Modifiable Risk Factors of Non-Syndromic Orofacial Clefts: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol and Registration
2.2. Search Processing
2.3. Inclusion Criteria
2.4. Data Processing
3. Results
3.1. Study Selection and Characteristics
3.2. Outcomes
4. Discussion
4.1. Non-Modifiable Risk Factors for Non-Syndromic Orofacial Clefts (NSOFCs)
4.1.1. Gender of the Newborn and Ethnicity
4.1.2. Familiarity
4.2. Modifiable Risk Factors for Non-Syndromic Orofacial Clefts (NSOFCs) before Conception
4.2.1. Consanguinity
4.2.2. Parental Age
4.2.3. Socioeconomic Level and Education
4.2.4. Area of Residency and Climate
4.3. Modifiable Risk Factors for Non-Syndromic Orofacial Clefts (NSOFCs) before and after Conception
4.3.1. Weight
4.3.2. Nutritional State
4.3.3. Psychophysical Stress
4.3.4. Acute and Chronic Diseases
Diabetes
Hypertension
Angina Pectoris
Infections
Folate and Cobalamin Deficiency
Other Diseases
4.3.5. Licit Drugs and Vitamins
| Drug | Type of OFCs | Gestational Period | References |
|---|---|---|---|
| Trimethoprim | CP | 1st trimester | [129] |
| Doxycycline/tetracycline | CL/P | 2nd month | [129] |
| Pivmecillinam | CP | 3rd month | [129] |
| Amoxicillin | CL/P CL/P, CP | 1st trimester 3rd trimester | [127] |
| Prednisone | CL/P | 1st trimester | [125] |
| Triamcinolone | CLP | 1st trimester | [125] |
| Beclomethasone | CLP, CP | 1st trimester | [125] |
| Ondansetron | CP | 1st trimester | [130] |
| Topiramate | CL/P | 1st trimester | [131] |
| Anticonvulsants | CL/P | Not reported | [34] |
| Retinoic acid | CL/P | Not reported | [34] |
4.3.6. Illicit Drugs
4.3.7. Smoke and Alcohol
4.3.8. Pollutants and Contaminants
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ADH1C | maternal alcohol metabolizing genes; |
| BMI | body mass index; |
| Cas | congenital abnormalities; |
| CL | cleft lip; |
| CL/P | cleft lip with or without cleft palate; |
| CLP | cleft lip and palate; |
| CO | carbon monoxide; |
| CP | cleft palate; |
| CTS | corticosteroids; |
| DBPs | water disinfection by products; |
| DM | diabetes mellitus; |
| GDM | gestational diabetes mellitus; |
| HAAs | haloacetic acids; |
| MAP | maternal angina pectoris; |
| NO2 | nitrogen dioxide; |
| NOWS | neonatal opiate abstinence syndrome; |
| NSCL/P | non syndromic cleft lip with or without cleft palate; |
| NSOFCs | non syndromic orofacial clefts; |
| OFCs | orofacial clefts; |
| PAHs | polycyclic aromatic hydrocarbons; |
| PM2.5 | particulate matter with aerodynamic diameter ≤2.5 μm; SO2: sulfur dioxide; |
| TFGA | transforming growth factor alfa; |
| THMs | trihalomethanes. |
References
- Sharp, G.C.; Ho, K.; Davies, A.; Stergiakouli, E.; Humphries, K.; McArdle, W.; Sandy, J.; Davey Smith, G.; Lewis, S.J.; Relton, C.L. Distinct DNA Methylation Profiles in Subtypes of Orofacial Cleft. Clin. Epigenet. 2017, 9, 63. [Google Scholar] [CrossRef] [PubMed]
- Wehby, G.; Cassell, C.H. The Impact of Orofacial Clefts on Quality of Life and Health Care Use and Costs. Oral Dis. 2010, 16, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Dixon, M.J.; Marazita, M.L.; Beaty, T.H.; Murray, J.C. Cleft Lip and Palate: Understanding Genetic and Environmental Influences. Nat. Rev. Genet. 2011, 12, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Sforza, C.; Ferrario, V.F. Three-Dimensional Analysis of Facial Morphology: Growth, Development and Aging of the Orolabial Region. Ital. J. Anat. Embryol. 2010, 115, 141–145. [Google Scholar]
- Kreiborg, S.; Herman, N.V.; Darvann, T.A. Characteristics of facial morphology and growth in infants with clefts. In Cleft Lip and Palate; Berkowitz, S., Ed.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 225–235. ISBN 978-3-540-30020-5. [Google Scholar]
- Mossey, P.A.; Little, J.; Munger, R.G.; Dixon, M.J.; Shaw, W.C. Cleft Lip and Palate. Lancet 2009, 374, 1773–1785. [Google Scholar] [CrossRef] [PubMed]
- Mcbride, W.A.; Mcintyre, G.T.; Carroll, K.; Mossey, P.A. Subphenotyping and Classification of Orofacial Clefts: Need for Orofacial Cleft Subphenotyping Calls for Revised Classification. 2016. Available online: https://journals.sagepub.com/doi/10.1597/15-029?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed (accessed on 3 October 2022).
- Houkes, R.; Smit, J.; Mossey, P.; Don Griot, P.; Persson, M.; Neville, A.; Ongkosuwito, E.; Sitzman, T.; Breugem, C. Classification Systems of Cleft Lip, Alveolus and Palate: Results of an International Survey. Cleft Palate Craniofac. J. 2021, 105566562110573. [Google Scholar] [CrossRef]
- Saxén, I. Epidemiology of Cleft Lip and Palate. An Attempt to Rule out Chance Correlations. Br. J. Prev. Soc. Med. 1975, 29, 103–110. [Google Scholar] [CrossRef]
- Hlongwa, P.; Levin, J.; Rispel, L.C. Epidemiology and Clinical Profile of Individuals with Cleft Lip and Palate Utilising Specialised Academic Treatment Centres in South Africa. PLoS ONE 2019, 14, e0215931. [Google Scholar] [CrossRef]
- Parada, C.; Chai, Y. Roles of BMP Signaling Pathway in Lip and Palate Development. Front. Oral Biol. 2012, 16, 60–70. [Google Scholar] [CrossRef]
- Stanier, P.; Moore, G.E. Genetics of Cleft Lip and Palate: Syndromic Genes Contribute to the Incidence of Non-Syndromic Clefts. Hum. Mol. Genet. 2004, 13, R73–R81. [Google Scholar] [CrossRef]
- Fraser, F.C. The Genetics of Cleft Lip and Cleft Palate. Am. J. Hum. Genet. 1970, 22, 336–352. [Google Scholar] [PubMed]
- Mitchell, L.E.; Risch, N. Mode of Inheritance of Nonsyndromic Cleft Lip with or without Cleft Palate: A Reanalysis. Am. J. Hum. Genet. 1992, 51, 323–332. [Google Scholar] [PubMed]
- Harville, E.W.; Wilcox, A.J.; Lie, R.T.; Vindenes, H.; Abyholm, F. Cleft Lip and Palate versus Cleft Lip Only: Are They Distinct Defects? Am. J. Epidemiol. 2005, 162, 448–453. [Google Scholar] [CrossRef] [PubMed]
- Kuriyama, M.; Udagawa, A.; Yoshimoto, S.; Ichinose, M.; Sato, K.; Yamazaki, K.; Matsuno, Y.; Shiota, K.; Mori, C. DNA Methylation Changes during Cleft Palate Formation Induced by Retinoic Acid in Mice. Cleft Palate Craniofac. J. 2008, 45, 545–551. [Google Scholar] [CrossRef]
- Radhakrishna, U. Small Players with a Big Role: MicroRNAs in Pathophysiology of Cleft Lip and Palate. Indian J. Hum. Genet. 2012, 18, 272–273. [Google Scholar] [CrossRef][Green Version]
- Peña-Soto, C.; Arriola-Guillén, L.-E.; Díaz-Suyo, A.; Flores-Fraile, J. Clinical and Epidemiological Profile of Cleft Lip and Palate Patients in Peru, 2006–2019. J. Clin. Exp. Dent. 2021, 13, e1118–e1123. [Google Scholar] [CrossRef]
- Habibi, M.; Vahdat, S. Evaluation of the Correlation between Socioeconomic Factors and Pediatric Cleft Lip and Palate. Mod. Med. Lab. J. 2021, 4, 40–49. [Google Scholar] [CrossRef]
- Murray, J. Gene/Environment Causes of Cleft Lip and/or Palate. Clin. Genet. 2002, 61, 248–256. [Google Scholar] [CrossRef]
- Li, Z.; Ren, A.; Liu, J.; Pei, L.; Zhang, L.; Guo, Z.; Li, Z. Maternal Flu or Fever, Medication Use, and Neural Tube Defects: A Population-Based Case-Control Study in Northern China. Birth Defects Res. A Clin. Mol. Teratol. 2007, 79, 295–300. [Google Scholar] [CrossRef]
- Jia, Z.-L.; Shi, B.; Chen, C.-H.; Shi, J.-Y.; Wu, J.; Xu, X. Maternal Malnutrition, Environmental Exposure during Pregnancy and the Risk of Non-Syndromic Orofacial Clefts. Oral Dis. 2011, 17, 584–589. [Google Scholar] [CrossRef]
- James, O.; Erinoso, O.; Ogunlewe, A.; Adeyemo, W.; Ladeinde, A.; Ogunlewe, M. Parental Age and the Risk of Cleft Lip and Palate in a Nigerian Population—A Case–Control Study. Ann. Maxillofac. Surg. 2020, 10, 429. [Google Scholar] [CrossRef] [PubMed]
- Berg, E.; Lie, R.T.; Sivertsen, Å.; Haaland, Ø.A. Parental Age and the Risk of Isolated Cleft Lip: A Registry-Based Study. Ann. Epidemiol. 2015, 25, 942–947.e1. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, P.H.P.; Machado, R.A.; de Reis, S.R.A.; Martelli, D.R.B.; Dias, V.O.; Martelli Júnior, H. Parental Age Is Related to the Occurrence of Cleft Lip and Palate in Brazilian Populations. Braz. J. Oral Sci. 2017, 15, 167. [Google Scholar] [CrossRef]
- Moore, L.L.; Singer, M.R.; Bradlee, M.L.; Rothman, K.J.; Milunsky, A. A Prospective Study of the Risk of Congenital Defects Associated with Maternal Obesity and Diabetes Mellitus. Epidemiology 2000, 11, 689–694. [Google Scholar] [CrossRef] [PubMed]
- Spilson, S.V.; Kim, H.J.; Chung, K.C. Association between Maternal Diabetes Mellitus and Newborn Oral Cleft. Ann. Plast. Surg. 2001, 47, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Meng, T.; Shi, B.; Zheng, Q.; Wang, Y.; Li, S. Clinical and Epidemiologic Studies of Nonsyndromic Cleft Lip and Palate in China: Analysis of 4268 Cases. Ann. Plast. Surg. 2006, 57, 264–269. [Google Scholar] [CrossRef]
- Czeizel, A.E.; Vereczkey, A.; Bánhidy, F. Higher Risk of Orofacial Clefts in Children Born to Mothers with Angina Pectoris: A Population-Based Case-Control Study. Congenit. Anom. 2015, 55, 49–54. [Google Scholar] [CrossRef]
- Kutbi, H.; Wehby, G.L.; Moreno Uribe, L.M.; Romitti, P.A.; Carmichael, S.; Shaw, G.M.; Olshan, A.F.; DeRoo, L.; Rasmussen, S.A.; Murray, J.C.; et al. Maternal Underweight and Obesity and Risk of Orofacial Clefts in a Large International Consortium of Population-Based Studies. Int. J. Epidemiol. 2017, 46, 190–199. [Google Scholar] [CrossRef][Green Version]
- Trindade-Suedam, I.K.; von Kostrisch, L.M.; Pimenta, L.A.F.; Negrato, C.A.; Franzolin, S.B.; Trindade, A.S. Diabetes Mellitus and Drug Abuse during Pregnancy and the Risk for Orofacial Clefts and Related Abnormalities. Rev. Lat. Am. Enferm. 2016, 24, e2701. [Google Scholar] [CrossRef]
- Neogi, S.B.; Singh, S.; Pallepogula, D.R.; Pant, H.; Kolli, S.R.; Bharti, P.; Datta, V.; Gosla, S.R.; Bonanthaya, K.; Ness, A.; et al. Risk Factors for Orofacial Clefts in India: A Case–Control Study. Birth Defects Res. 2017, 109, 1284–1291. [Google Scholar] [CrossRef]
- Butali, A.; Little, J.; Chevrier, C.; Cordier, S.; Steegers-Theunissen, R.; Jugessur, A.; Oladugba, B.; Mossey, P.A. Folic Acid Supplementation Use and the MTHFR C677T Polymorphism in Orofacial Clefts Etiology: An Individual Participant Data Pooled-Analysis: Folic Acid Supplementation and MTHFR C677T Polymorphism in OFC. Birth Defects Res. Part A Clin. Mol. Teratol. 2013, 97, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Nahas, L.D.; Alzamel, O.; Dali, M.Y.; Alsawah, R.; Hamsho, A.; Sulman, R.; Alzamel, M.; Omar, A. Distribution and Risk Factors of Cleft Lip and Palate on Patients from a Sample of Damascus Hospitals—A Case-Control Study. Heliyon 2021, 7, e07957. [Google Scholar] [CrossRef] [PubMed]
- Rozendaal, A.M.; van Essen, A.J.; te Meerman, G.J.; Bakker, M.K.; van der Biezen, J.J.; Goorhuis-Brouwer, S.M.; Vermeij-Keers, C.; de Walle, H.E.K. Periconceptional Folic Acid Associated with an Increased Risk of Oral Clefts Relative to Non-Folate Related Malformations in the Northern Netherlands: A Population Based Case-Control Study. Eur. J. Epidemiol. 2013, 28, 875–887. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.Y.; Little, J. Folate Intake, Markers of Folate Status and Oral Clefts: Is the Evidence Converging? Int. J. Epidemiol. 2008, 37, 1041–1058. [Google Scholar] [CrossRef] [PubMed]
- Ebadifar, A.; Hamedi, R.; KhorramKhorshid, H.R.; Kamali, K.; Moghadam, F.A. Parental Cigarette Smoking, Transforming Growth Factor-alpha Gene Variant and the Risk of Orofacial Cleft in Iranian Infants. Iran. J. Basic Med. Sci. 2016, 19, 9. [Google Scholar]
- Kummet, C.M.; Moreno, L.M.; Wilcox, A.J.; Romitti, P.A.; DeRoo, L.A.; Munger, R.G.; Lie, R.T.; Wehby, G.L. Passive Smoke Exposure as a Risk Factor for Oral Clefts—A Large International Population-Based Study. Am. J. Epidemiol. 2016, 183, 834–841. [Google Scholar] [CrossRef]
- Little, J.; Cardy, A.; Munger, R.G. Tobacco Smoking and Oral Clefts: A Meta-Analysis. Bull. World Health Organ. 2004, 82, 213–218. [Google Scholar]
- Lorente, C.; Cordier, S.; Goujard, J.; Aymé, S.; Bianchi, F.; Calzolari, E.; De Walle, H.E.; Knill-Jones, R. Tobacco and Alcohol Use during Pregnancy and Risk of Oral Clefts. Occupational Exposure and Congenital Malformation Working Group. Am. J. Public Health 2000, 90, 415–419. [Google Scholar]
- Welberg, L.A.M.; Thrivikraman, K.V.; Plotsky, P.M. Chronic Maternal Stress Inhibits the Capacity to Up-Regulate Placental 11beta-Hydroxysteroid Dehydrogenase Type 2 Activity. J. Endocrinol. 2005, 186, R7–R12. [Google Scholar] [CrossRef]
- O’Donnell, K.J.; Bugge Jensen, A.; Freeman, L.; Khalife, N.; O’Connor, T.G.; Glover, V. Maternal Prenatal Anxiety and Downregulation of Placental 11β-HSD2. Psychoneuroendocrinology 2012, 37, 818–826. [Google Scholar] [CrossRef]
- Jiang, W.; Xie, W.; Ni, B.; Zhou, H.; Liu, Z.; Li, X. First Trimester Exposure to Ambient Gaseous Air Pollutants and Risk of Orofacial Clefts: A Case–Control Study in Changsha, China. BMC Oral Health 2021, 21, 530. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhang, C.; Liu, D.; Grantz, K.L.; Wallace, M.; Mendola, P. Maternal Ambient Air Pollution Exposure Preconception and during Early Gestation and Offspring Congenital Orofacial Defects. Environ. Res. 2015, 140, 714–720. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Gilboa, S.M.; Herdt, M.L.; Lupo, P.J.; Flanders, W.D.; Liu, Y.; Shin, M.; Canfield, M.A.; Kirby, R.S. Maternal Exposure to Ozone and PM2.5 and the Prevalence of Orofacial Clefts in Four U.S. States. Environ. Res. 2017, 153, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Ahmed Sakran, K.; Mutahar Abotaleb, B.; Khaled Al-Rokhami, R.; Hsieh, T.; Al-Wesabi, M.A.; Mohammed, A.A.; Al-Sharani, H.M.; Shi, P.; He, D. Analysis of Environmental Exposures for Nonsyndromic Cleft Lip and/or Palate: A Case-Control Study. Iran. J. Public Health 2022, 51, 578. [Google Scholar] [CrossRef] [PubMed]
- Cheshmi, B.; Jafari, Z.; Naseri, M.A.; Davari, H.A. Assessment of the Correlation between Various Risk Factors and Orofacial Cleft Disorder Spectrum: A Retrospective Case-Control Study. Maxillofac. Plast. Reconstr. Surg. 2020, 42, 26. [Google Scholar] [CrossRef] [PubMed]
- Dien, V.H.A.; McKinney, C.M.; Pisek, A.; Pitiphat, W. Maternal Exposures and Risk of Oral Clefts in South Vietnam. Birth Defects Res. 2018, 110, 527–537. [Google Scholar] [CrossRef]
- Shibukawa, B.M.C.; Rissi, G.P.; Higarashi, I.H.; de Oliveira, R.R. Factors Associated with the Presence of Cleft Lip and/or Cleft Palate in Brazilian Newborns. Rev. Bras. Saude Mater. Infant. 2019, 19, 947–956. [Google Scholar] [CrossRef]
- Lei, R.-L.; Chen, H.-S.; Huang, B.-Y.; Chen, Y.-C.; Chen, P.K.-T.; Lee, H.-Y.; Chang, C.-W.; Wu, C.-L. Population-Based Study of Birth Prevalence and Factors Associated with Cleft Lip and/or Palate in Taiwan 2002–2009. PLoS ONE 2013, 8, e58690. [Google Scholar] [CrossRef]
- Malic, C.C.; Lam, M.; Donelle, J.; Richard, L.; Vigod, S.N.; Benchimol, E.I. Incidence, Risk Factors, and Mortality Associated With Orofacial Cleft Among Children in Ontario, Canada. JAMA Netw. Open 2020, 3, e1921036. [Google Scholar] [CrossRef]
- Figueiredo, J.C.; Ly, S.; Magee, K.S.; Ihenacho, U.; Baurley, J.W.; Sanchez-Lara, P.A.; Brindopke, F.; Nguyen, T.; Nguyen, V.; Tangco, M.I.; et al. Parental Risk Factors for Oral Clefts among Central Africans, Southeast Asians, and Central Americans. Birth Defects Res. Part A Clin. Mol. Teratol. 2015, 103, 863–879. [Google Scholar] [CrossRef]
- Salihu, S.; Krasniqi, B.; Sejfija, O.; Heta, N.; Salihaj, N.; Geci, A.; Sejdini, M.; Arifi, H.; Isufi, R.; Ueeck, B.A. Analysis of Potential Oral Cleft Risk Factors in the Kosovo Population. Int. Surg. 2014, 99, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Kapos, F.P.; White, L.A.; Schmidt, K.A.; Hawes, S.E.; Starr, J.R. Risk of Non-syndromic Orofacial Clefts by Maternal Rural-urban Residence and Race/Ethnicity: A Population-based Case-control Study in Washington State 1989–2014. Paediatr. Perinat. Epidemiol. 2021, 35, 292–301. [Google Scholar] [CrossRef]
- Da Silva, H.P.V.; Arruda, T.T.S.; de Souza, K.S.C.; Bezerra, J.F.; Leite, G.C.P.; de Brito, M.E.F.; Lima, V.M.G.D.M.; Luchessi, A.D.; Bortolin, R.H.; Ururahy, M.A.G.; et al. Risk Factors and Comorbidities in Brazilian Patients with Orofacial Clefts. Braz. Oral Res. 2018, 32, e24. [Google Scholar] [CrossRef] [PubMed]
- Ly, S.; Burg, M.; Ihenacho, U.; Brindopke, F.; Auslander, A.; Magee, K.; Sanchez-Lara, P.; Nguyen, T.-H.-D.; Nguyen, V.; Tangco, M.; et al. Paternal Risk Factors for Oral Clefts in Northern Africans, Southeast Asians, and Central Americans. Int. J. Environ. Res. Public Health 2017, 14, 657. [Google Scholar] [CrossRef]
- Noorollahian, M.; Nematy, M.; Dolatian, A.; Ghesmati, H.; Akhlaghi, S.; Khademi, G. Cleft Lip and Palate and Related Factors: A 10 Years Study in University Hospitalised Patients at Mashhad—Iran. Afr. J. Paediatr. Surg. 2015, 12, 286. [Google Scholar] [CrossRef] [PubMed]
- Heidari, F.; Dastgiri, S.; Akbari, R.; Khamnian, Z.; Khanlarzadeh, E.; Baradaran, M.; Jabbary-Fam, S.; Badrazar, S.; Heidarzadeh, M.; Tajaddini, N. Prevalence and Risk Factors of Consanguineous Marriage. Eur. J. Gen. Med. 2014, 11, 248–255. [Google Scholar] [CrossRef]
- Brito, L.A.; Cruz, L.A.; Rocha, K.M.; Barbara, L.K.; Silva, C.B.F.; Bueno, D.F.; Aguena, M.; Bertola, D.R.; Franco, D.; Costa, A.M.; et al. Genetic Contribution for Non-Syndromic Cleft Lip with or without Cleft Palate (NS CL/P) in Different Regions of Brazil and Implications for Association Studies. Am. J. Med. Genet. A 2011, 155, 1581–1587. [Google Scholar] [CrossRef]
- Martelli, D.-R.; Bonan, P.-R.-F.; Soares, M.-C.; Paranaíba, L.-R.; Martelli-Júnior, H. Analysis of Familial Incidence of Non-Syndromic Cleft Lip and Palate in a Brazilian Population. Med. Oral Patol. Oral Cir. Bucal 2010, 15, e898–e901. [Google Scholar] [CrossRef]
- Yang, J.; Carmichael, S.L.; Canfield, M.; Song, J.; Shaw, G.M.; National Birth Defects Prevention Study. Socioeconomic Status in Relation to Selected Birth Defects in a Large Multicentered US Case-Control Study. Am. J. Epidemiol. 2008, 167, 145–154. [Google Scholar] [CrossRef]
- Lupo, P.J.; Danysh, H.E.; Symanski, E.; Langlois, P.H.; Cai, Y.; Swartz, M.D. Neighborhood-Based Socioeconomic Position and Risk of Oral Clefts Among Offspring. Am. J. Public Health 2015, 105, 2518–2525. [Google Scholar] [CrossRef]
- Amidei, R.L.; Hamman, R.F.; Kassebaum, D.K.; Marshall, J.A. Birth Prevalence of Cleft Lip and Palate in Colorado by Sex Distribution, Seasonality, Race/Ethnicity, and Geographic Variation. Spec. Care Dent. 1994, 14, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Materna-Kiryluk, A.; Więckowska, B.; Wiśniewska, K.; Czyżewska, M.; Godula-Stuglik, U.; Jaworska-Bobkier, R.; Walencka, Z.; Kucharska, Z.; Latos-Bieleńska, A. Spatial and Temporal Clustering of Isolated Cleft Lip with or without Cleft Palate in Poland. Int. J. Environ. Health Res. 2014, 24, 567–579. [Google Scholar] [CrossRef] [PubMed]
- De Loffredo, L.C.; de Souza, J.M.; Yunes, J.; Freitas, J.A.; Spiri, W.C. Cleft lip and palate: Case-control study. Rev. Saude Publica 1994, 28, 213–217. [Google Scholar] [CrossRef]
- Messer, L.C.; Luben, T.J.; Mendola, P.; Carozza, S.E.; Horel, S.A.; Langlois, P.H. Urban-Rural Residence and the Occurrence of Cleft Lip and Cleft Palate in Texas, 1999–2003. Ann. Epidemiol. 2010, 20, 32–39. [Google Scholar] [CrossRef]
- Lynberg, M.C.; Khoury, M.J.; Lu, X.; Cocian, T. Maternal Flu, Fever, and the Risk of Neural Tube Defects: A Population-Based Case-Control Study. Am. J. Epidemiol. 1994, 140, 244–255. [Google Scholar] [CrossRef] [PubMed]
- Suarez, L.; Felkner, M.; Hendricks, K. The Effect of Fever, Febrile Illnesses, and Heat Exposures on the Risk of Neural Tube Defects in a Texas-Mexico Border Population. Birth Defects Res. A Clin. Mol. Teratol. 2004, 70, 815–819. [Google Scholar] [CrossRef] [PubMed]
- Soim, A.; Sheridan, S.C.; Hwang, S.; Hsu, W.; Fisher, S.C.; Shaw, G.M.; Feldkamp, M.L.; Romitti, P.A.; Reefhuis, J.; Langlois, P.H.; et al. A Population-based Case–Control Study of the Association between Weather-related Extreme Heat Events and Orofacial Clefts. Birth Defects Res. 2018, 110, 1468–1477. [Google Scholar] [CrossRef]
- Malik, V.S.; Willett, W.C.; Hu, F.B. Global Obesity: Trends, Risk Factors and Policy Implications. Nat. Rev. Endocrinol. 2013, 9, 13–27. [Google Scholar] [CrossRef]
- Stothard, K.J.; Tennant, P.W.G.; Bell, R.; Rankin, J. Maternal Overweight and Obesity and the Risk of Congenital Anomalies: A Systematic Review and Meta-Analysis. JAMA 2009, 301, 636–650. [Google Scholar] [CrossRef]
- Correa, A.; Gilboa, S.M.; Besser, L.M.; Botto, L.D.; Moore, C.A.; Hobbs, C.A.; Cleves, M.A.; Riehle-Colarusso, T.J.; Waller, D.K.; Reece, E.A. Diabetes Mellitus and Birth Defects. Am. J. Obstet. Gynecol. 2008, 199, 237.e1–237.e9. [Google Scholar] [CrossRef]
- King, J.C. Maternal Obesity, Metabolism, and Pregnancy Outcomes. Annu. Rev. Nutr. 2006, 26, 271–291. [Google Scholar] [CrossRef] [PubMed]
- Blanco, R.; Colombo, A.; Suazo, J. Maternal Obesity Is a Risk Factor for Orofacial Clefts: A Meta-Analysis. Br. J. Oral Maxillofac. Surg. 2015, 53, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Izedonmwen, O.M.; Cunningham, C.; Macfarlane, T.V. What Is the Risk of Having Offspring with Cleft Lip/Palate in Pre-Maternal Obese/Overweight Women When Compared to Pre-Maternal Normal Weight Women? A Systematic Review and Meta-Analysis. J. Oral Maxillofac. Res. 2015, 6, e1. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Yoshioka, E.; Saijo, Y.; Miyamoto, T.; Sengoku, K.; Azuma, H.; Tanahashi, Y.; Ito, Y.; Kobayashi, S.; Minatoya, M.; et al. Population Attributable Fractions of Modifiable Risk Factors for Nonsyndromic Orofacial Clefts: A Prospective Cohort Study From the Japan Environment and Children’s Study. J. Epidemiol. 2021, 31, 272–279. [Google Scholar] [CrossRef]
- Waller, D.K.; Shaw, G.M.; Rasmussen, S.A.; Hobbs, C.A.; Canfield, M.A.; Siega-Riz, A.-M.; Gallaway, M.S.; Correa, A.; National Birth Defects Prevention Study. Prepregnancy Obesity as a Risk Factor for Structural Birth Defects. Arch. Pediatr. Adolesc. Med. 2007, 161, 745–750. [Google Scholar] [CrossRef]
- Rankin, J.; Tennant, P.W.G.; Stothard, K.J.; Bythell, M.; Summerbell, C.D.; Bell, R. Maternal Body Mass Index and Congenital Anomaly Risk: A Cohort Study. Int. J. Obes. 2010, 34, 1371–1380. [Google Scholar] [CrossRef] [PubMed]
- Wehby, G.L.; Uribe, L.M.M.; Wilcox, A.J.; Christensen, K.; Romitti, P.A.; Munger, R.G.; Lie, R.T. Interaction between Smoking and Body Mass Index and Risk of Oral Clefts. Ann. Epidemiol. 2017, 27, 103–107.e2. [Google Scholar] [CrossRef] [PubMed]
- Godschalk, R.W.L.; Feldker, D.E.M.; Borm, P.J.A.; Wouters, E.F.M.; van Schooten, F.-J. Body Mass Index Modulates Aromatic DNA Adduct Levels and Their Persistence in Smokers. Cancer Epidemiol. Biomarkers Prev. 2002, 11, 790–793. [Google Scholar]
- Ács, L.; Bányai, D.; Nemes, B.; Nagy, K.; Ács, N.; Bánhidy, F.; Rózsa, N. Maternal-Related Factors in the Origin of Isolated Cleft Palate-A Population-Based Case-Control Study. Orthod. Craniofac. Res. 2020, 23, 174–180. [Google Scholar] [CrossRef]
- McKinney, C.M.; Chowchuen, B.; Pitiphat, W.; DeRouen, T.; Pisek, A.; Godfrey, K. Micronutrients and Oral Clefts: A Case-Control Study. J. Dent. Res. 2013, 92, 1089–1094. [Google Scholar] [CrossRef]
- Ni, W.; Tian, T.; Zhang, L.; Li, Z.; Wang, L.; Ren, A. Maternal Periconceptional Consumption of Sprouted Potato and Risks of Neural Tube Defects and Orofacial Clefts. Nutr. J. 2018, 17, 112. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, S.L.; Shaw, G.M.; Yang, W.; Abrams, B.; Lammer, E.J. Maternal Stressful Life Events and Risks of Birth Defects. Epidemiology 2007, 18, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Rowland, J.M.; Hendrickx, A.G. Corticosteroid Teratogenicity. Adv. Vet. Sci. Comp. Med. 1983, 27, 99–128. [Google Scholar] [PubMed]
- Fowden, A.L.; Sibley, C.; Reik, W.; Constancia, M. Imprinted Genes, Placental Development and Fetal Growth. Horm. Res. 2006, 65 (Suppl. S3), 50–58. [Google Scholar] [CrossRef] [PubMed]
- Ingstrup, K.G.; Liang, H.; Olsen, J.; Nohr, E.A.; Bech, B.H.; Wu, C.S.; Christensen, K.; Li, J. Maternal Bereavement in the Antenatal Period and Oral Cleft in the Offspring. Hum. Reprod. 2013, 28, 1092–1099. [Google Scholar] [CrossRef] [PubMed]
- Andrews, R.C.; Walker, B.R. Glucocorticoids and Insulin Resistance: Old Hormones, New Targets. Clin. Sci. 1999, 96, 513–523. [Google Scholar] [CrossRef]
- Yoneda, T.; Pratt, R.M. Vitamin B6 Reduces Cortisone-Induced Cleft Palate in the Mouse. Teratology 1982, 26, 255–258. [Google Scholar] [CrossRef] [PubMed]
- Istvan, J. Stress, Anxiety, and Birth Outcomes: A Critical Review of the Evidence. Psychol. Bull. 1986, 100, 331–348. [Google Scholar] [CrossRef]
- Sulik, K.K.; Cook, C.S.; Webster, W.S. Teratogens and Craniofacial Malformations: Relationships to Cell Death. Development 1988, 103 (Suppl. S1), 213–231. [Google Scholar] [CrossRef]
- Sakurai, K.; Nishi, A.; Kondo, K.; Yanagida, K.; Kawakami, N. Screening Performance of K6/K10 and Other Screening Instruments for Mood and Anxiety Disorders in Japan. Psychiatry Clin. Neurosci. 2011, 65, 434–441. [Google Scholar] [CrossRef]
- Furukawa, T.A.; Kawakami, N.; Saitoh, M.; Ono, Y.; Nakane, Y.; Nakamura, Y.; Tachimori, H.; Iwata, N.; Uda, H.; Nakane, H.; et al. The Performance of the Japanese Version of the K6 and K10 in the World Mental Health Survey Japan. Int. J. Methods Psychiatr. Res. 2008, 17, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Agopian, A.J.; Kim, J.; Langlois, P.H.; Lee, L.; Whitehead, L.W.; Symanski, E.; Herdt, M.L.; Delclos, G.L. Maternal Occupational Physical Activity and Risk for Orofacial Clefts. Am. J. Ind. Med. 2017, 60, 627–634. [Google Scholar] [CrossRef] [PubMed]
- Guariguata, L.; Whiting, D.R.; Hambleton, I.; Beagley, J.; Linnenkamp, U.; Shaw, J.E. Global Estimates of Diabetes Prevalence for 2013 and Projections for 2035. Diabetes Res. Clin. Pract. 2014, 103, 137–149. [Google Scholar] [CrossRef]
- Hurst, J.A.; Houlston, R.S.; Roberts, A.; Gould, S.J.; Tingey, W.G. Transverse Limb Deficiency, Facial Clefting and Hypoxic Renal Damage: An Association with Treatment of Maternal Hypertension? Clin. Dysmorphol. 1995, 4, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Hemingway, H.; McCallum, A.; Shipley, M.; Manderbacka, K.; Martikainen, P.; Keskimäki, I. Incidence and Prognostic Implications of Stable Angina Pectoris among Women and Men. JAMA 2006, 295, 1404–1411. [Google Scholar] [CrossRef]
- James, A.H.; Jamison, M.G.; Biswas, M.S.; Brancazio, L.R.; Swamy, G.K.; Myers, E.R. Acute Myocardial Infarction in Pregnancy: A United States Population-Based Study. Circulation 2006, 113, 1564–1571. [Google Scholar] [CrossRef] [PubMed]
- Métneki, J.; Puhó, E.; Czeizel, A.E. Maternal Diseases and Isolated Orofacial Clefts in Hungary. Birth Defects Res. A Clin. Mol. Teratol. 2005, 73, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Van den Boogaard, M.-J.H.; de Costa, D.; Krapels, I.P.C.; Liu, F.; van Duijn, C.; Sinke, R.J.; Lindhout, D.; Steegers-Theunissen, R.P.M. The MSX1 Allele 4 Homozygous Child Exposed to Smoking at Periconception Is Most Sensitive in Developing Nonsyndromic Orofacial Clefts. Hum. Genet. 2008, 124, 525–534. [Google Scholar] [CrossRef]
- Nygård, O.; Nordrehaug, J.E.; Refsum, H.; Ueland, P.M.; Farstad, M.; Vollset, S.E. Plasma Homocysteine Levels and Mortality in Patients with Coronary Artery Disease. N. Engl. J. Med. 1997, 337, 230–236. [Google Scholar] [CrossRef]
- Cleophas, T.J.; Hornstra, N.; van Hoogstraten, B.; van der Meulen, J. Homocysteine, a Risk Factor for Coronary Artery Disease or Not? A Meta-Analysis. Am. J. Cardiol. 2000, 86, 1005–1009. [Google Scholar] [CrossRef]
- Mills, J.L.; Kirke, P.N.; Molloy, A.M.; Burke, H.; Conley, M.R.; Lee, Y.J.; Mayne, P.D.; Weir, D.G.; Scott, J.M. Methylenetetrahydrofolate Reductase Thermolabile Variant and Oral Clefts. Am. J. Med. Genet. 1999, 86, 71–74. [Google Scholar] [CrossRef]
- Mostowska, A.; Hozyasz, K.K.; Wojcicka, K.; Lianeri, M.; Jagodzinski, P.P. Polymorphisms of Stress-Related Genes and the Risk of Nonsyndromic Cleft Lip with or without Cleft Palate. Birth Defects Res. A Clin. Mol. Teratol. 2011, 91, 948–955. [Google Scholar] [CrossRef] [PubMed]
- Czeizel, A.E.; Puhó, E.H.; Acs, N.; Bánhidy, F. High Fever-Related Maternal Diseases as Possible Causes of Multiple Congenital Abnormalities: A Population-Based Case-Control Study. Birth Defects Res. A Clin. Mol. Teratol. 2007, 79, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Carinci, F.; Palmieri, A.; Scapoli, L.; Cura, F.; Abenavoli, F.; Giannì, A.B.; Russillo, A.; Docimo, R.; Martinelli, M. Association between Oral Cleft and Transcobalamin 2 Polymorphism in a Sample Study from Nassiriya, Iraq. Int. J. Immunopathol. Pharmacol. 2019, 33, 205873841985557. [Google Scholar] [CrossRef] [PubMed]
- Guieu, R.; Ruf, J.; Mottola, G. Hyperhomocysteinemia and Cardiovascular Diseases. Ann. Biol. Clin. 2022, 80, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Komiyama, Y.; Koshiji, C.; Yoshida, W.; Natsume, N.; Kawamata, H. 5,10-Methylenetetrahydrofolate Reductase (MTHFR) C677T/A1298C Polymorphisms in Patients with Nonsyndromic Cleft Lip Palate. Biomed. Rep. 2020, 13, 1. [Google Scholar] [CrossRef]
- Rai, V. Strong Association of C677T Polymorphism of Methylenetetrahydrofolate Reductase Gene with Nosyndromic Cleft Lip/Palate (NsCL/P). Ind. J. Clin. Biochem. 2018, 33, 5–15. [Google Scholar] [CrossRef]
- Jahanbin, A.; Hasanzadeh, N.; Abdolhoseinpour, F.; Sadr-Nabavi, A.; Raisolsadat, M.-A.; Shamsian, K.; Mohajertehran, F.; Kianifar, H. Analysis of MTHFR Gene C.677C>T and C.1298A>C Polymorphisms in Iranian Patients with Non-Syndromic Cleft Lip and Palate. Iran. J. Public Health 2014, 43, 821–827. [Google Scholar]
- Machado, R.A.; Martelli-Junior, H.; de Reis, S.R.A.; Küchler, E.C.; Scariot, R.; das Neves, L.T.; Coletta, R.D. Identification of Novel Variants in Cleft Palate-Associated Genes in Brazilian Patients with Non-Syndromic Cleft Palate Only. Front. Cell Dev. Biol. 2021, 9, 638522. [Google Scholar] [CrossRef]
- Martinelli, M.; Girardi, A.; Cura, F.; Nouri, N.; Pinto, V.; Carinci, F.; Morselli, P.G.; Salehi, M.; Scapoli, L. Non-Syndromic Cleft Lip with or without Cleft Palate in Asian Populations: Association Analysis on Three Gene Polymorphisms of the Folate Pathway. Arch. Oral Biol. 2016, 61, 79–82. [Google Scholar] [CrossRef]
- Martinelli, M.; Scapoli, L.; Palmieri, A.; Pezzetti, F.; Baciliero, U.; Padula, E.; Carinci, P.; Giovanni Morselli, P.; Carinci, F. Study of Four Genes Belonging to the Folate Pathway: Transcobalamin 2 Is Involved in the Onset of Non-Syndromic Cleft Lip with or without Cleft Palate. Hum. Mutat. 2006, 27, 294. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, M.; Masiero, E.; Carinci, F.; Morselli, P.G.; Pezzetti, F.; Scapoli, L. New Evidence for the Role of Cystathionine Beta-Synthase in Non-Syndromic Cleft Lip with or without Cleft Palate: CBS Role in CL/P Onset. Eur. J. Oral Sci. 2011, 119, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Nagaoka, R.; Okuhara, S.; Sato, Y.; Amagasa, T.; Iseki, S. Effects of Embryonic Hypoxia on Lip Formation. Birth Defects Res. A Clin. Mol. Teratol. 2012, 94, 215–222. [Google Scholar] [CrossRef]
- Koenig, D.; Spreux, A.; Hiéronimus, S.; Chichmanian, R.-M.; Bastiani, F.; Fénichel, P.; Brucker-Davis, F. Birth Defects Observed with Maternal Carbimazole Treatment: Six Cases Reported to Nice’s Pharmacovigilance Center. Ann. Endocrinol. 2010, 71, 535–542. [Google Scholar] [CrossRef]
- Thomas, S.V.; Jose, M.; Divakaran, S.; Sankara Sarma, P. Malformation Risk of Antiepileptic Drug Exposure during Pregnancy in Women with Epilepsy: Results from a Pregnancy Registry in South India. Epilepsia 2017, 58, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Bánhidy, F.; Puhó, E.H.; Czeizel, A.E. Efficacy of Medical Care of Epileptic Pregnant Women Based on the Rate of Congenital Abnormalities in Their Offspring. Congenit. Anom. 2011, 51, 34–42. [Google Scholar] [CrossRef][Green Version]
- Castilla-Puentes, R.; Ford, L.; Manera, L.; Kwarta, R.F.; Ascher, S.; Li, Q. Topiramate Monotherapy Use in Women with and without Epilepsy: Pregnancy and Neonatal Outcomes. Epilepsy Res. 2014, 108, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Wainscott, G.; Sullivan, F.M.; Volans, G.N.; Wilkinson, M. The Outcome of Pregnancy in Women Suffering from Migraine. Postgrad. Med. J. 1978, 54, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Bánhidy, F.; Acs, N.; Horváth-Puhó, E.; Czeizel, A.E. Maternal Severe Migraine and Risk of Congenital Limb Deficiencies. Birth Defects Res. A Clin. Mol. Teratol. 2006, 76, 592–601. [Google Scholar] [CrossRef]
- Mines, D.; Tennis, P.; Curkendall, S.M.; Li, D.-K.; Peterson, C.; Andrews, E.B.; Calingaert, B.; Chen, H.; Deshpande, G.; Esposito, D.B.; et al. Topiramate Use in Pregnancy and the Birth Prevalence of Oral Clefts. Pharmacoepidemiol. Drug Saf. 2014, 23, 1017–1025. [Google Scholar] [CrossRef]
- Rockenbauer, M.; Olsen, J.; Czeizel, A.E.; Pedersen, L.; Sørensen, H.T.; EuroMAP Group. Recall Bias in a Case-Control Surveillance System on the Use of Medicine during Pregnancy. Epidemiology 2001, 12, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Proctor-Williams, K.; Louw, B. Cleft Lip and/or Palate in Infants Prenatally Exposed to Opioids. Cleft Palate Craniofac. J. 2022, 59, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Mølgaard-Nielsen, D.; Hviid, A. Maternal Use of Antibiotics and the Risk of Orofacial Clefts: A Nationwide Cohort Study. Pharmacoepidemiol. Drug Saf. 2012, 21, 246–253. [Google Scholar] [CrossRef]
- Van Gelder, M.M.H.J.; van Rooij, I.A.L.M.; Miller, R.K.; Zielhuis, G.A.; de Jong-van den Berg, L.T.W.; Roeleveld, N. Teratogenic Mechanisms of Medical Drugs. Hum. Reprod. Update 2010, 16, 378–394. [Google Scholar] [CrossRef]
- Lin, K.J.; Mitchell, A.A.; Yau, W.-P.; Louik, C.; Hernández-Díaz, S. Maternal Exposure to Amoxicillin and the Risk of Oral Clefts. Epidemiology 2012, 23, 699–705. [Google Scholar] [CrossRef] [PubMed]
- Skuladottir, H.; Wilcox, A.; McConnaughey, R.; Vindenes, H.; Lie, R.T. First-Trimester Nonsystemic Corticosteroid Use and the Risk of Oral Clefts in Norway. Ann. Epidemiol. 2014, 24, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Skuladottir, H.; Wilcox, A.J.; Ma, C.; Lammer, E.J.; Rasmussen, S.A.; Werler, M.M.; Shaw, G.M.; Carmichael, S.L. Corticosteroid Use and Risk of Orofacial Clefts. Birth Defects Res. Part A Clin. Mol. Teratol. 2014, 100, 499–506. [Google Scholar] [CrossRef]
- Huybrechts, K.F.; Hernández-Díaz, S.; Straub, L.; Gray, K.J.; Zhu, Y.; Patorno, E.; Desai, R.J.; Mogun, H.; Bateman, B.T. Association of Maternal First-Trimester Ondansetron Use With Cardiac Malformations and Oral Clefts in Offspring. JAMA 2018, 320, 2429. [Google Scholar] [CrossRef]
- Hernandez-Diaz, S.; Huybrechts, K.F.; Desai, R.J.; Cohen, J.M.; Mogun, H.; Pennell, P.B.; Bateman, B.T.; Patorno, E. Topiramate Use Early in Pregnancy and the Risk of Oral Clefts: A Pregnancy Cohort Study. Neurology 2018, 90, e342–e351. [Google Scholar] [CrossRef]
- Mullens, C.L.; McCulloch, I.L.; Hardy, K.M.; Mathews, R.E.; Mason, A.C. Associations between Orofacial Clefting and Neonatal Abstinence Syndrome. Plast. Reconstr. Surg. Glob. Open 2019, 7, e2095. [Google Scholar] [CrossRef]
- Auslander, A.; McKean-Cowdin, R.; Brindopke, F.; Sylvester, B.; DiBona, M.; Magee, K.; Kapoor, R.; Conti, D.V.; Rakotoarison, S.; Magee, W. The Role of Smoke from Cooking Indoors over an Open Flame and Parental Smoking on the Risk of Cleft Lip and Palate: A Case- Control Study in 7 Low-Resource Countries. J. Glob. Health 2020, 10, 020410. [Google Scholar] [CrossRef] [PubMed]
- Romitti, P.A.; Lidral, A.C.; Munger, R.G.; Daack-Hirsch, S.; Burns, T.L.; Murray, J.C. Candidate Genes for Nonsyndromic Cleft Lip and Palate and Maternal Cigarette Smoking and Alcohol Consumption: Evaluation of Genotype-Environment Interactions from a Population-Based Case-Control Study of Orofacial Clefts. Teratology 1999, 59, 39–50. [Google Scholar] [CrossRef]
- Shaw, G.M.; Wasserman, C.R.; Lammer, E.J.; O’Malley, C.D.; Murray, J.C.; Basart, A.M.; Tolarova, M.M. Orofacial Clefts, Parental Cigarette Smoking, and Transforming Growth Factor-Alpha Gene Variants. Am. J. Hum. Genet. 1996, 58, 551–561. [Google Scholar] [PubMed]
- Krapels, I.; van Rooij, I.; Ocke, M.; West, C.; Horst, C.; Steegers-Theunissen, R. Maternal Nutritional Status and the Risk for Orofacial Cleft Offspring in Humans. J. Nutr. 2004, 134, 3106–3113. [Google Scholar] [CrossRef] [PubMed]
- DeRoo, L.A.; Wilcox, A.J.; Lie, R.T.; Romitti, P.A.; Pedersen, D.A.; Munger, R.G.; Moreno Uribe, L.M.; Wehby, G.L. Maternal Alcohol Binge-Drinking in the First Trimester and the Risk of Orofacial Clefts in Offspring: A Large Population-Based Pooling Study. Eur. J. Epidemiol. 2016, 31, 1021–1034. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, E.M.; Abel, E.L. Fetal alcohol syndrome: The origins of a moral panic. Alcohol Alcohol. 2000, 35, 276–282. [Google Scholar] [CrossRef]
- Boyles, A.L.; DeRoo, L.A.; Lie, R.T.; Taylor, J.A.; Jugessur, A.; Murray, J.C.; Wilcox, A.J. Maternal Alcohol Consumption, Alcohol Metabolism Genes, and the Risk of Oral Clefts: A Population-Based Case-Control Study in Norway, 1996–2001. Am. J. Epidemiol. 2010, 172, 924–931. [Google Scholar] [CrossRef]
- Hobbs, C.A.; Chowdhury, S.; Cleves, M.A.; Erickson, S.; MacLeod, S.L.; Shaw, G.M.; Shete, S.; Witte, J.S.; Tycko, B. Genetic Epidemiology and Nonsyndromic Structural Birth Defects: From Candidate Genes to Epigenetics. JAMA Pediatr. 2014, 168, 371–377. [Google Scholar] [CrossRef]
- Kaufman, J.A.; Wright, J.M.; Evans, A.; Rivera-Núñez, Z.; Meyer, A.; Narotsky, M.G. Associations Between Disinfection By-Product Exposures and Craniofacial Birth Defects. J. Occup. Environ. Med. 2018, 60, 109–119. [Google Scholar] [CrossRef]
- Suhl, J.; Leonard, S.; Weyer, P.; Rhoads, A.; Siega-Riz, A.M.; Renée Anthony, T.; Burns, T.L.; Conway, K.M.; Langlois, P.H.; Romitti, P.A. Maternal Arsenic Exposure and Nonsyndromic Orofacial Clefts. Birth Defects Res. 2018, 110, 1455–1467. [Google Scholar] [CrossRef] [PubMed]
- Suhl, J.; Romitti, P.A.; Rocheleau, C.; Cao, Y.; Burns, T.L.; Conway, K.; Bell, E.M.; Stewart, P.; Langlois, P.; the National Birth Defects Prevention Study. Parental Occupational Pesticide Exposure and Nonsyndromic Orofacial Clefts. J. Occup. Environ. Hyg. 2018, 15, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, M.; Yoshida, S.; Kawakami, C.; Kawakami, K.; Ito, S. Association of Maternal Heavy Metal Exposure during Pregnancy with Isolated Cleft Lip and Palate in Offspring: Japan Environment and Children’s Study (JECS) Cohort Study. PLoS ONE 2022, 17, e0265648. [Google Scholar] [CrossRef] [PubMed]
- Spinder, N.; Bergman, J.E.H.; Boezen, H.M.; Vermeulen, R.C.H.; Kromhout, H.; de Walle, H.E.K. Maternal Occupational Exposure and Oral Clefts in Offspring. Environ. Health 2017, 16, 83. [Google Scholar] [CrossRef] [PubMed]
- Desrosiers, T.A.; Lawson, C.C.; Meyer, R.E.; Richardson, D.B.; Daniels, J.L.; Waters, M.A.; van Wijngaarden, E.; Langlois, P.H.; Romitti, P.A.; Correa, A.; et al. Maternal Occupational Exposure to Organic Solvents during Early Pregnancy and Risks of Neural Tube Defects and Orofacial Clefts. Occup. Environ. Med. 2012, 69, 493–499. [Google Scholar] [CrossRef] [PubMed]
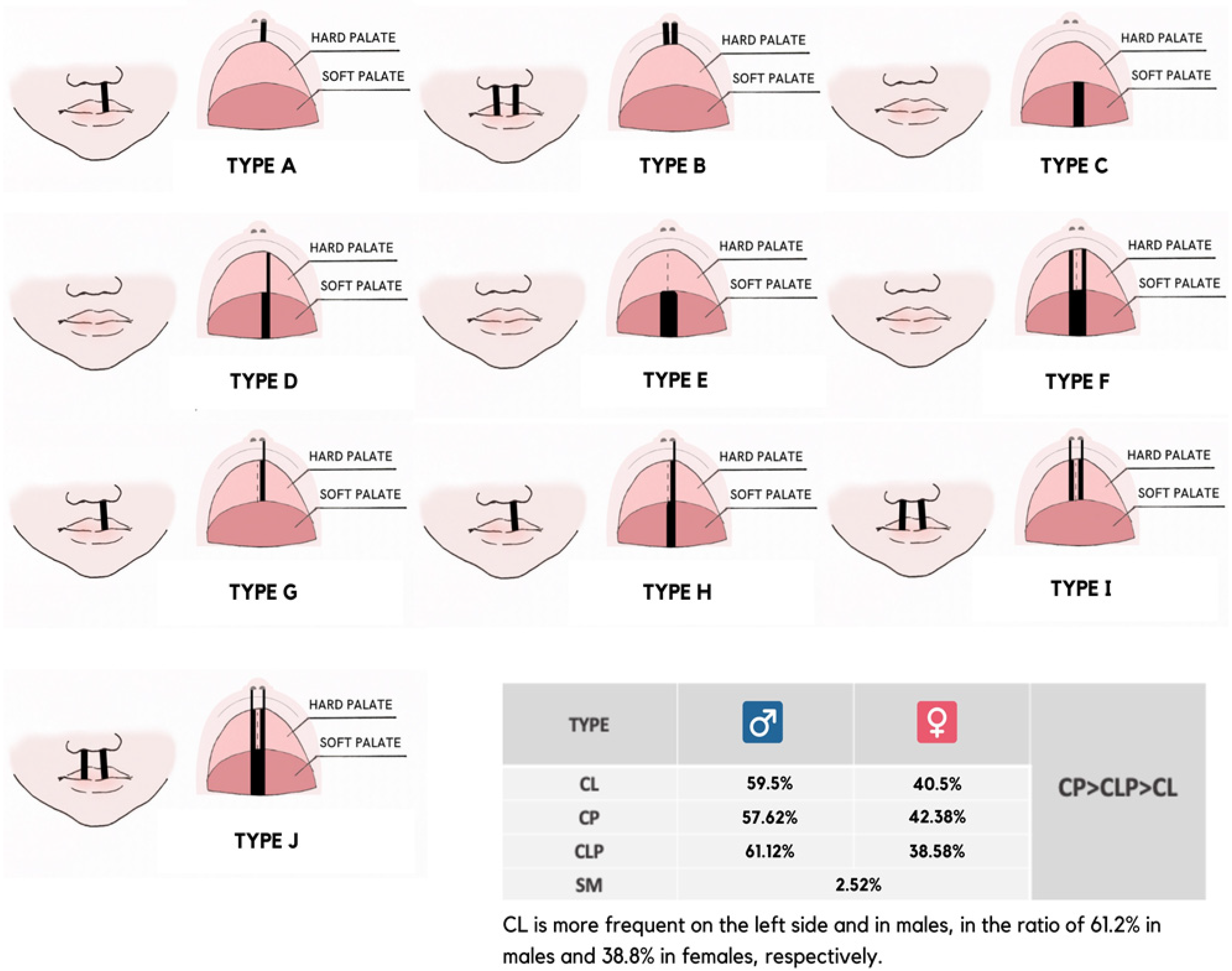


| LAHSAL 1 Abbreviated Notation | Phenotypic Description |
|---|---|
| [LAHS•••] | right unilateral complete CL, complete alveolus and complete “unilateral” CP |
| [laHS•••] | right unilateral incomplete CL and alveolus, complete “unilateral” CP |
| [•••SHal] | left unilateral incomplete CL and alveolus, with complete “unilateral” CP |
| [LAHSHAL] | bilateral symmetric complete CL, complete CA, and complete “bilateral” CP |
| [l*HSH*L] | bilateral symmetric incomplete CL, notched CA, and complete “bilateral” CP |
| [••HSH••] | complete “midline” cleft of hard and soft palate |
| [•••S•••] | complete “midline” cleft of soft palate |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inchingolo, A.M.; Fatone, M.C.; Malcangi, G.; Avantario, P.; Piras, F.; Patano, A.; Di Pede, C.; Netti, A.; Ciocia, A.M.; De Ruvo, E.; et al. Modifiable Risk Factors of Non-Syndromic Orofacial Clefts: A Systematic Review. Children 2022, 9, 1846. https://doi.org/10.3390/children9121846
Inchingolo AM, Fatone MC, Malcangi G, Avantario P, Piras F, Patano A, Di Pede C, Netti A, Ciocia AM, De Ruvo E, et al. Modifiable Risk Factors of Non-Syndromic Orofacial Clefts: A Systematic Review. Children. 2022; 9(12):1846. https://doi.org/10.3390/children9121846
Chicago/Turabian StyleInchingolo, Angelo Michele, Maria Celeste Fatone, Giuseppina Malcangi, Pasquale Avantario, Fabio Piras, Assunta Patano, Chiara Di Pede, Anna Netti, Anna Maria Ciocia, Elisabetta De Ruvo, and et al. 2022. "Modifiable Risk Factors of Non-Syndromic Orofacial Clefts: A Systematic Review" Children 9, no. 12: 1846. https://doi.org/10.3390/children9121846
APA StyleInchingolo, A. M., Fatone, M. C., Malcangi, G., Avantario, P., Piras, F., Patano, A., Di Pede, C., Netti, A., Ciocia, A. M., De Ruvo, E., Viapiano, F., Palmieri, G., Campanelli, M., Mancini, A., Settanni, V., Carpentiere, V., Marinelli, G., Latini, G., Rapone, B., ... Dipalma, G. (2022). Modifiable Risk Factors of Non-Syndromic Orofacial Clefts: A Systematic Review. Children, 9(12), 1846. https://doi.org/10.3390/children9121846




















