Minimally Invasive Approaches for Traumatic Rupture of the Pancreas in Children—A Case Series
Abstract
:1. Introduction
2. Material and Methods
3. Case Series and Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Iqbal, C.W.; Peter, S.D.S.; Tsao, K.; Cullinane, D.C.; Gourlay, D.M.; Ponsky, T.A.; Wulkan, M.L.; Adibe, O.O. Pancreatic Trauma in Children Study, G. Operative vs Nonoperative Management for Blunt Pancreatic Transection in Children: Multi-Institutional Outcomes. J. Am. Coll. Surg. 2014, 218, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Canty, T.G.; Weinman, D. Management of Major Pancreatic Duct Injuries in Children. J. Trauma Inj. Infect. Crit. Care 2001, 50, 1001–1007. [Google Scholar] [CrossRef] [PubMed]
- Koh, E.Y.; van Poll, D.; Goslings, J.C.; Busch, O.R.; Rauws, E.A.; Oomen, M.W.; Besselink, M.G. Operative Versus Nonoperative Management of Blunt Pancreatic Trauma in Children: A Systematic Review. Pancreas 2017, 46, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Haider, F.; Al Awadhi, M.A.; Abrar, E.; Al Dossari, M.; Isa, H.; Nasser, H.; Al Hashimi, H.; Al Arayedh, S. Pancreatic injury in children: A case report and review of the literature. J. Med. Case Rep. 2017, 11, 217. [Google Scholar] [CrossRef] [Green Version]
- Sutherland, I.; Ledder, O.; Crameri, J.; Nydegger, A.; Catto-Smith, A.; Cain, T.; Oliver, M. Pancreatic trauma in children. Pediatr. Surg. Int. 2010, 26, 1201–1206. [Google Scholar] [CrossRef]
- Jobst, M.A.; Canty, T.G.; Lynch, F.P. Management of pancreatic injury in pediatric blunt abdominal trauma. J. Pediatr. Surg. 1999, 34, 818–824. [Google Scholar] [CrossRef]
- Paul, M.D.; Mooney, D.P. The management of pancreatic injuries in children: Operate or observe. J. Pediatr. Surg. 2011, 46, 1140–1143. [Google Scholar] [CrossRef]
- Meier, D.E.; Coln, C.; Hicks, B.A.; Guzzetta, P.C. Early operation in children with pancreas transection. J. Pediatr. Surg. 2001, 36, 341–344. [Google Scholar] [CrossRef]
- Beres, A.L.; Wales, P.W.; Christison-Lagay, E.R.; McClure, M.E.; Fallat, M.E.; Brindle, M.E. Non-operative management of high-grade pancreatic trauma: Is it worth the wait? J. Pediatr. Surg. 2013, 48, 1060–1064. [Google Scholar] [CrossRef]
- Wood, J.H.; Partrick, D.A.; Bruny, J.L.; Sauaia, A.; Moulton, S.L. Operative vs nonoperative management of blunt pancreatic trauma in children. J. Pediatr. Surg. 2010, 45, 401–406. [Google Scholar] [CrossRef]
- 60th Annual Meeting of The Austrian Society of Surgery. Eur. Surg. 2019, 51, 1–107. [CrossRef]
- Mathuria, B.J.N.; Akinkuotu, A.C.; Wesson, D.E. Operative or Non-Operative Management for High-Grade Pediatric Pancreatic Trauma? A Systematic Review Still Leaves the Question Unanswered. Trauma Acute Care 2016, 1, 1–8. [Google Scholar] [CrossRef]
- Almaramhy, H.H.; Guraya, S.Y. Computed tomography for pancreatic injuries in pediatric blunt abdominal trauma. World J. Gastrointest. Surg. 2012, 4, 166–170. [Google Scholar] [CrossRef]
- Houben, C.H.; Ade-Ajayi, N.; Patel, S.; Kane, P.; Karani, J.; Devlin, J.; Harrison, P.; Davenport, M. Traumatic pancreatic duct injury in children: Minimally invasive approach to management. J. Pediatr. Surg. 2007, 42, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Rekhi, S.; Anderson, S.W.; Rhea, J.T.; Soto, J.A. Imaging of blunt pancreatic trauma. Emerg. Radiol. 2009, 17, 13–19. [Google Scholar] [CrossRef]
- Soto, J.A.; Alvarez, O.; Múnera, F.; Yepes, N.L.; Sepúlveda, M.E.; Pérez, J.M. Traumatic Disruption of the Pancreatic Duct: Diagnosis with MR pancreatography. Am. J. Roentgenol. 2001, 176, 175–178. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, E.H.; Vogel, A.M.; Klinkner, D.B.; Escobar, M.; Gaines, B.; Russell, R.; Campbell, B.; Wills, H.; Stallion, A.; Juang, D.; et al. The utility of ERCP in pediatric pancreatic trauma. J. Pediatr. Surg. 2017, 53, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Rescorla, F.J.; Plumley, D.A.; Sherman, S.; Scherer, L.R., 3rd; West, K.W.; Grosfeld, J.L. The efficacy of early ERCP in pediatric pancreatic trauma. J. Pediatr. Surg. 1995, 30, 336–340. [Google Scholar] [CrossRef]
- Subramanian, A.; Dente, C.J.; Feliciano, D.V. The Management of Pancreatic Trauma in the Modern Era. Surg. Clin. N. Am. 2007, 87, 1515–1532. [Google Scholar] [CrossRef]
- Beger, B.; Kızılyildiz, B.; Simsek, M.; Akdeniz, H.; Sonmez, B. Pancreatic injury due to blunt abdominal trauma in children. Med. Sci. Int. Med. J. 2019, 8, 225–228. [Google Scholar] [CrossRef]
- Kumar, A.; Panda, A.; Gamanagatti, S. Blunt pancreatic trauma: A persistent diagnostic conundrum? World J. Radiol. 2016, 8, 159–173. [Google Scholar] [CrossRef] [PubMed]
- Debi, U.; Kaur, R.; Prasad, K.K.; Sinha, S.K.; Sinha, A.; Singh, K. Pancreatic trauma: A concise review. World J. Gastroenterol. 2013, 19, 9003–9011. [Google Scholar] [CrossRef] [PubMed]
- Oniscu, G.C.; Parks, R.W.; Garden, O.J. Classification of liver and pancreatic trauma. HPB 2006, 8, 4–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moore, E.E.; Cogbill, T.H.; Malangoni, M.A.; Jurkovich, G.J.; Champion, H.R.; Gennarelli, T.A.; McAninch, J.W.; Pachter, H.L.; Shackford, S.R.; Trafton, P.G. Organ injury scaling, II: Pancreas, duodenum, small bowel, colon, and rectum. J. Trauma 1990, 30, 1427–1429. [Google Scholar] [CrossRef] [PubMed]
- Mattix, K.D.; Tataria, M.; Holmes, J.; Kristoffersen, K.; Brown, R.; Groner, J.; Scaife, E.; Mooney, D.; Nance, M.; Scherer, L. Pediatric pancreatic trauma: Predictors of nonoperative management failure and associated outcomes. J. Pediatr. Surg. 2007, 42, 340–344. [Google Scholar] [CrossRef] [PubMed]
- Haugaard, M.V.; Wettergren, A.; Hillingsø, J.G.; Gluud, C.; Penninga, L. Non-operative versus operative treatment for blunt pancreatic trauma in children. Cochrane Database Syst. Rev. 2014, 2, CD009746. [Google Scholar] [CrossRef] [Green Version]
- Goldberg-Murow, M.; Steiner, Z.; Lakovsky, Y.; Dlugy, E.; Baazov, A.; Freud, E.; Samuk, I. Blunt High-Grade Pancreatic Injury in Children: A 20-Year Experience in Two Pediatric Surgical Centers. ISR Med. Assoc. J. 2021, 23, 180–185. [Google Scholar]
- Naik-Mathuria, B.J.; Rosenfeld, E.H.; Gosain, A.; Burd, R.; Falcone, R.A., Jr.; Thakkar, R.; Gaines, B.; Mooney, D.; Escobar, M.; Jafri, M.; et al. Proposed clinical pathway for nonoperative management of high-grade pediatric pancreatic injuries based on a multicenter analysis: A pediatric trauma society collaborative. J. Trauma Acute Care Surg. 2017, 83, 589–596. [Google Scholar] [CrossRef]
- Naik-Mathuria, B. Practice variability exists in the management of high-grade pediatric pancreatic trauma. Pediatr. Surg. Int. 2016, 32, 789–794. [Google Scholar] [CrossRef]
- Mora, M.C.; Wong, K.E.; Friderici, J.; Bittner, K.; Moriarty, K.P.; Patterson, L.A.; Gross, R.I.; Tirabassi, M.V.; Tashjian, D.B. Operative vs Nonoperative Management of Pediatric Blunt Pancreatic Trauma: Evaluation of the National Trauma Data Bank. J. Am. Coll. Surg. 2016, 222, 977–982. [Google Scholar] [CrossRef]
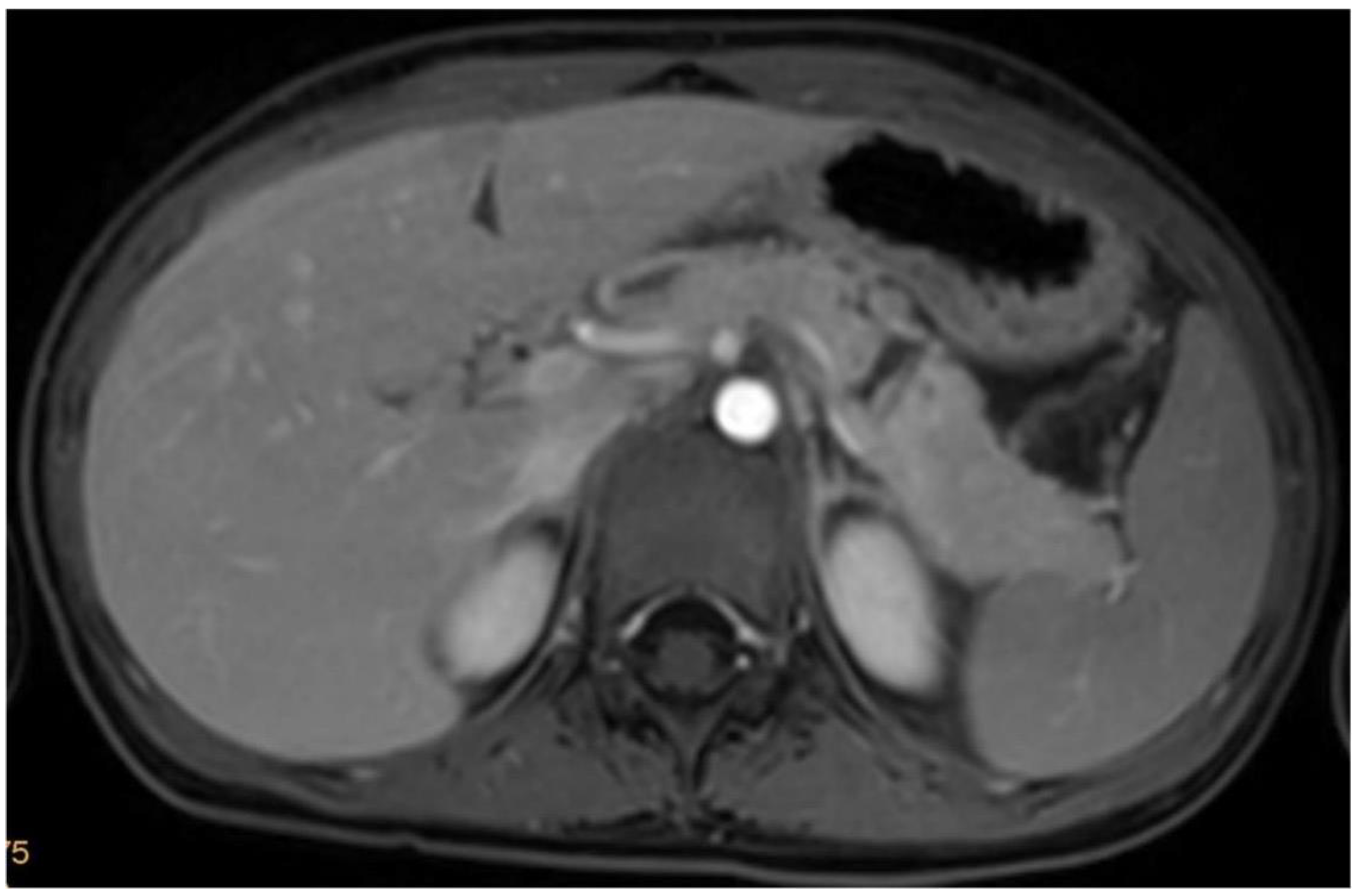
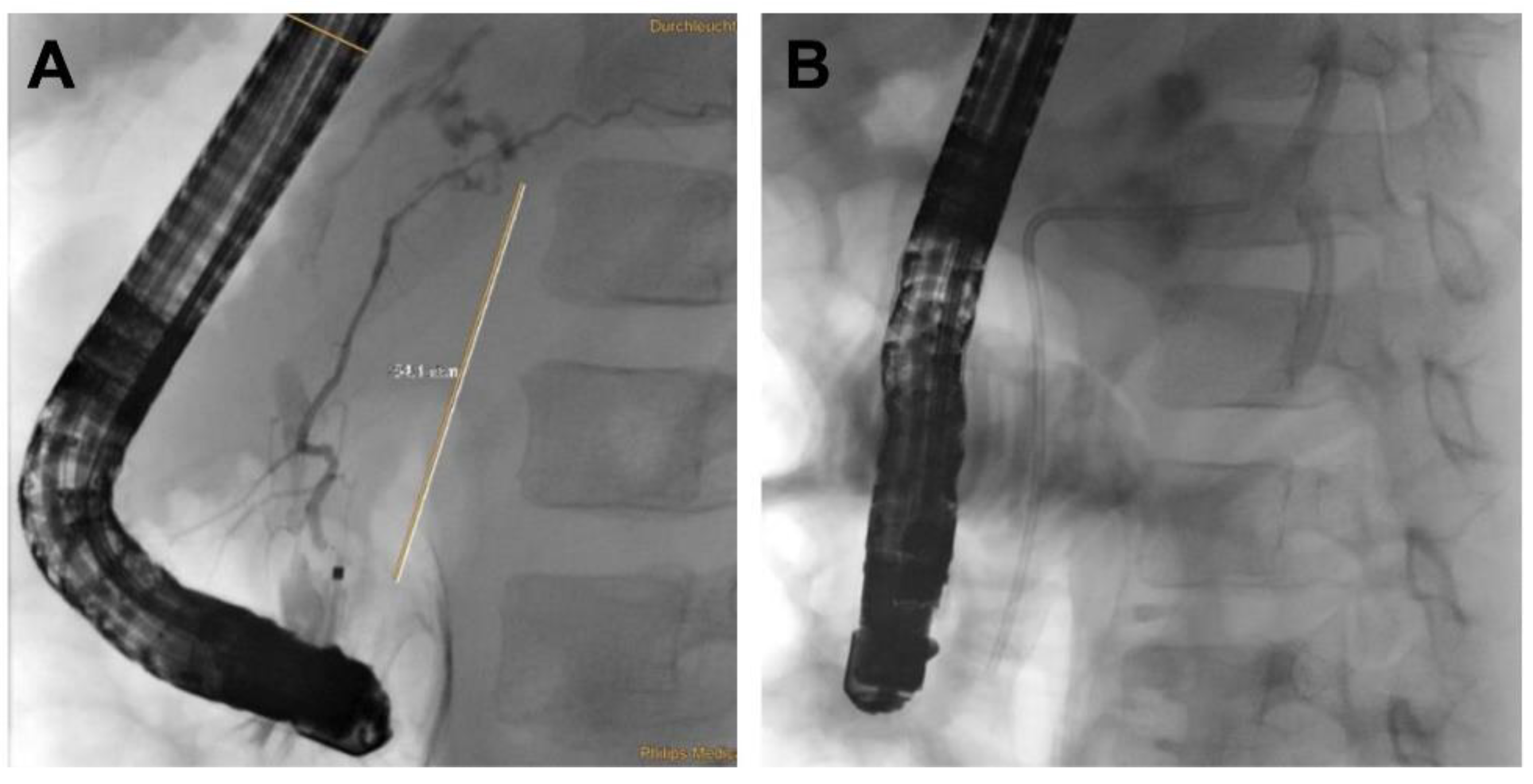
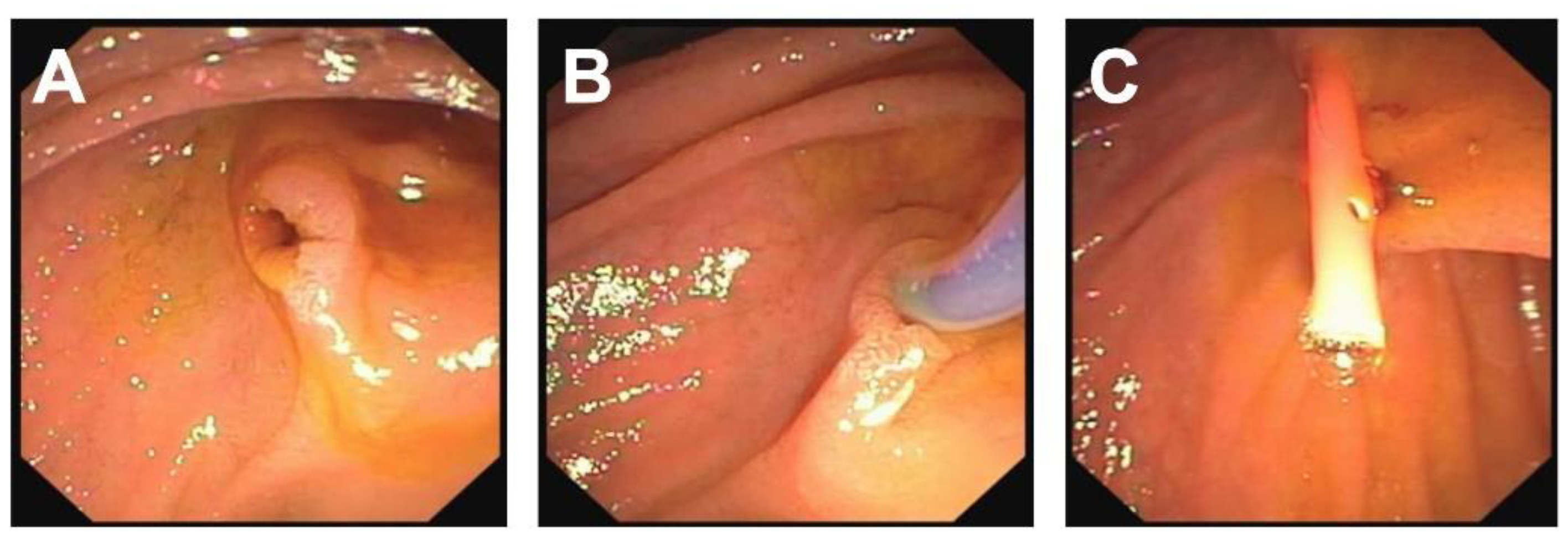
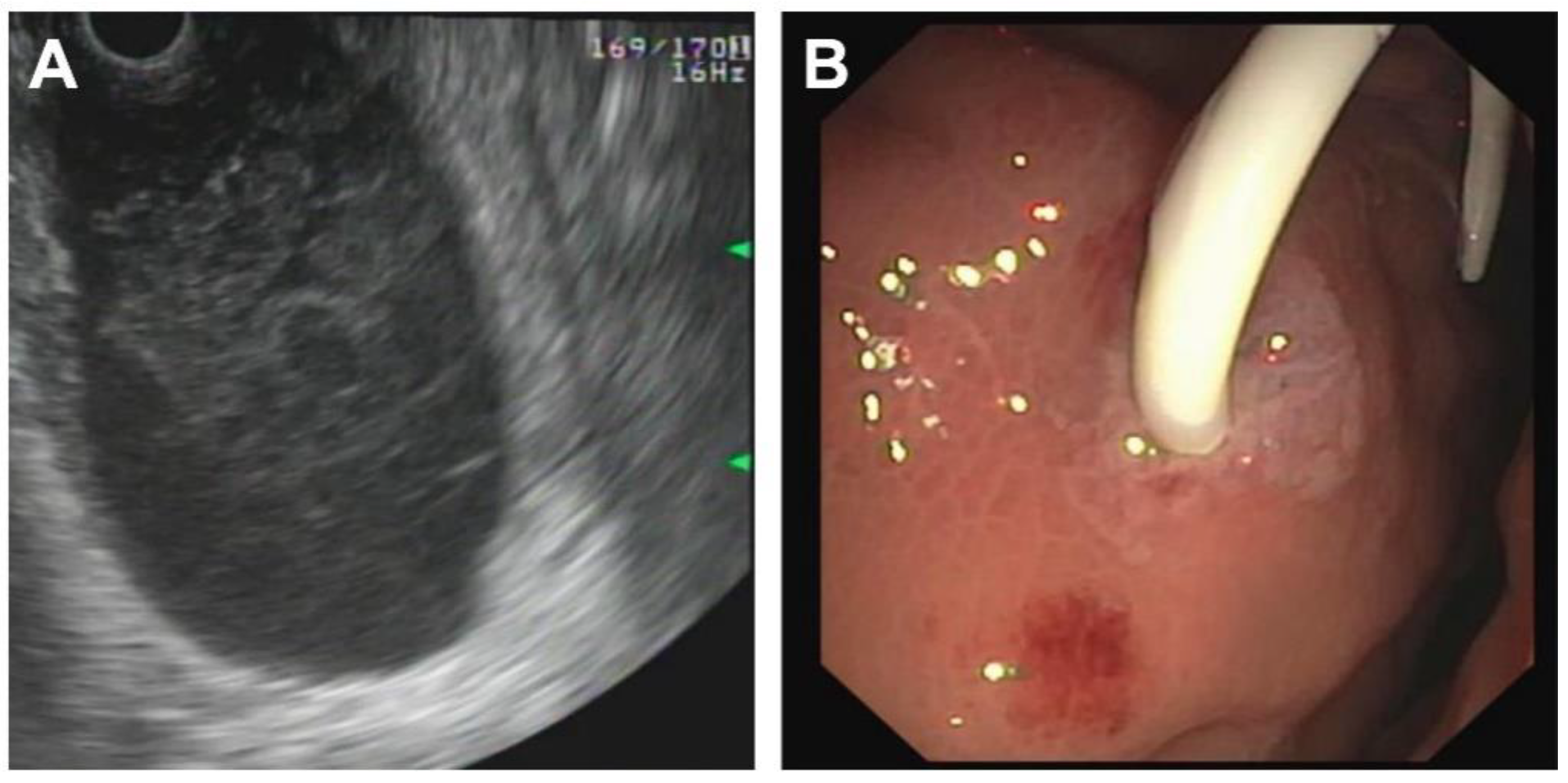
| Patient 1 | Patient 2 | Patient 3 | Patient 4 | |
|---|---|---|---|---|
| Age | 13 | 6 | 7 | 4 |
| Sex | Male | Male | Female | Male |
| Cause of trauma | Bike accident | Motor vehicle accident (patient run over by agricultural vehicle) | Non-motorized scooter accident | Motor vehicle accident (collision as car passenger) |
| Initial diagnosis obtained by | Computed Tomography | Computed Tomography | Magnetic Resonance Tomography | Computed Tomography |
| Additional injuries | Retroperitoneal Hematoma | Jejunal Perforation, Lung Contusions, Unstable Pelvic Fracture (External Fixation) | None | Lung Contusions, Hepatic rupture, left pneumothorax |
| Grade of Pancreatic Injury | IV | IV | III | III |
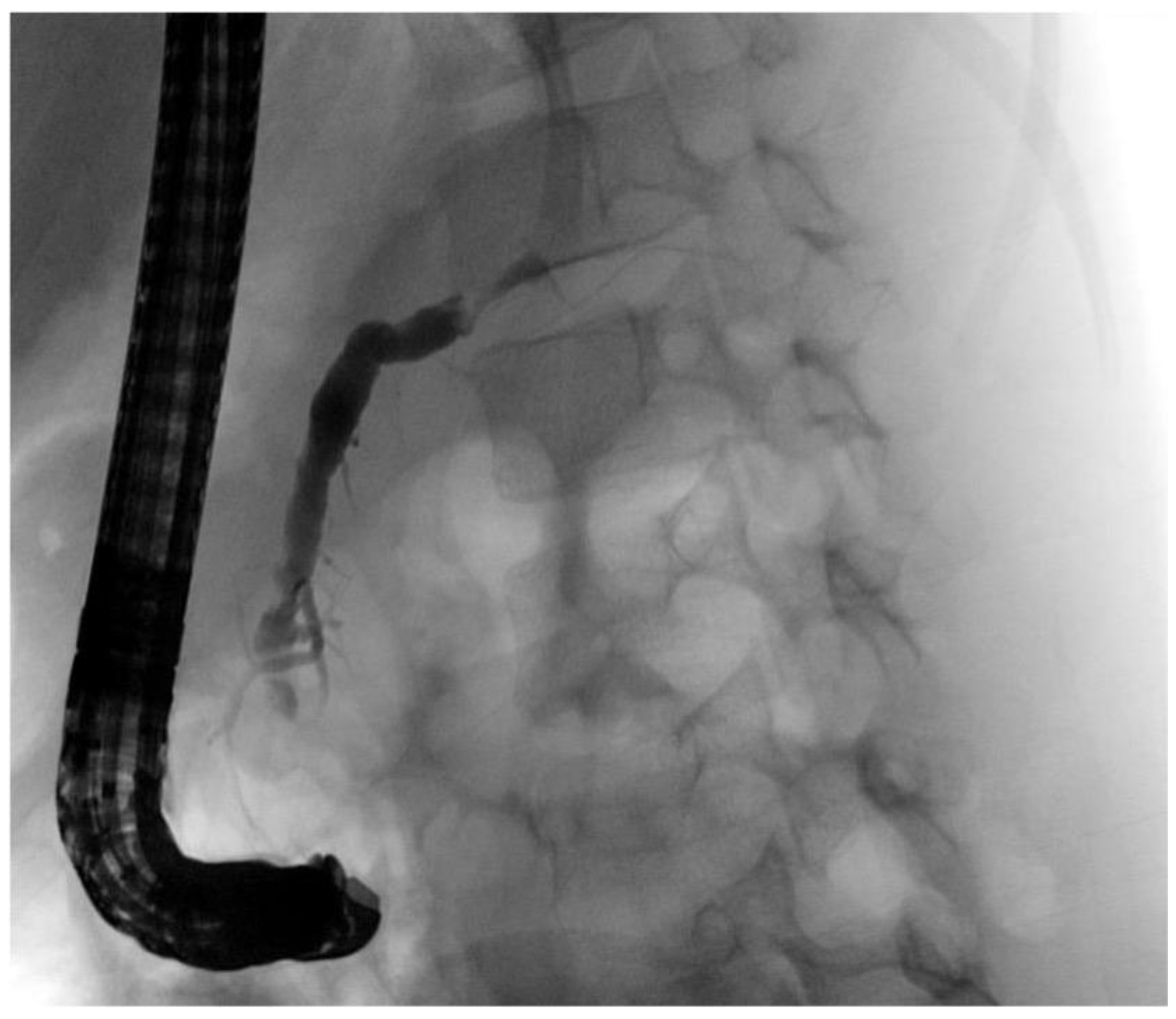
| Initial Management | Laparotomy, suture of the pancreatic head, distal pancreato-jejunostomy (Roux Y) | ERCP—complete dissection of the pancreas, Laparotomy, Jejunal repair, suture of the pancreatic head, distal pancreato-gastrostomy | ERCP—stenting of the ruptured pancreatic duct with 5 Ch pigtail drainage | ERCP unsuccessful, stenting of ruptured pancreatic duct not possible; chest drain (left side) |
| Postoperative Complications | Portal vein thrombosis with partial obstruction | None | Symptomatic pancreatic pseudocyst infection with Clostridium difficile | Symptomatic pancreatic pseudocyst Candida albicans sepsis; Posttraumatic stress disorder |
| Management of complications | Anticoagulant therapy | - | Transgastric punction and drainage of pseudocyst with double-pig tail drain Ch 7, spontaneous dislocation of pig tail Antibiotic therapy Persistent fistula of the ruptured pancreatic duct, recurrent stenting (2 times), removal of stent after 5 months | Transgastric punction and drainage of pseudocyst with double-pig tail Ch 7, spontaneous dislocation of pig tail Antifungal therapy |
| Duration of hospital stay | 21 days | 30 days | 26 days | 39 days |
| Follow Up | 12 yrs | 11.5 yrs | 6.5 yrs | 3.5 yrs |
| Grading | Type of Injury | Description |
|---|---|---|
| Grade I | Hematoma Laceration | Minor contusion without duct injury Superficial laceration without duct injury |
| Grade II | Hematoma Laceration | Major contusion without duct injury or tissue loss Major laceration without duct injury or tissue loss |
| Grade III | Laceration | Distal transection or parenchymal injury with duct injury |
| Grade IV | Laceration | Proximal transection or parenchymal injury involving the ampulla |
| Grade V | Laceration | Massive disruption of the pancreatic head |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stundner-Ladenhauf, H.N.; Bauer, L.; Heil, C.; Holzinger, J.; Stundner, O.; Metzger, R. Minimally Invasive Approaches for Traumatic Rupture of the Pancreas in Children—A Case Series. Children 2022, 9, 1102. https://doi.org/10.3390/children9081102
Stundner-Ladenhauf HN, Bauer L, Heil C, Holzinger J, Stundner O, Metzger R. Minimally Invasive Approaches for Traumatic Rupture of the Pancreas in Children—A Case Series. Children. 2022; 9(8):1102. https://doi.org/10.3390/children9081102
Chicago/Turabian StyleStundner-Ladenhauf, Hannah Noemi, Leopold Bauer, Christian Heil, Josef Holzinger, Ottokar Stundner, and Roman Metzger. 2022. "Minimally Invasive Approaches for Traumatic Rupture of the Pancreas in Children—A Case Series" Children 9, no. 8: 1102. https://doi.org/10.3390/children9081102
APA StyleStundner-Ladenhauf, H. N., Bauer, L., Heil, C., Holzinger, J., Stundner, O., & Metzger, R. (2022). Minimally Invasive Approaches for Traumatic Rupture of the Pancreas in Children—A Case Series. Children, 9(8), 1102. https://doi.org/10.3390/children9081102







