Fetal Congenital Complete Heart Block: A Rare Case with an Extremely Low Ventricular Rate and Review of Current Management Strategies
Abstract
1. Introduction
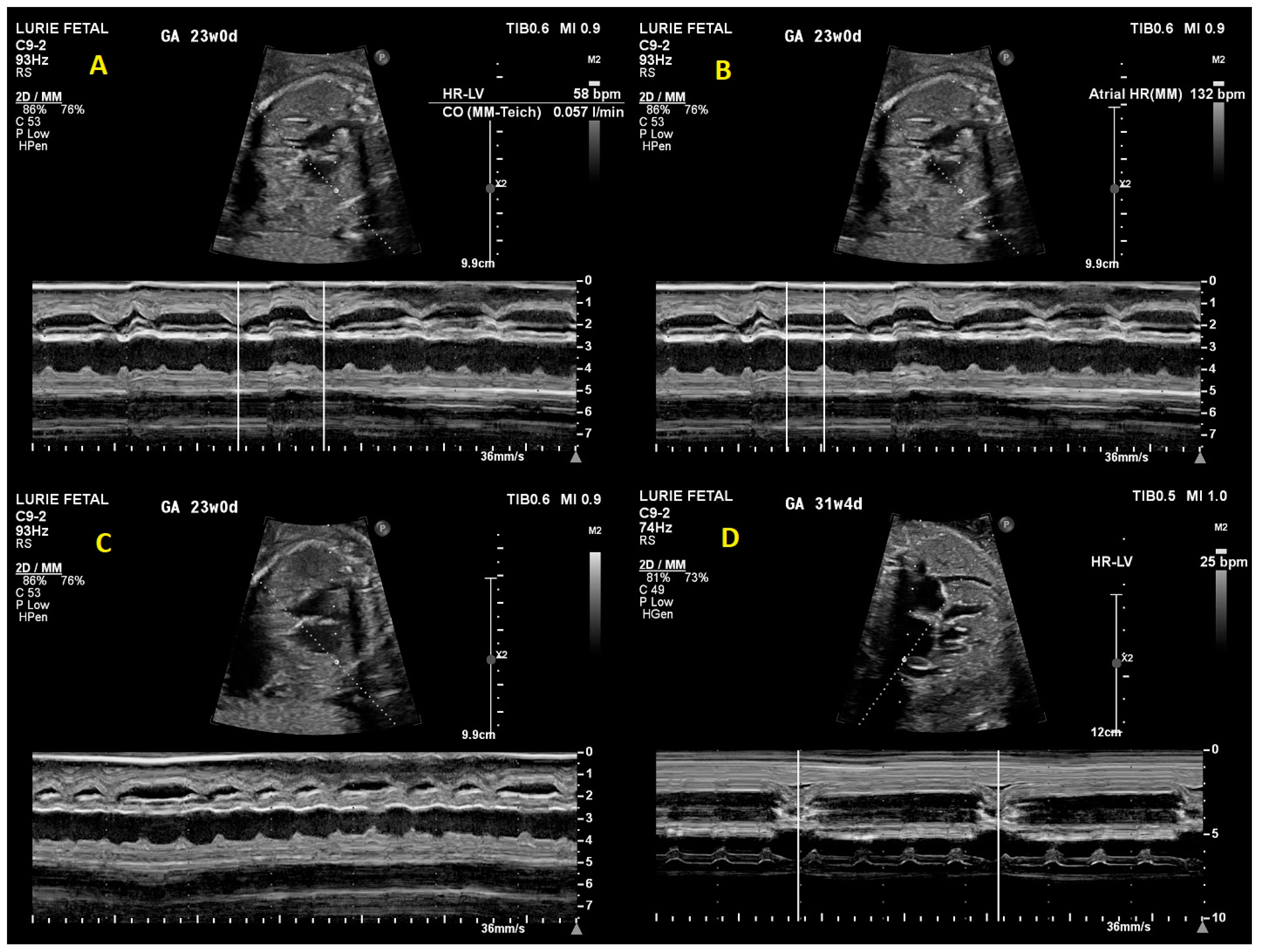
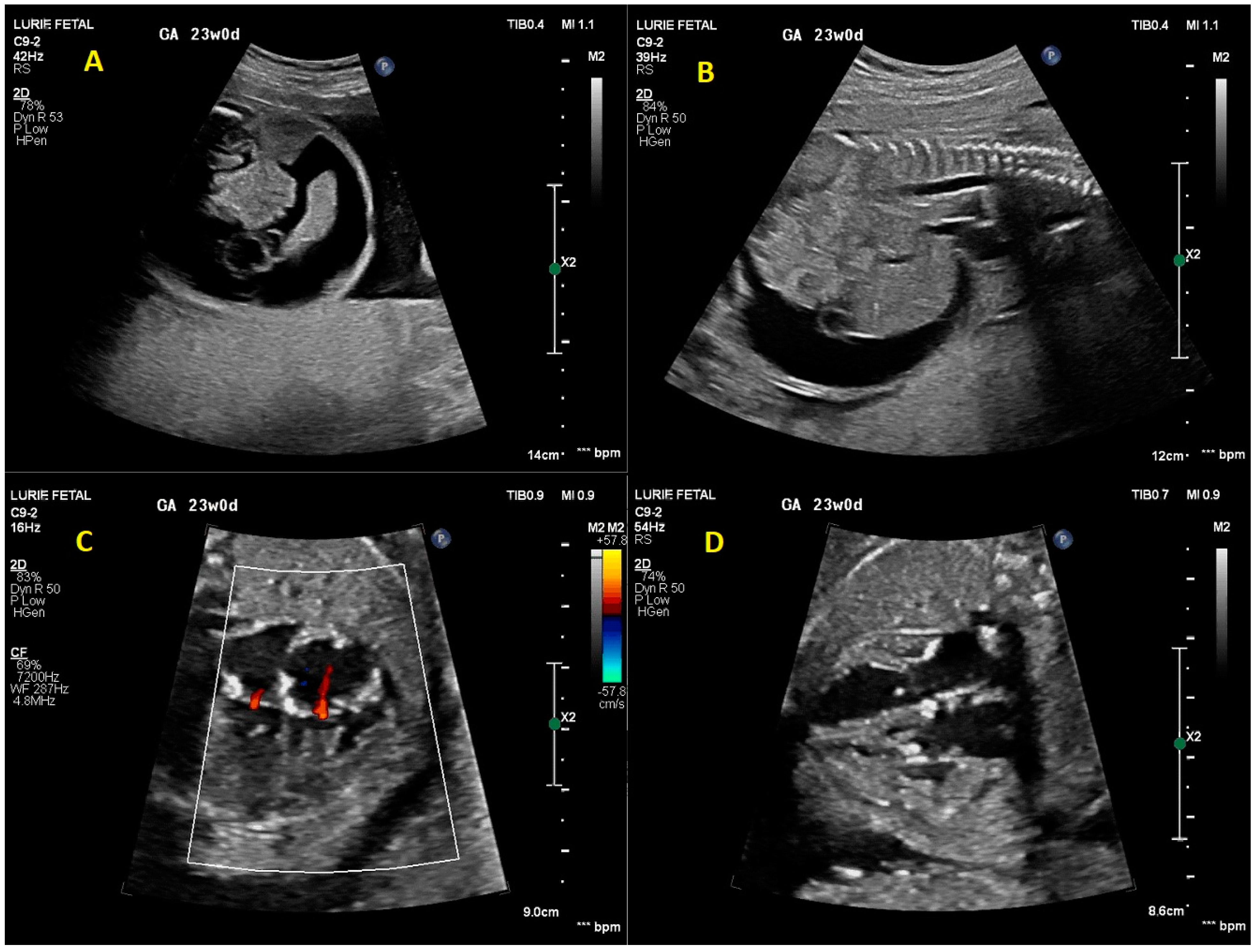
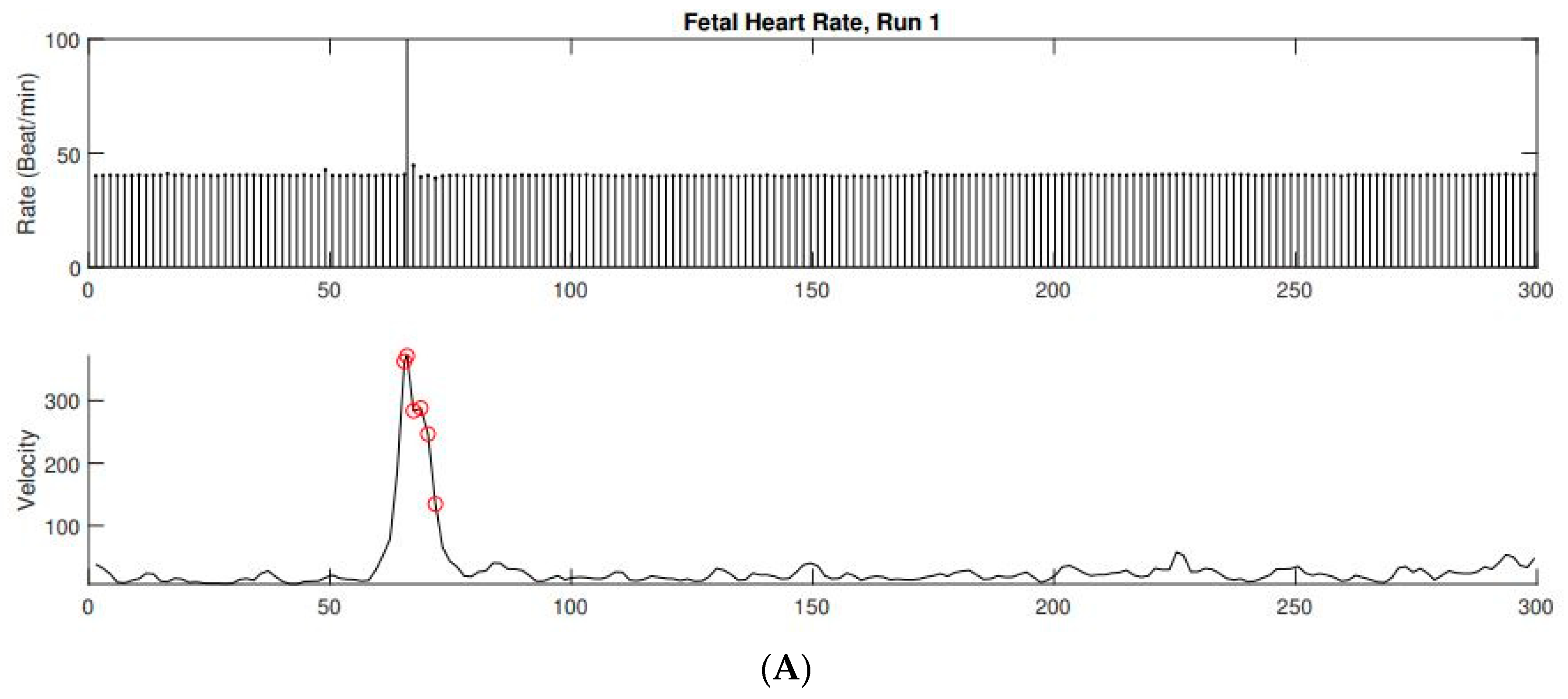
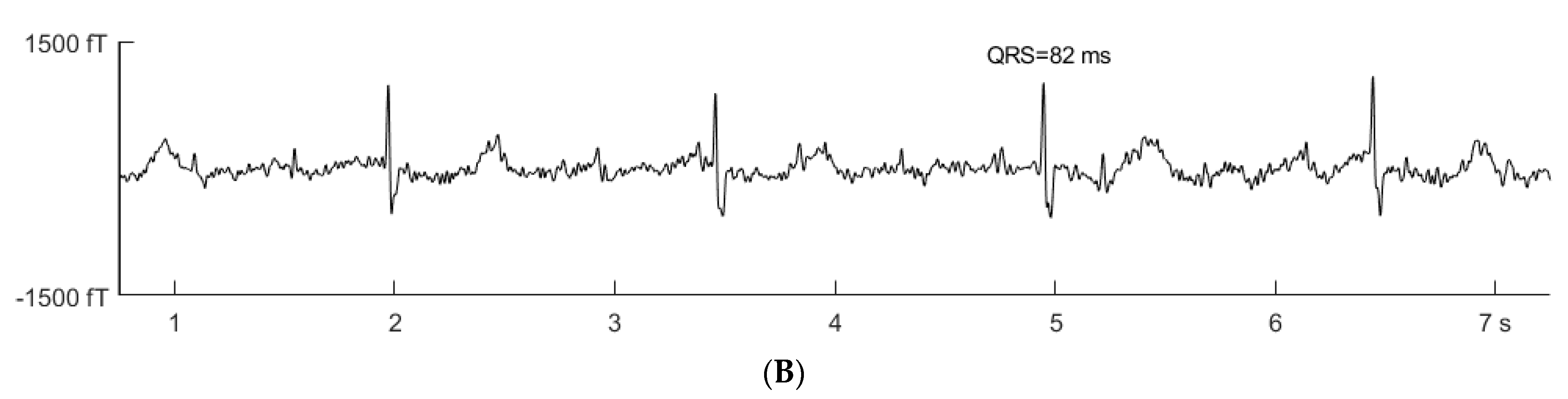
2. Case Presentation
3. Discussion
3.1. Etiology and Expected Outcomes
3.2. Prenatal Cardiac Evaluation
3.3. Prenatal Management
3.4. Delivery Room and Post-Natal Management
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Michaëlsson, M.; Engle, M.A. Congenital complete heart block: An international study of the natural history. Cardiovasc. Clin. 1972, 4, 85–101. [Google Scholar] [PubMed]
- Strasburger, J.F. Prenatal Diagnosis of Fetal Arrhythmias. Clin. Perinatol. 2005, 32, 891–912. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Cuneo, B.F.; Strasburger, J.F.; Huhta, J.C.; Gotteiner, N.L.; Wakai, R.T. Electrophysiological Characteristics of Fetal Atrioventricular Block. J. Am. Coll. Cardiol. 2008, 51, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Cuneo, B.F.; Zhao, H.; Strasburger, J.F.; Ovadia, M.; Huhta, J.C.; Wakai, R.T. Atrial and ventricular rate response and patterns of heart rate acceleration during maternal–fetal terbutaline treatment of fetal complete heart block. Am. J. Cardiol. 2007, 100, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Bordachar, P.; Zachary, W.; Ploux, S.; Labrousse, L.; Haissaguerre, M.; Thambo, J.-B. Pathophysiology, clinical course, and management of congenital complete atrioventricular block. Heart Rhythm. 2013, 10, 760–766. [Google Scholar] [CrossRef] [PubMed]
- Matta, M.J.; Cuneo, B.F. Doppler echocardiography for managing fetal cardiac arrhythmia. Clin. Obstet. Gynecol. 2010, 53, 899–914. [Google Scholar] [CrossRef] [PubMed]
- Eronen, M.; Sirèn, M.-K.; Ekblad, H.; Tikanoja, T.; Julkunen, H.; Paavilainen, T. Short- and long-term outcome of children with congenital complete heart block diagnosed in utero or as a newborn. Pediatrics 2000, 106 Pt 1, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Hernstadt, H.; Regan, W.; Bhatt, H.; Rosenthal, E.; Meau-Petit, V. Cohort study of congenital complete heart block among preterm neonates: A single-center experience over a 15-year period. Eur. J. Pediatr. 2021, 181, 1047–1054. [Google Scholar] [CrossRef] [PubMed]
- Eronen, M.; Heikkilä, P.; Teramo, K. Congenital Complete Heart Block in the Fetus: Hemodynamic Features, Antenatal Treatment, and Outcome in Six Cases. Pediatr. Cardiol. 2001, 22, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Vesel, S.; Završnik, T.; Podnar, T. Successful outcome in a fetus with an extremely low heart rate due to isolated complete congenital heart block. Ultrasound Obstet. Gynecol. 2003, 21, 189–191. [Google Scholar] [CrossRef] [PubMed]
- Pruetz, J.D.; Miller, J.C.; Loeb, G.E.; Silka, M.J.; Bar-Cohen, Y.; Chmait, R.H. Prenatal diagnosis and management of congenital complete heart block. Birth Defects Res. 2019, 111, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Wacker-Gussmann, A.; Cuneo, B.; Wakai, R.; Strasburger, J. Diagnosis and Treatment of Fetal Arrhythmia. Am. J. Perinatol. 2014, 31, 617–628. [Google Scholar] [CrossRef] [PubMed]
- Strasburger, J.F.; Eckstein, G.; Butler, M.; Noffke, P.; Wacker-Gussmann, A. Fetal Arrhythmia Diagnosis and Pharmacologic Management. J. Clin. Pharmacol. 2022, 62 (Suppl. S1), S53–S66. [Google Scholar] [CrossRef] [PubMed]
- Wacker-Gussmann, A.; Strasburger, J.F.; Wakai, R.T. Contribution of Fetal Magnetocardiography to Diagnosis, Risk Assessment, and Treatment of Fetal Arrhythmia. J. Am. Heart Assoc. 2022, 11, e025224. [Google Scholar] [CrossRef] [PubMed]
- Richards, D.S.; Wagman, A.J.; Cabaniss, M.L. Ascites not due to congestive heart failure in a fetus with lupus-induced heart block. Obstet. Gynecol. 1990, 76 Pt 2, 957–959. [Google Scholar]
- Ciardulli, A.; D’Antonio, F.; Magro-Malosso, E.R.; Manzoli, L.; Anisman, P.; Saccone, G.; Berghella, V. Maternal steroid therapy for fetuses with second-degree immune-mediated congenital atrioventricular block: A systematic review and meta-analysis. Acta Obstet. et Gynecol. Scand. 2018, 97, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Cuneo, B.F.; Ambrose, S.E.; Tworetzky, W. Detection and successful treatment of emergent anti-SSA mediated fetal atrioventricular block. Am. J. Obstet. Gynecol. 2016, 215, 527–528. [Google Scholar] [CrossRef] [PubMed]
- Donofrio, M.T. Predicting the Future: Delivery Room Planning of Congenital Heart Disease Diagnosed by Fetal Echocardiography. Am. J. Perinatol. 2018, 35, 549–552. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Fang, J.; Zhou, K.; Wang, C.; Hua, Y.; Shi, X.; Mu, D. Prediction of fetal outcome without intrauterine intervention using a cardiovascular profile score: A systematic review and meta-analysis. J. Matern. Neonatal Med. 2015, 28, 1965–1972. [Google Scholar] [CrossRef] [PubMed]
- Donofrio, M.T.; Gullquist, S.D.; Mehta, I.D.; Moskowitz, W.B.; Faap; Facc; Moskowitz, F.W.B. Congenital complete heart block: Fetal management protocol, review of the literature, and report of the smallest successful pacemaker implantation. J. Perinatol. 2004, 24, 112–117. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samples, S.; Fitt, C.; Satzer, M.; Wakai, R.; Strasburger, J.; Patel, S. Fetal Congenital Complete Heart Block: A Rare Case with an Extremely Low Ventricular Rate and Review of Current Management Strategies. Children 2023, 10, 1132. https://doi.org/10.3390/children10071132
Samples S, Fitt C, Satzer M, Wakai R, Strasburger J, Patel S. Fetal Congenital Complete Heart Block: A Rare Case with an Extremely Low Ventricular Rate and Review of Current Management Strategies. Children. 2023; 10(7):1132. https://doi.org/10.3390/children10071132
Chicago/Turabian StyleSamples, Stefani, Catherine Fitt, Michael Satzer, Ronald Wakai, Janette Strasburger, and Sheetal Patel. 2023. "Fetal Congenital Complete Heart Block: A Rare Case with an Extremely Low Ventricular Rate and Review of Current Management Strategies" Children 10, no. 7: 1132. https://doi.org/10.3390/children10071132
APA StyleSamples, S., Fitt, C., Satzer, M., Wakai, R., Strasburger, J., & Patel, S. (2023). Fetal Congenital Complete Heart Block: A Rare Case with an Extremely Low Ventricular Rate and Review of Current Management Strategies. Children, 10(7), 1132. https://doi.org/10.3390/children10071132





