | Summer | Blue | Brown |
| M.Z.K.7606 | Michael | USA | Agricultural institute of Türkiye | Spring | Blue | Brown |
| M.Z.K.7607 | Konya Beyazı | Türkiye | Agricultural institute of Türkiye | Summer | Blue | Brown |
| M.Z.K.7608 | Florinda | Romania | Agricultural institute of Türkiye | Spring | Blue | Brown |
| M.Z.K.7609 | Lirina | Russian | Agricultural institute of Türkiye | Spring | White | Brown |
| M.Z.K.76010 | Nareum | USA | Agricultural institute of Türkiye | Spring | Blue | Brown |
| M.Z.K.76011 | Barbara | Romania | Agricultural institute of Türkiye | Spring | Blue | Brown |
| M.Z.K.76012 | Atalanta | Türkiye | Agricultural institute of Türkiye | Summer | Blue | Brown |
| M.Z.K.76013 | Milas | Türkiye | Agricultural institute of Türkiye | Spring | White | Brown |
| M.Z.K.76014 | Zoltan | Hungary | Agricultural institute of Türkiye | Summer | Blue | Brown |
| M.Z.K.76015 | Mcduff | Canada | Agricultural institute of Türkiye | Summer | Blue | Brown |
| Variety | Total Phenolic Content (mg GAE/g) | Total Flavonoid Content (mg QE/g) | IC50 for DPPH Radical Scavenging (µg/mL) | IC50 for ABTS Radical Scavenging (µg/mL) | IC50 for Ferrous Ion Chelating Activity (µg/mL) | Cupric Reducing Capacity (µmol TE/mg) |
|---|---|---|---|---|---|---|
| Karakız | 2117.4 ± 253.2 a–d | 442.14 ± 71.37 a–d | 1.89 | 1.88 | 1.56 | 10.04 |
| BonnyDoon | 761.9 ± 16.1 d | 176.25 ± 19.17 a–d | 4.75 | 3.21 | 1.91 | 10.35 |
| Norman | 768.7 ± 76.0 e | 286.98 ± 9.59 a–c | 3.91 | 2.19 | 1.59 | 2.23 |
| McGregor | 1808.2 ± 168.8 e | 390.17 ± 87.35 d | 2.24 | 0.61 | 1.92 | 11.78 |
| Dillman | 1127.1 ± 203.5 f | 217.93 ± 48.98 a–d | 3.35 | 2.15 | 0.92 | 8.82 |
| Michael | 613.6 ± 108.2 bc | 187.55 ± 9.59 ab | 4.41 | 2.24 | 2.01 | 6.34 |
| Konya Beyazı | 616.2 ± 100.9 d | 230.49 ± 44.74 a–c | 5.9 | 2.21 | 2.25 | 8.22 |
| Florinda | 702.6 ± 85.6 cd | 326.9 ± 74.56 a–c | 4.55 | 2.04 | 3.84 | 10.17 |
| Lirina | 689.4 ± 79.8 b | 338.20 ± 39.41 a | 3.78 | 1.68 | 2.74 | 7.99 |
| Nareum | 1303.2 ± 191.7 f | 586.01 ± 90.96 a–d | 2.29 | 1.10 | 3.71 | 11.12 |
| Barbara | 1802.1 ± 209.7 f | 396.20 ± 90.40 cd | 3.82 | 1.49 | 1.04 | 9.85 |
| Atalanta | 3164.6 ± 459.6 f | 372.09 ± 61.78 d | 3.11 | 1.69 | 2.3 | 10.00 |
| Milas | 2227.6 ± 398.5 f | 641.00 ± 90.72 d | 4.01 | 1.43 | 3.72 | 8.38 |
| Zoltan | 2045.6 ± 206.2 f | 595.05 ± 92.74 b–d | 6.03 | 1.30 | 2.3 | 10.25 |
| Mcduff | 2421.5 ± 189.2 a | 689.20 ± 98.21 a–d | 2.13 | 1.41 | 3.78 | 11.23 |
| Ascorbic acid | - | - | 15.51 | 20.49 | - | - |
| α-tocopherol | - | - | - | - | 12.46 | - |
| Trolox | - | - | 15.00 | 2.04 | 3.22 | - |
| Variety | Crude Oil Yield (g/100 g) | Oil Content (%) | Palmitic Acid (C16.0) | Stearic Acid (C18.0) | Oleic Acid (C18.1) | Linoleic Acid (C18.2) | α-Linolenic Acid (C18.3) |
|---|---|---|---|---|---|---|---|
| Karakız | 1.12 b | 19.6 ef | 11.65 a | 7.12 a | 24.75 ab | 14.83 c–e | 39.49 e |
| BonnyDoon | 2.11 a | 36.8 b | 6.93 d–f | 5.59 a–d | 21.61 c–e | 17.89 a | 47.95 cd |
| Norman | 0.89 b | 18.4 ef | 6.84 d–f | 5.62 a–d | 23.36 bc | 17.65 ab | 46.51 cd |
| McGregor | 2.14 a | 42.6 a | 17.59 a | 6.57 ab | 18.12 fg | 13.61 de | 39.21 e |
| Dillman | 0.91 b | 19.8 ef | 8.02 c–e | 5.33 b–e | 20.89 d–f | 17.25 ab | 47.87 cd |
| Michael | 0.82 b | 17.4 f | 6.55 d–f | 5.72 a–d | 20.27 d–g | 15.24 cd | 52.18 ab |
| Konya Beyazı | 0.93 b | 19.3 ef | 8.96 c | 4.73 c–e | 20.81 d–f | 17.44 ab | 46.62 cd |
| Florinda | 1.03 b | 20.6 e | 8.35 cd | 6.07 a–d | 18.74 fg | 16.22 a–c | 49.19 bc |
| Lirina | 1.81 a | 27.8 c | 5.76 f | 4.57 de | 20.55 d–f | 16.02 bc | 53.08 a |
| Nareum | 0.98 b | 19.7 ef | 5.73 f | 3.71 e | 19.88 f–g | 16.53 a–c | 54.13 a |
| Barbara | 1.94 a | 37.6 b | 11.04 a | 6.77 ab | 21.37 c–e | 13.23 e | 45.35 d |
| Atalanta | 0.96 b | 24.8 d | 6.34 ef | 5.34 b–d | 20.8 d–f | 13.3 e | 54.2 a |
| Milas | 0.97 b | 17.4 f | 6.55 d–f | 7.31 a | 26.17 a | 15.24 cd | 44.71 d |
| Zoltan | 1.05 b | 19.4 ef | 6.2 ef | 6.42 a–c | 23.91 b | 16.5 a–c | 46.94 cd |
| Mcduff | 1.48 a | 21.3 e | 6.26 ef | 6.97 ab | 22.64 b–d | 16.29 a–c | 47.81 cd |
| p-values | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 |
| Variety | Chlorogenic Acid | Catechine Hydrate | 4-Hydroxy Benzoic Acid | Vanillin | Rutin | Naringin | o-Coumaric Acid | Salicylic Acid | Resveratrol | Quercetin | t-Cinamic Acid | Naringenin |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Karakız | 1.88 de | 5.83 b | 0.00 d | 0.00 d | 0.00 f | 67.39 a | 3.11 a | 0.00 e | 1.79 c | 3.99 b | 9.51 a | 11.40 a |
| BonnyDoon | 12.08 a | 3.97 c | 1.38 c | 5.63 b | 4.09 b | 6.78 b | 0.00 d | 0.00 e | 0.00 e | 0.00 e | 7.79 bc | 4.01 b |
| Norman | 2.20 d | 3.62 cd | 7.09 a | 5.73 b | 1.26 e | 0.00 e | 0.00 d | 0.00 e | 1.94 bc | 0.00 e | 10.49 a | 9.45 a |
| McGregor | 7.56 a | 6.48 a | 0.00 d | 2.02 c | 0.00 f | 0.00 e | 0.00 d | 0.00 e | 1.02 d | 0.00 e | 8.66 a | 4.83 a |
| Dillman | 8.82 a | 9.33 a | 0.00 d | 7.09 a | 0.00 f | 0.00 e | 0.00 d | 0.00 e | 0.00 e | 0.00 e | 8.25 ab | 4.29 ab |
| Michael | 1.14 f | 10.21 a | 0.00 d | 0.00 d | 0.00 f | 6.83 b | 0.00 d | 6.37 b | 1.29 d | 1.57 d | 6.81 de | 2.57 c |
| Konya Beyazı | 6.87 a | 0.00 f | 0.00 d | 0.00 d | 24.46 a | 1.66 c | 2.65 b | 4.11 c | 8.03 a | 4.56 a | 6.88 de | 6.88 a |
| Florinda | 2.06 de | 1.48 e | 1.33 c | 0.00 d | 0.00 f | 0.00 e | 0.00 d | 0.00 e | 0.00 e | 1.36 d | 6.47 e | 8.89 a |
| Lirina | 3.22 c | 6.93 a | 1.45 c | 0.00 d | 3.46 c | 0.00 e | 0.00 d | 0.00 e | 0.00 e | 1.43 d | 8.95 a | 1.56 de |
| Nareum | 1.92 de | 3.60 cd | 0.00 d | 0.00 d | 0.00 f | 0.00 e | 0.00 d | 0.00 e | 1.06 d | 0.00 e | 7.35 cd | 2.08 cd |
| Barbara | 2.39 d | 3.24 d | 0.00 d | 0.00 d | 0.00 f | 0.00 e | 0.00 d | 0.00 e | 1.70 c | 0.00 e | 7.96 bc | 2.50 c |
| Atalanta | 5.05 b | 10.23 a | 0.00 d | 5.80 b | 1.86 d | 1.07 d | 0.00 d | 8.98 a | 2.24 b | 0.00 e | 12.52 a | 1.35 e |
| Milas | 1.54 ef | 3.58 cd | 0.00 d | 0.00 d | 0.00 f | 0.00 e | 0.00 d | 0.00 e | 0.00 e | 1.32 d | 6.21 e | 0.00 f |
| Zoltan | 6.62 a | 6.60 a | 1.78 b | 0.00 d | 0.00 f | 6.56 b | 0.00 d | 1.04 d | 1.01 d | 1.29 d | 5.20 f | 4.89 a |
| Mcduff | 1.13 f | 8.12 a | 0.00 d | 0.00 d | 8.65 a | 1.94 c | 1.53 c | 0.00 e | 1.03 d | 2.27 c | 10.28 a | 0.00 f |
| p-values | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koçak, M.Z. Phenolic Compounds, Fatty Acid Composition, and Antioxidant Activities of Some Flaxseed (Linum usitatissimum L.) Varieties: A Comprehensive Analysis. Processes 2024, 12, 689. https://doi.org/10.3390/pr12040689
Koçak MZ. Phenolic Compounds, Fatty Acid Composition, and Antioxidant Activities of Some Flaxseed (Linum usitatissimum L.) Varieties: A Comprehensive Analysis. Processes. 2024; 12(4):689. https://doi.org/10.3390/pr12040689
Chicago/Turabian StyleKoçak, Mehmet Zeki. 2024. "Phenolic Compounds, Fatty Acid Composition, and Antioxidant Activities of Some Flaxseed (Linum usitatissimum L.) Varieties: A Comprehensive Analysis" Processes 12, no. 4: 689. https://doi.org/10.3390/pr12040689
APA StyleKoçak, M. Z. (2024). Phenolic Compounds, Fatty Acid Composition, and Antioxidant Activities of Some Flaxseed (Linum usitatissimum L.) Varieties: A Comprehensive Analysis. Processes, 12(4), 689. https://doi.org/10.3390/pr12040689








