Invasive Infections Associated with the Use of Probiotics in Children: A Systematic Review
Abstract
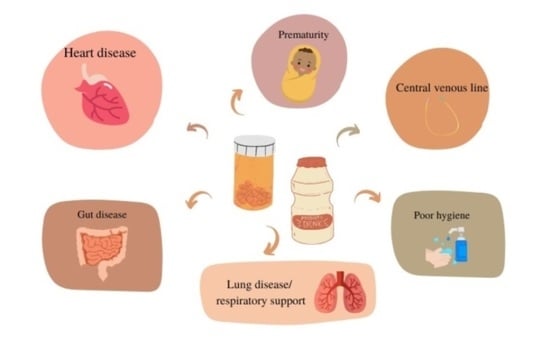
1. Introduction
2. Materials and Methods
- −
- Publication data: title and authors of the article, name and year of the journal, volume, number, and pages of the articles.
- −
- Patient information: age, gender, underlying condition(s), exposure time to probiotics (in days), type and duration of the treatment (in days), and outcome (favourable or death).
- −
- Type of infection: identified microorganisms.
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The international scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Fijan, S. Microorganisms with claimed probiotic properties: An overview of recent literature. Int. J. Environ. Res. Public Health 2014, 11, 4745–4767. [Google Scholar] [CrossRef]
- Bernardo, W.; Aires, F.T.; Carneiro, R.M.; Sá, F.P.; Rullo, V.E.; Burns, D.A. Effectiveness of probiotics in the prophylaxis of necrotizing enterocolitis in preterm neonates: A systematic review and meta-analysis. J. Pediatr. 2013, 89, 18–24. [Google Scholar] [CrossRef]
- AlFaleh, K.; Anabrees, J. Probiotics for prevention of necrotizing enterocolitis in preterm infants. Cochrane Database Syst. Rev. 2014, 4. [Google Scholar]
- Rao, S.C.; Athalye-Jape, G.K.; Deshpande, G.C.; Simmer, K.N.; Patole, S.K. Probiotic supplementation and late-onset Sepsis in preterm infants: A meta-analysis. Pediatrics 2016, 137, e20153684. [Google Scholar] [CrossRef] [PubMed]
- Bi, L.W.; Yan, B.L.; Yang, Q.Y.; Li, M.M.; Cui, H.L. Probiotic strategies to prevent necrotizing enterocolitis in preterm infants: A meta-analysis. Pediatr. Surg. Int. 2019, 35, 1143–1162. [Google Scholar] [CrossRef] [PubMed]
- Schnadower, D.; Tarr, P.I.; Casper, T.C.; Gorelick, M.H.; Dean, J.M.; O’Connell, K.J.; Mahajan, P.; Levine, A.C.; Bhatt, S.R.; Roskind, C.G.; et al. Lactobacillus rhamnosus GG versus Placebo for Acute Gastroenteritis in Children. N. Engl. J. Med. 2018, 379, 2002–2014. [Google Scholar] [CrossRef] [PubMed]
- Collinson, S.; Deans, A.; Padua-Zamora, A.; Gregorio, G.V.; Li, C.; Dans, L.F.; Allen, S.J. Probiotics for treating acute infectious diarrhoea. Cochrane Database Syst. Rev. 2020, 12, CD003048. [Google Scholar] [CrossRef]
- Francavilla, R.; Miniello, V.; Magistà, A.M.; De Canio, A.; Bucci, N.; Gagliardi, F.; Lionetti, E.; Castellaneta, S.; Polimeno, L.; Peccarisi, L.; et al. A randomized controlled trial of Lactobacillus GG in children with functional abdominal pain. Pediatrics 2010, 126, e1445–e1452. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Ni, B.; Liu, Z.; Liu, X.; Xie, W.; Wu, I.X.Y.; Li, X. The Role of Probiotics in the Prevention and Treatment of Atopic Dermatitis in Children: An Updated Systematic Review and Meta-Analysis of Randomized Controlled Trials. Paediatr. Drugs. 2020, 22, 535–549. [Google Scholar] [CrossRef] [PubMed]
- Sung, V.; Hiscock, H.; Tang, M.L.; Mensah, F.; Nation, M.L.; Satzke, C.; Heine, R.G.; Stock, A.; Barr, R.G.; Wake, M. Treating infant colic with the probiotic Lactobacillus reuteri: Double blind, placebo controlled randomised trial. BMJ 2014, 348, g2107. [Google Scholar] [CrossRef]
- Braga, V.L.; Rocha, L.P.D.S.; Bernardo, D.D.; Cruz, C.O.; Riera, R. What do Cochrane systematic reviews say about probiotics as preventive interventions? Sao Paulo Med. J. 2017, 135, 578–586. [Google Scholar] [CrossRef]
- Probiotics Market Size, Share & Trends Analysis Report By Product (Food & Beverages, Dietary Supplements), By Ingredient (Bacteria, Yeast), By End Use (Human, Animal), By Distribution Channel, And Segment Forecasts, 2021–2028, Grand View Research. 2021. Available online: https://www.grandviewresearch.com/industry-analysis/probiotics-market (accessed on 1 August 2021).
- Snydman, D.R. The safety of probiotics. Clin. Infect. Dis. 2008, 46 (Suppl. 2), S104–S111; discussion S144–S151. [Google Scholar] [CrossRef]
- Didari, T.; Solki, S.; Mozaffari, S.; Nikfar, S.; Abdollahi, M. A systematic review of the safety of probiotics. Expert Opin. Drug Saf. 2014, 13, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Sotoudegan, F.; Daniali, M.; Hassani, S.; Nikfar, S.; Abdollahi, M. Reappraisal of probiotics’ safety in human. Food Chem. Toxicol. 2019, 129, 22. [Google Scholar] [CrossRef] [PubMed]
- Bafeta, A.; Koh, M.; Riveros, C.; Ravaud, P. Harms Reporting in Randomized Controlled Trials of Interventions Aimed at Modifying Microbiota: A Systematic Review. Ann. Intern. Med. 2018, 169, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Yelin, I.; Flett, K.B.; Merakou, C.; Mehrotra, P.; Stam, J.; Snesrud, E.; Hinkle, M.; Lesho, E.; McGann, P.; McAdam, A.J.; et al. Genomic and epidemiological evidence of bacterial transmission from probiotic capsule to blood in ICU patients. Nat. Med. 2019, 25, 1728–1732. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.A.; Woodfield, M.C.; Boyle, N.; Stednick, Z.; Boeckh, M.; Pergam, S.A. Incidence and outcomes of bloodstream infections among hematopoietic cell transplant recipients from species commonly reported to be in over-the-counter probiotic formulations. Transpl. Infect. Dis. 2016, 18, 699–705. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Goldenberg, J.Z.; Humphrey, C.; El Dib, R.; Johnston, B.C. Probiotics for the prevention of pediatric antibiotic-associated diarrhea. Cochrane Database Syst. Rev. 2019, 4, CD004827. [Google Scholar] [CrossRef] [PubMed]
- Mackay, A.D.; Taylor, M.B.; Kibbler, C.C.; Hamilton-Miller, J.M. Lactobacillus endocarditis caused by a probiotic organism. Clin. Microbiol Infect. 1999, 5, 290–292. [Google Scholar] [CrossRef]
- Presterl, E.; Kneifel, W.; Mayer, H.K.; Zehetgruber, M.; Makristathis, A.; Graninger, W. Endocarditis by Lactobacillus rhamnosus due to yogurt ingestion? Scand J. Infect. Dis. 2001, 33, 710–714. [Google Scholar]
- Pasala, S.; Singer, L.; Arshad, T.; Roach, K. Lactobacillus endocarditis in a healthy patient with probiotic use. IDCases 2020, 22, e00915. [Google Scholar] [CrossRef]
- Conen, A.; Zimmerer, S.; Trampuz, A.; Frei, R.; Battegay, M.; Elzi, L. A pain in the neck: Probiotics for ulcerative colitis. Ann. Intern. Med. 2009, 151, 895–897, Erratum in: Ann Intern Med. 2010, 152, 200. [Google Scholar] [CrossRef]
- Costa, R.L.; Moreira, J.; Lorenzo, A.; Lamas, C.C. Infectious complications following probiotic ingestion: A potentially underestimated problem? A systematic review of reports and case series. BMC Complement. Altern. Med. 2018, 18, 329. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef] [PubMed]
- Kunz, A.N.; Noel, J.M.; Fairchok, M.P. Two cases of Lactobacillus bacteremia during probiottreatment of short gut syndrome. J. Pediatr. Gastroenterol. Nutr. 2004, 38, 457–45821. [Google Scholar] [CrossRef] [PubMed]
- Land, M.H.; Rouster-Stevens, K.; Woods, C.R.; Cannon, M.L.; Cnota, J.; Shetty, A.K. Lactobacillus sepsis associated with probiotic therapy. Pediatrics 2005, 115, 178–181. [Google Scholar] [CrossRef]
- Dani, C.; Coviello, C.; Corsini, I.; Arena, F.; Antonelli, A.; Rossolini, G.M. Lactobacillus sepsis and probiotic therapy in newborns: Two new cases and literature review. Am. J. Perinatol. Rep. 2016, 6, e25. [Google Scholar]
- De Groote, M.A.; Frank, D.N.; Dowell, E.; Glode, M.P.; Pace, N.R. Lactobacillus rhamnosus GG bacteremia associatedwith probiotic use in a child with short gut syndrome. Pediatr. Infect. Dis. J. 2005, 24, 278–280. [Google Scholar] [CrossRef]
- Sadowska-Krawczenko, I.; Paprzycka, M.; Korbal, P.; Wiatrzyk, A.; Krysztopa-Grzybowska, K.; Polak, M.; Czajka, U.; Lutyńska, A. Lactobacillus rhamnosus GG suspected infection in a newbornwith intrauterine growth restriction. Benef. Microbes 2014, 5, 397–402. [Google Scholar] [CrossRef]
- Cavicchiolo, M.E.; Magnani, M.; Calgaro, S.; Bonadies, L.; Castagliulo, I.; Morelli, L.; Verlato, G.; Baraldi, E. Neonatal sepsis associated with Lactobacillus supplementation. J. Perinat. Med. 2019, 48, 87–88. [Google Scholar] [CrossRef] [PubMed]
- Vahabnezhad, E.; Mochon, A.B.; Wozniak, L.J.; Ziring, D.A. Lactobacillus bacteremia associated with probiotic use in a pediatric patient with ulcerative colitis. J. Clin. Gastroenterol. 2013, 47, 437–439. [Google Scholar] [CrossRef] [PubMed]
- Brecht, M.; Garg, A.; Longstaff, K.; Cooper, C.; Andersen, C. Lactobacillus sepsis following a laparotomy in a preterm infant: A note of caution. Neonatology 2016, 109, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Chiang, M.C.; Chen, C.L.; Feng, Y.; Chen, C.C.; Lien, R.; Chiu, C.H. Lactobacillus rhamnosus Sepsis Associated with Probiotic Therapy in an Extremely Preterm Infant: Pathogenesis and a Review for Clinicians. J. Microbiol. Immunol. Infect. 2020, 54, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Doern, C.D.; Nguyen, S.R.; Afolabi, F.; Burnham, C.A. Probiotic-associated aspiration pneumonia due to Lactobacillus rhamnosus. J. Clin. Microbiol. 2014, 52, 3124–3126. [Google Scholar] [CrossRef] [PubMed]
- Molinaro, M.; Aiazzi, M.; La Torre, A.; Cini, E.; Banfi, R. Lactobacillus Rhamnosus sepsis in a preterm infant associated with probiotic integrator use: A case report. Recenti. Prog. Med. 2016, 107, 485–486. [Google Scholar]
- Celis Castañeda, L.A.; Morales Camacho, W.J.; Durán Ochoa, N.M. Sepsis due to Lactobacillus reuteri in an extreme preterm newborn: Case report. Arch. Argent. Pediatr. 2019, 117, e509–e513. [Google Scholar]
- Viggiano, M.; Badetti, C.; Bernini, V.; Garabedian, M.; Manelli, J.C. Saccharomyces boulardii fungemia in a patient with severe burns. Ann. Fr. Anesth Reanim 1995, 14, 356–358. [Google Scholar] [CrossRef]
- Pletincx, M.; Legein, J.; Vandenplas, Y. Fungemia with Saccharomyces boulardii in a 1-year-old girl with protracted diarrhea. J. Pediatr. Gastroenterol. Nutr. 1995, 21, 113–115. [Google Scholar] [CrossRef]
- Chioukh, F.Z.; Ben Hmida, H.; Ben Ameur, K.; Toumi, A.; Monastiri, K. Septicémie à Saccharomyces cerevisiae chez un prématuré traité par Ultra-Levure® [Saccharomyces cerevisiae fungemia in a premature neonate treated receiving probiotics]. Med. Mal. Infect. 2013, 43, 359–360. [Google Scholar] [CrossRef]
- Roy, U.; Jessani, L.G.; Rudramurthy, S.M.; Gopalakrishnan, R.; Dutta, S.; Chakravarty, C.; Jillwin, J.; Chakrabarti, A. Seven cases of Saccharomyces fungaemia related to use of probiotics. Mycoses 2017, 60, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Cesaro, S.; Chinello, P.; Rossi, L.; Zanesco, L. Saccharomyces cerevisiae fungemia in a neutropenic patient treated with Saccharomyces boulardii. Support. Care Cancer 2000, 8, 504–505. [Google Scholar] [CrossRef] [PubMed]
- Lungarotti, M.; Mezzetti, D.; Radicioni, M. Methaemoglobinaemia with concurrent blood isolation of Saccharomyces and Candida. Arch. Dis. Child. Fetal. Neonatal. Ed. 2003, 88, F446. [Google Scholar] [CrossRef] [PubMed]
- Chakravarty, S.; Parashar, A.; Acharyya, S. Saccharomyces cerevisiae Sepsis Following Probiotic Therapy in an Infant. Indian Pediatrics 2019, 56, 971–972. [Google Scholar] [CrossRef] [PubMed]
- Atıcı, S.; Soysal, A.; Karadeniz Cerit, K.; Yılmaz, Ş.; Aksu, B.; Kıyan, G.; Bakır, M. Catheter-related Saccharomyces cerevisiae Fungemia Following Saccharomyces boulardii Probiotic Treatment: In a child in intensive care unit and review of the literature. Med. Mycol. Case Rep. 2017, 15, 33–35. [Google Scholar] [CrossRef] [PubMed]
- Belet, N.; Dalgc¸, N.; Oncel, S.; Ciftçi, E.; Ince, E.; Güriz, H.; Barlas, M.; Doğru, U. Catheter-related fungemia caused by Saccharomyces cerevisiae in a newborn. Pediatr. Infect. Dis. J. 2005, 24, 1125. [Google Scholar] [CrossRef][Green Version]
- Romanio, M.R.; Coraine, L.A.; Maielo, V.P.; Abramczyc, M.L.; de Souza, R.L.; Oliveira, N.F. Saccharomyces cerevisiae fungemia in a pediatric patient after treatment with probiotics. Rev. Paul. Pediatr. 2017, 35, 361–364. [Google Scholar] [CrossRef]
- Hennequin, C.; Kauffmann-Lacroix, C.; Jobert, A.; Viard, J.P.; Ricour, C.; Jacquemin, J.L.; Berche, P. Possible role of catheters in Saccharomyces boulardii fungemia. Eur. J. Clin. Microbiol. Infect. Dis. 2000, 19, 16–20. [Google Scholar] [CrossRef]
- Perapoch, J.; Planes, A.; Querol, A.; Tormo, R.; Peguero, G.; Salcedo, S. Fungemia with Saccharomyces cerevisiae in two newborns, only one of whom had been treated with ultra-levura. Eur. J. Clin. Microbiol. Infect. Dis. 2000, 19, 468–470. [Google Scholar] [CrossRef]
- Bertelli, C.; Pillonel, T.; Torregrossa, A.; Prod’hom, G.; Fischer, C.J.; Greub, G.; Giannoni, E. Bifidobacterium longum bacteremia in preterm infants receiving probiotics. Clin. Infect. Dis. 2015, 60, 924–927. [Google Scholar] [CrossRef]
- Zbinden, A.; Zbinden, R.; Berger, C.; Arlettaz, R. Case series of Bifidobacterium longum bacteremia in three preterm infants on probiotic therapy. Neonatology 2015, 107, 56–59. [Google Scholar] [CrossRef]
- Esaiassen, E.; Hjerde, E.; Cavanagh, J.P.; Simonsen, G.S.; Klingenberg, C. Norwegian Study Group on Invasive Bifidobacterial I. Bifidobacterium Bacteremia: Clinical characteristics and a genomic approach to assess pathogenicity. J. Clin. Microbiol. 2017, 55, 2234–2248. [Google Scholar] [CrossRef]
- Jenke, A.; Ruf, E.M.; Hoppe, T.; Heldmann, M.; Wirth, S. Bifidobacterium septicaemia in an extremely low-birthweight infant under probiotic therapy. Arch. Dis. Child. Fetal. Neonatal. Ed. 2012, 97, F217–F218. [Google Scholar] [CrossRef]
- Pruccoli, G.; Silvestro, E.; Pace Napoleone, C.; Aidala, E.; Garazzino, S.; Scolfaro, C. Are probiotics safe? Bifidobacterium bacteremia in a child with severe heart failure. Infez. Med. 2019, 27, 175–178. [Google Scholar]
- Sato, S.; Uchida, T.; Kuwana, S.; Sasaki, K.; Watanabe, T.; Saito, J.; Kawaji, T. Bacteremia induced by Bifidobacterium breve in a newborn with cloacal exstrophy. Pediatr. Int. 2016, 58, 1226–1228. [Google Scholar] [CrossRef]
- Avcin, S.L.; Pokorn, M.P.; Kitanovski, L.; Premru, M.M.; Jazbec, J. Bifidobacterium breve sepsis in child with high-risk acute lymphoblastic leukemia. Emerg. Infect. Dis. 2015, 21, 1674–1675. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, H.; Sumida, Y.; Tanaka, R.; Yuki, N.; Takayama, H.; Fujimura, M. Early administration of Bifidobacterium breve to preterm infants: Randomised controlled trial. Arch. Dis. Child. Fetal. Neonatal. Ed. 1997, 76, F101–F107. [Google Scholar] [CrossRef] [PubMed]
- Ohishi, A.; Takahashi, S.; Ito, Y.; Ohishi, Y.; Tsukamoto, K.; Nanba, Y.; Ito, N.; Kakiuchi, S.; Saitoh, A.; Morotomi, M.; et al. Bifidobacterium septicemia associated with postoperative probiotic therapy in a neonate with omphalocele. J. Pediatr. 2011, 156, 679–681. [Google Scholar] [CrossRef] [PubMed]
- Khatri, A.M.; Rai, S.; Shank, C.; McInerney, A.; Kaplan, B.; Hagmann, S.H.F.; Kainth, M.K. A tale of caution: Prolonged Bacillus clausii bacteraemia after probiotic use in an immunocompetent child. Access Microbiol. 2021, 3, 000205. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Udani, S.; Sen, S.; Kirolikar, S.; Shetty, A. Bacillus Clausii septicemia in a pediatric patient after treatment with probiotics. Pediatr. Infect. Dis. J. 2019, 38, e228–e230. [Google Scholar] [CrossRef] [PubMed]
- Guenther, K.; Straube, E.; Pfister, W.; Guenther, A.; Huebler, A. Severe sepsis after probiotic treatment with Escherichia coli NISSLE 1917. Pediatr. Infect. Dis. J. 2010, 29, 188–189. [Google Scholar] [CrossRef]
- Borriello, S.P.; Hammes, W.P.; Holzapfel, W.; Marteau, P.; Schrezenmeir, J.; Vaara, M.; Valtonen, V. Safety of probiotics that contain lactobacilli or bifidobacteria. Clin. Infect. Dis. 2003, 36, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Manzoni, P.; Lista, G.; Gallo, E.; Marangione, P.; Priolo, C.; Fontana, P.; Guardione, R.; Farina, D. Routine Lactobacillus rhamnosus GG administration in VLBW infants: A retrospective, 6-year cohort study. Early Hum. Dev. 2011, 87 (Suppl. 1), S35–S38. [Google Scholar] [CrossRef] [PubMed]
- Melville, J.M.; Moss, T.J. The immune consequences of preterm birth. Front. Neurosci. 2013, 7, 79. [Google Scholar] [CrossRef] [PubMed]
- Liong, M.T. Safety of probiotics: Translocation and infection. Nutr. Rev. 2008, 66, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Berg, R.D. Bacterial translocation from the gastrointestinal tract. J. Med. 1992, 23, 217–244. [Google Scholar]
- Kolaček, S.; Hojsak, I.; Berni Canani, R.; Guarino, A.; Indrio, F.; Orel, R.; Pot, B.; Shamir, R.; Szajewska, H.; Vandenplas, Y.; et al. ESPGHAN working Group for Probiotics and Prebiotics. Commercial probiotic products: A call for improved quality control. A position paper by the ESPGHAN working Group for Probiotics and Prebiotics. J. Pediatr. Gastroenterol. Nutr. 2017, 65, 117–124. [Google Scholar] [CrossRef]
- Sanders, M.E.; Shane, A.L.; Merenstein, D.J. Advancing probiotic research in humans in the United States: Challenges and strategies. Gut Microbes 2016, 7, 97–100. [Google Scholar] [CrossRef]
- Theunissen, J.; Britz, T.J.; Torriani, S.; Witthuhn, R.C. Identification of probiotic microorganisms in South African products using PCR-based DGGE analysis. Int. J. Food Microbiol. 2005, 98, 11–21. [Google Scholar] [CrossRef]
| Ref. | Etiologic Agent * | Infection Type | Sex, Age | Underlying Condition(s) | Lenght of Probiotic Intake (Days) | Treatment (Days) | Outcome |
|---|---|---|---|---|---|---|---|
| [27] | L. rhamnosus GG | Sepsis | M, 4 months | Prematurity (36 weeks), short bowel syndrome, gastrostomy, cholestasis, chronic intestinal inflammation | 23 | cro+ amp (10) | Favourable |
| [27] | L. rhamnosus GG (ATCC53103) | Sepsis | M, 6 months | Prematurity (34 weeks), gastroschisis, short bowel syndrome, TPN, cholestasis, chronic intestinal inflammation | 169 | cro+ amp (10) | Favourable |
| [28] | L. rhamnosus GG (ATCC53103) | Sepsis | F, 6 years | Cerebral palsy, epilepsy, jejunostomy feeding, antibiotic-associated diarrhoea, CVC | 44 | NA (10) | Favourable |
| [28] | L. rhamnosus GG (ATCC53103) | Sepsis | M, 6 weeks | CHD, cardiac surgery, epilepsy, AKI, respiratory support, antibiotic-associated diarrhoea, CVC | 20 | pen G+ gen (24) | Favourable |
| [29] | L. rhamnosus GG (ATCC53103) | Sepsis | F, 3 months | Trisomy 18 and triple-X syndromes, CHD, respiratory support, CVC | 88 | cli (10) | Favourable |
| [29] | L. rhamnosus GG (ATCC53103) | Sepsis | M, 18 days | Prematurity (23 weeks), non-invasive respiratory support, CVC | 16 | gen (10) | Favourable |
| [30] | L. rhamnosus GG (ATCC53103) | Sepsis | M, 11 months | Prematurity (26 weeks), short bowel syndrome, cholestasis, cirrhosis, hypothyroidism, megaloblastic anaemia, CLD of infancy, CVC | 35 | amp+ gen (7) | Favourable |
| [31] | L. rhamnosus GG (ATCC53103) | Sepsis | M, 6 days | IUGR | 4 | tic+ca (14) | Favourable |
| [32] | L. rhamnosus GG (ATCC53103) | Sepsis | F, 18 days | Prematurity (25 weeks) | 15 | amp (17) | Favourable |
| [32] | L. rhamnosus GG (ATCC53103) | Sepsis | M, NA | Prematurity, CVC | neighbour | amp | Favourable |
| [32] | L. rhamnosus GG (ATCC53103) | Sepsis | M, NA | Prematurity, CVC | neighbour | amp | Favourable |
| [33] | L. rhamnosus GG (ATCC53103) | Bacteremia | M, 17 years | UC, concurrent enteric infection, immunosuppressive treatment, C. difficile colitis | 5 | tzp+ gen (5) | Favourable |
| [34] | L. rhamnosus GG (ATCC53103) | Sepsis | M, 2 months | Prematurity (25 weeks), spontaneous intestinal perforation, ileostomy | 45 | pen G (10) | Favourable |
| [35] | L. rhamnosus GG (ATCC53103) | Sepsis | F, 26 days | Prematurity (26 weeks), CVC | 12 | amp+ tzp (10) | Favourable |
| [36] | L. rhamnosus GG (ATCC53103) | Pneumonia | F, 11 months | Trisomy 21, esophageal surgery, gastrostomy, RSV infection | 90 | sam (10) | Favourable |
| [37] | L. rhamnosus GG (ATCC53103) | Sepsis | NA, 20 days | Prematurity (23 weeks) | 19 | NA | Favourable |
| [38] | L. reuteri (ATCC55730) | Sepsis | M, 2 days | Prematurity (27 weeks), respiratory support, UC | 2 | NA | death |
| Ref. | Etiologic Agent | Infection Type | Sex, Age | Underlying Condition(s) | Lenght of Probiotic Intake (Days) | Treatment (Days) | Outcome |
|---|---|---|---|---|---|---|---|
| [39] | S. boulardii | Sepsis | M, 14 years | Burn, CVC | 7 | fc+ amB | favourable |
| [40] | S. boulardii | Fungemia | F, 1 year | Gastroenteritis, malnutrition, CVC | 13 | flz | favourable |
| [41] | S. cerevisiae | Sepsis | F, 17 days | Prematurity (26 weeks), CVC | 1 | NA | favourable |
| [42] | S. boulardii | Sepsis | M, NA | Prematurity (27 weeks), UVC, TPN | NA | mica | death |
| [42] | S. boulardii | Sepsis | M, NA | Prematurity (31 weeks), late-onset sepsis | NA | mica (14) | favourable |
| [43] | S. cerevisiae | Fungemia | NA, 8 months | AML, chemotherapy, neutropenia, CVC | 1 | L-amB (14) | favourable |
| [44] | S. cerevisiae | Sepsis | M, 3 weeks | Prematurity (30 weeks, IUGR) | 4 | NA (14) | favourable |
| [45] | S. cerevisiae | Sepsis | M, 3.5 months | Undiagnosed combined immunodeficiency | NA | amB (20) | favourable |
| [46] | S. cerevisiae | Sepsis | M, 8 years | Cerebral palsy, gastrostomy, aspiration pneumonia, CLD, CVC | NA | amB (14) | favourable |
| [47] | S. cerevisiae | Sepsis | F, 32 days | Esophageal atresia, tracheoesophageal fistula, CVC | neighbour | amB (21) | favourable |
| [48] | S. cerevisiae | Sepsis | M, 1 year | Trisomy 21, cardiac surgery, respiratory support, tracheostomy, dialysis catheter, malnutrition, CVC | 4 | amB (15) | favourable |
| [49] | S. boulardii | Sepsis | M, 30 months | Ileal atresia, small bowel resection, cystic fibrosis, malnutrition, TPN, CVC | 300 | amB (21) | favourable |
| [50] | S. cerevisiae | Fungemia | M, 3 months | CHD, TPN, CVC | 10 | L-amB | favourable |
| [50] | S. cerevisiae | Fungemia | F, 1 month | Intestinal atresia, small bowel resection, TPN, CVC | neighbour | NA | favourable |
| Ref. | Etiologic Agent * | Infection Type | Sex, Age | Underlying Condition(s) | Lenght of Probiotic Intake (Days) | Treatment (DAYs) | Outcome |
|---|---|---|---|---|---|---|---|
| [51] | B. longum infantis BIC 1206122787 | Sepsis | F, 14 days | Prematurity (26 weeks), intussusception | 9 | caz+van (7), then ipm (7) | favourable |
| [51] | B. longum infantis BIC 140111125 | Sepsis | F, 10 days | Prematurity (28 weeks), NEC | 4 | caz+amk+mtz | favourable |
| [52] | B. longum | Bacteriemia | F, 20 days | Prematurity (30 weeks), respiratory support, periumbilical infection (Staphylococcus aureus) | 19 | flx+gen (2) | favourable |
| [52] | B. longum | Bacteriemia | M, 20 days | Prematurity (28 weeks), respiratory support, BPD, UVC | 14 | amx+gen (2) | favourable |
| [52] | B. longum | Bacteriemia | F, 11 days | Prematurity (29 weeks), respiratory support, NEC | 10 | amc+gen | favourable |
| [53] | B. longum infantis ATCC15697 | Sepsis | M, 2 weeks | Prematurity (23 weeks), spontaneous intestinal perforation | NA | ctx+gen | favourable |
| [53] | B. longum infantis ATCC15697 | Sepsis | F, 5 weeks | Prematurity (24 weeks), NEC | NA | amp+gen+mtz | favourable |
| [53] | B. longum infantis ATCC15697 | Bacteremia | M, 2 weeks | Prematurity (24 weeks) | NA | none | favourable |
| [54] | B. longum infantis ATCC15697 | Sepsis | NA, 18 days | Prematurity (27 weeks) | 9 | ctx+van+mtz | favourable |
| [55] | B. spp | Bacteriemia | NA, 15 months | Heart disease (dilated cardiomyopathy, valvopathies, heart failure), ECMO, CVC | 8 | van+mem (7) | favourable |
| [56] | B. breve BBG-01 | Bacteriemia | M, 8 days | Prematurity (36 weeks), cloacal exstrophy, omphalocele, imperforate anus, cystourethroplasty and colostomy, resection of the small intestine | 8 | cez+van | favourable |
| [57] | B. breve | Sepsis | M, 2 years | Acute lymphoblastic leukaemia, chemotherapy | NA | tpz+van+gen, then pen | favourable |
| [58] | B. breve BBG-01 | Bacteriemia | F, 10 days | IUGR, abdominal surgery for omphalocele | NA | NA | favourable |
| [58] | B. breve BBG-01 | Bacteriemia | M, 23 days | Preterm, Trisomy 21, Hirschsprung disease | NA | NA | favourable |
| [59] | B. breve BBG-01 | Sepsis | M, 10 days | Prematurity (37 weeks), abdominal surgery for omphalocele, CVC | 8 | sam (2), amk (8), mem (10) | favourable |
| Ref. | Etiologic Agent * | Infection Type | Sex, Age | Underlying Condition(s) | Lenght of Probiotic Intake (Days) | Treatment (DAYs) | Outcome |
|---|---|---|---|---|---|---|---|
| [60] | Bacillus clausii | Bacteremia | F, 17 months | No comorbidity, immunocompetent | 4 | amp, then lvx + gen, then van+ gen | favourable |
| [61] | Bacillus clausii | Sepsis | M, 5 months | Surgically corrected CHD | 58 | van (21) then mem+ cst | death |
| [62] | E. coli NISSLE 1917 | Sepsis | NA 25 days | Prematurity (28 weeks) | 10 | mem+ van+ IVIg | favourable |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Agostin, M.; Squillaci, D.; Lazzerini, M.; Barbi, E.; Wijers, L.; Da Lozzo, P. Invasive Infections Associated with the Use of Probiotics in Children: A Systematic Review. Children 2021, 8, 924. https://doi.org/10.3390/children8100924
D’Agostin M, Squillaci D, Lazzerini M, Barbi E, Wijers L, Da Lozzo P. Invasive Infections Associated with the Use of Probiotics in Children: A Systematic Review. Children. 2021; 8(10):924. https://doi.org/10.3390/children8100924
Chicago/Turabian StyleD’Agostin, Martina, Domenica Squillaci, Marzia Lazzerini, Egidio Barbi, Lotte Wijers, and Prisca Da Lozzo. 2021. "Invasive Infections Associated with the Use of Probiotics in Children: A Systematic Review" Children 8, no. 10: 924. https://doi.org/10.3390/children8100924
APA StyleD’Agostin, M., Squillaci, D., Lazzerini, M., Barbi, E., Wijers, L., & Da Lozzo, P. (2021). Invasive Infections Associated with the Use of Probiotics in Children: A Systematic Review. Children, 8(10), 924. https://doi.org/10.3390/children8100924