Juvenile Idiopathic Arthritis Associated Uveitis
Abstract
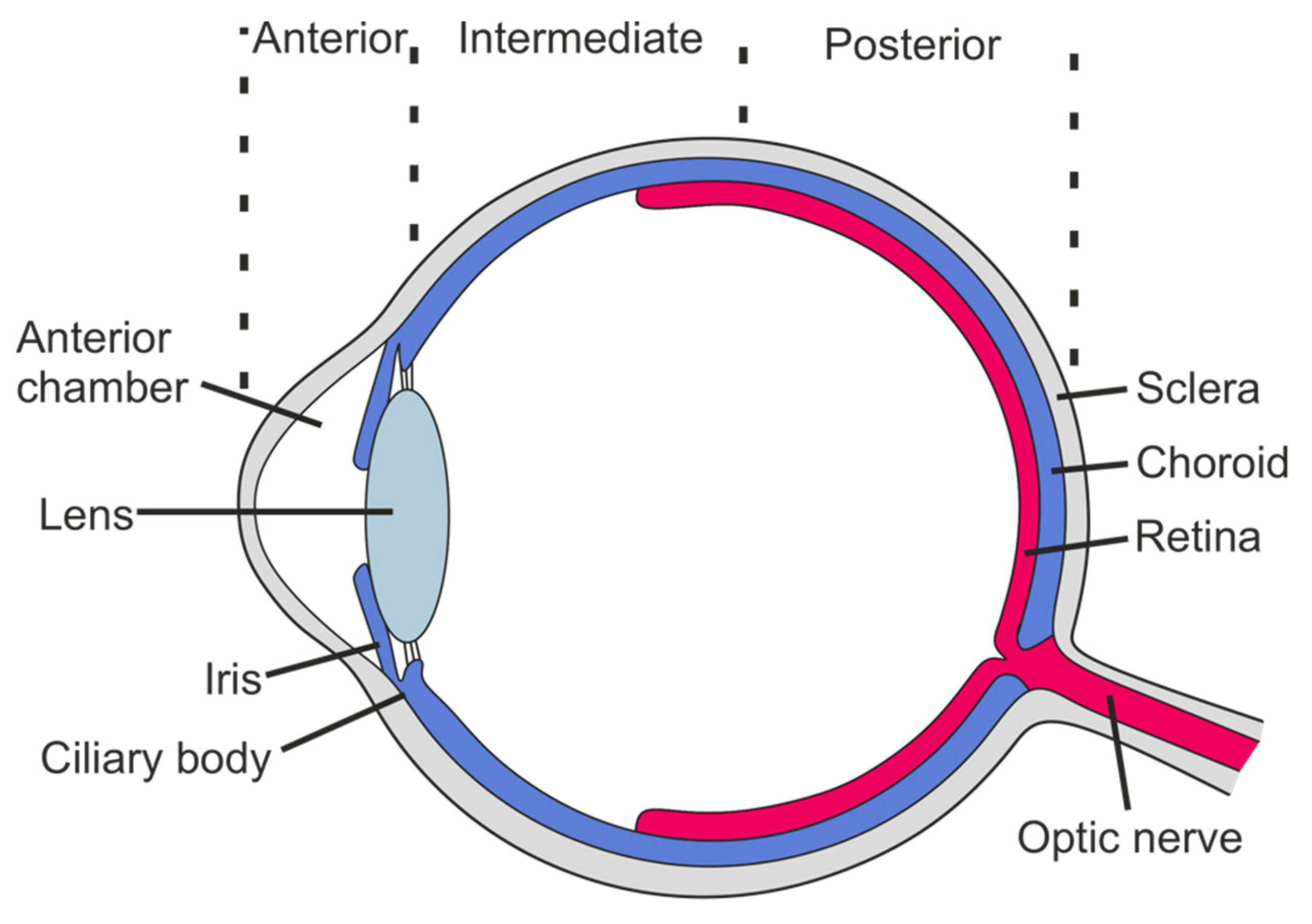
:1. Background
2. Epidemiology
3. Pathophysiology and Systemic Risk Factors
3.1. Demographic Risk Factors
3.2. Immune Cells and Serum Proteins
3.3. Environmental Factors
4. Genetic Factors
4.1. Human Leukocyte Antigen (HLA)
4.2. Non-HLA Genes
5. Laboratory Biomarkers
5.1. Anti-Nuclear Antibodies (ANAs)
5.2. Rheumatoid Factor (RF)
5.3. Erythrocyte Sedimentation Rate (ESR)
5.4. Ocular Proteins
6. Treatment
7. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Search Strategy and Review Criteria
References
- Prakken, B.; Albani, S.; Martini, A. Juvenile idiopathic arthritis. Lancet 2011, 377, 2138–2149. [Google Scholar] [CrossRef] [Green Version]
- Carvounis, P.E.; Herman, D.C.; Cha, S.; Burke, J.P. Incidence and outcomes of uveitis in juvenile rheumatoid arthritis, a synthesis of the literature. Graefes Arch. Clin. Exp. Ophthalmol. 2006, 244, 281–290. [Google Scholar] [CrossRef]
- Ravelli, A.; Martini, A. Juvenile idiopathic arthritis. Lancet 2007, 369, 767–778. [Google Scholar] [CrossRef] [Green Version]
- Ravelli, A.; Felici, E.; Magni-Manzoni, S.; Pistorio, A.; Novarini, C.; Bozzola, E.; Viola, S.; Martini, A. Patients with antinuclear antibody-positive juvenile idiopathic arthritis constitute a homogeneous subgroup irrespective of the course of joint disease. Arthritis Rheum. 2005, 52, 826–832. [Google Scholar] [CrossRef]
- Angeles-Han, S.T.; Pelajo, C.F.; Vogler, L.B.; Rouster-Stevens, K.; Kennedy, C.; Ponder, L.; McCracken, C.; Lopez-Benitez, J.; Drews-Botsch, C.; Prahalad, S.; et al. Risk markers of juvenile idiopathic arthritis-associated uveitis in the Childhood Arthritis and Rheumatology Research Alliance (CARRA) Registry. J. Rheumatol. 2013, 40, 2088–2096. [Google Scholar] [CrossRef] [Green Version]
- Berthold, E.; Mansson, B.; Kahn, R. Outcome in juvenile idiopathic arthritis: A population-based study from Sweden. Arthritis Res. Ther. 2019, 21, 218. [Google Scholar] [CrossRef] [Green Version]
- Castagna, I.; Roszkowska, A.M.; Alessandrello, F.; Oliverio, G.W.; Tumminello, G.; Gallizzi, R.; Conti, G.; Aragona, P. Juvenile idiopathic arthritis-associated uveitis: A retrospective analysis from a centre of South Italy. Int. Ophthalmol. 2020, 40, 335–342. [Google Scholar] [CrossRef] [Green Version]
- Cosickic, A.; Halilbasic, M.; Selimovic, A.; Avdagic, H. Uveitis Associated with Juvenile Idiopathic Arthritis, our Observations. Med. Arch. 2017, 71, 52–55. [Google Scholar] [CrossRef] [Green Version]
- Heiligenhaus, A.; Klotsche, J.; Niewerth, M.; Horneff, G.; Ganser, G.; Haas, J.P.; Minden, K. Similarities in clinical course and outcome between juvenile idiopathic arthritis (JIA)-associated and ANA-positive idiopathic anterior uveitis: Data from a population-based nationwide study in Germany. Arthritis Res. Ther. 2020, 22, 81. [Google Scholar] [CrossRef] [Green Version]
- Nordal, E.; Rypdal, V.; Christoffersen, T.; Aalto, K.; Berntson, L.; Fasth, A.; Herlin, T.; Nielsen, S.; Peltoniemi, S.; Straume, B.; et al. Incidence and predictors of Uveitis in juvenile idiopathic arthritis in a Nordic long-term cohort study. Pediatr. Rheumatol. Online J. 2017, 15, 66. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, M.; Zetterberg, M.; Oskarsdottir, S.; Andersson Gronlund, M. Assessment of the outcome of ophthalmological screening for uveitis in a cohort of Swedish children with juvenile idiopathic arthritis. Acta Ophthalmol. 2017, 95, 741–747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sabri, K.; Saurenmann, R.K.; Silverman, E.D.; Levin, A.V. Course, complications, and outcome of juvenile arthritis-related uveitis. J. AAPOS 2008, 12, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Tappeiner, C.; Klotsche, J.; Schenck, S.; Niewerth, M.; Minden, K.; Heiligenhaus, A. Temporal change in prevalence and complications of uveitis associated with juvenile idiopathic arthritis:data from a cross-sectional analysis of a prospective nationwide study. Clin. Exp. Rheumatol. 2015, 33, 936–944. [Google Scholar]
- Tappeiner, C.; Klotsche, J.; Sengler, C.; Niewerth, M.; Liedmann, I.; Walscheid, K.; Lavric, M.; Foell, D.; Minden, K.; Heiligenhaus, A. Risk Factors and Biomarkers for the Occurrence of Uveitis in Juvenile Idiopathic Arthritis: Data From the Inception Cohort of Newly Diagnosed Patients With Juvenile Idiopathic Arthritis Study. Arthritis Rheumatol. 2018, 70, 1685–1694. [Google Scholar] [CrossRef] [PubMed]
- Consolaro, A.; Giancane, G.; Alongi, A.; van Dijkhuizen, E.H.P.; Aggarwal, A.; Al-Mayouf, S.M.; Bovis, F.; De Inocencio, J.; Demirkaya, E.; Flato, B.; et al. Phenotypic variability and disparities in treatment and outcomes of childhood arthritis throughout the world: An observational cohort study. Lancet Child. Adolesc. Health 2019, 3, 255–263. [Google Scholar] [CrossRef]
- Yasumura, J.; Yashiro, M.; Okamoto, N.; Shabana, K.; Umebayashi, H.; Iwata, N.; Okura, Y.; Kubota, T.; Shimizu, M.; Tomiita, M.; et al. Clinical features and characteristics of uveitis associated with juvenile idiopathic arthritis in Japan: First report of the pediatric rheumatology association of Japan (PRAJ). Pediatr. Rheumatol. Online J. 2019, 17, 15. [Google Scholar] [CrossRef]
- Hyrich, K.L.; Lal, S.D.; Foster, H.E.; Thornton, J.; Adib, N.; Baildam, E.; Gardner-Medwin, J.; Wedderburn, L.R.; Chieng, A.; Davidson, J.; et al. Disease activity and disability in children with juvenile idiopathic arthritis one year following presentation to paediatric rheumatology. Results from the Childhood Arthritis Prospective Study. Rheumatology 2010, 49, 116–122. [Google Scholar] [CrossRef] [Green Version]
- Oen, K.; Duffy, C.M.; Tse, S.M.; Ramsey, S.; Ellsworth, J.; Chedeville, G.; Chetaille, A.L.; Saint-Cyr, C.; Cabral, D.A.; Spiegel, L.R.; et al. Early outcomes and improvement of patients with juvenile idiopathic arthritis enrolled in a Canadian multicenter inception cohort. Arthritis Care Res. 2010, 62, 527–536. [Google Scholar] [CrossRef]
- Saurenmann, R.K.; Rose, J.B.; Tyrrell, P.; Feldman, B.M.; Laxer, R.M.; Schneider, R.; Silverman, E.D. Epidemiology of juvenile idiopathic arthritis in a multiethnic cohort: Ethnicity as a risk factor. Arthritis Rheum. 2007, 56, 1974–1984. [Google Scholar] [CrossRef]
- Walscheid, K.; Glandorf, K.; Rothaus, K.; Niewerth, M.; Klotsche, J.; Minden, K.; Heiligenhaus, A. Enthesitis-related Arthritis: Prevalence and Complications of Associated Uveitis in Children and Adolescents From a Population-based Nationwide Study in Germany. J. Rheumatol. 2021, 48, 262–269. [Google Scholar] [CrossRef]
- Kalinina Ayuso, V.; Makhotkina, N.; van Tent-Hoeve, M.; de Groot-Mijnes, J.D.; Wulffraat, N.M.; Rothova, A.; de Boer, J.H. Pathogenesis of juvenile idiopathic arthritis associated uveitis: The known and unknown. Surv. Ophthalmol. 2014, 59, 517–531. [Google Scholar] [CrossRef] [PubMed]
- Heiligenhaus, A.; Minden, K.; Foll, D.; Pleyer, U. Uveitis in juvenile idiopathic arthritis. Dtsch. Arztebl. Int. 2015, 112, 92–100. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.S.; Roberton, D.; Hammerton, M.E. Juvenile arthritis-associated uveitis: Visual outcomes and prognosis. Can. J. Ophthalmol. 2004, 39, 614–620. [Google Scholar] [CrossRef]
- Rosenberg, A.M.; Oen, K.G. The relationship between ocular and articular disease activity in children with juvenile rheumatoid arthritis and associated uveitis. Arthritis Rheum. 1986, 29, 797–800. [Google Scholar] [CrossRef] [PubMed]
- Heiligenhaus, A.; Niewerth, M.; Ganser, G.; Heinz, C.; Minden, K.; German Uveitis in Childhood Study, G. Prevalence and complications of uveitis in juvenile idiopathic arthritis in a population-based nation-wide study in Germany: Suggested modification of the current screening guidelines. Rheumatology 2007, 46, 1015–1019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kotaniemi, K.; Kautiainen, H.; Karma, A.; Aho, K. Occurrence of uveitis in recently diagnosed juvenile chronic arthritis: A prospective study. Ophthalmology 2001, 108, 2071–2075. [Google Scholar] [CrossRef]
- Edelsten, C.; Lee, V.; Bentley, C.R.; Kanski, J.J.; Graham, E.M. An evaluation of baseline risk factors predicting severity in juvenile idiopathic arthritis associated uveitis and other chronic anterior uveitis in early childhood. Br. J. Ophthalmol. 2002, 86, 51–56. [Google Scholar] [CrossRef] [Green Version]
- Heinz, C.; Mingels, A.; Goebel, C.; Fuchsluger, T.; Heiligenhaus, A. Chronic uveitis in children with and without juvenile idiopathic arthritis: Differences in patient characteristics and clinical course. J. Rheumatol. 2008, 35, 1403–1407. [Google Scholar]
- Bolt, I.B.; Cannizzaro, E.; Seger, R.; Saurenmann, R.K. Risk factors and longterm outcome of juvenile idiopathic arthritis-associated uveitis in Switzerland. J. Rheumatol. 2008, 35, 703–706. [Google Scholar]
- Boone, M.I.; Moore, T.L.; Cruz, O.A. Screening for uveitis in juvenile rheumatoid arthritis. J. Pediatr. Ophthalmol. Strabismus 1998, 35, 41–43. [Google Scholar] [CrossRef]
- Demirkaya, E.; Ozen, S.; Bilginer, Y.; Ayaz, N.A.; Makay, B.B.; Unsal, E.; Erguven, M.; Poyrazoglu, H.; Kasapcopur, O.; Gok, F.; et al. The distribution of juvenile idiopathic arthritis in the eastern Mediterranean: Results from the registry of the Turkish Paediatric Rheumatology Association. Clin. Exp. Rheumatol. 2011, 29, 111–116. [Google Scholar] [PubMed]
- Kanski, J.J. Screening for uveitis in juvenile chronic arthritis. Br. J. Ophthalmol. 1989, 73, 225–228. [Google Scholar] [CrossRef] [Green Version]
- Sim, K.T.; Venning, H.E.; Barrett, S.; Gregson, R.M.; Amoaku, W.M. Extended oligoarthritis and other risk factors for developing JIA-associated uveitis under ILAR classification and its implication for current screening guideline. Ocul. Immunol. Inflamm. 2006, 14, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Bloom, J.N.; Ni, M.; Moore, T.L.; Osborn, T.G.; Hageman, G.S. Serum antiocular antibodies in patients with juvenile rheumatoid arthritis. J. Pediatr. Ophthalmol. Strabismus 1993, 30, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Busch, M.; Wefelmeyer, K.L.; Walscheid, K.; Rothaus, K.; Bauer, D.; Deeg, C.A.; Degroote, R.L.; Ackermann, D.; Konig, S.; Thanos, S.; et al. Identification of Ocular Autoantigens Associated With Juvenile Idiopathic Arthritis-Associated Uveitis. Front. Immunol. 2019, 10, 1793. [Google Scholar] [CrossRef] [Green Version]
- Uchiyama, R.C.; Osborn, T.G.; Moore, T.L. Antibodies to iris and retina detected in sera from patients with juvenile rheumatoid arthritis with iridocyclitis by indirect immunofluorescence studies on human eye tissue. J. Rheumatol. 1989, 16, 1074–1078. [Google Scholar]
- Walscheid, K.; Hennig, M.; Heinz, C.; Wasmuth, S.; Busch, M.; Bauer, D.; Dietzel, M.; Deeg, C.A.; Heiligenhaus, A. Correlation between disease severity and presence of ocular autoantibodies in juvenile idiopathic arthritis-associated uveitis. Investig. Ophthalmol. Vis. Sci. 2014, 55, 3447–3453. [Google Scholar] [CrossRef] [Green Version]
- Walscheid, K.; Neekamp, L.; Heiligenhaus, A.; Weinhage, T.; Heinz, C.; Foell, D. Increased Circulating Proinflammatory T Lymphocytes in Children with Different Forms of Anterior Uveitis: Results from a Pilot Study. Ocul. Immunol. Inflamm. 2019, 27, 788–797. [Google Scholar] [CrossRef]
- Caspi, R.R. Understanding autoimmune uveitis through animal models. The Friedenwald Lecture. Investig. Ophthalmol. Vis. Sci. 2011, 52, 1872–1879. [Google Scholar] [CrossRef]
- Forrester, J.V.; Klaska, I.P.; Yu, T.; Kuffova, L. Uveitis in mouse and man. Int. Rev. Immunol. 2013, 32, 76–96. [Google Scholar] [CrossRef]
- Kasper, M.; Walscheid, K.; Laffer, B.; Bauer, D.; Busch, M.; Loser, K.; Vogl, T.; Langmann, T.; Ganser, G.; Rath, T.; et al. Phenotype of Innate Immune Cells in Uveitis Associated with Axial Spondyloarthritis- and Juvenile Idiopathic Arthritis-associated Uveitis. Ocul. Immunol. Inflamm. 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Moschovakis, G.L.; Bubke, A.; Friedrichsen, M.; Ristenpart, J.; Back, J.W.; Falk, C.S.; Kremmer, E.; Forster, R. The chemokine receptor CCR7 is a promising target for rheumatoid arthritis therapy. Cell Mol. Immunol. 2019, 16, 791–799. [Google Scholar] [CrossRef]
- Wildschutz, L.; Ackermann, D.; Witten, A.; Kasper, M.; Busch, M.; Glander, S.; Melkonyan, H.; Walscheid, K.; Tappeiner, C.; Thanos, S.; et al. Transcriptomic and proteomic analysis of iris tissue and aqueous humor in juvenile idiopathic arthritis-associated uveitis. J. Autoimmun. 2019, 100, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Austermann, J.; Spiekermann, C.; Roth, J. S100 proteins in rheumatic diseases. Nat. Rev. Rheumatol. 2018, 14, 528–541. [Google Scholar] [CrossRef]
- Kessel, C.; Holzinger, D.; Foell, D. Phagocyte-derived S100 proteins in autoinflammation: Putative role in pathogenesis and usefulness as biomarkers. Clin. Immunol. 2013, 147, 229–241. [Google Scholar] [CrossRef]
- Walscheid, K.; Heiligenhaus, A.; Holzinger, D.; Roth, J.; Heinz, C.; Tappeiner, C.; Kasper, M.; Foell, D. Elevated S100A8/A9 and S100A12 Serum Levels Reflect Intraocular Inflammation in Juvenile Idiopathic Arthritis-Associated Uveitis: Results From a Pilot Study. Investig. Ophthalmol. Vis. Sci. 2015, 56, 7653–7660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ehrchen, J.M.; Sunderkotter, C.; Foell, D.; Vogl, T.; Roth, J. The endogenous Toll-like receptor 4 agonist S100A8/S100A9 (calprotectin) as innate amplifier of infection, autoimmunity, and cancer. J. Leukoc. Biol. 2009, 86, 557–566. [Google Scholar] [CrossRef]
- Foell, D.; Wittkowski, H.; Vogl, T.; Roth, J. S100 proteins expressed in phagocytes: A novel group of damage-associated molecular pattern molecules. J. Leukoc. Biol. 2007, 81, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.L.N.; Mageean, K.S.; Carlton, H.; Simonini, G.; Sharp, G.C.; Relton, C.L.; Ramanan, A.V. Environmental risk factors associated with juvenile idiopathic arthritis associated uveitis: A systematic review of the literature. J. Ophthalmic. Inflamm. Infect. 2021, 11, 15. [Google Scholar] [CrossRef]
- Christakos, S.; Dhawan, P.; Verstuyf, A.; Verlinden, L.; Carmeliet, G. Vitamin D: Metabolism, Molecular Mechanism of Action, and Pleiotropic Effects. Physiol. Rev. 2016, 96, 365–408. [Google Scholar] [CrossRef]
- Nisar, M.K.; Masood, F.; Cookson, P.; Sansome, A.; Ostor, A.J. What do we know about juvenile idiopathic arthritis and vitamin D? A systematic literature review and meta-analysis of current evidence. Clin. Rheumatol. 2013, 32, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Sengler, C.; Zink, J.; Klotsche, J.; Niewerth, M.; Liedmann, I.; Horneff, G.; Kessel, C.; Ganser, G.; Thon, A.; Haas, J.P.; et al. Vitamin D deficiency is associated with higher disease activity and the risk for uveitis in juvenile idiopathic arthritis-data from a German inception cohort. Arthritis Res. Ther. 2018, 20, 276. [Google Scholar] [CrossRef] [Green Version]
- Mason, R.S.; Sequeira, V.B.; Gordon-Thomson, C. Vitamin D: The light side of sunshine. Eur. J. Clin. Nutr. 2011, 65, 986–993. [Google Scholar] [CrossRef] [PubMed]
- Clemens, L.E.; Albert, E.; Ansell, B.M. Sibling pairs affected by chronic arthritis of childhood: Evidence for a genetic predisposition. J. Rheumatol. 1985, 12, 108–113. [Google Scholar]
- Julian, K.; Terrada, C.; Quartier, P.; Lehoang, P.; Bodaghi, B. Uveitis related to juvenile idiopathic arthritis: Familial cases and possible genetic implication in the pathogenesis. Ocul. Immunol. Inflamm. 2010, 18, 172–177. [Google Scholar] [CrossRef]
- Moroldo, M.B.; Chaudhari, M.; Shear, E.; Thompson, S.D.; Glass, D.N.; Giannini, E.H. Juvenile rheumatoid arthritis affected sibpairs: Extent of clinical phenotype concordance. Arthritis Rheum. 2004, 50, 1928–1934. [Google Scholar] [CrossRef]
- Moroldo, M.B.; Tague, B.L.; Shear, E.S.; Glass, D.N.; Giannini, E.H. Juvenile rheumatoid arthritis in affected sibpairs. Arthritis Rheum. 1997, 40, 1962–1966. [Google Scholar] [CrossRef]
- Rosenberg, A.M.; Petty, R.E. Similar patterns of juvenile rheumatoid arthritis within families. Arthritis Rheum 1980, 23, 951–953. [Google Scholar] [CrossRef]
- Saila, H.; Kotaniemi, K.; Savolainen, A.; Kautiainen, H.; Leirisalo-Repo, M.; Aho, K. Uveitis in sibling pairs with juvenile idiopathic arthritis. Rheumatology 2001, 40, 221–224. [Google Scholar] [CrossRef] [Green Version]
- Haasnoot, A.J.W.; Schilham, M.W.; Kamphuis, S.; Hissink Muller, P.C.E.; Heiligenhaus, A.; Foell, D.; Minden, K.; Ophoff, R.A.; Radstake, T.; Den Hollander, A.I.; et al. Identification of an Amino Acid Motif in HLA-DRbeta1 That Distinguishes Uveitis in Patients With Juvenile Idiopathic Arthritis. Arthritis Rheumatol. 2018, 70, 1155–1165. [Google Scholar] [CrossRef]
- Viallard, J.F.; Bloch-Michel, C.; Neau-Cransac, M.; Taupin, J.L.; Garrigue, S.; Miossec, V.; Mercie, P.; Pellegrin, J.L.; Moreau, J.F. HLA-DR expression on lymphocyte subsets as a marker of disease activity in patients with systemic lupus erythematosus. Clin. Exp. Immunol. 2001, 125, 485–491. [Google Scholar] [CrossRef]
- Wakiguchi, H.; Hasegawa, S.; Suzuki, Y.; Kudo, K.; Ichiyama, T. Relationship between T-cell HLA-DR expression and intravenous immunoglobulin treatment response in Kawasaki disease. Pediatr. Res. 2015, 77, 536–540. [Google Scholar] [CrossRef] [Green Version]
- Yanagimachi, M.; Miyamae, T.; Naruto, T.; Hara, T.; Kikuchi, M.; Hara, R.; Imagawa, T.; Mori, M.; Kaneko, T.; Goto, H.; et al. Association of HLA-A*02:06 and HLA-DRB1*04:05 with clinical subtypes of juvenile idiopathic arthritis. J. Hum. Genet. 2011, 56, 196–199. [Google Scholar] [CrossRef]
- Weiss, P.F.; Klink, A.J.; Behrens, E.M.; Sherry, D.D.; Finkel, T.H.; Feudtner, C.; Keren, R. Enthesitis in an inception cohort of enthesitis-related arthritis. Arthritis Care Res. 2011, 63, 1307–1312. [Google Scholar] [CrossRef] [PubMed]
- Petty, R.E.; Southwood, T.R.; Manners, P.; Baum, J.; Glass, D.N.; Goldenberg, J.; He, X.; Maldonado-Cocco, J.; Orozco-Alcala, J.; Prieur, A.M.; et al. International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: Second revision, Edmonton, 2001. J. Rheumatol. 2004, 31, 390–392. [Google Scholar] [PubMed]
- Kavadichanda, C.G.; Seth, G.; Kumar, G.; Gulati, R.; Negi, V.S. Clinical correlates of HLA-B*27 and its subtypes in enthesitis-related arthritis variant of juvenile idiopathic arthritis in south Indian Tamil patients. Int. J. Rheum. Dis. 2019, 22, 1289–1296. [Google Scholar] [CrossRef]
- Jhaj, G.; Kopplin, L.J. Ocular features of the HLA-B27-positive seronegative spondyloarthropathies. Curr. Opin. Ophthalmol. 2018, 29, 552–557. [Google Scholar] [CrossRef] [PubMed]
- McDowell, T.L.; Symons, J.A.; Ploski, R.; Forre, O.; Duff, G.W. A genetic association between juvenile rheumatoid arthritis and a novel interleukin-1 alpha polymorphism. Arthritis Rheum. 1995, 38, 221–228. [Google Scholar] [CrossRef]
- Donn, R.P.; Barrett, J.H.; Farhan, A.; Stopford, A.; Pepper, L.; Shelley, E.; Davies, N.; Ollier, W.E.; Thomson, W. Cytokine gene polymorphisms and susceptibility to juvenile idiopathic arthritis. British Paediatric Rheumatology Study Group. Arthritis Rheum. 2001, 44, 802–810. [Google Scholar] [CrossRef]
- Donn, R.P.; Farhan, A.J.; Barrett, J.H.; Thomson, W.; Worthington, J.; Ollier, W.E. Absence of association between interleukin 1 alpha and oligoarticular juvenile chronic arthritis in UK patients. Rheumatology 1999, 38, 171–175. [Google Scholar] [CrossRef] [Green Version]
- Pers, Y.M.; Le Blay, P.; Ludwig, C.; Rittore, C.; Tejedor, G.; Foliwe, R.; Rodiere, M.; Jorgensen, C.; Touitou, I. Association of TRAF1-C5 with risk of uveitis in juvenile idiopathic arthritis. Joint. Bone Spine 2017, 84, 305–308. [Google Scholar] [CrossRef]
- Han, T.U.; Bang, S.Y.; Kang, C.; Bae, S.C. TRAF1 polymorphisms associated with rheumatoid arthritis susceptibility in Asians and in Caucasians. Arthritis Rheum. 2009, 60, 2577–2584. [Google Scholar] [CrossRef] [PubMed]
- Angeles-Han, S.T.; Yeh, S.; Vogler, L.B. Updates on the risk markers and outcomes of severe juvenile idiopathic arthritis-associated uveitis. Int. J. Clin. Rheumtol. 2013, 8. [Google Scholar] [CrossRef]
- Alberdi-Saugstrup, M.; Enevold, C.; Zak, M.; Nielsen, S.; Nordal, E.; Berntson, L.; Fasth, A.; Rygg, M.; Muller, K.; Nordic Study Group of Pediatric, R. Non-HLA gene polymorphisms in juvenile idiopathic arthritis: Associations with disease outcome. Scand. J. Rheumatol. 2017, 46, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Y.; Wang, W.P. B7-H4, a promising target for immunotherapy. Cell Immunol. 2020, 347, 104008. [Google Scholar] [CrossRef]
- Cassidy, J.; Kivlin, J.; Lindsley, C.; Nocton, J. Ophthalmologic examinations in children with juvenile rheumatoid arthritis. Pediatrics 2006, 117, 1843–1845. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.J.Y.; Duffy, C.M.; Guzman, J.; Oen, K.; Barrowman, N.; Rosenberg, A.M.; Shiff, N.J.; Boire, G.; Stringer, E.; Spiegel, L.; et al. Prospective Determination of the Incidence and Risk Factors of New-Onset Uveitis in Juvenile Idiopathic Arthritis: The Research in Arthritis in Canadian Children Emphasizing Outcomes Cohort. Arthritis Care Res. 2019, 71, 1436–1443. [Google Scholar] [CrossRef]
- Nordal, E.B.; Songstad, N.T.; Berntson, L.; Moen, T.; Straume, B.; Rygg, M. Biomarkers of chronic uveitis in juvenile idiopathic arthritis: Predictive value of antihistone antibodies and antinuclear antibodies. J. Rheumatol. 2009, 36, 1737–1743. [Google Scholar] [CrossRef] [PubMed]
- Grassi, A.; Corona, F.; Casellato, A.; Carnelli, V.; Bardare, M. Prevalence and outcome of juvenile idiopathic arthritis-associated uveitis and relation to articular disease. J. Rheumatol. 2007, 34, 1139–1145. [Google Scholar]
- Haasnoot, A.J.; van Tent-Hoeve, M.; Wulffraat, N.M.; Schalij-Delfos, N.E.; Los, L.I.; Armbrust, W.; Zuithoff, N.P.; de Boer, J.H. Erythrocyte sedimentation rate as baseline predictor for the development of uveitis in children with juvenile idiopathic arthritis. Am. J. Ophthalmol. 2015, 159, 372–377 e371. [Google Scholar] [CrossRef] [Green Version]
- Kotaniemi, K.; Arkela-Kautiainen, M.; Haapasaari, J.; Leirisalo-Repo, M. Uveitis in young adults with juvenile idiopathic arthritis: A clinical evaluation of 123 patients. Ann. Rheum. Dis. 2005, 64, 871–874. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kotaniemi, K.; Kotaniemi, A.; Savolainen, A. Uveitis as a marker of active arthritis in 372 patients with juvenile idiopathic seronegative oligoarthritis or polyarthritis. Clin. Exp. Rheumatol. 2002, 20, 109–112. [Google Scholar]
- Pelegrin, L.; Casaroli-Marano, R.; Anton, J.; Garcia de Vicuna, M.C.; Molina-Prat, N.; Ignacio Arostegui, J.; Yague, J.; Rios, J.; Adan, A. Predictive value of selected biomarkers, polymorphisms, and clinical features for oligoarticular juvenile idiopathic arthritis-associated uveitis. Ocul. Immunol. Inflamm. 2014, 22, 208–212. [Google Scholar] [CrossRef]
- Zulian, F.; Martini, G.; Falcini, F.; Gerloni, V.; Zannin, M.E.; Pinello, L.; Fantini, F.; Facchin, P. Early predictors of severe course of uveitis in oligoarticular juvenile idiopathic arthritis. J. Rheumatol. 2002, 29, 2446–2453. [Google Scholar] [PubMed]
- Haasnoot, A.M.; Kuiper, J.J.; Hiddingh, S.; Schellekens, P.A.; de Jager, W.; Imhof, S.M.; Radstake, T.R.; de Boer, J.H. Ocular Fluid Analysis in Children Reveals Interleukin-29/Interferon-lambda1 as a Biomarker for Juvenile Idiopathic Arthritis-Associated Uveitis. Arthritis Rheumatol. 2016, 68, 1769–1779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalinina Ayuso, V.; de Boer, J.H.; Byers, H.L.; Coulton, G.R.; Dekkers, J.; de Visser, L.; van Loon, A.M.; Schellekens, P.A.; Rothova, A.; de Groot-Mijnes, J.D. Intraocular biomarker identification in uveitis associated with juvenile idiopathic arthritis. Investig. Ophthalmol. Vis. Sci. 2013, 54, 3709–3720. [Google Scholar] [CrossRef] [Green Version]
- Angeles-Han, S.T.; Yeh, S.; Patel, P.; Duong, D.; Jenkins, K.; Rouster-Stevens, K.A.; Altaye, M.; Fall, N.; Thornton, S.; Prahalad, S.; et al. Discovery of tear biomarkers in children with chronic non-infectious anterior uveitis: A pilot study. J. Ophthalmic. Inflamm. Infect. 2018, 8, 17. [Google Scholar] [CrossRef] [Green Version]
- Godfrey, W.A.; Lindsley, C.B.; Cuppage, F.E. Localization of IgM in plasma cells in the iris of a patient with iridocyclitis and juvenile rheumatoid arthritis. Arthritis Rheum. 1981, 24, 1195–1198. [Google Scholar] [CrossRef]
- Kalinina Ayuso, V.; van Dijk, M.R.; de Boer, J.H. Infiltration of Plasma Cells in the Iris of Children With ANA-Positive Anterior Uveitis. Investig. Ophthalmol. Vis. Sci. 2015, 56, 6770–6778. [Google Scholar] [CrossRef] [Green Version]
- Merriam, J.C.; Chylack, L.T., Jr.; Albert, D.M. Early-onset pauciarticular juvenile rheumatoid arthritis. A histopathologic study. Arch. Ophthalmol. 1983, 101, 1085–1092. [Google Scholar] [CrossRef]
- Parikh, J.G.; Tawansy, K.A.; Rao, N.A. Immunohistochemical study of chronic nongranulomatous anterior uveitis in juvenile idiopathic arthritis. Ophthalmology 2008, 115, 1833–1836. [Google Scholar] [CrossRef] [PubMed]
- Sabates, R.; Smith, T.; Apple, D. Ocular histopathology in juvenile rheumatoid arthritis. Ann. Ophthalmol. 1979, 11, 733–737. [Google Scholar]
- Packham, J.C.; Hall, M.A. Long-term follow-up of 246 adults with juvenile idiopathic arthritis: Functional outcome. Rheumatology 2002, 41, 1428–1435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castro, C.; Gourley, M. Diagnostic testing and interpretation of tests for autoimmunity. J. Allergy Clin. Immunol. 2010, 125, S238–S247. [Google Scholar] [CrossRef] [Green Version]
- Harrison, M. Erythrocyte sedimentation rate and C-reactive protein. Aust. Prescr. 2015, 38, 93–94. [Google Scholar] [CrossRef] [PubMed]
- Lazear, H.M.; Daniels, B.P.; Pinto, A.K.; Huang, A.C.; Vick, S.C.; Doyle, S.E.; Gale, M., Jr.; Klein, R.S.; Diamond, M.S. Interferon-lambda restricts West Nile virus neuroinvasion by tightening the blood-brain barrier. Sci. Transl. Med. 2015, 7, 284ra259. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, S.; Padmanabhan, A.; Vaidya, T.; Watson, A.M.; Bhutto, I.A.; Hose, S.; Shang, P.; Stepicheva, N.; Yazdankhah, M.; Weiss, J.; et al. Neutrophils homing into the retina trigger pathology in early age-related macular degeneration. Commun. Biol. 2019, 2, 348. [Google Scholar] [CrossRef] [PubMed]
- Buxbaum, J.N.; Reixach, N. Transthyretin: The servant of many masters. Cell Mol. Life Sci. 2009, 66, 3095–3101. [Google Scholar] [CrossRef] [Green Version]
- Mullins, R.F.; Russell, S.R.; Anderson, D.H.; Hageman, G.S. Drusen associated with aging and age-related macular degeneration contain proteins common to extracellular deposits associated with atherosclerosis, elastosis, amyloidosis, and dense deposit disease. FASEB J. 2000, 14, 835–846. [Google Scholar] [CrossRef]
- Carreno, E.; Portero, A.; Herreras, J.M.; Garcia-Vazquez, C.; Whitcup, S.M.; Stern, M.E.; Calonge, M.; Enriquez-de-Salamanca, A. Cytokine and chemokine tear levels in patients with uveitis. Acta Ophthalmol. 2017, 95, e405–e414. [Google Scholar] [CrossRef] [Green Version]
- Constantin, T.; Foeldvari, I.; Anton, J.; de Boer, J.; Czitrom-Guillaume, S.; Edelsten, C.; Gepstein, R.; Heiligenhaus, A.; Pilkington, C.A.; Simonini, G.; et al. Consensus-based recommendations for the management of uveitis associated with juvenile idiopathic arthritis: The SHARE initiative. Ann. Rheum. Dis. 2018, 77, 1107–1117. [Google Scholar] [CrossRef]
- Amin, R.M.; Miserocchi, E.; Thorne, J.E.; Hornbeak, D.; Jabs, D.A.; Zierhut, M. Treatment Options for Juvenile Idiopathic Arthritis (JIA) Associated Uveitis. Ocul. Immunol. Inflamm. 2016, 24, 81–90. [Google Scholar] [CrossRef]
- Asproudis, I.; Katsanos, A.; Kozeis, N.; Tantou, A.; Konstas, A.G. Update on the Treatment of Uveitis in Patients with Juvenile Idiopathic Arthritis: A Review. Adv. Ther. 2017, 34, 2558–2565. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.L.; Abiri, P.; Tsui, E. Recent advances in the treatment of juvenile idiopathic arthritis-associated uveitis. Ther. Adv. Ophthalmol. 2021, 13, 2515841420984572. [Google Scholar] [CrossRef]
- Gaggiano, C.; Rigante, D.; Tosi, G.M.; Vitale, A.; Frediani, B.; Grosso, S.; Fabiani, C.; Cantarini, L. Treating juvenile idiopathic arthritis (JIA)-related uveitis beyond TNF-alpha inhibition: A narrative review. Clin. Rheumatol. 2020, 39, 327–337. [Google Scholar] [CrossRef]
- Schnabel, A.; Unger, E.; Bruck, N.; Berner, R.; Range, U.; Holl-Wieden, A.; Morbach, H.; Leszczynska, A.; Bau, V.; Hedrich, C.M. High-dose intravenous methylprednisolone in juvenile non-infectious uveitis: A retrospective analysis. Clin. Immunol. 2020, 211, 108327. [Google Scholar] [CrossRef]
- Bou, R.; Adan, A.; Borras, F.; Bravo, B.; Calvo, I.; De Inocencio, J.; Diaz, J.; Escudero, J.; Fonollosa, A.; de Vicuna, C.G.; et al. Clinical management algorithm of uveitis associated with juvenile idiopathic arthritis: Interdisciplinary panel consensus. Rheumatol. Int. 2015, 35, 777–785. [Google Scholar] [CrossRef]
- Heiligenhaus, A.; Michels, H.; Schumacher, C.; Kopp, I.; Neudorf, U.; Niehues, T.; Baus, H.; Becker, M.; Bertram, B.; Dannecker, G.; et al. Evidence-based, interdisciplinary guidelines for anti-inflammatory treatment of uveitis associated with juvenile idiopathic arthritis. Rheumatol. Int. 2012, 32, 1121–1133. [Google Scholar] [CrossRef] [PubMed]
- Nussenblatt, R.B.; Palestine, A.G.; Chan, C.C.; Stevens, G., Jr.; Mellow, S.D.; Green, S.B. Randomized, double-masked study of cyclosporine compared to prednisolone in the treatment of endogenous uveitis. Am. J. Ophthalmol. 1991, 112, 138–146. [Google Scholar] [CrossRef]
- Tappeiner, C.; Roesel, M.; Heinz, C.; Michels, H.; Ganser, G.; Heiligenhaus, A. Limited value of cyclosporine A for the treatment of patients with uveitis associated with juvenile idiopathic arthritis. Eye 2009, 23, 1192–1198. [Google Scholar] [CrossRef] [PubMed]
- Ramanan, A.V.; Dick, A.D.; Jones, A.P.; McKay, A.; Williamson, P.R.; Compeyrot-Lacassagne, S.; Hardwick, B.; Hickey, H.; Hughes, D.; Woo, P.; et al. Adalimumab plus Methotrexate for Uveitis in Juvenile Idiopathic Arthritis. N. Engl. J. Med. 2017, 376, 1637–1646. [Google Scholar] [CrossRef]
- Quartier, P.; Baptiste, A.; Despert, V.; Allain-Launay, E.; Kone-Paut, I.; Belot, A.; Kodjikian, L.; Monnet, D.; Weber, M.; Elie, C.; et al. ADJUVITE: A double-blind, randomised, placebo-controlled trial of adalimumab in early onset, chronic, juvenile idiopathic arthritis-associated anterior uveitis. Ann. Rheum. Dis. 2018, 77, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- Ramanan, A.V.; Dick, A.D.; Guly, C.; McKay, A.; Jones, A.P.; Hardwick, B.; Lee, R.W.J.; Smyth, M.; Jaki, T.; Beresford, M.W.; et al. Tocilizumab in patients with anti-TNF refractory juvenile idiopathic arthritis-associated uveitis (APTITUDE): A multicentre, single-arm, phase 2 trial. Lancet Rheumatol. 2020, 2, e135–e141. [Google Scholar] [CrossRef] [Green Version]
- Clarke, S.L.; Sen, E.S.; Ramanan, A.V. Juvenile idiopathic arthritis-associated uveitis. Pediatr. Rheumatol. Online J. 2016, 14, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foster, C.S.; Barrett, F. Cataract development and cataract surgery in patients with juvenile rheumatoid arthritis-associated iridocyclitis. Ophthalmology 1993, 100, 809–817. [Google Scholar] [CrossRef]
| Type of JIA | Sex Ratio (F:M) | Age of Uveitis Onset | Uveitis Risk | Type of Uveitis |
|---|---|---|---|---|
| Systemic arthritis | 1:1 | N/A | Low | N/A |
| Oligoarthritis (persistent) | 3:1 | Early childhood | 10–20% | Chronic asymptomatic |
| Oligoarthritis (extended) | 3:1 | Early childhood | 20–30% | Chronic asymptomatic |
| Polyarthritis (RF-) | 3:1 | Early childhood | 10–20% | Chronic asymptomatic |
| Polyarthritis (RF+) | 4:1 | N/A | Low | N/A |
| Psoriatic arthritis | 2:1 | Early childhood or school-age | 5–20% | Chronic asymptomatic or acute symptomatic |
| Enthesitis related arthritis | 1:4 | School-age | 5–20% | Acute symptomatic |
| Risk Factor | Tissue | Status in JIA-Uveitis | Comparator Group | Reference |
|---|---|---|---|---|
| ANA | Blood | Higher proportion positive | JIA | [2,4,10,11,14,16,19,25,26,28,76,77,78] |
| ESR | Blood | Increased rate | JIA | [14,79,80,81,82,83,84] |
| Anti-ocular antibodies | Serum | Higher proportion positive | JIA, idiopathic arthritis and healthy controls | [34,35,36,37] |
| BAFF | AqH | Increased | POAG | [43] |
| APRIL | AqH | Increased | POAG | [43] |
| IL-6 | AqH | Increased | POAG | [43] |
| IL-29 | AqH | Reduced | JIA | [85] |
| OPG | AqH | Increased | Healthy controls and idiopathic arthritis | [85] |
| LAP | AqH | Increased | Healthy controls and idiopathic arthritis | [85] |
| TTR | AqH | Increased | Other uveitis and non-inflammatory controls. | [86] |
| S100A8/A9 | AqH/Serum | Increased | JIA | [46] |
| S100A12 | AqH/Serum | Increased | JIA | [46] |
| SEMA3G | Tears | Increased | Idiopathic uveitis | [87] |
| TIMP1 | Tears | Increased | Idiopathic uveitis | [87] |
| HEXB | Tears | Increased | Idiopathic uveitis | [87] |
| ERN1 | Tears | Increased | Idiopathic uveitis | [87] |
| SAA1 | Tears | Increased | Idiopathic uveitis | [87] |
| sCD14 | Tears | Increased | Idiopathic uveitis | [87] |
| S100A8 | Tears | Increased | Idiopathic uveitis | [87] |
| S100A9 | Tears | Reduced | Idiopathic uveitis | [87] |
| LAP3 | Tears | Reduced | Idiopathic uveitis | [87] |
| TTR | Tears | Reduced | Idiopathic uveitis | [87] |
| MIF | Tears | Reduced | Idiopathic uveitis | [87] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carlsson, E.; Beresford, M.W.; Ramanan, A.V.; Dick, A.D.; Hedrich, C.M. Juvenile Idiopathic Arthritis Associated Uveitis. Children 2021, 8, 646. https://doi.org/10.3390/children8080646
Carlsson E, Beresford MW, Ramanan AV, Dick AD, Hedrich CM. Juvenile Idiopathic Arthritis Associated Uveitis. Children. 2021; 8(8):646. https://doi.org/10.3390/children8080646
Chicago/Turabian StyleCarlsson, Emil, Michael W. Beresford, Athimalaipet V. Ramanan, Andrew D. Dick, and Christian M. Hedrich. 2021. "Juvenile Idiopathic Arthritis Associated Uveitis" Children 8, no. 8: 646. https://doi.org/10.3390/children8080646







