Endocrine Disruptors and Autism Spectrum Disorder in Pregnancy: A Review and Evaluation of the Quality of the Epidemiological Evidence
Abstract
1. Introduction
1.1. Environmental Factors and ASD Causation
1.2. EDCs and Human Exposure
1.3. Prenatal EDC Exposure and Risk for ASD
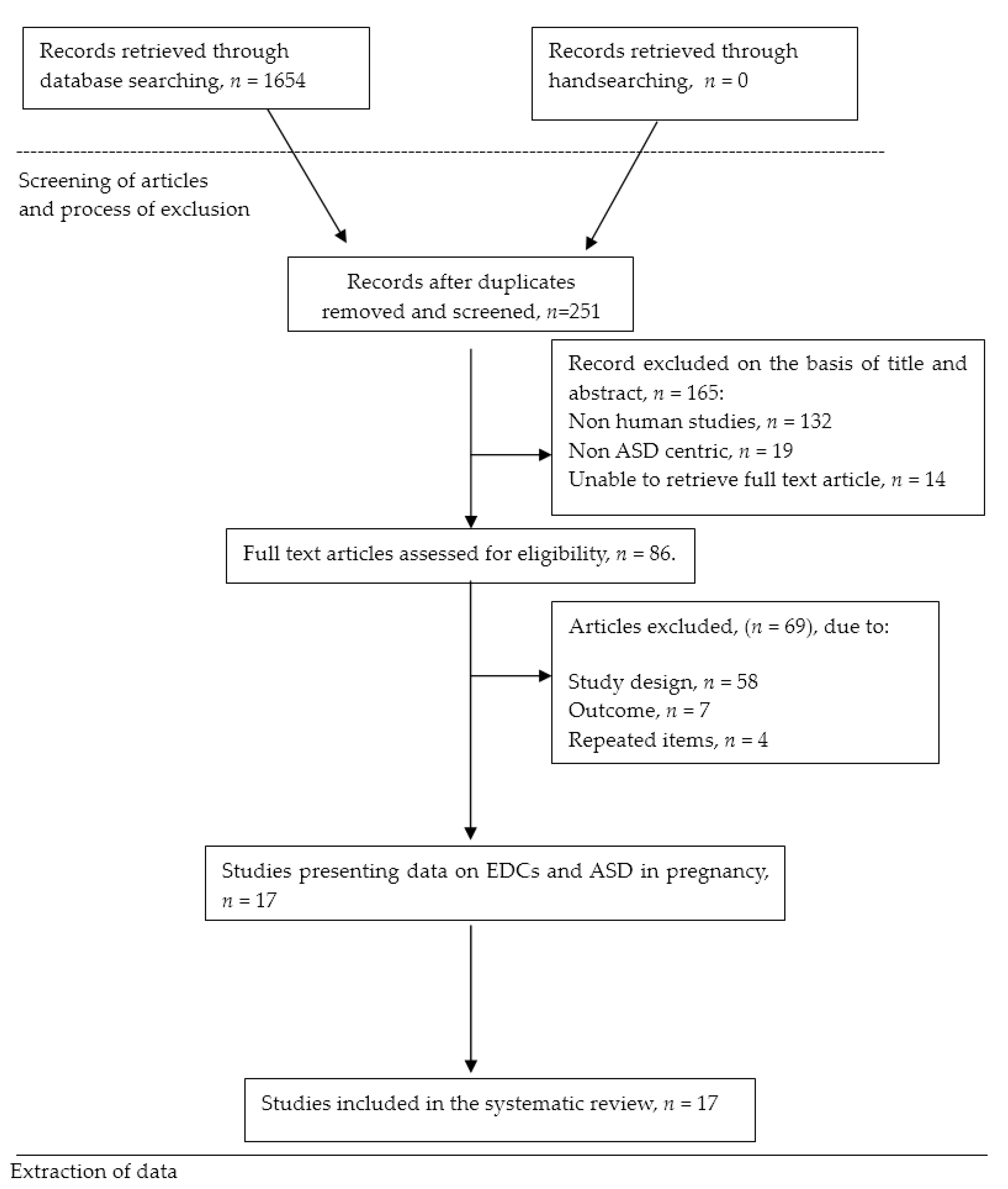
2. Materials and Methods
2.1. Search Strategy
2.2. Specify the Study Question
- Participants: pregnant women and their children of any age.
- Exposure: exposure to EDCs during pregnancy. The EDCs exposure was measured either through questionnaires/interviews held with parents, estimations provided by environmental agencies (Toxic Release Inventory (TRI), the US EPA National-scale Air Toxics Assessment (NATA), or analyses of biological samples.
- Comparator: works defined by ASD observational studies, and comparing the EDCs exposure levels for people with ASD versus those without.
- Outcomes: children of any age classified as having ASD disorder.
2.3. Study Identification and Eligibility Criteria
2.4. Data Extraction
3. Results
3.1. Characteristics of Studies
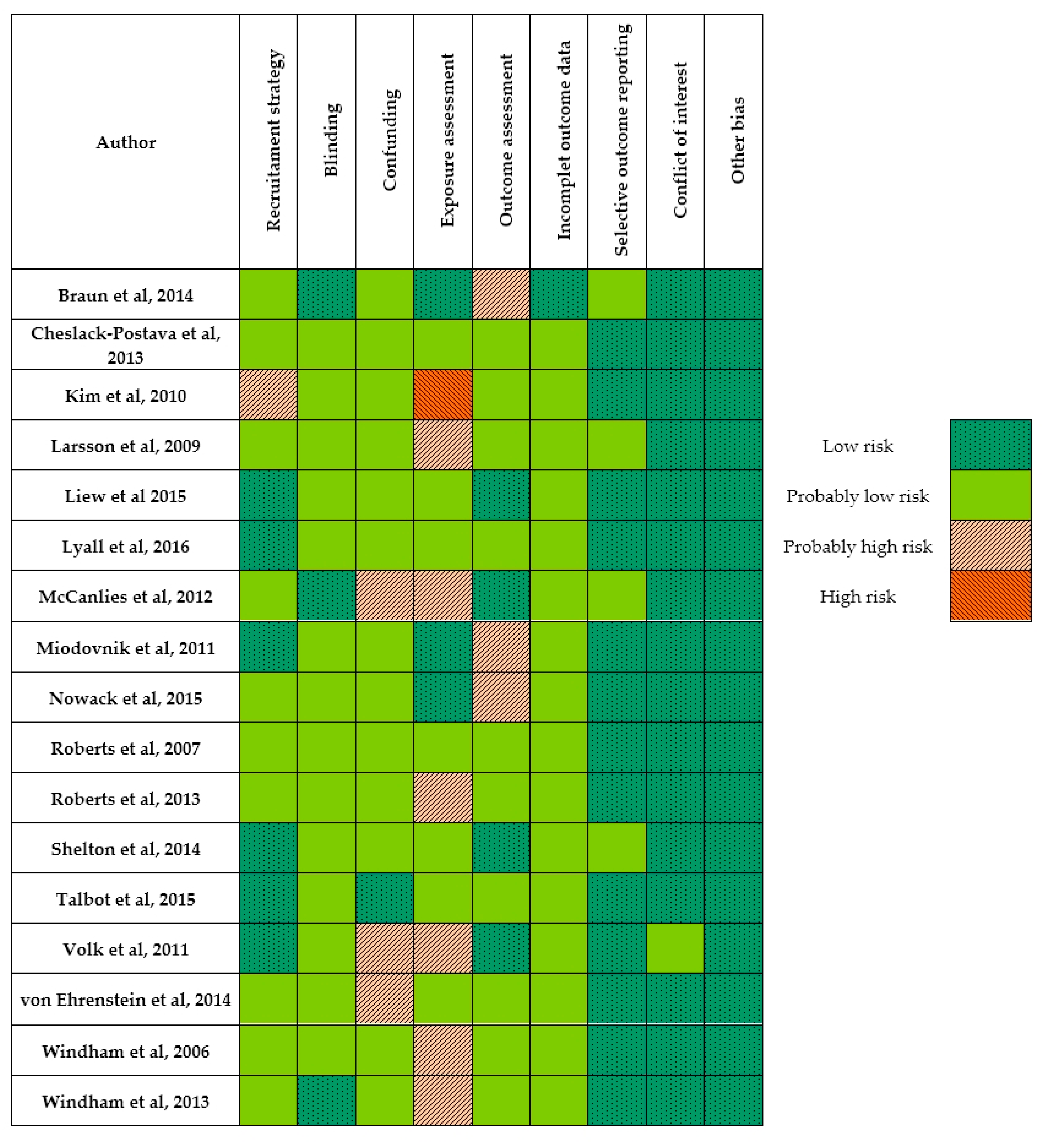
3.2. Internal Validity for Individual Studies
3.3. Risk of Bias Exposure Assessment for Individual Studies
3.4. Summary of Results
3.5. Quality of the Overall Body of Evidence
3.6. Strength of the Overall Body of Evidence
4. Discussion
4.1. Limitations of the Review Process
4.2. Overview of the Topic, Recommendations and Implications
4.3. Biological Plausibility
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kohane, I.S.; McMurry, A.; Weber, G.; MacFadden, D.; Rappaport, L.; Kunkel, L.; Bickel, J.; Wattanasin, N.; Spence, S.; Murphy, S.; et al. The Co-Morbidity Burden of Children and Young Adults with Autism Spectrum Disorders. PLoS ONE 2012, 7, e33224. [Google Scholar] [CrossRef] [PubMed]
- Lai, M.-C.; Lombardo, M.V.; Baron-Cohen, S. Autism. Lancet 2014, 383, 896–910. [Google Scholar] [CrossRef]
- Matson, J.L.; Kozlowski, A.M. The increasing prevalence of autism spectrum disorders. Res. Autism Spectr. Disord. 2011, 5, 418–425. [Google Scholar] [CrossRef]
- Baxter, A.J.; Brugha, T.S.; Erskine, H.E.; Scheurer, R.W.; Vos, T.; Scott, J.G. The epidemiology and global burden of autism spectrum disorders. Psychol. Med. 2014, 45, 601–613. [Google Scholar] [CrossRef] [PubMed]
- Elsabbagh, M.; Divan, G.; Koh, Y.-J.; Kim, Y.S.; Kauchali, S.; Marcín, C.; Montiel-Nava, C.; Patel, V.; Paula, C.S.; Wang, C.; et al. Global Prevalence of Autism and Other Pervasive Developmental Disorders. Autism Res. 2012, 5, 160–179. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Prevalence of Autism Spectrum Disorder Among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2014. MMWR Surveill. Summ. 2018, 67, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Christensen, D.L.; Baio, J.; Braun, K.V.N.; Bilder, D.; Charles, J.; Constantino, J.N.; Daniels, J.; Durkin, M.S.; Fitzgerald, R.T.; Kurzius-Spencer, M.; et al. Prevalence and Characteristics of Autism Spectrum Disorder Among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2012. MMWR Surveill. Summ. 2016, 65, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Christensen, D.L.; Bilder, D.A.; Zahorodny, W.; Pettygrove, S.; Durkin, M.S.; Fitzgerald, R.T.; Rice, C.; Kurzius-Spencer, M.; Baio, J.; Yeargin-Allsopp, M. Prevalence and characteristics of autism spectrum disorder among 4-year-old children in the autism and developmental disabilities monitoring network. J. Dev. Behav. Pediatr. 2016, 37, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Newschaffer, C.J.; Croen, L.A.; Daniels, J.; Giarelli, E.; Grether, J.K.; Levy, S.E.; Mandell, D.S.; Miller, L.A.; Pinto-Martin, J.; Reaven, J.; et al. The Epidemiology of Autism Spectrum Disorders. Annu. Rev. Public Health 2007, 28, 235–258. [Google Scholar] [CrossRef] [PubMed]
- Geschwind, D.H. Autism: Many Genes, Common Pathways? Cell 2008, 135, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Bailey, A.; Le Couteur, A.; Gottesman, I.; Bolton, P.; Simonoff, E.; Yuzda, E.; Rutter, M. Autism as a strongly genetic disorder: Evidence from a British twin study. Psychol. Med. 1995, 25, 63. [Google Scholar] [CrossRef] [PubMed]
- Steffenburg, S.; Gillberg, C.; Hellgren, L.; Andersson, L.; Gillberg, I.C.; Jakobsson, G.; Bohman, M. A Twin Study of Autism in Denmark, Finland, Iceland, Norway and Sweden. J. Child Psychol. Psychiatry 1989, 30, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Hallmayer, J. Genetic Heritability and Shared Environmental Factors Among Twin Pairs with Autism. Arch. Gen. Psychiatry 2011, 68, 1095. [Google Scholar] [CrossRef] [PubMed]
- Tordjman, S.; Somogyi, E.; Coulon, N.; Kermarrec, S.; Cohen, D.; Bronsard, G.; Bonnot, O.; Weismann-Arcache, C.; Botbol, M.; Lauth, B.; et al. Gene × Environment Interactions in Autism Spectrum Disorders: Role of Epigenetic Mechanisms. Front. Psychiatry 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Hu, V.W. From Genes to Environment: Using Integrative Genomics to Build a “Systems-Level” Understanding of Autism Spectrum Disorders. Child Dev. 2012, 84, 89–103. [Google Scholar] [CrossRef] [PubMed]
- LaSalle, J.M. Epigenomic strategies at the interface of genetic and environmental risk factors for autism. J. Hum. Genet. 2013, 58, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Ronald, A.; Pennell, C.E.; Whitehouse, A.J.O. Prenatal Maternal Stress Associated with ADHD and Autistic Traits in early Childhood. Front. Psychol. 2011, 1. [Google Scholar] [CrossRef] [PubMed]
- Roth, T.L. Epigenetics of neurobiology and behavior during development and adulthood. Dev. Psychobiol. 2012, 54, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.M.; Kalkbrenner, A.E.; Just, A.C.; Yolton, K.; Calafat, A.M.; Sjödin, A.; Hauser, R.; Webster, G.M.; Chen, A.; Lanphear, B.P. Gestational Exposure to Endocrine-Disrupting Chemicals and Reciprocal Social, Repetitive, and Stereotypic Behaviors in 4- and 5-Year-Old Children: The HOME Study. Environ. Health Perspect. 2014, 122, 513–520. [Google Scholar] [CrossRef] [PubMed]
- De Cock, M.; Maas, Y.G.H.; van de Bor, M. Does perinatal exposure to endocrine disruptors induce autism spectrum and attention deficit hyperactivity disorders? Review. Acta Paediatr. 2012, 101, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Kajta, M.; Wójtowicz, A.K. Impact of endocrine-disrupting chemicals on neural development and the onset of neurological disorders. Pharmacol. Rep. 2013, 65, 1632–1639. [Google Scholar] [CrossRef]
- Kim, S.M.; Han, D.H.; Lyoo, H.S.; Min, K.J.; Kim, K.H.; Renshaw, P. Exposure to Environmental Toxins in Mothers of Children with Autism Spectrum Disorder. Psychiatry Investig. 2010, 7, 122. [Google Scholar] [CrossRef] [PubMed]
- Lanphear, B.P. The Impact of Toxins on the Developing Brain. Annu. Rev. Public Health 2015, 36, 211–230. [Google Scholar] [CrossRef] [PubMed]
- Lyall, K.; Schmidt, R.J.; Hertz-Picciotto, I. Maternal lifestyle and environmental risk factors for autism spectrum disorders. Int. J. Epidemiol. 2014, 43, 443–464. [Google Scholar] [CrossRef] [PubMed]
- Sandin, S.; Lichtenstein, P.; Kuja-Halkola, R.; Larsson, H.; Hultman, C.M.; Reichenberg, A. The Familial Risk of Autism. JAMA 2014, 311, 1770. [Google Scholar] [CrossRef] [PubMed]
- Shelton, J.F.; Hertz-Picciotto, I.; Pessah, I.N. Tipping the Balance of Autism Risk: Potential Mechanisms Linking Pesticides and Autism. Environ. Health Perspect. 2012, 120, 944–951. [Google Scholar] [CrossRef] [PubMed]
- Volk, H.E.; Hertz-Picciotto, I.; Delwiche, L.; Lurmann, F.; McConnell, R. Residential Proximity to Freeways and Autism in the CHARGE Study. Environ. Health Perspect. 2011, 119, 873–877. [Google Scholar] [CrossRef] [PubMed]
- Siniscalco, D.; Cirillo, A.; Bradstreet, J.; Antonucci, N. Epigenetic findings in autism: New perspectives for therapy. Int. J. Environ. Res. Public Health 2013, 10, 4261–4273. [Google Scholar] [CrossRef] [PubMed]
- Birnbaum, L.S. State of the Science of Endocrine Disruptors. Environ. Health Perspect. 2013, 121. [Google Scholar] [CrossRef] [PubMed]
- Zoeller, R.T.; Brown, T.R.; Doan, L.L.; Gore, A.C.; Skakkebaek, N.E.; Soto, A.M.; Woodruff, T.J.; Vom Saal, F.S. Endocrine-Disrupting Chemicals and Public Health Protection: A Statement of Principles from The Endocrine Society. Endocrinology 2012, 153, 4097–4110. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization; United Nations Environment Programme. State of the Science of Endocrine Disrupting Chemicals; United Nations Environment Programme and the World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- Brouwers, M.M.; van Tongeren, M.; Hirst, A.A.; Bretveld, R.W.; Roeleveld, N. Occupational exposure to potential endocrine disruptors: Further development of a job exposure matrix. Occup. Environ. Med. 2009, 66, 607–614. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization; International Labour Organisation; United Nations Environment Programme. Global Assessment of the State-of-the-Science of Endocrine Disruptors; World Health Organization: Geneva, Switzerland, 2002. [Google Scholar]
- Goldman, L. New approaches for assessing the etiology and risks of developmental abnormalities from chemical exposure. Reprod. Toxicol. 1997, 11, 443–451. [Google Scholar] [CrossRef]
- Sharara, F.I.; Seifer, D.B.; Flaws, J.A. Environmental toxicants and female reproduction 44Additional references are available from the authors. Fertil. Steril. 1998, 70, 613–622. [Google Scholar] [CrossRef]
- Braun, J.M.; Yolton, K.; Stacy, S.L.; Erar, B.; Papandonatos, G.D.; Bellinger, D.C.; Lanphear, B.P.; Chen, A. Prenatal environmental chemical exposures and longitudinal patterns of child neurobehavior. NeuroToxicology 2017, 62, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Vrijheid, M.; Casas, M.; Gascon, M.; Valvi, D.; Nieuwenhuijsen, M. Environmental pollutants and child health—A review of recent concerns. Int. J. Hyg. Environ. Health 2016, 219, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.M.; Yolton, K.; Dietrich, K.N.; Hornung, R.; Ye, X.; Calafat, A.M.; Lanphear, B.P. Prenatal Bisphenol A Exposure and Early Childhood Behavior. Environ. Health Perspect. 2009, 117, 1945–1952. [Google Scholar] [CrossRef] [PubMed]
- Herbstman, J.B.; Kurzon, M.; Lederman, S.A.; Rauh, V.; Tang, D.; Perera, F. Prenatal PBDEs and Neurodevelopment: Herbstman et al. Respond to Goodman et al. and to Banasik and Strosznajder. Environ. Health Perspect. 2010, 118, a469–a470. [Google Scholar] [CrossRef]
- Kim, Y.; Ha, E.; Kim, E.; Park, H.; Ha, M.; Kim, J.; Hong, Y.; Chang, N.; Kim, B. Prenatal Exposure to Phthalates and Infant Development at 6 Months: Prospective Mothers and Children’s Environmental Health (MOCEH) Study. Environ. Health Perspect. 2011, 119, 1495–1500. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J.P. Sir Richard Doll Lecture. Developmental origins of chronic disease. Public Health 2012, 126, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Heindel, J.J.; Balbus, J.; Birnbaum, L.; Brune-Drisse, M.N.; Grandjean, P.; Gray, K.; Landrigan, P.J.; Sly, P.D.; Suk, W.; Cory Slechta, D.; et al. Developmental Origins of Health and Disease: Integrating Environmental Influences. Endocrinology 2015, 156, 3416–3421. [Google Scholar] [CrossRef] [PubMed]
- Adinolfi, M. The development of the human blood-CSF-brain barrier. Dev. Med. Child Neurol. 1985, 27, 532–537. [Google Scholar] [CrossRef] [PubMed]
- Meeker, J.D. Exposure to Environmental Endocrine Disruptors and Child Development. Arch. Pediatr. Adolesc. Med. 2012, 166. [Google Scholar] [CrossRef] [PubMed]
- Moosa, A.; Shu, H.; Sarachana, T.; Hu, V.W. Are endocrine disrupting compounds environmental risk factors for autism spectrum disorder? Horm. Behav. 2018, 101, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Palanza, P.; Morellini, F.; Parmigiani, S.; vom Saal, F. Prenatal exposure to endocrine disrupting chemicals: Effects on behavioral development. Neurosci. Biobehav. Rev. 1999, 23, 1011–1027. [Google Scholar] [CrossRef]
- Sadler, T. Langman’s Medical Embryology, 12th ed.; Lippincott Williams & Wilkins: Baltimore, MD, USA, 2011. [Google Scholar]
- Andersen, H.R.; Nielsen, J.B.; Grandjean, P. Toxicologic evidence of developmental neurotoxicity of environmental chemicals. Toxicology 2000, 144, 121–127. [Google Scholar] [CrossRef]
- Sakamoto, M.; Kubota, M.; Liu, X.J.; Murata, K.; Nakai, K.; Satoh, H. Maternal and Fetal Mercury andn-3 Polyunsaturated Fatty Acids as a Risk and Benefit of Fish Consumption to Fetus. Environ. Sci. Technol. 2004, 38, 3860–3863. [Google Scholar] [CrossRef] [PubMed]
- Hakkola, J.; Tanaka, E.; Pelkonen, O. Developmental Expression of Cytochrome P450 Enzymes in Human Liver. Pharmacol. Toxicol. 1998, 82, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Koufaris, C.; Sismani, C. Modulation of the Genome and Epigenome of Individuals Susceptible to Autism by Environmental Risk Factors. Int. J. Mol. Sci. 2015, 16, 8699–8718. [Google Scholar] [CrossRef] [PubMed]
- Matelski, L.; Van de Water, J. Risk factors in autism: Thinking outside the brain. J. Autoimmun. 2016, 67, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Prusinski, L.; Al-Hendy, A.; Yang, Q. Developmental Exposure to Endocrine Disrupting Chemicals Alters the Epigenome: Identification of Reprogrammed Targets. Gynecol. Obstet. Res. Open J. 2016, 3, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Schug, T.T.; Blawas, A.M.; Gray, K.; Heindel, J.J.; Lawler, C.P. Elucidating the Links Between Endocrine Disruptors and Neurodevelopment. Endocrinology 2015, 156, 1941–1951. [Google Scholar] [CrossRef] [PubMed]
- Sealey, L.A.; Hughes, B.W.; Sriskanda, A.N.; Guest, J.R.; Gibson, A.D.; Johnson-Williams, L.; Pace, D.G.; Bagasra, O. Environmental factors in the development of autism spectrum disorders. Environ. Int. 2016, 88, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Craig, Z.R.; Wang, W.; Flaws, J.A. Endocrine-disrupting chemicals in ovarian function: Effects on steroidogenesis, metabolism and nuclear receptor signaling. Reproduction 2011, 142, 633–646. [Google Scholar] [CrossRef] [PubMed]
- Ghisari, M.; Bonefeld-Jorgensen, E.C. Effects of plasticizers and their mixtures on estrogen receptor and thyroid hormone functions. Toxicol. Lett. 2009, 189, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, M.E.; Gore, A.C. Endocrine disruptors: Chemical contaminants—A toxic mixture for neurodevelopment. Nat. Rev. Endocrinol. 2017, 13, 322–323. [Google Scholar] [CrossRef] [PubMed]
- Lanphear, B.P.; Hornung, R.; Khoury, J.; Yolton, K.; Baghurst, P.; Bellinger, D.C.; Canfield, R.L.; Dietrich, K.N.; Bornschein, R.; Greene, T.; et al. Low-Level Environmental Lead Exposure and Children’s Intellectual Function: An International Pooled Analysis. Environ. Health Perspect. 2005, 113, 894–899. [Google Scholar] [CrossRef] [PubMed]
- Axelrad, D.A.; Bellinger, D.C.; Ryan, L.M.; Woodruff, T.J. Dose–Response Relationship of Prenatal Mercury Exposure and IQ: An Integrative Analysis of Epidemiologic Data. Environ. Health Perspect. 2007, 115, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Hoover, R.N.; Hyer, M.; Pfeiffer, R.M.; Adam, E.; Bond, B.; Cheville, A.L.; Colton, T.; Hartge, P.; Hatch, E.E.; Herbst, A.L.; et al. Adverse Health Outcomes in Women Exposed in Utero to Diethylstilbestrol. N. Engl. J. Med. 2011, 365, 1304–1314. [Google Scholar] [CrossRef] [PubMed]
- Eubig, P.A.; Aguiar, A.; Schantz, S.L. Lead and PCBs as Risk Factors for Attention Deficit/Hyperactivity Disorder. Environ. Health Perspect. 2010, 118, 1654–1667. [Google Scholar] [CrossRef] [PubMed]
- Eskenazi, B.; Marks, A.R.; Bradman, A.; Fenster, L.; Johnson, C.; Barr, D.B.; Jewell, N.P. In Utero Exposure to Dichlorodiphenyltrichloroethane (DDT) and Dichlorodiphenyldichloroethylene (DDE) and Neurodevelopment Among Young Mexican American Children. Pediatrics 2006, 118, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Rauh, V.A.; Garfinkel, R.; Perera, F.P.; Andrews, H.F.; Hoepner, L.; Barr, D.B.; Whitehead, R.; Tang, D.; Whyatt, R.W. Impact of Prenatal Chlorpyrifos Exposure on Neurodevelopment in the First 3 Years of Life Among Inner-City Children. Pediatrics 2006, 118, e1845–e1859. [Google Scholar] [CrossRef] [PubMed]
- Gore, A.C.; Martien, K.M.; Gagnidze, K.; Pfaff, D. Implications of Prenatal Steroid Perturbations for Neurodevelopment, Behavior, and Autism. Endocr. Rev. 2014, 35, 961–991. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S.; Knickmeyer, R.; Belmonte, M. Sex Differences in the Brain: Implications for Explaining Autism. Science 2005, 310, 819–823. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S.; Auyeung, B.; Nørgaard-Pedersen, B.; Hougaard, D.M.; Abdallah, M.W.; Melgaard, L.; Cohen, A.S.; Chakrabarti, B.; Ruta, L.; Lombardo, M.V. Elevated fetal steroidogenic activity in autism. Mol. Psychiatry 2014, 20, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.M. Endocrine disrupting compounds, gonadal hormones, and autism. Dev. Med. Child Neurol. 2012, 54, 1068. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Harney, J.W.; Zavacki, A.M.; Sajdel-Sulkowska, E.M. Disrupted brain thyroid hormone homeostasis and altered thyroid hormone-dependent brain gene expression in autism spectrum disorders. J. Physiol. Pharmacol. 2014, 65, 257–272. [Google Scholar] [PubMed]
- Andersen, S.; Laurberg, P.; Wu, C.; Olsen, J. Attention deficit hyperactivity disorder and autism spectrum disorder in children born to mothers with thyroid dysfunction: A Danish nationwide cohort study. BJOG Int. J. Obstet. Gynaecol. 2014, 121, 1365–1374. [Google Scholar] [CrossRef] [PubMed]
- Chevrier, J.; Gunier, R.B.; Bradman, A.; Holland, N.T.; Calafat, A.M.; Eskenazi, B.; Harley, K.G. Maternal Urinary Bisphenol A during Pregnancy and Maternal and Neonatal Thyroid Function in the CHAMACOS Study. Environ. Health Perspect. 2013, 121, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Johns, L.E.; Ferguson, K.K.; Soldin, O.P.; Cantonwine, D.E.; Rivera-González, L.O.; Del Toro, L.V.; Calafat, A.M.; Ye, X.; Alshawabkeh, A.N.; Cordero, J.F.; et al. Urinary phthalate metabolites in relation to maternal serum thyroid and sex hormone levels during pregnancy: A longitudinal analysis. Reprod. Biol. Endocrinol. 2015, 13, 4. [Google Scholar] [CrossRef] [PubMed]
- Skinner, M.K.; Anway, M.D.; Savenkova, M.I.; Gore, A.C.; Crews, D. Transgenerational Epigenetic Programming of the Brain Transcriptome and Anxiety Behavior. PLoS ONE 2008, 3, e3745. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, T.J.; Sutton, P. An Evidence-Based Medicine Methodology to Bridge The Gap Between Clinical And Environmental Health Sciences. Health Aff. 2011, 30, 931–937. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, T.J.; Sutton, P. The Navigation Guide Systematic Review Methodology: A Rigorous and Transparent Method for Translating Environmental Health Science into Better Health Outcomes. Environ. Health Perspect. 2014, 122, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Atkins, D.; Best, D.; Briss, P.A.; Eccles, M.; Falck-Ytter, Y.; Flottorp, S.; Guyatt, G.H.; Harbour, R.T.; Haugh, M.; Henry, D.; GRADE Working Group. Grading quality of evidence and strength of recommendations. BMJ 2004, 328, 1490. [Google Scholar] [CrossRef] [PubMed]
- Atkins, D.; Eccles, M.; Flottorp, S.; Guyatt, G.H.; Henry, D.; Hill, S.; Liberati, A.; O’Connell, D.; Oxman, A.D.; Phillips, B.; et al. Systems for grading the quality of evidence and the strength of recommendations I: Critical appraisal of existing approaches The GRADE Working Group. BMC Health Serv. Res. 2004, 4, 38. [Google Scholar] [CrossRef] [PubMed]
- Balshem, H.; Helfand, M.; Schünemann, H.J.; Oxman, A.D.; Kunz, R.; Brozek, J.; Vist, G.E.; Falck-Ytter, Y.; Meerpohl, J.; Norris, S. GRADE guidelines: 3. Rating the quality of evidence. J. Clin. Epidemiol. 2011, 64, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Atkins, D.; Brozek, J.; Vist, G.; Alderson, P.; Glasziou, P.; Falck-Ytter, Y.; Schünemann, H.J. GRADE guidelines: 2. Framing the question and deciding on important outcomes. J. Clin. Epidemiol. 2011, 64, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; deBeer, H. GRADE guidelines: 1. Introduction—GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Green, S. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- National Research Council. Science and Decisions: Advancing Risk Assessment; National Academies Press: Washington, DC, USA, 2009. [Google Scholar]
- Viswanathan, M.; Patnode, C.D.; Berkman, N.D.; Bass, E.B.; Chang, S.; Hartling, L.; Murad, M.H.; Treadwell, J.R.; Kane, R.L. Assessing the Risk of Bias of Individual Studies in Systematic Reviews of Health Care Interventions; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2012.
- Friedenreich, C.M. Methods for Pooled Analyses of Epidemiologic Studies. Epidemiology 1993, 4, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Froom, P.; Froom, J. Deficiencies in structured medical abstracts. J. Clin. Epidemiol. 1993, 46, 591–594. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Prevalence of autism spectrum disorders—Autism and developmental disabilities monitoring network, 14 sites, United States, 2002. MMWR Surveill. Summ. 2007, 56, 12–28. [Google Scholar]
- Centers for Disease Control and Prevention. Prevalence of Autism Spectrum Disorders—Autism and Developmental Disabilities Monitoring Network, 14 Sites, United States, 2008. MMWR Surveill. Summ. 2012, 61, 1–19. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; Text Revised DSM-IV-TR; American Psychiatric Association: Washington, DC, USA, 2000. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; DSM-V; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Bölte, S.; Poustka, F.; Constantino, J.N. Assessing autistic traits: Cross-cultural validation of the social responsiveness scale (SRS). Autism Res. 2008, 1, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Constantino, J.N.; Davis, S.A.; Todd, R.D.; Schindler, M.K.; Gross, M.M.; Brophy, S.L.; Metzger, L.M.; Shoushtari, C.S.; Splinter, R.; Reich, W. Validation of a Brief Quantitative Measure of Autistic Traits: Comparison of the Social Responsiveness Scale with the Autism Diagnostic Interview-Revised. J. Autism Dev. Disord. 2003, 33, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Vandenbroucke, J.P. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and Elaboration. Ann. Intern. Med. 2007, 147. [Google Scholar] [CrossRef] [PubMed]
- Larsson, M.; Weiss, B.; Janson, S.; Sundell, J.; Bornehag, C.-G. Associations between indoor environmental factors and parental-reported autistic spectrum disorders in children 6–8 years of age. Neurotoxicology 2009, 30, 822–831. [Google Scholar] [CrossRef] [PubMed]
- McCanlies, E.C.; Fekedulegn, D.; Mnatsakanova, A.; Burchfiel, C.M.; Sanderson, W.T.; Charles, L.E.; Hertz-Picciotto, I. Parental Occupational Exposures and Autism Spectrum Disorder. J. Autism Dev. Disord. 2012, 42, 2323–2334. [Google Scholar] [CrossRef] [PubMed]
- Roberts, A.L.; Lyall, K.; Hart, J.E.; Laden, F.; Just, A.C.; Bobb, J.F.; Koenen, K.C.; Ascherio, A.; Weisskopf, M.G. Perinatal Air Pollutant Exposures and Autism Spectrum Disorder in the Children of Nurses’ Health Study II Participants. Environ. Health Perspect. 2013, 121, 978–984. [Google Scholar] [CrossRef] [PubMed]
- Windham, G.C.; Zhang, L.; Gunier, R.; Croen, L.A.; Grether, J.K. Autism Spectrum Disorders in Relation to Distribution of Hazardous Air Pollutants in the San Francisco Bay Area. Environ. Health Perspect. 2006, 114, 1438–1444. [Google Scholar] [CrossRef] [PubMed]
- Windham, G.C.; Sumner, A.; Li, S.X.; Anderson, M.; Katz, E.; Croen, L.A.; Grether, J.K. Use of Birth Certificates to Examine Maternal Occupational Exposures and Autism Spectrum Disorders in Offspring. Autism Res. 2013, 6, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Roberts, E.; English, P.; Grether, J.; Windham, G.; Somberg, L. Maternal Residence Near Agricultural Pesticide Applications and Autism Among Children in the California Central Valley. Epidemiology 2007, 18, S52. [Google Scholar] [CrossRef]
- Shelton, J.F.; Geraghty, E.M.; Tancredi, D.J.; Delwiche, L.D.; Schmidt, R.J.; Ritz, B.; Hansen, R.L.; Hertz-Picciotto, I. Neurodevelopmental Disorders and Prenatal Residential Proximity to Agricultural Pesticides: The CHARGE Study. Environ. Health Perspect. 2014, 122, 1103–1109. [Google Scholar] [CrossRef] [PubMed]
- Talbott, E.O.; Marshall, L.P.; Rager, J.R.; Arena, V.C.; Sharma, R.K.; Stacy, S.L. Air toxics and the risk of autism spectrum disorder: The results of a population based case–control study in southwestern Pennsylvania. Environ. Health 2015, 14. [Google Scholar] [CrossRef] [PubMed]
- Von Ehrenstein, O.S.; Aralis, H.; Cockburn, M.; Ritz, B. In Utero Exposure to Toxic Air Pollutants and Risk of Childhood Autism. Epidemiology 2014, 25, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Miodovnik, A.; Engel, S.M.; Zhu, C.; Ye, X.; Soorya, L.V.; Silva, M.J.; Calafat, A.M.; Wolff, M.S. Endocrine disruptors and childhood social impairment. Neurotoxicology 2011, 32, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Nowack, N.; Wittsiepe, J.; Kasper-Sonnenberg, M.; Wilhelm, M.; Schölmerich, A. Influence of Low-Level Prenatal Exposure to PCDD/Fs and PCBs on Empathizing, Systemizing and Autistic Traits: Results from the Duisburg Birth Cohort Study. PLoS ONE 2015, 10, e0129906. [Google Scholar] [CrossRef] [PubMed]
- Cheslack-Postava, K.; Rantakokko, P.V.; Hinkka-Yli-Salomäki, S.; Surcel, H.-M.; McKeague, I.W.; Kiviranta, H.A.; Sourander, A.; Brown, A.S. Maternal serum persistent organic pollutants in the Finnish Prenatal Study of Autism: A pilot study. Neurotoxicol. Teratol. 2013, 38, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Liew, Z.; Ritz, B.; von Ehrenstein, O.S.; Bech, B.H.; Nohr, E.A.; Fei, C.; Bossi, R.; Henriksen, T.B.; Bonefeld-Jørgensen, E.C.; Olsen, J. Attention Deficit/Hyperactivity Disorder and Childhood Autism in Association with Prenatal Exposure to Perfluoroalkyl Substances: A Nested Case–Control Study in the Danish National Birth Cohort. Environ. Health Perspect. 2015, 123, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Sultan, S.; Glasziou, P.; Akl, E.A.; Alonso-Coello, P.; Atkins, D.; Kunz, R.; Brozek, J.; Montori, V.; et al. GRADE guidelines: 9. Rating up the quality of evidence. J. Clin. Epidemiol. 2011, 64, 1311–1316. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.I.; Sutton, P.; Atchley, D.S.; Koustas, E.; Lam, J.; Sen, S.; Robinson, K.A.; Axelrad, D.A.; Woodruff, T.J. The Navigation Guide—Evidence-Based Medicine Meets Environmental Health: Systematic Review of Human Evidence for PFOA Effects on Fetal Growth. Environ. Health Perspect. 2014, 122, 1028–1039. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.I.; Sutton, P.; Koustas, E.; Vesterinen, H.M.; Woodruff, T.J. Response to correspondence by Heather Lynch, Julie Goodman and Nancy Beck Re: “Application of the Navigation Guide systematic review methodology to the evidence for developmental and reproductive toxicity of triclosan”. Environ. Int. 2017, 102, 76–78. [Google Scholar] [CrossRef] [PubMed]
- Koustas, E.; Lam, J.; Sutton, P.; Johnson, P.I.; Atchley, D.S.; Sen, S.; Robinson, K.A.; Axelrad, D.A.; Woodruff, T.J. The Navigation Guide—Evidence-Based Medicine Meets Environmental Health: Systematic Review of Nonhuman Evidence for PFOA Effects on Fetal Growth. Environ. Health Perspect. 2014, 122, 1015–1027. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.; Koustas, E.; Sutton, P.; Johnson, P.I.; Atchley, D.S.; Sen, S.; Robinson, K.A.; Axelrad, D.A.; Woodruff, T.J. The Navigation Guide—Evidence-Based Medicine Meets Environmental Health: Integration of Animal and Human Evidence for PFOA Effects on Fetal Growth. Environ. Health Perspect. 2014, 122, 1040–1051. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.; Sutton, P.; Kalkbrenner, A.; Windham, G.; Halladay, A.; Koustas, E.; Lawler, C.; Davidson, L.; Daniels, N.; Newschaffer, C.; et al. A Systematic Review and Meta-Analysis of Multiple Airborne Pollutants and Autism Spectrum Disorder. PLoS ONE 2016, 11, e0161851. [Google Scholar] [CrossRef] [PubMed]
- Vesterinen, H.M.; Johnson, P.I.; Atchley, D.S.; Sutton, P.; Lam, J.; Zlatnik, M.G.; Sen, S.; Woodruff, T.J. Fetal growth and maternal glomerular filtration rate: A systematic review. J. Matern.-Fetal Neonatal Med. 2015, 28, 2176–2181. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Yang, D.; Wang, C.; Liu, W.; Liao, J.; Xu, T.; Bai, C.; Chen, J.; Lin, K.; Huang, C.; et al. Chronic zebrafish low dose decabrominated diphenyl ether (BDE-209) exposure affected parental gonad development and locomotion in F1 offspring. Ecotoxicology 2011, 20, 1813–1822. [Google Scholar] [CrossRef] [PubMed]
- Frye, C.; Bo, E.; Calamandrei, G.; Calzà, L.; Dessì-Fulgheri, F.; Fernández, M.; Fusani, L.; Kah, O.; Kajta, M.; Le Page, Y.; et al. Endocrine Disrupters: A Review of Some Sources, Effects, and Mechanisms of Actions on Behaviour and Neuroendocrine Systems. J. Neuroendocrinol. 2011, 24, 144–159. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Mitra, P.S.; Loffredo, C.A.; Trnovec, T.; Murinova, L.; Sovcikova, E.; Ghimbovschi, S.; Zang, S.; Hoffman, E.P.; Dutta, S.K. Transcriptional profiling and biological pathway analysis of human equivalence PCB exposure in vitro: Indicator of disease and disorder development in humans. Environ. Res. 2015, 138, 202–216. [Google Scholar] [CrossRef] [PubMed]
- Ciernia, A.V.; LaSalle, J. The landscape of DNA methylation amid a perfect storm of autism aetiologies. Nat. Rev. Neurosci. 2016, 17, 411–423. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Poschmann, J.; Cruz-Herrera del Rosario, R.; Parikshak, N.N.; Hajan, H.S.; Kumar, V.; Ramasamy, R.; Belgard, T.G.; Elanggovan, B.; Wong, C.C.Y.; et al. Histone Acetylome-wide Association Study of Autism Spectrum Disorder. Cell 2016, 167, 1385–1397.e11. [Google Scholar] [CrossRef] [PubMed]
- Schumann, C.M.; Sharp, F.R.; Ander, B.P.; Stamova, B. Possible sexually dimorphic role of miRNA and other sncRNA in ASD brain. Mol. Autism 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.E.; Parikshak, N.N.; Belgard, T.G.; Geschwind, D.H. Genome-wide, integrative analysis implicates microRNA dysregulation in autism spectrum disorder. Nat. Neurosci. 2016, 19, 1463–1476. [Google Scholar] [CrossRef] [PubMed]
- Skinner, M.K.; Savenkova, M.I.; Zhang, B.; Gore, A.C.; Crews, D. Gene bionetworks involved in the epigenetic transgenerational inheritance of altered mate preference: Environmental epigenetics and evolutionary biology. BMC Genom. 2014, 15, 377. [Google Scholar] [CrossRef] [PubMed]
- Siu, M.T.; Weksberg, R. Epigenetics of Autism Spectrum Disorder. Adv. Exp. Med. Biol. 2017, 63–90. [Google Scholar] [CrossRef]
| Chemical Group | Subgroups | Description | Reported Endocrine Disrupting Effects |
|---|---|---|---|
| Polycyclic aromatic hydrocarbons | None | Formed by incomplete combustion of carbon-containing fuels. | Anti-estrogenic effects in vitro |
| Polychlorinated organic compounds | Polychlorinated biphenyls (PCBs) Dioxins, furans, polychlorinated naphthalene (PCN) Hexachlorobenzene (HCB) Octachlorostyrene (OCS) | Produced as by-products during waste incineration and industrial processes involving carbon and chlorine (eg., during metal, solvent or pesticide manufacturing) PCBs: until 1970s widely used as insulating and cooling fluids | PCBs, dioxins, furans, PCN: interfere with steroid synthesis through aryl hydrocarbon receptor binding; HCB: affects male and female fertility in animal studies; OCS: their metabolites possibly interfere with thyroid homeostasis through binding to plasma proteins |
| Pesticides | Organochlorines CarbamatesOrganophosphatesTributyltin Pyrethroids Other pesticides | Used in agriculture.Other purposes include wood preservation, anti-fouling, parasite treatment and public hygiene | Estrogenic or anti-androgenic effects in vitro, reproductive toxicity in animal models, and fertility or endocrine alterations in human studies |
| Phthalates | Di-2-ethylhexyl phthalate (DEHP), di-isononyl phthalate (DNP), di-n-hexyl phthalate (DHP) Benzylbutyl phthalate (BBP) Dibutylphthalate(DBP) Diethyl phthalate (DEP) | Many industrial applications:High molecular weight phthalates (DEHP, DNP, DHP) primarily used a plasticisers in polyvinyl chloride(PVC).Low molecular weight phthalates (BBP, DBP, DEP) used as solvents and plasticisers in cosmetics, adhesives, ink, dyes and plastic packaging | DEHP, DNP, DHP, BBP, DBP: affect the development of male reproductive organs in animal studies; DEP, DBP, BBP: suggested to interfere with male reproductive hormone levels in children |
| Organic solvents | Ethylene glycol ethers (EGEs) Styrene 5oluene Xylene Trichloroethylene (TCE) Perchloroethylene (PCE) | EGEs, toluene, xylene: widely used in, for example, paints, adhesives, thinners, lacquers and resinsStyrene: used for producing polystyrene plastics and resinsTCE, PCE: used for metal degreasing and other industrial cleaning purposes | EGEs: reproductive toxicity in animal studies and possibly associated with reduced fertility and menstrual length variability in women; Styrene: styrene dimers and trimers bind to estrogen receptors in vitro; Toluene, xylene, TCE: suggested to interfere with reproductive hormone levels in humans. PCE: dry cleaning has been associated with menstrual disorders, infertility and delayed conception in women |
| Bisphenol A | None | Used in the production of polycarbonate plastic and epoxy resins | Estrogenic effects according to in vitro and in vivo studies |
| Alkylphenolic compounds | Alkylphenolic ethoxylates (APEs) Alkylphenols (APs) | APEs: commonly used surfactants in, for example, detergents, pesticides and cosmetics; APs: primarily used to produce APEs | APE metabolites, which include APs and short chain APEs, interact with estrogen receptors in vitro |
| Flame retardants | Tetrabromobisphenol A (TBBPA) Hexabromocyclodecane (HBCD) Polybrominated disphenyl ethe r(PBDEs) | Widely used in the polymer industry, for example in the production of PVC, epoxy resins, polyesteand rubber | TBBPA, HBCD, PBDEs: interfere with thyroid hormone levels. TBBPA, PBDEs: possibly interfere with estrogen metabolism through estrogen sulfotransferase inhibition |
| Metals | Arsenic Cadmium Copper Lead Mercury | E.g., used in the electrical/electronics industry, for construction, in batteries, dyes, pesticides and dental amalgam, and as chemical intermediates | Arsenic: inhibits glucocorticoid gene transcription in vitro and thought to have similar effects on other steroid receptors. Cadmium, copper, lead, mercury: testicular toxicity in animal models or altered hormone levels and/or male subfertility in humans. |
| Miscellaneous | Benzophenones Parabens Siloxanes Phytoestrgens Pharmaceutical chemicals | Benzophenones: UV screens used in cosmetics and the plastic industry Parabens: widely used preservatives in cosmetics and the pharmaceutical industry Siloxanes: intermediates in the polymer industry and ingredients in personal care products and precision cleaning agents Soy and other plant products. | Benzophenones: bind to estrogen receptors in vitro and exert estrogenic effects in animal studies. Parabens: estrogenic activity in vitro and in animal Studies. Siloxanes: estrogenic and anti-estrogenic activity in animal studies |
| Risk of Bias a | Quality of Evidence b | Strength of Evidence b |
|---|---|---|
Domains:
Determined for each risk of bias domain
| Human evidence begins as moderate Downgrade criteria −1 or −2 according these factors:
+1 or +2 according these factors:
| The final rating represent the level of certainty of toxicity. Quality of body evidence:
|
| Terms | |
|---|---|
| #1 | “Autism spectrum disorder” [MeSH] OR “Autistic disorder”[MeSH] OR “Child Development Disorders, Pervasive”[Mesh] OR “Child Behavior Disorders”[Mesh] OR “ autistic traits” |
| #2 | “Environmental Exposure”[Mesh] OR “Endocrine Disruptors”[Mesh] OR “Pesticides”[Mesh] OR “Polychlorinated Biphenyls”Mesh] OR “Hydrocarbons, Chlorinated”[Mesh] OR “Dichlorodiphenyl Dichloroethylene”[Mesh] OR “DDT”[Mesh] OR “Hexachlorobenzene”[Mesh] OR “Flame Retardants”[Mesh] OR “Polybrominated Biphenyls”[Mesh] OR “Perfluorooctane sulfonic acid” [Supplementary Concept] OR “Bisphenol A” [Supplementary Concept] OR “Perfluorooctanoic acid” [Supplementary Concept] |
| #3 | “pregnancy” OR “prenatal” OR “in utero” |
| #4 | #1 AND #2 AND #3 |
| Author and Year | Study Population and Sample Size (N) | Study Design | Exposure | Outcome | Results |
|---|---|---|---|---|---|
| Kim et al., 2010 | 106 mothers of children with ADS and 324 mothers of typically developing (TD).children, and were recruited from special and elementary schools respectively in Seoul, Chungju, and Chuncheon, South Korea | Case-control study. | Self-reported exposure. Two questionnaires (knowledge/exposure) asking about the potential risk to EDCs. These questions regarding possible exposure to PBDEs, PCBs, BPA and PCDD were selected based on the guidelines provided by the study ‘Ministry of Environment for the Republic of Korea’ | The Child Behavior Checklist Korean version (K-CBCL) was used to assess the diagnosis and severity of behavioral traits of ASD in children | The knowledge regarding the possible toxicity to EDCs, such as PBDEs, PCBs BPA, PCDD was significantly higher in cases than controls (t = 2.9, p = 0.001) and self-reported exposure was significantly higher in cases than controls (t = 5.6, p = 0.001) |
| Larsson et al., 2009 | 72 children (60 boys, 12 girls) with ASD in the county of Värmland, Sweden | Retrospective study based on the DBH longitudinal cohort study | Questionnaire asking for type of floor material used at home (PVC, wood, linoleum, etc.) as source of phthalates. | Parentally-reported ASD | ASD aOR 1.66 (95% CI: 1.02–2.7) for children with PVC floor at home in comparison with those with other floor material. Poor ventilation was also associated with ASD. |
| McCanlies et al., 2012 | 174 families: 93 children with ASD and 81 TD children born and living in California, and enrolled in the CHARGE study | Case-control study | Industrial-Hygienist Evaluation Exposures, i.e., occupational exposure to asphalt and several solvents including nickel, chromium, iron, aluminum, lead, toluene, xylene, phthalate, PCBs), and collected retrospectively. | ASD were assessed on the Autism Diagnostic Interview Revised (ADI-R) and the Autism Diagnostic Observation Schedules (ADOS) | Higher exposure (OR ≥ 2) to asphalt and solvents were observed among parents with ASD children compared with parents of TD children. But no significant associations after correcting |
| Author and Year | Study Population and Sample Size (N) | Study Design | Exposure | Outcome | Results |
|---|---|---|---|---|---|
| Roberts et al., 2007 | Cases: 465 children with ASD. Controls: 6975 paired TD children Central Valley (California) | Case-control study. | Residential proximity of sources of agricultural pesticides: organochlorines, organophosphates, tTrifluralin) | Children with ASD were identified through electronic files of the California Department of Developmental Services according the Diagnostic and Statistical Manual of Mental Disorders (DSM IV-R) | In children of mothers living within 500 m of field sites (the fourth quartile vs. the lowest non-zero quartile of organochlorine poundage) to those with mothers not living near field sites the aOR was for ASD of 6.1 (95% CI: 2.4–15.3). |
| Roberts et al., 2013 | Cases: 325 children with ASD (46 girls, 279 boys). Controls: 22,101 TD children. From all 50 U.S. states. | Case-control study from the Nurses’ Health Study II cohort | US EPA concentrations of several pollutants according to residency: Antimony, arsenic, cadmium, chromium, lead, manganese, mercury, nickel, all metals, diesel particulate, styrene, and methylene chloride. | ASD diagnosis validated by telephone administration of the Autism Diagnostic Interview–Revised (ADI-R) to 50 randomly selected case mothers | Comparing the higher quintile score and the lowest quintile Lead: aOR = 1.6; 95% CI: 1.1, 2.3 Manganese: aOR = 1.5; 95% CI: 1.1, 2.2 Mercury: aOR = 2; 95% CI: 1.2, 3. NickelL: aOR = 1.7; 95% CI: 1.1, 2.5 Cadmium: aOR = 1.5; 95% CI: 1.0, 2.1 Total metals: aOR = 1.5; 95% CI: 1.0, 2.3 Styrene: aOR = 1.4; 95% CI: 1.0, 2.1 Methylenechloride: aOR = 1.8; 95% CI: 1.2,2.8 Diesel particulate: aOR = 2; 95% CI: 1.0,4.0 |
| Shelton et al., 2014 | 486 cases (children with ASD) and 316 controls (TD children) CHARGE study California | Case–control study | Proximity of homes to agricultural pesticides is used to estimate pesticide exposure using the Pesticide Use Report (PUR). Pesticides included are organophosphates, carbamates, pyrethroids, organochlorates and chlorpyrifos | Children are administered the Autism Diagnostic Observation Schedule (ADOS), combined with the ADI-R | Residential proximity (within 1.5 km) to agricultural pesticides it was compared with binary (1 = exposed vs. 0 = not exposed) indicators during pregnancy and his association with ASD Organophosphate pesticides: aOR = 2.07; (95% CI: 1.23, 3.50) Chlorpyrifos: aOR = 3.31; (95% CI: 1.48, 7.42) Pyrethroids: aOR 1.87; (95% CI: 1.02, 3.43) |
| Talbott et al., 2015 | 217 cases (children with ASD) and two different control groups: 1) 224 matched TD children and 2) 5,007 controls generated from a random sample using birth certificates (BC). Pennsylvania | Case–control study conducted by the EPA-NATA | Exposure to arsenic, chromium, methylene chloride, styrene, lead, cyanide, PAHs among other from ambient air pollution concentrations are estimated using modelled data from the 2005 NATA data. | ASD self-reported by family is diagnosed according to specific tests either such as ADOS or the Social Communication Questionnaire (SCQ) | Comparing fourth to first quartile of exposures: Styrene aOR 1.61 (95% CI = 1.08–2.38) Chromium aOR 1.60 (95% CI = 1.08–2.38). Methylene chloride aOR 1.41; 95% CI = 0.96–2.07) PAHs aOR 1.44; 95% CI = 0.98–2.11 Remaining compounds were not statistically significant. |
| Volk et al., 2011 | Cases: 304 children with ASD. Controls: 259 TD children. California | Case-control study based on the CHARGE study. | Residential proximity to a freeway during pregnancy as a surrogate for air pollution (traffic-related pollutants) | The diagnosis of ASD was evaluated from both the ADOS and the ADI-R | Residential proximity (≤309 m) was compared to distance to the nearest freeway during the third trimester of pregnancy and was associated with ASD in offspring (aOR = 2.22; 95% CI: 1.16–4.42). No association with living close to other main roads during pregnancy and ASD. |
| von Ehrenstein et al., 2014 | Cohort of children (n = 148,722) of which 768 were diagnosed with ASD. Los Angeles County, California | Observational cohort study | 1,3-butadiene, lead, benzene, toluene, ethyl-benzene, xylenes, formaldehyde, and chlorinated solvents measured by community-based air-monitoring stationsin mothers residing at 5km from air-toxics during pregnancy. | ASD cases are identified through records maintained by the California Department of Developmental Services and diagnosed according the DSM IV-R | ASD increased risk per interquartile-range increase of exposures: 1,3-butadiene: aOR = 1.59; 95% CI: 1.18–2.15 Meta/para-xylene: aOR = 1.51; 95% CI: 1.26–1.82 Lead: aOR = 1.49; 95% CI = 1.23–1.81 Perchloroethylene: aOR = 1.40; 95% CI: 1.09–1.80 Formaldehyde: aOR = 1.34; 95% CI: 1.17–1.52 |
| Windham et al., 2006 | Cases: 284 children with ASD. Controls: 657 TD children. Born in 1994 and live in San Francisco. California | Case-control study | Exposure to 25 environmental pollutants is estimated by the US EPA according to place of residence. | The diagnosis of ASD is made by qualified medical professionals according to the criteria of DSM-IV | ASD risk in the upper quartiles of chemical concentrations compared with those below the median. Chlorinated solvents: Methylene chloride: aOR = 1.50; 95% CI: 1.06, 2.13 Trichloroethylene: aOR = 1.47; 95% CI: 1.03, 2.08 Vinyl chloride: aOR = 1.75; 95% CI: 1.25, 2.43 Metals: Cadmium: aOR 1.54; 95% CI: 1.08, 2.20 Mercury: aOR 1.92; 95% CI: 1.36, 2.71 Nickel: aOR 1.46; 95% CI: 1.04, 2.06 Other exposures were not associated with ASD |
| Windham et al., 2013 | Parental occupation was obtained from birth certificates for 284 children with autism and 659 controls, born in 1994 in the San Francisco Bay Area (California) | Case-control study | Self-reported occupation and industry exposures are coded into eight chemical groups (exhaust/combustion products, disinfectants, metals, pesticides, solvents, cooling fluids, and auto paint) | Autism cases are identified according the DSM-IV by qualified medical professionals | Mothers of children with ASD had a higher probability (aOR = 2.3; 95% CI: 1.3, 4.2) of working in occupations considered exposed compared to mothers of controls (non-exposed). The exposure categories of the greatest frequency among case mothers were exhaust and combustion products (aOR 12.0; 95% CI: 1.4, 104.6) and disinfectants (aOR 4.0; 95% CI: 1.4, 12.0). |
| Author and Year | Study Population and Sample Size (N) | Study Design | Exposure | Outcome | Results |
|---|---|---|---|---|---|
| Braun et al., 2014 | 175 pregnant women ≥18 year from Cincinnati. | Observational study with the prospective birth cohort HOME | 8 phthalate metabolites, BPA, 25 PCBs, 6 organochlorine pesticides, 8 brominated flame retardants and 4 PFAS in maternal serum or urine samples taken at gestation weeks 16–26. | Mothers completed the SRS questionnaire when children were 4–5 years old to evaluate autistic behavior | Trans-nonachlor and PBDE-28 were associated with autistic behaviors, β = 4.1; 95% CI: 0.8–7.3 and β = 2.5; 95% CI: 0.6–5.6, respectively. Weak associations (not reaching the statistically significance) were observed for PCB-178 (β = −3.0; 95% CI: −6.3, 0.2), β-HCH (β = −3.3; 95% CI: −6.1, −0.5), PBDE-85 (β = −3.2; 95% CI: −5.9, −0.5) and PFOA (β = −2.0; 95% CI: −4.4, 0.4). |
| Cheslack-Postava et al., 2013 | Cases: 75 children with ASD. Controls: 75 TD children. Finland | Nested case-control pilot study in the Finnish Maternity Cohort | It is measured different PCB congeners, PBDE, HCB, DDT, and its metabolite (DDE) in maternal serum samples taken during pregnancy. | ASD in children validated by the ADI-R. | No significant association with ASD was found for any compound. The aOR of ASD in the >90th of exposure was compared to the lower end of the control distributions: PCBs aOR 1.91 (95% CI: 0.57, 6.39) HCB aOR 0.89 (95% CI: 0.28, 2.76) DDE aOR 1.79 (95% CI: 0.51, 6.21) |
| Liew et al., 2015 | 220 cases (children with ASD), and 550 TD children (controls) are selected from the Danish National Birth Cohort, Denmark | Nested case-control study. | Six PFASs measured in maternal plasma collected in early or mid-pregnancy | All diagnoses are based on ICD-10, code F84.0 | No associations were observed for any of the PFAS assessed in relation to ASD. |
| Lyall et al., 2017 | Cases: 545 children with ASD Controls: 418 TD children. California | Population-based case–control study | Concentrations of 11 PCB congeners and 2 OCPs measured in banked second-trimester serum samples and was compared between both groups | The diagnosis of ASD based on DSM-IV-TR criteria | OCPs were no associated with ASD and only 2 PCB congeners showed significant association. Comparing highest with the first quartile of PCBs: the OR of ASD were PCB138/158: aOR = 1.79 (95% CI: 1.10, 2.92) and PCB153: aOR = 1.82 (95% CI: 1.10, 3.02). |
| Miodovnik et al., 2011 | 137 mothers and their children born at Hospital Mount Sinai. New York City | Observational prospective cohort study. | Concentration of 10 metabolites of phthalates and BPA of maternal urine samples taken during the third trimester of pregnancy. | Mothers completed the SRS for detecting and measuring the severity of autistic behavior. | ΣLMWP β 1.53; 95% CI: 0.25–2.8 MEP β = 1.38; 95% CI: 0.23,2.53 |
| Nowack et al., 2015 | Out of 133 invited parents, 100 filled out the questionnaire SRS (N = 100) Duisburg, (Germany) | Observational cohort study | Concentrations of PCDD/Fs and PCBs measured in maternal whole blood samples during pregnancy. | Diagnosis and measurement of autistic behavior by the SRS. | Overall PCDD/Fs and PCBs were negatively associated with autistic behavior, PCDD/Fs: β = −6.66 (95% CI: −11.88, 1.44. p < 0.05); PCB: β = −3.99 (95% CI:−8.61, 0.64. p = 0.09). |
| Factor | Explanation | |
|---|---|---|
| Downgrades | ||
| Risk of bias | 0 to −1 | Based on the high/probably high risk of bias across the studies, mostly driven by the exposure assessment methods and the outcome evaluation (DSM, ICD-10, SRS, ADOS, ADI-R). The lack of specificity across different types of EDCs was of special concern. |
| Indirectness | 0 to −1 | Based on the adequate assessment of the exposure at individual level. There is a lack of individual EDC (or metabolites) and/or the exposures are not directly measured; for instance, Larsson et al. (2009) uses the floor material, McConlies et al. (2012) the occupational exposure and Volk et al. (2011) the residential proximity as a indicators of EDC. |
| Inconsistency | 0 | With few exceptions [Liew et al. (2015), Cheslack-Postava et al. (2013), and Mc Canlies et al. (2012)] results across studies are generally consistent in the magnitude and direction. Although most of the studies showed positive EDC-ASD associations, the magnitude of the effect was small and the statistical significance was not reached in many of them. |
| Imprecision | 0 | We judged that the CIs for ASD risk were considered as being excessively large. |
| Publication bias | 0 | There was no reason to suspect of publication bias. The search was comprehensive, and the most studies were generally consistent among their findings. |
| Upgrades | ||
| Magnitude of effect | 0 to +1 | Most of the studies found slight effects (i.e., OR < 2). However, several studies showed greater evidence of risk of ASD (RR or OR > 2) |
| Dose-response | 0 | The authors considered that there was some evidence of a dose-response relationship. |
| Confounding | 0 | We did not find evidence that possible residual confounding influenced results. In the studies retrieved potential confounders and effect modifiers they were examined including population characteristics such as race and ethnicity distribution, whether the tract was urban or rural, level of education, age of participants, percentage of the population below the poverty line, and median household income, among other. |
| Overall quality of evidence | Moderate | We judge that the results obtained from the retrieved studies not change the quality of the initial evidence |
| Overall strength of evidence | Limited | Although there is a trend of a positive association between the prenatal exposure to certain EDCs and following risk of ASD in the offspring, because the limitations present in the available studies so far, any conclusion can be drawn. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marí-Bauset, S.; Donat-Vargas, C.; Llópis-González, A.; Marí-Sanchis, A.; Peraita-Costa, I.; Llopis-Morales, J.; Morales-Suárez-Varela, M. Endocrine Disruptors and Autism Spectrum Disorder in Pregnancy: A Review and Evaluation of the Quality of the Epidemiological Evidence. Children 2018, 5, 157. https://doi.org/10.3390/children5120157
Marí-Bauset S, Donat-Vargas C, Llópis-González A, Marí-Sanchis A, Peraita-Costa I, Llopis-Morales J, Morales-Suárez-Varela M. Endocrine Disruptors and Autism Spectrum Disorder in Pregnancy: A Review and Evaluation of the Quality of the Epidemiological Evidence. Children. 2018; 5(12):157. https://doi.org/10.3390/children5120157
Chicago/Turabian StyleMarí-Bauset, Salvador, Carolina Donat-Vargas, Agustín Llópis-González, Amelia Marí-Sanchis, Isabel Peraita-Costa, Juan Llopis-Morales, and María Morales-Suárez-Varela. 2018. "Endocrine Disruptors and Autism Spectrum Disorder in Pregnancy: A Review and Evaluation of the Quality of the Epidemiological Evidence" Children 5, no. 12: 157. https://doi.org/10.3390/children5120157
APA StyleMarí-Bauset, S., Donat-Vargas, C., Llópis-González, A., Marí-Sanchis, A., Peraita-Costa, I., Llopis-Morales, J., & Morales-Suárez-Varela, M. (2018). Endocrine Disruptors and Autism Spectrum Disorder in Pregnancy: A Review and Evaluation of the Quality of the Epidemiological Evidence. Children, 5(12), 157. https://doi.org/10.3390/children5120157






