Video Modeling and Social Skills Learning in ASD-HF
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Instruments
2.3. Procedures and Tasks
3. Methods
4. Results
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5®); Psychiatric Association: Arlington, VA, USA, 2013; pp. 1–947. [Google Scholar]
- Wang, S.-Y.; Cui, Y.; Parrila, R. Examining the effectiveness of peer-mediated and video-modeling social skills interventions for children with autism spectrum disorders: A meta-analysis in single-case research using HLM. Res. Autism Spectr. Disord. 2011, 5, 562–569. [Google Scholar] [CrossRef]
- Lawrence, D.H.; Alleckson, D.A.; Bjorklund, P. Beyond the Roadblocks: Transitioning to Adulthood With Asperger’s Disorder. Arch. Psychiatr. Nurs. 2010, 24, 227–238. [Google Scholar] [CrossRef]
- Bass, J.D.; Mulick, J.A. Social play skill enhancement of children with autism using peers and siblings as therapists. Psychol. Sch. 2007, 44, 727–735. [Google Scholar] [CrossRef]
- Matson, J.L.; Matson, M.L.; Rivet, T.T. Social-skills treatments for children with autism spectrum disorders: An overview. Behav. Modif. 2007, 31, 682–707. [Google Scholar] [CrossRef] [PubMed]
- Nikopoulos, C.; Keenan, M. Effects of video modeling on social initiations by children with autism. J. Appl. Behav. Anal. 2004, 37, 93–96. [Google Scholar] [CrossRef]
- Scattone, D. Social skills interventions for children with autism. Psychol. Sch. 2007, 44, 717–726. [Google Scholar] [CrossRef]
- Rayner, C.; Denholm, C.; Sigafoos, J. Video-based intervention for individuals with autism: Key questions that remain unanswered. Res. Autism Spectr. Disord. 2009, 3, 291–303. [Google Scholar] [CrossRef]
- Charlop, M.H.; Milstein, J.P. Teaching Autistic Children Conversational Speech Using Video Modeling. J. Appl. Behav. Anal. 1989, 22, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Sherer, M.; Pierce, K.L.; Paredes, S.; Kisacky, K.L.; Ingersoll, B.; Schreibman, L. Enhancing conversation skills in children with autism via video technology: Which is better,“self” or “other” as a model? Behav. Modif. 2001, 25, 140–158. [Google Scholar] [CrossRef]
- Apple, A.; Billingsley, F.; Schwartz, I. Effects of video modeling alone and with self-management on compliment-giving behaviours of children with highfunctioning ASD. J. Posit. Behav. Interv. 2005, 7, 33–46. [Google Scholar] [CrossRef]
- Gena, A.; Couloura, S.; Kymissis, E. Modifying the affective behaviour of preschoolers with autism using in-vivo or video modeling and reinforcement contingencies. J. Autism Dev. Disord. 2005, 35, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Nikopoulos, C.K.; Keenan, M. Promoting social initiation in children with autism using video modeling. Behav. Interv. 2003, 18, 87–108. [Google Scholar] [CrossRef]
- Wert, B.Y.; Neisworth, J.T. Effects of Video Self-Modeling on Spontaneous Requesting in Children with Autism. J. Posit. Behav. Interv. 2003, 5, 30–34. [Google Scholar] [CrossRef]
- Buggey, T. Video Self-Modeling Applications With Students With Autism Spectrum Disorder in a Small Private School Setting. Focus Autism Other Dev. Disabil. 2005, 20, 52–63. [Google Scholar] [CrossRef]
- Bellini, S.; Akullian, J. A Meta-Analysis of Video Modeling and Video Self-Modeling Interventions for Children and Adolescents with Autism Spectrum Disorders. Except. Child. 2007, 73, 264–287. [Google Scholar] [CrossRef]
- Hayes, G.R.; Custodio, V.E.; Haimson, O.L.; Nguyen, K.; Ringland, K.E.; Ulgado, R.R.; Waterhouse, A.; Weiner, R. Mobile video modeling for employment interviews for individuals with autism. J. Vocat. Rehabilitation 2015, 43, 275–287. [Google Scholar] [CrossRef]
- Williams, J.H.; Whiten, A.; Singh, T. A Systematic Review of Action Imitation in Autistic Spectrum Disorder. J. Autism Dev. Disord. 2004, 34, 285–299. [Google Scholar] [CrossRef]
- Baron-Cohen, S.; Belmonte, M.K. AUTISM: A Window Onto the Development of the Social and the Analytic Brain. Annu. Rev. Neurosci. 2005, 28, 109–126. [Google Scholar] [CrossRef]
- Gallese, V.; Migone, P.; Eagle, M.N. La simulazione incarnata: I neuroni specchio, le basi neurofisiologiche dell’intersoggettività ed alcune implicazioni per la psicoanalisi. Psicoterapia e Scienze Umane. 2006, 40, 543–580. [Google Scholar]
- Cattaneo, L.; Fabbri-Destro, M.; Boria, S.; Pieraccini, C.; Monti, A.; Cossu, G.; Rizzolatti, G. Impairment of actions chains in autism and its possible role in intention understanding. Proc. Natl. Acad. Sci. USA 2007, 104, 17825–17830. [Google Scholar] [CrossRef]
- Frolli, A.; ILa Penna Cavallaro, A.; Ricci, M.C. Theory of Mind: Autism and Typical Developmental. Acad. J. Ped. Neonatol. 2019, 8, 555799. [Google Scholar] [CrossRef]
- Orsini, A.; Pezzuti, L.; Picone, L. WISC-IV: Contributo alla taratura Italiana; Giunti O. S. Organizzazioni Specialib: Florence, Italy, 2012; pp. 1–140. [Google Scholar]
- IBM to Acquire SPSS Inc. to Provide Clients Predictive Analytics Capabilities. Available online: https://newsroom.ibm.com/2009-07-28-IBM-to-Acquire-SPSS-Inc-to-Provide-Clients-Predictive-Analytics-Capabilities (accessed on 28 August 2020).
- Wong, C.; Odom, S.L.; Hume, K.A.; Cox, A.W.; Fettig, A.; Kucharczyk, S.; Brock, M.E.; Plavnick, J.B.; Fleury, V.P.; Schultz, T.R. Evidence-Based Practices for Children, Youth, and Young Adults with Autism Spectrum Disorder: A Comprehensive Review. J. Autism Dev. Disord. 2015, 45, 1951–1966. [Google Scholar] [CrossRef] [PubMed]
- Delano, M. Video Modeling Interventions for Individuals with Autism. Remedial Spéc. Educ. 2007, 28, 33–42. [Google Scholar] [CrossRef]
- Wang, P.; Spillane, A. Evidence-based social skills interventions for children with autism: A meta-analysis. Educ. Train. Dev. Disabil. 2009, 44, 318–342. [Google Scholar]
- McCoy, K.; Hermansen, E. Video Modeling for Individuals with Autism: A Review of Model Types and Effects. Educ. Treat. Child. 2007, 30, 183–213. [Google Scholar] [CrossRef]
- Chartrand, T.L.; van Baaren, R. Human mimicry. Adv. Exp. Soc. Psychol. 2009, 41, 219–274. [Google Scholar]
- Heyes, C.M. What’s social about social learning? J. Comp. Psychol. 2012, 126, 193–202. [Google Scholar] [CrossRef]
- Van Baaren, R.; Janssen, L.; Chartrand, T.L.; Dijksterhuis, A. Where is the love? The social aspects of mimicry. Philos. Trans. R. Soc. B: Biol. Sci. 2009, 364, 2381–2389. [Google Scholar] [CrossRef] [PubMed]
- Rizzolatti, G.; Fadiga, L.; Gallese, V.; Fogassi, L. Premotor cortex and the recognition of motor actions. Cogn. Brain Res. 1996, 3, 131–141. [Google Scholar] [CrossRef]
- Rizzolatti, G.; Craighero, L. THE MIRROR-NEURON SYSTEM. Annu. Rev. Neurosci. 2004, 27, 169–192. [Google Scholar] [CrossRef]
- Iacoboni, M.; Dapretto, M. The mirror neuron system and the consequences of its dysfunction. Nat. Rev. Neurosci. 2006, 7, 942–951. [Google Scholar] [CrossRef] [PubMed]
- Cattaneo, L.; Rizzolatti, G. The Mirror Neuron System. Arch. Neurol. 2009, 66, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Rizzolatti, G.; Fabbri-Destro, M.; Cattaneo, L. Mirror neurons and their clinical relevance. Nat. Clin. Pr. Neurol. 2009, 5, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Rizzolatti, G.; Sinigaglia, C. The functional role of the parieto-frontal mirror circuit: Interpretations and misinterpretations. Nat. Rev. Neurosci. 2010, 11, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Perkins, T.; Stokes, M.; McGillivray, J.; Bittar, R. Mirror neuron dysfunction in autism spectrum disorders. J. Clin. Neurosci. 2010, 17, 1239–1243. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, V.S.; Oberman, L.M. Broken Mirrors: A Theory of Autism. Sci. Am. 2006, 295, 62–69. [Google Scholar] [CrossRef]
- Raymaekers, R.; Wiersema, J.R.; Roeyers, H. EEG study of the mirror neuron system in children with high functioning autism. Brain Res. 2009, 1304, 113–121. [Google Scholar] [CrossRef]
- Rizzolatti, G.; Fabbri-Destro, M. Mirror neurons: From discovery to autism. Exp. Brain Res. 2010, 200, 223–237. [Google Scholar] [CrossRef] [PubMed]
- Iacoboni, M.; Molnar-Szakacs, I.; Gallese, V.; Buccino, G.; Mazziotta, J.C.; Rizzolatti, G. Grasping the intentions of others with one’s own mirror neuron system. PLoS Biol. 2005, 3, e79. [Google Scholar] [CrossRef] [PubMed]
- Gowen, E.; Poliakoff, E. How does visuomotor priming differ for biological and non-biological stimuli? A review of the evidence. Psychol. Res. 2012, 76, 407–420. [Google Scholar] [CrossRef]
- Klapper, A.; Ramsey, R.; Wigboldus, D.; Cross, E.S. The Control of Automatic Imitation Based on Bottom–Up and Top–Down Cues to Animacy: Insights from Brain and Behavior. J. Cogn. Neurosci. 2014, 26, 2503–2513. [Google Scholar] [CrossRef] [PubMed]
- Brass, M.; Heyes, C. Imitation: Is cognitive neuroscience solving the correspondence problem? Trends Cogn. Sci. 2005, 9, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Liepelt, R.; Brass, M. Top-Down Modulation of Motor Priming by Belief About Animacy. Exp. Psychol. 2010, 57, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Longo, M.R.; Bertenthal, B.I. Attention modulates the specificity of automatic imitation to human actors. Exp. Brain Res. 2008, 192, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.C.; Brass, M. Does the human motor system simulate Pinocchio’s actions? Coacting with a human hand versus a wooden hand in a dyadic interaction. Psychol. Sci. 2007, 18, 1058–1062. [Google Scholar] [CrossRef]
- Oberman, L.M.; Ramachandran, V.S.; Pineda, J.A. Modulation of mu suppression in children with autism spectrum disorders in response to familiar or unfamiliar stimuli: The mirror neuron hypothesis. Neuropsychologia 2008, 46, 1558–1565. [Google Scholar] [CrossRef]
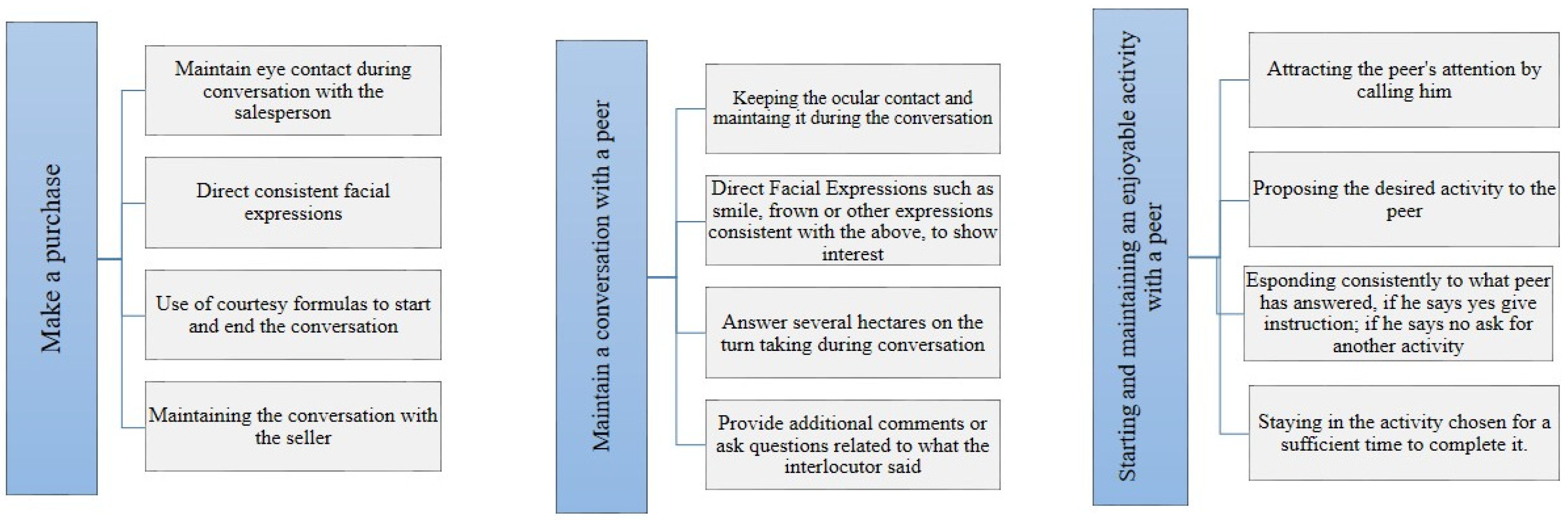

| Maintain a Conversation with a Peer | |
|---|---|
| (1) Keep ocular contact during the conversation | 0 = The subject does not maintain eye contact during the conversation 1 = The subject uses eye contact but in an inflexible way or in a limited number of exchanges 2 = The subject uses eye contact flexibly and during all conversational exchanges |
| (2) Direct facial expressions, such as a smile, frown, or other expressions consistent with the above, to show interest | 0 = The subject does not express interest in what was said in the conversation through facial expressions 1 = The subject directs other facial expressions in a limited or exaggerated way or uses only some facial expressions (e.g., smiles in hands is contextual, but does not use expressions to express disappointment, sadness, etc.) 2 = The subject uses a wide range of facial expressions in a flexible and coherent way with the speech |
| (3) Answer several hectares on the turn taking during conversation | 0 = The subject is unable to respect the conversational rules, for example speaking while the other is completing the speech or responding only after long pauses 1 = The subject occasionally uses non-verbal elements that allow him to understand conversational exchanges 2 = The subject can read the main forms of non-verbal communication to regulate conversational shifts |
| (4) Provide additional comments or ask questions related to what the interlocutor said | 0 = The subject does not continue with comments or questions what the interlocutor said 1 = The subject provides additional comments on an occasional basis or not always in context with what was said by the interlocutor 2 = The subject provides additional comments or shows interest in what the interlocutor said with coherent questions. |
| Interacting with a salesperson while making a purchase |
|
| Initiating and maintaining a conversation with peers |
|
| Starting and maintaining an enjoyable activity with a peer |
|
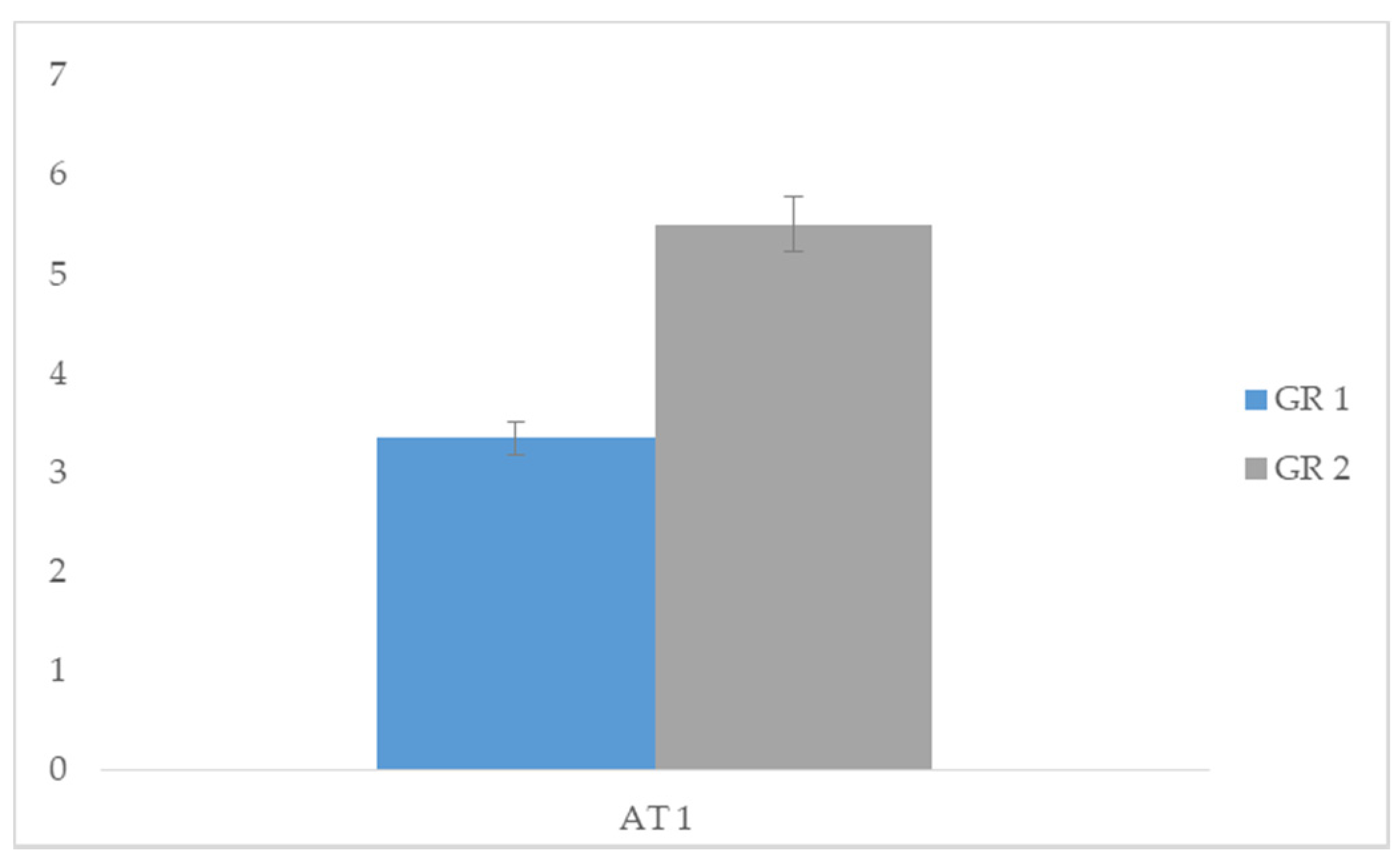
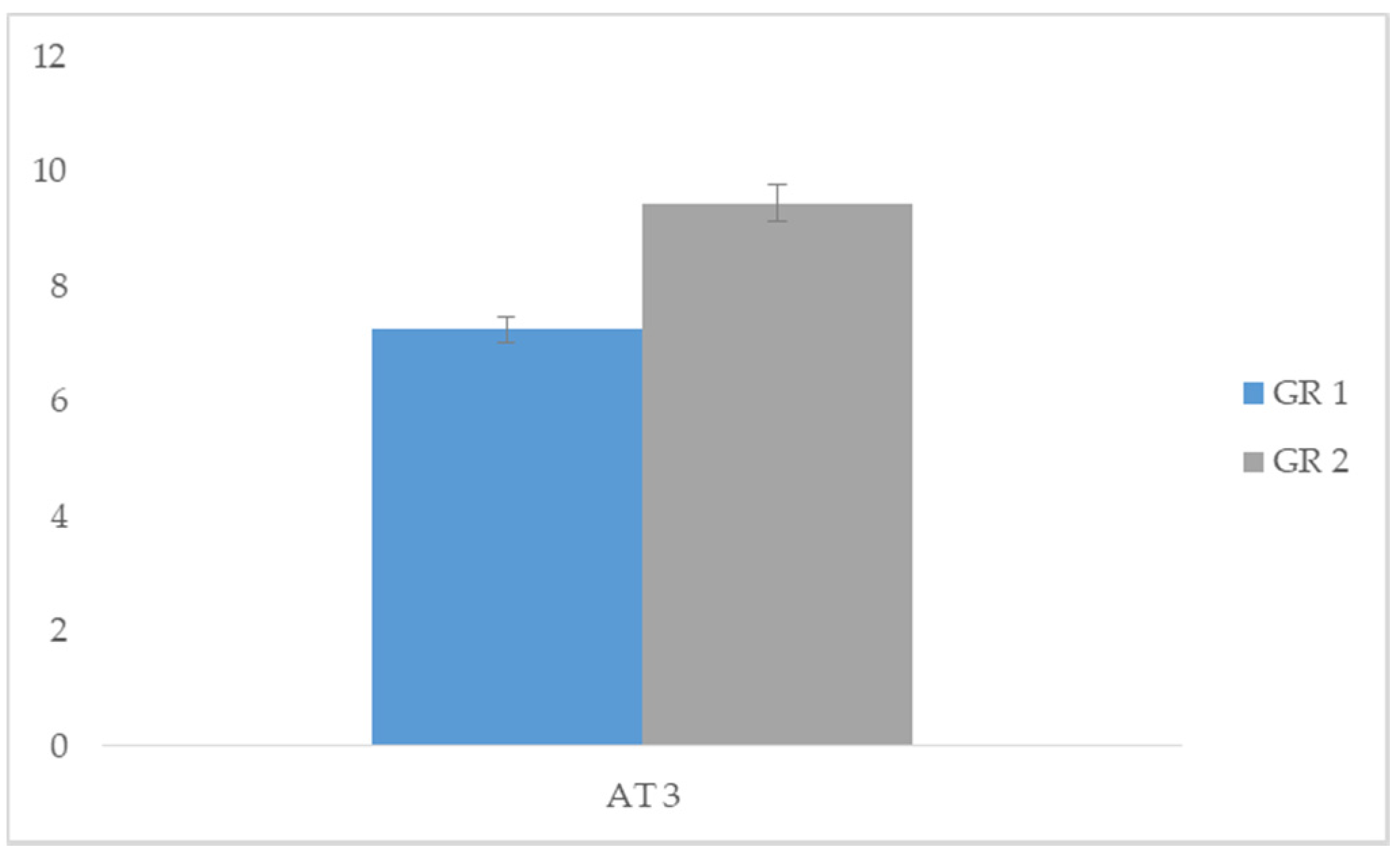
| Group 1 | Group 2 | |||||
|---|---|---|---|---|---|---|
| Means | SD | Means | SD | t | p | |
| AT 1 | 3.35 | 0.489 | 5.50 | 0.513 | −11.831 | 0.000 * |
| AT 2 | 5.35 | 0.489 | 7.40 | 0.503 | −11.105 | 0.000 * |
| AT 3 | 7.25 | 0.444 | 9.45 | 0.510 | −14.139 | 0.000 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frolli, A.; Ricci, M.C.; Bosco, A.; Lombardi, A.; Cavallaro, A.; Operto, F.F.; Rega, A. Video Modeling and Social Skills Learning in ASD-HF. Children 2020, 7, 279. https://doi.org/10.3390/children7120279
Frolli A, Ricci MC, Bosco A, Lombardi A, Cavallaro A, Operto FF, Rega A. Video Modeling and Social Skills Learning in ASD-HF. Children. 2020; 7(12):279. https://doi.org/10.3390/children7120279
Chicago/Turabian StyleFrolli, Alessandro, Maria Carla Ricci, Antonia Bosco, Agnese Lombardi, Antonella Cavallaro, Francesca Felicia Operto, and Angelo Rega. 2020. "Video Modeling and Social Skills Learning in ASD-HF" Children 7, no. 12: 279. https://doi.org/10.3390/children7120279
APA StyleFrolli, A., Ricci, M. C., Bosco, A., Lombardi, A., Cavallaro, A., Operto, F. F., & Rega, A. (2020). Video Modeling and Social Skills Learning in ASD-HF. Children, 7(12), 279. https://doi.org/10.3390/children7120279







