Diving into Fish Valorisation: Review Opportunities and Analyzing Azorean Fish Data
Abstract
1. Introduction
1.1. Blue Economy
1.2. Fish Industry in Portugal
1.3. Environmental Impacts Associated with Fish Industry
2. Gifts from the Sea: Azorean Fish
3. Fish Waste Valorisation
3.1. Fishery Environmental Impact
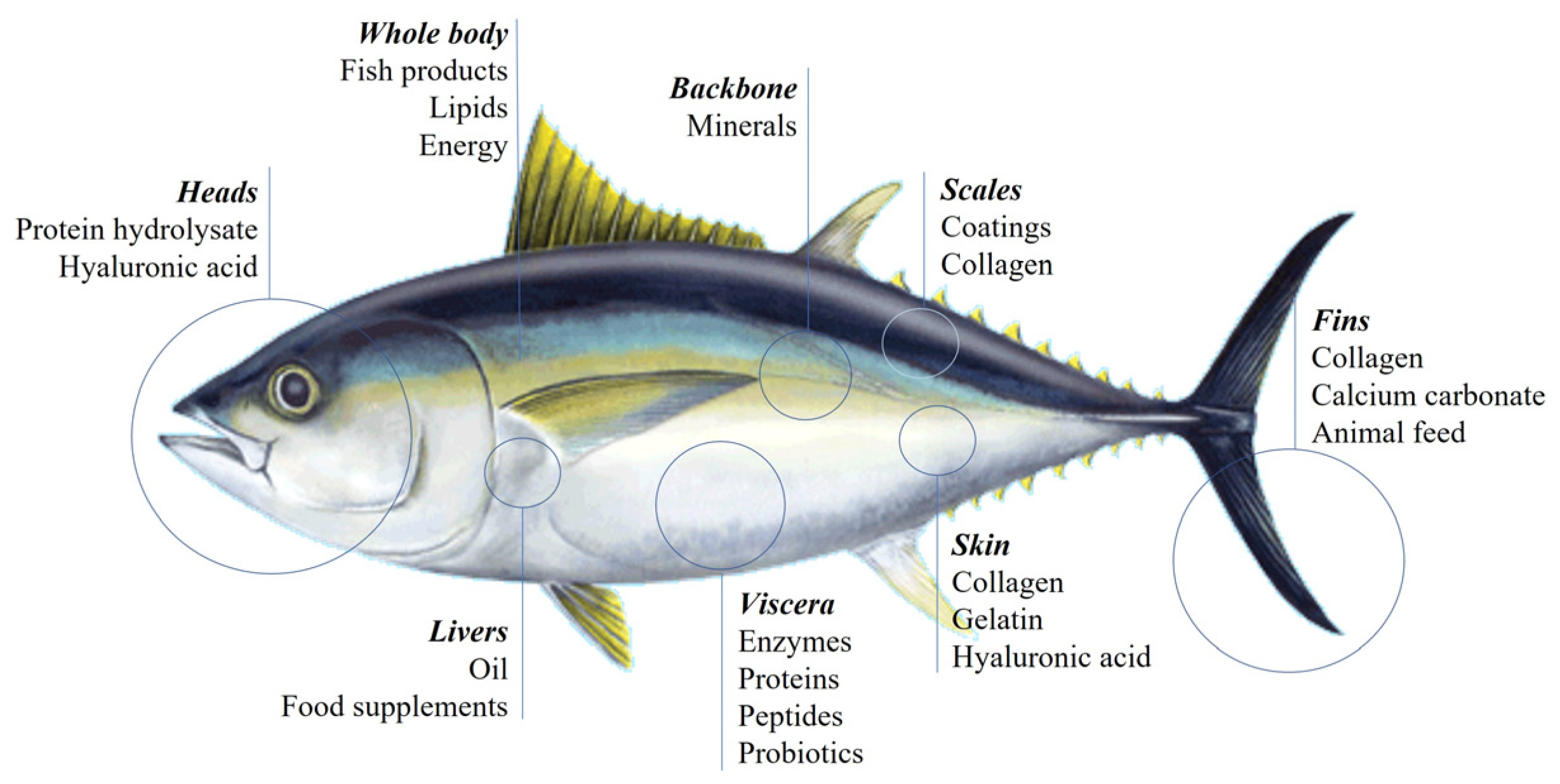
3.2. Biomass Valorisation
3.3. High-Added Value Biomaterials
3.3.1. Hydroxyapatite
3.3.2. Fish Protein Hydrolysate
3.3.3. Collagen and Gelatin
Extraction Process
Applications
3.3.4. Fish Oil
Extraction Process
Applications
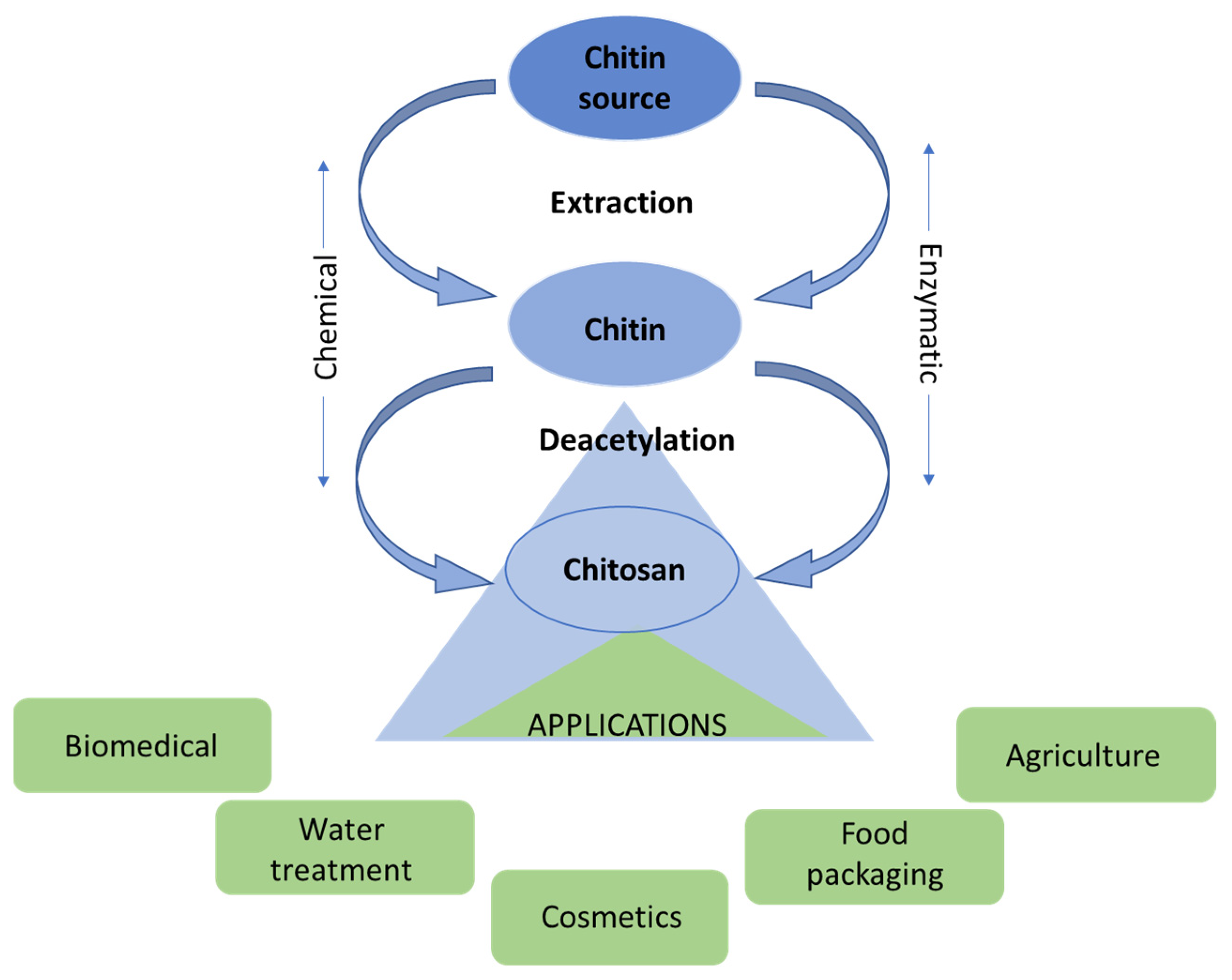
3.3.5. Chitin and Chitosan
Chitin Extraction and Purification
Chitin/Chitosan Applications
3.4. Azorean Fish Waste Potential Valorisation
4. Final Remarks
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Almeida, C.; Karadzic, V.; Vaz, S. The seafood market in Portugal: Driving forces and consequences. Mar. Policy 2015, 61, 87–94. [Google Scholar] [CrossRef]
- European Commission. Blue Economy Report; European Commission: Brussels, Belgium, 2020. [Google Scholar]
- DGRM; INE. Estatísticas da Pesca, 2021; DGRM: Leça da Palmeira, Portugal, 2022. [Google Scholar]
- INE. Boletim Mensal da Agricultura e Pescas; INE: Lisboa, Portugal, 2023. [Google Scholar]
- Wang, S.-P.; Maunder, M.N.; Aires-Da-Silva, A.; Bayliff, W.H. Evaluating fishery impacts: Application to bigeye tuna (Thunnus obesus) in the eastern Pacific Ocean. Fish. Res. 2009, 99, 106–111. [Google Scholar] [CrossRef]
- Aranda, M.; Le Gallic, B.; Ulrich, C.; Borges, L.; Metz, S.; Prellezo, R.; Santurtún, M. EU Fisheries Policy—Latest Developments and Future Challenges; European Parliament: Strasbourg, France, 2019. [Google Scholar]
- FAO. The State of World Fisheries and Aquaculture 2020. Sustainability in Action; FAO: Rome, Italy, 2020. [Google Scholar]
- Kaiser, M.J.; Collie, J.S.; Hall, S.J.; Jennings, S.; Poiner, I.R. Impacts of fishing gear on marine benthic habitats. In Responsible Fisheries in the Marine Ecosystem; CABI: Oxfordshire, UK, 2010; pp. 197–217. [Google Scholar] [CrossRef]
- The Council of the European Union, European Parliament. Regulation (EU) No 1380/2013 of the European Parliament and of the Council of 11 December 2013; European Union: Luxembourg, 2013; Volume 354, pp. 22–61. [Google Scholar] [CrossRef]
- Cámara, A.; Santero-Sánchez, R. Economic, Social, and Environmental Impact of a Sustainable Fisheries Model in Spain. Sustainability 2019, 11, 6311. [Google Scholar] [CrossRef]
- Silva, H.M.; Pinho, M.R. Small-Scale Fishing on Seamounts. In Seamounts: Ecology, Fisheries and Conservation; Pitcher, T.J., Morato, T., Hart, P.J.B., Clark, M.R., Haggan, N., Santos, R.S., Eds.; Blackwell Publishing Ltd.: Oxford, UK, 2007; pp. 330–360. [Google Scholar]
- Ruiz-Salmón, I.; Laso, J.; Margallo, M.; Villanueva-Rey, P.; Rodríguez, E.; Quinteiro, P.; Dias, A.C.; Almeida, C.; Nunes, M.L.; Marques, A.; et al. Life cycle assessment of fish and seafood processed products—A review of methodologies and new challenges. Sci. Total Environ. 2021, 761, 144094. [Google Scholar] [CrossRef] [PubMed]
- Filipa, A. The Economic, Social and Territorial Situation of the Azores (Portugal); European Parliament: Strasbourg, France, 2017. [Google Scholar]
- European Parliament. Directiva do Conselho 75/442/CEE. of. das Comunidades Eur.; European Parliament: Strasbourg, France, 1975. [Google Scholar]
- Directorate-General for Maritime Affairs and Fisheries. Blue Bioeconomy: Situation Report and Perspectives; European Commission: Brussels, Belgium, 2018. [Google Scholar]
- Ahuja, I.; Dauksas, E.; Remme, J.F.; Richardsen, R.; Løes, A.-K. Fish and fish waste-based fertilizers in organic farming—With status in Norway: A review. Waste Manag. 2020, 115, 95–112. [Google Scholar] [CrossRef]
- Guillen, J.; Holmes, S.J.; Carvalho, N.; Casey, J.; Dörner, H.; Gibin, M.; Mannini, A.; Vasilakopoulos, P.; Zanzi, A. A Review of the European Union Landing Obligation Focusing on Its Implications for Fisheries and the Environment. Sustainability 2018, 10, 900. [Google Scholar] [CrossRef]
- Martín-Sánchez, A.; Navarro, C.; Pérez-Álvarez, J.; Kuri, V. Alternatives for Efficient and Sustainable Production of Surimi: A Review. Compr. Rev. Food Sci. Food Saf. 2009, 8, 359–374. [Google Scholar] [CrossRef]
- Jannat-Alipour, H.; Rezaei, M.; Shabanpour, B.; Tabarsa, M. Edible green seaweed, Ulva intestinalis as an ingredient in surimi-based product: Chemical composition and physicochemical properties. J. Appl. Phycol. 2019, 31, 2529–2539. [Google Scholar] [CrossRef]
- Alipour, H.J.; Rezaei, M.; Shabanpour, B.; Tabarsa, M. Effects of sulfated polysaccharides from green alga Ulva intestinalis on physicochemical properties and microstructure of silver carp surimi. Food Hydrocoll. 2018, 74, 87–96. [Google Scholar] [CrossRef]
- FAO. Production and Utilization of Fish Silage; FAO: Rome, Italy, 2018; pp. 1–28. [Google Scholar]
- Islam, J.; Yap, E.E.S.; Krongpong, L.; Toppe, J.; Peñarubia, O.R. Fish Waste Management; FAO: Rome, Italy, 2021. [Google Scholar]
- Shahidi, F.; Varatharajan, V.; Peng, H.; Senadheera, R. Utilization of marine by-products for the recovery of value-added products. J. Food Bioact. 2019, 6. [Google Scholar] [CrossRef]
- Kuley, E.; Özyurt, G.; Özogul, I.; Boga, M.; Akyol, I.; Rocha, J.M.; Özogul, F. The Role of Selected Lactic Acid Bacteria on Organic Acid Accumulation during Wet and Spray-Dried Fish-Based Silages. Contributions to the Winning Combination of Microbial Food Safety and Environmental Sustainability. Microorganisms 2020, 8, 172. [Google Scholar] [CrossRef]
- Le Gouic, A.V.; Harnedy, P.A.; FitzGerald, R.J. Bioactive Peptides from Fish Protein By-Products. In Bioactive Molecules in Food; Springer: Cham, Switzerland, 2018; pp. 1–35. [Google Scholar]
- Ambigaipalan, P.; Shahidi, F. Bioactive peptides from shrimp shell processing discards: Antioxidant and biological activities. J. Funct. Foods 2017, 34, 7–17. [Google Scholar] [CrossRef]
- Maschmeyer, T.; Luque, R.; Selva, M. Upgrading of marine (fish and crustaceans) biowaste for high added-value molecules and bio(nano)-materials. Chem. Soc. Rev. 2020, 49, 4527–4563. [Google Scholar] [CrossRef] [PubMed]
- Xiao, W.; Bal, B.S.; Rahaman, M.N. Preparation of resorbable carbonate-substituted hollow hydroxyapatite microspheres and their evaluation in osseous defects in vivo. Mater. Sci. Eng. C 2016, 60, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Boutinguiza, M.; Pou, J.; Comesaña, R.; Lusquiños, F.; de Carlos, A.; León, B. Biological hydroxyapatite obtained from fish bones. Mater. Sci. Eng. C 2012, 32, 478–486. [Google Scholar] [CrossRef]
- Zamora-Sillero, J.; Gharsallaoui, A.; Prentice, C. Peptides from Fish By-product Protein Hydrolysates and Its Functional Properties: An Overview. Mar. Biotechnol. 2018, 20, 118–130. [Google Scholar] [CrossRef]
- Fernandes, P. Enzymes in Fish and Seafood Processing. Front. Bioeng. Biotechnol. 2016, 4, 59. [Google Scholar] [CrossRef]
- Petrova, I.; Tolstorebrov, I.; Eikevik, T.M. Production of fish protein hydrolysates step by step: Technological aspects, equipment used, major energy costs and methods of their minimizing. Int. Aquat. Res. 2018, 10, 223–241. [Google Scholar] [CrossRef]
- Ehrlich, H.; Wysokowski, M.; Żółtowska-Aksamitowska, S.; Petrenko, I.; Jesionowski, T. Collagens of Poriferan Origin. Mar. Drugs 2018, 16, 79. [Google Scholar] [CrossRef]
- Kwak, H.W.; Kim, J.E.; Lee, K.H. Green fabrication of antibacterial gelatin fiber for biomedical application. React. Funct. Polym. 2019, 136, 86–94. [Google Scholar] [CrossRef]
- Jafari, H.; Lista, A.; Siekapen, M.M.; Ghaffari-Bohlouli, P.; Nie, L.; Alimoradi, H.; Shavandi, A. Fish Collagen: Extraction, Characterization, and Applications for Biomaterials Engineering. Polymers 2020, 12, 2230. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.M.; Dornelles, R.C.P.; Mello, R.O.; Kubota, E.H.; Mazutti, M.A.; Kempka, A.P.; Demiate, I.M. Collagen extraction process. Int. Food Res. J. 2016, 23, 913–922. [Google Scholar]
- Liu, D.; Wei, G.; Li, T.; Hu, J.; Lu, N.; Regenstein, J.M.; Zhou, P. Effects of alkaline pretreatments and acid extraction conditions on the acid-soluble collagen from grass carp (Ctenopharyngodon idella) skin. Food Chem. 2017, 172, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Żelechowska, E.; Sadowska, M.; Turk, M. Isolation and some properties of collagen from the backbone of Baltic cod (Gadus morhua). Food Hydrocoll. 2010, 24, 325–329. [Google Scholar] [CrossRef]
- Bai, C.; Wei, Q.; Ren, X. Selective Extraction of Collagen Peptides with High Purity from Cod Skins by Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2017, 5, 7220–7227. [Google Scholar] [CrossRef]
- Ibáñez, E.; Mendiola, J.A.; Castro-Puyana, M. Supercritical Fluid Extraction. In Encyclopedia of Food and Health; Elsevier: Amsterdam, The Netherlands, 2016; pp. 227–233. [Google Scholar]
- Atay, E.; Fabra, M.J.; Martínez-Sanz, M.; Gomez-Mascaraque, L.G.; Altan, A.; Lopez-Rubio, A. Development and characterization of chitosan/gelatin electrosprayed microparticles as food grade delivery vehicles for anthocyanin extracts. Food Hydrocoll. 2018, 77, 699–710. [Google Scholar] [CrossRef]
- Rahman, M.A. Collagen of Extracellular Matrix from Marine Invertebrates and Its Medical Applications. Mar. Drugs 2019, 17, 118. [Google Scholar] [CrossRef]
- Bernhardt, A.; Paul, B.; Gelinsky, M. Biphasic Scaffolds from Marine Collagens for Regeneration of Osteochondral Defects. Mar. Drugs 2018, 16, 91. [Google Scholar] [CrossRef]
- Araujo, P.; Zhu, H.; Breivik, J.F.; Hjelle, J.I.; Zeng, Y. Determination and Structural Elucidation of Triacylglycerols in Krill Oil by Chromatographic Techniques. Lipids 2014, 49, 163–172. [Google Scholar] [CrossRef]
- Olsen, R.L.; Toppe, J.; Karunasagar, I. Challenges and realistic opportunities in the use of by-products from processing of fish and shellfish. Trends Food Sci. Technol. 2014, 36, 144–151. [Google Scholar] [CrossRef]
- Chakraborty, K.; Joseph, D. Production and Characterization of Refined Oils Obtained from Indian Oil Sardine (Sardinella longiceps). J. Agric. Food Chem. 2015, 63, 998–1009. [Google Scholar] [CrossRef] [PubMed]
- Suseno, S.H.; Fitriana, N.; Jacoeb, A.M.; Saraswati, S. Optimization of Sardine Oil Neutralization Process from Fish Meal Industry By-product. Orient. J. Chem. 2015, 31, 2507–2514. [Google Scholar] [CrossRef]
- Monte, M.L.; Monte, M.L.; Pohndorf, R.S.; Crexi, V.T.; Pinto, L.A.A. Bleaching with blends of bleaching earth and activated carbon reduces color and oxidation products of carp oil. Eur. J. Lipid Sci. Technol. 2015, 117, 829–836. [Google Scholar] [CrossRef]
- de Oliveira, D.A.; Minozzo, M.G.; Licodiedoff, S.; Waszczynskyj, N. Physicochemical and sensory characterization of refined and deodorized tuna (Thunnus albacares) by-product oil obtained by enzymatic hydrolysis. Food Chem. 2016, 207, 187–194. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Novel functional food ingredients from marine sources. Curr. Opin. Food Sci. 2015, 2, 123–129. [Google Scholar] [CrossRef]
- Suleria, H.; Osborne, S.; Masci, P.; Gobe, G. Marine-Based Nutraceuticals: An Innovative Trend in the Food and Supplement Industries. Mar. Drugs 2015, 13, 6336–6351. [Google Scholar] [CrossRef]
- Abraha, B.; Admassu, H.; Mahmud, A.; Tsighe, N.; Shui, X.W.; Fang, Y. Effect of processing methods on nutritional and physico-chemical composition of fish: A review. MOJ Food Process. Technol. 2018, 6, 1. [Google Scholar] [CrossRef]
- Karkal, S.S.; Kudre, T.G. Valorization of fish discards for the sustainable production of renewable fuels. J. Clean. Prod. 2020, 275, 122985. [Google Scholar] [CrossRef]
- Dave, D.; Ramakrishnan, V.V.; Trenholm, S.; Manuel, H.; Murphy, J.P.W. Marine Oils as Potential Feedstock for Biodiesel Production: Physicochemical Characterization. J. Bioprocess. Biotech. 2014, 4, 168. [Google Scholar] [CrossRef]
- Doan, C.T.; Tran, T.N.; Nguyen, V.B.; Vo, T.P.K.; Nguyen, A.D.; Wang, S.-L. Chitin extraction from shrimp waste by liquid fermentation using an alkaline protease-producing strain, Brevibacillus parabrevis. Int. J. Biol. Macromol. 2019, 131, 706–715. [Google Scholar] [CrossRef]
- Sudatta, B.; Sugumar, V.; Varma, R.; Nigariga, P. Extraction, characterization and antimicrobial activity of chitosan from pen shell, Pinna bicolor. Int. J. Biol. Macromol. 2020, 163, 423–430. [Google Scholar] [CrossRef]
- Mami, S.; Yeganeh, F.; Salari, A.-A.; Anissian, A.; Azizi, M.; Hajimollahoseini, M. Oral chitin treatment improved demyelination in murine autoimmune encephalomyelitis model by inhibition of inflammatory responses. Int. Immunopharmacol. 2020, 84, 106536. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.A.; Gould, M. Untapped potentials of hazardous nanoarchitectural biopolymers. J. Hazard. Mater. 2021, 411, 124740. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Dhillon, G.S. Recent trends in biological extraction of chitin from marine shell wastes: A review. Crit. Rev. Biotechnol. 2013, 35, 44–61. [Google Scholar] [CrossRef]
- Santos, V.P.; Marques, N.S.S.; Maia, P.C.S.V.; De Lima, M.A.B.; de Oliveira Franco, L.; De Campos-Takaki, G.M. Seafood Waste as Attractive Source of Chitin and Chitosan Production and Their Applications. Int. J. Mol. Sci. 2020, 21, 4290. [Google Scholar] [CrossRef]
- Berezina, N. Production and application of chitin. Phys. Sci. Rev. 2016, 1, 20160048. [Google Scholar] [CrossRef]
- Lopes, C.; Antelo, L.T.; Franco-Uría, A.; Alonso, A.A.; Pérez-Martín, R. Chitin production from crustacean biomass: Sustainability assessment of chemical and enzymatic processes. J. Clean. Prod. 2018, 172, 4140–4151. [Google Scholar] [CrossRef]
- Yadav, M.; Goswami, P.; Paritosh, K.; Kumar, M.; Pareek, N.; Vivekanand, V. Seafood waste: A source for preparation of commercially employable chitin/chitosan materials. Bioresour. Bioprocess. 2019, 6, 8. [Google Scholar] [CrossRef]
- Lokman, I.H.; Ibitoye, E.B.; Hezmee, M.N.M.; Goh, Y.M.; Zuki, A.B.Z.; Jimoh, A.A. Effects of chitin and chitosan from cricket and shrimp on growth and carcass performance of broiler chickens. Trop. Anim. Health Prod. 2019, 51, 2219–2225. [Google Scholar] [CrossRef] [PubMed]
- Andonegi, M.; Heras, K.L.; Santos-Vizcaíno, E.; Igartua, M.; Hernandez, R.M.; de la Caba, K.; Guerrero, P. Structure-properties relationship of chitosan/collagen films with potential for biomedical applications. Carbohydr. Polym. 2020, 237, 116159. [Google Scholar] [CrossRef]
- Rezakazemi, M.; Shirazian, S. Lignin-chitosan blend for methylene blue removal: Adsorption modeling. J. Mol. Liq. 2018, 274, 778–791. [Google Scholar] [CrossRef]
- Wen, L.; Liang, Y.; Lin, Z.; Xie, D.; Zheng, Z.; Xu, C.; Lin, B. Design of multifunctional food packaging films based on carboxymethyl chitosan/polyvinyl alcohol crosslinked network by using citric acid as crosslinker. Polymer 2021, 230, 124048. [Google Scholar] [CrossRef]
- Cadano, J.R.; Jose, M.; Lubi, A.G.; Maling, J.N.; Moraga, J.S.; Shi, Q.Y.; Vegafria, H.M.; VinceCruz-Abeledo, C.C. A comparative study on the raw chitin and chitosan yields of common bio-waste from Philippine seafood. Environ. Sci. Pollut. Res. 2020, 28, 11954–11961. [Google Scholar] [CrossRef] [PubMed]
- Balitaan, J.N.I.; Yeh, J.-M.; Santiago, K.S. Marine waste to a functional biomaterial: Green facile synthesis of modified-β-chitin from Uroteuthis duvauceli pens (gladius). Int. J. Biol. Macromol. 2020, 154, 1565–1575. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.N.; Lee, P.P.; Chen, W.N. Microbial extraction of chitin from seafood waste using sugars derived from fruit waste-stream. AMB Express 2020, 10, 17. [Google Scholar] [CrossRef] [PubMed]
- EL Knidri, H.; Dahmani, J.; Addaou, A.; Laajeb, A.; Lahsini, A. Rapid and efficient extraction of chitin and chitosan for scale-up production: Effect of process parameters on deacetylation degree and molecular weight. Int. J. Biol. Macromol. 2019, 139, 1092–1102. [Google Scholar] [CrossRef]
- Vázquez, J.A.; Rodríguez-Amado, I.; Sotelo, C.G.; Sanz, N.; Pérez-Martín, R.I.; Valcárcel, J. Production, Characterization, and Bioactivity of Fish Protein Hydrolysates from Aquaculture Turbot (Scophthalmus maximus) Wastes. Biomolecules 2020, 10, 310. [Google Scholar] [CrossRef]
- Taati, M.M.; Shabanpour, B.; Ojagh, M. Investigation on fish oil extraction by enzyme extraction and wet reduction methods and quality analysis. AACL Bioflux 2018, 11, 83–90. [Google Scholar]
- Routray, W.; Dave, D.; Ramakrishnan, V.V.; Murphy, W. Production of High Quality Fish Oil by Enzymatic Protein Hydrolysis from Cultured Atlantic Salmon By-Products: Investigation on Effect of Various Extraction Parameters Using Central Composite Rotatable Design. Waste Biomass Valorization 2017, 9, 2003–2014. [Google Scholar] [CrossRef]
- Ahmed, R.; Haq, M.; Cho, Y.-J.; Chun, B.-S. Quality Evaluation of Oil Recovered from By-products of Bigeye Tuna Using Supercritical Carbon Dioxide Extraction. Turkish J. Fish. Aquat. Sci. 2017, 17, 663–672. [Google Scholar] [CrossRef]
- Ayala-Barajas, D.; Gonzalez-Velez, V.; Velez-Tirado, M.; Aguilar-Pliego, J. Hydroxyapatite extraction from fish scales of Tilapia. In Proceedings of the 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Montreal, QC, Canada, 20–24 July 2020; pp. 2206–2208. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, M.; Ji, S.; Zhang, L.; Cao, W.; Wang, H.; Wang, S. Preparation and application of hydroxyapatite extracted from fish scale waste using deep eutectic solvents. Ceram. Int. 2020, 47, 9366–9372. [Google Scholar] [CrossRef]
- Arumugam, G.K.S.; Sharma, D.; Balakrishnan, R.M.; Ettiyappan, J.B.P. Extraction, optimization and characterization of collagen from sole fish skin. Sustain. Chem. Pharm. 2018, 9, 19–26. [Google Scholar] [CrossRef]
- Menezes, M.D.L.L.R.; Ribeiro, H.L.; Abreu, F.D.O.M.D.S.; Feitosa, J.P.D.A.; Filho, M.D.S.M.D.S. Optimization of the collagen extraction from Nile tilapia skin (Oreochromis niloticus) and its hydrogel with hyaluronic acid. Colloids Surf. B Biointerfaces 2020, 189, 110852. [Google Scholar] [CrossRef] [PubMed]
- Bhuimbar, M.V.; Bhagwat, P.K.; Dandge, P.B. Extraction and characterization of acid soluble collagen from fish waste: Development of collagen-chitosan blend as food packaging film. J. Environ. Chem. Eng. 2019, 7, 102983. [Google Scholar] [CrossRef]
- Sousa, R.O.; Martins, E.; Carvalho, D.N.; Alves, A.L.; Oliveira, C.; Duarte, A.R.C.; Silva, T.H.; Reis, R.L. Collagen from Atlantic cod (Gadus morhua) skins extracted using CO2 acidified water with potential application in healthcare. J. Polym. Res. 2020, 27, 73. [Google Scholar] [CrossRef]

| Main Species | 2017 | 2018 | 2019 | 2020 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Portugal | Azores | Portugal | Azores | Portugal | Azores | Portugal | Azores | |||||||||
| t | 1000€ | t | 1000€ | t | 1000€ | t | 1000€ | t | 1000€ | t | 1000€ | t | 1000€ | t | 1000€ | |
| Marine Fish | 99,834 | 191,800 | 6048 | 26,572 | 107,996 | 191,107 | 11,204 | 32,280 | 119,534 | 201,760 | 6960 | 23,788 | 92,606 | 179,629 | 6890 | 24,266 |
| Fork-beard fish (Phycis phycis) | 309 | 1374 | 103 | 576 | 282 | 1298 | 97 | 579 | 289 | 1294 | 98 | 580 | 325 | 1373 | 129 | 671 |
| Megrim (Lepidorhombus whiffiagonis) | 160 | 451 | ə | ə | 138 | 387 | ə | 2 | 124 | 369 | ə | 1 | 151 | 411 | ə | 2 |
| Bigeye tuna (Thunnus obesus), blue fin tuna (Thunnus thynnus thynnus), bullet tuna (Auxis rochei rochei), skipjack tuna (Katsuwonus pelamis), yellowfin tuna (Thunnus albacares) | 8236 | 21,845 | 2052 | 4299 | 13,229 | 27,481 | 7335 | 12,309 | 9966 | 25,491 | 3390 | 5440 | 6822 | 19,231 | 3507 | 6942 |
| Badejo (Mycteroperca fusca) | 19 | 108 | 2 | 14 | 16 | 91 | 2 | 9 | 18 | 72 | 1 | 8 | 21 | 84 | 1 | 6 |
| Axillary sea-bream (Pagellus acarne) | 596 | 2774 | 37 | 167 | 672 | 2956 | 36 | 108 | 533 | 2838 | 14 | 63 | 486 | 2308 | 22 | 92 |
| Bogue (Boops boops) | 605 | 171 | 64 | 45 | 604 | 144 | 81 | 37 | 368 | 92 | 15 | 8 | 341 | 80 | 13 | 8 |
| Tope (Galeorhinus galeus) | 131 | 416 | 75 | 174 | 101 | 321 | 41 | 81 | 88 | 292 | 27 | 50 | 93 | 310 | 21 | 42 |
| Offshore rockfish (Pontinus kuhlii) | 600 | 3149 | 374 | 2207 | 502 | 2826 | 312 | 1962 | 374 | 2327 | 214 | 1588 | 339 | 1934 | 169 | 1191 |
| Blue jack mackerel (Trachurus picturatus) | 4573 | 3128 | 602 | 1318 | 3738 | 2920 | 848 | 1478 | 3635 | 2677 | 1040 | 1336 | 3472 | 2853 | 854 | 1352 |
| Chub mackerel (Scomber japonicus) | 19,482 | 8282 | 197 | 305 | 33,564 | 10,401 | 202 | 267 | 46,314 | 17,878 | 227 | 304 | 23,666 | 9348 | 299 | 394 |
| Wreck-fish (Polyprion americanus) | 215 | 3674 | 128 | 1975 | 174 | 3223 | 89 | 1477 | 157 | 3134 | 80 | 1423 | 172 | 2927 | 81 | 1201 |
| Conger eel (Conger conger) | 1302 | 3422 | 318 | 748 | 1012 | 2930 | 211 | 566 | 975 | 2854 | 173 | 517 | 1001 | 2666 | 163 | 440 |
| Dory (Zeus faber) | 352 | 4132 | 19 | 232 | 328 | 4052 | 10 | 138 | 384 | 4469 | 4 | 46 | 359 | 4087 | 4 | 53 |
| Blacktail comber (Serranus atricauda) | 80 | 435 | 76 | 408 | 69 | 367 | 62 | 324 | 38 | 253 | 31 | 209 | 29 | 200 | 25 | 176 |
| Red sea-bream (Pagellus bogaraveo) | 568 | 8006 | 499 | 7030 | 504 | 7458 | 446 | 6449 | 510 | 7197 | 473 | 6550 | 534 | 7054 | 491 | 6328 |
| Red bream (Beryx decadactylus) | 169 | 1420 | 149 | 1152 | 179 | 2003 | 157 | 1655 | 148 | 1785 | 138 | 1598 | 156 | 2220 | 139 | 1966 |
| Red porgy (Pagrus pagrus) | 277 | 3764 | 131 | 1510 | 239 | 3012 | 83 | 919 | 179 | 2355 | 39 | 478 | 152 | 1979 | 41 | 467 |
| Scabbardfish (Lepidopus caudatus) | 152 | 796 | 100 | 367 | 98 | 517 | 73 | 283 | 104 | 497 | 65 | 170 | 171 | 812 | 88 | 181 |
| Black scabbard fish (Aphanopus carbo) | 4342 | 14,053 | 63 | 205 | 3940 | 13,972 | 14 | 47 | 4565 | 15,450 | 17 | 51 | 4505 | 14,740 | ə | ə |
| Blue ling (Molva macrophthalma) | 1494 | 4895 | 10 | 44 | 1499 | 4267 | 11 | 47 | 1917 | 4906 | 10 | 51 | 1902 | 5627 | 9 | 33 |
| Skate (Raja (Dipturus) batis), painted ray (Raja (Raja) microocellata), long-nosed skate (Raja (Dipturus) oxyrinchus) | 1213 | 3019 | 69 | 110 | 1167 | 3139 | 60 | 95 | 1175 | 3144 | 41 | 74 | 1289 | 3198 | 60 | 85 |
| Red gurnard (Aspitrigla cuculus) | 346 | 651 | 1 | 1 | 322 | 604 | 1 | 1 | 334 | 609 | ə | ə | 341 | 594 | ə | ə |
| Salema (Sarpa salpa) | 263 | 155 | 6 | 5 | 252 | 136 | 6 | 4 | 171 | 96 | 2 | 2 | 297 | 123 | 1 | 1 |
| Red stripped mullet (Mullus surmuletus) | 171 | 2371 | 10 | 117 | 180 | 2604 | 14 | 151 | 175 | 2759 | 8 | 103 | 152 | 2371 | 6 | 58 |
| Sardine (Sardina pilchardus) | 14,557 | 23,868 | 32 | 73 | 9694 | 21,873 | 25 | 39 | 9700 | 19,039 | 22 | 37 | 14,526 | 22,087 | 22 | 39 |
| White sea bream (Diplodus sargus cadenati) | 921 | 4120 | 70 | 245 | 809 | 3411 | 79 | 238 | 767 | 3446 | 49 | 149 | 684 | 3027 | 43 | 127 |
| Thick-lipped grey mullet (Chelon labrosus) | 280 | 386 | 27 | 68 | 530 | 491 | 29 | 63 | 342 | 469 | 16 | 50 | 289 | 433 | 13 | 38 |
| Monkfish (Lophius piscatorius) | 544 | 3196 | 6 | 11 | 341 | 2265 | 4 | 12 | 308 | 2071 | 3 | 9 | 604 | 3671 | 2 | 5 |
| Atlantic pomfret (Brama brama) | 2 | 6 | ə | 1 | 2 | 5 | ə | 1 | 6 | 18 | ə | 1 | 1 | 4 | ə | 1 |
| Waste | Biomaterial | Protocol (Briefing) | Reference |
|---|---|---|---|
| Mussel Perna viridis; Tropical oyster Crassostrea iredalei; | Chitin | Chemical
| [68] |
| Shrimp, crab, and squid shells | Chitin | Biological
| [55] |
| Pinna bicolor pen shells | Chitin | Chemical
| [56] |
| Cephalopod Uroteuthis duvauceli | Chitin | Chemical
| [69] |
| Shrimp shells | Chitin | Biological
| [70] |
| Shrimp shells | Chitin | Chemical
| [71] |
| Turbot fish By-products | Fish hydrolyzate | Alcalase hydrolysis
| [72] |
| Tuna By-Products | Fish oil | Enzymatic Extraction
| [73] |
| Salmon By-products | Fish oil | Enzymatic Extraction
| [74] |
| Tuna By-Products | Fish oil | Supercritical carbon dioxide extraction
| [75] |
| Tilapia scales | Hydroxyapatite |
| [76] |
| Fish scales | Hydroxyapatite | Eutectic solvents
| [77] |
| Flounder fish skin | Collagen |
| [78] |
| Nile tilapia skin | Collagen |
| [79] |
| Medusa skin | Collagen |
| [80] |
| Atlantic cod skin | Collagen | Supercritical Fluids
| [81] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valério, N.; Soares, M.; Vilarinho, C.; Correia, M.; Carvalho, J. Diving into Fish Valorisation: Review Opportunities and Analyzing Azorean Fish Data. Processes 2023, 11, 1998. https://doi.org/10.3390/pr11071998
Valério N, Soares M, Vilarinho C, Correia M, Carvalho J. Diving into Fish Valorisation: Review Opportunities and Analyzing Azorean Fish Data. Processes. 2023; 11(7):1998. https://doi.org/10.3390/pr11071998
Chicago/Turabian StyleValério, Nádia, Margarida Soares, Cândida Vilarinho, Manuela Correia, and Joana Carvalho. 2023. "Diving into Fish Valorisation: Review Opportunities and Analyzing Azorean Fish Data" Processes 11, no. 7: 1998. https://doi.org/10.3390/pr11071998
APA StyleValério, N., Soares, M., Vilarinho, C., Correia, M., & Carvalho, J. (2023). Diving into Fish Valorisation: Review Opportunities and Analyzing Azorean Fish Data. Processes, 11(7), 1998. https://doi.org/10.3390/pr11071998






