Abstract
This article describes the performance of hemp shives, a co-product of the hemp industry, when used as an adsorbent to recover copper present in aqueous solutions by a batch method. Two materials provided by an industrial partner, namely water-washed hemp shives (SHI-W) and Na2CO3-activated shives (SHI-C), were studied. Two important variables in the water treatment industry, i.e., pH and ionic strength of the solution, were studied to evaluate their impact on the purification performance of the materials. The results obtained clearly indicated that the performance in terms of Cu removal obtained from the SHI-C material was significantly higher than that obtained with SHI-W, mainly due to the structural and chemical modifications after carbonate treatment. For each dose of this adsorbent, a percentage of recovery between 90 and 100% was achieved, independently of the pH value in the range 3–5. In the case of SHI-W, the highest values were between 60 and 75% and were both dependent on the pH and the dose used. SHI-C was also able to selectively remove 70% of copper in the presence of NaCl 1 M. The analysis of the isotherms indicated the presence of a complex adsorption mechanism that cannot be described by only one isotherm model.
1. Introduction
Copper is a chemical substance and a material widely used in industry due to its numerous applications such as electrical and electronic equipment, construction, cooking utensils, energy sector, automobiles, food supplements, animal husbandry, cosmetology, and jewelry. All these applications lead to a constant increase in the global demand for copper, which is why the recycling and recovery of this element as a secondary source of supply is becoming, more than ever, a major issue. In particular, the recovery of copper is very interesting because the metal can be recycled indefinitely and easily reused without any loss of quality or performance, its properties being comparable to those of mined copper [1,2,3,4,5].
One possible source of copper recovery is wastewater from the metallurgical and surface treatment industries. Techniques for the recovery of metals from aqueous solutions such as electrolysis, adsorption, ion exchange, reverse osmosis, or selective extraction by complexation are well known [6,7,8,9,10]. These methods are certainly efficient and rapid but only profitable for high added value metals such as gold, silver, palladium or platinum, or for the treatment of particular effluents, e.g., photographic solutions and printed circuits. Indeed, for more conventional metals such as copper, manganese or nickel, electrolysis, which is very energy consuming, has problems of profitability. Moreover, adsorption on activated carbon and ion-exchange using organic resins are very sensitive to several characteristics of wastewater, e.g., the presence of a very high organic load, highly variable pH, and a high salinity close to that of marine water. In addition, the metal to be recovered is often complexed to the organic load or to other mineral elements such as boron and fluorides and in competition with other metal cations. Membrane techniques are also energy intensive, non-selective, and require skilled personnel. Therefore, there is a need to continue to develop materials capable of complexing and selectively recovering copper from wastewater while taking into account the characteristics of the wastewater to be treated [11,12,13].
In the last decade, many non-conventional materials have been studied for their ability to complex copper [14,15,16,17,18,19,20,21,22,23,24,25]. In spite of the numerous bibliographic data on the subject, there are still no technical options that are cheap, simple to use, and efficient to recover copper from metallurgical waters. Lignocellulosic materials based on hemp, flax, or wood have been proposed to clean up water contaminated by metals or dyes [18,19,20,24]. For about ten years, our group has been developing hemp fiber-based materials in the form of felt as adsorbent systems to treat polycontaminated waters [26,27,28,29]. Another topic of our work is focalized on hemp co-products such as shives produced by the hemp industry during the processing of the plant. These wastes have found applications in soil improvement, animal bedding, and mulching, and more recently in construction and bioplastics, as well as in the production of energy, oils, biofuel, and biodiesel [29]. In the current study, we test the adsorption capacities of shives for metal complexation using characteristics specific to these natural co-products, such as their particular cellulosic structure and the presence and reactivity of numerous chemical functions such as carboxylic groups.
Here, the adsorption properties of hemp shives to recover copper from aqueous solutions was investigated using the batch method. Two materials, namely water-washed hemp shives (SHI-W) and Na2CO3-activated shives (SHI-C), were studied and the effect of initial copper concentration, solution pH, and solution ionic strength were evaluated. The experimental adsorption data were modeled using the following common isotherm models: Langmuir [30], Freundlich [31], Tempkin [32], Redlich [33] Tóth [34], Hill [35], and Dubinin-Radushkevich [36].
2. Materials and Methods
2.1. Hemp Shives and Reagents
Raw shives as a co-product of the hemp industry were provided by Eurochanvre (Arc-les-Gray, Haute-Saône, France). Two materials, namely water-washed hemp shives (SHI-W) and Na2CO3-activated shives (SHI-C), were used. These commercial shives are intended for plant and animal mulching, and insulation. They are constituted of parallelepiped particles varying in length, from 5 up to 25 mm. Other characteristics are given in Table 1. The two materials were prepared as follows: raw shives were washed several times with osmosed water under agitation at room temperature until the wash water became clear; the shives (noted SHI-W) were then separated by filtration and dried under vacuum at 80 °C for one day; the dry SHI-W sample was then activated using sodium carbonate 1 M during 4 h at 40 °C; after cooling, the mixture was filtered, and the recovered shives (SHI-C) were extensively washed with osmosed water (until neutral pH) and dried under vacuum at 80 °C for one day.

Table 1.
Characteristics of the hemp shives samples: SHI-R: raw shives; SHI-W: water-washed hemp shives; SHI-C: Na2CO3-activated shives [37,38].
Copper(II) sulfate was purchased from Sigma-Aldrich (Saint-Quentin Fallavier, France) and used as received. Appropriate weight of this salt was dissolved in water to obtain a stock solution containing 300 mgCu/L (initial pH of the metal solution 4.5 ± 0.1). Solutions with lower copper concentrations were obtained by diluting the stock solution. The copper concentration in all solutions was determined by inductively coupled plasma atomic emission spectroscopy (ICP-AES) following a standard protocol prior to each experiment [37,38].
2.2. In-Batch Copper Adsorption Experiments
The adsorption experiments were determined by the batch technique, which consists of contacting the solution to be treated at a known concentration with a given mass of adsorbent and stirring for a defined time. At the end of the experiment, the material is easily removed by filtration and the treated solution analyzed to determine the adsorption capacity of the material. Indeed, the copper concentration in solutions before and after adsorption was determined by ICP-AES, and these values were used to calculate the amount of metal adsorbed per gram of hemp shives, i.e., q in mg/g. At equilibrium time (te), qe is calculated from the mass balance equation given by Equation (1), where C0 and Ce are the initial and final copper concentrations in liquid phase (mg/L), respectively, V is the volume of metal solution (L), and m the mass of adsorbent used (g) [37,38].
A recent study showed that the optimal batch processing conditions for complexing copper present in 100 mL of aqueous solution at a concentration of 25 mg/L and a pH 5.5 (similar to the concentration and pH of the industrial wastewaters) were as follows [37]: adsorbent dose = 2 g, contact time = 2 h, rotating shaker speed = 250 rpm, temperature = 22 ± 1 °C. These same conditions were used in this study to evaluate the effect of the initial pH of the solution (range 2–5) as well as the presence of two salts, CaCl2 and NaCl (concentration range 0.1–1 mol/L). These salts are widely used in the formulations used by the surface treatment industry, and concentrations of the cations in the order of several dozen grams are found in the wastewater [37,38]. All experiments were replicated (n = 3) under identical conditions.
2.3. Adsorption Models
To describe the experimental adsorption data obtained by varying copper concentrations between 1 and 150 mg/L [37], seven commonly used adsorption isotherm models were tested. The equations in non-linear form and the associated parameters are described in Table 2. To evaluate the validity of each model, the results obtained by the fitting of experimental data with each equation were analyzed using the non-linear correlation coefficients of determination (R2) and the Chi-square test (χ2). The lowest χ2 value indicates that the applied model is more appropriate than others [39,40].

Table 2.
List of seven adsorption isotherm models used in this study [30,31,32,33,34,35,36,41,42,43,44,45,46,47,48,49,50]. The non-linear equation of each model expresses the amount of pollutant adsorbed at equilibrium (qe in mg/g) in function of liquid phase pollutant concentration at equilibrium (Ce in mg/L).
3. Results and Discussion
3.1. Effect of Solution pH on Hemp Shives Performances
One of the most important parameters in treating industrial wastewater is the initial pH of the water, especially in the case of metal pollution. In fact, at low pH, metallic species are found in cationic form and are more soluble and mobile in aqueous environments. Moreover, these species form more or less stable complexes with the organic substances (e.g., complexing agents and surfactants) present in industrial formulations. As the pH increases, other types of complexes begin to form and the metals take the form of hydroxides, sulfides, or anions depending on the type of alkalizing agent used. Finally, at a specific pH value, they also start to precipitate. In addition, pH can also play an important role on the surface charge present on the adsorbent material by changing the protonation state of functional groups such as those found in lignin, polyphenols, pectins, and proteins constituents [51,52].
Figure 1 shows the Cu removal percentages obtained by varying the pH solution in the range 2–5 and using three different doses of adsorbent material. The adsorbent doses of 0.5, 1, and 1.5 g in 100 mL were chosen to highlight any differences in decontamination for the two materials. pH values above 5 were not studied due to the onset of copper precipitation at this pH. These results showed that the SHI-C material was much more efficient than the SHI-W material. Indeed, for each dose of SHI-C, a removal percentage between 90 and 100% is achieved, independently of the pH value in the range 3–5. This pH range is generally that of industrial waste baths in which the copper to be recovered is found [2,12]. Furthermore, 0.5 g of material is sufficient to achieve high purification efficiency, so there is no need to increase the dose in this pH range, which can be an interesting economic argument. In the case of SHI-W, the highest values were between 60 and 75% and mostly dependent on the dose used (Figure 1). These results can be explained by the differences in chemical composition and structure of the two materials, in accordance with the data reported in Table 1. Indeed, the ion-exchange capacity of the SHI-C sample is three times greater than that of SHI-W, suggesting the possible presence of a chemisorption mechanism. The SHI-C material can be considered as a weakly acidic cation exchanger due to the presence of carboxylate groups in the Na+ form. In addition, it should be noted that at the end of each experiment, only for an initial pH of 5, the final pH of the solutions increased by 0.5 unit, suggesting acid-base interactions. Another observation concerned a little increase in sodium concentrations after copper adsorption. These two observations also suggested the presence of chemical interactions between the material and the metal ions.

Figure 1.
Influence of the initial pH of the solution on copper removal for three different doses of (a) washed hemp shives (SHI-W) and (b) Na2CO3 activated hemp shives (SHI-C). Other experimental conditions: solution volume = 100 mL, [Cu2+] = 25 mg/L, contact time = 120 min, stirring speed = 250 rpm, temperature = 22 ± 1 °C, n = 3.
However, this cannot be the only explanation, as the values of ion-exchange capacities are relatively low (Table 1). In addition, the values of point of zero charge are identical for the two materials. At the pH values of the study, the materials should have a positive charge, therefore they should not attract metal cations. This implies the presence of other interactions and, therefore, a different kind of mechanism. Recently, using both microscopic and spectroscopic data, we have demonstrated significant changes in the morphology and structure of the material upon activation by carbonate [37]. These data were in agreement with the hypothesis of a physisorption mechanism through a diffusion process into the polysaccharide network. The mechanism is thus much more complex than a simple physisorption or chemisorption. Another observation indicated the presence of several physical and chemical interactions, which could take place simultaneously. Even at a pH of 3 (in which case the carboxylate functions are protonated), the SHI-C sample was able to adsorb some amount of copper, regardless of the dose used, confirming a possible diffusion mechanism [38]. In addition, when the pH was below 3, the removal significantly decreased due to the high concentration of H+ ions, which decreased the probability of encounter between metal cations and the adsorbent material. Nevertheless, even at a pH of 2 and a dose of 1.5 g, SHI-C is able to retain 45% copper (Figure 1). This phenomenon has been observed in many similar studies conducted using agricultural low-cost materials such as lignocellulosics as adsorbents for metal removal [28,29,37,51,52,53,54].
3.2. Effect of Ionic Strength of Solution on Hemp Shives Performances
The ionic strength of the aqueous solution can impact the performance of an adsorbent to remove metals. It is known that metallurgical effluents and surface treatment effluents have very high ionic conductivities due to the wide use of salts and leaching formulations in industrial processes [2,12,29,37,52]. To evaluate the influence of the ionic strength, experimental tests were carried out by adding different concentrations of NaCl or CaCl2 to the copper solution. The results showed a decrease in adsorption capacity as the salt concentration increased both for washed and activated hemp shives (Figure 2). This can be explained by a decrease in electrostatic interactions as the ionic strength increases. However, the high removal showed the selectivity of the materials to remove copper even in the presence of high salinity, especially in the case of NaCl. For example, SHI-C was able to remove 70% of copper even in the presence of NaCl 1 M. The difference in the impact of the two salts could be explained by considering the characteristics in size and charge of the cations: Ca2+ is a bivalent ion (as well as copper), and its ionic radius is closer to Cu2+ than Na+. Therefore, there is a greater competition towards the active sites of shives [27]. Moreover, the presence of a larger amount of counter ions in the case of CaCl2 salt should be considered. Indeed, chloride can lead to the formation of neutral or negatively-charged complexes with metals that are soluble and difficult to remove [27,52,55].
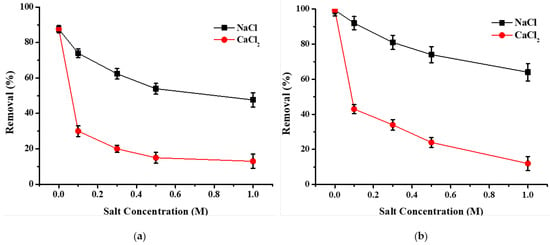
Figure 2.
Effect of the ionic strength of solution on copper adsorption onto (a) washed hemp shives (SHI-W) and (b) Na2CO3 activated hemp shives (SHI-C). Experimental conditions: adsorbent dose = 2 g; solution volume = 100 mL, [Cu2+] = 25 mg/L, [NaCl] = [CaCl2] = 0.1–1.0 M, contact time = 120 min, stirring speed = 250 rpm, pH = 5.2 ± 0.1, temperature = 22 ± 1 °C, n = 3.
3.3. Adsorption Isotherm of Copper Removal Using Hemp Shives
The modelling of results using equilibrium adsorption isotherms is an essential step to designing and optimizing each adsorption process. Different equations can be used to determine the adsorption capacity of a material and to better understand the interactions between the adsorbate and the adsorbent [42,46,56,57,58]. Our experimental data were fitted using various common models having two or three parameters (Table 2). Figure 3 shows the adsorption isotherms plots obtained for copper onto the two materials, and Table 3 summarizes all the constants calculated from the different non-linear equations.
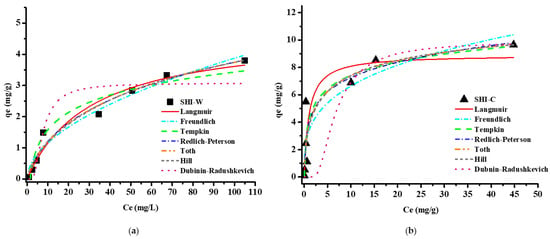
Figure 3.
Adsorption isotherm plots of seven common isotherm models for copper adsorption onto (a) washed hemp shives (SHI-W) and (b) Na2CO3 activated hemp shives (SHI-C).

Table 3.
Isotherm parameter constants for copper adsorption onto washed (SHI-W) and Na2CO3 activated (SHI-C) hemp shives obtained using seven common isotherm models.
The Langmuir isotherm is an empirical model (Equation (2) in Table 2) assuming that adsorption is homogenous with all site having equal energy and affinity towards the adsorbate. This model also implies that the adsorbate molecules are distributed in a monolayer onto the surface of the adsorbent material, and there is no interaction between them [41,42,43,44,45,46]. Two interesting parameters can be obtained from the Langmuir equation: the maximum adsorption capacity (qmax in mg/g) and the separation factor RL (Equation (3) in Table 2). The first one represents the highest metal ion uptake per unit mass of adsorbent: for hemp shives, qmax increased from 6.1 mg/g for washed shives (SHI-W) to 10.2 mg/g for activated shives (SHI-C). This increase is due to the structural and chemical modifications of the adsorbent caused by the alkaline agent Na2CO3, which promotes both ion exchange properties and diffusion in the material, as recently reported [59]. The RL constant gives information about the linear (RL = 1), irreversible (RL = 0), unfavourable (RL > 1), or favourable (0 < RL < 1) adsorption nature of the process [44,56]. For SHI-W, RL values were found in the range 0.0488–0.6060, meanwhile for SHI-C, the range was 0.0008–0.0223. These results suggested that copper adsorption onto hemp shives was a favourable process, especially after carbonate treatment [38,44,56,59].
Another widely used model is the Freundlich equation (Equation (4)), and its applicability suggests the presence of a heterogeneous system with a different energy distribution for active sites and a multilayer adsorption. KF ((mg/g)(L/mg)1/n) and nF are the Freundlich constants, indicating the adsorption capacity and the adsorption intensity of the system, respectively [41,42,43,44,45,46]. When 1/nF ranges between 0 and 1, the adsorption is favourable; when 1/nF is greater than 1, the adsorption process is unfavourable; and when 1/nF is equal 1, the adsorption is irreversible [44,53,57]. The Freundlich parameters shown in Table 3 indicated that SHI-C had higher adsorption capacity than SHI-W. For both samples, the adsorption process was favourable, with 1/nF values of 0.56 and 0.30 for washed and activated hemp shives, respectively.
The Tempkin isotherm model is suitable for a multilayer process, and it takes into account the interaction between the adsorbent and the adsorbate [32,43,44,45,46]. The model described by Equation (5) assumes that adsorption heat decreases linearly as the coverage surface increases, and it permits to calculate the Tempkin constant b (J/mol) related to the heat of adsorption. According to Atkins [60], when the heat of adsorption values are less than 20 kJ/mol, the nature of the adsorption process is physical. The b values found for SHI-C and SHI-W (Table 3) indicate a physisorption mechanism for copper removal onto the two materials, confirming our experimental data. This is in agreement with previous studies on the adsorption kinetics of copper on the same materials and their characterisation by spectroscopic and microscopic methods after adsorption [38,59]. Other researchers working on the adsorption of Zn and Cd by lignocellulosic materials have reported similar conclusions [61,62].
The Redlich–Peterson isotherm is a three versatile parameters equation (Equation (6)) combining Langmuir and Freundlich features and applicable for demonstrating adsorption equilibrium over a wide range of adsorbate concentrations. This isotherm model is also applicable to homogenous and heterogeneous systems but does not follow ideal monolayer adsorption. In this model, particularly interesting is the value of exponent β: for β = 1 the Redlich–Peterson equation reduces to the Langmuir one, for β = 0 it reduces to the one parameter Henry’s model [42,46,47,58]. In our case, β values comprised between 0 and 1 were found for both SHI-C and SHI-W, which means that data are adequately explained by the Redlich–Peterson isotherm [58].
The Tόth isotherm is another empirical modification of the Langmuir equation permitting to reduce the error between experimental data and predicted value of equilibrium data [44]. This equation (Equation (7)) permits to describe heterogeneous adsorption systems, and it is valid for both low and high adsorbate concentration. The values of Tόth maximum adsorption capacity (qmT in mg/g) for SHI-W and SHI-C given in Table 3 were greater if compared to the Langmuir qmax. Moreover, the qmT values for SHI-C were higher than those for SHI-W, similar to those found for the Langmuir model. In addition, the values of Tόth exponent were not equal to unity for both samples, indicating the heterogeneity of the adsorption system [44,46,47].
The Hill model (Equation (8)) assumes that adsorption is a cooperative phenomenon characterized by a cooperative binding constant (nH) and describes the binding of species onto homogeneous systems. Three cases are possible: a positive cooperativity in binding when nH > 1, a non-cooperative or hyperbolic binding when nH = 1, and negative cooperativity in binding when nH < 1 [45,46,58]. In our case (Table 3), the values of nH indicates that adsorption is not a cooperative phenomenon.
The last equation used (Equation (9)) is that proposed in the Dubinin–Radushkevich model. This isotherm, originally designed for adsorption of vapours onto solid surface and assuming a physisorption mechanism, is often used for the estimation of apparent free energy. The Dubinin–Radushkevich model is based on the adsorption potential theory and assumes a Gaussian energy distribution onto a heterogeneous surface and the presence of a multilayer process involving van der Waal’s forces [49]. This model is also applied for the adsorption of pollutants in a solid/liquid system. In this case, the determination of the Dubinin–Radushkevich parameters permits to highlight the nature, physical or chemical, of the adsorption process, thanks to the determination of the apparent free energy (E in kJ/mol obtained using Equation (10)). If the value of E is below 8 kJ/mol, the adsorption is physical, whereas if it ranges between 8 and 16 kJ/mol, the adsorption is chemical [44,50,51,53]. In our case, the E values calculated for SHI-C and SHI-W were 0.25 kJ/mol and 0.28 kJ/mol (Table 3), respectively, suggesting a physical adsorption mechanism.
In order to verify the validity of all the adsorption models and their applicability to experimental data, the values of correlation coefficient (R2) and the chi-square test (χ2) were analysed (Table 4) [39,40,63,64,65]. The order of fitting found was:

Table 4.
Non-linear correlation coefficient R2 and Chi-square χ2 of isotherms models for copper adsorption onto washed (SHI-W) and Na2CO3 activated (SHI-C) hemp shives.
- -
- Redlich-Peterson > Tόth > Hill > Freundlich > Langmuir > Tempkin > Dubinin-Radushkevich for SHI W sample;
- -
- Tempkin > Freundlich > Langmuir > Redlich-Peterson > Toth > Hill > Dubinin-Radushkevich for SHI-C sample.
However, the close similarity in R2 and χ2 values found for each model suggested the presence of a complex adsorption mechanism that cannot be described by only one isotherm. Indeed, even if the applicability of some models suggests the predominance of a physical mechanism, the results of the study on pH and ionic strength also suggest the presence of chemical interactions. In this work, we concluded that the mechanism on this type of adsorbent is complex, involving several physical and chemical interactions simultaneously, although it seems that the physical nature is predominant due to the diffusion of cations in the polymeric network of the material.
4. Conclusions
The results described in this study confirmed that hemp shives, considered as low-cost by-products of the hemp industry, can be used as adsorbents for the recovery of copper ions from solutions. For the hemp sample activated by sodium carbonate, the performance is independent of the pH of the solution between 3 and 5, a pH range characteristic of industrial waste baths in which the copper to be recovered is found. The adsorption results also indicated that 0.5 g of material were sufficient to achieve high purification efficiency, so there is no need to increase the dose in this pH range, which can be an interesting economic argument. Furthermore, even under high salinity conditions, the material shows excellent capacity and selectivity for copper recovery. The material was able to remove 70% of copper even in the presence of NaCl 1 M. The batch technique is also simple and the process proposed in this work could find applications in the treatment of real effluents containing high copper concentrations. The adsorption mechanism is nevertheless complicated, involving several specific interactions such as surface adsorption, interactions with the carboxylate groups, and diffusion into the macromolecular network. These interactions can occur simultaneously and are intimately linked to the conditions of the solution to be treated. The analysis of the isotherms confirmed the presence of a complex adsorption mechanism that cannot be described by only one isotherm model, due to the simultaneous presence of several interactions. The next step will be to carry out a test on a semi-industrial scale on effluents from the surface treatment sector to study the feasibility of its implementation.
Author Contributions
Conceptualization, methodology, resources, funding acquisition, G.C.; formal analysis, investigation, writing-original draft, C.M.; validation, data curation, writing-review and editing, visualization, C.M. and G.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the FINEAU project (FINEAU Program 2021–2024: An European project focused on “Plant-Based Cellulosic Materials for Wastewater Treatment” _ 2020-0058).
Data Availability Statement
Not applicable.
Acknowledgments
The authors thank Eurochanvre Arc-les-Gray (France) for the gift of the two materials, Région Bourgogne Franche-Comté, FEDER, and Université de Franche-Comté (FINEAU and MYDREAU Projects) for financial support, and the PEA2t Plateform (Chrono-environnement, Besançon, Université de Franche-Comté), which manages and maintains the analytical equipment used in this work. Chiara Mongioví thanks the Région Bourgogne Franche-Comté for awarding her a research grant. The authors also acknowledge the scientific and technical expertise of Nadia Morin-Crini, Dario Lacalamita, Vincent Placet and Xavier Gabrion (Besançon, France), Corina Bradu (Budapest, Romania), Aleksandra Ivanovska and Mirjana Kostić (Belgrade, Serbia), and Giangiacomo Torri (Milan, Italy).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chowdhury, S.; Jafar Mazumder, M.A.; Al-Attas, O.; Husain, T. Heavy metals in drinking water: Occurrences, implications, and future needs in developing countries. Sci. Total Environ. 2016, 569–570, 476–488. [Google Scholar] [CrossRef] [PubMed]
- Al-Saydeh, S.A.; El-Naas, M.H.; Zaidi, S.J. Copper removal from industrial wastewater: A comprehensive review. J. Ind. Eng. Chem. 2017, 56, 35–44. [Google Scholar] [CrossRef]
- Rehman, M.; Liu, L.; Wang, Q.; Saleem, M.H.; Bashir, S.; Ullah, S.; Peng, D.X. Copper environmental toxicology, recent advances, and future outlook: A review. Environ. Sci. Pollut. Res. 2019, 26, 18003–18016. [Google Scholar] [CrossRef] [PubMed]
- Botelho Junior, A.B.; Dreisinger, D.B.; Espinosa, D.C. A review of nickel, copper and cobalt recovery by chelating ion exchange resins from mining processes and mining tailings. Min. Met. Explor. 2019, 36, 199–213. [Google Scholar] [CrossRef]
- Kute, A.D.; Gaikwad, R.P.; Warkad, I.R.; Gawande, M.B. A review on the synthesis and applications of sustainable copper-based nanomaterials. Green Chem. 2022, 24, 3502–3573. [Google Scholar] [CrossRef]
- Gupta, V.K.; Nayak, A.; Agarwal, S. Bioadsorbents for remediation of heavy metals: Current status and their future prospects. Environ. Eng. Res. 2015, 20, 1–18. [Google Scholar] [CrossRef]
- Malik, D.S.; Jain, C.K.; Yadav, A.K. Removal of heavy metals from emerging cellulosic low-cost biosorbents: A review. Appl. Water Sci. 2017, 7, 2113–2136. [Google Scholar] [CrossRef]
- Jamshaid, A.; Hamid, A.; Muhammad, N.; Naseer, A.; Ghauri, M.; Iqbal, J.; Rafiq, S.; Shah, N.S. Cellulose-based materials for the removal of heavy metals from wastewater—An overview. ChemBioEng 2017, 4, 240–256. [Google Scholar] [CrossRef]
- Qasem, N.A.A.; Mohammed, R.H.; Lawal, D.U. Removal of heavy metal ions from wastewater: A comprehensive and critical review. NPJ Clean Water 2021, 4, 36. [Google Scholar] [CrossRef]
- Alsafran, M.; Saleem, M.H.; Al Jabri, H.; Rizwan, M.; Usman, K. Principles and applicability of integrated remediation strategies for heavy metal removal/recovery from contaminated environments. J. Plant Growth Regul. 2022, 1–22. [Google Scholar] [CrossRef]
- Barakat, M.A.; Kumar, R. Modified and New Adsorbents for Removal of Heavy Metals from Wastewater. In Heavy Metals in Water: Presence, Removal and Safety; Chapter 10; Sharma, S.K., Ed.; The Royal Society of Chemistry: London, UK, 2015; pp. 193–212. [Google Scholar]
- Crini, G.; Lichtfouse, E.; Wilson, L.D.; Morin-Crini, N. Conventional and non-conventional adsorbents for wastewater treatment. Environ. Chem. Lett. 2019, 17, 195–213. [Google Scholar] [CrossRef]
- Torres, E. Biosorption: A review of the latest advances. Processes 2020, 8, 1584. [Google Scholar] [CrossRef]
- Abdolali, A.; Guo, W.S.; Ngo, H.H.; Chen, S.S.; Nguyen, N.C.; Tung, K.L. Typical lignocellulosic wastes and by-products for biosorption process in water and wastewater treatment: A critical review. Bioresour. Technol. 2014, 160, 57–66. [Google Scholar] [CrossRef]
- Vijayaraghavan, K.; Balasubramanian, R. Is biosorption suitable for decontamination of metal-bearing wastewaters? A critical review on the state-of-the-art of biosorption processes and future directions. J. Environ. Manag. 2015, 160, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Azarova, Y.A.; Pestov, A.V.; Bratskaya, S.Z. Application of chitosan and its derivatives for solid-phase extraction of metal and metalloid ions: A mini-review. Cellulose 2016, 23, 2273–2289. [Google Scholar] [CrossRef]
- Emenike, P.C.; Omole, D.O.; Ngene, B.U.; Tenebe, I.T. Potentiality of agricultural adsorbent for the sequestering of metal ions from wastewater. Glob. J. Environ. Sci. Manag. 2016, 2, 411–442. [Google Scholar]
- Kumar, R.; Sharma, R.K.; Singh, A.P. Cellulose based grafted biosorbents—Journey from lignocellulose biomass to toxic metal ions sorption applications—A review. J. Mol. Liq. 2017, 232, 62–93. [Google Scholar] [CrossRef]
- Afroze, S.; Sen, T.K. A review on heavy metal ions and dye adsorption from water by agricultural solid waste adsorbents. Water Air Soil Pollut. 2018, 229, 225. [Google Scholar] [CrossRef]
- Rozumová, L.; Legátová, B. Biosorbent from industrial hemp hurds for copper ions removal. Int. J. Mater. Sci. Eng. 2018, 6, 114–125. [Google Scholar] [CrossRef]
- Bashir, M.; Tyagi, S.; Annachhatre, A.P. Adsorption copper from aqueous solution onto agricultural adsorbent: Kinetics and isotherm studies. Mater. Today Proc. 2020, 28, 1833–1840. [Google Scholar] [CrossRef]
- Begum, B.; Yuhana, N.Y.; Saleh, N.M.; Kamarudin, N.H.N.; Sulong, A.B. Review of chitosan composite as a heavy metal adsorbent: Material preparation and properties. Carbohydr. Polym. 2021, 259, 117613. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Liu, R.; Yang, J.; Shuai, Q.; Yuliarto, B.; Kaneti, Y.V.; Yamauchi, Y. Nanoarchitectured porous organic polymers and their environmental applications for removal of toxic metal ions. Chem. Eng. J. 2021, 408, 127991. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, V. A comprehensive review on application of lignocellulose derived nanomaterial in heavy metals removal from wastewater. Chem. Afr. 2022, 1–40. [Google Scholar] [CrossRef]
- Chakraborty, R.; Asthana, A.; Singh, A.K.; Jain, B.; Susan, A.B.H. Adsorption of heavy metal ions by various low-cost adsorbents: A review. Int. J. Environ. Anal. Chem. 2022, 102, 342–379. [Google Scholar] [CrossRef]
- Bugnet, J.; Morin-Crini, N.; Cosentino, C.; Chanet, G.; Winterton, P.; Crini, G. Hemp decontamination of poly-metallic aqueous solutions. Environ. Eng. Manag. J. 2017, 16, 535–542. [Google Scholar]
- Loiacono, S.; Crini, G.; Chanet, G.; Raschetti, M.; Placet, V.; Morin-Crini, N. Metals in aqueous solutions and real effluents: Biosorption behavior onto a hemp-based felt. J. Chem. Technol. Biotechnol. 2018, 93, 2592–2601. [Google Scholar] [CrossRef]
- Crini, G.; Bradu, C.; Cosentino, C.; Staelens, J.N.; Martel, B.; Fourmentin, M.; Loiacono, S.; Chanet, G.; Torri, G.; Morin-Crini, N. Simultaneous removal of inorganic and organic pollutants from polycontaminated wastewaters on modified hemp-based felts. Rev. Chim. 2021, 72, 25–43. [Google Scholar] [CrossRef]
- Mongioví, C.; Morin-Crini, N.; Lacalamita, D.; Bradu, C.; Raschetti, M.; Placet, V.; Ribeiro, A.R.L.; Ivanovska, A.; Kostić, M.; Crini, G. Biosorbents from plant fibers of hemp and flax for metal removal: Comparison of their biosorption properties. Molecules 2021, 26, 4199. [Google Scholar] [CrossRef]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. J. Am. Chem. Soc. 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Über die adsorption in lösungen. Z. Phys. Chem. 1906, 57, 385–471. [Google Scholar] [CrossRef]
- Tempkin, M.J.; Pyzhev, V. Kinetics of ammonia synthesis on promoted iron catalysts. Acta Phys. URSS 1940, 12, 217–256. [Google Scholar]
- Redlich, O.; Peterson, D.L. A useful adsorption isotherm. J. Phys. Chem. 1959, 63, 1024–1026. [Google Scholar] [CrossRef]
- Tόth, J. State equations of the solid gas interface layer. Acta Chem. Acad. Sci. Hung. 1971, 69, 311–317. [Google Scholar]
- Hill, A.V. The possible effects of the aggregation of the molecules of hæmoglobin on its dissociation curves. J. Physiol. 1910, 40, 4–7. [Google Scholar]
- Dubinin, M.M.; Radushkevich, L.V. The equation of the characteristic curve of activated charcoal. Proc. Acad. Sci. Phys. Chem. Sect. 1947, 55, 331–333. [Google Scholar]
- Mongioví, C.; Lacalamita, D.; Morin-Crini, N.; Gabrion, X.; Placet, V.; Ribeiro, A.R.L.; Ivanovska, A.; Kostić, M.; Bradu, C.; Staelens, J.N.; et al. Use of chènevotte, a valuable co-product of industrial hemp fiber, as adsorbent for copper ions: Kinetic studies and modelling. Arab. J. Chem. 2022, 15, 103742. [Google Scholar] [CrossRef]
- Mongioví, C.; Crini, G.; Gabrion, X.; Placet, V.; Blondeau-Patissier, V.; Krystianiak, A.; Durand, S.; Beaugrand, J.; Dorlando, A.; Rivard, C.; et al. Revealing the adsorption mechanism of copper on hemp-based materials through EDX, nano-CT, XPS, FTIR, Raman, and XANES characterization techniques. Chem. Eng. J. Adv. 2022, 10, 100282. [Google Scholar] [CrossRef]
- Hadi, M.; McKay, G.; Samarghandi, M.R.; Maleki, A.; Aminabad, M.S. Prediction of optimimum adsorption isotherm: Comparison of chi-square and Log-likelihood statistics. Desalin. Water Treat. 2012, 49, 81–94. [Google Scholar] [CrossRef]
- Kaushal, A.; Singh, S.K. Critical analysis of adsorption data statistically. Appl. Water Sci. 2017, 7, 3191–3196. [Google Scholar] [CrossRef]
- González-López, M.E.; Laureano-Anzaldo, C.M.; Pérez-Fonseca, A.A.; Arellano, M.; Robledo-Ortíz, J.R. A critical overview of adsorption models linearization: Methodological and statistical inconsistencies. Sep. Purif. Rev. 2021, 51, 358–372. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.J.; Hosseini-Bandegharaei, A.; Chao, H.P. Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: A critical review. Water Res. 2017, 120, 88–116. [Google Scholar] [CrossRef] [PubMed]
- Ayawei, N.; Ebelegi, A.N.; Wankasi, D. Modelling and interpretation of adsorption isotherms. J. Chem. 2017, 2017, 3039817. [Google Scholar] [CrossRef]
- Al-Ghouti, M.A.; Da’ana, D.A. Guidelines for the use and interpretation of adsorption isotherms models: A review. J. Hazard. Mater. 2020, 393, 122383. [Google Scholar] [CrossRef] [PubMed]
- Saadi, R.; Saadi, Z.; Fazaeli, R.; Fard, N.E. Monolayer and multilayer adsorption isotherm models for sorption from aqueous media. Korean J. Chem. Eng. 2015, 32, 787–799. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Hamdaoui, O.; Naffrechoux, E. Modeling of adsorption isotherms of phenol and chlorophenols onto granular activated carbon: Part II. Models with more than two parameters. J. Hazard. Mater. 2007, 147, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Dubinin, M.M. Physical Adsorption of Gases and Vapors in Micropores. In Progress in Surface and Membrane Science; Cadenhead, D.A., Danielli, J.F., Rosenberg, M.D., Eds.; Academic Press: New York, NY, USA, 1975; Volume 9, pp. 1–70. [Google Scholar]
- Dubinin, M.M. The potential theory of adsorption of gases and vapors for adsorbents with energetically non-uniform surfaces. Chem. Rev. 1960, 60, 235–241. [Google Scholar] [CrossRef]
- Hu, Q.; Zhang, Z. Application of Dubinin-Radushkevich isotherm model at solid/solution interface: A theoretical analysis. J. Mol. Liq. 2019, 277, 646–648. [Google Scholar] [CrossRef]
- Batool, F.; Akbar, J.; Iqbal, S.; Noreen, S.; Bukhari, S.N.A. Study of isothermal, kinetic, and thermodynamic parameters for adsorption of cadmium: An overview of linear and nonlinear approach and error analysis. Bioinorg. Chem. Appl. 2018, 2018, 3463724. [Google Scholar] [CrossRef]
- Joseph, L.; Jun, B.M.; Flora, J.R.V.; Park, C.M.; Yoon, Y. Removal of heavy metals from water sources in the developing world using low-cost materials: A review. Chemosphere 2019, 229, 142–159. [Google Scholar] [CrossRef]
- Rahman, M.S.; Sathasivam, K.V. Heavy metal adsorption onto Kappaphycus sp. from aqueous solutions: The use of error functions for validation of isotherm and kinetics models. Biomed. Res. Int. 2015, 2015, 126298. [Google Scholar] [CrossRef] [PubMed]
- Das, J.; Saha, R.; Nath, H.; Mondal, A.; Nag, S. An eco-friendly removal of Cd(II) utilizing banana pseudo-fibre and Moringa bark as indigenous green adsorbent and modelling of adsorption by artificial neural network. Environ. Sci. Pollut. Res. 2022, 29, 86528–86549. [Google Scholar] [CrossRef] [PubMed]
- Villaescusa, I.; Fiol, N.; Martínez, M.; Miralles, N.; Poch, J.; Serarols, J. Removal of copper and nickel ions from aqueous solutions by grape stalks wastes. Water Res. 2004, 38, 992–1002. [Google Scholar] [CrossRef] [PubMed]
- Khee, Y.L.; Kiew, P.L.; Chung, Y.T. Valorizing papaya seed waste for wastewater treatment: A review. Int. J. Environ. Sci. Technol. 2022, 1–20. [Google Scholar] [CrossRef]
- Zeng, H.Q.; Hao, H.Y.; Wang, X.; Shao, Z. Chitosan-based composite film adsorbents reinforced with nanocellulose for removal of Cu (II) ion from wastewater: Preparation, characterization, and adsorption mechanism. Int. J. Biol. Macromol. 2022, 213, 369–380. [Google Scholar] [CrossRef]
- Rangabhashiyam, S.; Anu, N.; Giri Nandagopal, M.S.; Selvaraju, N. Relevance of isotherm models in biosorption of pollutants by agricultural byproducts. J. Environ. Chem. Eng. 2014, 2, 398–414. [Google Scholar] [CrossRef]
- Mongiovì, C.; Lacalamita, D.; Morin-Crini, N.; Gabrion, X.; Ivanovska, A.; Sala, F.; Placet, V.; Rizzi, V.; Gubitosa, J.; Mesto, E.; et al. Use of chènevotte, a valuable coproduct of industrial hemp fiber, as adsorbent for pollutant removal. Part 1: Chemical microscopic, spectroscopic and thermogravimetric characterization of raw and modified samples. Molecules 2021, 26, 4574. [Google Scholar] [CrossRef]
- Atkins, P. Physical Chemistry, 6th ed.; Oxford University Press: London, UK, 1999; pp. 857–864. [Google Scholar]
- Nagy, B.; Mânzatu, C.; Török, A.; Indolean, C.; Măicăneanu, A.; Tonk, S.; Majdik, C. Isotherm and thermodynamic studies of Cd(II) removal process using chemically modified lignocellulosic adsorbent. Rev. Roum. Chim. 2015, 60, 257–264. [Google Scholar]
- Dada, A.O.; Olalekan, A.P.; Olatunya, A.M.; Dada, O. Langmuir, Freundlich, Temkin and Dubinin-Radushkevich isotherms studies of equilibrium sorption of Zn2+ onto phosphoric acid modified rice husk. J. Appl. Chem. 2012, 3, 38–45. [Google Scholar]
- Ratkowski, D.A. Handbook of Nonlinear Regression Models; Marcel Dekker: New York, NY, USA, 1990. [Google Scholar]
- Myers, R.H. Classical and Modern Regression with Applications; PWS-KENT: Boston, MA, USA, 1990; Volume 444–445, pp. 297–298. [Google Scholar]
- Crini, G.; Peindy, H.N.; Gimbert, F.; Robert, C. Removal of C.I. Basic Green 4 (Malachite Green) from aqueous solutions by adsorption using cyclodextrin-based adsorbent: Kinetic and equilibrium studies. Sep. Purif. Technol. 2007, 53, 97–110. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).