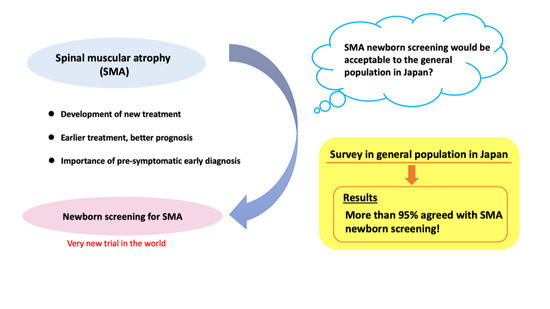
Views of the General Population on Newborn Screening for Spinal Muscular Atrophy in Japan
Abstract
:1. Introduction
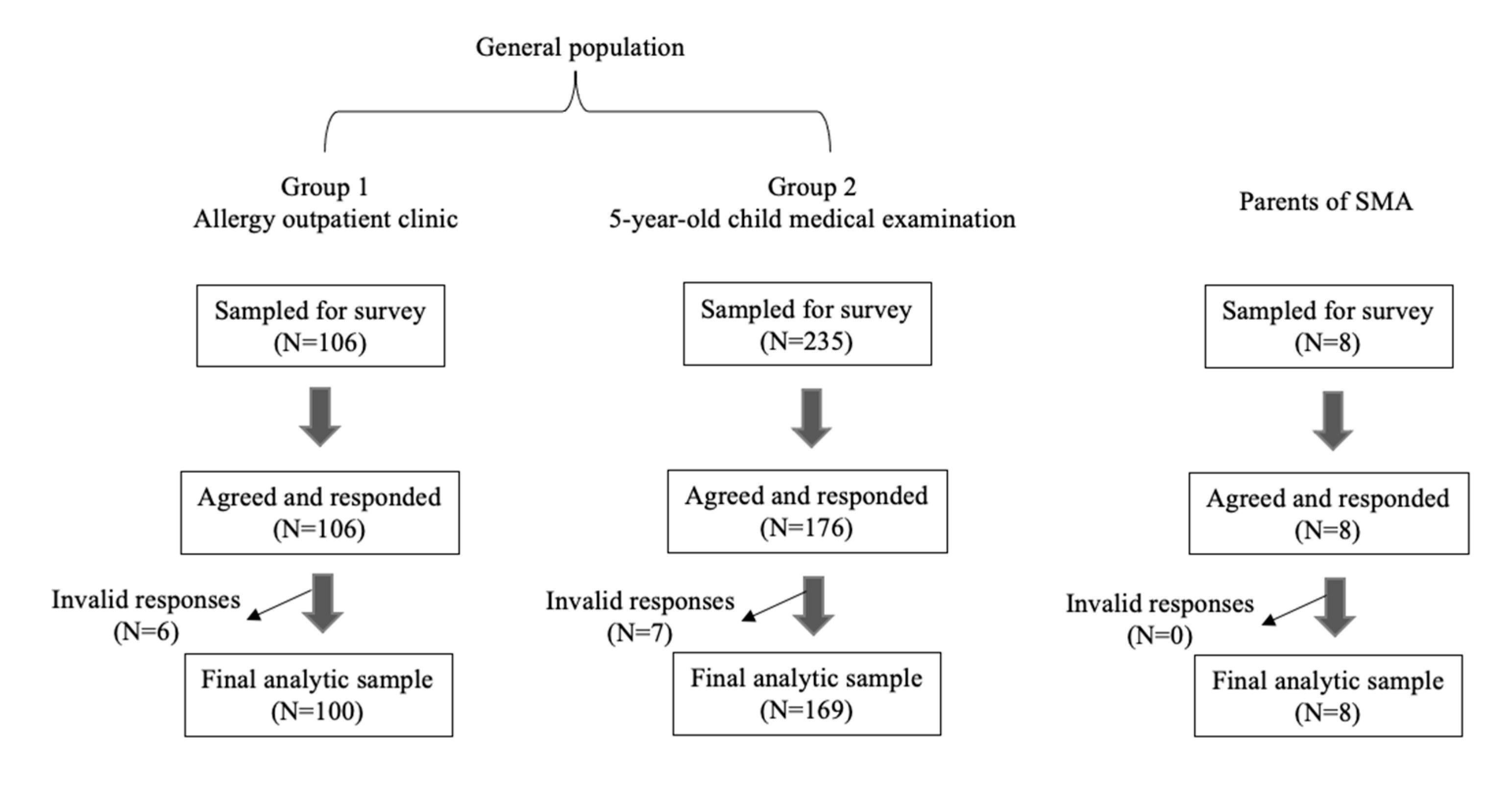
2. Materials and Methods
2.1. Survey Design
2.2. Statical Analysis
2.3. Ethics
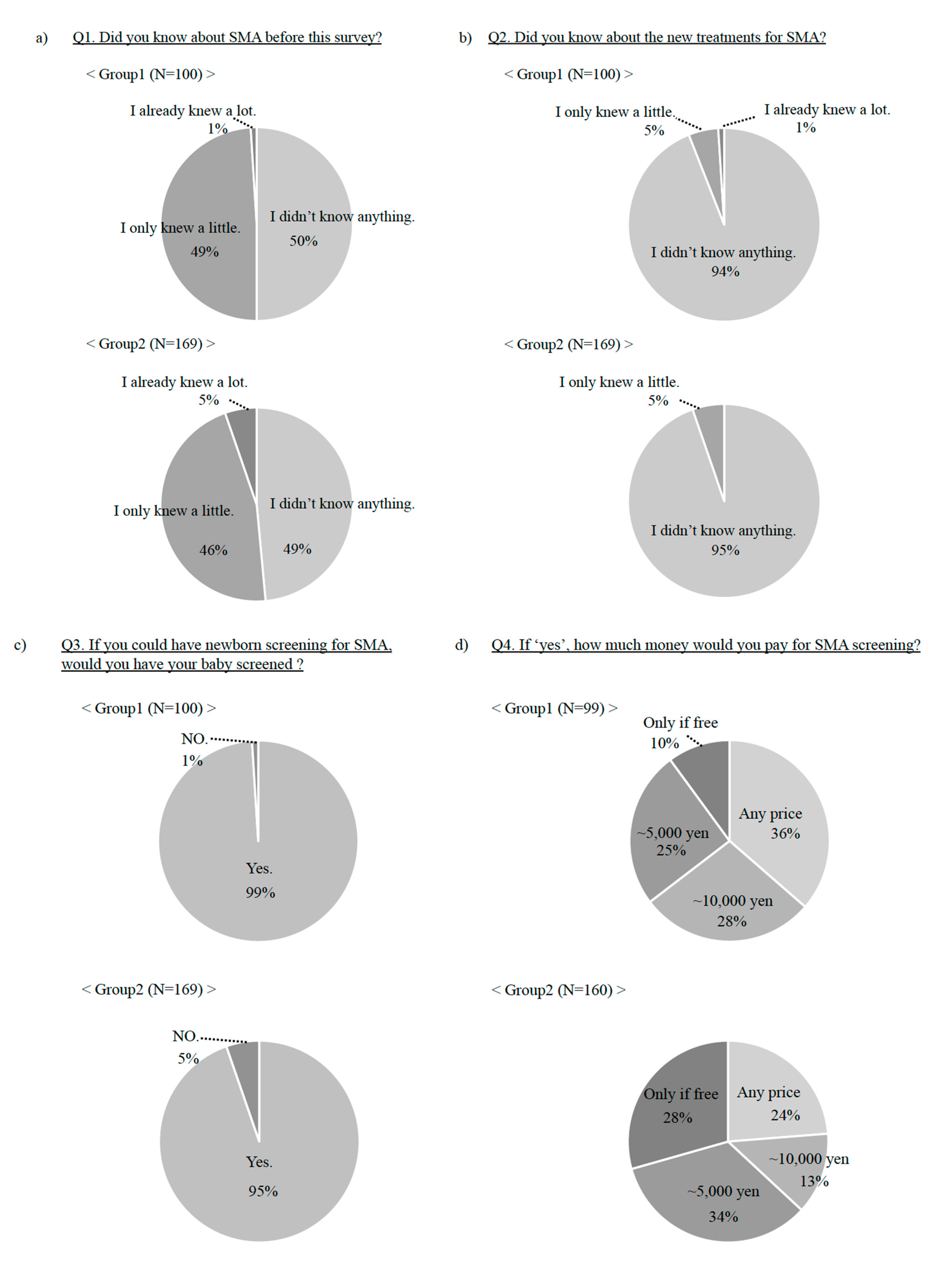
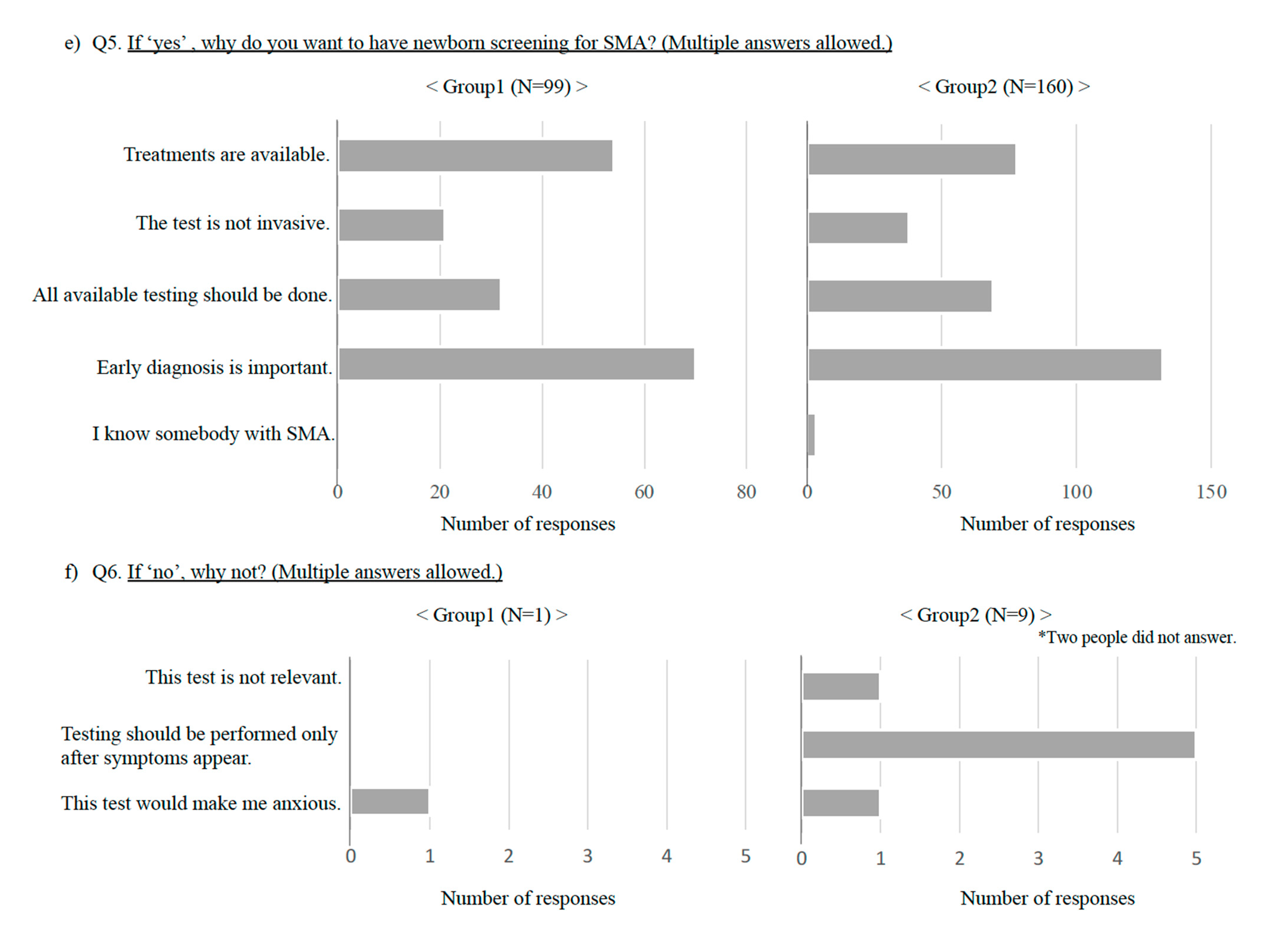
3. Results
3.1. Results in General Population
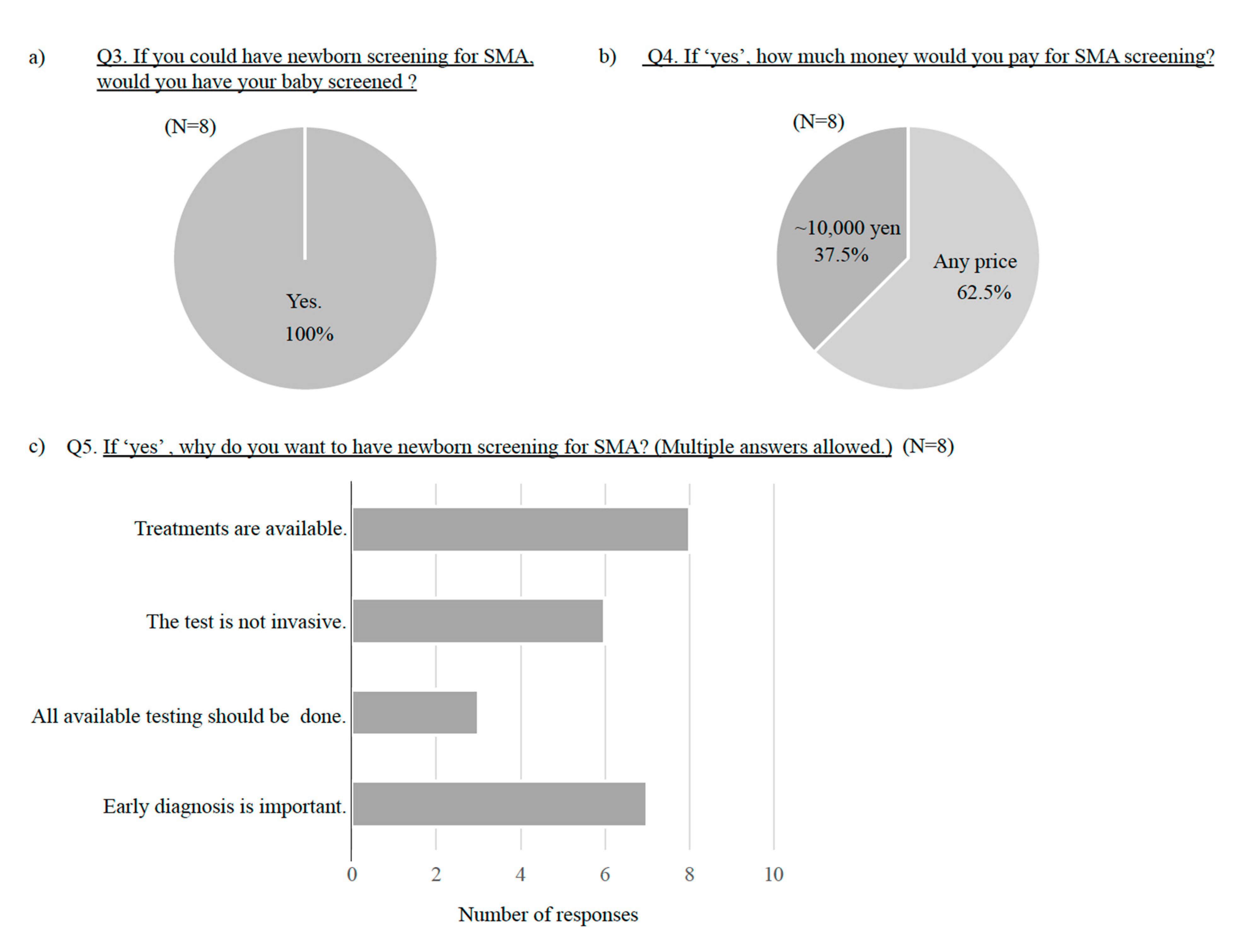
3.2. Results in Parents of SMA Patients
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Verhaart, I.E.C.; Robertson, A.; Wilson, I.J.; Aartsma-Rus, A.; Cameron, S.; Jones, C.C.; Cook, S.F.; Lochmüller, H. Prevalence, Incidence and Carrier Frequency of 5q-Linked Spinal Muscular Atrophy-a Literature Review. Orphanet J. Rare Dis. 2017, 12, 124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lefebvre, S.; Bürglen, L.; Reboullet, S.; Clermont, O.; Burlet, P.; Viollet, L.; Benichou, B.; Cruaud, C.; Millasseau, P.; Zeviani, M. Identification and characterization of a spinal muscular atrophy-determining gene. Cell 1995, 80, 155–165. [Google Scholar] [CrossRef] [Green Version]
- Harada, Y.; Sutomo, R.; Sadewa, A.H.; Akutsu, T.; Takeshima, Y.; Wada, H.; Matsuo, M.; Nishio, H. Correlation between SMN2 copy number and clinical phenotype of spinal muscular atrophy: Three SMN2 copies fail to rescue some patients from the disease severity. J. Neurol. 2002, 249, 1211–1219. [Google Scholar] [CrossRef] [PubMed]
- Ojala, K.S.; Reedich, E.J.; DiDonato, C.J.; Meriney, S.D. In search of a cure: The development of therapeutics to alter the progression of spinal muscular atrophy. Brain Sci. 2021, 11, 194. [Google Scholar] [CrossRef] [PubMed]
- Munsat, T.L.; Davies, K.E. International SMA consortium meeting (26–28 June 1992, Bonn, Germany). Neuromuscul. Disord. 1992, 2, 423–428. [Google Scholar] [CrossRef]
- Farrar, M.A.; Vucic, S.; Johnston, H.M.; du Sart, D.; Kiernan, M.C. Pathophysiologocal insights derived by natural history and motor function of spinal muscular atrophy. J Pediatr. 2013, 162, 155–159. [Google Scholar] [CrossRef]
- Arnold, W.D.; Kassar, D.; Kissel, J.T. Spinal muscular atrophy: Diagnosis and management in a new therapeutic era. Muscle Nerve 2015, 51, 157–167. [Google Scholar] [CrossRef]
- SMA News Today. Zolgensma. Available online: https://smanewstoday.com/zolgensma/ (accessed on 20 June 2021).
- SMA News Today.Evrysdi (Risdiplam). Available online: https://smanewstoday.com/evrysdi-risdiplam/ (accessed on 20 June 2021).
- Mercuri, E.; Finkel, R.S.; Muntoni, F.; Wirth, B.; Montes, J.; Main, M.; Mazzone, E.S.; Vitale, M.; Snyder, B.; Quijano-Roy, S.; et al. Diagnosis and management of spinal muscular atrophy: Part 1: Recommendations for diagnosis, rehabilitation, orthopedic and nutritional care. Neuromuscul. Disord. 2018, 28, 103–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finkel, R.S.; Mercuri, E.; Meyer, O.H.; Simonds, A.K.; Schroth, M.K.; Graham, R.J.; Kirschner, J.; Iannaccone, S.T.; Crawford, T.O.; Sejersen, T.; et al. Diagnosis and management of spinal muscular atrophy: Part 2: Pulmonary and acute care; medications, supplements and immunizations; other organ systems; and ethics. Neuromuscul. Disord. 2018, 28, 197–207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finkel, R.S.; Mercuri, E.; Darras, B.T.; Connolly, A.M.; Kuntz, N.L.; Kirschner, J.; Chiriboga, C.A.; Saito, K.; Servais, L.; Tizzano, E.; et al. Nusinersen versus sham control in infantile-onset spinal muscular atrophy. N. Engl. J. Med. 2017, 377, 1723–1732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mendell, J.R.; Al-Zaidy, S.; Shell, R.; Arnold, W.D.; Rodino-Klapac, L.R.; Prior, T.W.; Lowes, L.; Alfano, L.; Berry, K.; Church, K.; et al. Single-dose gene-replacement therapy for spinal muscular atrophy. N. Engl. J. Med. 2017, 377, 1713–1722. [Google Scholar] [CrossRef]
- De Vivo, D.C.; Bertini, E.; Swoboda, K.J.; Hwu, W.L.; Crawford, T.O.; Finkel, R.S.; Kirschner, J.; Kuntz, N.L.; Parsons, J.A.; Ryan, M.M.; et al. Nusinersen was initiated in infants during the presymptomatic stage of spinal muscular atrophy: Interim efficacy and safety results from the Phase 2 NURTURE study. Neuromuscul. Disord. 2019, 29, 842–856. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kolb, S.J.; Coffey, C.S.; Yankey, J.W.; Krosschell, K.; Arnold, W.D.; Rutkove, S.B.; Swoboda, K.J.; Reyna, S.P.; Sakonju, A.; Darras, B.T.; et al. Natural history of infantile-inset spinal muscular atrophy. Ann. Neurol. 2017, 82, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Jędrzejowska, M. Advances in newborn screening and presymptomatic diagnosis of spinal muscular atrophy. Degener. Neurol. Neuromuscul. Dis. 2020, 10, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.H.; Chiang, S.C.; Weng, W.C.; Lee, N.C.; Lin, C.J.; Hsieh, W.S.; Lee, W.T.; Jong, Y.J.; Ko, T.M.; Hwu, W.L. Presymptomatic diagnosis of spinal muscular atrophy through newborn screening. J. Pediatr. 2017, 190, 124–129. [Google Scholar] [CrossRef]
- Kraszewski, J.N.; Kay, D.M.; Stevens, C.F.; Koval, C.; Haser, B.; Ortiz, V.; Albertorio, A.; Cohen, L.L.; Jain, R.; Andrew, S.P.; et al. Pilot study of population-based newborn screening for spinal muscular atrophy in New York state. Genet. Med. 2018, 20, 608–613. [Google Scholar] [CrossRef] [Green Version]
- Vill, K.; Kölbel, H.; Schwartz, O.; Blaschek, A.; Olgemöller, B.; Harms, E.; Burggraf, S.; Röschinger, W.; Durner, J.; Gläser, D.; et al. One year of newborn screening for SMA–results of a German pilot project. J. Neuromuscul. Dis. 2019, 6, 503–515. [Google Scholar] [CrossRef] [Green Version]
- Kariyawasam, D.S.T.; Russell, J.S.; Wiley, V.; Alexander, I.E.; Farrar, M.A. Implementation of newborn screening for spinal muscular atrophy: Australian experience. Genet. Med. 2020, 22, 557–565. [Google Scholar] [CrossRef]
- Shinohara, M.; Niba, E.T.E.; Wijaya, Y.O.S.; Takayama, I.; Mitsuishi, C.; Kumasaka, S.; Kondo, Y.; Takatera, A.; Hokuto, I.; Morioka, I.; et al. A novel system for spinal muscular atrophy screening in newborns: A Japanese pilot study. Int. J. Neonat. Screen. 2019, 5, 41. [Google Scholar] [CrossRef] [Green Version]
- Boardman, F.K.; Sadler, C.; Young, P. Newborn genetic screening for spinal muscular atrophy in the UK: Views of the general population. J. Mol. Genet. Genomic Med. 2018, 6, 99–108. [Google Scholar] [CrossRef] [Green Version]
- Lin, P.J.; Yeh, W.S.; Neumann, P.J. Willingness to pay for a newborn screening test for spinal muscular atrophy. Pediatr. Neurol. 2017, 66, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Glascock, J.; Sampson, J.; Haidet-Phillips, A.; Connolly, A.; Darras, B.; Day, J.; Finkel, R.; Howell, R.R.; Klinger, K.; Kuntz, N.; et al. Treatment algorithm for infants diagnosed with spinal muscular atrophy through newborn Sscreening. J. Neuromuscul. Dis. 2018, 5, 145–158. [Google Scholar] [CrossRef] [Green Version]
- Glascock, J.; Sampson, J.; Connolly, A.M.; Darras, B.T.; Day, J.W.; Finkel, R.; Howell, R.R.; Klinger, K.W.; Kuntz, N.; Prior, T.; et al. Revised recommendations for the treatment of infants diagnosed with spinal muscular atrophy via newborn screening who have 4 copies of SMN2. J. Neuromuscul. Dis. 2020, 7, 97–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Müller-Felber, W.; Vill, K.; Schwartz, O.; Gläser, D.; Nennstiel, U.; Wirth, B.; Burggraf, S.; Röschinger, W.; Becker, M.; Durner, J.; et al. Infants Diagnosed with Spinal Muscular Atrophy and 4 SMN2 Copies through Newborn Screening-Opportunity or Burden? J. Neuromuscul. Dis. 2020, 7, 109–117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Characteristics | All n = 269 (%) | Group 1: Outpatient Allergy Clinic n = 100 (%) | Group 2: 5-Year-Old Checkup n = 169 (%) | |
|---|---|---|---|---|
| Relationship with the child | Father | 4.8 | 7.0 | 3.6 |
| Mother | 94.4 | 91.0 | 96.4 | |
| Grandparent | 0.4 | 1.0 | 0 | |
| Not specified | 0.4 | 1.0 | 0 | |
| Age (years) | 20–29 | 3.7 | 6.0 | 2.4 |
| 30–39 | 62.5 | 53.0 | 68.0 | |
| 40–49 | 32.0 | 38.0 | 28.4 | |
| Over 50 | 1.9 | 3.0 | 1.2 | |
| Is the respondent close with an SMA patient? | Yes | 0.7 | 2.0 | 0 |
| No | 99.3 | 98.0 | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, T.; Tokunaga, S.; Taniguchi, N.; Fujino, T.; Saito, M.; Shimomura, H.; Takeshima, Y. Views of the General Population on Newborn Screening for Spinal Muscular Atrophy in Japan. Children 2021, 8, 694. https://doi.org/10.3390/children8080694
Lee T, Tokunaga S, Taniguchi N, Fujino T, Saito M, Shimomura H, Takeshima Y. Views of the General Population on Newborn Screening for Spinal Muscular Atrophy in Japan. Children. 2021; 8(8):694. https://doi.org/10.3390/children8080694
Chicago/Turabian StyleLee, Tomoko, Sachi Tokunaga, Naoko Taniguchi, Tetsuro Fujino, Midori Saito, Hideki Shimomura, and Yasuhiro Takeshima. 2021. "Views of the General Population on Newborn Screening for Spinal Muscular Atrophy in Japan" Children 8, no. 8: 694. https://doi.org/10.3390/children8080694
APA StyleLee, T., Tokunaga, S., Taniguchi, N., Fujino, T., Saito, M., Shimomura, H., & Takeshima, Y. (2021). Views of the General Population on Newborn Screening for Spinal Muscular Atrophy in Japan. Children, 8(8), 694. https://doi.org/10.3390/children8080694