Need for a Next Generation of Chromatography Models—Academic Demands for Thermodynamic Consistency and Industrial Requirements in Everyday Project Work
Abstract
:1. Introduction
2. Materials and Methods
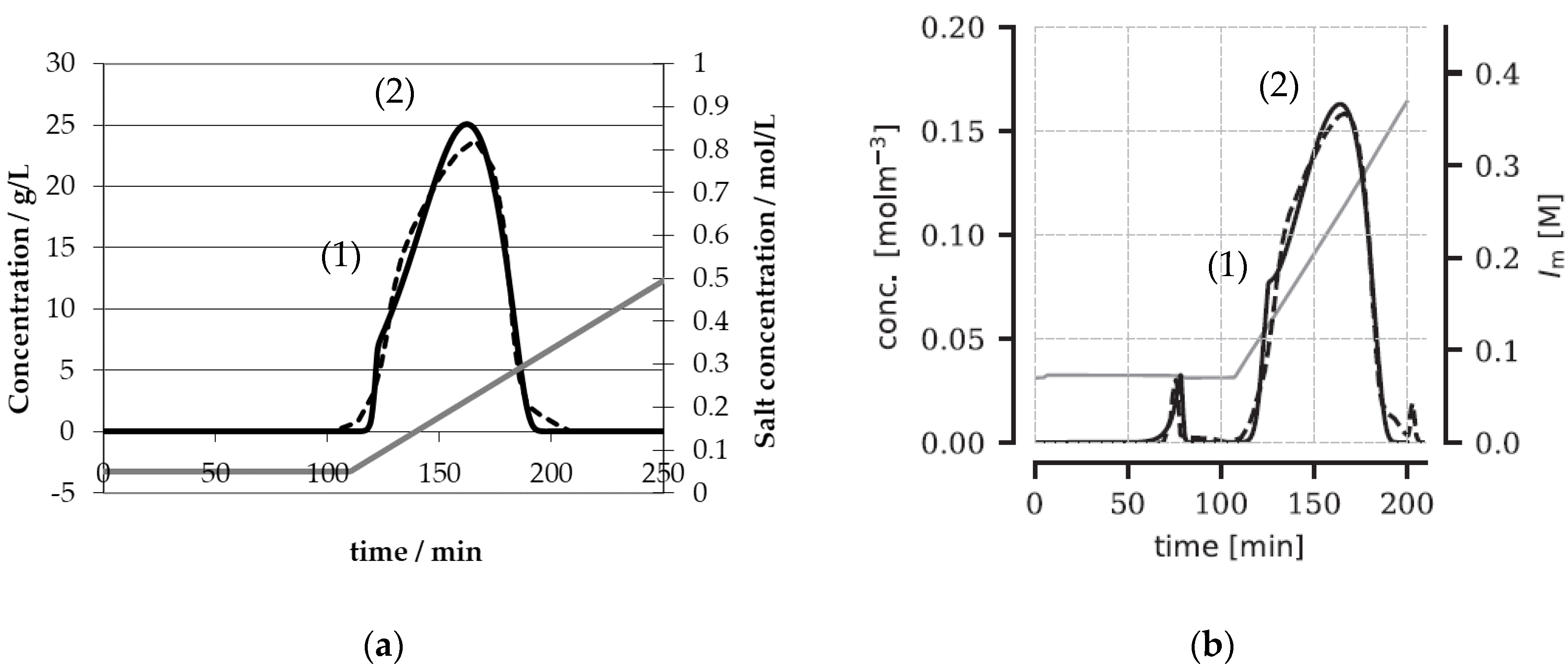
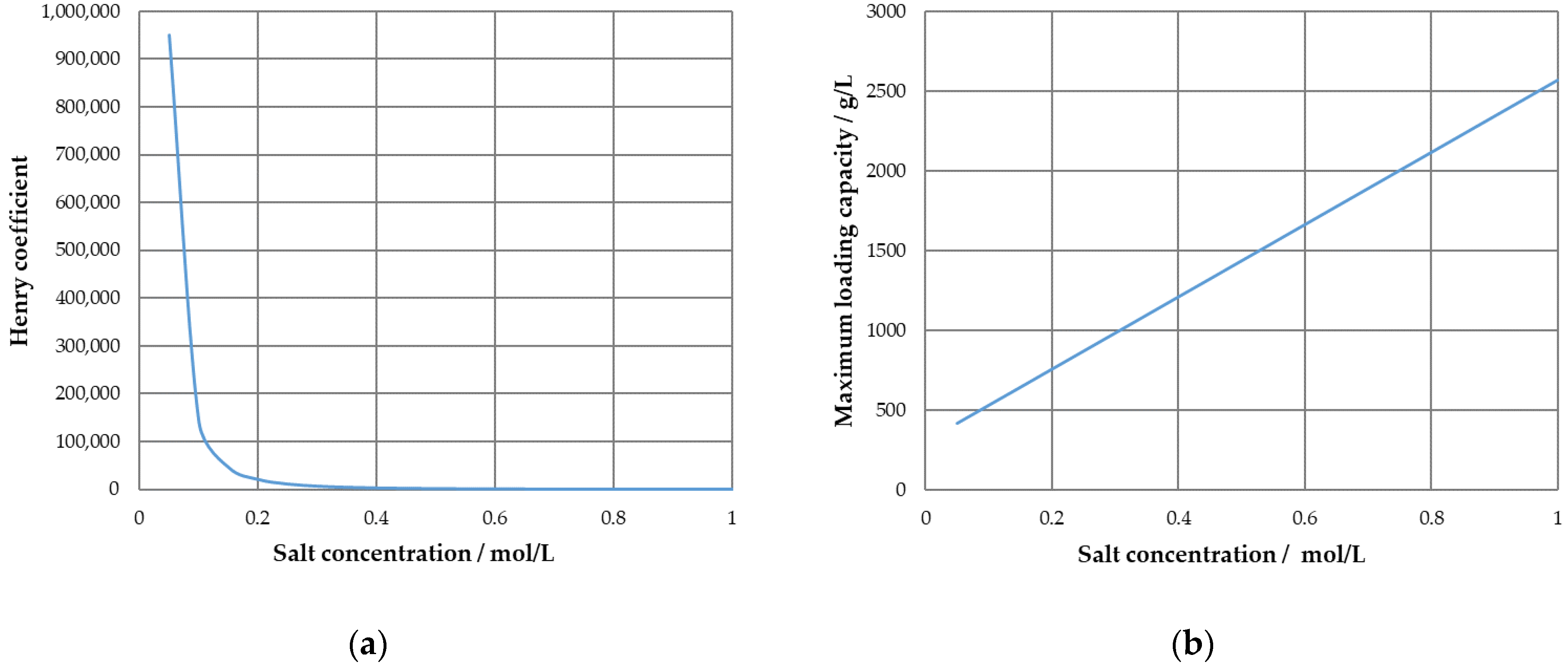
3. Results
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Strube, J.; Jupke, A.; Epping, A.; Schmidt-Traub, H.; Schulte, M.; Devant, R. Design, optimization, and operation of SMB chromatography in the production of enantiomerically pure pharmaceuticals. Chirality 1999, 11, 440–450. [Google Scholar] [CrossRef]
- Strube, J. Technische Chromatographie: Auslegung, Optimierung, Betrieb und Wirtschaftlichkeit; Zugl.: Dortmund, Univ., Habil.-Schr., 1999, Als Ms. gedr; Shaker: Aachen, Germany, 2000; ISBN 978-3-8265-6897-8. [Google Scholar]
- Seidel-Morgenstern, A. Mathematische Modellierung der Präparativen Flüssigchromatographie; Zugl.: Berlin, Techn. Univ., Habil.-Schr., 1994; DUV Dt. Univ.: Wiesbaden, Germany, 1995; ISBN 9783824420643. [Google Scholar]
- Dünnebier, G.; Engell, S.; Epping, A.; Hanisch, F.; Jupke, A.; Klatt, K.-U.; Schmidt-Traub, H. Model-based control of batch chromatography. AIChE J. 2001, 47, 2493–2502. [Google Scholar] [CrossRef]
- Engell, S.; Toumi, A. Optimisation and control of chromatography. Comput. Chem. Eng. 2005, 29, 1243–1252. [Google Scholar] [CrossRef]
- Klatt, K.-U.; Hanisch, F.; Dünnebier, G.; Engell, S. Model-based optimization and control of chromatographic processes. Comput. Chem. Eng. 2000, 24, 1119–1126. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Klatt, K.-U.; Dünnebier, G.; Engell, S.; Hanisch, F. Neural network-based identification of SMB chromatographic processes. Control. Eng. Pract. 2003, 11, 949–959. [Google Scholar] [CrossRef]
- Erdem, G.; Abel, S.; Morari, M.; Mazzotti, M.; Morbidelli, M.; Lee, J.H. Automatic Control of Simulated Moving Beds. Ind. Eng. Chem. Res. 2004, 43, 405–421. [Google Scholar] [CrossRef]
- Strube, J.; Zobel-Roos, S.; Ditz, R. Process-Scale Chromatography. Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH GmbH & Co. KGaA: Weinheim, Germany, 2000; pp. 1–47. ISBN 9783527306732. [Google Scholar]
- Guiochon, G.; Felinger, A.; Shirazi, D.G.; Katti, A.M. Fundamentals of Preparative and Nonlinear Chromatography, 2nd ed.; Elsevier Academic Press: Amsterdam, The Netherlands, 2006; ISBN 9780123705372. [Google Scholar]
- Brooks, C.A.; Cramer, S.M. Steric mass-action ion exchange: Displacement profiles and induced salt gradients. AIChE J. 1992, 38, 1969–1978. [Google Scholar] [CrossRef]
- Rajendran, A.; Maruyama, R.T.; Landa, H.O.R.; Seidel-Morgenstern, A. Modelling binary non-linear chromatography using discrete equilibrium data. Adsorption 2020, 26, 973–987. [Google Scholar] [CrossRef]
- Ortner, F.; Ruppli, C.; Mazzotti, M. Description of Adsorption in Liquid Chromatography under Nonideal Conditions. Langmuir 2018, 34, 5655–5671. [Google Scholar] [CrossRef]
- Ilić, M.; Flockerzi, D.; Seidel-Morgenstern, A. A thermodynamically consistent explicit competitive adsorption isotherm model based on second-order single component behaviour. J. Chromatogr. A 2010, 1217, 2132–2137. [Google Scholar] [CrossRef]
- Guélat, B.; Ströhlein, G.; Lattuada, M.; Morbidelli, M. Electrostatic model for protein adsorption in ion-exchange chromatography and application to monoclonal antibodies, lysozyme and chymotrypsinogen A. J. Chromatogr. A 2010, 1217, 5610–5621. [Google Scholar] [CrossRef] [PubMed]
- Streb, A.; Mazzotti, M. Adsorption for efficient low carbon hydrogen production: Part 1—Adsorption equilibrium and breakthrough studies for H2/CO2/CH4 on zeolite 13X. Adsorption 2021, 27, 541–558. [Google Scholar] [CrossRef]
- Subramanian, G. (Ed.) Process Control, Intensification, and Digitalisation in Continuous Biomanufacturing; Wiley-VCH: Weinheim, Germany, 2022; ISBN 3-527-82734-X. [Google Scholar]
- Bendersky, M.; Davis, J.M. DLVO interaction of colloidal particles with topographically and chemically heterogeneous surfaces. J. Colloid Interface Sci. 2011, 353, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Galushko, S.V.; Shishkina, I.; Urtans, E.; Rotkaja, O. ChromSword®: Software for Method Development in Liquid Chromatography. In Software-Assisted Method Development in High Performance Liquid Chromatography; Fekete, S., Molnár, I., Eds.; World Scientific (Europe): London, UK, 2018; pp. 53–94. ISBN 978-1-78634-545-5. [Google Scholar]
- Mazza, C.B.; Sukumar, N.; Breneman, C.M.; Cramer, S.M. Prediction of protein retention in ion-exchange systems using molecular descriptors obtained from crystal structure. Anal. Chem. 2001, 73, 5457–5461. [Google Scholar] [CrossRef]
- Salvalaglio, M.; Paloni, M.; Guelat, B.; Morbidelli, M.; Cavallotti, C. A two level hierarchical model of protein retention in ion exchange chromatography. J. Chromatogr. A 2015, 1411, 50–62. [Google Scholar] [CrossRef]
- Grimes, B.A.; Skudas, R.; Unger, K.K.; Lubda, D. Pore structural characterization of monolithic silica columns by inverse size-exclusion chromatography. J. Chromatogr. A 2007, 1144, 14–29. [Google Scholar] [CrossRef]
- Liapis, A.I.; Grimes, B.A. Modeling the velocity field of the electroosmotic flow in charged capillaries and in capillary columns packed with charged particles: Interstitial and intraparticle velocities in capillary electrochromatography systems. J. Chromatogr. A 2000, 877, 181–215. [Google Scholar] [CrossRef]
- Skudas, R.; Grimes, B.A.; Thommes, M.; Unger, K.K. Flow-through pore characteristics of monolithic silicas and their impact on column performance in high-performance liquid chromatography. J. Chromatogr. A 2009, 1216, 2625–2636. [Google Scholar] [CrossRef]
- Leinweber, F.C.; Lubda, D.; Cabrera, K.; Tallarek, U. Characterization of silica-based monoliths with bimodal pore size distribution. Anal. Chem. 2002, 74, 2470–2477. [Google Scholar] [CrossRef]
- Ndocko Ndocko, E.; Ditz, R.; Josch, J.-P.; Strube, J. New Material Design Strategy for Chromatographic Separation Steps in Bio-Recovery and Downstream Processing. Chem. Ing. Tech. 2011, 83, 113–129. [Google Scholar] [CrossRef]
- Reich, S.-J.; Svidrytski, A.; Höltzel, A.; Wang, W.; Kübel, C.; Hlushkou, D.; Tallarek, U. Transport under confinement: Hindrance factors for diffusion in core-shell and fully porous particles with different mesopore space morphologies. Microporous Mesoporous Mater. 2019, 282, 188–196. [Google Scholar] [CrossRef]
- Walther, C.; Voigtmann, M.; Bruna, E.; Abusnina, A.; Tscheließnig, A.-L.; Allmer, M.; Schuchnigg, H.; Brocard, C.; Föttinger-Vacha, A.; Klima, G. Smart process development: Application of machine-learning and integrated process modeling for inclusion body purification processes. Biotechnol. Prog. 2022, e3249. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Seth, S.; Scharl, T.; Schroeder, T.; Jungbauer, A.; Dimartino, S. Prediction of the performance of pre-packed purification columns through machine learning. J. Sep. Sci. 2022, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hubbuch, J. Editorial: High-throughput process development. Biotechnol. J. 2012, 7, 1185. [Google Scholar] [CrossRef]
- Mouellef, M.; Vetter, F.L.; Zobel-Roos, S.; Strube, J. Fast and Versatile Chromatography Process Design and Operation Optimization with the Aid of Artificial Intelligence. Processes 2021, 9, 2121. [Google Scholar] [CrossRef]
- Mouellef, M.; Szabo, G.; Vetter, F.L.; Siemers, C.; Strube, J. Artificial Neural Network for Fast and Versatile Model Param-eter Adjustment utilizing PAT signals of Chromatography Processes for Process Control under Production Conditions. Processes 2022, 10, 709. [Google Scholar] [CrossRef]
- Briskot, T.; Hahn, T.; Huuk, T.; Hubbuch, J. Protein adsorption on ion exchange adsorbers: A comparison of a stoichiometric and non-stoichiometric modeling approach. J. Chromatogr. A 2021, 1653, 462397. [Google Scholar] [CrossRef]
- Briskot, T.; Hahn, T.; Huuk, T.; Wang, G.; Kluters, S.; Studts, J.; Wittkopp, F.; Winderl, J.; Schwan, P.; Hagemann, I.; et al. Analysis of complex protein elution behavior in preparative ion exchange processes using a colloidal particle adsorption model. J. Chromatogr. A 2021, 1654, 462439. [Google Scholar] [CrossRef]
- Zobel-Roos, S. Entwicklung, Modellierung und Validierung von Integrierten Kontinuierlichen Gegenstrom-Chromatographie-Prozessen. Ph.D. Thesis, Universitätsbibliothek der TU Clausthal, Clausthal-Zellerfeld, Germany, 2018. [Google Scholar]
- Zobel-Roos, S.; Mouellef, M.; Ditz, R.; Strube, J. Distinct and Quantitative Validation Method for Predictive Process Modelling in Preparative Chromatography of Synthetic and Bio-Based Feed Mixtures Following a Quality-by-Design (QbD) Approach. Processes 2019, 7, 580. [Google Scholar] [CrossRef] [Green Version]
- Carta, G.; Jungbauer, A. Protein Chromatography: Process Development and Scale-Up; Wiley-VCH: Weinheim, Germany, 2010; ISBN 978-3-527-31819-3. [Google Scholar]
- Seidel-Morgenstern, A.; Guiochon, G. Modelling of the competitive isotherms and the chromatographic separation of two enantiomers. Chem. Eng. Sci. 1993, 48, 2787–2797. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef] [Green Version]
- Seidel-Morgenstern, A. Experimental determination of single solute and competitive adsorption isotherms. J. Chromatogr. A 2004, 1037, 255–272. [Google Scholar] [CrossRef] [PubMed]
- Mollerup, J.M. A Review of the Thermodynamics of Protein Association to Ligands, Protein Adsorption, and Adsorption Isotherms. Chem. Eng. Technol. 2008, 31, 864–874. [Google Scholar] [CrossRef]
- Carta, G.; Rodrigues, A.E. Diffusion and convection in chromatographic processes using permeable supports with a bidisperse pore structure. Chem. Eng. Sci. 1993, 48, 3927–3935. [Google Scholar] [CrossRef]
- Wilson, E.J.; Geankoplis, C.J. Liquid Mass Transfer at Very Low Reynolds Numbers in Packed Beds. Ind. Eng. Chem. Fund. 1966, 5, 9–14. [Google Scholar] [CrossRef]
- Sixt, M.; Uhlenbrock, L.; Strube, J. Toward a Distinct and Quantitative Validation Method for Predictive Process Modelling—On the Example of Solid-Liquid Extraction Processes of Complex Plant Extracts. Processes 2018, 6, 66. [Google Scholar] [CrossRef] [Green Version]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vetter, F.L.; Strube, J. Need for a Next Generation of Chromatography Models—Academic Demands for Thermodynamic Consistency and Industrial Requirements in Everyday Project Work. Processes 2022, 10, 715. https://doi.org/10.3390/pr10040715
Vetter FL, Strube J. Need for a Next Generation of Chromatography Models—Academic Demands for Thermodynamic Consistency and Industrial Requirements in Everyday Project Work. Processes. 2022; 10(4):715. https://doi.org/10.3390/pr10040715
Chicago/Turabian StyleVetter, Florian Lukas, and Jochen Strube. 2022. "Need for a Next Generation of Chromatography Models—Academic Demands for Thermodynamic Consistency and Industrial Requirements in Everyday Project Work" Processes 10, no. 4: 715. https://doi.org/10.3390/pr10040715
APA StyleVetter, F. L., & Strube, J. (2022). Need for a Next Generation of Chromatography Models—Academic Demands for Thermodynamic Consistency and Industrial Requirements in Everyday Project Work. Processes, 10(4), 715. https://doi.org/10.3390/pr10040715





